Modification of Taxifolin Properties by Spray Drying
Abstract
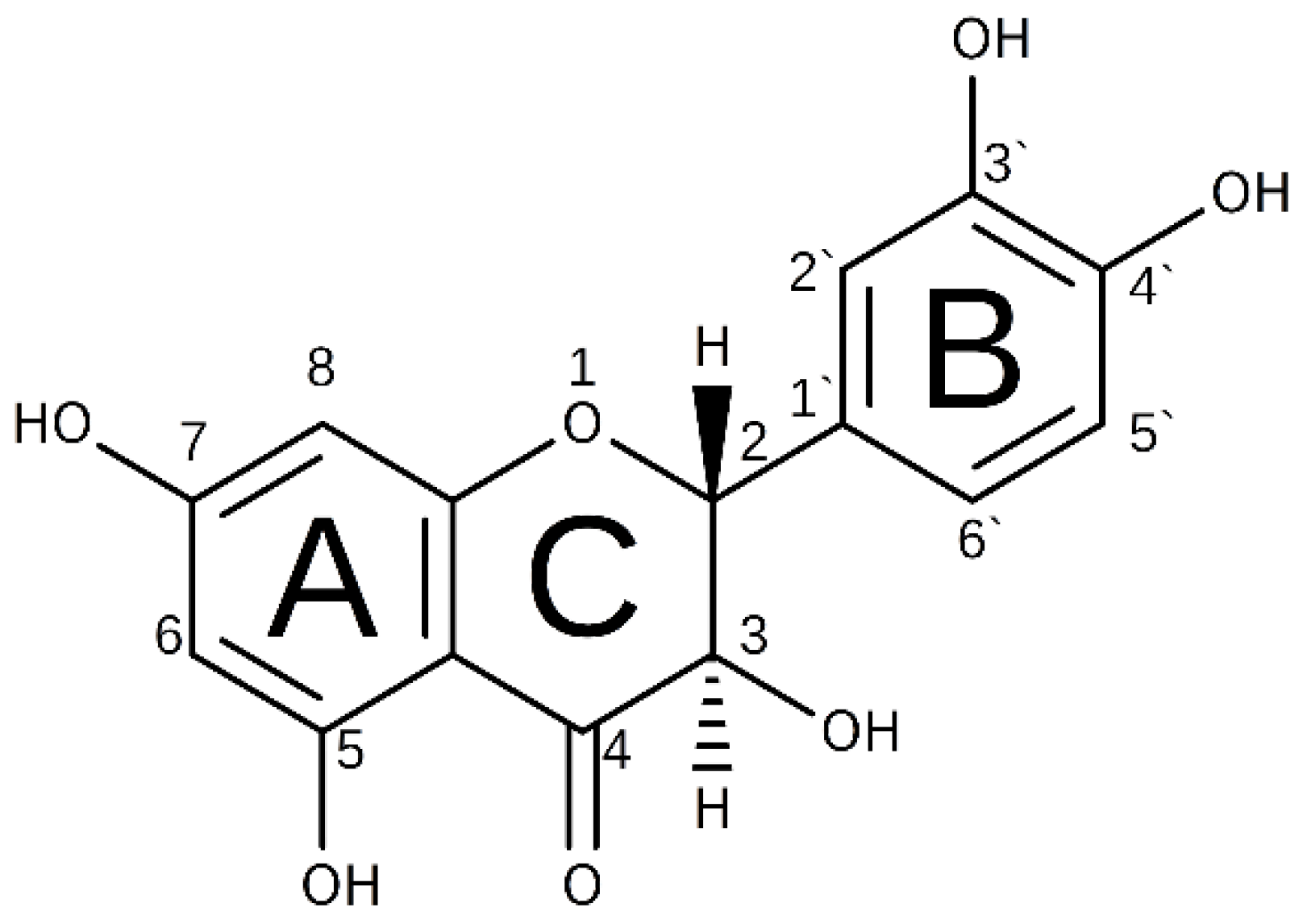
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Spray Drying of Taxifolin
2.3. Tablet Preparation
2.4. Cell Culture
2.5. Antimicrobial Activity
2.6. Fourier Transform Infrared Spectroscopy (FTIR)
2.7. Solubility
2.8. Cell Viability Test
2.9. Permeability Assay
2.10. Flowability
2.11. Disintegration of Tablets
2.12. Tablet Hardness
2.13. Friability Test
2.14. Dissolution Test
2.15. Statistical Analysis
3. Results
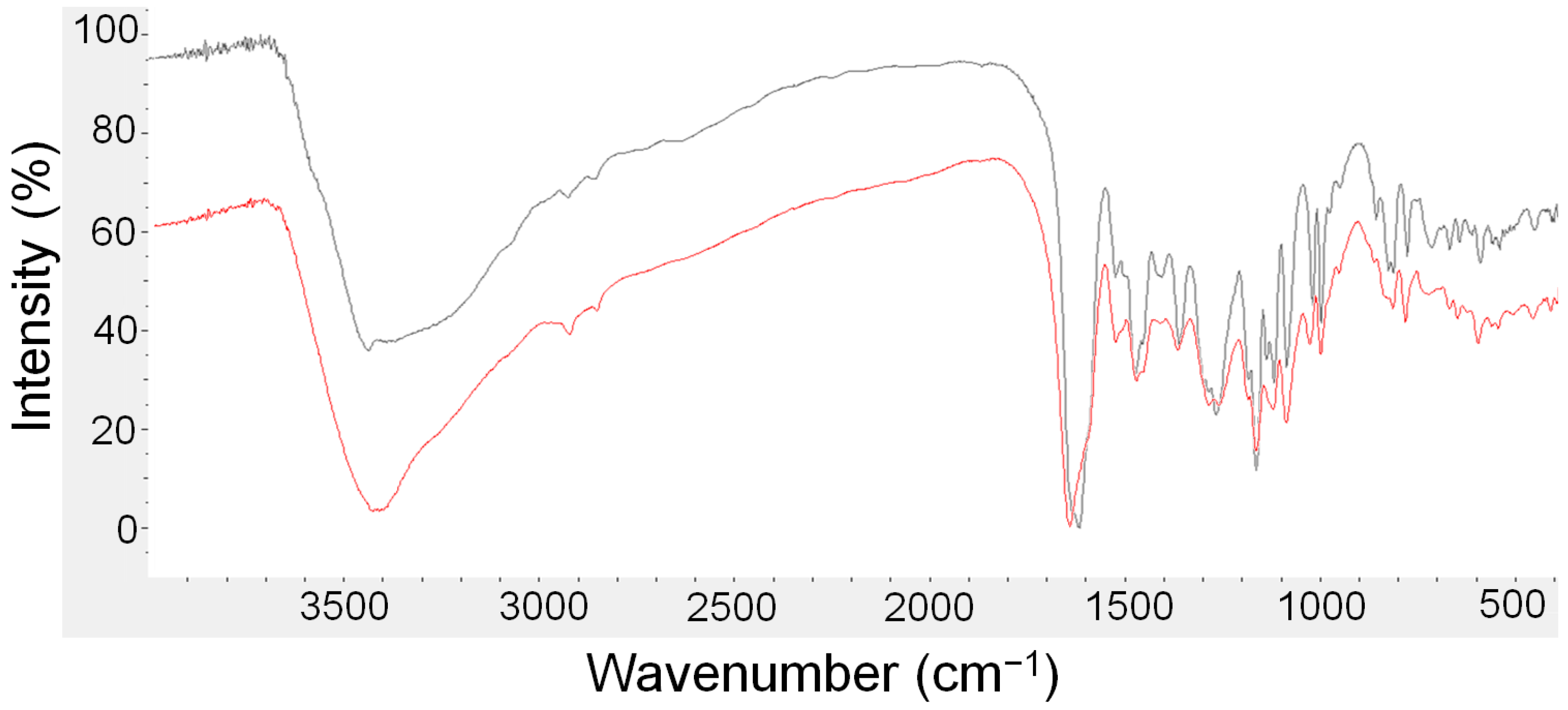
3.1. FTIR Spectroscopy
3.2. Solubility
3.3. Antimicrobial Activity
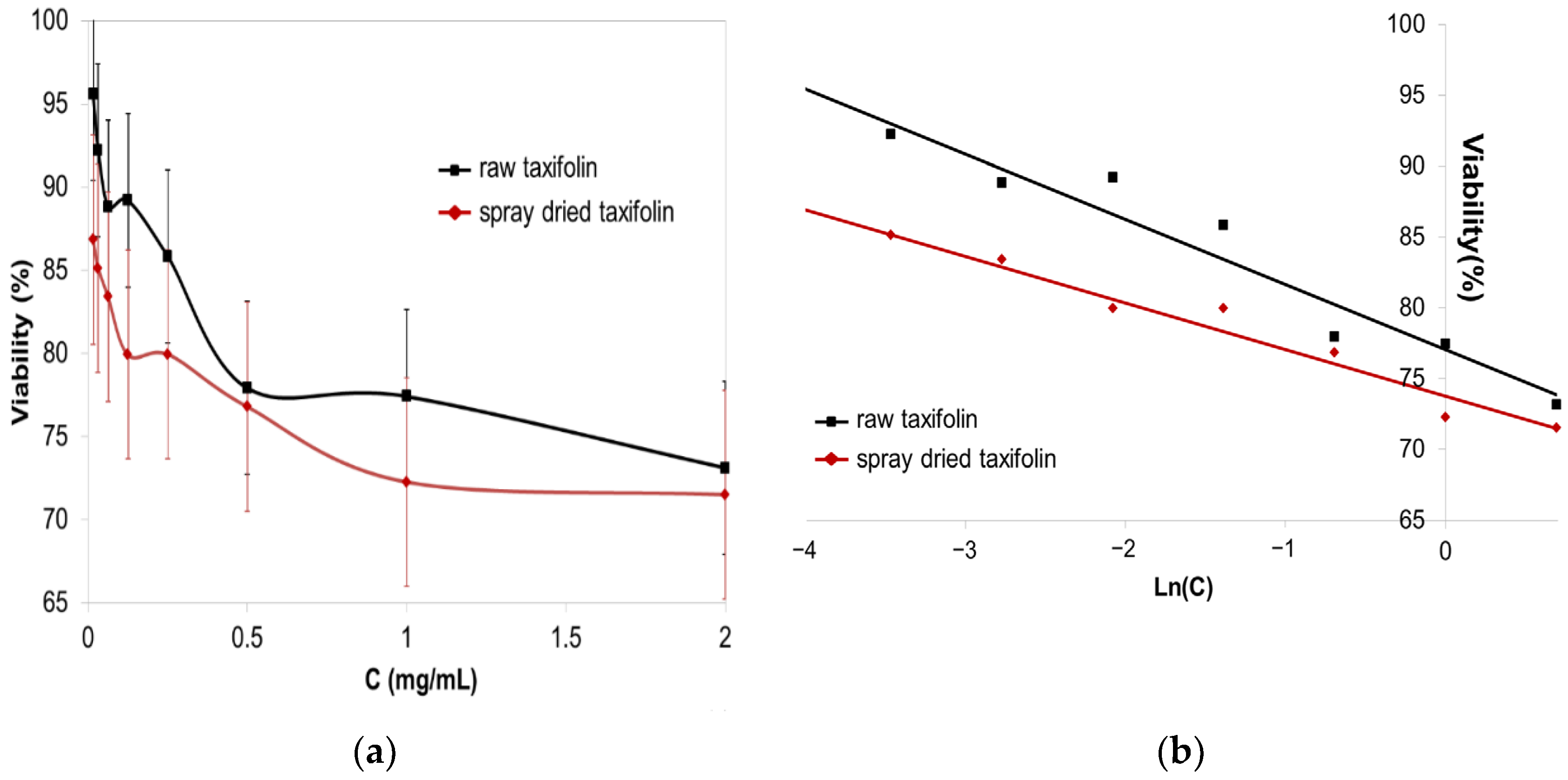
3.4. Cell Viability Test
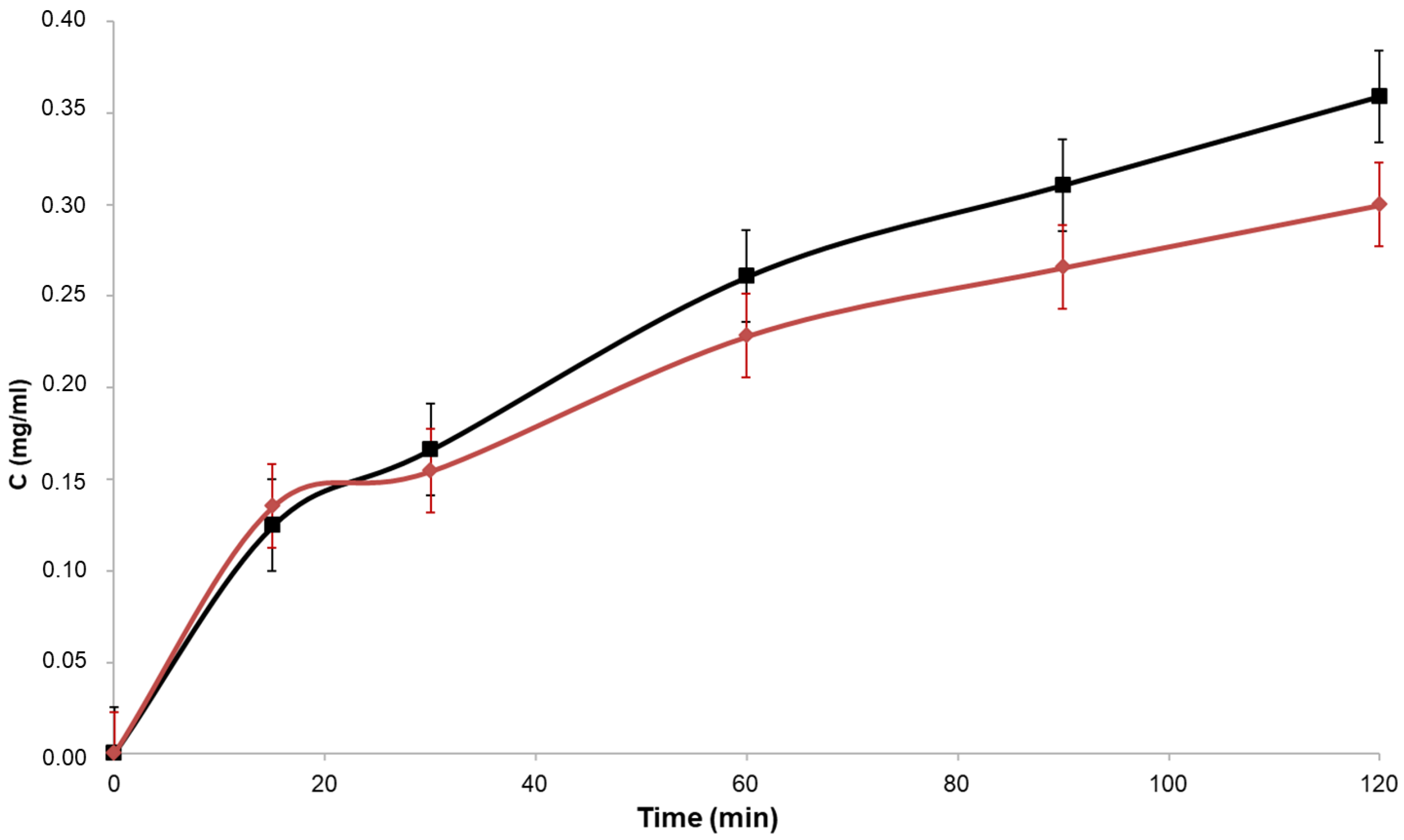
3.5. Permeability Assay
3.6. Flowability
3.7. Design of Orally Dispersed Tablets with TS
3.8. Characteristics of Tablets
3.9. Dissolution Test
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Deng, M.; Dong, L.; Jia, X.; Huang, F.; Chi, J.; Muhammad, Z.; Ma, Q.; Zhao, D.; Zhang, M.; Zhang, R. The flavonoid profiles in the pulp of different pomelo (Citrus grandis L. Osbeck) and grapefruit (Citrus paradise Mcfad) cultivars and their in vitro bioactivity. Food Chem. X 2022, 15, 100368. [Google Scholar] [CrossRef] [PubMed]
- Zhao, C.; Wang, F.; Lian, Y.; Xiao, H.; Zheng, J. Biosynthesis of citrus flavonoids and their health effects. Crit. Rev. Food SciNutr. 2020, 60, 566–583. [Google Scholar] [CrossRef]
- Wang, S.; Yang, C.; Tu, H.; Zhou, J.; Liu, X.; Cheng, Y.; Luo, J.; Deng, X.; Zhang, H.; Xu, J. Characterization and Metabolic Diversity of Flavonoids in Citrus Species. Sci. Rep. 2017, 7, 10549. [Google Scholar] [CrossRef] [PubMed]
- Chen, X.; Wang, P.; Zheng, Y.; Gu, M.; Lin, X.; Wang, S.; Jin, S.; Ye, N. Comparison of Metabolome and Transcriptome of Flavonoid Biosynthesis Pathway in a Purple-Leaf Tea Germplasm Jinmingzaoand a Green-Leaf Tea Germplasm Huangdanreveals Their Relationship with Genetic Mechanisms of Color Formation. Int. J. Mol. Sci. 2020, 21, 4167. [Google Scholar] [CrossRef] [PubMed]
- Zheng, C.; Ma, J.-Q.; Ma, C.-L.; Shen, S.-Y.; Liu, Y.-F.; Chen, L. Regulation of Growth and Flavonoid Formation of Tea Plants (Camellia sinensis) by Blue and Green Light. J. Agric. Food Chem. 2019, 67, 2408–2419. [Google Scholar] [CrossRef] [PubMed]
- Lu, S.; Wang, J.; Zhuge, Y.; Zhang, M.; Liu, C.; Jia, H.; Fang, J. Integrative Analyses of Metabolomes and Transcriptomes Provide Insights into Flavonoid Variation in Grape Berries. J. Agric. Food Chem. 2021, 69, 12354–12367. [Google Scholar] [CrossRef]
- Liu, J.; Hefni, M.E.; Witthöft, C.M. Characterization of Flavonoid Compounds in Common Swedish Berry Species. Food 2020, 9, 358. [Google Scholar] [CrossRef]
- Cui, Q.; Liu, J.-Z.; Wang, L.-T.; Kang, Y.-F.; Meng, Y.; Jiao, J.; Fu, Y.-J. Sustainable deep eutectic solvents preparation and their efficiency in extraction and enrichment of main bioactive flavonoids from sea buckthorn leaves. J. Clean. Prod. 2018, 184, 826–835. [Google Scholar] [CrossRef]
- Chen, C.; Zhang, H.; Xiao, W.; Yong, Z.-P.; Bai, N. High-performance liquid chromatographic fingerprint analysis for different origins of sea buckthorn berries. J. Chromatogr. A 2007, 1154, 250–259. [Google Scholar] [CrossRef]
- Bagdonaitė, E.; Mártonfi, P.; Repčák, M.; Labokas, J. Variation in concentrations of major bioactive compounds in Hypericum perforatum L. from Lithuania. Ind. Crops Prod. 2012, 35, 302–308. [Google Scholar] [CrossRef]
- Smelcerovic, A.; Spiteler, M.; Zuehlke, S. Comparison of Methods for the Exhaustive Extraction of Hypericins, Flavonoids, and Hyperforin from Hypericum perforatum L. J. Agric. Food Chem. 2006, 54, 2750–2753. [Google Scholar] [CrossRef] [PubMed]
- Gou, K.-J.; Zeng, R.; Ma, Y.; Li, A.-N.; Yang, K.; Yan, H.-X.; Jin, S.-R.; Qu, Y. Traditional uses, phytochemistry, and pharmacology of Persicaria orientalis (L.) Spach—A review. J. Ethnopharmacol. 2020, 249, 112407. [Google Scholar] [CrossRef] [PubMed]
- Kurkina, A.V.; Ryazanova, T.K.; Kurkin, V.A. Flavonoids from the Aerial Part of Polygonum persicaria. Chem. Nat. Compd. 2013, 49, 845–847. [Google Scholar] [CrossRef]
- Sharma, A.; Shahzad, B.; Rehman, A.; Bhardwaj, R.; Landi, M.; Zheng, B. Response of Phenylpropanoid Pathway and the Role of Polyphenols in Plants under Abiotic Stress. Molecules 2019, 24, 2452. [Google Scholar] [CrossRef]
- Sathishkumar, P.; Gu, F.L.; Zhan, Q.; Palvannan, T.; MohdYusoff, A.R. Flavonoids mediated ‘Green’ nanomaterials: A novel nanomedicine system to treat various diseases—Current trends and future perspective. Mater. Lett. 2018, 210, 26–30. [Google Scholar] [CrossRef]
- Yoon, K.D.; Lee, J.Y.; Kim, T.Y.; Kang, H.; Ha, K.S.; Ham, T.H.; Ryu, S.N.; Kang, M.Y.; Kim, Y.H.; Kwon, Y.I. In Vitro and in Vivo Anti-Hyperglycemic Activities of Taxifolin and Its Derivatives Isolated from Pigmented Rice (Oryzae sativa L. cv. Superhongmi). J. Agric. Food Chem. 2020, 68, 742–750. [Google Scholar] [CrossRef]
- Kim, J.W.; Im, S.; Jeong, H.R.; Jung, Y.S.; Lee, I.; Kim, K.J.; Park, S.K.; Kim, D.O. Neuroprotective effects of korean red pine (Pinus densiflora) bark extract and its phenolics. J. Microbiol. Biotechnol. 2018, 28, 679–687. [Google Scholar] [CrossRef]
- Xia, Y.; Wang, Y.; Li, W.; Ma, C.; Liu, S. Homogenization-assisted cavitation hybrid rotation extraction and macroporous resin enrichment of dihydroquercetin from Larixgmelinii. J. Chromatogr. B 2017, 1070, 62–69. [Google Scholar] [CrossRef]
- Ilyasov, I.R.; Beloborodov, V.L.; Selivanova, I.A. Three ABTS•+ radical cation-based approaches for the evaluation of antioxidant activity: Fast- and slow-reacting antioxidant behavior. Chem. Paper 2018, 72, 1917–1925. [Google Scholar] [CrossRef]
- Ilyasov, I.; Beloborodov, V.; Antonov, D.; Dubrovskaya, A.; Terekhov, R.; Zhevlakova, A.; Saydasheva, A.; Evteev, V.; Selivanova, I. Flavonoids with Glutathione Antioxidant Synergy: Influence of Free Radicals Inflow. Antioxidants 2020, 9, 695. [Google Scholar] [CrossRef]
- Schauss, A.G.; Tselyico, S.S.; Kuznetsova, V.A.; Yegorova, I. Toxicological and genotoxicity assessment of a dihydroquercetin-rich Dahurian larch tree (Larix gmelinii Rupr) extract (Lavitol). Int. J. Toxicol. 2015, 34, 162–181. [Google Scholar] [CrossRef] [PubMed]
- Taldaev, A.; Terekhov, R.; Nikitin, I.; Zhevlakova, A.; Selivanova, I. Insights into the Pharmacological Effects of Flavonoids: The Systematic Review of Computer Modeling. Int. J. Mol. Sci. 2022, 23, 6023. [Google Scholar] [CrossRef] [PubMed]
- Fischer, A.; Sellner, M.; Neranjian, S.; Smieško, M.; Lill, M.A. Potential Inhibitors for Novel Coronavirus Protease Identified by Virtual Screening of 606 Million Compounds. Int. J. Mol. Sci. 2020, 21, 3626. [Google Scholar] [CrossRef] [PubMed]
- Raj, U.; Varadwaj, P.K. Flavonoids as multi-target inhibitors for proteins associated with Ebola virus: In-silico discovery using virtual screening and molecular docking studies. Interdiscip. Sci. 2016, 8, 132–141. [Google Scholar] [CrossRef]
- Oi, N.; Chen, H.; Kim, M.O.; Kim, M.O.; Lubert, R.A.; Bode, A.M.; Dong, Z. Taxifolin Suppresses UV-Induced Skin Carcinogenesis by Targeting EGFR and PI3K. Cancer Prev. Res. 2012, 5, 1103–1114. [Google Scholar] [CrossRef]
- Dias, J.L.; Lenza, M.; Ferreira, R.S. Cocrystallization: A tool to modulate physicochemical and biological properties of food-relevant polyphenols. Trends Food Sci. Technol. 2021, 110, 13–27. [Google Scholar] [CrossRef]
- Selivanova, I.A.; Terekhov, R.P. Crystal engineering as a scientific basis for modification of physicochemical properties of bioflavonoids. Russ. Chem. Bull. 2019, 68, 2155–2162. [Google Scholar] [CrossRef]
- Li, W.; Pi, J.; Zhang, Y.; Ma, X.; Zhang, B.; Wang, S.; Qi, D.; Li, N.; Guo, P.; Liu, Z. A strategy to improve the oral availability of baicalein: The baicalein-theophylline cocrystal. Fitoterapia 2018, 129, 85–93. [Google Scholar] [CrossRef]
- Terekhov, R.; Selivanova, I. Fractal Aggregation of Dihydroquercetin After Lyophilization. J. Pharm. Innov. 2018, 13, 313–320. [Google Scholar] [CrossRef]
- Liu, H.; Lin, H.; Zhou, Z.; Li, L. Bergenin-isonicotinamide (1:1) cocrystal with enhanced solubility and investigation of its solubility behaviour. J. Drug Deliv. Sci. Tech. 2021, 64, 102556. [Google Scholar] [CrossRef]
- Terekhov, R.P.; Selivanova, I.A.; Tyukavkina, N.A.; Shylov, G.V.; Utenishev, A.N.; Porozov, Y.B. Taxifolin tubes: Crystal engineering and characteristics. Acta Crystallogr. B 2019, 75, 175–182. [Google Scholar] [CrossRef] [PubMed]
- Patil, S.; Chaudhari, K.; Kamble, R. Electrospray technique for cocrystallization of phytomolecules. J. King Saud. Univ. Sci. 2018, 30, 138–141. [Google Scholar] [CrossRef]
- An, H.; Lee, Y.; Liu, L.; Lee, S.; Lee, J.; Yi, Y. Physical and Chemical Stability of Formulations Loaded with Taxifolin Tetra-octanoate. Chem. Pharm. Bull. 2019, 67, 985–991. [Google Scholar] [CrossRef]
- Zu, Y.; Wu, W.; Zhao, X.; Li, Y.; Wang, W.; Zhong, C.; Zhang, Y. The high water solubility of inclusion complex of taxifolin-γ-CD prepared and characterized by the emulsion solvent evaporation and the freeze drying combination method. Int. J. Parm. 2014, 477, 148–158. [Google Scholar] [CrossRef]
- Shikov, A.N.; Pozharitskaya, O.N.; Miroshnyk, I.; Mirza, S.; Hirsjärvi, S.; Makarov, V.G.; Heinämäki, J.; Yliruusi, J.; Hiltunen, R. Nanodispersions of taxifolin: Impact of solid-state properties on dissolution behavior. Int. J. Pharm. 2009, 377, 148–152. [Google Scholar] [CrossRef] [PubMed]
- Hasabi, F.; Nasipour, A.; Varshosaz, J.; Gacia-Manrique, P.; Blanco-López, M.C.; Gutiérrez, G.; Matos, M. Formulation and Characterization of Taxifolin-Loaded Lipid Nanovesicles (Liposomes, Niosomes, and Transfersomes) for Beverage Fortification. Eur. J. Lipid Sci. Technol. 2020, 122, 1900105. [Google Scholar] [CrossRef]
- Shubina, V.S.; Shatalin, Y.V. Skin Regeneration after Chemical Burn under the Effect of Taxifolin-Based Preparations. Bull. Exp. Biol. Med. 2012, 154, 152–157. [Google Scholar] [CrossRef]
- Guo, Y.; Wang, C.; Dun, J.; Du, L.; Hawley, M.; Sun, C.C. Mechanism for the Reduced Dissolution of Ritonavir Tablets by Sodium Lauryl Sulfate. J. Pharm. Sci. 2019, 108, 516–524. [Google Scholar] [CrossRef]
- Sayatan, C.; Chadan, B.; Zheng, J.; Sun, C.C. Effect of Heating Rate and Kinetic Model Selection on Activation Energy of Nonisothermal Crystallization of Amorphous Felodipine. J. Pharm. Sci. 2014, 103, 3950–3957. [Google Scholar] [CrossRef]
- Poozesh, S.; Bilgili, E. Scale-up of pharmaceutical spray drying using scale-up rules: A review. Int. J. Pharm. 2019, 562, 271–292. [Google Scholar] [CrossRef]
- Ziaee, A.; Albadarin, A.B.; Padrela, L.; Femmer, T.; O’Reily, E.; Walker, G. Spray drying of pharmaceuticals and biopharmaceuticals: Critical parameters and experimental process optimization approaches. Eur. J. Phar. Sci. 2019, 127, 300–318. [Google Scholar] [CrossRef] [PubMed]
- Celik, M.; Wendell, S.C. Spray Drying and Pharmaceutical Applications. In Handbook of Pharmaceutical Granulation Technology; Parikh, D.M., Ed.; CRC Press: Boca Raton, FL, USA, 2016. [Google Scholar]
- Singh, A.; den Mooter, G.V. Spray drying formulation of amorphous solid dispersions. Adv. Drug Deliv. Rev. 2015, 100, 27–50. [Google Scholar] [CrossRef] [PubMed]
- Miao, X.; Zhou, J.; Liao, Y.; Zheng, Y. Chinese Medicine in Inhalation Therapy: A Review of Clinical Application and Formulation Development. Curr. Parm. Des. 2015, 21, 3917–3931. [Google Scholar] [CrossRef] [PubMed]
- Gharsallaoui, A.; Roudaut, G.; Chambin, O.; Voilley, A.; Saurel, R. Applications of spray-drying in microencapsulation of food ingredients: An overview. Food Res. Int. 2007, 40, 1107–1121. [Google Scholar] [CrossRef]
- Zhao, Y.; Chang, Y.-X.; Hu, X.; Liu, C.-Y.; Quan, L.-H.; Liao, Y.-H. Solid lipid nanoparticles for sustained pulmonary delivery of Yuxingcao essential oil: Preparation, characterization and in vivo evaluation. Int. J. Pharm. 2017, 516, 364–371. [Google Scholar] [CrossRef]
- Vehring, R. Pharmaceutical Particle Engineering via Spray Drying. Pharm. Res. 2008, 25, 999–1022. [Google Scholar] [CrossRef]
- Lauro, M.R.; Maggi, L.; Conte, U.; De Simone, F.; Aquino, R.P. Rutin and quercetin gastro-resistant microparticles obtained by spray-drying technique. J. Drug Deliv. Sci. Technol. 2005, 15, 363–369. [Google Scholar] [CrossRef]
- Sansone, F.; Rossi, A.; Del Gaudio, P.; De Simone, F.; Aquino, R.P.; Lauro, M.R. Hesperidin Gastroresistant Microparticles by Spray-Drying: Preparation, Characterization, and Dissolution Profiles. AAPS ParmSciTech 2009, 10, 391–401. [Google Scholar] [CrossRef]
- Sansone, F.; Picerno, P.; Mencherini, T.; Villecco, F.; D’Ursi, A.M.D.; Aquino, R.P.; Lauro, M.R. Flavonoid microparticles by spray-drying: Influence of enhancers of the dissolution rate on properties and stability. J. Food Eng. 2011, 103, 188–196. [Google Scholar] [CrossRef]
- Lauro, M.R.; De Simone, F.; Sansaone, F.; Iannelli, P.; Aquino, R.P. Preparations and release characteristics of naringin and naringenin gastro-resistant microparticles by spray-drying. J. Drug Deliv. Sci. Technol. 2007, 17, 119–124. [Google Scholar] [CrossRef]
- Terekhov, R.P.; Selivanova, I.A.; Tyukavkina, N.A.; Ilyasov, I.R.; Zhevlakova, A.K.; Dzuban, A.V.; Bogdanov, A.G.; Davidovich, G.N.; Shylov, G.V.; Utenishev, A.N.; et al. Assembling the Puzzle of Taxifolin Polymorphism. Molecules 2020, 25, 5437. [Google Scholar] [CrossRef] [PubMed]
- Terekhov, R.P.; Selivanova, I.A.; Anurova, M.N.; Zhevlakova, A.K.; Nikitin, I.D.; Cong, Z.; Ma, S.; Yang, F.; Dong, Z.; Liao, Y. Comparative Study of Wound-Healing Activity of Dihydroquercetin Pseudopolymorphic Modifications. Bull. Exp. Biol. Med. 2021, 170, 444–447. [Google Scholar] [CrossRef] [PubMed]
- Mizina, P.G.; Gulenkov, A.S. The orally disintegrating tablets: The achievements and prospects (review). Probl. Bio. Med. Pharm. Chem. 2018, 21, 3–11. (In Russian) [Google Scholar] [CrossRef]
- Ku, M.S. Use of the biopharmaceutical classification system in early drug development. AAPS J. 2008, 10, 208–212. [Google Scholar] [CrossRef]
- Zuikina, Y.; Polovko, N.; Strilets, O.; Strelnikov, L. The in vitro release testing and the antimicrobial activity of semi-solid dosage forms which contain salicylic acid. Farmacia 2021, 69, 1073–1079. [Google Scholar] [CrossRef]
- Thomson, K.S.; Thomson, G.K.; Biehle, J.; Deeb, A.; Crawford, J.; Herrera, R. A novel Topical Combination Ointment with Antimicrobial Activity against Methicillin-Resistant Streptococcus aureus, Gram-Negative Superburgs, Yeasts, and Dermatophytic Fungi. Curr. Ther. Res. Clin. Exp. 2016, 83, 8–12. [Google Scholar] [CrossRef][Green Version]
- European Pharmacopoeia, 10th ed.; European Directorate for the Quality of Medicines & HealthCare: Strasbourg, France, 2019.
- Petrie, A.; Sabin, C. Medical Statistics at a Glance, 3rd ed.; John Wiley & Sons: New York, NY, USA, 2009. [Google Scholar]
- MacFarland, T.W.; Yates, J.M. Mann-Whitney U Test. In Introduction to Nonparametric Statistics for the Biological Science Using R; Springer International Publishing AG: Cham, Switzerland, 2016; pp. 103–132. [Google Scholar]
- Shkarenkov, A.A.; Beloshapko, A.A.; Kuznetsov, Y.B.; Borovkova, M.V.; Anikanova, V.V.; Krepkova, L.V. Preclinical toxicological study of Diquertin. Probl. Biol. Med. Pharm. Chem. 1998, 3, 36–39. (In Russian) [Google Scholar]
- Kolkhir, V.K.; Tyukavkina, N.A.; Bykov, V.A.; Glyzin, V.I.; Arzamastsev, A.P.; Baginskaya, A.I.; Sokolov, S.Y.; Kolesnik, Y.A.; Glazova, N.G.; Rulenko, I.A.; et al. Dicvertin: A New Antioxidant and Capillary-Protecting Drugs. Pharm. Chem. J. 1995, 29, 657–660. [Google Scholar] [CrossRef]
- Kolhir, V.K.; Bykov, V.A.; Teselkin, Y.O.; Babenkova, I.V.; Tjukavkina, N.A.; Rulenko, I.A.; Kolesnik, Y.A.; Eichholz, A.A. Use of a new antioxidant diquertin as an adjuvant in the therapy of patients with acute pneumonia. Phytother. Res. 1998, 12, 606–608. [Google Scholar] [CrossRef]
- Fang, Y.; Cao, W.; Xia, M.; Pan, S.; Xu, X. Study of structure and permeability relationship of flavonoids in caco-2 cells. Nutrients 2017, 9, 1301. [Google Scholar] [CrossRef]
- Wong, M.; McAllister, M. Lead Identification/Optimization. In Oral Formulation Roadmap from Early Drug Discovery to Development; Kwong, E., Ed.; John Wiley & Sons Inc: Hoboken, NJ, USA, 2017; pp. 9–38. [Google Scholar]
- Liu, W.; Okochi, H.; Benet, L.Z.; Zhai, S.-D. Sotalol permeability in cultured-cell, rat intestine and PAMPA system. Pharm. Res. 2012, 29, 1768–1774. [Google Scholar] [CrossRef] [PubMed]
- Zu, Y.; Wu, W.; Zhao, X.; Li, Y.; Wang, W.; Zhong, C.; Zhang, Y.; Zhao, X. Enhancement of solubility, antioxidant ability and bioavailability of taxifolin nanoparticles by liquid antisolvent precipitation technique. Int. J. Pharm. 2014, 471, 366–376. [Google Scholar] [CrossRef]
- Zu, S.; Yang, L.; Huang, J.; Ma, C.; Wang, W.; Zhao, C.; Zu, Y. Micronization of Taxifolin by Supercritical Antisolvent Process and Evaluation of Radical Scavenging Activity. Int. J. Mol. Sci. 2012, 13, 8869–8881. [Google Scholar] [CrossRef] [PubMed]
- Potoroko, I.Y.; Kalinina, I.V.; Naumenko, N.V.; Fatkullin, R.I.; Nenasheva, A.V.; Uskova, D.G.; Sonawane, S.H.; Ivanova, D.G.; Velyamov, M.T. Sonochemical micronization of taxifolin aimed at improving its bioavalability in drinks for athletes. Hum. Sport Med. 2018, 18, 90–100. [Google Scholar] [CrossRef]
- Bavishi, D.D.; Borkhataria, C.H. Spring and parachute: How cocrystals enhance solubility. Prog. Cryst. Growth Charact. Mater. 2016, 62, 1–8. [Google Scholar] [CrossRef]
- Smith, A.J.; Kavuru, P.; Arora, K.K.; Kesani, S.; Tan, J.; Zaworotko, M.J.; Shytle, R.D. Crystal engineering of green tea epigallocatechin-3-gallate (EGCg) cocrystals and pharmacokinetic modulation in rats. Mol. Pharm. 2013, 10, 2948–2961. [Google Scholar] [CrossRef]
- Wang, C.; Tong, Q.; Hou, X.; Hu, S.; Fang, J.; Sun, C.C. Enhancing Bioavailability of Dihydromyricetin through Inhibiting Precipitation of Soluble Cocrystals by a Crystallization Inhibitor. Cryst. Growth Des. 2016, 16, 5030–5039. [Google Scholar] [CrossRef]
- Kassem, F.A.; Abdelaziz, A.E.; El Maghraby, G.M. Ethanol-assisted kneading of apigenin with arginine for enhanced dissolution rate of apigenin: Development of rapidly disintegrating tablets. Pharm. Dev. Tech. 2021, 26, 693–700. [Google Scholar] [CrossRef]
- Shevelev, A.B.; la Porta, N.; Isakova, E.P.; Martens, S.; Biryukova, Y.K.; Belous, A.S.; Sivokhin, D.A.; Trubnikova, E.V.; Zylkova, M.V.; Belyakova, A.V.; et al. In Vivo Antimicrobial and Wound-Healing Activity of Resveratrol, Dihydroquercetin, and Dihydromyricetin against Staphylococcus aureus, Pseudomonas aeruginosa, and Candida albicans. Pathogens 2020, 9, 296. [Google Scholar] [CrossRef]
- Ivanov, M.; Novović, K.; Malešević, M.; Dinić, M.; Stojković, D.; Jovčić, B.; Soković, M. Polyphenols as Inhibitors of Antibiotic Resistant Bacteria-Mechanisms Underlying Rutin Interference with Bacterial Virulence. Pharmaceuticals 2022, 15, 385. [Google Scholar] [CrossRef]
- Abugri, D.A.; Witola, W.H.; Russel, A.E.; Troy, R.M. In vitro activity of the interaction between taxifolin (dihydroquercetin) and pyrimethamine against Toxoplasma gondii. Chem. Biol. Drug Des. 2018, 91, 194–201. [Google Scholar] [CrossRef]
- Taldaev, A.K.; Terekhov, R.P.; Selivanova, I.A. Antimicrobial activity of dihydroqurcetin against E. coli. In Mendeleev 2021. Book of Abstracts XII International Conference on Chemistry for Young Scientists; VVM Publishing LLC: Saint-Petersburg, Russia, 2021; p. 172. [Google Scholar]
- Terekhov, R.P.; Selivanova, I.A. Molecular modeling of the interaction of the dihydroquercetin and its metabolites with cyclooxygenase-2. Bull. Sib. Med. 2019, 18, 101–106. (In Russian) [Google Scholar] [CrossRef]
- Ha, C.H.H.; Fatima, A.; Gaurav, A. In silico investigation of flavonoids as potential trypanosomal nucleoside hydrolase inhibitors. Adv. Bioinforma 2015, 12, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Abdulrazzak, S.A.; Vorobyev, A.N.; Sinitsyna, N.I.; Sharutina, I.V.; Menshova, O.V.; Elizarova, E.V.; Ivanova, Y.V.; Karamyan, A.S.; Abramovich, R.A.; Potanina, O.G.; et al. Development of Dihydroquercetin-based Oral tablets and Evalution of the General Toxic Effect. Drug Dev. Regist. 2022, 11, 126–138. (In Russian) [Google Scholar] [CrossRef]
- Peltonen, L.; Strachan, C.J. Degrees of order: A comparison of nanocrystal and amorphous solids for poorly soluble drugs. Int. J. Pharm. 2020, 586, 119492. [Google Scholar] [CrossRef]
- Murdande, S.B.; Pikal, M.J.; Shanker, R.M.; Bogner, R.H. Solubility advantage of amorphous pharmaceuticals: I. A thermodynamic analysis. J. Pharm. Sci. 2010, 99, 1254–1264. [Google Scholar] [CrossRef]

| No | Mass of Tablet, mg | Mass of Component, mg | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| TS | Saccharose | Mannitol | Sorbitol | Sodium Starch Glycolate | Crospovidone | Calcium Stearate | Aspartame | Menthol | ||
| 1 | 150.0 | 20.0 | - | 125.0 | - | 3.0 | - | 2.0 | - | - |
| 2 | 150.0 | 20.0 | - | 125.0 | - | - | 3.0 | 2.0 | - | - |
| 3 | 150.0 | 20.0 | 125.0 | - | - | 3.0 | - | 2.0 | - | - |
| 4 | 150.0 | 20.0 | 125.0 | - | - | - | 3.0 | 2.0 | - | - |
| 5 | 150.0 | 20.0 | - | - | 125.0 | 3.0 | - | 2.0 | - | - |
| 6 | 150.0 | 20.0 | - | - | 125.0 | - | 3.0 | 2.0 | - | - |
| 7 | 150.0 | 20.0 | - | 120.0 | - | 3.0 | - | 2.0 | - | - |
| 8 | 150.0 | 20.0 | - | 120.0 | - | - | 3.0 | 2.0 | - | - |
| 9 | 150.0 | 20.0 | 125.0 | - | - | 3.0 | - | 2.0 | - | - |
| 10 | 150.0 | 20.0 | 125.0 | - | - | - | 3.0 | 2.0 | - | - |
| 11 | 150.0 | 20.0 | - | - | 120.0 | 3.0 | - | 2.0 | - | - |
| 12 | 150.0 | 20.0 | - | - | 120.0 | - | 3.0 | 2.0 | - | - |
| 13 | 150.0 | 20.0 | - | 120.0 | - | - | 3.0 | 2.0 | 5.0 | - |
| 14 | 150.0 | 20.0 | 110.0 | - | - | - | 8.0 | 2.0 | 5.0 | 5.0 |
| 15 | 150.0 | 20.0 | 125.0 | - | - | - | 8.0 | 2.0 | 5.0 | - |
| 16 | 150.0 | 20.0 | - | 120.0 | - | - | 3.0 | 2.0 | - | 5.0 |
| 17 | 150.0 | 20.0 | 115.0 | - | - | - | 8.0 | 2.0 | - | 5.0 |
| 18 | 300.0 | 20.0 | - | 270.0 | - | - | 3.0 | 2.0 | - | 5.0 |
| 19 | 300.0 | 20.0 | 265.0 | - | - | - | 8.0 | 2.0 | - | 5.0 |
| Sample | Taxifolin Concentration, mg/mL | ||||
|---|---|---|---|---|---|
| 0 h | 1 h | 2 h | 4 h | 6 h | |
| RT | 0.700 ± 0.016 | 0.764 ± 0.017 | 0.785 ± 0.018 | 0.884 ± 0.020 | 0.956 ± 0.023 |
| TS | 1.642 ± 0.177 | 1.764 ± 0.180 | 1.822 ± 0.196 | 2.099 ± 0.226 | 2.127 ± 0.229 |
| Sample | Growth Inhibition Zone, mm | |||
|---|---|---|---|---|
| Staphylococcus aureus | Escherichia coli | Micrococcus luteus | Bacillus cereus | |
| RT | 13.67 ± 0.58 | 14.00 ± 1.00 | 14.00 ± 0.00 | 14.33 ± 0.58 |
| TS | 13.00 ± 0.00 | 13.33 ± 1.15 | 13.67 ± 0.58 | 14.00 ± 0.00 |
| Sample | HR | CI, % | Flow Description |
|---|---|---|---|
| RT | 1.421 ± 0.149 | 29.63 ± 3.11 | poor |
| TS | 1.219 ± 0.113 | 19.00 ± 1.77 | fair |
| No. | Disintegration Time, min | Hardness, N | Taste | ||
|---|---|---|---|---|---|
| Measurement | Regulatory Requirements | Measurement | Regulatory Requirements | ||
| 1 | 9.3 ± 0.6 | [15.0–45.0] | - | >40.0 | - |
| 2 | 13.8 ± 0.9 | - | - | ||
| 3 | 4.4 ± 0.3 | - | - | ||
| 4 | 4.7 ± 0.3 | - | - | ||
| 5 | 7.3 ± 0.5 | - | - | ||
| 6 | 4.9 ± 0.3 | - | - | ||
| 7 | 1.3 ± 0.1 | - | - | ||
| 8 | 7.3 ± 0.5 | - | - | ||
| 9 | 9.8 ± 0.6 | - | - | ||
| 10 | 18.1 ± 1.1 | 71.9 ± 20.8 | bittersweet, astringent | ||
| 11 | 3.8 ± 0.4 | - | - | ||
| 12 | 6.2 ± 2.3 | - | - | ||
| 13 | 39.9 ± 2.5 | 22.2 ± 6.4 | - | ||
| 14 | 17.0 ± 1.1 | 25.1 ± 7.3 | - | ||
| 15 | 23.1 ± 1.5 | 26.0 ± 7.5 | - | ||
| 16 | 10.1 ± 0.6 | - | - | ||
| 17 | 40.2 ± 2.3 | 34.5 ± 10.0 | - | ||
| 18 | 8.0 ± 0.5 | - | - | ||
| 19 | 36.4 ± 2.3 | 81.0 ± 23.4 | cooling, spicy | ||
| Sample | Hardness, N | Friability, % | ||
|---|---|---|---|---|
| Measurement | Regulatory Requirements | Measurement | Regulatory Requirements | |
| RT | 74.9 ± 33.6 | >40.0 | 0.3 | <3.0 |
| TS | 81.0 ± 23.4 | 1.3 | ||
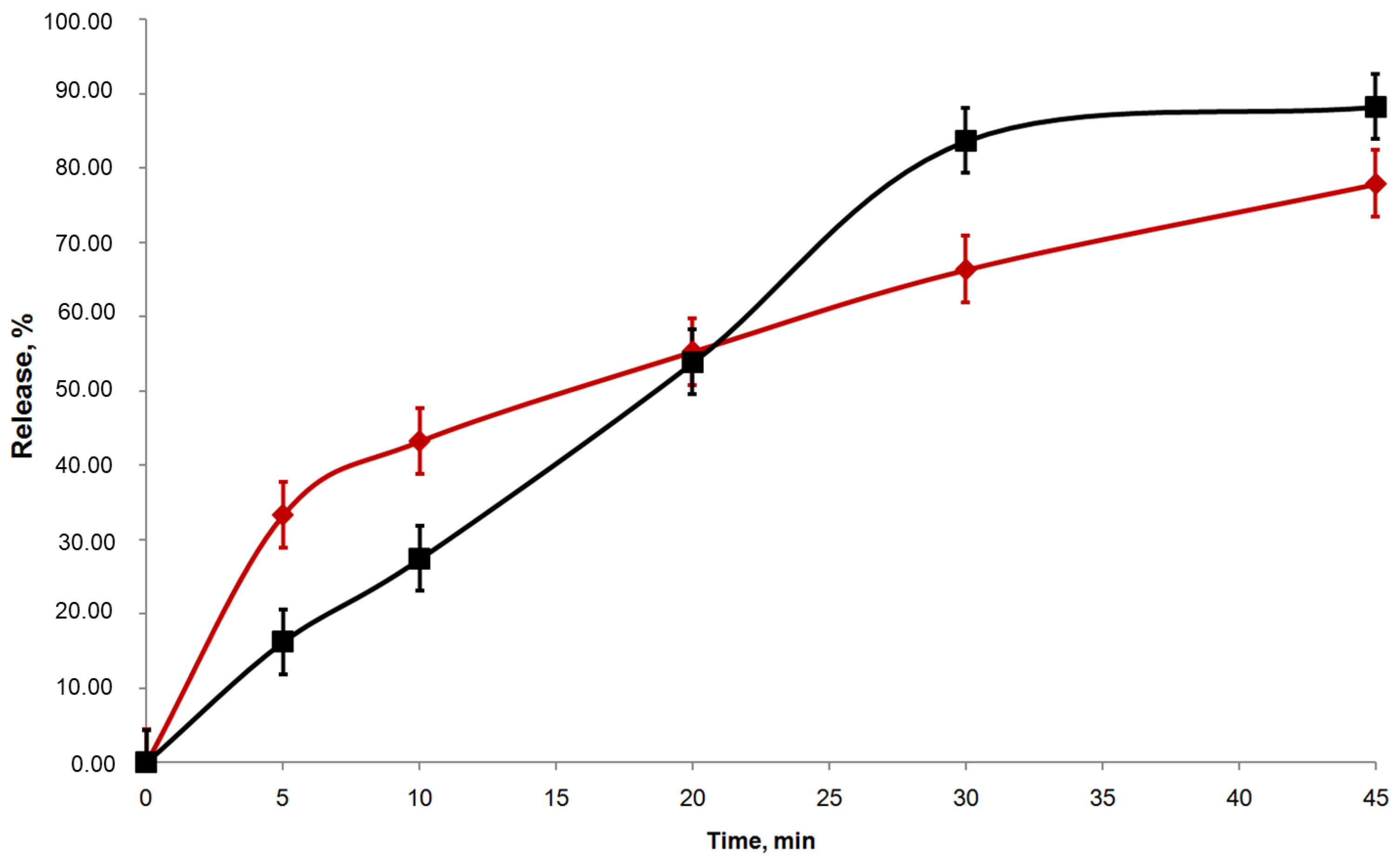
| Sample | Parameters of Release Kinetic | f1 | f2 | |
|---|---|---|---|---|
| K, min−1 | T50%, min | |||
| RT | 0.034 ± 0.002 | 20.63 ± 1.13 | 23.0 ± 1.7 | 42.8 ± 6.0 |
| TS | 0.048 ± 0.002 | 14.56 ± 0.48 | ||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Taldaev, A.; Terekhov, R.P.; Selivanova, I.A.; Pankov, D.I.; Anurova, M.N.; Markovina, I.Y.; Cong, Z.; Ma, S.; Dong, Z.; Yang, F.; et al. Modification of Taxifolin Properties by Spray Drying. Sci. Pharm. 2022, 90, 67. https://doi.org/10.3390/scipharm90040067
Taldaev A, Terekhov RP, Selivanova IA, Pankov DI, Anurova MN, Markovina IY, Cong Z, Ma S, Dong Z, Yang F, et al. Modification of Taxifolin Properties by Spray Drying. Scientia Pharmaceutica. 2022; 90(4):67. https://doi.org/10.3390/scipharm90040067
Chicago/Turabian StyleTaldaev, Amir, Roman P. Terekhov, Irina A. Selivanova, Denis I. Pankov, Maria N. Anurova, Irina Yu. Markovina, Zhaoqing Cong, Siqi Ma, Zhengqi Dong, Feifei Yang, and et al. 2022. "Modification of Taxifolin Properties by Spray Drying" Scientia Pharmaceutica 90, no. 4: 67. https://doi.org/10.3390/scipharm90040067
APA StyleTaldaev, A., Terekhov, R. P., Selivanova, I. A., Pankov, D. I., Anurova, M. N., Markovina, I. Y., Cong, Z., Ma, S., Dong, Z., Yang, F., & Liao, Y. (2022). Modification of Taxifolin Properties by Spray Drying. Scientia Pharmaceutica, 90(4), 67. https://doi.org/10.3390/scipharm90040067







