Sodium-Glucose Cotransporter-2 Inhibitors Improve Cardiovascular Dysfunction in Type 2 Diabetic East Asians
Abstract
:1. Introduction
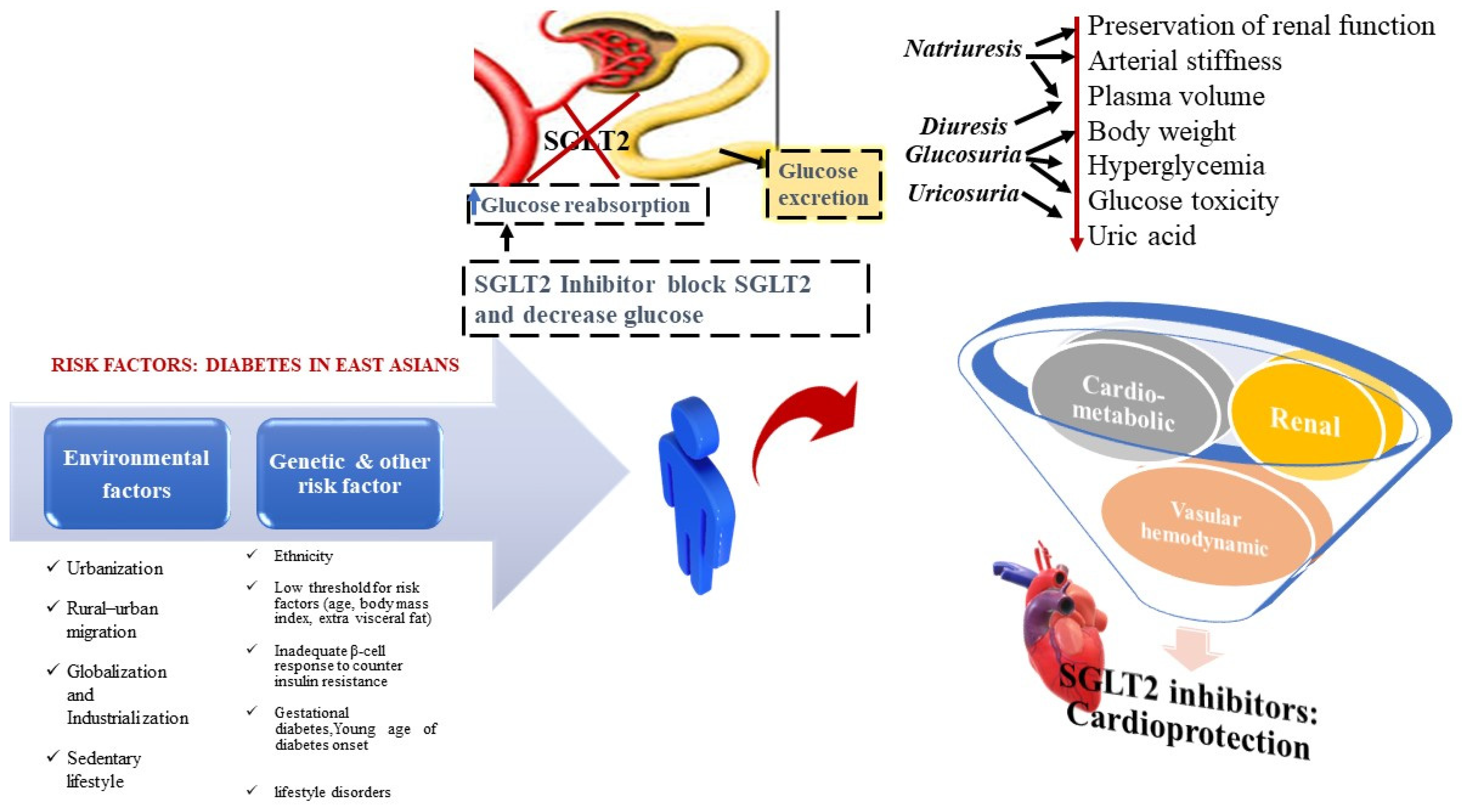
2. Feature Affects the High Occurrence of Diabetes in East Asians
Genetic Factors
3. Risk Factors Associated with Incidence of T2DM and Complications
4. SGLT2 Inhibitors in T2DM Cure in Asians
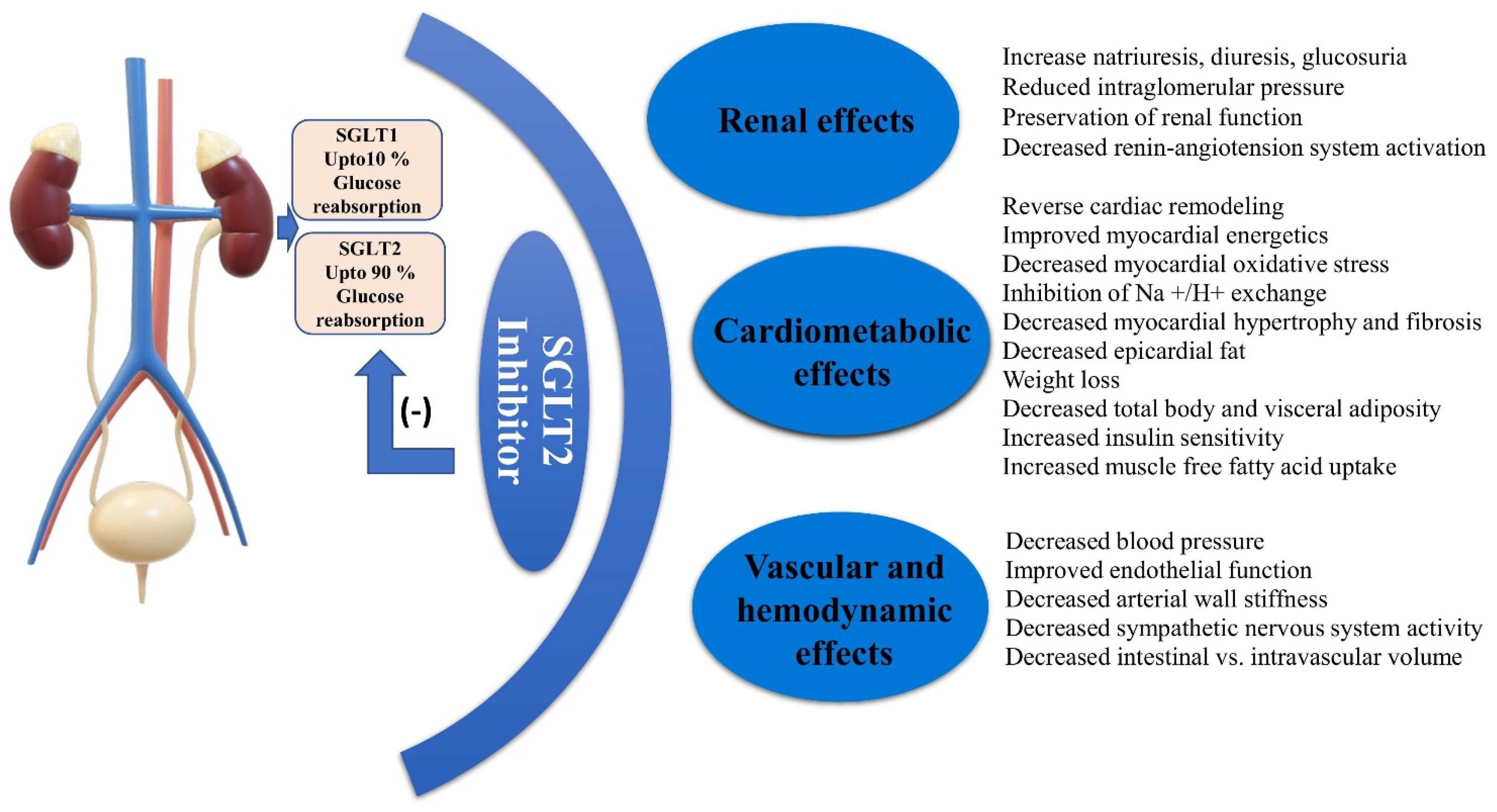
5. Cardiovascular Axis of SGLT2 Inhibitors
6. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
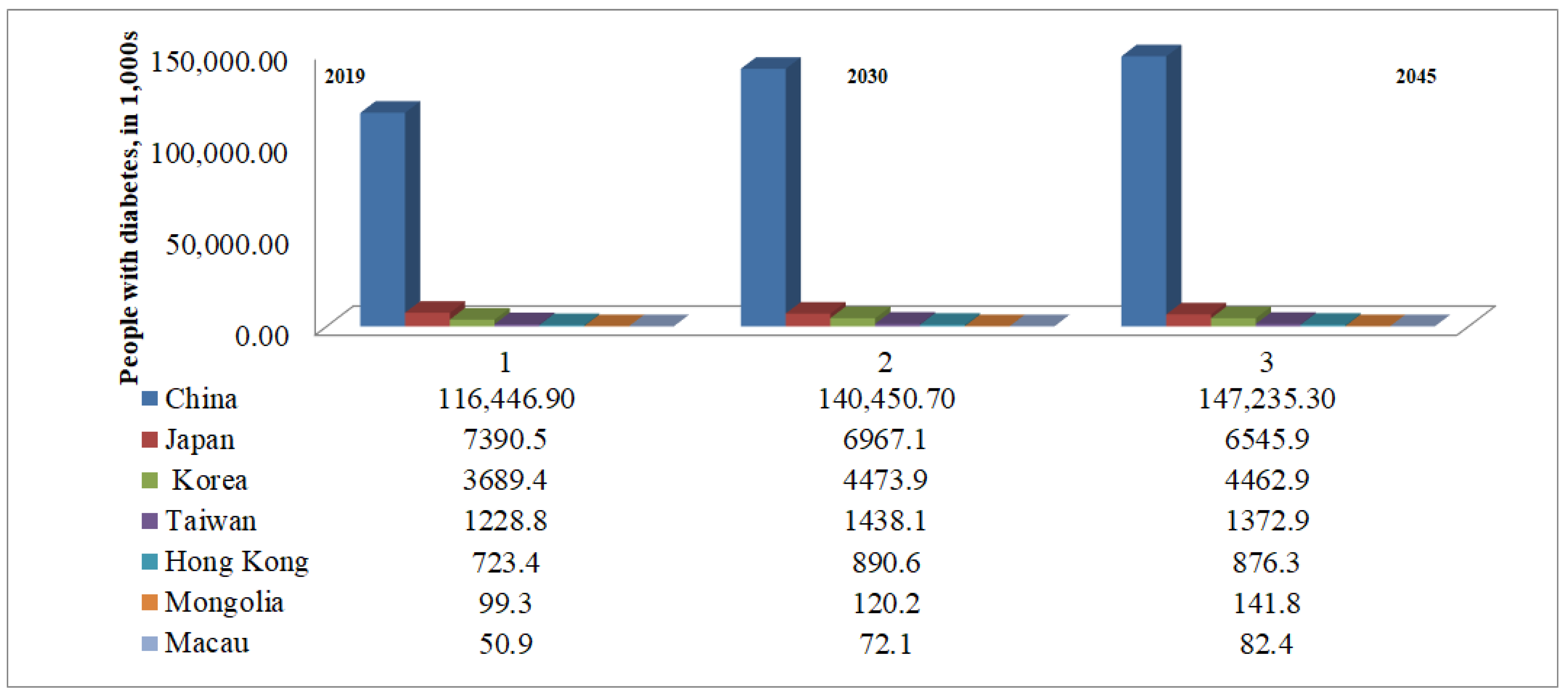
- Saeedi, P.; Petersohn, I.; Salpea, P.; Malanda, B.; Karuranga, S.; Unwin, N.; Colagiuri, S.; Guariguata, L.; Motalae, A.A.; Ogurtsova, K. Global and regional diabetes prevalence estimates for 2019 and projections for 2030 and 2045: Results from the International Diabetes Federation Diabetes Atlas. Diabetes Res. Clin. Pract. 2019, 157, 107843. [Google Scholar] [CrossRef] [Green Version]
- Priyadi, A.; Muhtadi, A.; Suwantika, A.A.; Sumiwi, S.A. An economic evaluation of diabetes mellitus management in South East Asia. J. Adv. Pharm. Edu. Res. 2019, 9, 53–74. [Google Scholar]
- Hu, F.B. Globalization of diabetes: The role of diet, lifestyle, and genes. Diabetes Care 2011, 34, 1249–1257. [Google Scholar] [CrossRef] [Green Version]
- Nanditha, A.; Ma, R.C.; Ramachandran, A.; Snehalatha, C.; Chan, J.C.; Chia, K.S.; Zimmet, P.Z. Diabetes in Asia and the Pacific: Implications for the global epidemic. Diabetes Care 2016, 39, 472–485. [Google Scholar] [CrossRef] [Green Version]
- Lim, L.L.; Tan, A.; Moses, K.; Rajadhyaksha, V.; Chan, S.P. Place of sodium-glucose cotransporter-2 inhibitors in East Asian subjects with type 2 diabetes mellitus: Insights into the management of Asian phenotype. J. Diabetes Its Complicat. 2016, 31, 494–503. [Google Scholar] [CrossRef] [PubMed]
- Yoon, K.H.; Lee, J.H.; Kim, J.W.; Cho, J.H.; Choi, Y.H.; Ko, S.H.; Zimmet, P.; Son, H.Y. Epidemic obesity and type 2 diabetes in Asia. Lancet 2006, 368, 1681–1688. [Google Scholar] [CrossRef]
- Chan, J.C.; Malik, V.; Jia, W.; Kadowaki, T.; Yajnik, C.S.; Yoon, K.H.; Hu, F.B. Diabetes in Asia: Epidemiology, risk factors, and pathophysiology. JAMA 2009, 301, 2129–2140. [Google Scholar] [CrossRef]
- Bragg, F.; Holmes, M.V.; Iona, A.; Guo, Y.; Du, H.; Chen, Y.; Bian, Z.; Yang, L.; Herrington, W.; Bennett, D.; et al. Association Between Diabetes and Cause-Specific Mortality in Rural and Urban Areas of China. JAMA 2017, 317, 280–289. [Google Scholar] [CrossRef] [PubMed]
- Ng, M.C.; Park, K.S.; Oh, B.; Tam, C.H.; Cho, Y.M.; Shin, H.D.; Cho, N.H. Implication of ge netic variants near TCF7L2, SLC30A8, HHEX, CDKAL1,CDKN2A/B, IGF2BP2, and FTO in type 2 diabetes and obesity in 6719 Asians. Diabetes 2008, 57, 2226–2233. [Google Scholar] [CrossRef] [Green Version]
- Chang, Y.C.; Chang, T.J.; Jiang, Y.D.; Kuo, S.S.; Lee, K.C.; Chiu, K.C.; Chuang, L.M. Associa tion study of the genetic polymorphisms of the tran scription factor 7-like 2 (TCF7L2) gene and type 2 dia betes in the Chinese population. Diabetes 2007, 56, 2631–2637. [Google Scholar] [CrossRef] [Green Version]
- Ma, R.C.; Chan, J.C. Type 2 diabetes in East Asians: Similarities and differences with populations in Europe and the United States. Ann. N. Y. Acad Sci. 2013, 1281, 64–91. [Google Scholar] [CrossRef] [Green Version]
- Cho, Y.M. Characteristics of the pathophysiology of type 2 diabetes in Asians. Ann. Laparosc. Endosc. Surg. 2017, 2, 14. [Google Scholar] [CrossRef]
- Jensen, E.T.; Dabelea, D. Type 2 Diabetes in Youth: New Lessons from the SEARCH Study. Curr. Diabetes Rep. 2018, 18, 36. [Google Scholar] [CrossRef]
- Lee, K.W.; Ching, S.M.; Ramachandran, V.; Yee, A.; Hoo, F.K.; Chia, Y.C.; Veettil, S.K. Prevalence and risk factors of gestational diabetes mellitus in Asia: A systematic review and meta-analysis. BMC Pregnancy Childbirth 2018, 18, 1–20. [Google Scholar] [CrossRef] [Green Version]
- Sone, H. Clinical and pathophysiological features of Japanese patients with type 2 diabetes mellitus and their risk factors for diabetic complication. Nihon Rinsho. Jpn. J. Clin. Med. 2015, 73, 1979–1987. [Google Scholar]
- Chan, J.C.Y.; Chee, M.L.; Tan, N.Y.Q.; Cheng, C.-Y.; Wong, T.Y.; Sabanayagam, C. Differential effect of body mass index on the incidence of diabetes and diabetic retinopathy in two Asian populations. Nutr. Diabetes 2018, 8, 16. [Google Scholar] [CrossRef] [Green Version]
- Unger, R.H.; Clark, G.O.; Scherer, P.E.; Orci, L. Lipid homeostasis, lipotoxicity and the metabolic syndrome. Biochim. Biophys. Acta (BBA)-Mol. Cell Biol. Lipids 2010, 1801, 209–214. [Google Scholar] [CrossRef] [PubMed]
- Yabe, D.; Seino, Y.; Fukushima, M.; Seino, S. β Cell Dysfunction Versus Insulin Resistance in the Pathogenesis of Type 2 Diabetes in East Asians. Curr. Diabetes Rep. 2015, 15, 1–9. [Google Scholar] [CrossRef] [Green Version]
- Cheng, H.; Weiping, J. Diabetes in China: Epidemiology and Genetic Risk Factors and Their Clinical Utility in Personalized Medication. Diabetes 2018, 67, 3. [Google Scholar]
- International Diabetes Federation. IDF Diabetes Atlas, 8th ed.; International Diabetes Federation: Brussels, Belgium, 2017. [Google Scholar]
- Chawla, A.; Chawla, R.; Jaggi, S. Microvasular and macrovascular complications in diabetes mellitus: Distinct or continuum? Indian J. Endocrinol. Metab. 2016, 20, 546–551. [Google Scholar] [CrossRef] [PubMed]
- Coope, A.; Torsoni, A.S.; Velloso, L.A. Mechanisms in Endocrinology: Metabolic and inflammatory pathways on the pathogenesis of type 2 diabetes. Eur. J. Endocrinol. 2016, 174, R175–R187. [Google Scholar] [CrossRef] [Green Version]
- Sanada, F.; Taniyama, Y.; Muratsu, J.; Otsu, R.; Shimizu, H.; Rakugi, H.; Morishita, R. Source of Chronic Inflammation in Aging. Front. Cardiovasc. Med. 2018, 5, 12. [Google Scholar] [CrossRef] [Green Version]
- Eguchi, K.; Nagai, R. Islet inflammation in type 2 diabetes and physiology. J. Clin. Investig. 2017, 127, 14–23. [Google Scholar] [CrossRef] [Green Version]
- Lascar, N.; Afzal, I.; Nevill, A.M.; Shabir, K.; Ioannis Kyrou Brown, J.E.; Bellary, S. Increased Circulating Levels of Inflammatory Markers in A Cohort of Adults with Youth-Onset Type 2 Diabetes Mellitus. J. Diabetes Metab. 2020, 11, 853. [Google Scholar]
- Chaudhury, A.; Duvoor, C.; Reddy Dendi, V.S.; Kraleti, S.; Chada, A.; Ravilla, R.; Mirza, W. Clinical review of antidiabetic drugs: Implications for type 2 diabetes mellitus management. Front. Endocrinol. 2017, 8, 6. [Google Scholar] [CrossRef] [Green Version]
- Chan, J.C.; Bunnag, P.; Chan, S.P.; Tan, I.T.; Tsai, S.-T.; Gao, L.; Landgraf, W. Glycaemic responses in Asian and non-Asian people with type 2 diabetes initiating insulin glargine 100 units/mL: A patient-level pooled analysis of 16 randomised controlled trials. Diabetes Res. Clin. Pract. 2017, 135, 199–205. [Google Scholar] [CrossRef] [PubMed]
- Ni, L.; Yuan, C.; Chen, G.; Zhang, C.; Wu, X. SGLT2i: Beyond the glucose-lowering effect. Cardiovasc. Diabetol. 2020, 19, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Yang, L.; Zhang, L.; He, H.; Zhang, M.; An, Z. Efficacy and Safety of Sodium-Glucose Cotransporter 2 Inhibitors in East Asians with Type 2 Diabetes: A Systematic Review and Meta-Analysis. Diabetes Ther. 2019, 10, 1921–1934. [Google Scholar] [CrossRef] [Green Version]
- Frampton, J.E. Empagliflozin: A Review in Type 2 Diabetes. Drugs 2018, 78, 1037–1048. [Google Scholar] [CrossRef]
- Zelniker, T.A.; Wiviott, S.D.; Raz, I.; Im, K.; Goodrich, E.L.; Bonaca, M.P. SGLT2 inhibitors for primary and secondary prevention of cardiovascular and renal outcomes in type 2 diabetes: A systematic review and meta-analysis of cardiovascular outcome trials. Lancet 2019, 393, 31–39. [Google Scholar] [CrossRef]
- Sjöström, C.D.; Johansson, P.; Ptaszynska, A.; List, J.; Johnsson, E. Dapagliflozin lowers blood pressure in hypertensive and non-hypertensive patients with type 2 diabetes. Diabetes Vasc. Dis. Res. 2015, 12, 352–358. [Google Scholar] [CrossRef] [Green Version]
- Pancholia, A. Sodium-glucose cotransporter-2 inhibition for the reduction of cardiovascular events in high-risk patients with diabetes mellitus. Indian Heart J. 2018, 70, 915–921. [Google Scholar] [CrossRef]
- Merovci, A.; Mari, A.; Solis, C.; Xiong, J.; Daniele, G.; Chavez-Velazquez, A.; DeFronzo, R.A. Dapagliflozin lowers plasma glucose concentration and improves β-cell function. J. Clin. Endocrinol. Metabolism. 2015, 100, 1927–1932. [Google Scholar] [CrossRef] [Green Version]
- Jurczak, M.J.; Saini, S.; Ioja, S.; Costa, D.K.; Udeh, N.; Zhao, X.; Whaley, J.M.; Kibbey, R.G. SGLT2 knockout prevents hyperglycemia and is associated with reduced pancreatic β-cell death in genetically obese mice. Islets 2018, 10, 181–189. [Google Scholar] [CrossRef]
- Bonner, C.; Kerr-Conte, J.J.; Gmyr, V.V.; Queniat, G.G.; Moerman, E.E.; Thévenet, J.J.; Beaucamps, C.C.; Delalleau, N.N.; Popescu, I.I.; Malaisse, W.J.; et al. Inhibition of the glucose transporter SGLT2 with dapagliflozin in pancreatic alpha cells triggers glucagon secretion. Nat. Med. 2015, 21, 512–517. [Google Scholar] [CrossRef] [PubMed]
- E Inzucchi, S.; Zinman, B.; Wanner, C.; Ferrari, R.; Fitchett, D.; Hantel, S.; Espadero, R.-M.; Woerle, H.-J.; Broedl, U.C.; Johansen, O.E. SGLT-2 inhibitors and cardiovascular risk: Proposed pathways and review of ongoing outcome trials. Diabetes Vasc. Dis. Res. 2015, 12, 90–100. [Google Scholar] [CrossRef] [Green Version]
- Sonesson, C.; Johansson, P.A.; Johnsson, E.; Gause-Nilsson, I. Cardiovascular effects of dapagliflozin in patients with type 2 diabetes and different risk categories: A meta-analysis. Cardiovasc. Diabetol. 2016, 15, 37. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Inzucchi, S.E.; Bergenstal, R.M.; Buse, J.B.; Diamant, M.; Ferrannini, E.; Nauck, M.; Peters, A.L.; Tsapas, A.; Wender, R.; Matthews, D.R. Management of Hyperglycemia in Type 2 Diabetes, 2015: A Patient-Centered Approach: Update to a Position Statement of the American Diabetes Association and the European Association for the Study of Diabetes. Diabetes Care 2014, 38, 140–149. [Google Scholar] [CrossRef] [Green Version]
- Araki, E.; Onishi, Y.; Asano, M.; Kim, H.; Ekholm, E.; Johnsson, E.; Yajima, T. Efficacy and safety of dapagliflozin in addition to insulin therapy in Japanese patients with type 2 diabetes: Results of the interim analysis of 16-week double-blind treatment period. J. Diabetes Investig. 2016, 7, 555–564. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Handelsman, Y.; Bloomgarden, Z.T.; Grunberger, G.; Umpierrez, G.; Zimmerman, R.S.; Bailey, T.S.; Blonde, L.; Bray, G.A.; Cohen, A.J.; Dagogo-Jack, S.; et al. American Association Of Clinical Endocrinologists And American College Of Endocrinology -Clinical Practice Guidelines For Developing A Diabetes Mellitus Comprehensive Care Plan—2015. Endocr. Pract. 2015, 21, 1–87. [Google Scholar] [CrossRef] [Green Version]
- Watts, N.B.; Bilezikian, J.P.; Usiskin, K.; Edwards, R.; Desai, M.; Law, G.; Meininger, G. Effects of Canagliflozin on Fracture Risk in Patients With Type 2 Diabetes Mellitus. J. Clin. Endocrinol. Metab. 2016, 101, 157–166. [Google Scholar] [CrossRef] [Green Version]
- Shubrook, J.H.; Bokaie, B.B.; E Adkins, S. Empagliflozin in the treatment of type 2 diabetes: Evidence to date. Drug Des. Dev. Ther. 2015, 9, 5793–5803. [Google Scholar] [CrossRef] [Green Version]
- Yoon, K.-H.; Nishimura, R.; Lee, J.; Crowe, S.; Salsali, A.; Hach, T.; Woerle, H.J. Efficacy and safety of empagliflozin in patients with type 2 diabetes from Asian countries: Pooled data from four phase III trials. Diabetes Obes. Metab. 2016, 18, 1045–1049. [Google Scholar] [CrossRef]
- Yang, Y.; Chen, S.; Pan, H.; Zou, Y.; Wang, B.; Wang, G.; Zhu, H. Safety and efficiency of SGLT2 inhibitor combining with insulin in subjects with diabetes. Medicine 2017, 96, e6944. [Google Scholar] [CrossRef]
- Molugulu, N.; Yee, L.S.; Ye, Y.T.; Khee, T.C.; Nie, L.Z.; Yee, N.J.; Yee, T.K.; Liang, T.C.; Kesharwani, P. Systematic review of metformin monotherapy and dual therapy with sodium glucose co-transporter 2 inhibitor (SGLT-2) in treatment of type 2 diabetes mellitus. Diabetes Res. Clin. Pract. 2017, 132, 157–168. [Google Scholar] [CrossRef]
- Han, E.; Kim, A.; Lee, S.J.; Kim, J.Y.; Kim, J.H.; Lee, W.J.; Lee, B.W. Characteristics of dapagliflozin responders: A longitudinal, prospective, nationwide dapagliflozin surveillance study in Korea. Diabetes Ther. 2018, 9, 1689–1701. [Google Scholar] [CrossRef] [Green Version]
- Hong, A.R.; Koo, B.K.; Kim, S.W.; Yi, K.H.; Moon, M.K. Efficacy and safety of sodiumglucose cotransporter-2 inhibitors in Korean patients with type 2 diabetes mellitus in real-world clinical practice. Diabetes Metab. J. 2019, 43, 590–606. [Google Scholar] [CrossRef] [PubMed]
- Huang, C.H.; Huang, Y.Y.; Hsu, B.R. A short-term follow-up of glycemic and body weight changes in diabetic patients who replaced dipeptidyl peptidase-4 inhibitors with the sodium-glucose cotransporter 2. Ann. Saudi Med. 2018, 38, 420–426. [Google Scholar] [CrossRef] [PubMed]
- Huang, H.; Bell, K.F.; Gani, R.; Tugwell, C.W.; Eudicone, J.M.; Krukas-Hampel, M.R. A retrospective real-world study of dapagliflozin versus other oral antidiabetic drugs added to metformin in patients with type 2 diabetes. Am. J. Manag. Care. 2018, 24, S132–S137. [Google Scholar] [PubMed]
- Seino, Y.; Yabe, D.; Sasaki, T.; Fukatsu, A.; Imazeki, H.; Ochiai, H.; Sakai, S. Sodium-glucose cotransporter-2 inhibitor luseogliflozin added to glucagon-like peptide 1 receptor agonist liraglutide improves glycemic control with bodyweight and fat mass reductions in Japanese patients with type 2 diabetes: A 52-week, open-label, single-arm study. J. Diabetes Investig. 2018, 9, 332–340. [Google Scholar]
- Tobe, K.; Suganami, H.; Kaku, K. Sodium–glucose cotransporter 2 inhibitor, tofogliflozin, shows better improvements of blood glucose and insulin secretion in patients with high insulin levels at baseline. J. Diabetes Investig. 2017, 9, 862–869. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yabe, D.; Yasui, A.; Ji, L.; Lee, M.-K.; Ma, R.C.W.; Chang, T.-J.; Okamura, T.; Zeller, C.; Kaspers, S.; Lee, J.; et al. Safety and tolerability of empagliflozin in East Asian patients with type 2 diabetes: Pooled analysis of phase I–III clinical trials. J. Diabetes Investig. 2018, 10, 418–428. [Google Scholar] [CrossRef] [PubMed]
- Yang, W.; Ma, J.; Li, Y.; Li, Y.; Zhou, Z.; Kim, J.H.; Ptaszynska, A. Dapagliflozin as add-on therapy in Asian patients with type 2 diabetes inadequately controlled on insulin with or without oral antihyperglycemic drugs: A randomized controlled trial. J. Diabetes 2018, 10, 589–599. [Google Scholar] [CrossRef] [PubMed]
- Kawamori, R.; Haneda, M.; Suzaki, K.; Cheng, G.; Shiki, K.; Miyamoto, Y.; Solimando, F.; Lee, C.; Lee, J.; George, J. Empagliflozin as add-on to linagliptin in a fixed-dose combination in Japanese patients with type 2 diabetes: Glycaemic efficacy and safety profile in a 52-week, randomized, placebo-controlled trial. Diabetes Obes. Metab. 2018, 20, 2200–2209. [Google Scholar] [CrossRef]
- Kutoh, E.; Wada, A.; Murayama, T.; Hayashi, J. Two Glucose-Lowering Mechanisms of Canagliflozin Depending on Body Weight Changes in Drug-Naïve Subjects with Type 2 Diabetes. Drugs R D 2018, 18, 309–315. [Google Scholar] [CrossRef] [Green Version]
- Osonoi, T.; Gouda, M.; Kubo, M.; Arakawa, K.; Hashimoto, T.; Abe, M. Effect of Canagliflozin on Urinary Albumin Excretion in Japanese Patients with Type 2 Diabetes Mellitus and Microalbuminuria: A Pilot Study. Diabetes Technol. Ther. 2018, 20, 681–688. [Google Scholar] [CrossRef] [Green Version]
- Furukawa, S.; Miyake, T.; Senba, H.; Sakai, T.; Furukawa, E.; Yamamoto, S.; Niiya, T.; Matsuura, B.; Hiasa, Y. The effectiveness of dapagliflozin for sleep-disordered breathing among Japanese patients with obesity and type 2 diabetes mellitus. Endocr. J. 2018, 65, 953–961. [Google Scholar] [CrossRef] [Green Version]
- Han, K.A.; Chon, S.; Chung, C.H.; Lim, S.; Lee, K.W.; Baik, S.; Lee, M.K. Efficacy and safety of ipragliflozin as an add-on therapy to sitagliptin and metformin in Korean patients with inadequately controlled type 2 diabetes mellitus: A randomized controlled trial. Diabetes Obes. Metab. 2018, 20, 2408–2415. [Google Scholar] [CrossRef] [PubMed]
- Lu, C.H.; Min, K.W.; Chuang, L.M.; Kokubo, S.; Yoshida, S.; Cha, B.S. Efficacy, safety, and tolerability of ipragliflozin in Asian patients with type 2 diabetes mellitus and inadequate glycemic control with metformin: Results of a phase 3 randomized, placebo-controlled, double-blind, multicenter trial. J. Diabetes Investig. 2016, 7, 366–373. [Google Scholar] [CrossRef]
- Zinman, B.; Wanner, C.; Lachin, J.M.; Fitchett, D.; Bluhmki, E.; Hantel, S.; Inzucchi, S.E. Empagliflozin, cardiovascular outcomes, and mortality in type 2 diabetes. N. Engl. J. Med. 2015, 373, 2117–2128. [Google Scholar] [CrossRef]
- Baglioni, P.; Wiviott, S.D.; Raz, I.; Sabatine, M.S.; Akinci, B. Dapagliflozin and Cardiovascular Outcomes in Type 2 Diabetes. N. Engl. J. Med. 2019, 380, 1880–1881. [Google Scholar] [CrossRef]
- Neal, B.; Perkovic, V.; Matthews, D.R. Canagliflozin and cardiovascular and renal events in type 2 diabetes. N. Engl. J. Med. 2017, 377, 2099. [Google Scholar] [CrossRef] [PubMed]
- Kashiwagi, A.; Araki, S.; Maegawa, H. Sodium-glucose cotransporter 2 inhibitors represent a paradigm shift in the prevention of heart failure in type 2 diabetes patients. J. Diabetes Investig. 2021, 12, 6–20. [Google Scholar] [CrossRef] [PubMed]
- Shah, K.S.; Fang, J.C. Sodium-Glucose Cotransporter 2 Inhibitors in Heart Failure. Annu. Rev. Pharmacol. Toxicol. 2021, 62. [Google Scholar] [CrossRef] [PubMed]
- Dekkers, C.C.J.; Sjöström, C.D.; Greasley, P.J.; Cain, V.; Boulton, D.W.; Heerspink, H.J.L. Effects of the sodium-glucose co-transporter-2 inhibitor dapagliflozin on estimated plasma volume in patients with type 2 diabetes. Diabetes Obes. Metab. 2019, 21, 2667–2673. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Nojima, T.; Matsubayashi, Y.; Yoshida, A.; Suganami, H.; Abe, T.; Ishizawa, M.; Sone, H. Influence of an SGLT2 inhibitor, tofogliflozin, on the resting heart rate in relation to adipose tissue insulin resistance. Diabet. Med. 2020, 37, 1316–1325. [Google Scholar] [CrossRef]
- Núñez, J.; Palau, P.; Domínguez, E.; Mollar, A.; Núñez, E.; Ramón, J.M.; Miñana, G.; Santas, E.; Fácila, L.; Górriz, J.L.; et al. Early effects of empagliflozin on exercise tolerance in patients with heart failure: A pilot study. Clin. Cardiol. 2018, 41, 476–480. [Google Scholar] [CrossRef]
- Carbone, S.; Canada, J.M.; Billingsley, H.E.; Kadariya, D.; Dixon, D.L.; Trankle, C.R.; Abbate, A. Effects of empagliflozin on cardiorespiratory fitness and significant interaction of loop diuretics. Diabetes Obes. Metab. 2018, 20, 2014–2018. [Google Scholar] [CrossRef] [Green Version]
- Packer, M. SGLT2 Inhibitors Produce Cardiorenal Benefits by Promoting Adaptive Cellular Reprogramming to Induce a State of Fasting Mimicry: A Paradigm Shift in Understanding Their Mechanism of Action. Diabetes Care 2020, 43, 508–511. [Google Scholar] [CrossRef] [Green Version]
- Scheen, A.J. Effect of SGLT2 Inhibitors on the Sympathetic Nervous System and Blood Pressure. Curr. Cardiol. Rep. 2019, 21, 70. [Google Scholar] [CrossRef]
- Kario, K.; Ferdinand, K.C.; O’Keefe, J.H. Control of 24-hour blood pressure with SGLT2 inhibitors to prevent cardiovascular disease. Prog. Cardiovasc. Dis. 2020, 63, 249–262. [Google Scholar] [CrossRef] [PubMed]
- Li, H.; Shin, S.E.; Seo, M.S. The anti-diabetic drug dapagliflozin induces vasodilation via activation of PKG and Kv channels. Life Sci. 2018, 197, 46–55. [Google Scholar] [CrossRef]
- Packer, M.; Anker, S.D.; Butler, J.; Filippatos, G.; Zannad, F. Effects of sodiumglucose cotransporter 2 inhibitors for the treatment of patients with heart failure: Proposal of a novel mechanism of action. JAMA Cardiol. 2017, 2, 1025–1029. [Google Scholar] [CrossRef]
- Mabillard, H.; Sayer, J.A. SGLT2 inhibitors–a potential treatment for Alport syndrome. Clin. Sci. 2020, 134, 379–388. [Google Scholar] [CrossRef]
- Kaplan, A.; Abidi, E.; El-Yazbi, A.; Eid, A.; Booz, G.W.; Zouein, F.A. Direct cardiovascular impact of SGLT2 inhibitors: Mechanisms and effects. Hear. Fail. Rev. 2018, 23, 419–437. [Google Scholar] [CrossRef]
- Mathieu, C.; Van Den Mooter, L.; Eeckhout, B. Empagliflozin in type 1 diabetes. Diabetes Metab Syndr. Obes. 2019, 12, 1555–1561. [Google Scholar] [CrossRef] [Green Version]
- Vallon, V.; Thomson, S.C. Targeting renal glucose reabsorption to treat hyperglycaemia: The pleiotropic effects of SGLT2 inhibition. Diabetologia 2017, 60, 215–225. [Google Scholar] [CrossRef] [PubMed]
- Lee, T.M.; Chang, N.C.; Lin, S.Z. Dapagliflozin, a selective SGLT2 Inhibitor, attenuated cardiac fibrosis by regulating the macrophage polarization via STAT3 signaling in infarcted rat hearts. Free Radic. Biol. Med. 2017, 104, 298–310. [Google Scholar] [CrossRef] [PubMed]
- Lim, V.G.; Bell, R.M.; Arjun, S.; Kolatsi-Joannou, M.; Long, D.A.; Yellon, D.M. SGLT2 inhibitor, canagliflozin, attenuates myocardial infarction in the diabetic and nondiabetic heart. J. Am. Coll. Cardiol. Basic. Trans. Science. 2019, 4, 15–26. [Google Scholar] [CrossRef]
- Sato, T.; Aizawa, Y.; Yuasa, S.; Kishi, S.; Fuse, K.; Fujita, S.; Ikeda, Y.; Kitazawa, H.; Takahashi, M.; Sato, M.; et al. The effect of dapagliflozin treatment on epicardial adipose tissue volume. Cardiovasc. Diabetol. 2018, 17, 1–9. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lin, B.; Koibuchi, N.; Hasegawa, Y.; Sueta, D.; Toyama, K.; Uekawa, K.; Ma, M.; Nakagawa, T.; Kusaka, H.; Kim-Mitsuyama, S. Glycemic control with empagliflozin, a novel selective SGLT2 inhibitor, ameliorates cardiovascular injury and cognitive dysfunction in obese and type 2 diabetic mice. Cardiovasc. Diabetol. 2014, 13, 1–15. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Verma, S.; Rawat, S.; Ho, K.L.; Wagg, C.S.; Zhang, L.; Teoh, H.; Lopaschuk, G.D. Empagliflozin increases cardiac energy production in diabetes: Novel translational insights into the heart failure benefits of SGLT2 inhibitors. J. Am. Coll. Cardiol. Basic Trans. Sci. 2018, 3, 575–587. [Google Scholar]
- Cavender, M.A.; Norhammar, A.; Birkeland, K.I.; Jorgensen, M.E.; Wilding, J.P.; Khunti, K.; Fu, A.Z.; Bodegard, J.; Blak, B.T.; Wittbrodt, E.; et al. SGLT-2 inhibitors and cardiovascular risk: An analysis of CVD-REAL. J. Am. Coll. Cardiol. 2018, 71, 2497–2506. [Google Scholar] [CrossRef]
- Norhammar, A.; Bodegard, J.; Nystrom, T.; Thuresson, M.; Nathanson, D.; Eriksson, J.W. Dapagliflozin and cardiovascular mortality and disease outcomes in a population with type 2 diabetes similar to that of the DECLARE-TIMI 58 trial: A nationwide observational study. Diabetes Obes. Metabolism. 2019, 21, 1136–1145. [Google Scholar] [CrossRef] [Green Version]
- Wiviott, S.D.; Raz, I.; Bonaca, M.P.; Mosenzon, O.; Kato, E.T.; Cahn, A.; Silverman, M.G.; Zelniker, T.A.; Kuder, J.F.; Murphy, S.A.; et al. Dapagliflozin and cardiovascular outcomes in type 2 diabetes. N. Engl. J. Med. 2019, 380, 347–357. [Google Scholar] [CrossRef]
- Cai, X.; Yang, W.; Gao, X.; Chen, Y.; Zhou, L.; Zhang, S.; Han, X.; Wenjia, Y. The Association Between the Dosage of SGLT2 Inhibitor and Weight Reduction in Type 2 Diabetes Patients: A Meta-Analysis. Obesity 2017, 26, 70–80. [Google Scholar] [CrossRef] [Green Version]
- Woo, V.C. Cardiovascular Effects of Sodium-Glucose Cotransporter-2 Inhibitors in Adults with Type 2 Diabetes. Can. J. Diabetes 2019, 44, 61–67. [Google Scholar] [CrossRef] [Green Version]
- Kim, E.J.; Choi, M.J.; Lee, J.H.; Oh, J.E.; Seo, J.W.; Lee, Y.K.; Koo, J.R. Extracellular fluid/intracellular fluid volume ratio as a novel risk indicator for all-cause mortality and cardiovascular disease in hemodialysis patients. PLoS ONE 2017, 12, e0170272. [Google Scholar] [CrossRef] [Green Version]
- Rosenstein, R.; Hough, A. Empagliflozin, cardiovascular outcomes, and mortality in type 2 diabetes. N. Engl. J. Med. 2016, 374, 1093–1094. [Google Scholar] [PubMed]
| Author; Region | Study Design and Population | Drug Therapy | Key Findings |
|---|---|---|---|
| Yang et al. China [54] | 24-week, randomized, Phase 3, double-blind, placebo-controlled (n = 272) | Dapagliflozin | Significant reduction with Dapagliflozin HbA1c Fasting plasma glucose Body weight |
| Kawamori et al. Japanese [55] | 52-week, randomized, double-blind, placebocontrolled study (n = 433) | Empagliflozin | Significant reduction with Empagliflozin HbA1c Fasting plasma glucose Body weight Systolic blood pressure |
| Kutoh et al. Japanese [56] | 3-month, observational (n = 36) | Canagliflozin | Significant reduction with Canagliflozin HbA1c Free fatty acid Body weight insulin resistance |
| Osonoi et al. Japanese [57] | 12-week, open label study (n = 20) | Canagliflozin | Significant reduction with Canagliflozin HbA1c Fasting plasma glucose Body weight |
| Furukawa et al. Japanese [58] | 24-week, open label study (n = 104) | Dapagliflozin | Significant reduction with Dapagliflozin HbA1c Fasting plasma glucose Body weight low-density lipoprotein cholesterol |
| Han et al. Korean [59] | 24-week, randomized, placebo-controlled, double-blind study (n = 104) | Ipragliflozin | Sustained reduction with Ipragliflozin HbA1c Fasting plasma glucose Body weight Fasting serum insulin |
| Chieh-Hsiang Lu et al. Korea and Taiwan [60] | 24-week, multicenter, placebo-controlled, double-blind, parallel-group (n = 87) | Ipragliflozin | Significant reduction with Ipragliflozin HbA1 Fasting plasma glucose Body weight and waist circumference low-density lipoprotein cholesterol |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Afzal, M.; Al-Abbasi, F.A.; Nadeem, M.S.; Alshehri, S.; Ghoneim, M.M.; Imam, S.S.; Almalki, W.H.; Kazmi, I. Sodium-Glucose Cotransporter-2 Inhibitors Improve Cardiovascular Dysfunction in Type 2 Diabetic East Asians. Metabolites 2021, 11, 794. https://doi.org/10.3390/metabo11110794
Afzal M, Al-Abbasi FA, Nadeem MS, Alshehri S, Ghoneim MM, Imam SS, Almalki WH, Kazmi I. Sodium-Glucose Cotransporter-2 Inhibitors Improve Cardiovascular Dysfunction in Type 2 Diabetic East Asians. Metabolites. 2021; 11(11):794. https://doi.org/10.3390/metabo11110794
Chicago/Turabian StyleAfzal, Muhammad, Fahad A. Al-Abbasi, Muhammad Shahid Nadeem, Sultan Alshehri, Mohammed M. Ghoneim, Syed Sarim Imam, Waleed Hassan Almalki, and Imran Kazmi. 2021. "Sodium-Glucose Cotransporter-2 Inhibitors Improve Cardiovascular Dysfunction in Type 2 Diabetic East Asians" Metabolites 11, no. 11: 794. https://doi.org/10.3390/metabo11110794
APA StyleAfzal, M., Al-Abbasi, F. A., Nadeem, M. S., Alshehri, S., Ghoneim, M. M., Imam, S. S., Almalki, W. H., & Kazmi, I. (2021). Sodium-Glucose Cotransporter-2 Inhibitors Improve Cardiovascular Dysfunction in Type 2 Diabetic East Asians. Metabolites, 11(11), 794. https://doi.org/10.3390/metabo11110794








