The Bioassay-Guided Fractionation and Identification of Potent Acetylcholinesterase Inhibitors from Narcissus c.v. ‘Hawera’ Using Optimized Vacuum Liquid Chromatography, High Resolution Mass Spectrometry and Bioautography
Abstract
:1. Introduction
2. Results and Discussion
2.1. Extraction and Optimization of Fractionation by VLC for the Isolation of Alkaloids from Narcissus triandrus L. c.v. ‘Hawera’
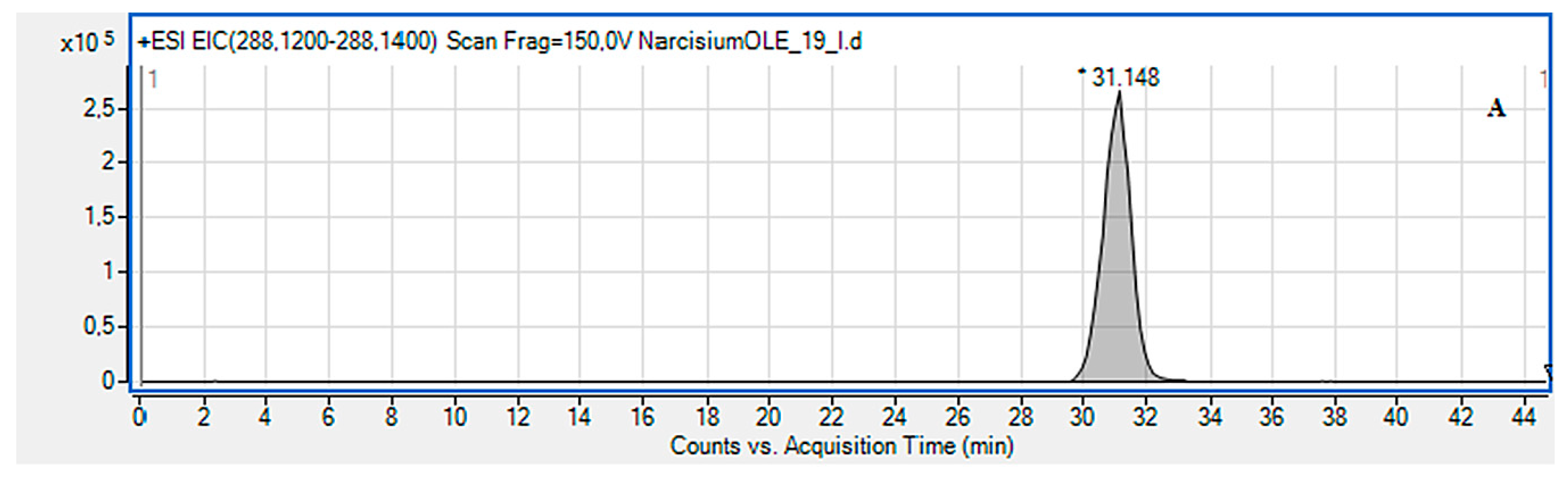
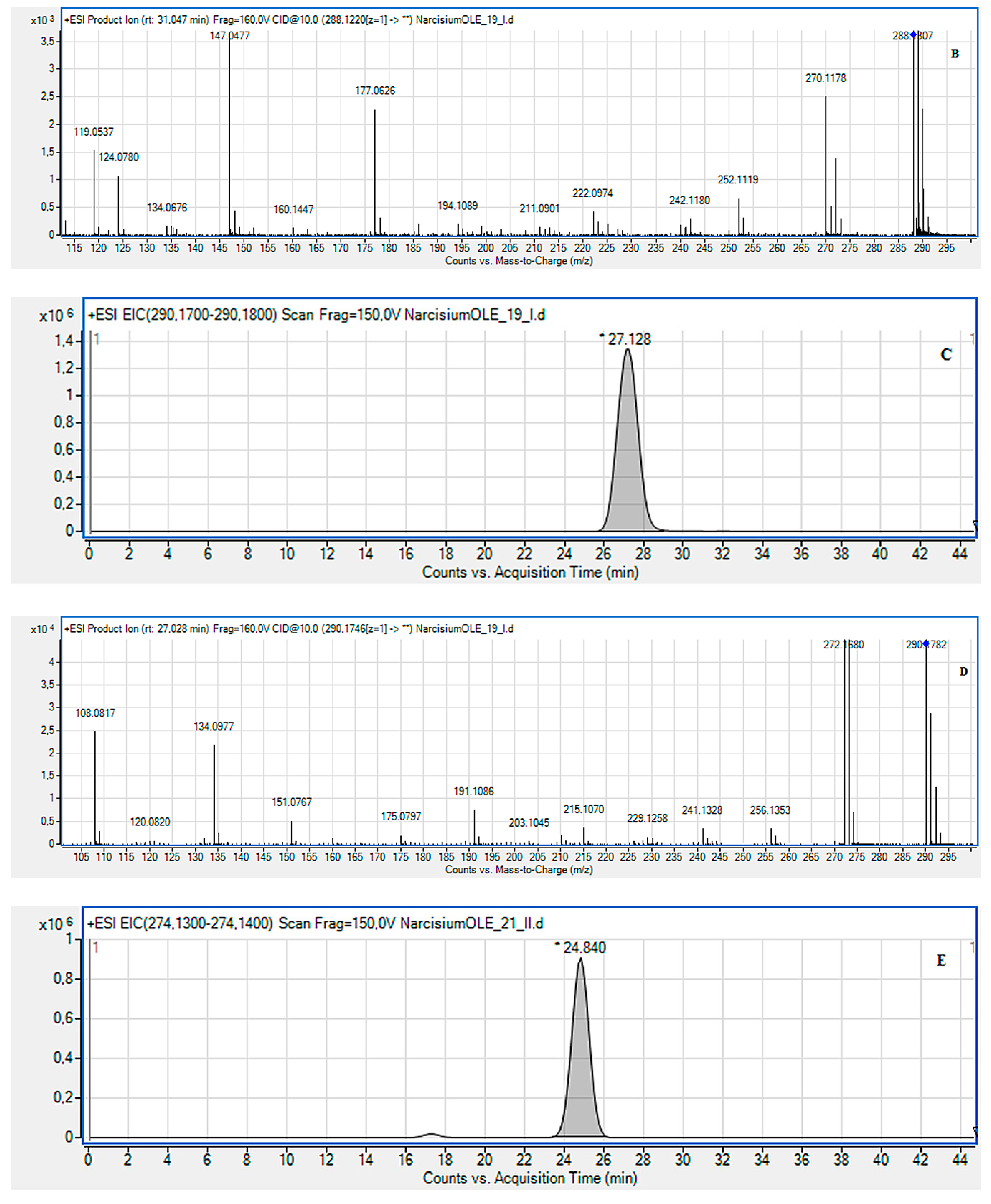
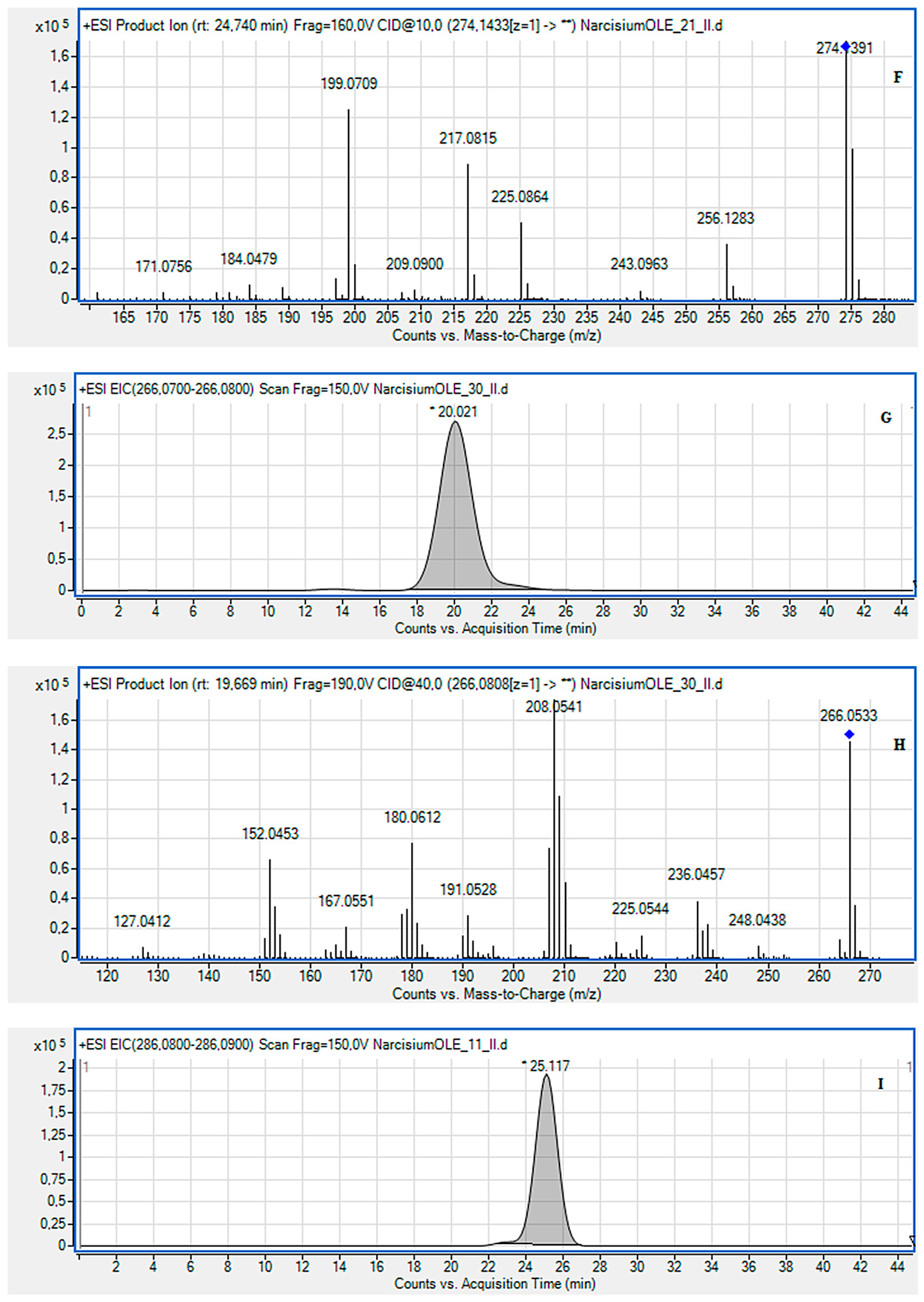
2.2. LC-MS Identification of the Isolated Compounds
2.3. TLC-Bioautography for the Detection of Potent AChE Inhibitors
3. Materials and Methods
3.1. Plant Material
3.2. Sample Preparation and Alkaloid Extraction
3.3. Method Optimization of VLC
3.4. LC-MS Identification of the Isolated Compounds
3.5. TLC with Bioautography of Anticholinesterase Activity
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Li, Y.; ShI, X.; Zang, J.; Zang, X.; Martin, R.C.G. Hepatic protection and anticancer activity of curcuma: A potential chemopreventive strategy against hepatocellular carcinoma. Int. J. Oncol. 2014, 44, 505–513. [Google Scholar] [CrossRef] [PubMed]
- Bräunlich, M.; Slimestad, R.; Wangensteen, H.; Brede, C.; Malterud, K.E.; Barsett, H. Extracts, Anthocyanins and Procyanidins from Aronia melanocarpa as Radical Scavengers and Enzyme Inhibitors. Nutrients 2013, 5, 663–678. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kasi, P.D.; Tamilselvan, R.; Skalicka-Woźniak, K.; Nabavi, S.F.; Daglia, M.; Bishayee, A.; Pazoki-Toroudi, H.; Nabavi, S.M. Molecular targets of curcumin for cancer therapy: An updated review. Tumor Biol. 2016, 37, 13017–13028. [Google Scholar] [CrossRef] [PubMed]
- Kukula-Koch, W.; Grabarska, A.; Łuszczki, J.; Czernicka, L.; Nowosadzka, E.; Gumbarewicz, E.; Jarząb, A.; Audo, G.; Upadhyay, S.; Głowniak, K.; et al. Superior anticancer activity is demonstrated by total extract of Curcuma longa L. as opposed to individual curcuminoid sseparated by centrifugal partition chromatography. Phytother. Res. 2018, 5, 933–942. [Google Scholar] [CrossRef] [Green Version]
- Oniszczuk, T.; Widelska, G.; Oniszczuk, A.; Kasprzak, K.; Wójtowicz, A.; Olech, M.; Nowak, R.; Wojtunik-Kulesza, K.; Jozwiak, G.; Waksmundzka-Hajnos, M. Influence of Production Parameters on the Content of Polyphnolic Compounds in Extruded Porridge Enriched with Chokeberry Fruit (Aronia melanocarpa (Michx.) Elliott). Open Chem. 2019, 17, 166–167. [Google Scholar] [CrossRef]
- Skalicka-Woźniak, K.; Orhan, I.E.; Cordell, G.A.; Nabavi, S.M.; Budzyńska, B. Implication of coumarins towards central nervous system disorders. Pharmacol. Res. 2016, 103, 188–203. [Google Scholar] [CrossRef]
- Chaves, S.K.M.; Feitosa, C.M.; da S. Araujo, L. Alkaloids Pharmacological Activities Prospects for the Development of Phytopharmaceuticals for Neurodegenerative Diseases. Curr. Pharm. Biotechnol. 2016, 17, 629–695. [Google Scholar] [CrossRef]
- Berkov, S.; Martinez-Frances, V.; Bastida, J.; Codina, C.; Rios, S. Evolution of alkaloid biosynthesis in the genus Narcissus. Phytochemistry 2014, 99, 95–106. [Google Scholar] [CrossRef]
- Lopez, S.; Bastida, J.; Viladomat, F.; Codina, C. Acetylcholinesterase inhibitory activity of some Amaryllidaceae alkaloids and Narcissus extracts. Life Sci. 2002, 71, 2521–2529. [Google Scholar] [CrossRef]
- Vergura, S.; Santoro, E.; Masi, M.; Evidente, A.; Scafato, P.; Superchi, S.; Mazzeo, G.; Longhi, G.; Abbate, S. Absolute configuration assignment of anticancer Amaryllidaceae alkaloid jonquailine. Fitoterapia 2018, 129, 78–84. [Google Scholar] [CrossRef]
- Zabłocka, A. Choroba Alzheimera jako przykład schorzenia neurodegeneracyjnego. Postepy Hig. Med. Dosw. 2006, 60, 209–216. [Google Scholar]
- Ghisalberti, E. Detection and Isolation of Bioactive Natural Products. In Bioactive Natural Products: Detection, Isolation, and Structural Determination, 2nd ed.; Colegate, S.M., Molyneux, R.J., Eds.; Taylor & Francis: Philadelphia, PA, USA, 2008; pp. 11–76. [Google Scholar]
- Maurya, A.; Kalani, K.; Verma, S.C.; Singh, R.; Srivastava, A. Vacuum Liquid Chromatography: Simple, Efficient and Versatile Separation Technique for Natural Products. Org. Med. Chem. IJ. 2018, 7, 1–3. [Google Scholar] [CrossRef]
- Targett, N.M.; Kilcoyne, J.P.; Green, B. Vacuum Liquid Chromatography: An Alternative to Common Chromatographic Methods. J. Org. Chem. 1979, 44, 4962–4964. [Google Scholar] [CrossRef]
- Upadhyay, H.C.; Saini, D.C.; Srivastava, S.K. Phytochemical Analysis of Ammannia multiflora. Res. J. Phytochem. 2011, 5, 170–176. [Google Scholar] [CrossRef]
- Bucar, F.; Wube, A.; Schmid, M. Natural products isolation–how to get from biological material to pure compounds. Nat. Prod. Rep. 2013, 30, 525–545. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Noviany, N.; Hadi, S. The Isolation of α-viniferin, a Timer Stilbene, from Shorea ovalis Blume. Adv. Nat. Appl. Sci. 2009, 3, 107–112. [Google Scholar] [CrossRef]
- Zarate, R.; Sukrasno; Yeoman, M.M. Application of two rapid techniques of column chromatography to separate the pungent principles of ginger, Zingiber officinale Roscoe. J. Chromatogr. A 1992, 609, 407–413. [Google Scholar] [CrossRef]
- Pelletier, S.W.; Chokshi, H.P.; Desai, H.K. Separation of diterpenoid alkaloid mixtures using vacuum liquid chromatography. J. Nat. Prod. 1986, 49, 892–900. [Google Scholar] [CrossRef]
- Pigni, N.B.; Berkov, S.; Elamrani, A.; Benaissa, M.; Viladomat, F.; Codina, C.; Bastida, J. Two New Alkaloids from Narcissus serotinus L. Molecules 2010, 15, 7083–7089. [Google Scholar] [CrossRef] [Green Version]
- Tallini, L.R.; Torras-Claveria, L.; de Souza Borges, W.; Kaiser, M.; Viladomat, F.; Zuanazzi, J.A.S.; Bastida, J. N-oxide alkaloids from Crinum amabile (Amaryllidaceae). Molecules 2018, 23, 1277. [Google Scholar] [CrossRef] [Green Version]
- Mroczek, T.; Mazurek, J. Pressurized liquid extraction and anticholinesterase activity-based this-layer-chromatography with bioautography of Amaryllidaceae alklaloids. Anal. Chim. Acta 2009, 633, 188–196. [Google Scholar] [CrossRef] [PubMed]
- Mroczek, T. Qualitative and quantitative two-dimensional thin-layerchromatography/high performance liquid chromatography/diode-array/electrospray-ionization-time-of-flight mass spectrometry of cholinesterase inhibitors. J. Pharm. Biomed. Anal. 2016, 129, 155–162. [Google Scholar] [CrossRef] [PubMed]
- Mroczek, T. Highly Efficient, Selective and Sensitive Molecular Screening of Acetylcholinesterase Inhibitors of Natural Origin by Solid-Phase Extraction-Liquid Chromatography/Electrospray Ionisation-Octopole-Orthogonal Acceleration Time-of-Flight-Mass Spectrometry and Novel Thin-Layer Chromatography-Based. Bioautography J. Chromatogr. A 2009, 1216, 2519–2528. [Google Scholar] [CrossRef] [PubMed]
- Marston, A. Thin-layer chromatography with biological detection in phytochemistry. J. Chromatogr. A 2011, 1218, 2676–2683. [Google Scholar] [CrossRef]
- Guillou, C.; Beunard, J.L.; Gras, E.; Thal, C. An Efficient Total Synthesis of (±) galanthamine, Angew. Chem. Int. Ed. 2001, 40, 4745–4746. [Google Scholar] [CrossRef]
- Heinrich, M.; Teoh, H.L. Galanthamine from snowdrop–the development of a modern drug against Alzheimer’s disease from local Caucasian knowledge. J. Ethnopharmacol. 2004, 92, 147–162. [Google Scholar] [CrossRef]
- Russo, P.; Frustaci, A.; Fini, M.; Cesario, A. From Traditional European Medicine to Discovery of New Drug Candidates for the Treatment of Dementia and Alzheimer’s Disease: Acetylcholinesterase Inhibitors. Curr. Med. Chem. 2013, 20, 976–983. [Google Scholar] [CrossRef]
- Coyle, J.; Kershaw, P. Galantamine, a cholinesterase inhibitor that allosterically modulates nicotinic receptors: Effect on the course of Alzheimer’s disease. Biol. Psychiat. 2001, 49, 289–299. [Google Scholar] [CrossRef]
- Sticher, O. Natural products isolation. Nat. Prod. Rep. 2008, 7, 517–554. [Google Scholar] [CrossRef]
- Bores, G.M.; Huger, F.P.; Petko, W.; Mutlib, A.E.; Camacho, F.; Rush, D.K.; Selk, D.E.; Wolf, V.; Kosley, R.W.; Davis, L.; et al. Pharmacological evaluation of novel Alzheimer’s disease therapeutics: Acetylcholinesterase inhibitors related to galanthamine. J. Pharmacol. Exp. Ther. 1996, 277, 728–738. [Google Scholar]


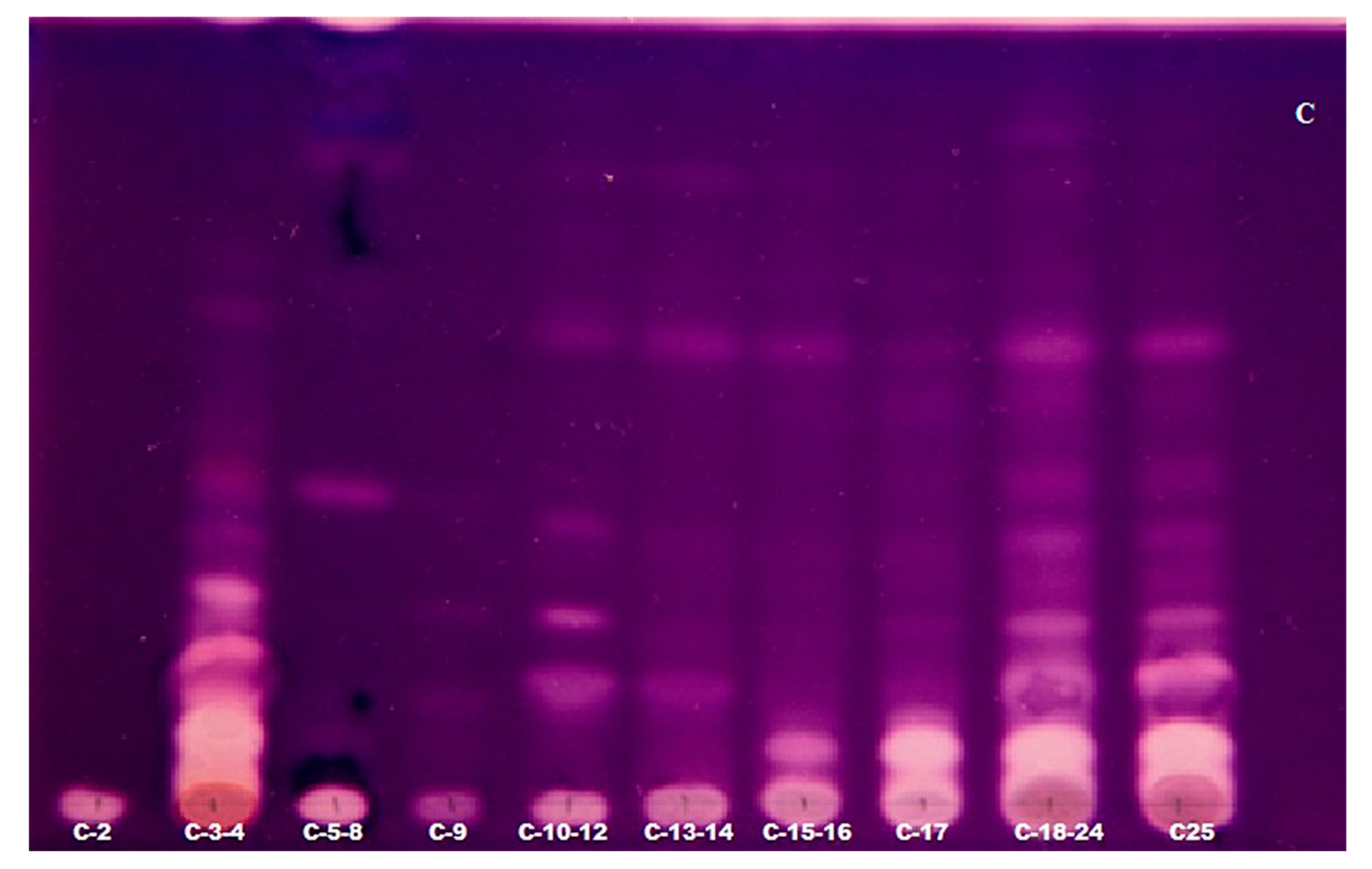
| The Number of Experiments | 1-A | 2-B | 3-C |
|---|---|---|---|
| Type of column | glass | polypropylene cartridge | polypropylene cartridge |
| Sorbent filling ratio (Al2O3(150 MeSh): silica gel (60 F254)) | 1:1 Al2O3(25 g): silica gel (57 g) | 1:3 Al2O3(17 g): silica gel (25 g) | 3:1 Al2O3(57 g): silica gel (8 g) |
| Fractions obtained | A-19 A-20-22 A-23-24 A-25 A-26 A-27-33 A-34 | B-3 B-4-7 B-8-9 B-10 B-11 B-12 B-13-14 B-15-17 B-18-20 B-21 B-22-25 B-26 B-27-29 B-30 B-31 | C-2 C-3-4 C-5-8 C-9 C-10-12 C-13-14 C-15-16 C-17 |
| % of Isolated Compounds to the Total Amount of All Alkaloid Compounds Obtained from Individual Fractions [%] | Fraction Number | ||||||
|---|---|---|---|---|---|---|---|
| A-19 | A-20-22 | A-23-24 | A-25 | A-26 | A-27-33 | A-34 | |
| Sanguinine | 1.2 | ||||||
| Lycoramine | 54.3 | 9.9 | 27.5 | 16.4 | 5.6 | ||
| Lycorine | 7.2 | 52.9 | 41.2 | 23.6 | 17.8 | 10.0 | 10.0 |
| Ungeremine | 9.9 | 16.0 | 16.8 | 18.6 | 22.6 | 14.6 | |
| 4,N-didehydro-nor-augustamine | 17.7 | 15.5 | 7.2 | 15.9 | 18.1 | 11.9 | |
| Tetrahydro-nor-augustamine | 4.7 | 10.7 | 8.8 | 12.5 | 21.4 | 16.6 | |
| Mesembrinole | 6.2 | 7.1 | 24.6 | ||||
| Galanthamine | 1.4 | 3.3 | |||||
| % of Isolated Compounds to the Total Amount of All Alkaloid Compounds Obtained from Individual Fractions [%] | Fraction Number | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| B-10 | B-11 | B-12 | B-13-14 | B-15-17 | B-18-20 | B-21 | B-22-25 | B-26 | B-27-29 | B-30 | B-31 | |
| Sanguinine | 3.4 | 9.2 | 9.1 | 5.8 | 7.5 | 1.4 | 2.9 | |||||
| Lycoramine | 39.0 | 38.1 | 36.9 | |||||||||
| Lycorine | 11.6 | 26.6 | 29.2 | 22.6 | 17.1 | 21.2 | 22.2 | 22.8 | 26.1 | 9.1 | ||
| Ungeremine | 7.7 | 4.5 | 5.8 | 9.2 | 7.6 | 7.2 | 6.3 | 11.5 | 9.2 | 24.9 | 26.1 | |
| 4,N-didehydro-nor-augustamine | 10.8 | 4.7 | 6.6 | 6.0 | 3.6 | 5.9 | 4.0 | 8.1 | ||||
| Tetrahydro-nor-augustamine | 5.8 | 3.4 | 4.3 | 4.7 | 0.8 | 0.8 | ||||||
| Mesembrinole | 27.4 | 15.6 | 12.1 | 9.0 | 4.6 | 9.7 | ||||||
| Haemanthamine | 3.5 | |||||||||||
| Lycorine-N-oxide | 0.7 | |||||||||||
| Galanthamine-N-oxide | 2.1 | 1.6 | ||||||||||
| Galanthamine | 1.9 | 1.7 | ||||||||||
| Tazettine | 3.4 | 8.3 | 9.9 | 5.7 | ||||||||
| % of Isolated Compounds to the Total Amount of All Alkaloid Compounds Obtained from Individual Fractions [%] | Fraction Number | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| C-2 | C-3-4 | C-5-8 | C-9 | C-10-12 | C-13-14 | C-15-16 | C-17 | C-18-24 | C-25 | |
| Sanguinine | 1.7 | 8.7 | ||||||||
| Lycoramine | 7.5 | 26.5 | 53.5 | |||||||
| Lycorine | 23.8 | 23.3 | 2.4 | 20.2 | 25.4 | 22.1 | 15.6 | 30.1 | 7.8 | |
| Ungeremine | 4.2 | 8.2 | 6.7 | 4.1 | 4.1 | 10.3 | 11.2 | 7.8 | 2.0 | 10.0 |
| 4,N-didehydro-nor-augustamine | 4.2 | 9.7 | 12.9 | 4.3 | ||||||
| Tetrahydro-nor-augustamine | 5.5 | 2.2 | 3.3 | 2.8 | 1.3 | 9.3 | 3.7 | |||
| Mesembrinole | 6.7 | 13.4 | 36.2 | 10.3 | 4.3 | |||||
| Galanthamine | 5.9 | |||||||||
| Lycoramine-N-oxide | 2.1 | 2.4 | ||||||||
| Haemanthamine | 12.2 | 6.9 | 3.4 | 2.6 | ||||||
| Lycorine-N-oxide | 2.8 | |||||||||
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mroczek, T.; Dymek, A.; Widelski, J.; Wojtanowski, K.K. The Bioassay-Guided Fractionation and Identification of Potent Acetylcholinesterase Inhibitors from Narcissus c.v. ‘Hawera’ Using Optimized Vacuum Liquid Chromatography, High Resolution Mass Spectrometry and Bioautography. Metabolites 2020, 10, 395. https://doi.org/10.3390/metabo10100395
Mroczek T, Dymek A, Widelski J, Wojtanowski KK. The Bioassay-Guided Fractionation and Identification of Potent Acetylcholinesterase Inhibitors from Narcissus c.v. ‘Hawera’ Using Optimized Vacuum Liquid Chromatography, High Resolution Mass Spectrometry and Bioautography. Metabolites. 2020; 10(10):395. https://doi.org/10.3390/metabo10100395
Chicago/Turabian StyleMroczek, Tomasz, Aleksandra Dymek, Jarosław Widelski, and Krzysztof Kamil Wojtanowski. 2020. "The Bioassay-Guided Fractionation and Identification of Potent Acetylcholinesterase Inhibitors from Narcissus c.v. ‘Hawera’ Using Optimized Vacuum Liquid Chromatography, High Resolution Mass Spectrometry and Bioautography" Metabolites 10, no. 10: 395. https://doi.org/10.3390/metabo10100395







