The Determinants of the Human Milk Metabolome and Its Role in Infant Health
Abstract
1. Introduction
2. Human Milk Metabolome
3. Specific Human Milk (HM) Metabolites and Their Role in Health
3.1. Carbohydrates
3.1.1. Lactose
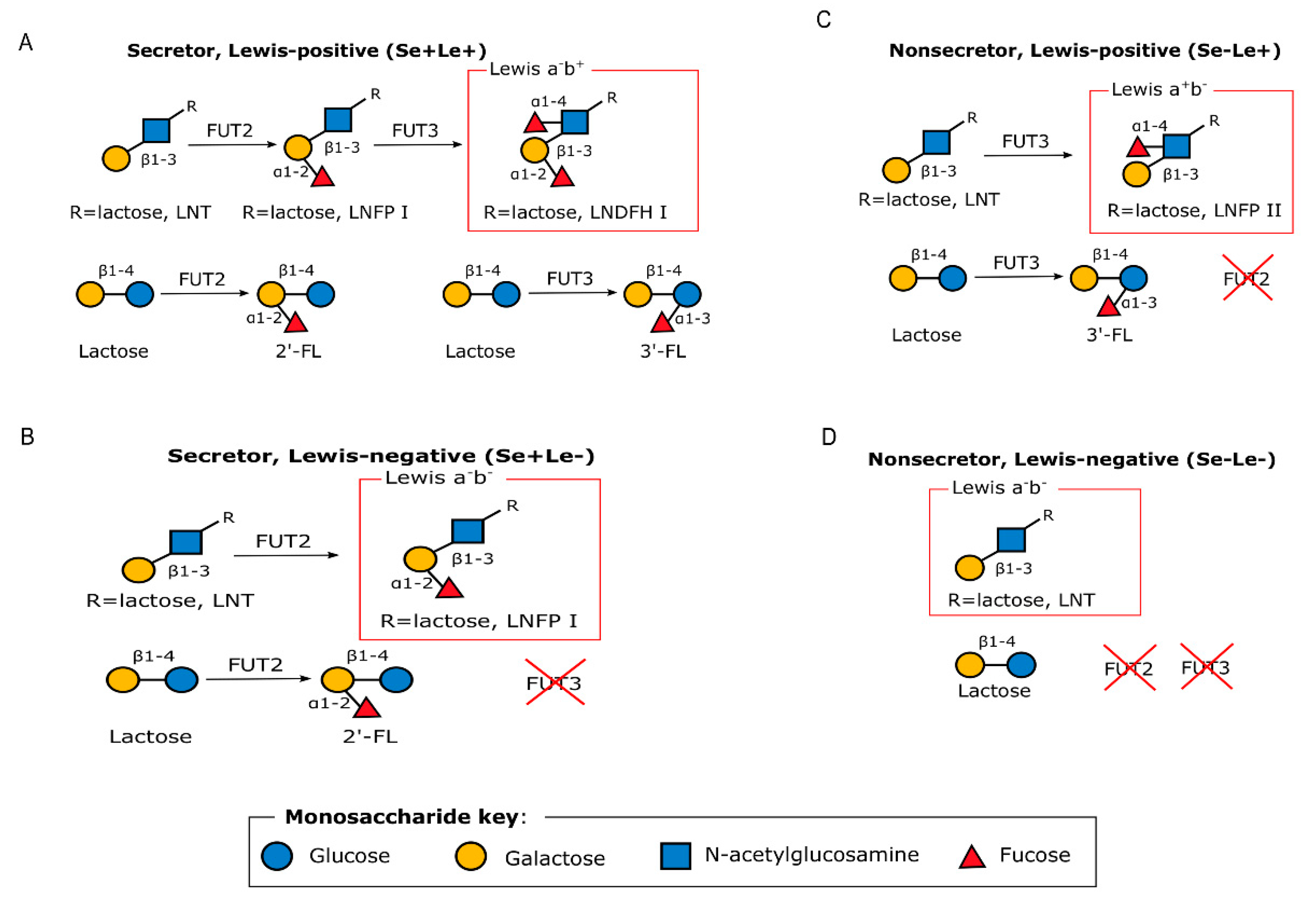
3.1.2. Human Milk Oligosaccharides
3.2. Lipids
3.3. Biogenic Amines
3.4. Nonprotein Nitrogen Molecules
3.4.1. Free Amino Acids
3.4.2. Creatine
4. Determinants of the Human Milk Metabolome
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Goldman, A.S. Evolution of immune functions of the mammary gland and protection of the infant. Breastfeed. Med. 2012, 7, 132–142. [Google Scholar] [CrossRef]
- Zalewski, B.M.; Patro, B.; Veldhorst, M.; Kouwenhoven, S.; Crespo Escobar, P.; Calvo Lerma, J.; Koletzko, B.; van Goudoever, J.B.; Szajewska, H. Nutrition of infants and young children (one to three years) and its effect on later health: A systematic review of current recommendations (earlynutrition project). Crit. Rev. Food Sci. Nutr. 2017, 57, 489–500. [Google Scholar] [CrossRef]
- Lamberti, L.M.; Fischer Walker, C.L.; Noiman, A.; Victora, C.; Black, R.E. Breastfeeding and the risk for diarrhea morbidity and mortality. BMC Public Health 2011, 11 (Suppl. S3), S15. [Google Scholar] [CrossRef] [PubMed]
- Quigley, M.A.; Kelly, Y.J.; Sacker, A. Breastfeeding and hospitalization for diarrheal and respiratory infection in the United Kingdom millennium cohort study. Pediatrics 2007, 119, e837. [Google Scholar] [CrossRef] [PubMed]
- Villamor-Martínez, E.; Pierro, M.; Cavallaro, G.; Mosca, F.; Kramer, B.W.; Villamor, E. Donor human milk protects against bronchopulmonary dysplasia: A systematic review and meta-analysis. Nutrients 2018, 10, 238. [Google Scholar] [CrossRef] [PubMed]
- Miller, J.; Tonkin, E.; Damarell, R.A.; McPhee, A.J.; Suganuma, M.; Suganuma, H.; Middleton, P.F.; Makrides, M.; Collins, C.T. A systematic review and meta-analysis of human milk feeding and morbidity in very low birth weight infants. Nutrients 2018, 10, 707. [Google Scholar] [CrossRef]
- Hylander, M.A.; Strobino, D.M.; Pezzullo, J.C.; Dhanireddy, R. Association of human milk feedings with a reduction in retinopathy of prematurity among very low birthweight infants. J. Perinatol. 2001, 21, 356–362. [Google Scholar] [CrossRef]
- Garrido, D.; Dallas, D.C.; Mills, D.A. Consumption of human milk glycoconjugates by infant-associated bifidobacteria: Mechanisms and implications. Microbiology 2013, 159, 649–664. [Google Scholar] [CrossRef]
- Expert panel on integrated guidelines for cardiovascular health and risk reduction in children and adolescents: Summary report. Available online: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4536582/ (accessed on 1 December 2011).
- Uwaezuoke, S.N.; Eneh, C.I.; Ndu, I.K. Relationship between exclusive breastfeeding and lower risk of childhood obesity: A narrative review of published evidence. Clin. Med. Insights Pediatr. 2017, 11, 1179556517690196. [Google Scholar] [CrossRef]
- Victora, C.G.; Bahl, R.; Barros, A.J.; Franca, G.V.; Horton, S.; Krasevec, J.; Murch, S.; Sankar, M.J.; Walker, N.; Rollins, N.C.; et al. Breastfeeding in the 21st century: Epidemiology, mechanisms, and lifelong effect. Lancet 2016, 387, 475–490. [Google Scholar] [CrossRef]
- Martin, C.R.; Ling, P.-R.; Blackburn, G.L. Review of infant feeding: Key features of breast milk and infant formula. Nutrients 2016, 8, 279. [Google Scholar] [CrossRef]
- Wishart, D.S. Proteomics and the human metabolome project. Expert Rev. Proteom. 2007, 4, 333–335. [Google Scholar] [CrossRef]
- Kresge, N.; Simoni, R.D. Lactose synthesis in the mammary gland: Lactose synthase and the work of Robert L. Hill. J. Biol. Chem. 2006, 281, e6. [Google Scholar]
- Sundekilde, U.K.; Larsen, L.B.; Bertram, H.C. NMR-based milk metabolomics. Metabolites 2013, 3, 204–222. [Google Scholar] [CrossRef]
- Smilowitz, J.T.; O’Sullivan, A.; Barile, D.; German, J.B.; Lonnerdal, B.; Slupsky, C.M. The human milk metabolome reveals diverse oligosaccharide profiles. J. Nutr. 2013, 143, 1709–1718. [Google Scholar] [CrossRef]
- Alonso, A.; Marsal, S.; Julià, A. Analytical methods in untargeted metabolomics: State of the art in 2015. Front. Bioeng. Biotechnol. 2015, 3, 23. [Google Scholar] [CrossRef]
- Wishart, D.S. Quantitative metabolomics using NMR. TrAC Trends Anal. Chem. 2008, 27, 228–237. [Google Scholar] [CrossRef]
- Emwas, A.-H.M. The strengths and weaknesses of NMR spectroscopy and mass spectrometry with particular focus on metabolomics research. In Metabonomics: Methods and Protocols; Bjerrum, J.T., Ed.; Springer: New York, NY, USA, 2015; pp. 161–193. [Google Scholar]
- Dunn, W.B.; Bailey, N.J.; Johnson, H.E. Measuring the metabolome: Current analytical technologies. Analyst 2005, 130, 606–625. [Google Scholar] [CrossRef]
- Marincola, F.C.; Noto, A.; Caboni, P.; Reali, A.; Barberini, L.; Lussu, M.; Murgia, F.; Santoru, M.L.; Atzori, L.; Fanos, V. A metabolomic study of preterm human and formula milk by high resolution NMR and GC/MS analysis: Preliminary results. J. Matern. Fetal Neonatal Med. 2012, 25, 62–67. [Google Scholar] [CrossRef]
- Longini, M.; Tataranno, M.L.; Proietti, F.; Tortoriello, M.; Belvisi, E.; Vivi, A.; Tassini, M.; Perrone, S.; Buonocore, G. A metabolomic study of preterm and term human and formula milk by proton MRS analysis: Preliminary results. J. Matern. Fetal Neonatal Med. 2014, 27, 27–33. [Google Scholar] [CrossRef]
- Pratico, G.; Capuani, G.; Tomassini, A.; Baldassarre, M.E.; Delfini, M.; Miccheli, A. Exploring human breast milk composition by NMR-based metabolomics. Nat. Prod. Res. 2014, 28, 95–101. [Google Scholar] [CrossRef]
- Qian, L.; Zhao, A.; Zhang, Y.; Chen, T.; Zeisel, S.H.; Jia, W.; Cai, W. Metabolomic approaches to explore chemical diversity of human breast-milk, formula milk and bovine milk. Int. J. Mol. Sci. 2016, 17, 2128. [Google Scholar] [CrossRef]
- Hennet, T.; Borsig, L. Breastfed at tiffany’s. Trends Biochem. Sci. 2016, 41, 508–518. [Google Scholar] [CrossRef]
- Koletzko, B.; Agostoni, C.; Bergmann, R.; Ritzenthaler, K.; Shamir, R. Physiological aspects of human milk lipids and implications for infant feeding: A workshop report. Acta Paediatr. 2011, 100, 1405–1415. [Google Scholar] [CrossRef]
- Ballard, O.; Morrow, A.L. Human milk composition: Nutrients and bioactive factors. Pediatr. Clin. 2013, 60, 49–74. [Google Scholar]
- Cederlund, A.; Kai-Larsen, Y.; Printz, G.; Yoshio, H.; Alvelius, G.; Lagercrantz, H.; Strömberg, R.; Jörnvall, H.; Gudmundsson, G.H.; Agerberth, B. Lactose in human breast milk an inducer of innate immunity with implications for a role in intestinal homeostasis. PLoS ONE 2013, 8, e53876. [Google Scholar] [CrossRef]
- Boix-Amoros, A.; Collado, M.C.; Mira, A. Relationship between milk microbiota, bacterial load, macronutrients, and human cells during lactation. Front. Microbiol. 2016, 7, 492. [Google Scholar] [CrossRef]
- Davin-Regli, A.; Pagès, J.-M. Enterobacter aerogenes and enterobacter cloacae; versatile bacterial pathogens confronting antibiotic treatment. Front. Microbiol. 2015, 6. [Google Scholar] [CrossRef]
- Könönen, E.; Wade, W.G. Actinomyces and related organisms in human infections. Clin. Microbiol. Rev. 2015, 28, 419–442. [Google Scholar] [CrossRef]
- Prentice, P.; Ong, K.K.; Schoemaker, M.H.; van Tol, E.A.; Vervoort, J.; Hughes, I.A.; Acerini, C.L.; Dunger, D.B. Breast milk nutrient content and infancy growth. Acta Paediatr. 2016, 105, 641–647. [Google Scholar] [CrossRef] [PubMed]
- Khan, S.; Hepworth, A.R.; Prime, D.K.; Lai, C.T.; Trengove, N.J.; Hartmann, P.E. Variation in fat, lactose, and protein composition in breast milk over 24 hours: Associations with infant feeding patterns. J. Hum. Lact. 2013, 29, 81–89. [Google Scholar] [CrossRef] [PubMed]
- Nilsson, A.; Mardinoglu, A.; Nielsen, J. Predicting growth of the healthy infant using a genome scale metabolic model. NPJ Syst. Biol. Appl. 2017, 3, 1–8. [Google Scholar] [CrossRef]
- Ziegler, E.E.; Fomon, S.J. Lactose enhances mineral absorption in infancy. J. Pediatr. Gastroenterol. Nutr. 1983, 2, 288–294. [Google Scholar] [CrossRef]
- Goehring, K.C.; Kennedy, A.D.; Prieto, P.A.; Buck, R.H. Direct evidence for the presence of human milk oligosaccharides in the circulation of breastfed infants. PLoS ONE 2014, 9, e101692. [Google Scholar] [CrossRef]
- Urashima, T.; Hirabayashi, J.; Sato, S.; Kobata, A. Human milk oligosaccharides as essential tools for basic and application studies on galectins. Trends Glycosci. Glycotechnol. 2018, 30, SE51–SE65. [Google Scholar] [CrossRef]
- Aakko, J.; Kumar, H.; Rautava, S.; Wise, A.; Autran, C.; Bode, L.; Isolauri, E.; Salminen, S. Human milk oligosaccharide categories define the microbiota composition in human colostrum. Benef. Microbes 2017, 8, 563–567. [Google Scholar] [CrossRef]
- Dessì, A.; Briana, D.; Corbu, S.; Gavrili, S.; Cesare Marincola, F.; Georgantzi, S.; Pintus, R.; Fanos, V.; Malamitsi-Puchner, A. Metabolomics of breast milk: The importance of phenotypes. Metabolites 2018, 8, 79. [Google Scholar] [CrossRef]
- Bode, L. The functional biology of human milk oligosaccharides. Early Hum. Dev. 2015, 91, 619–622. [Google Scholar] [CrossRef]
- Bode, L. Human milk oligosaccharides: Every baby needs a sugar mama. Glycobiology 2012, 22, 1147–1162. [Google Scholar] [CrossRef]
- Walker, W.A.; Iyengar, R.S. Breast milk, microbiota, and intestinal immune homeostasis. Pediatr. Res. 2015, 77, 220–228. [Google Scholar] [CrossRef]
- Smilowitz, J.T.; Lebrilla, C.B.; Mills, D.A.; German, J.B.; Freeman, S.L. Breast milk oligosaccharides: Structure-function relationships in the neonate. Annu. Rev. Nutr. 2014, 34, 143–169. [Google Scholar] [CrossRef]
- Sun, Y.; O’Riordan, M.X. Regulation of bacterial pathogenesis by intestinal short-chain fatty acids. Adv. Appl. Microbiol. 2013, 85, 93–118. [Google Scholar]
- Smith, P.M.; Howitt, M.R.; Panikov, N.; Michaud, M.; Gallini, C.A.; Bohlooly, Y.M.; Glickman, J.N.; Garrett, W.S. The microbial metabolites, short-chain fatty acids, regulate colonic treg cell homeostasis. Science 2013, 341, 569–573. [Google Scholar] [CrossRef]
- Fukuda, S.; Toh, H.; Hase, K.; Oshima, K.; Nakanishi, Y.; Yoshimura, K.; Tobe, T.; Clarke, J.M.; Topping, D.L.; Suzuki, T. Bifidobacteria can protect from enteropathogenic infection through production of acetate. Nature 2011, 469, 543. [Google Scholar] [CrossRef]
- Berni Canani, R.; Di Costanzo, M.; Leone, L. The epigenetic effects of butyrate: Potential therapeutic implications for clinical practice. Clin. Epigenet. 2012, 4, 4. [Google Scholar] [CrossRef]
- Canani, R.B.; Costanzo, M.D.; Leone, L.; Pedata, M.; Meli, R.; Calignano, A. Potential beneficial effects of butyrate in intestinal and extraintestinal diseases. World J. Gastroenterol. 2011, 17, 1519–1528. [Google Scholar] [CrossRef]
- Newburg, D.S.; Pickering, L.K.; McCluer, R.H.; Cleary, T.G. Fucosylated oligosaccharides of human milk protect suckling mice from heat-stabile enterotoxin of escherichia coli. J. Infect. Dis. 1990, 162, 1075–1080. [Google Scholar] [CrossRef]
- Ruiz-Palacios, G.M.; Cervantes, L.E.; Ramos, P.; Chavez-Munguia, B.; Newburg, D.S. Campylobacter jejuni binds intestinal h (o) antigen (fuca1, 2galb1, 4glcnac), and fucosyloligosaccharides of human milk ihibit its binding and infection. J. Biol. Chem. 2003, 278, 14112–14120. [Google Scholar] [CrossRef]
- Jiang, X.; Huang, P.; Zhong, W.; Tan, M.; Farkas, T.; Morrow, A.L.; Newburg, D.S.; Ruiz-Palacios, G.M.; Pickering, L.K. Human milk contains elements that block binding of noroviruses to human histo—Blood group antigens in saliva. J. Infect. Dis. 2004, 190, 1850–1859. [Google Scholar] [CrossRef]
- Bode, L. Recent advances on structure, metabolism, and function of human milk oligosaccharides. J. Nutr. 2006, 136, 2127–2130. [Google Scholar] [CrossRef]
- Jantscher-Krenn, E.; Lauwaet, T.; Bliss, L.A.; Reed, S.L.; Gillin, F.D.; Bode, L. Human milk oligosaccharides reduce entamoeba histolytica attachment and cytotoxicity in vitro. Br. J. Nutr. 2012, 108, 1839–1846. [Google Scholar] [CrossRef]
- Gonia, S.; Tuepker, M.; Heisel, T.; Autran, C.; Bode, L.; Gale, C.A. Human milk oligosaccharides inhibit candida albicans invasion of human premature intestinal epithelial cells, 2. J. Nutr. 2015, 145, 1992–1998. [Google Scholar] [CrossRef]
- Van Niekerk, E.; Autran, C.A.; Nel, D.G.; Kirsten, G.F.; Blaauw, R.; Bode, L. Human milk oligosaccharides differ between hiv-infected and hiv-uninfected mothers and are related to necrotizing enterocolitis incidence in their preterm very-low-birth-weight infants–3. J. Nutr. 2014, 144, 1227–1233. [Google Scholar] [CrossRef]
- Yuhas, R.; Pramuk, K.; Lien, E.L. Human milk fatty acid composition from nine countries varies most in dha. Lipids 2006, 41, 851–858. [Google Scholar] [CrossRef]
- Dizdar, E.A.; Sari, F.N.; Degirmencioglu, H.; Canpolat, F.E.; Oguz, S.S.; Uras, N.; Dilmen, U. Effect of mode of delivery on macronutrient content of breast milk. J. Matern. Fetal Neonatal Med. 2014, 27, 1099–1102. [Google Scholar] [CrossRef]
- Delplanque, B.; Gibson, R.; Koletzko, B.; Lapillonne, A.; Strandvik, B. Lipid quality in infant nutrition: Current knowledge and future opportunities. J. Pediatr. Gastroenterol. Nutr. 2015, 61, 8–17. [Google Scholar] [CrossRef]
- Jensen, R.G. Lipids in human milk. Lipids 1999, 34, 1243–1271. [Google Scholar] [CrossRef]
- Schmidt, R.; Meier, U.; Markart, P.; Grimminger, F.; Velcovsky, H.; Morr, H.; Seeger, W.; Gunther, A. Altered fatty acid composition of lung surfactant phospholipidsin interstitial lung disease. Am. J. Physiol. Lung Cell. Mol. Physiol. 2002, 283, L1079–L1085. [Google Scholar] [CrossRef]
- Andreas, N.J.; Kampmann, B.; Le-Doare, K.M. Human breast milk: A review on its composition and bioactivity. Early Hum. Dev. 2015, 91, 629–635. [Google Scholar] [CrossRef]
- Tanaka, K.; Hosozawa, M.; Kudo, N.; Yoshikawa, N.; Hisata, K.; Shoji, H.; Shinohara, K.; Shimizu, T. The pilot study: Sphingomyelin-fortified milk has a positive association with the neurobehavioural development of very low birth weight infants during infancy, randomized control trial. Brain Dev. 2013, 35, 45–52. [Google Scholar] [CrossRef] [PubMed]
- Zou, X.; Ali, A.H.; Abed, S.M.; Guo, Z. Current knowledge of lipids in human milk and recent innovations in infant formulas. Curr. Opin. Food Sci. 2017, 16, 28–39. [Google Scholar] [CrossRef]
- Bar-Yoseph, F.; Lifshitz, Y.; Cohen, T. Review of sn-2 palmitate oil implications for infant health. PLEFA Prostaglandins Leukot. Essent. Fat. Acids 2013, 89, 139–143. [Google Scholar] [CrossRef] [PubMed]
- Mehrotra, V.; Sehgal, S.K.; Bangale, N.R. Fat structure and composition in human milk and infant formulas: Implications in infant health. Clin. Epidemiol. Glob. Health 2018, 7, 153–159. [Google Scholar] [CrossRef]
- Koletzko, B.; Lien, E.; Agostoni, C.; Bohles, H.; Campoy, C.; Cetin, I.; Decsi, T.; Dudenhausen, J.W.; Dupont, C.; Forsyth, S.; et al. The roles of long-chain polyunsaturated fatty acids in pregnancy, lactation and infancy: Review of current knowledge and consensus recommendations. J. Perinat. Med. 2008, 36, 5–14. [Google Scholar] [CrossRef]
- Waidyatillake, N.T.; Dharmage, S.C.; Allen, K.J.; Lodge, C.J.; Simpson, J.A.; Bowatte, G.; Abramson, M.J.; Lowe, A.J. Association of breast milk fatty acids with allergic disease outcomes-a systematic review. Allergy 2017, 73, 295–312. [Google Scholar] [CrossRef]
- Logan, C.A.; Brandt, S.; Wabitsch, M.; Brenner, H.; Wiens, F.; Stahl, B.; Marosvoelgyi, T.; Decsi, T.; Rothenbacher, D.; Genuneit, J. New approach shows no association between maternal milk fatty acid composition and childhood wheeze or asthma. Allergy 2017, 72, 1374–1383. [Google Scholar] [CrossRef]
- Demmelmair, H.; Koletzko, B. Lipids in human milk. Best Pract. Res. Clin. Endocrinol. Metab. 2018, 32, 57–68. [Google Scholar] [CrossRef]
- Jensen, C.L.; Voigt, R.G.; Prager, T.C.; Zou, Y.L.; Fraley, J.K.; Rozelle, J.C.; Turcich, M.R.; Llorente, A.M.; Anderson, R.E.; Heird, W.C. Effects of maternal docosahexaenoic acid intake on visual function and neurodevelopment in breastfed term infants–. Am. J. Clin. Nutr. 2005, 82, 125–132. [Google Scholar] [CrossRef]
- Yu, G.; Bjorksten, B. Serum levels of phospholipid fatty acids in mothers and their babies in relation to allergic disease. Eur. J. Pediatr. 1998, 157, 298–303. [Google Scholar] [CrossRef]
- Duchen, K.; Casas, R.; Fageras-Bottcher, M.; Yu, G.; Bjorksten, B. Human milk polyunsaturated long-chain fatty acids and secretory immunoglobulin A antibodies and early childhood allergy. Pediatr. Allergy Immunol. 2000, 11, 29–39. [Google Scholar] [CrossRef] [PubMed]
- Perez, M.; Ladero, V.; Redruello, B.; Del Rio, B.; Fernandez, L.; Rodriguez, J.M.; Martin, M.C.; Fernandez, M.; Alvarez, M.A. Mastitis modifies the biogenic amines profile in human milk, with significant changes in the presence of histamine, putrescine and spermine. PLoS ONE 2016, 11, e0162426. [Google Scholar] [CrossRef] [PubMed]
- Jiménez, E.; Ladero, V.; Chico, I.; Maldonado-Barragán, A.; López, M.; Martín, V.; Fernández, L.; Fernández, M.; Álvarez, M.A.; Torres, C.; et al. Antibiotic resistance, virulence determinants and production of biogenic amines among enterococci from ovine, feline, canine, porcine and human milk. BMC Microbiol. 2013, 13, 288. [Google Scholar] [CrossRef] [PubMed]
- Plaza-Zamora, J.; Sabater-Molina, M.; Rodriguez-Palmero, M.; Rivero, M.; Bosch, V.; Nadal, J.M.; Zamora, S.; Larque, E. Polyamines in human breast milk for preterm and term infants. Br. J. Nutr. 2013, 110, 524–528. [Google Scholar] [CrossRef] [PubMed]
- Zhang, M.; Caragine, T.; Wang, H.; Cohen, P.S.; Botchkina, G.; Soda, K.; Bianchi, M.; Ulrich, P.; Cerami, A.; Sherry, B.; et al. Spermine inhibits proinflammatory cytokine synthesis in human mononuclear cells: A counterregulatory mechanism that restrains the immune response. J. Exp. Med. 1997, 185, 1759–1768. [Google Scholar] [CrossRef]
- Ali, M.A.; Strandvik, B.; Palme-Kilander, C.; Yngve, A. Lower polyamine levels in breast milk of obese mothers compared to mothers with normal body weight. J. Hum. Nutr. Diet. 2013, 26 (Suppl. S1), 164–170. [Google Scholar] [CrossRef]
- Gómez-Gallego, C.; Kumar, H.; García-Mantrana, I.; du Toit, E.; Suomela, J.-P.; Linderborg, K.M.; Zhang, Y.; Isolauri, E.; Yang, B.; Salminen, S. Breast milk polyamines and microbiota interactions: Impact of mode of delivery and geographical location. Ann. Nutr. Metab. 2017, 70, 184–190. [Google Scholar] [CrossRef]
- Peulen, O.; Dewé, W.; Dandrifosse, G.; Henrotay, I.; Romain, N. The relationship between spermine content of human milk during the first postnatal month and allergy in children. Public Health Nutr. 1998, 1, 181–184. [Google Scholar] [CrossRef]
- Duchén, K.; Thorell, L. Nucleotide and polyamine levels in colostrum and mature milk in relation to maternal atopy and atopic development in the children. Acta Paediatr. 1999, 88, 1338–1343. [Google Scholar] [CrossRef]
- Edison, E.E.; Brosnan, M.E.; Aziz, K.; Brosnan, J.T. Creatine and guanidinoacetate content of human milk and infant formulas: Implications for creatine deficiency syndromes and amino acid metabolism. Br. J. Nutr. 2013, 110, 1075–1078. [Google Scholar] [CrossRef]
- Xiao, G.; Xiao, H.; Zhu, Y.; You, Y. Determination of nucleotides in chinese human milk by high-performance liquid chromatography–tandem mass spectrometry. Dairy Sci. Technol. 2014, 94, 591–602. [Google Scholar] [CrossRef]
- Agostoni, C.; Carratu, B.; Boniglia, C.; Riva, E.; Sanzini, E. Free amino acid content in standard infant formulas: Comparison with human milk. J. Am. Coll. Nutr. 2000, 19, 434–438. [Google Scholar] [CrossRef] [PubMed]
- Larnkjær, A.; Bruun, S.; Pedersen, D.; Zachariassen, G.; Barkholt, V.; Agostoni, C.; Christian, M.; Husby, S.; Michaelsen, K.F. Free amino acids in human milk and associations with maternal anthropometry and infant growth. J. Pediatr. Gastroenterol. Nutr. 2016, 63, 374–378. [Google Scholar] [CrossRef] [PubMed]
- Burrin, D.G.; Stoll, B. Metabolic fate and function of dietary glutamate in the gut. Am. J. Clin. Nutr. 2009, 90, 850S–856S. [Google Scholar] [CrossRef] [PubMed]
- Emmett, P.M.; Rogers, I.S. Properties of human milk and their relationship with maternal nutrition. Early Hum. Dev. 1997, 49, S7–S28. [Google Scholar] [CrossRef]
- Chawla, D. Taurine and neonatal nutrition. Indian J. Pediatr. 2018, 85, 829. [Google Scholar] [CrossRef]
- Sánchez, C.L.; Cubero, J.; Sánchez, J.; Franco, L.; Rodríguez, A.B.; Rivero, M.; Barriga, C. Evolution of the circadian profile of human milk amino acids during breastfeeding. J. Appl. Biomed. 2013, 11, 59–70. [Google Scholar] [CrossRef]
- da Silva, R.P.; Nissim, I.; Brosnan, M.E.; Brosnan, J.T. Creatine synthesis: Hepatic metabolism of guanidinoacetate and creatine in the rat in vitro and in vivo. Am. J. Physiol. Endocrinol. Metab. 2009, 296, E256–E261. [Google Scholar] [CrossRef]
- Foxman, B.; D’Arcy, H.; Gillespie, B.; Bobo, J.K.; Schwartz, K. Lactation mastitis: Occurrence and medical management among 946 breastfeeding women in the united states. Am. J. Epidemiol. 2002, 155, 103–114. [Google Scholar] [CrossRef]
- Bermúdez, R.; Lorenzo, J.M.; Fonseca, S.; Franco, I.; Carballo, J. Strains of staphylococcus and bacillus isolated from traditional sausages as producers of biogenic amines. Front. Microbiol. 2012, 3, 151. [Google Scholar] [CrossRef]
- Hunt, K.M.; Williams, J.E.; Shafii, B.; Hunt, M.K.; Behre, R.; Ting, R.; McGuire, M.K.; McGuire, M.A. Mastitis is associated with increased free fatty acids, somatic cell count, and interleukin-8 concentrations in human milk. Breastfeed. Med. 2013, 8, 105–110. [Google Scholar] [CrossRef] [PubMed]
- Kelsey, J.; Bayles, K.W.; Shafii, B.; McGuire, M. Fatty acids and monoacylglycerols inhibit growth ofstaphylococcus aureus. Lipids 2006, 41, 951–961. [Google Scholar] [CrossRef] [PubMed]
- Bender, J.M.; Li, F.; Martelly, S.; Byrt, E.; Rouzier, V.; Leo, M.; Tobin, N.; Pannaraj, P.S.; Adisetiyo, H.; Rollie, A.; et al. Maternal HIV infection influences the microbiome of HIV uninfected infants. Sci. Transl. Med. 2016, 8, 349ra100. [Google Scholar] [CrossRef] [PubMed]
- Bode, L.; Kuhn, L.; Kim, H.-Y.; Hsiao, L.; Nissan, C.; Sinkala, M.; Kankasa, C.; Mwiya, M.; Thea, D.M.; Aldrovandi, G.M. Human milk oligosaccharide concentration and risk of postnatal transmission of HIV through breastfeeding. Am. J. Clin. Nutr. 2012, 96, 831–839. [Google Scholar] [CrossRef]
- Kuhn, L.; Kim, H.-Y.; Hsiao, L.; Nissan, C.; Kankasa, C.; Mwiya, M.; Thea, D.M.; Aldrovandi, G.M.; Bode, L. Oligosaccharide composition of breast milk influences survival of uninfected children born to HIV-infected mothers in lusaka, zambia, 2. J. Nutr. 2014, 145, 66–72. [Google Scholar] [CrossRef]
- Makela, J.; Linderborg, K.; Niinikoski, H.; Yang, B.; Lagstrom, H. Breast milk fatty acid composition differs between overweight and normal weight women: The steps study. Eur. J. Nutr. 2013, 52, 727–735. [Google Scholar] [CrossRef]
- Urbaniak, C.; McMillan, A.; Angelini, M.; Gloor, G.B.; Sumarah, M.; Burton, J.P.; Reid, G. Effect of chemotherapy on the microbiota and metabolome of human milk, a case report. Microbiome 2014, 2, 24. [Google Scholar] [CrossRef]
- Sanders, T.A.; Reddy, S. The influence of a vegetarian diet on the fatty acid composition of human milk and the essential fatty acid status of the infant. J. Pediatr. 1992, 120, S71–S77. [Google Scholar] [CrossRef]
- Nayak, U.; Kanungo, S.; Zhang, D.; Ross Colgate, E.; Carmolli, M.P.; Dey, A.; Alam, M.; Manna, B.; Nandy, R.K.; Kim, D.R.; et al. Influence of maternal and socioeconomic factors on breast milk fatty acid composition in urban, low-income families. Matern. Child Nutr. 2017, 13, e12423. [Google Scholar] [CrossRef]
- Gómez-Gallego, C.; Morales, J.; Monleón, D.; du Toit, E.; Kumar, H.; Linderborg, K.; Zhang, Y.; Yang, B.; Isolauri, E.; Salminen, S.; et al. Human breast milk nmr metabolomic profile across specific geographical locations and its association with the milk microbiota. Nutrients 2018, 10, 1355. [Google Scholar] [CrossRef]
- Kumar, H.; du Toit, E.; Kulkarni, A.; Aakko, J.; Linderborg, K.M.; Zhang, Y.; Nicol, M.P.; Isolauri, E.; Yang, B.; Collado, M.C. Distinct patterns in human milk microbiota and fatty acid profiles across specific geographic locations. Front. Microbiol. 2016, 7, 1619. [Google Scholar] [CrossRef] [PubMed]
- Gay, M.; Koleva, P.; Slupsky, C.; Toit, E.; Eggesbo, M.; Johnson, C.; Wegienka, G.; Shimojo, N.; Campbell, D.; Prescott, S.; et al. Worldwide variation in human milk metabolome: Indicators of breast physiology and maternal lifestyle? Nutrients 2018, 10, 1151. [Google Scholar] [CrossRef] [PubMed]
- Sundekilde, U.K.; Downey, E.; O’Mahony, J.A.; O’Shea, C.A.; Ryan, C.A.; Kelly, A.L.; Bertram, H.C. The effect of gestational and lactational age on the human milk metabolome. Nutrients 2016, 8, 304. [Google Scholar] [CrossRef] [PubMed]
- Lapillonne, A.; Groh-Wargo, S.; Gonzalez, C.H.; Uauy, R. Lipid needs of preterm infants: Updated recommendations. J. Pediatr. 2013, 162, S37–S47. [Google Scholar] [CrossRef]
- Thurl, S.; Munzert, M.; Henker, J.; Boehm, G.; Muller-Werner, B.; Jelinek, J.; Stahl, B. Variation of human milk oligosaccharides in relation to milk groups and lactational periods. Br. J. Nutr. 2010, 104, 1261–1271. [Google Scholar] [CrossRef]
- Spevacek, A.R.; Smilowitz, J.T.; Chin, E.L.; Underwood, M.A.; German, J.B.; Slupsky, C.M. Infant maturity at birth reveals minor differences in the maternal milk metabolome in the first month of lactation. J. Nutr. 2015, 145, 1698–1708. [Google Scholar] [CrossRef]
- Coppa, G.V.; Gabrielli, O.; Pierani, P.; Catassi, C.; Carlucci, A.; Giorgi, P.L. Changes in carbohydrate composition in human milk over 4 months of lactation. Pediatrics 1993, 91, 637–641. [Google Scholar]
- van Sadelhoff, J.; Mastorakou, D.; Weenen, H.; Stahl, B.; Garssen, J.; Hartog, A. Short communication: Differences in levels of free amino acids and total protein in human foremilk and hindmilk. Nutrients 2018, 10, 1828. [Google Scholar] [CrossRef]
- Dangat, K.; Upadhyay, D.; Kilari, A.; Sharma, U.; Kemse, N.; Mehendale, S.; Lalwani, S.; Wagh, G.; Joshi, S.; Jagannathan, N.R. Altered breast milk components in preeclampsia; an in-vitro proton nmr spectroscopy study. Clin. Chim. Acta 2016, 463, 75–83. [Google Scholar] [CrossRef]
- Atiya Ali, M.; Strandvik, B.; Sabel, K.G.; Palme Kilander, C.; Stromberg, R.; Yngve, A. Polyamine levels in breast milk are associated with mothers’ dietary intake and are higher in preterm than full-term human milk and formulas. J. Hum. Nutr. Diet. 2014, 27, 459–467. [Google Scholar] [CrossRef]
- Bauer, J.; Gerss, J. Longitudinal analysis of macronutrients and minerals in human milk produced by mothers of preterm infants. Clin. Nutr. 2011, 30, 215–220. [Google Scholar] [CrossRef] [PubMed]
- Gabrielli, O.; Zampini, L.; Galeazzi, T.; Padella, L.; Santoro, L.; Peila, C.; Giuliani, F.; Bertino, E.; Fabris, C.; Coppa, G.V. Preterm milk oligosaccharides during the first month of lactation. Pediatrics 2011, 128, e1520–e1531. [Google Scholar] [CrossRef] [PubMed]
- Perrone, S.; Longini, M.; Zollino, I.; Bazzini, F.; Tassini, M.; Vivi, A.; Bracciali, C.; Calderisi, M.; Buonocore, G. Breast milk: To each his own. From metabolomic study, evidence of personalized nutrition in preterm infants. Nutrition 2019, 62, 158–161. [Google Scholar] [CrossRef] [PubMed]
- Villasenor, A.; Garcia-Perez, I.; Garcia, A.; Posma, J.M.; Fernandez-Lopez, M.; Nicholas, A.J.; Modi, N.; Holmes, E.; Barbas, C. Breast milk metabolome characterization in a single-phase extraction, multiplatform analytical approach. Anal. Chem. 2014, 86, 8245–8252. [Google Scholar] [CrossRef] [PubMed]
- Boersma, E.R.; Offringa, P.J.; Muskiet, F.A.; Chase, W.M.; Simmons, I.J. Vitamin e, lipid fractions, and fatty acid composition of colostrum, transitional milk, and mature milk: An international comparative study. Am. J. Clin. Nutr. 1991, 53, 1197–1204. [Google Scholar] [CrossRef] [PubMed]
| Lipid Classes | Proportion of Total Lipids in HM (%) |
|---|---|
| Triacylglycerides | 98.1–98.8 |
| Phospholipids | 0.26–0.8 |
| Cholesterol | 0.25–0.34 |
| Non-esterified fatty acids (free fatty acids) | 0.08–0.4 |
| Diacylglycerides | 0.01–0.7 |
| Monoacylglycerides | Traces |
| Long Chain-Polyunsaturated Fatty Acids (LC-PUFA) | Source, Functions and Determinants | References |
|---|---|---|
| n-3 series | ||
| Docohexaenoic acid (DHA) | Higher DHA concentration in HM leads to improved neurodevelopmental and vision outcomes in infants. DHA concentration is greatly influenced by dietary intake especially of fish or fish oil supplements in lactating mothers. A dose dependent relationship exists between maternal DHA intake and its concentration in HM, as an increased DHA concentration in HM has been observed in lactating women supplemented with DHA. A lower AA to DHA ratio is found in HM of women from Asian and Scandinavian countries due to higher feeding of DHA-rich fish. | [56,58,66,69,70] |
| Eicosapentaenoic acid (EPA) | EPA competes with AA for 5-lipoxygenase and cyclooxygenase enzymes needed for the metabolism of AA, thereby antagonizing the pro-inflammatory effects of AA. Lower concentrations of EPA and DHA in HM have been associated with allergy in children. | [71,72] |
| n-6 series | ||
| Arachidonic acid (AA) | AA is the most abundant of the PUFA with ~0.5% of total fatty acids in HM. AA content of HM is relatively stable among women despite variations in diet and lifestyles as it is derived from pre-existing maternal stores. AA serves as a progenitor to signaling molecules, leukotrienes (LTs), thromboxane and the prostaglandins. These products have inflammatory and atherogenic effects on cells. High AA:EPA ratio in HM has been associated with the development of allergy symptoms at 18 months. | [58,71,72] |
| Factors Influencing Human Milk (HM) Composition | Metabolome | References |
|---|---|---|
| Maternal Health | ||
| HIV | ↑ 3′-SL, ↓ LNT ↓LNnT | [55,94,95] |
| Mastitis | ↑ spermine, ↑ putrescine, ↑ histamine, ↑ FFA | [73] |
| Pre-eclampsia | ↓ Oligosaccharides, ↓ lactose, ↓ glutamate, ↓ glutamine and ↓ glycerophosphocholine | [110] |
| Maternal weight | ||
| Overweight mothers | ↑ SFA, ↓ n-3 PUFA, ↓ Unsaturated FA: Saturated FA | [97] |
| Obese mothers | ↓ Polyamines | [77] |
| Medication | ||
| Chemotherapy | ↓ Docosahexaenoic acid, ↓ Inositol | [98] |
| Sociodemographic factors | ||
| Maternal diet | ||
| Vegetarian diet | ↑ LA, ↑ ALA, ↓ DHA | [66,99] |
| Maternal education | ||
| ↑ Higher education | ↑ PUFA: SFA | [100] |
| Geographical location | ||
| Chinese women | ↑ n-6 PUFA, ↓ SFA, ↑ 3′FL, ↑ LNFP III | [101,102] |
| Spanish women | ↑ 2′-FL, ↑ Putrescine | [78,101] |
| Finland women | ↑ LNFP I, ↑ spermidine | [78,101] |
| South African women | ↑ Lactose, ↑ 2-oxoglutarate, ↑ citrate | [103] |
| Infant factors | ||
| Gestational age | ||
| Preterm | ↑ DHA, ↑lactose, ↑ HMO conc., ↑phosphocholine, ↑choline, ↑glutamate, ↑3′-SL, ↑ 6′-SL, ↑ polyamine conc., ↓ spermidine/spermine | [75,104,105,111,112,113,114] |
| Full term | ↑carnitine, ↑caprylate, ↑caprate, ↑pantothenate, ↑β-hydroxybutyrate, ↑urea | [104] |
| HM factors | ||
| Lactational stage | ||
| Colostrum | ↑ HMOs, ↑ LNnT, ↑ 2′-FL, ↑ 3′-SL, ↓ Lactose, ↑ Leucine, ↑ Betaine, and ↑ Creatinine | [43,104,107] |
| Mature HM | ↓ Total HMOs, ↑ 3′-FL, ω6/ ω3 PUFA, ↑ oleic acid, ↑ palmitoleic acid, ↑ linoleic acid, ↑ tri-, di-, mono-glycerides, ↓ cholesterol, ↓ phospholipids, ↓ α-tocopherol, ↓ fucose, ↓ furanose isomers, ↓ D-glucosaminic acid, ↑ Alanine, ↑ caprylate, ↑ caprate, ↑ glutamate | [16,25,100,104,107,115,116] |
| Course of lactation | ||
| Foremilk | ↑ free amino acids, ↑ phenylalanine, ↑ threonine, ↑ valine, ↑ alanine, ↑ glutamine, ↑ serine | [109] |
| Delivery mode | ||
| Vaginal delivery | ↑ 3-hydroxybutyrate and ↑ LNFP III | [101] |
| CS delivery | ↑ Butyrate, ↑ urea, ↑ putrescine, ↑ ethanolamine and ↑ proline | [101] |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ojo-Okunola, A.; Cacciatore, S.; Nicol, M.P.; du Toit, E. The Determinants of the Human Milk Metabolome and Its Role in Infant Health. Metabolites 2020, 10, 77. https://doi.org/10.3390/metabo10020077
Ojo-Okunola A, Cacciatore S, Nicol MP, du Toit E. The Determinants of the Human Milk Metabolome and Its Role in Infant Health. Metabolites. 2020; 10(2):77. https://doi.org/10.3390/metabo10020077
Chicago/Turabian StyleOjo-Okunola, Anna, Stefano Cacciatore, Mark P. Nicol, and Elloise du Toit. 2020. "The Determinants of the Human Milk Metabolome and Its Role in Infant Health" Metabolites 10, no. 2: 77. https://doi.org/10.3390/metabo10020077
APA StyleOjo-Okunola, A., Cacciatore, S., Nicol, M. P., & du Toit, E. (2020). The Determinants of the Human Milk Metabolome and Its Role in Infant Health. Metabolites, 10(2), 77. https://doi.org/10.3390/metabo10020077






