A Systematic Review of Metabolite-to-Drug Ratios of Pharmaceuticals in Hair for Forensic Investigations
Abstract
1. Introduction
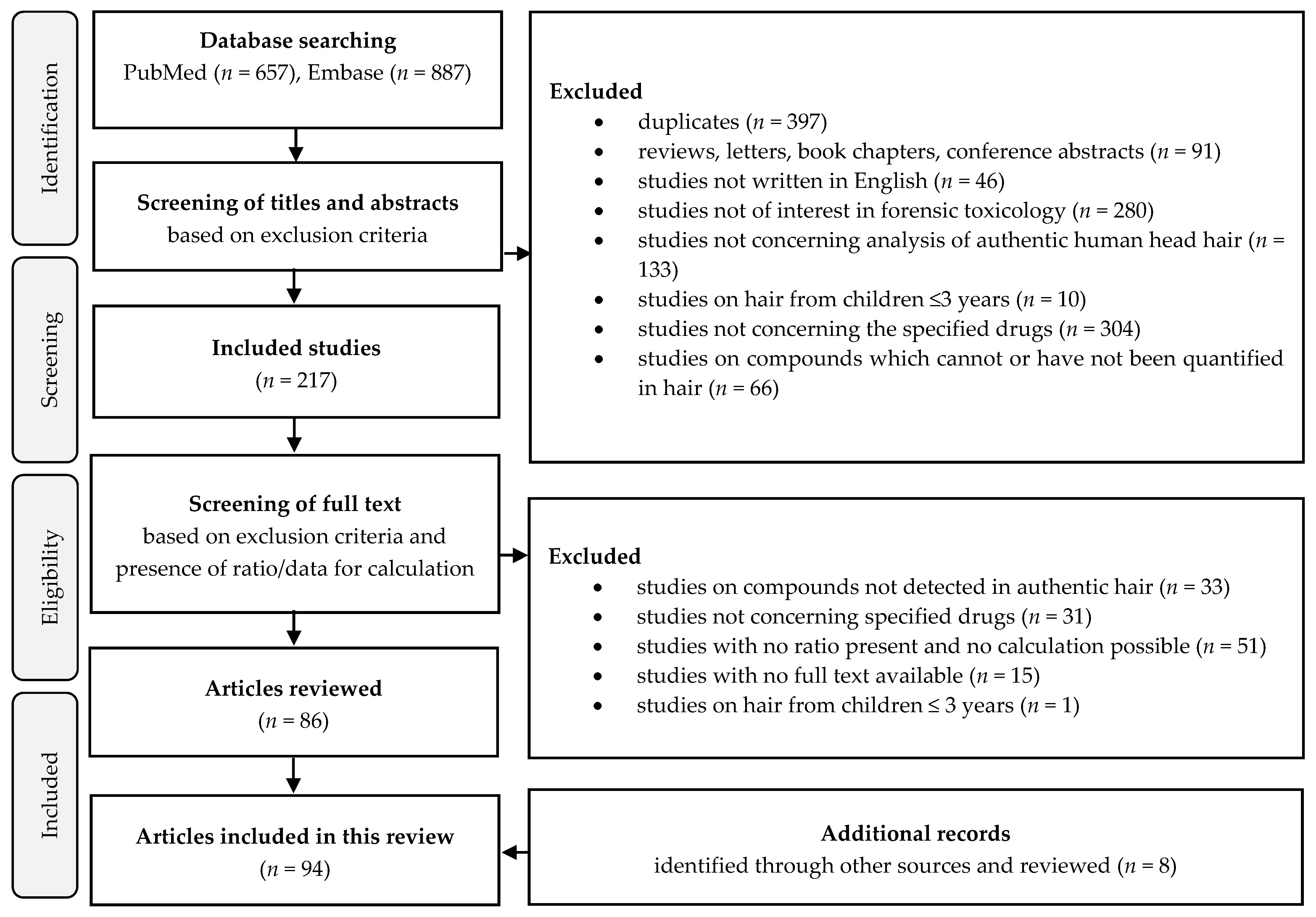
2. Methods
2.1. Information Sources and Search Strategy
2.2. Exclusion Criteria
- Studies not written in English;
- Reviews, letters, book chapters, or conference abstracts;
- Studies not of interest in forensic toxicology, e.g., cancer-related studies;
- Studies not concerning the analysis of authentic human head hair;
- Studies on hair from children ≤3 years old or unreported age;
- Studies not concerning antipsychotics, antidepressants, benzodiazepines, opioids/opiates (Except for morphine and codeine. See in Section 2.3);
- Studies not concerning other pharmaceuticals of interest in forensic toxicology, such as ketamine and methylphenidate;
- Studies on illegal drugs (New Psychoactive Substances (NPS)) or non-marketed drugs were excluded;
- Studies on compounds that cannot or have not been quantified in authentic hair (e.g., ethanol).
2.3. Study Selection
2.4. Data Collection
3. Results and Discussion
3.1. MD Ratios in Hair
3.2. Factors Affecting the MD Ratios in Hair
3.2.1. Inter- and Intraindividual Differences
3.2.2. Hair Characteristic
3.2.3. Drug Intake
3.2.4. Pre-Analytical Workflow and Analytics
3.3. Limitations
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Kintz, P. Value of hair analysis in postmortem toxicology. Forensic Sci. Int. 2004, 142, 127–134. [Google Scholar] [CrossRef]
- Hansen, S.H.; Pedersen-Bjergaard, S.; Reubsaet, L.; Grønhaug Halvorsen, T.; Gjelstad, A.; Jørgensen, M.; Kall, M.A. Bioanalysis of Pharmaceuticals: Sample Preparation, Separation Techniques and Mass Spectrometry, 1st ed.; Wiley: Hoboken, NJ, USA, 2015; ISBN 9781118716830. [Google Scholar]
- Pragst, F.; Balikova, M.A. State of the art in hair analysis for detection of drug and alcohol abuse. Clin. Chim. Acta 2006, 370, 17–49. [Google Scholar] [CrossRef]
- Wennig, R. Potential problems with the interpretation of hair analysis results. Forensic Sci. Int. 2000, 107, 5–12. [Google Scholar] [CrossRef]
- Cooper, G.A.A.; Kronstrand, R.; Kintz, P. Society of hair testing guidelines for drug testing in hair. Forensic Sci. Int. 2012, 218, 20–24. [Google Scholar] [CrossRef]
- Kintz, P. Segmental hair analysis can demonstrate external contamination in postmortem cases. Forensic Sci. Int. 2012, 215, 73–76. [Google Scholar] [CrossRef]
- Society of Hair Testing. Statement of the society of hair testing concerning the examination of drugs in human hair. Forensic Sci. Int. 1997, 84, 3–6. [Google Scholar]
- Nielsen, M.K.K.; Johansen, S.S.; Linnet, K. Evaluation of poly-drug use in methadone-related fatalities using segmental hair analysis. Forensic Sci. Int. 2015, 248, 134–139. [Google Scholar] [CrossRef]
- Zhuo, Y.; Xiang, P.; Wu, J.; Wang, X. Segmental hair analysis for flunitrazepam and 7-aminoflunitrazepam in users: A comparison to existing literature. Forensic Sci. Res. 2020, 1–9. [Google Scholar] [CrossRef]
- Morris-Kukoski, C.L.; Montgomery, M.A.; Hammer, R.L. Analysis of extensively washed hair from cocaine users and drug chemists to establishnew reporting criteria. J. Anal. Toxicol. 2014, 38, 628–636. [Google Scholar] [CrossRef]
- Han, E.; Park, Y.; Yang, W.; Lee, J.; Lee, S.; Kim, E.; Lim, M.; Chung, H. The study of metabolite-to-parent drug ratios of methamphetamine and methylenedioxymethamphetamine in hair. Forensic Sci. Int. 2006, 161, 124–129. [Google Scholar] [CrossRef]
- Franz, T.; Scheufler, F.; Stein, K.; Uhl, M.; Dame, T.; Schwarz, G.; Sachs, H.; Skopp, G.; Musshoff, F. Determination of hydroxy metabolites of cocaine from hair samples and comparison with street cocaine samples. Forensic Sci. Int. 2018, 288, 223–226. [Google Scholar] [CrossRef]
- Ropero-Miller, J.D.; Huestis, M.A.; Stout, P.R. Cocaine analytes in human hair: Evaluation of concentration ratios in different cocaine sources, drug-user populations and surface-contaminated specimens. J. Anal. Toxicol. 2012, 36, 390–398. [Google Scholar] [CrossRef]
- Methling, M.; Krumbiegel, F.; Hartwig, S. Hair analysis of antidepressants and antipsychotics—Overview of quantitative data. Drug Test. Anal. 2020, 12, 659–676. [Google Scholar] [CrossRef]
- Vogliardi, S.; Favretto, D.; Tucci, M.; Stocchero, G.; Ferrara, S.D. Simultaneous LC-HRMS determination of 28 benzodiazepines and metabolites in hair. Anal. Bioanal. Chem. 2011, 400, 51–67. [Google Scholar] [CrossRef]
- Moher, D.; Liberati, A.; Tetzlaff, J.; Altman, D.G.; Group, T.P. Preferred reporting items for systematic reviews and meta-analyses: The PRISMA Statement. PLoS Med. 2009, 6, e1000097. [Google Scholar] [CrossRef]
- Liberati, A.; Altman, D.G.; Tetzlaff, J.; Mulrow, C.; Gøtzsche, P.C.; Ioannidis, J.P.A.; Clarke, M.; Devereaux, P.J.; Kleijnen, J.; Moher, D. The PRISMA statement for reporting systematic reviews and meta-analyses of studies that evaluate health care interventions: Explanation and elaboration. BMJ 2009, 339, b2700. [Google Scholar] [CrossRef]
- Methling, M.; Krumbiegel, F.; Alameri, A.; Hartwig, S.; Parr, M.; Tsokos, M. Concentrations of antidepressants, antipsychotics, and benzodiazepines in hair samples from postmortem cases. SN Compr. Clin. Med. 2020, 2, 284–300. [Google Scholar] [CrossRef]
- Fernández, M.D.M.R.; Wille, S.M.R.R.; Hill, V.; Samyn, N.; Fernádez, M.d.M.; Wille, S.M.R.R.; Hill, V.; Samyn, N. Determination of antidepressants in hair via UHPLC-MS/MS as a complementary informative tool for clinical and forensic toxicological assessments. Ther. Drug Monit. 2016, 38, 751–760. [Google Scholar] [CrossRef]
- Licata, M.; Rustichelli, C.; Palazzoli, F.; Ferrari, A.; Baraldi, C.; Vandelli, D.; Verri, P.; Marchesi, F.; Silingardi, E. Hair testing in clinical setting: Simultaneous determination of 50 psychoactive drugs and metabolites in headache patients by LC tandem MS. J. Pharm. Biomed. Anal. 2016, 126, 14–25. [Google Scholar] [CrossRef]
- Fisichella, M.; Morini, L.; Sempio, C.; Groppi, A. Validation of a multi-analyte LC-MS/MS method for screening and quantification of 87 psychoactive drugs and their metabolites in hair. Anal. Bioanal. Chem. 2014, 406, 3497–3506. [Google Scholar] [CrossRef]
- Nielsen, M.K.K.; Johansen, S.S.; Dalsgaard, P.W.; Linnet, K. Simultaneous screening and quantification of 52 common pharmaceuticals and drugs of abuse in hair using UPLC-TOF-MS. Forensic Sci. Int. 2010, 196, 85–92. [Google Scholar] [CrossRef]
- Pragst, F.; Rothe, M.; Hunger, J.; Thor, S. Structural and concentration effects on the deposition of tricyclic antidepressants in human hair. Forensic Sci. Int. 1997, 84, 225–236. [Google Scholar] [CrossRef]
- Ishiyama, I.; Nagai, T.; Toshida, S. Detection of basic drugs (methamphetamine, antidepressants, and nicotine) from human hair. J. Forensic Sci. 1983, 28, 380–385. [Google Scholar] [CrossRef]
- Wille, S.M.R.; De Letter, E.A.; Piette, M.H.A.; Van Overschelde, L.K.; Van Peteghem, C.H.; Lambert, W.E. Determination of antidepressants in human postmortem blood, brain tissue, and hair using gas chromatography-mass spectrometry. Int. J. Legal Med. 2009, 123, 451–458. [Google Scholar] [CrossRef]
- Kłys, M.; Scisłowski, M.; Rojek, S.; Kołodziej, J. A fatal clomipramine intoxication case of a chronic alcoholic patient: Application of postmortem hair analysis method of clomipramine and ethyl glucuronide using LC/APCI/MS. Leg. Med. 2005, 7, 319–325. [Google Scholar] [CrossRef]
- Krumbiegel, F.; Hastedt, M.; Westendorf, L.; Niebel, A.; Methling, M.; Parr, M.K.; Tsokos, M. The use of nails as an alternative matrix for the long-term detection of previous drug intake: Validation of sensitive UHPLC-MS/MS methods for the quantification of 76 substances and comparison of analytical results for drugs in nail and hair samples. Forensic Sci. Med. Pathol. 2016, 12, 416–434. [Google Scholar] [CrossRef]
- Wang, X.; Johansen, S.S.; Nielsen, M.K.K.; Linnet, K. Hair analysis in toxicological investigation of drug-facilitated crimes in Denmark over a 8-year period. Forensic Sci. Int. 2018, 285, e1–e12. [Google Scholar] [CrossRef]
- Couper, F.; Mclntyre, I.; Drummer, O. Detection of antidepressant and antipsychotic drugs in postmortem human scalp hair. J. Forensic Sci. 1995, 40, 87–90. [Google Scholar] [CrossRef]
- Kintz, P.; Raul, S.; Ameline, A. Evidence of repeated mirtazapine poisoning in children by hair analysis. J. Forensic Sci. 2021, 66, 1165–1170. [Google Scholar] [CrossRef]
- Wang, X.; Zhuo, Y.; Tang, X.; Qiang, H.; Liu, W.; Wu, H.; Xiang, P.; Duan, G.; Shen, M. Segmental analysis of antidepressant and antipsychotic drugs in the hair of schizophrenic patients. Drug Test. Anal. 2019, 12, 1–13. [Google Scholar] [CrossRef]
- Marchei, E.; Palmi, I.; Pichini, S.; Pacifici, R.; Anton Airaldi, I.-R.; Costa Orvay, J.A.; Garcia Serra, J.; Bonet Serra, B.; Garcia-Algar, O. Segmental hair testing to disclose chronic exposure to psychoactive drugs. Adicciones 2016, 28, 158–162. [Google Scholar] [CrossRef]
- Pichini, S.; Cortes, L.; Marchei, E.; Solimini, R.; Pacifici, R.; Gomez-Roig, M.D.; García-Algar, O. Ultra-high-pressure liquid chromatography tandem mass spectrometry determination of antidepressant and anxiolytic drugs in neonatal meconium and maternal hair. J. Pharm. Biomed. Anal. 2016, 118, 9–16. [Google Scholar] [CrossRef]
- Rygaard, K.; Linnet, K.; Banner, J.; Johansen, S.S. Concentrations of aripiprazole and dehydroaripiprazole in hair segments from deceased individuals with mental disorders. Forensic Sci. Int. 2020, 317, 110523. [Google Scholar] [CrossRef]
- Günther, K.N.; Johansen, S.S.; Wicktor, P.; Banner, J.; Linnet, K. Segmental analysis of chlorprothixene and desmethylchlorprothixene in postmortem hair. J. Anal. Toxicol. 2018, 42, 642–649. [Google Scholar] [CrossRef]
- Kronstrand, R.; Roman, M.; Hedman, M.; Ahlner, J.; Dizdar, N. Dose–hair concentration relationship and pigmentation effects in patients on low-dose clozapine. Forensic Sci. Med. Pathol. 2007, 3, 107–114. [Google Scholar] [CrossRef]
- Günther, K.N.; Banner, J.; Linnet, K.; Johansen, S.S. Segmental hair analysis of olanzapine and N-desmethyl-olanzapine in postmortem hair from mentally ill patients by LC–MS/MS. J. Pharm. Biomed. Anal. 2020, 190, 113510. [Google Scholar] [CrossRef]
- Schneider, S.; Sibille, E.; Yegles, M.; Neels, H.; Wennig, R.; Mühe, A. Time resolved analysis of risperidone and 9-hydroxy-risperidone in hair using LC/MS-MS. J. Chromatogr. B Anal. Technol. Biomed. Life Sci. 2009, 877, 2589–2592. [Google Scholar] [CrossRef]
- Binz, T.M.; Yegles, M.; Schneider, S.; Neels, H.; Crunelle, C.L. Time resolved analysis of quetiapine and 7-OH-quetiapine in hair using LC/MS-MS. Forensic Sci. Int. 2014, 242, 200–203. [Google Scholar] [CrossRef]
- Wang, X.; Johansen, S.S.; Nielsen, M.K.K.; Linnet, K. Targeted analysis of 116 drugs in hair by UHPLC-MS/MS and its application to forensic cases. Drug Test. Anal. 2017, 9, 1137–1151. [Google Scholar] [CrossRef]
- Madry, M.M.; Kraemer, T.; Baumgartner, M.R. Large scale consumption monitoring of benzodiazepines and z-drugs by hair analysis. J. Pharm. Biomed. Anal. 2020, 183, 113151. [Google Scholar] [CrossRef]
- Shin, Y.; Kong, T.Y.; Cheong, J.C.; Kim, J.Y.; Lee, J.; Lee, H.S. Simultaneous determination of 75 abuse drugs including amphetamines, benzodiazepines, cocaine, opioids, piperazines, zolpidem and metabolites in human hair samples using liquid chromatography–tandem mass spectrometry. Biomed. Chromatogr. 2019, 33, e4600. [Google Scholar] [CrossRef]
- Chéze, M.; Duffort, G.; Deveaux, M.; Pépin, G. Hair analysis by liquid chromatography—Tandem mass spectrometry in toxicological investigation of drug-facilitated crimes: Report of 128 cases over the period June 2003–May 2004 in metropolitan Paris. Forensic Sci. Int. 2005, 153, 3–10. [Google Scholar] [CrossRef]
- Negrusz, A.; Moore, C.M.; Kern, J.L.; Philip, G.; Strong, M.J.; Levy, N.A. Quantitation of clonazepam and its major metabolite 7-aminoclonazepam in hair. J. Anal. Toxicol. 2000, 24, 614–620. [Google Scholar] [CrossRef]
- Lendoiro, E.; de Castro, A.; Jiménez-Morigosa, C.; Gomez-Fraguela, X.A.; López-Rivadulla, M.; Cruz, A. Usefulness of hair analysis and psychological tests for identification of alcohol and drugs of abuse consumption in driving license regranting. Forensic Sci. Int. 2018, 286, 239–244. [Google Scholar] [CrossRef]
- Morini, L.; Pozzi, F.; Groppi, A. Stability of benzodiazepines in hair after prolonged exposure to chlorinated water. Forensic Sci. Int. 2017, 278, 217–220. [Google Scholar] [CrossRef]
- Wang, X.; Johansen, S.S.; Zhang, Y.; Jia, J.; Rao, Y.; Jiang, F.; Linnet, K. Deposition of diazepam and its metabolites in hair following a single dose of diazepam. Int. J. Legal Med. 2017, 131, 131–141. [Google Scholar] [CrossRef]
- Kim, J.; Yum, H.; Jang, M.; Shin, I.; Yang, W.; Baeck, S.; Suh, J.H.; Lee, S.; Han, S.B. A comprehensive and sensitive method for hair analysis in drug-facilitated crimes and incorporation of zolazepam and tiletamine into hair after a single exposure. Anal. Bioanal. Chem. 2016, 408, 251–263. [Google Scholar] [CrossRef]
- Maublanc, J.; Dulaurent, S.; Morichon, J.; King, L. Identification and quantification of 35 psychotropic drugs and metabolites in hair by LC-MS/MS: Application in forensic toxicology. Int. J. Leg. Med. 2015, 129, 259–268. [Google Scholar] [CrossRef]
- Lendoiro, E.; Quintela, Ó.; de Castro, A.; Cruz, A.; López-Rivadulla, M.; Concheiro, M. Target screening and confirmation of 35 licit and illicit drugs and metabolites in hair by LC-MSMS. Forensic Sci. Int. 2012, 217, 207–215. [Google Scholar] [CrossRef]
- Favretto, D.; Vogliardi, S.; Stocchero, G.; Nalesso, A.; Tucci, M.; Ferrara, S.D. High performance liquid chromatography-high resolution mass spectrometry and micropulverized extraction for the quantification of amphetamines, cocaine, opioids, benzodiazepines, antidepressants and hallucinogens in 2.5 mg hair samples. J. Chromatogr. A 2011, 1218, 6583–6595. [Google Scholar] [CrossRef]
- Kim, J.; Lee, S.; In, S.; Choi, H.; Chung, H. Validation of a simultaneous analytical method for the detection of 27 benzodiazepines and metabolites and zolpidem in hair using LC–MS/MS and its application to human and rat hair. J. Chromatogr. B 2011, 879, 878–886. [Google Scholar] [CrossRef]
- Lee, S.; Han, E.; In, S.; Choi, H.; Chung, H.; Chung, K.H. Determination of illegally abused sedative-hypnotics in hair samples from drug offenders. J. Anal. Toxicol. 2011, 35, 312–315. [Google Scholar] [CrossRef] [PubMed]
- Miller, E.I.; Wylie, F.M.; Oliver, J.S. Simultaneous detection and quantification of amphetamines, diazepam and its metabolites, cocaine and its metabolites, and opiates in hair by LC–ESI-MS–MS using a single extraction method. J. Anal. Toxicol. 2008, 32, 457–469. [Google Scholar] [CrossRef] [PubMed]
- Ariffin, M.M.; Miller, E.I.; Cormack, P.A.G.; Anderson, R.A. Molecularly imprinted solid-phase extraction of diazepam and its metabolites from hair samples. Anal. Chem. 2007, 79, 256–262. [Google Scholar] [CrossRef] [PubMed]
- Yegles, M.; Mersch, F.; Wennig, R. Detection of benzodiazepines and other psychotropic drugs in human hair by GC/MS. Forensic Sci. Int. 1997, 84, 211–218. [Google Scholar] [CrossRef]
- Negrusz, A.; Moore, C.; Deitermann, D.; Lewis, D.; Kaleciak, K.; Kronstrand, R.; Feeley, B.; Niedbala, R.S. Highly sensitive micro-plate enzyme immunoassay screening and NCI-GC-MS confirmation of flunitrazepam and its major metabolite 7-aminoflunitrazepam in hair. J. Anal. Toxicol. 1999, 23, 429–435. [Google Scholar] [CrossRef]
- Cirimele, V.; Kintz, P.; Staub, C.; Mangin, P. Testing human hair for flunitrazepam and 7-amino-flunitrazepam by GC/MS-NCI. Forensic Sci. Int. 1997, 84, 189–200. [Google Scholar] [CrossRef]
- Cirimele, V.; Kintz, P.; Mangin, P. Determination of chronic flunitrazepam abuse by hair analysis using GC-MS-NCi. J. Anal. Toxicol. 1996, 20, 20–22. [Google Scholar] [CrossRef][Green Version]
- Wiart, J.; Hakim, F.; Andry, A.; Eiden, C.; Drevin, G.; Lelièvre, B.; Rougé-maillart, C.; Decourcelle, M.; Allorge, D. Pitfalls of toxicological investigations in hair, bones and nails in extensively decomposed bodies: Illustration with two cases. Int. J. Leg. Med. 2020, 134, 1339–1344. [Google Scholar] [CrossRef]
- Kintz, P.; Cirimele, V.; Vayssette, F.; Mangin, P. Hair analysis for nordiazepam and oxazepam by gas chromatography-negative-ion chemical ionization mass spectrometry. J. Chromatogr. B Biomed. Sci. Appl. 1996, 677, 241–244. [Google Scholar] [CrossRef]
- Toyo, T.; Kanbori, M.; Kumaki, Y.; Nakahara, Y. Determination of triazolam involving its hydroxy metabolites in hair shaft and hair root by reversed-phase liquid chromatography with electrospray ionization mass spectrometry and application to human hair analysis. Anal. Biochem. 2001, 179, 172–179. [Google Scholar] [CrossRef]
- Hansen, S.L.; Johansen, S.S.; Nielsen, M.K.K.; Nilsson, G.; Kronstrand, R. Distribution of zopiclone and main metabolites in hair following a single dose. Forensic Sci. Int. 2020, 306, 110074. [Google Scholar] [CrossRef]
- Belivanis, S.; Tzatzarakis, M.N.; Vakonaki, E.; Kovatsi, L.; Mantsi, M.; Alegakis, A.; Kavvalakis, M.P.; Vynias, D.; Tsatsakis, A.M. Buprenorphine and nor-buprenorphine levels in head hair samples from former heroin users under Suboxone® treatment. Drug Test. Anal. 2014, 6 (Suppl. 1), 93–100. [Google Scholar] [CrossRef]
- Vincent, F.; Bessard, J.; Vacheron, J.; Mallaret, M.; Bessard, G. Determination of buprenorphine and norbuprenorphine in urine and hair by gas chromatography-mass spectrometry. J. Anal. Toxicol. 1999, 23, 270–279. [Google Scholar] [CrossRef] [PubMed]
- Kintz, P.; Cirimele, V.; Edel, Y.; Jamey, C. Hair analysis for buprenorphine and its dealkylated metabolite by RIA and confirmation by LC/ECD. J. Forensic Sci. 1994, 39, 1497–1503. [Google Scholar] [CrossRef]
- Kim, J.; Ji, D.; Kang, S.; Park, M.; Yang, W.; Kim, E.; Choi, H.; Lee, S. Simultaneous determination of 18 abused opioids and metabolites in human hair using LC–MS/MS and illegal opioids abuse proven by hair analysis. J. Pharm. Biomed. Anal. 2014, 89, 99–105. [Google Scholar] [CrossRef]
- Kim, J.Y.; Suh, S.I.; Paeng, K.; In, M.K. Determination of dextromethorphan and its metabolite dextrorphan in human hair by gas chromatography–mass spectrometry. Chromatographia 2004, 60, 703–707. [Google Scholar] [CrossRef]
- Salomone, A.; Bigiarini, R.; Palamar, J.J.; McKnight, C.; Vinsick, L.; Amante, E.; Di Corcia, D.; Vincenti, M. Toward the interpretation of positive testing for fentanyl and its analogs in real hair samples: Preliminary considerations. J. Anal. Toxicol. 2020, 44, 362–369. [Google Scholar] [CrossRef] [PubMed]
- Palamar, J.J.; Salomone, A.; Bigiarini, R.; Vincenti, M.; Acosta, P.; Tofighi, B.; Palamar, J.J.; Salomone, A.; Bigiarini, R.; Vincenti, M. Testing hair for fentanyl exposure: A method to inform harm reduction behavior among individuals who use heroin. Am. J. Drug Alcohol Abuse 2019, 45, 90–96. [Google Scholar] [CrossRef] [PubMed]
- Min, S.; Ping, X.; Baohua, S. Detection of meperidine and its metabolites in the hair of meperidine addicts. Forensic Sci. Int. 1999, 103, 159–171. [Google Scholar] [CrossRef]
- Rosado, T.; Gallardo, E. Microextraction by packed sorbent as a novel strategy for sample clean-up in the determination of methadone and EDDP in hair. J. Anal. Toxicol. 2020, 44, 840–850. [Google Scholar] [CrossRef]
- Cappelle, D.; De Keukeleire, S.; Neels, H.; Been, F.; De Doncker, M.; Dom, G.; Crunelle, C.L.; Covaci, A.; van Nuijs, A.L.N. Keratinous matrices for the assessment of drugs of abuse consumption: A correlation study between hair and nails. Drug Test. Anal. 2018, 10, 1110–1118. [Google Scholar] [CrossRef]
- Cappelle, D.; De Doncker, M.; Gys, C.; Krysiak, K.; De Keukeleire, S.; Maho, W.; Crunelle, C.L.; Dom, G.; Covaci, A.; van Nuijs, A.L.N.; et al. A straightforward, validated liquid chromatography coupled to tandem mass spectrometry method for the simultaneous detection of nine drugs of abuse and their metabolites in hair and nails. Anal. Chim. Acta 2017, 960, 101–109. [Google Scholar] [CrossRef] [PubMed]
- Kintz, P.; Farrugia, A.; Ameline, A.; Eibel, A.; Raul, J. High risk of misinterpreting hair analysis results for children tested for methadone. Forensic Sci. Int. 2017, 280, 176–180. [Google Scholar] [CrossRef]
- Joya, X.; Marchei, E.; Salat-batlle, J.; García-Algar, O. Drugs of abuse in maternal hair and paired neonatal meconium: An objective assessment of foetal exposure to gestational consumption. Drug Test. Anal. 2016, 8, 864–868. [Google Scholar] [CrossRef] [PubMed]
- Koster, R.A.; Alffenaar, J.-W.C.; Greijdanus, B.; VanDernagel, J.E.L.; Uges, D.R.A. Fast and highly selective LC-MS/MS screening for THC and 16 other abused drugs and metabolites in human hair to monitor patients for drug abuse. Ther. Drug Monit. 2014, 36, 234–243. [Google Scholar] [CrossRef] [PubMed]
- Tournel, G.; Pollard, J.; Humbert, L.; Wiart, J.-F.; Héouin, V.; Allorge, D. Use of hair testing to determine methadone exposure in pediatric deaths. J. Forensic Sci. 2014, 59, 1436–1440. [Google Scholar] [CrossRef] [PubMed]
- Favretto, D.; Tucci, M.; Monaldi, A.; Ferrara, S.D.; Miolo, G.; Davide, S.; Miolo, G. A study on photodegradation of methadone, EDDP, and other drugs of abuse in hair exposed to controlled UVB radiation. Drug Test. Anal. 2014, 6, 78–84. [Google Scholar] [CrossRef]
- Fucci, N.; De Giovanni, N. Methadone in hair and sweat from patients in long-term maintenance therapy. Ther. Drug Monit. 2007, 29, 452–454. [Google Scholar] [CrossRef]
- Lucas, A.C.S.; Bermejo, A.M.; Tabernero, M.J.; Fernandez, P. Use of solid-phase microextraction (SPME) for the determination of methadone and EDDP in human hair by GC–MS. Forensic Sci. Int. 2000, 107, 225–232. [Google Scholar] [CrossRef]
- Sporkert, F.; Pragst, F. Determination of methadone and its metabolites EDDP and EMDP in human hair by headspace solid-phase microextraction and gas chromatography–mass spectrometry. J. Chromatogr. B Biomed. Sci. Appl. 2000, 746, 255–264. [Google Scholar] [CrossRef]
- Moeller, M.; Fey, P.; Wennig, R. Simultaneous determination of drugs of abuse in human hair by GC/MS and its application to a methadone treatment program. Forensic Sci. Int. 1993, 63, 185–206. [Google Scholar] [CrossRef]
- Reisfield, G.M.; Jones, J.T. the disposition of oxycodone and metabolite in human hair. J. Anal. Toxicol. 2015, 39, 746–750. [Google Scholar] [CrossRef]
- Johansen, S.S.; Le Dang, L.T.V.; Nielsen, M.K.K.; Haage, P.; Kugelberg, F.C.; Kronstrand, R. Temporal patterns of tramadol in hair after a single dose. Forensic Sci. Int. 2020, 316, 110546. [Google Scholar] [CrossRef]
- Yu, H.; Choi, M.; Jang, J.-H.; Park, B.; Seo, Y.H.; Jeong, C.-H.; Bae, J.-W.; Lee, S. Development of a column-switching LC-MS/MS method of tramadol and its metabolites in hair and application to a pharmacogenetic study. Arch. Pharm. Res. 2018, 41, 554–563. [Google Scholar] [CrossRef] [PubMed]
- Verri, P.; Rustichelli, C.; Palazzoli, F.; Vandelli, D.; Marchesi, F.; Ferrari, A.; Licata, M. Tramadol chronic abuse: An evidence from hair analysis by LC tandem MS. J. Pharm. Biomed. Anal. 2015, 102, 450–458. [Google Scholar] [CrossRef] [PubMed]
- Madry, M.M.; Rust, K.Y.; Guglielmello, R.; Baumgartner, M.R.; Kraemer, T. Metabolite to parent drug concentration ratios in hair for the differentiation of tramadol intake from external contamination and passive exposure. Forensic Sci. Int. 2012, 223, 330–334. [Google Scholar] [CrossRef] [PubMed]
- Musshoff, F.; Schwarz, G.; Sachs, H.; Skopp, G.; Franz, T. Concentration distribution of more than 100 drugs and metabolites in forensic hair samples. Int. J. Legal Med. 2020, 134, 989–995. [Google Scholar] [CrossRef]
- Wang, X.; Johansen, S.S.; Nielsen, M.K.K.; Linnet, K. Segmental hair analysis—Interpretation of the time of drug intake in two patients undergoing drug treatment. J. Forensic Sci. 2019, 64, 950–955. [Google Scholar] [CrossRef] [PubMed]
- Pinho, S.; Oliveira, A.; Costa, I.; Alexandra, C.; Carvalho, F.; Falcão, R. Simultaneous quantification of tramadol and O-desmethyltramadol in hair samples by gas chromatography—Electron impact/mass spectrometry. Biomed. Chromatogr. 2013, 23, 1003–1011. [Google Scholar] [CrossRef] [PubMed]
- Papaseit, E.; Marchei, E.; Mortali, C.; Aznar, G.; Garcia-Algar, O.; Farrè, M.; Pacifici, R.; Pichini, S. Development and validation of a liquid chromatography-tandem mass spectrometry assay for hair analysis of atomoxetine and its metabolites: Application in clinical practice. Forensic Sci. Int. 2012, 218, 62–67. [Google Scholar] [CrossRef]
- Kim, J.Y.; In, M.K.; Paeng, K.; Chung, B.C. Simultaneous determination of carisoprodol and meprobamate in human hair using solid-phase extraction and gas chromatography / mass spectrometry of the trimethylsilyl derivatives. Rapid Commun. Mass Spectrom. 2005, 19, 3056–3062. [Google Scholar] [CrossRef]
- Zhuo, Y.; Wang, X.; Wu, J.; Zhang, S.; Deng, H.; Liu, W.; Wu, H.; Xiang, P.; Shen, M. Simultaneous quantitative determination of amphetamines, opiates, ketamine, cocaine and metabolites in human hair: Application to forensic cases of drug abuse. J. Forensic Sci. 2020, 65, 563–569. [Google Scholar] [CrossRef]
- Miolo, G.; Tucci, M.; Id, L.M.; Stocchero, G.; Vogliardi, S.; Scrivano, S.; Montisci, M.; Favretto, D. A Study on photostability of amphetamines and ketamine in hair irradiated under artificial sunlight. Brain Sci. 2018, 8, 96. [Google Scholar] [CrossRef]
- Leung, K.W.; Wong, Z.C.F.; Ho, J.Y.M.; Yip, A.W.S.; Ng, J.S.C.; Ip, S.P.H.; Ng, W.Y.Y.; Ho, K.K.L.; Duan, R.; Zhu, K.Y.; et al. Determination of hair ketamine cut-off value from Hong Kong ketamine users by LC–MS/MS analysis. Forensic Sci. Int. 2016, 259, 53–58. [Google Scholar] [CrossRef]
- Salomone, A.; Gerace, E.; Diana, P.; Romeo, M.; Malvaso, V.; Di Corcia, D.; Vincenti, M. Cut-off proposal for the detection of ketamine in hair. Forensic Sci. Int. 2015, 248, 119–123. [Google Scholar] [CrossRef] [PubMed]
- Chang, Y.; Chao, M.; Chen, S.; Chen, C. A high-throughput method based on microwave-assisted extraction and liquid chromatography–tandem mass spectrometry for simultaneous analysis of amphetamines, ketamine, opiates, and their metabolites in hair. Anal. Bioanal. Chem. 2014, 406, 2445–2455. [Google Scholar] [CrossRef] [PubMed]
- Favretto, D.; Vogliardi, S.; Stocchero, G.; Nalesso, A.; Tucci, M.; Terranova, C.; Davide, S. Determination of ketamine and norketamine in hair by micropulverized extraction and liquid chromatography–high resolution mass spectrometry. Forensic Sci. Int. 2013, 226, 88–93. [Google Scholar] [CrossRef] [PubMed]
- Zhu, K.Y.; Leung, K.W.; Ting, A.K.L.; Wong, Z.C.F.; Fu, Q.; Ng, W.Y.Y.; Choi, R.C.Y.; Dong, T.T.X.; Wang, T.; Lau, D.T.W.; et al. The establishment of a highly sensitive method in detecting ketamine and norketamine simultaneously in human hairs by HPLC-Chip–MS/MS. Forensic Sci. Int. 2011, 208, 53–58. [Google Scholar] [CrossRef] [PubMed]
- Harun, N.; Anderson, R.A.; Cormack, P.A.G. Analysis of ketamine and norketamine in hair samples using molecularly imprinted solid-phase extraction (MISPE) and liquid chromatography–tandem mass spectrometry (LC-MS/MS). Anal. Bioanal. Chem. 2010, 396, 2449–2459. [Google Scholar] [CrossRef] [PubMed]
- Leong, H.S.; Tan, N.L.; Lui, C.P.; Lee, T.K. Evaluation of ketamine abuse using hair analysis: Concentration trends in a Singapore population. J. Anal. Toxicol. 2010, 29, 314–318. [Google Scholar] [CrossRef] [PubMed]
- Tabernero, M.J.; Felli, M.L.; Bermejo, A.M.; Chiarotti, M. Determination of ketamine and amphetamines in hair by LC/MS/MS. Anal. Bioanal. Chem. 2009, 395, 2547–2557. [Google Scholar] [CrossRef]
- Wu, Y.-H.; Lin, K.; Chen, S.-C.; Chang, Y.-Z. Integration of GC/EI-MS and GC/NCI-MS for simultaneous quantitative determination of opiates, amphetamines, MDMA, ketamine, and metabolites in human hair. J. Chromatogr. B 2008, 870, 192–202. [Google Scholar] [CrossRef] [PubMed]
- Wu, Y.; Lin, K.; Chen, S.; Chang, Y. Simultaneous quantitative determination of amphetamines, ketamine, opiates and metabolites in human hair by gas chromatography/mass spectrometry. Rapid Commun. Mass Spectrom. 2008, 22, 887–897. [Google Scholar] [CrossRef]
- Xiang, P.; Shen, M.; Zhuo, X. Hair analysis for ketamine and its metabolites. Forensic Sci. Int. 2006, 162, 131–134. [Google Scholar] [CrossRef] [PubMed]
- Jang, M.; Kim, J.; Shin, I.; Kang, S.; Choi, H.; Yang, W. Simultaneous determination of methylphenidate and ritalinic acid in hair using LC–MS/MS. Forensic Sci. Int. 2019, 294, 183–188. [Google Scholar] [CrossRef] [PubMed]
- Kłys, M.; Rojek, S.; Bolechała, F. Determination of oxcarbazepine and its metabolites in postmortem blood and hair by means of liquid chromatography with mass detection (HPLC/APCI/MS). J. Chromatogr. B 2005, 825, 38–46. [Google Scholar] [CrossRef]
- Frison, G.; Favretto, D.; Tedeschi, L.; Ferrara, S.D. Detection of thiopental and pentobarbital in head and pubic hair in a case of drug-facilitated sexual assault. Forensic Sci. Int. 2003, 133, 171–174. [Google Scholar] [CrossRef]
- Wishart, D.S.; Feunang, Y.D.; Guo, A.C.; Lo, E.J.; Marcu, A.; Grant, J.R.; Sajed, T.; Johnson, D.; Li, C.; Sayeeda, Z.; et al. DrugBank 5.0: A major update to the DrugBank database for 2018. Nucleic Acids Res. 2018, 46, D1074–D1082. [Google Scholar] [CrossRef]
- Stowe, G.N.; Paulsen, R.B.; Hill, V.A.; Schaffer, M.I.; Corporation, P.; Way, U.; City, C. A retrospective analysis of selected opioids in hair of workplace drug testing subjects. J. Anal. Toxicol. 2019, 43, 553–563. [Google Scholar] [CrossRef]
- Baselt, R.C. Disposition of Toxic Drugs and Chemicals in Man, 10th ed.; Biomedical Publications: Seal Beach, CA, USA, 2014; pp. 1846–1848. [Google Scholar]
- Schulz, M.; Iwersen-Bergmann, S.; Andresen, H.; Schmoldt, A. Therapeutic and toxic blood concentrations of nearly 1000 drugs and other xenobiotics. Crit. Care 2012, 16, 2–5. [Google Scholar] [CrossRef] [PubMed]
- Ramírez, M.; Baumgartner, W.A.; Wille, S.M.R.; Farabee, D.; Samyn, N.; Baumgartner, A.M.; Fernández, M.d.M.R.; Baumgartner, W.A.; Wille, S.M.R.; Farabee, D.; et al. A different insight in hair analysis: Simultaneous measurement of antipsychotic drugs and metabolites in the protein and melanin fraction of hair from criminal justice patients. Forensic Sci. Int. 2020, 312, 110337. [Google Scholar] [CrossRef]
- Skopp, G.; Kniest, A.; Haisser, J.; Mann, K.; Hermann, D. Buprenorphine and norbuprenorphine findings in hair during constant maintenance dosage. Int. J. Legal Med. 2011, 125, 277–281. [Google Scholar] [CrossRef] [PubMed]
- Baumgartner, W.A.; Hill, V.A.; Hillb, V.A. Sample preparation techniques. Forensic Sci. Int. 1993, 63, 121–135. [Google Scholar] [CrossRef]
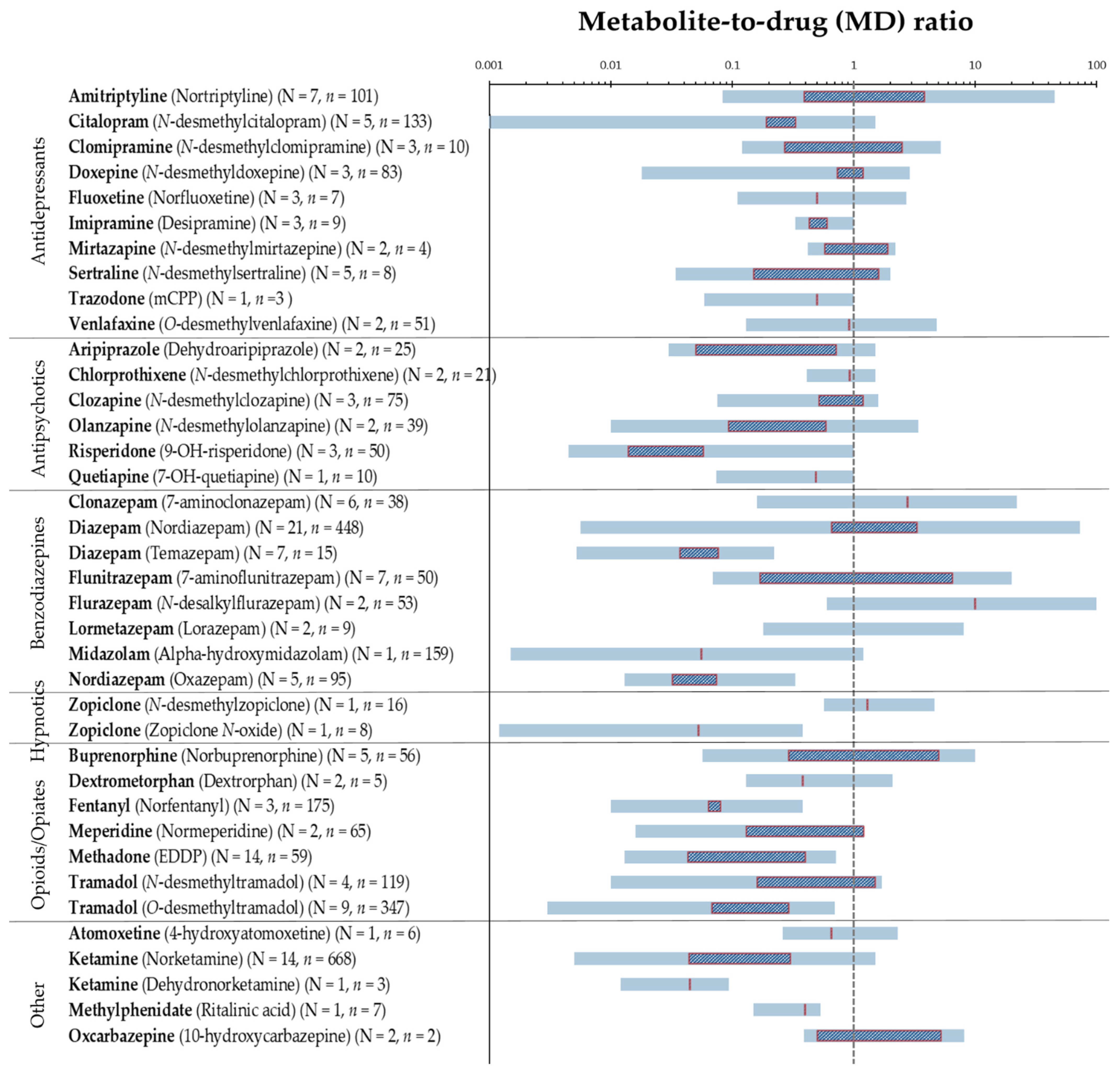
| Pharmaceutical (Metabolite) | MD Ratio | No. of Subjects | No. of Seg. Per Subject (Seg. Length) | Total No. of Seg. (Whole Strand) | Case Description | Analytical Method | Reference | |||
|---|---|---|---|---|---|---|---|---|---|---|
| Range | 10–90 PCTL (A) | Mean | Median | |||||||
| ANTIDEPRESSANTS | ||||||||||
| Amitriptyline (Nortriptyline) | 0.083–5.5 | 0.15–5.0 (B) | 1.4 | 1.1 | 45 | 2 (2 cm) or full length | 42 (24) | PM cases, presumed intake | LC–MS/MS | Methling (2020) [18] |
| 0.63–45 | - | 14 | 3.8 | 2 | 2–5 (1 cm) | 7 | AD users, presumed intake | LC–MS/MS | Fernández (2016) [19] | |
| 0.24–3.2 | - | 24 | 1 (3 cm) | 24 | Headache patients | LC–MS/MS | Licata (2016) [20] | |||
| 2.4 | - | 2.4 | - | 1 | 1 (9 cm) | 1 | Patient under treatment with psychoactive drugs | LC–MS/MS | Fisichella (2014) [21] | |
| 0.97–3.3 | - | 2.2 | 2.2 | 2 | 1 (1–3) | 2 | PM cases | LC–HRMS | Nielsen (2010) [22] | |
| 0.1–2.6 | 0.17–0.80 | 0.56 | 0.39 | 25 | 1 (3 cm) | 25 | Psychiatric patients, presumed intake | GC–MS | Pragst (1997) [23] | |
| 0.88–2.9 | - | 1.9 | 1.9 | 2 | Unspecified | ≥2 | Patients under long-term treatment | GC–MS | Ishiyama (1983) [24] | |
| 0.083–45 | 0.39–3.8 | 101 | 103 (24) | Combined | ||||||
| Citalopram (N-desmethyl-citalopram) | 0.066–0.87 (B) | 0.39 | 0.33 | 108 | 2 (2 cm) or full length | 136 (40) | PM cases, presumed intake | LC–MS/MS | Methling (2020) [18] | |
| 0.13–1.5 | 0.14–0.62 | 0.33 | 0.23 | 3 | 4–10 (1 cm) | 20 | AD users, presumed intake | LC–MS/MS | Fernández (2016) [19] | |
| 0.35–0.88 | - | - | - | 16 | 1 (3 cm) | 16 | Headache patients | LC–MS/MS | Licata (2016) [20] | |
| 0.00069–0.67 | - | 0.25 | 0.19 | 5 | 1 (9 cm) | 5 | PM cases and patients under treatment with psychoactive drugs | LC–MS/MS | Fisichella (2014) [21] | |
| 0.76 | - | 0.76 | - | 1 | 1 (2 cm) | 1 | PM case, polydrug intoxication | GC–MS | Wille (2009) [25] | |
| 0.00069–1.5 | 0.19–0.33 | 133 | 178 (40) | Combined | ||||||
| Clomipramine (N-desmethyl-clomipramine) | 1.8–5.2 | 1.8–5.2 (B) | 3 | 2.5 | 4 | 2 (2 cm) or full length | 4 (2) | PM cases, presumed intake | LC–MS/MS | Methling (2020) [18] |
| 0.75–2.2 | - | 1.7 | 2.1 | 1 | 3 (4 cm) | 3 | PM case, under treatment, alcohol abuse | LC–MS | Klys (2005) [26] | |
| 0.12–0.86 | - | 0.37 | 0.27 | 5 | 1 (3 cm) | 5 | Psychiatric patients, presumed intake | GC–MS | Pragst (1997) [23] | |
| 0.12–5.2 | 0.27–2.5 | 10 | 12 (2) | Combined | ||||||
| Doxepine (N-desmethyldoxepine) | 0.018–2.9 | 0.14–2.3 (B) | 1.2 | 1.2 | 76 | 2 (2 cm) or full length | 98 (27) | PM cases, presumed intake | LC–MS/MS | Methling (2020) [18] |
| 0.85 | - | 0.85 | - | 1 | 1 (6 cm) | 1 | PM cases, drug abuse history | LC–MS/MS | Krumbiegel (2016) [27] | |
| 0.33–1.4 | - | 0.81 | 0.73 | 6 | 1 (3 cm) | 6 | Psychiatric patients, presumed intake | GC–MS | Pragst (1997) [23] | |
| 0.018–2.9 | - | 0.73–1.2 | 83 | 105 (27) | Combined | |||||
| Fluoxetine (Norfluoxetine) | 0.15 | - | 0.15 | - | 1 | 1 (2 cm) | 1 | Suspected DFC case | LC–MS/MS | Wang (2018) [28] |
| 0.11–0.67 | 0.12–0.61 | 0.38 | 0.50 | 2 | 4–6 (1 cm) | 10 | AD users, presumed intake | LC–MS/MS | Fernández (2016) [19] | |
| 0.93–2.7 | - | - | - | 4 | 1 (3 cm) | 4 | Headache patients | LC–MS/MS | Licata 2016 [20] | |
| 0.11–2.7 | 0.50 | 7 | 15 | Combined | ||||||
| Imipramine (Desipramine) | 0.36–0.8 | - | 0.62 | 0.6 | 5 | 1 (3 cm) | 5 | Psychiatric patients, presumed intake | GC–MS | Pragst (1997) [23] |
| 0.85 | - | 0.85 | - | 1 | Unspecified | ≥1 | PM case | GC–MS + LC | Couper (1995) [29] | |
| 0.33–1.0 | - | 0.59 | 0.43 | 3 | Unspecified | ≥3 | Patients under long-term treatment | GC–MS | Ishiyama (1983) [24] | |
| 0.33–1.0 | 0.43–0.6 | 9 | ≥9 | Combined | ||||||
| Mirtazapine (N-desmethyl-mirtazapine) | 0.42–0.90 | - | 0.60 | 0.58 | 2 | 3 (1 cm) | 6 | Children administered mirtazapine without consent | LC–MS/MS | Kintz (2021) [30] |
| 1.6–2.2 | - | 1.9 | 1.9 | 2 | 1 (3 cm) | 2 | Headache patients | LC–MS/MS | Licata (2016) [20] | |
| 0.42–2.2 | 0.58–1.9 | 4 | 8 | Combined | ||||||
| Sertraline (N-desmethylsertraline) | 1.7 | - | 1.7 | - | 1 | 1 (2 cm) | 1 | Schizophrenic patients under treatment, in compliance | LC–MS/MS | Wang (2019) [31] |
| 0.18–0.89 | - | 0.50 | 0.50 | 1 | 6 (2 cm) | 6 | Child suspected accidental intoxication | LC–MS/MS | Marchei (2016) [32] | |
| 0.79–2.0 | - | - | - | 4 | 1 (3 cm) | 4 | Headache patients | LC–MS/MS | Licata (2016) [20] | |
| 0.034–0.16 | - | 0.12 | 0.15 | 1 | 3 (3 cm) | 3 | AD and anxiolytic drug consumption during pregnancy | LC–MS/MS | Pichini (2016) [33] | |
| 0.83–1.8 | 1.4 | 1.6 | 1 | 3 (1.5–2 cm) | 3 | PM case, polydrug intoxication | GC–MS | Wille (2009) [25] | ||
| 0.034–2.0 | 0.15–1.6 | 8 | 17 | Combined | ||||||
| Trazodone (mCPP (M-chloro-phenylpiperazine)) | 0.059–1.0 | 0.088–0.50 | 0.37 | 0.50 | 3 | 1–10 (1 cm) | 13 | AD users, presumed intake | LC–MS/MS | Fernández (2016) [19] |
| Venlafaxine (O-desmethyl-venlafaxine) | - | 0.13–3.5 | 1.5 | 0.92 | 45 | 2 (2 cm) or full length | 50 (20) | PM case, presumed intake | LC–MS/MS | Methling (2020) [18] |
| 1.2–4.8 | - | - | - | 6 | 1 (3 cm) | 6 | Headache patients | LC–MS/MS | Licata (2016) [20] | |
| 0.13–4.8 | 0.92 | 51 | 56 (20) | Combined | ||||||
| ANTIPSYCHOTICS | ||||||||||
| Aripiprazole (Dehydroaripiprazole) | 0.21–1.5 | 0.3–1.3 | 0.76 | 0.72 | 16 | 1–6 (1 cm) | 71 | PM cases of psychiatric patients with presumed intake | LC–MS/MS | Rygaard (2020) [34] |
| 0.030–0.093 | 0.034–0.083 | - | 0.050 | 9 | 1–2 (2 cm) | 11 | Schizophrenic patients under drug treatment, in compliance | LC–MS/MS | Wang (2019) [31] | |
| 0.030–1.5 | 0.050–0.72 | 25 | 82 | Combined | ||||||
| Chlorprothixene (N-desmethyl-chlorprothixene) | - | 0.41–1.5 | - | 0.93 | 20 | 3–6 (1 cm) | ≥60 | PM cases of psychiatric patients with presumed intake | LC–MS/MS | Günther (2018) [35] |
| 1.5 | - | 1.5 | - | 1 | 1 (2 cm) | 1 | Suspected DFC case | LC–MS/MS | Wang (2018) [28] | |
| 0.41–1.5 | - | 0.93 | 21 | ≥61 | Combined | |||||
| Clozapine (N-desmethylclozapine) | - | 0.075–1.3 (B) | 0.57 | 0.52 | 25 | 2 (2 cm) or full length | 28 (11) | PM cases presumed intake | LC–MS/MS | Methling (2020) [18] |
| 0.52–1.5 | 0.77–1.4 | - | 1.1 | 27 | 1–3 (2 cm) | 54 | Schizophrenic patients under drug treatment, in compliance | LC–MS/MS | Wang (2019) [31] | |
| 0.97–1.6 0.70–1.3 | 1.0–1.6 0.75–1.1 | 1.2 0.96 | 1.2 0.97 | 12 11 | 1 (2–7 cm) 1 (2–7 cm) | 12 11 | Patients in low-dose treatment, pigmented hair and non-pigmented hair | LC–MS/MS | Kronstrand (2007) [36] | |
| 0.075–1.6 | 0.52–1.2 | 75 | 105 (11) | Combined | ||||||
| Olanzapine (N-desmethylolanzapine) | 0.010–3.4 | - | - | 0.59 | 34 | 1–6 (1 cm) or full length (if <2 cm) | 105 | PM cases of psychiatric patients, presumed intake | LC–MS/MS | Günter (2020) [37] |
| 0.063–0.23 | 0.063–0.23 | - | 0.093 | 5 | 1–2 (2 cm) | 7 | Schizophrenic patients under drug treatment, in compliance | LC–MS/MS | Wang (2019) [31] | |
| 0.010–3.4 | 0.093–0.59 | 39 | 112 | Combined | ||||||
| Risperidone (9-OH-risperidone) | 0.0045–0.093 | 0.0055–0.031 | - | 0.014 | 12 | 1–3 (2 cm) | 27 | Schizophrenic patients under drug treatment, in compliance | LC–MS/MS | Wang (2019) [31] |
| - | 0.0047–1 (B) | 0.17 | 0.058 | 35 | 2 (2 cm) or full length | 30 (20) | PM cases, presumed intake | LC–MS/MS | Methling (2020) [18] | |
| 0.012–1.0 | - | 0.31 | 0.023 | 3 | 1–4 (2–10 cm) | 8 | Psychiatric patients | LC–MS/MS | Schneider (2009) [38] | |
| 0.0045–1.0 | 0.014–0.058 | 50 | 65 (20) | Combined | ||||||
| Quetiapine (7-OH-quetiapine) | 0.074–1 | 0.12–0.91 | 0.50 | 0.49 | 10 | 1–6 (2 cm) | 26 | Patients under drug treatment | LC–MS/MS | Binz (2014) [39] |
| BENZODIAZEPINES | ||||||||||
| Alprazolam (Alpha-hydroxyalprazolam) | 0.056–0.062 | - | 0.059 | 0.059 | 1 | 3 (2 cm) | 3 | PM case, presumed intake, drug addiction | LC–MS/MS | Wang (2017) [40] |
| Clonazepam (7-aminoclonazepam) | 0.16–17 | - | - | 2.8 | 33 | 1 (≤5 cm) | 33 | Individuals undergoing toxicological investigation | LC–MS/MS | Madry (2020) [41] |
| 82 | - | 82 | - | 1 | Unspecified | ≥1 | Suspected drug abuser | LC–MS/MS | Shin (2019) [42] | |
| 1.6 | - | 1.6 | - | 1 | 1 (2 cm) | 1 | Suspected DFC case | LC–MS/MS | Wang (2018) [28] | |
| 3.7 | - | 3.7 | - | 1 | 1 (3 cm) | 1 | Headache patient | LC–MS/MS | Licata (2016) [20] | |
| 22 | - | 22 | - | 1 | Unspecified | ≥1 | DFC case | LC–MS/MS | Chéze 2005 [43] | |
| 7.0 | - | 7.0 | - | 1 | 1 (2 cm) | 1 | Individuals under treatment | GC–MS | Negruz (2000) [44] | |
| 0.16–22 | 2.8 | 38 | ≥38 | Combined | ||||||
| Delorazepam (Lorezepam) | ≤0.090 | - | - | - | 23 | 1 (3 cm) | 23 | Headache patients | LC–MS/MS | Licata (2016) [20] |
| Diazepam (Nordiazepam) | 0.0056–26 | - | - | 1.6 | 293 | 1 (≤5 cm) | 293 | Individuals undergoing toxicological investigation | LC–MS/MS | Madry (2020) [41] |
| - | 0.26–2.9 (B) | 1.3 | 1.1 | 71 | 2 (2 cm) or full length | 70 (36) | PM cases, presumed intake | LC–MS/MS | Methling (2020) [18] | |
| 0.40 | - | 0.40 | - | 1 | Unspecified | ≥1 | Individuals with suspected drug abuse | LC–MS/MS | Shin (2019) [42] | |
| 0.96 | - | 0.96 | - | 1 | 1 (3–6 cm) | 1 | Driving licence regranting | LC–MS/MS | Lendoiro (2018) [45] | |
| 0.58–1.6 | - | 1.0 | 0.98 | 2 | 1–3 (0.8–1.5 cm) | 4 | Suspected DFC case | LC–MS/MS | Wang (2018) [28] | |
| 0.98 | - | 0.98 | - | 1 | 1 (3–6 cm) | 1 | PM case | LC–MS/MS | Morini (2017) [46] | |
| 1.3–4.1 | - | 2.3 | 2.1 | 8 | 1 (2 cm) | 8 | Controlled single dose study, proximal seg. (1 month after intake) and | LC–MS/MS | Wang (2017) [47] | |
| 0.77–1.1 | - | 0.91 | 0.88 | 6 | 1 (2 cm) | 6 | second seg. | |||
| 6 | - | 6 | - | 1 | 1 (2 cm) | 1 | DFC case | LC–MS/MS | Kim (2016) [48] | |
| 0.23–1.6 | 0.39–1.3 | 0.89 | 0.94 | 5 | 1–4 (1.5–8 cm) | 10 | PM cases | LC–MS/MS | Krumbiegel (2016) [27] | |
| 0.30–2.2 | - | - | - | 3 | 1 (3 cm) | 3 | Headache patients | LC–MS/MS | Licata (2016) [20] | |
| 2.0–2.8 | - | 2.4 | 2.5 | 1 | 3 (3 cm) | 3 | AD and anxiolytic drug consumption during pregnancy | LC–MS/MS | Pichini (2016) [33] | |
| 1.1–1.1 | - | 1.1 | 1.1 | 1 | 2 (4 cm) | 2 | PM case | LC–MS/MS | Maublanc 2015 [49] | |
| 0.31–5.5 | - | 2.1 | 0.66 | 5 | 1 (3 or 9 cm) | 5 | PM cases and patients under treatment with psychoactive drugs | LC–MS/MS | Fisichella (2014) [21] | |
| 1.3–1.7 | - | 1.5 | 1.5 | 2 | Unspecified | ≥2 | Patient following withdrawal treatment and individual with suspected drug abuse | LC–MS/MS | Lendoiro (2012) [50] | |
| 0.67–5 | - | 2.8 | 3.3 | 5 | Unspecified | ≥5 | Presumed drug use | LC–HRMS | Favretto (2011) [51] | |
| 0.35–1.6 | - | 0.96 | 0.96 | 2 | Unspecified | ≥2 | Individuals with suspected drug abuse | LC–MS/MS | Kim (2011) [52] | |
| 0.35 0.27 | - - | 0.35 0.27 | - - | 1 | Unspecified (10–35 cm) | ≥1 | Individuals with suspected drug abuse non-pigmented hair and pigmented hair | GC–MS | Lee (2011) [53] | |
| 0.13–6.7 | 0.36–5.0 | 2.2 | 1.6 | 14 | 1–3 (1–3 cm) | 16 | PM cases and individuals undergoing toxicological investigation | LC–HRMS | Vogliardi (2011) [15] | |
| 0.94–7.3 | 1.1–6.7 | 3.1 | 2.8 | 8 | 1–2 (0.5–3 cm) | 10 | PM cases undergoing toxicological investigation | LC–MS/MS | Miller (2008) [54] | |
| 0.46–8 | - | 3.3 | 1.5 | 3 | Unspecified | ≥3 | PM cases, presumed intake | LC–MS/MS | Ariffin (2007) [55] | |
| 0.21–73 | 0.53–40 | 15 | 2.4 | 14 | 1 (3 cm) | 14 | PM cases, dead from a drug overdose | GC–MS | Yegles (1997) [56] | |
| 0.0056–73 | 0.66–3.3 | 448 | ≥461 (36) | Combined | ||||||
| Diazepam (Temazepam) | 0.023 | - | 0.023 | - | 1 | 1 (0.8 cm) | 1 | Suspected DFC case | LC–MS/MS | Wang (2018) [28] |
| 0.048–0.090 | - | 0.066 | 0.064 | 4 | 1–4 (1.5–6 cm) | 9 | PM cases | LC–MS/MS | Krumbiegel (2016) [27] | |
| 0.076–0.078 | - | 0.077 | 0.077 | 1 | 2 (4 cm) | 2 | PM case | LC–MS/MS | Maublanc 2015 [49] | |
| 0.0052– 0.068 | - | 0.037 | 0.037 | 2 | unspecified | ≥2 | Individuals with suspected drug abuse | LC–MS/MS | Kim (2011) [52] | |
| 0.050–0.11 | - | 0.069 | 0.063 | 5 | 1 (1–3 cm) | 5 | PM cases and individuals undergoing toxicological investigation | LC–HRMS | Vogliardi (2011) [15] | |
| 0.19 | - | 0.19 | - | 1 | 1 (3 cm) | 1 | PM cases undergoing toxicological investigation | LC–MS/MS | Miller (2008) [54] | |
| 0.22 | - | 0.22 | - | 1 | unspecified | ≥1 | PM cases, presumed intake | LC–MS/MS | Ariffin (2007) [55] | |
| 0.0052–0.22 | 0.037–0.077 | 15 | ≥21 | Combined | ||||||
| Flunitrazepam (7-amino-flunitrazepam) | 2.1–20 | - | - | 6.5 | 8 | 1 (≤5 cm) | 8 | Individuals undergoing toxicological investigation | LC–MS/MS | Madry (2020) [41] |
| - | 1.3–5.0 (B) | 3.2 | 3.1 | 3 | 2 (2 cm) or full length | 4 (1) | PM cases, presumed intake | LC–MS/MS | Methling (2020) [18] | |
| - | 0.51–8.3 | - | 3.07 | 22 | 3–6 (1 cm) | 76 | Individuals undergoing toxicological investigation, repeated use | LC–MS/MS | Zhuo (2020) [9] | |
| 1.1 | - | 1.1 | - | 1 | 1 (3 cm) | 1 | DFC case | LC–MS/MS | Kim (2016) [48] | |
| 1.1 | - | 1.1 | - | 1 | unspecified | ≥1 | PM case | GC–MS | Negrusz (1999) [57] | |
| 0.069–3.5 | 0.27–2.5 | 1.2 | 0.90 | 14 | Whole strand | (14) | PM cases, polydrug abusers | GC–MS | Cirimele (1997) [58] | |
| 0.11–0.27 | - | 0.18 | 0.17 | 1 | 3 (3–4 cm) | 3 | Individual with a suspected drug abuse | GC–MS | Cirimele (1996) [59] | |
| 0.069–20 | 0.17–6.5 | 50 | 93 (15) | Combined | ||||||
| Flurazepam (2-hydroxyethyl-flurazepam) | 0.033 | - | 0.033 | - | 1 | 1 (3 cm) | 1 | Headache patients | LC–MS/MS | Licata (2016) [20] |
| Flurazepam (N-desalkylflurazepam) | 0.60–110 | - | - | 10 | 52 | 1 (≤5 cm) | 52 | Individuals undergoing toxicological investigation | LC–MS/MS | Madry (2020) [41] |
| 2.4 | - | 2.4 | - | 1 | 1 (3 cm) | 1 | Headache patients | LC–MS/MS | Licata (2016) [20] | |
| 0.60–110 | 10 | 53 | 53 | Combined | ||||||
| Lormetazepam (Lorazepam) | 0.18–8 | - | - | - | 8 | 1 (≤5 cm) | 8 | Individuals undergoing toxicological investigation | LC–MS/MS | Madry (2020) [41] |
| 0.42 | - | 0.42 | - | 1 | 1 (3 cm) | 1 | Headache patients | LC–MS/MS | Licata (2016) [20] | |
| 0.18–8 | - | 9 | 9 | Combined | ||||||
| Midazolam (alpha-hydroxymidazolam) | 0.0015–1.2 | - | - | 0.056 | 159 | 1 (≤5 cm) | 159 | Cases of interest in forensic toxicolgy | LC–MS/MS | Madry (2020) [41] |
| Nordiazepam (Oxazepam) | 0.017–0.28 | - | - | 0.074 | 86 | 1 (≤5 cm) | 86 | Individuals undergoing toxicological investigation | LC–MS/MS | Madry (2020) [41] |
| 0.040–0.10 | - | 0.070 | 0.070 | 2 | Full length (5–7 cm) | (2) | Individuals with past history depressive disorder | LC–MS/MS | Wiart (2020) [60] | |
| 0.030 | - | 0.030 | - | 1 | 1 (0.8 cm) | 1 | Suspected DFC case | LC–MS/MS | Wang (2018) [28] | |
| 0.33 | - | 0.33 | - | 1 | 1 (3 cm) | 1 | PM cases undergoing toxicological investigation | LC–MS/MS | Miller (2008) [54] | |
| 0.013–0.056 | - | 0.034 | 0.032 | 5 | Unspecified | ≥5 | Individuals with polydrug abuse | GC–MS | Kintz (1996) [61] | |
| 0.013–0.33 | 0.032–0.074 | 95 | ≥93 (2) | Combined | ||||||
| Triazolam (1-hydroxymethyl-triazolam) | 2.1 | - | 2.1 | - | 1 | Hair shaved | (1) | PM case, individual with drug addiction | LC–MS | Toyo’oka (2001) [62] |
| Triazolam (4-hydroxy triazolam) | 14 | - | 14 | - | 1 | Hair shaved | (1) | PM case, individual with drug addiction | LC–MS | Toyo’oka (2001) [62] |
| HYPNOTICS | ||||||||||
| Zolpidem (zolpidem phenyl 4carboxylic acid) | 0.0018 | - | 0.0018 | - | 1 | Unspecified | ≥1 | Individuals with suspected drug abuse | LC–MS/MS | Shin (2019) [42] |
| Zopiclone (N-desmethylzopiclone) | 0.57–4.6 | - | - | 1.3 | 16 | 2–12 (0.5 cm) | 56 | Controlled single dose study | LC–MS/MS | Hansen (2020) [63] |
| Zopiclone (zopiclone N-oxide) | 0.0012–0.38 | - | - | 0.053 | 8 | 2–12 (0.5 cm) | 31 | Controlled single dose study | LC–MS/MS | Hansen (2020) [63] |
| OPIOIDS/OPIATES | ||||||||||
| Buprenorphine (Norbuprenorphine) | 1.1 | - | 1.1 | - | 1 | 1 (3.5 cm) | 1 | PM case, drug abuse history | LC–MS/MS | Krumbiegel (2016) [27] |
| - - - | 1.7–5 (C) 3.3–10 (C) 1.4–10 (C) | 1.7 2.5 2.5 | 3.3 5 3.3 | 36 | 1–3 (3.9 ± 1.2 cm) (5.0 ± 2.1 cm) (4.5 ± 2.0 cm) | 19 17 15 | Individuals under treatment, proximal seg. and middle seg. and distal seg. | LC–MS | Belivanis (2013) [64] | |
| 2.0–3.0 | - | 2.7 | 2.7 | 3 | Unspecified | ≥3 | Presumed drug use | LC–HRMS | Favretto (2011) [51] | |
| 0.49–2.9 | - | 1.9 | 1.5 | 5 | Unspecified(1 cm) | ≥5 | Long history of buprenorphine abuse | GC–MS | Vincent (1999) [65] | |
| 0.057–0.64 | 0.11–0.44 | 0.28 | 0.29 | 11 | Unspecified | ≥11 | Drug abuse history (Heroin) | LC–ECD | Kintz (1994) [66] | |
| 0.057–10 | 0.29–5 | 56 | ≥71 | Combined | ||||||
| Dextrometorphan (Dextrorphan) | 0.40 | - | 0.40 | - | 1 | 1 (12 cm) | 1 | Individuals with suspected drug abuse | LC–MS/MS | Kim (2014) [67] |
| 0.13–2.1 | - | 0.76 | 0.38 | 4 | Whole strand | (4) | Individuals with suspected abuse | GC–MS | Kim (2004) [68] | |
| 0.13–2.1 | 0.38 | 5 | 1 (4) | Combined | ||||||
| Fentanyl (Norfentanyl) | 0.01–0.38 | - | 0.10 | 0.08 | 154 | Full length (1–20 cm) | (154) | Suspected intake | LC–MS/MS | Salomone (2020) [69] |
| 0.031–0.15 | 0.034–0.096 | 0.069 | 0.064 | 20 | Full length (1–20 cm) | (20) | Individuals using heroine | LC–MS/MS | Palamar (2019) [70] | |
| 0.03 | - | 0.03 | - | 1 | 1 (3.5 cm) | 1 | PM case, drug abuse history | LC–MS/MS | Krumbiegel (2016) [27] | |
| 0.01–0.38 | 0.08–0.064 | 175 | 1 (174) | Combined | ||||||
| Meperidine (Normeperidine) | 0.016–0.39 | - | 0.17 | 0.13 | 5 | 1 (9–22 cm) | 5 | Individuals with suspected abuse | LC–MS/MS | Kim (2014) [67] |
| - | - | 1.40 | 1.21 | 60 | Unspecified(1–5 cm) | ≥71 | Individuals with meperidine addiction | GC–MS | Min (1999) [71] | |
| 0.016–0.39 | 0.13–1.21 | 65 | ≥76 | Combined | ||||||
| Methadone (EDDP (2-ethyl-1,5-dimethyl- 3,3-diphenylpyrrolidine)) | 0.30 | - | 0.30 | - | 1 | Full length | (1) | Individual undergoing methadone treatment program | GC–MS/MS | Rosado (2020) [72] |
| 0.063 | - | 0.063 | - | 5 | 1 (3 cm) | 5 | Patients in treatment for substance-use disorders | LC–MS/MS | Cappelle (2018) [73] | |
| 0.063 | - | 0.063 | - | 1 | Unspecified | ≥1 | Individuals with self-reported drug use | LC–MS/MS | Cappelle (2017) [74] | |
| 0.11–0.15 | - | 0.13 | 0.12 | 1 | 4 (1 cm) | 4 | Child sedated | LC–MS/MS | Kintz (2017) [75] | |
| 0.060–0.099 | - | 0.075 | 0.074 | 3 | 1–4 (2–6 cm) | 6 | PM cases, known intake | LC–MS/MS | Krumbiegel (2016) [27] | |
| 1 | - | 1 | - | 1 | 1 (3 cm) | 1 | Maternal hair sample | LC–MS/MS | Joya (2015) [76] | |
| 0.035 | - | 0.035 | - | 1 | 1 (10 cm) | 1 | Psychiatric patient | LC–MS/MS | Koster (2014) [77] | |
| 0.013–0.39 | - | 0.15 | 0.043 | 1 | 3 (1.5 cm) | 3 | Maternal hair samples, seg. 2–4 | LC–MS/MS | Tournel (2014) [78] | |
| 0.037–0.48 | - | 0.23 | 0.19 | 7 | 1 (5–7 cm) | 7 | Cases undergoing toxicological investigation, presumed drug use | LC–HRMS | Favretto (2014) [79] | |
| 0.26–0.53 | - | 0.40 | 0.40 | 2 | unspecified | ≥2 | Presumed drug use | LC–HRMS | Favretto (2011) [51] | |
| 0.1–0.3 | - | 0.2 | 0.2 | 9 | 1 (3 cm) | 9 | Methadone maintenance therapy | GC–MS | Fucci (2007) [80] | |
| 0.099–0.23 | - | 0.16 | 0.16 | 7 | Whole strand | (7) | Methadone maintenance therapy | GC–MS | Lucas (2000) [81] | |
| 0.19–0.67 | - | 0.39 | - | 17 | 1 (3 cm) | 17 | PM cases, died from overdose | GC–MS | Sporkert (2000) [82] | |
| 0.14–0.67 0.14–0.19 0.031–0.71 | - - - | 0.18 0.14 0.062 | - - - | 3 | 10–29 (2 cm) | 29 10 23 | Methadone maintenance therapy | GC–MS | Moeller (1993) [83] | |
| 0.013–0.71 | 0.043–0.40 | 59 | 118 (8) | Combined | ||||||
| Oxycodone (Oxymorphone) | ≤0.034 | - | - | - | 2 | 1 (3 cm) | 2 | Headache patients | LC–MS/MS | Licata (2016) [20] |
| >1.0 (n = 2) | - | 0.18 | - | 47 | 1 (app 4 cm) | 47 | Individuals ongoing monitoring program | LC–MS/MS | Reisfield (2015) [84] | |
| ≤0.034–>1 | 49 | 49 | Combined | |||||||
| Tramadol (N-desmethyltramadol) | 0.11–0.83 0.10–0.69 1.4–1.7 | - - - | 0.27 0.34 1.5 | 0.26 0.30 1.5 | 8 8 1 | ≤4 (0.5 cm) | 30 30 4 | Controlled single dose study, extensive metabolizers, intermediate metabolizers and primary metabolizers | LC–MS/MS | Johansen (2020) [85] |
| - - - | - - - | 0.05 0.12 0.24 | - - - | 14 8 1 | Whole strand | (14) (8) (1) | Controlled study, normal CYP2D6 activity, reduced CYP2D6 activity and no CYP2D6 activity | LC–MS/MS | Yu (2018) [86] | |
| 0.093–0.44 | 0.12–0.44 | 0.31 | 0.40 | 4 | 2–8 (2 cm) | 14 | Individuals with kown intake | LC–MS/MS | Verri (2015) [87] | |
| 0.01–0.97 | 0.037–0.87 | - | 0.16 | 75 | 1 (3 cm) | 75 | Patients with intake | LC–MS/MS | Madry (2012) [88] | |
| 0.01–1.7 | 0.16–1.5 | 119 | 153 (23) | Combined | ||||||
| Tramadol (O-desmethyltramadol) | 0.037–0.70 0.080–0.48 0.064–0.086 | - - - | 0.30 0.21 0.072 | 0.29 0.16 0.068 | 8 8 1 | ≤4 (0.5 cm) | 30 30 4 | Controlled single dose study, extensive metabolizers, intermediate metabolizers and primary metabolizers | LC–MS/MS | Johansen (2020) [85] |
| 0.16–0.22 | - | - | - | 205 | 1 (1–6 cm) | 205 | Individuals undergoing toxicological investigation | LC–MS/MS | Musshoff (2020) [89] | |
| 0.25 | - | 0.25 | - | 1 | 1 (0–2 cm) | 1 | Patient undergoing treatment | LC–MS/MS | Wang (2019) [90] | |
| 0.11–0.18 | - | 0.14 | 0.15 | 2 | 1–2 (2–2.5 cm) | 3 | Suspected DFC and DFSA cases | LC–MS/MS | Wang (2018) [28] | |
| - | - | 0.13 0.04 0.02 | - | 14 8 1 | Whole strand | (14) (8) (1) | Controlled study Normal CYP2D6 activity Reduced CYP2D6 activity No CYP2D6 activity | LC–MS/MS | Yu (2018) [86] | |
| ≤0.050 | - | - | - | 14 | 1 (3 cm) | 14 | Headache patients | LC–MS/MS | Licata (2016) [20] | |
| 0.051–0.11 | 0.060–0.10 | 0.077 | 0.069 | 4 | 2–8 (2 cm) | 14 | Individuals with kown intake | LC–MS/MS | Verri (2015) [87] | |
| 0.021–0.45 | - | 0.21 | 0.17 | 6 | Unspecified | ≥6 | Patients undergoing tramadol therapy | GC–MS | Pinho (2013) [91] | |
| 0.003–0.43 | 0.021–0.29 | - | 0.11 | 75 | 1 (3 cm) | 75 | Patients with intake | LC–MS/MS | Madry (2012) [88] | |
| 0.003–0.70 | 0.068–0.29 | 347 | ≥382 (23) | Combined | ||||||
| OTHER PHARMACEUTICALS | ||||||||||
| Atomoxetine (4-hydroxyatomoxetine) | 0.26–2.3 | 0.33–1.6 | 0.87 | 0.66 | 6 | 1–3 (1.5 cm) | 10 | Adolescents + children under treatment | LC–MS/MS | Papaseit (2012) [92] |
| Carisoprodol (Meprobamate) | 1.4 | - | 1.4 | - | 1 | Unspecified | ≥1 | Suspected carisoprodol abuser | GC–MS | Kim (2005) [93] |
| Ketamine (Norketamine) | ≤0.41 | - | 0.12 | 0.06 | 19 | 1 (3 cm) | 19 | Individuals with suspected drug abuse | LC–MS/MS | Zhuo (2020) [94] |
| 0.059–1 | - | 0.29 | 0.17 | 9 | Unspecified (5–7 cm) | 9 | Individuals previously tested positive for ketamine | LC–HRMS | Miolo (2018) [95] | |
| 0.010–1.3 | - | 0.30 | - | 526 | 1 (3 cm) | 526 | Individuals with ketamine abuse | LC–MS/MS | Leung (2016) [96] | |
| 0.06–0.29 0.09–0.26 0.32–0.80 | - - - | 0.14 0.19 0.56 | - - - | 6 8 2 | 1 (0–6 cm) | 6 8 2 | Driving relicensing, driving relicensing, medical cases | LC–MS/MS | Salomone (2015) [97] | |
| 0.033–0.20 | - | 0.12 | 0.12 | 3 | Whole strand | (3) | Individuals with known drug abuse | LC–MS/MS | Chang (2014) [98] | |
| 0.039–0.13 | - | 0.069 | 0.044 | 2 | 1–2 (1.5–3) | 3 | Suspected drug abuser, polydrug intoxication | LC–HRMS | Favretto (2013) [99] | |
| 0.67 | - | 0.67 | - | 1 | unspecified | ≥1 | Presumed drug user | LC–HRMS | Favretto (2011) [51] | |
| 0.08–1.1 | 0.084–0.54 | 0.28 | 0.18 | 10 | 1 (3 cm) | 10 | Individuals with drug abuse | LC–MS/MS | Zhu (2011) [100] | |
| 0.045–0.5 | - | 0.26 | 0.24 | 4 | Whole strand | (4) | Hair from drug misuse prevention center | LC–MS/MS | Harun (2010) [101] | |
| 0.05–0.84 | - | 0.33 | - | 51 | ≥2 (1–3 cm) | 91 | Individuals with suspected ketamine abuse | GC–MS | Leong (2010) [102] | |
| 0.005–1.5 | - | 0.31 | 0.052 | 6 | Whole strand | (6) | Multi drug abusers | LC–MS/MS | Tabernero (2009) [103] | |
| 0.10–0.38 | - | 0.21 | 0.15 | 3 | Full length | (3) | Individuals with a suspected drug abuse | GC–MS | Wu (2008) [104] | |
| 0.043–0.27 | - | 0.16 | 0.17 | 4 | Full length | (4) | Individuals with a drug abuse | GC–MS | Wu (2008) [105] | |
| 0.03–0.88 | 0.043–0.77 | 0.32 | 0.30 | 14 | 1 (3 cm) | 14 | Individuals with a ketamine abuse | GC–MS | Xiang (2006) [106] | |
| 0.005–1.5 | 0.044–0.30 | 668 | ≥686 (20) | Combined | ||||||
| Ketamine (Dehydronorketamine) | 0.012–0.094 | - | 0.050 | 0.045 | 3 | Whole strand | (3) | Individuals with known drug abuse | LC–MS/MS | Chang (2014) [98] |
| Methylphenidate (Ritalinic acid) | 0.15–0.53 | 0.18–0.52 | 0.35 | 0.40 | 7 | 1–4 (3–14 cm) | 17 | Illegal use of methylphenidate | LC–MS/MS | Jang (2019) [107] |
| Oxcarbazepine (10-hydroxycarbamazepine) | 0.39–0.64 | - | 0.51 | 0.50 | 1 | 3 (2 cm) | 3 | PM case, drug addiction | LC–MS/MS | Wang 2017 [40] |
| 4.7–8.1 | - | 6.0 | 5.2 | 1 | 3 (2 cm) | 3 | PM case, psychiatric patient in the past | LC–MS | Klys (2005) [108] | |
| 0.39–8.1 | 0.50–5.2 | 2 | 6 | Combined | ||||||
| Oxcarbazepine (Trans-diol-carbazepine) | 0.12–0.23 | - | 0.16 | 0.13 | 1 | 3 (2 cm) | 3 | PM case, psychiatric patient in the past | LC–MS | Klys (2005) [108] |
| Thiopental (Pentobarbital) | 1.0–1.3 | - | 1.2 | 1.3 | 1 | 3 (1.5) | 3 | DFSA, proximal segments analyzed | GC–MS/MS | Frison (2003) [109] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rygaard, K.; Linnet, K.; Johansen, S.S. A Systematic Review of Metabolite-to-Drug Ratios of Pharmaceuticals in Hair for Forensic Investigations. Metabolites 2021, 11, 686. https://doi.org/10.3390/metabo11100686
Rygaard K, Linnet K, Johansen SS. A Systematic Review of Metabolite-to-Drug Ratios of Pharmaceuticals in Hair for Forensic Investigations. Metabolites. 2021; 11(10):686. https://doi.org/10.3390/metabo11100686
Chicago/Turabian StyleRygaard, Karen, Kristian Linnet, and Sys Stybe Johansen. 2021. "A Systematic Review of Metabolite-to-Drug Ratios of Pharmaceuticals in Hair for Forensic Investigations" Metabolites 11, no. 10: 686. https://doi.org/10.3390/metabo11100686
APA StyleRygaard, K., Linnet, K., & Johansen, S. S. (2021). A Systematic Review of Metabolite-to-Drug Ratios of Pharmaceuticals in Hair for Forensic Investigations. Metabolites, 11(10), 686. https://doi.org/10.3390/metabo11100686






