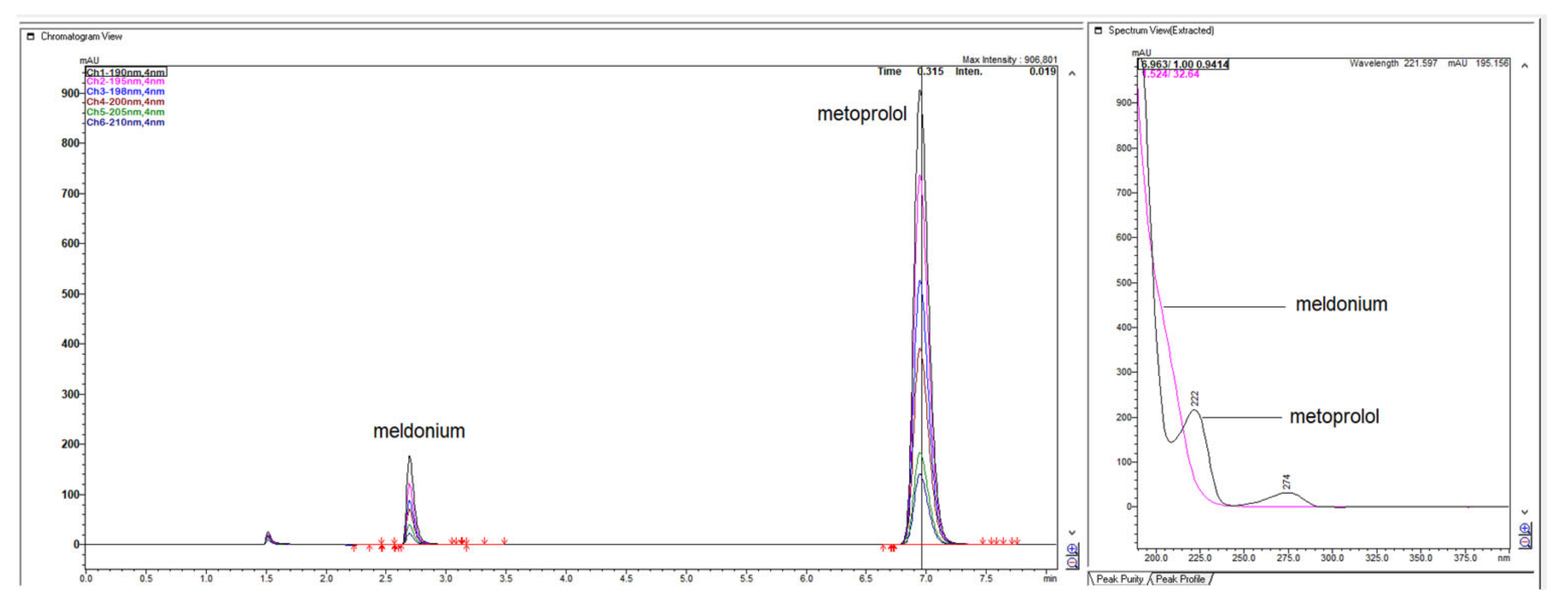
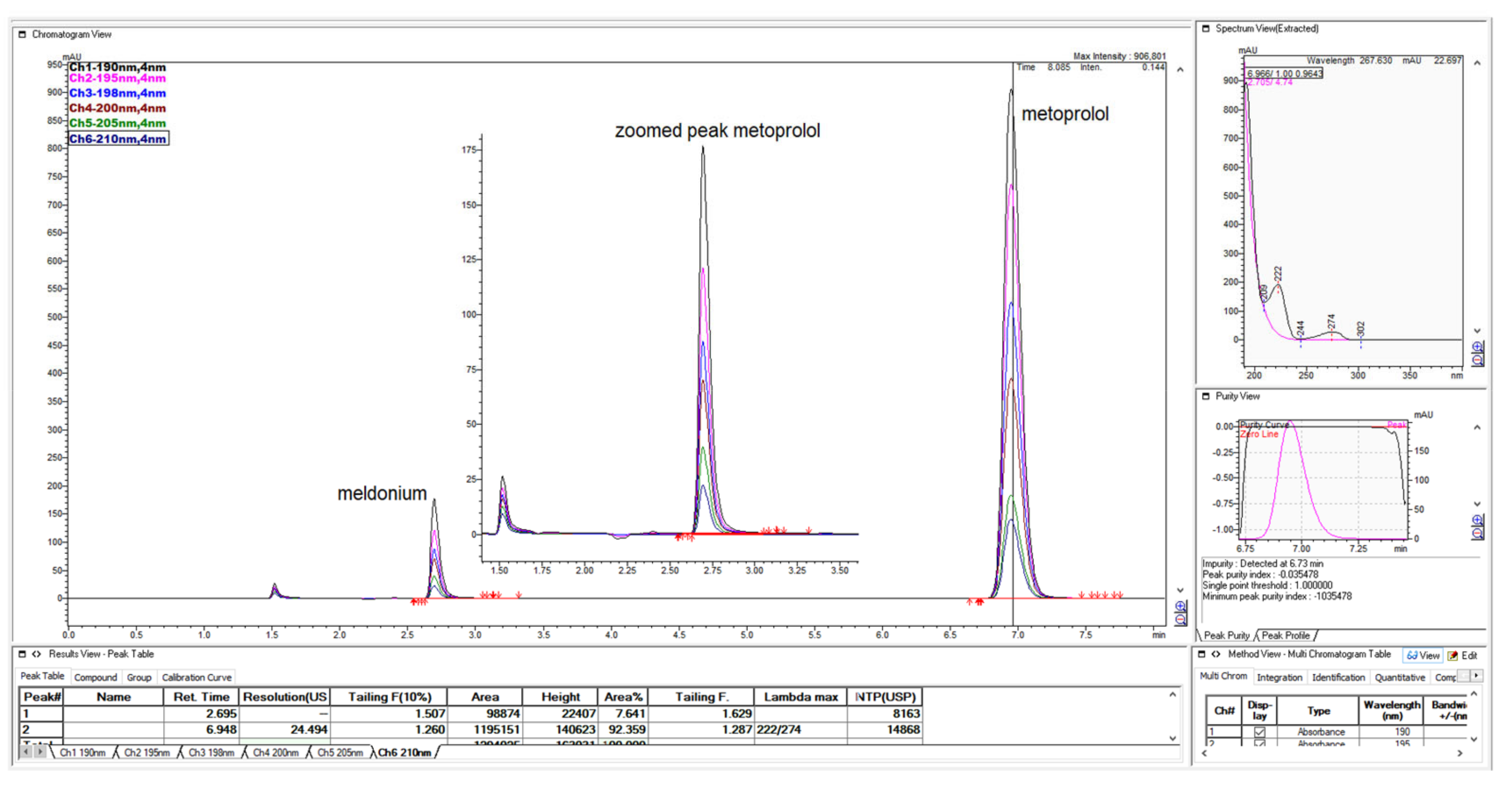
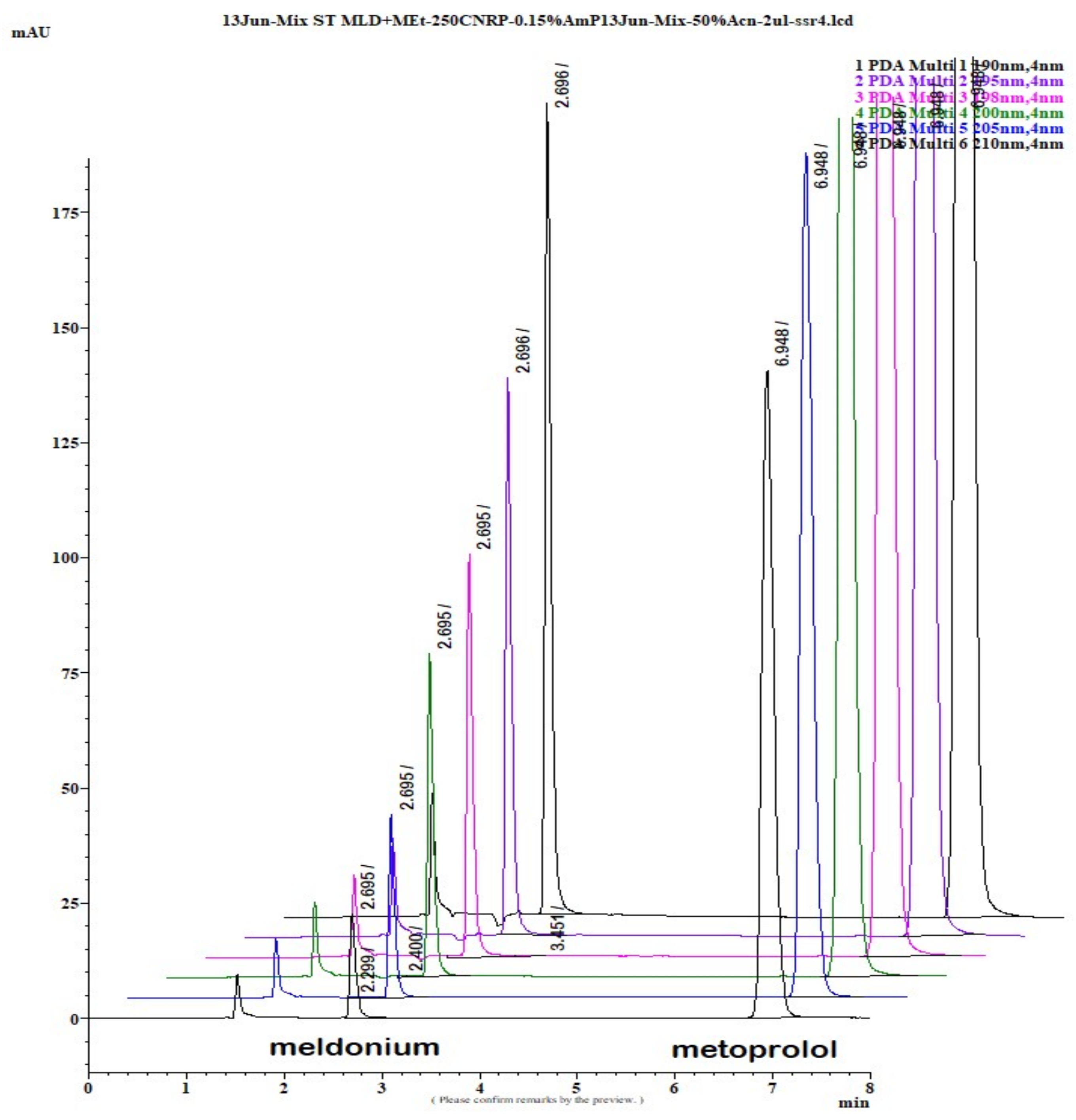
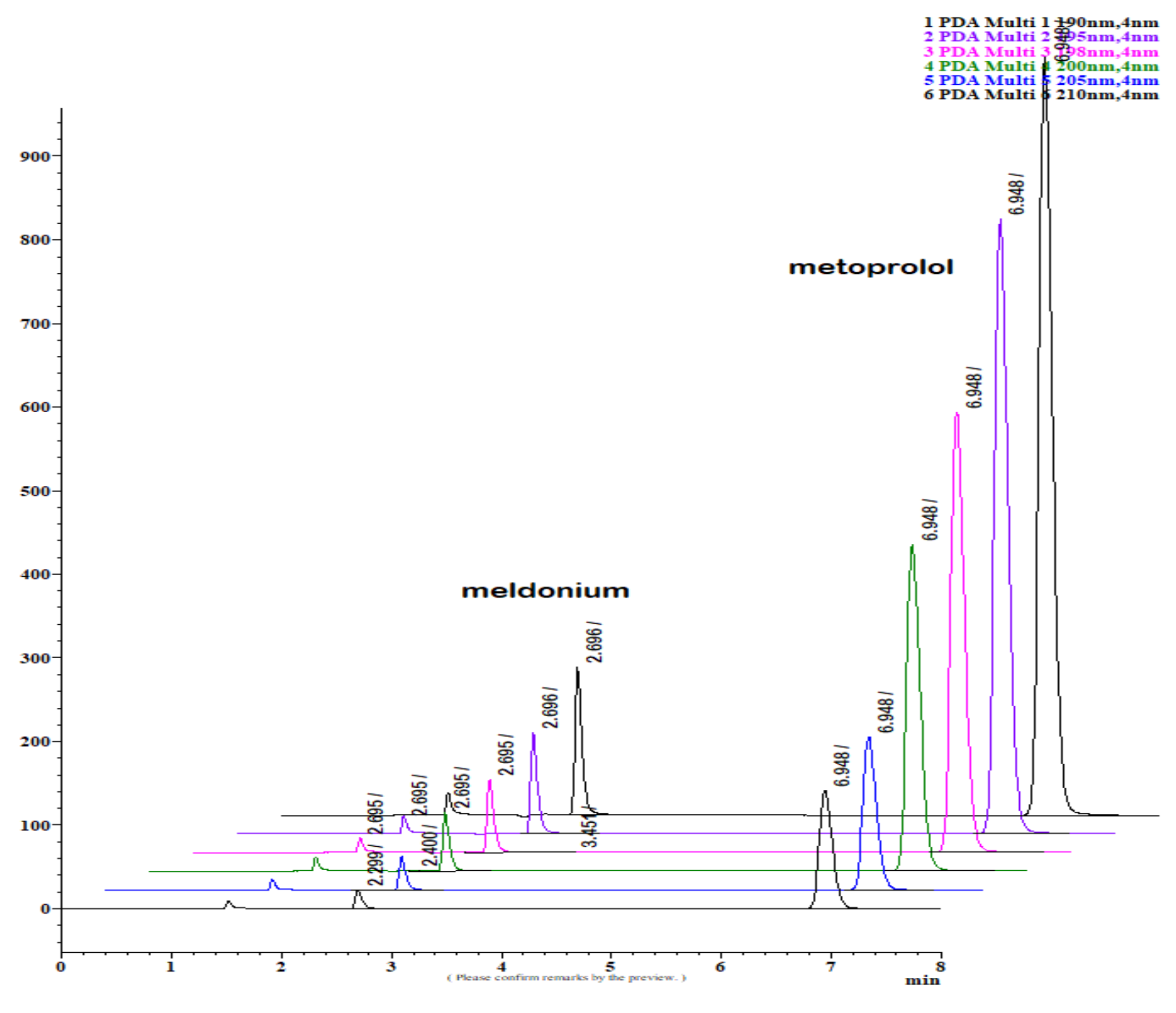
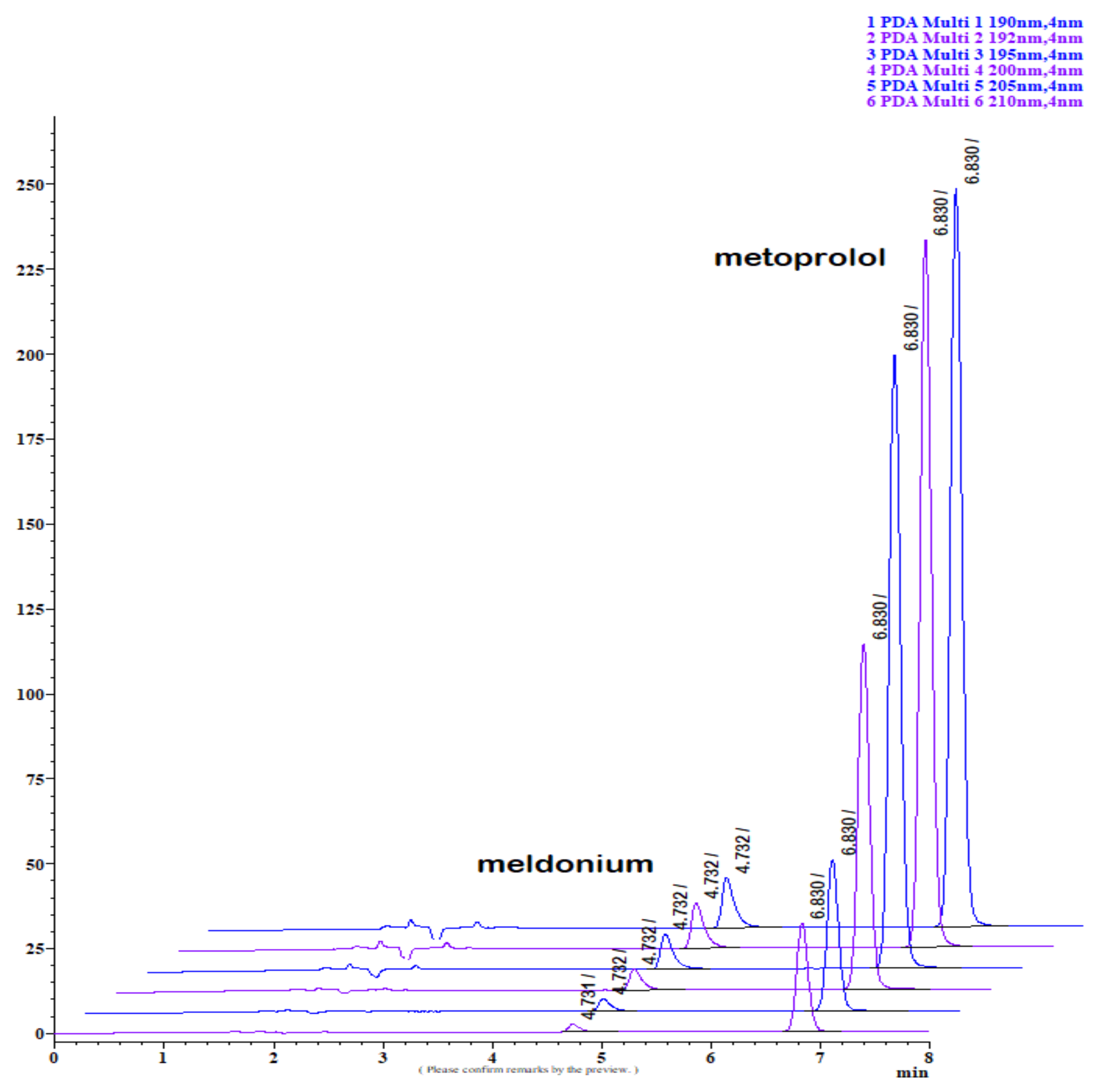
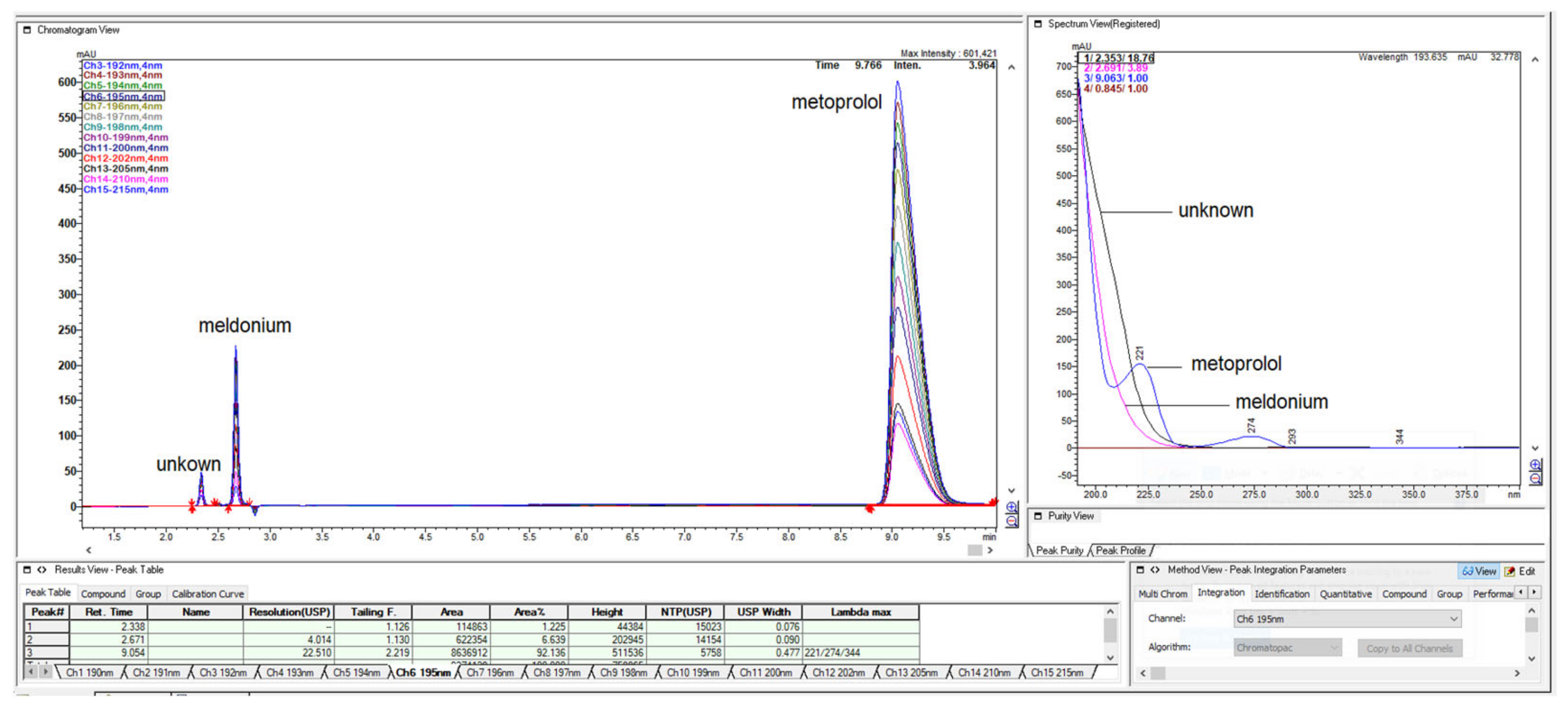
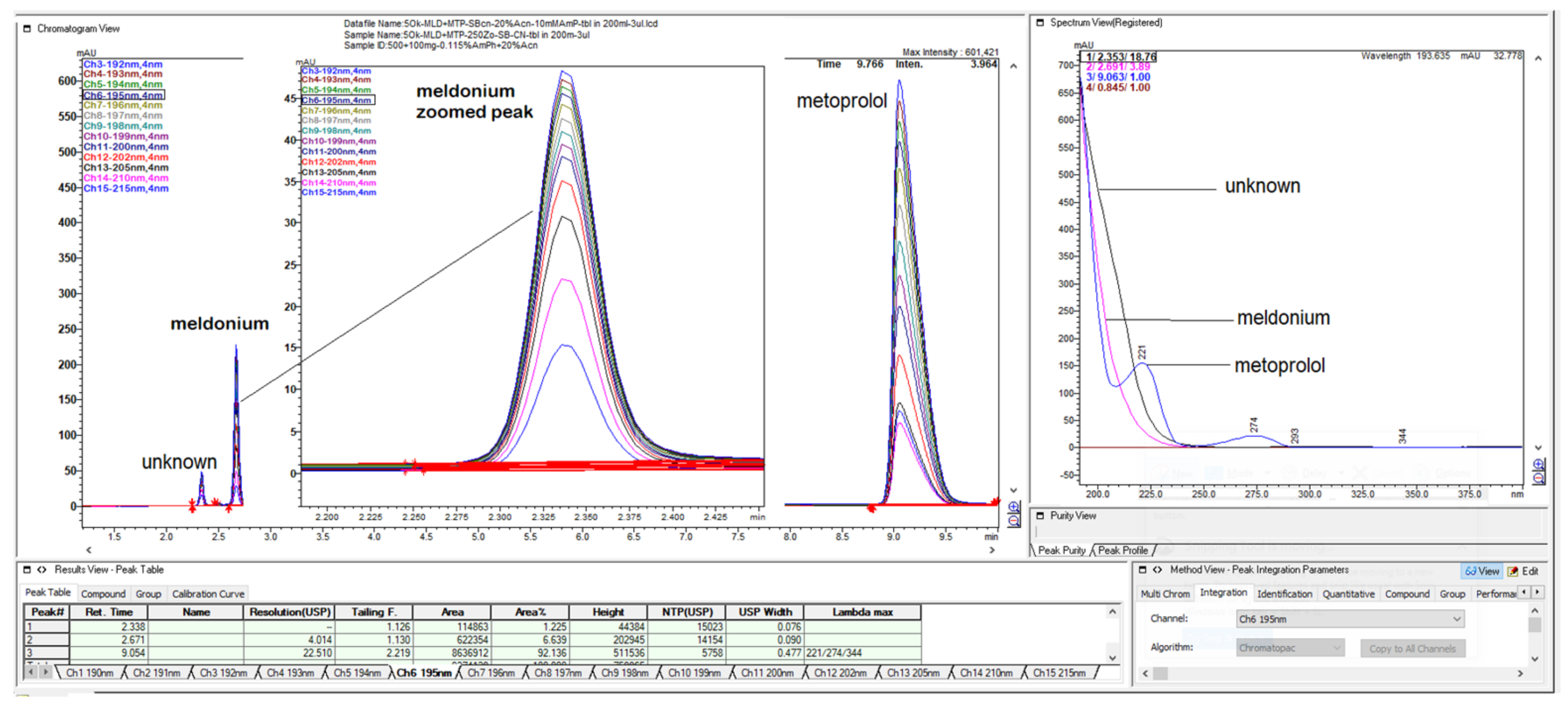
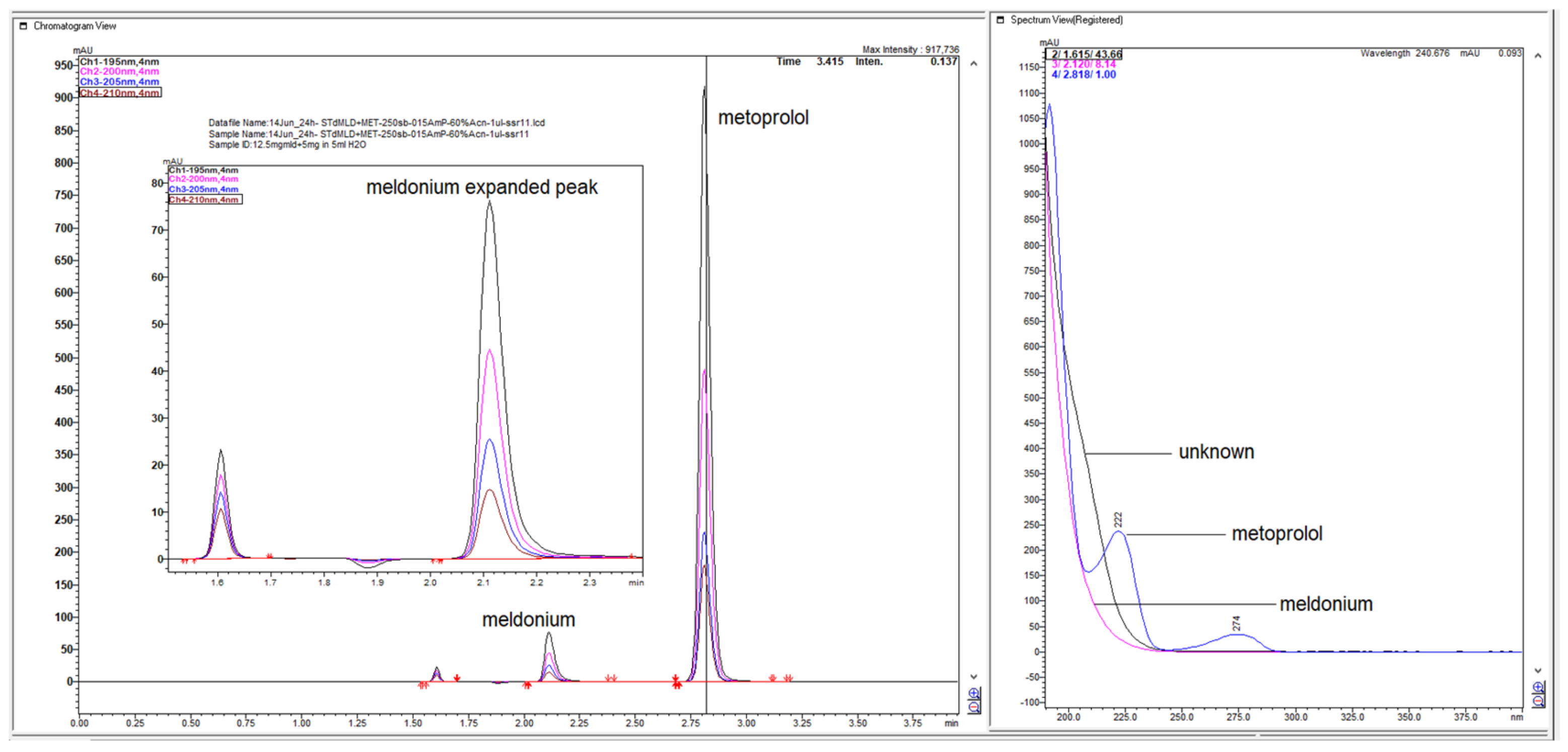
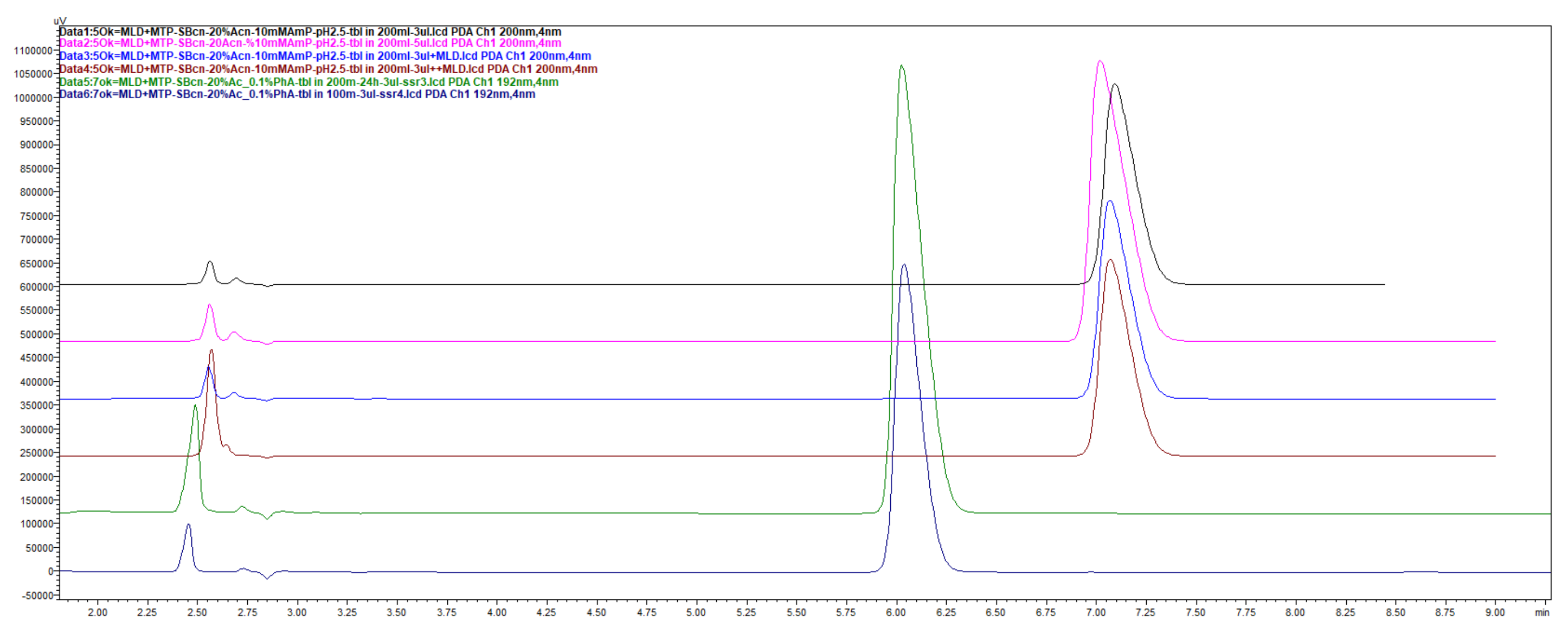
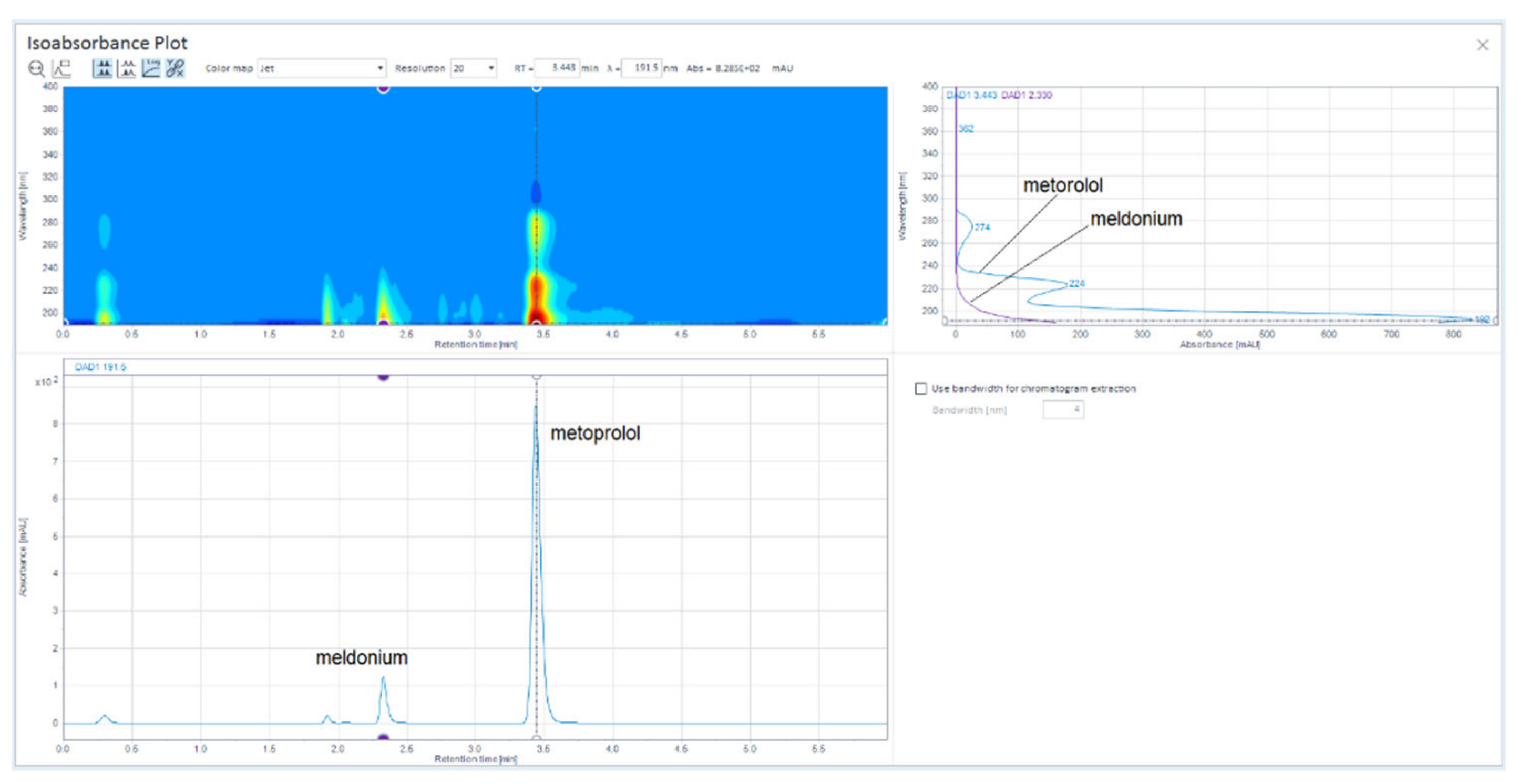
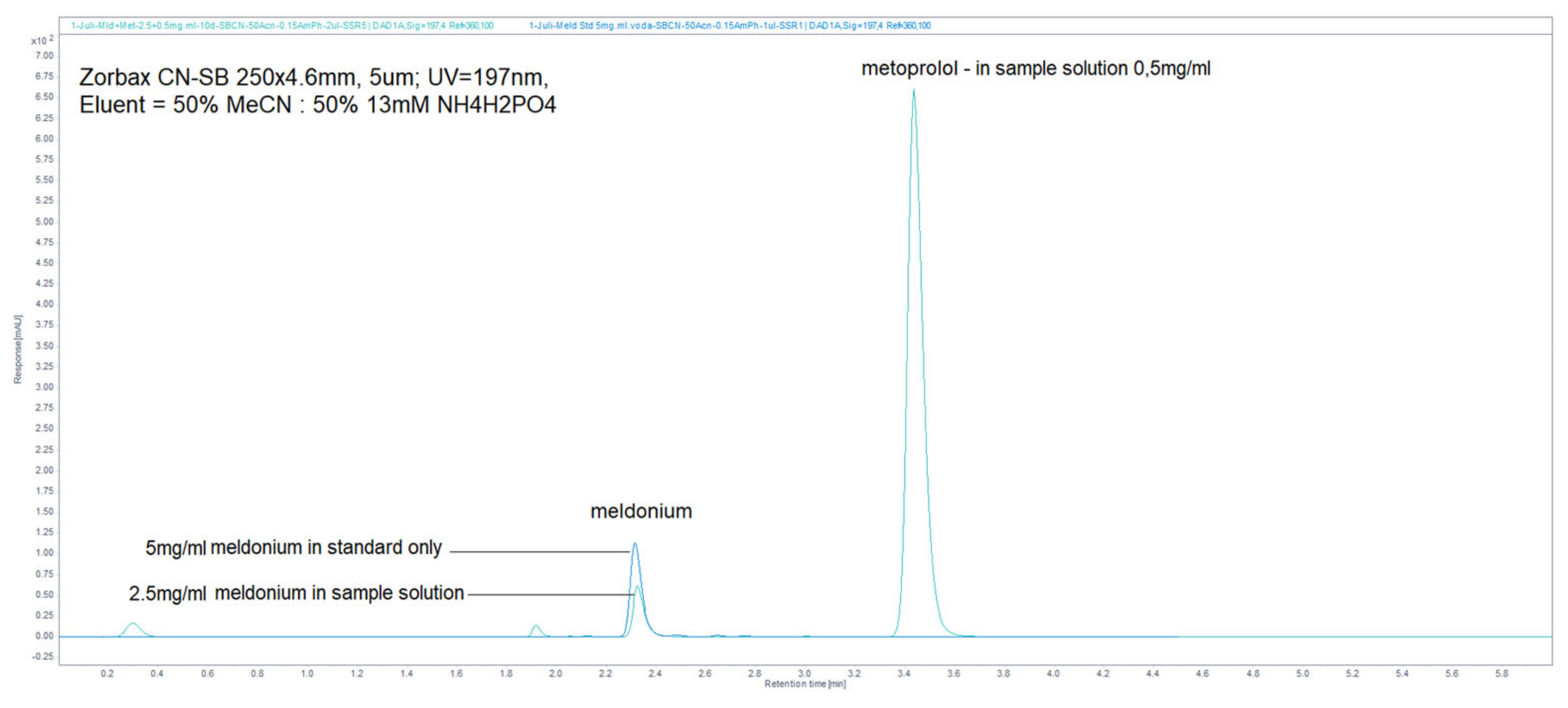
Figure 1.
LiChrospher 100 CN column 125 × 4 mm, 2-wavelength comparison chromatogram, worked out with 20% ACN and 80% 12 mM (0.15% s/w) NH4H2PO4 (Shimadzu LC-40).
Figure 1.
LiChrospher 100 CN column 125 × 4 mm, 2-wavelength comparison chromatogram, worked out with 20% ACN and 80% 12 mM (0.15% s/w) NH4H2PO4 (Shimadzu LC-40).
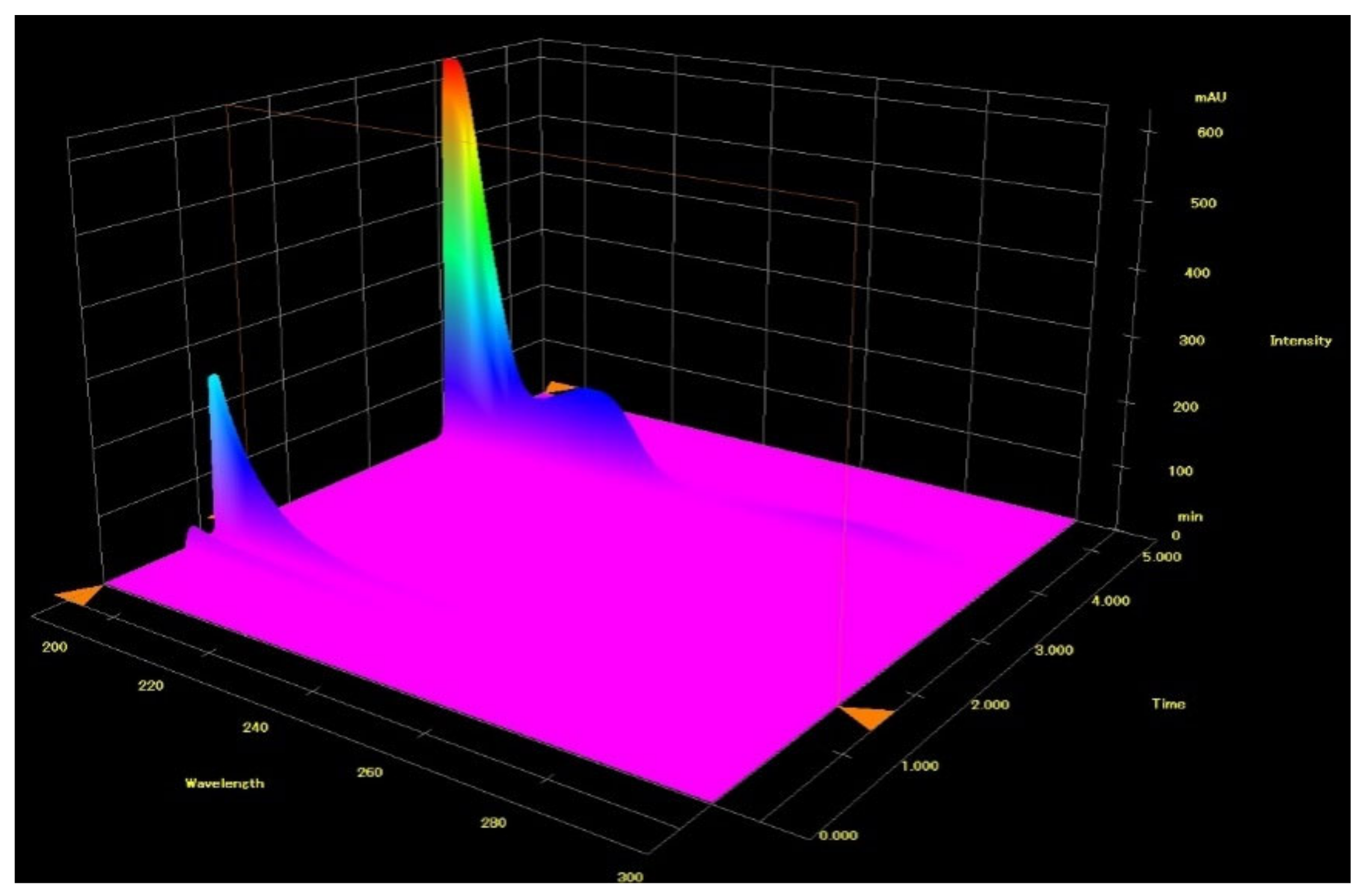
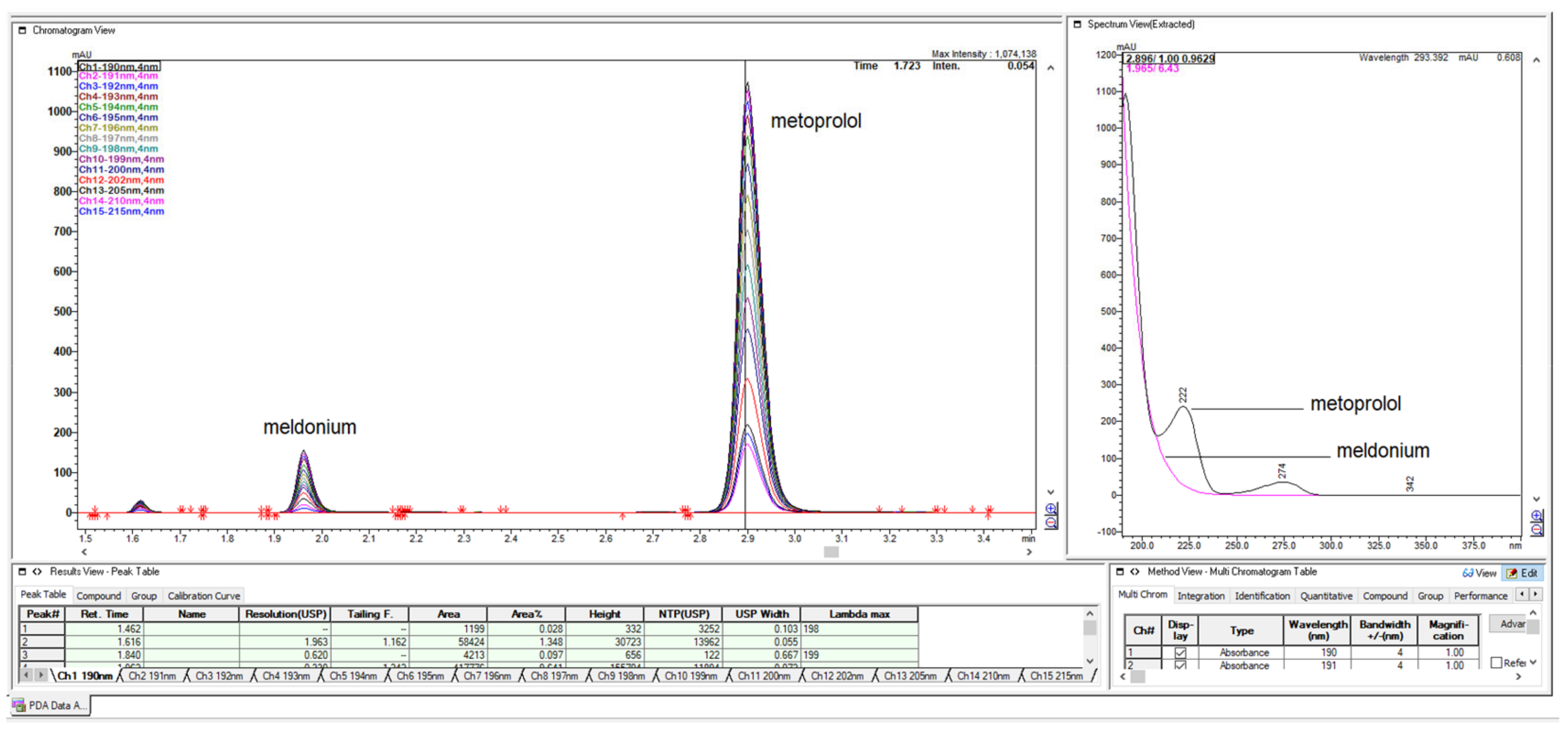
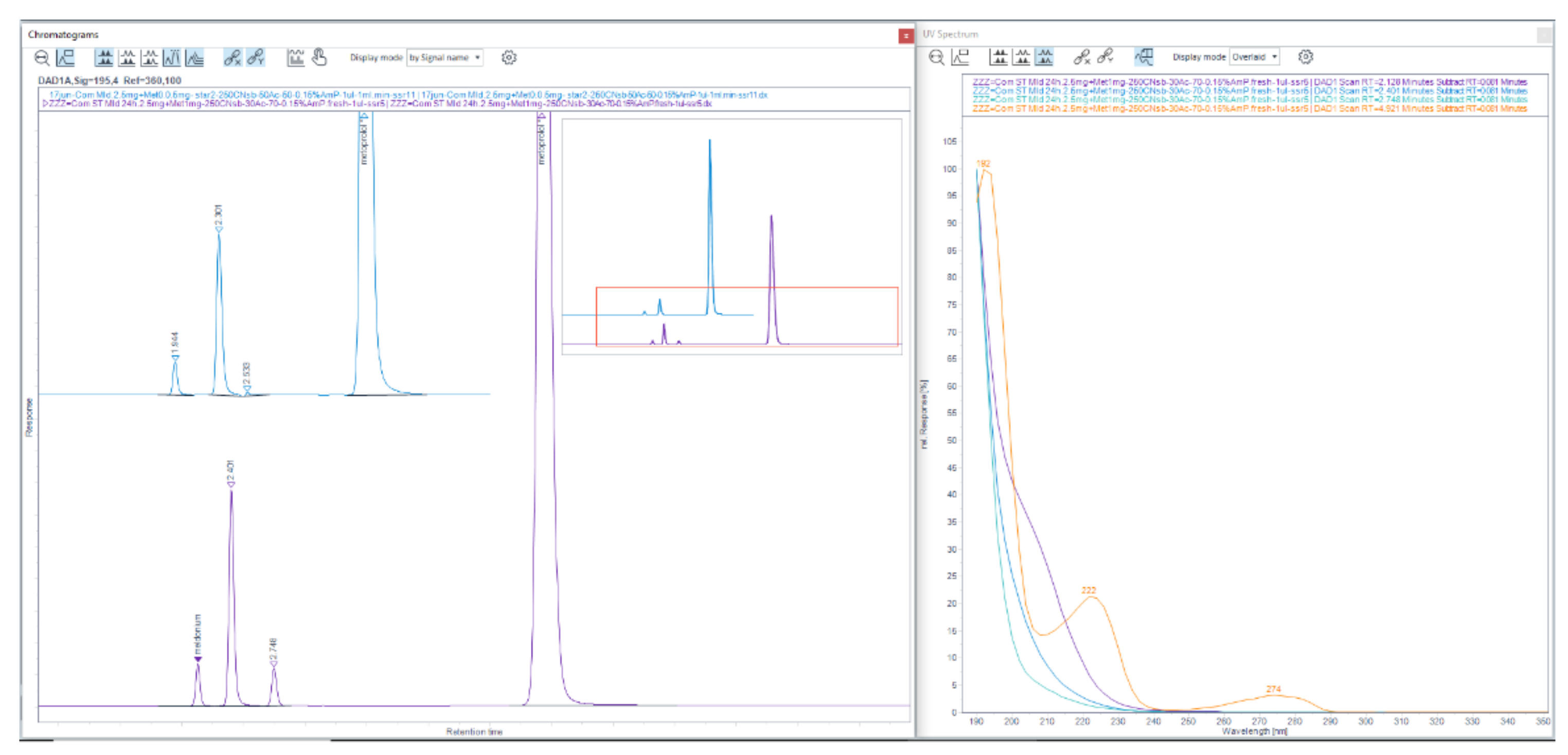
Figure 2.
Full 3-D chromatogram of LiChrospher 125 × 4 mm column in acetonitrile and from the upper figure, got with 20% ACN and 80% 13 mM NH4H2PO4. The first meldonium peak eluted at retention time about 1.25 min, which showed poor UV-abs spectrum exponentially falling in value as the wavelength increased from 190 nm to about 230–235 nm, while meldonium shows intense three max Abs peaks.
Figure 2.
Full 3-D chromatogram of LiChrospher 125 × 4 mm column in acetonitrile and from the upper figure, got with 20% ACN and 80% 13 mM NH4H2PO4. The first meldonium peak eluted at retention time about 1.25 min, which showed poor UV-abs spectrum exponentially falling in value as the wavelength increased from 190 nm to about 230–235 nm, while meldonium shows intense three max Abs peaks.
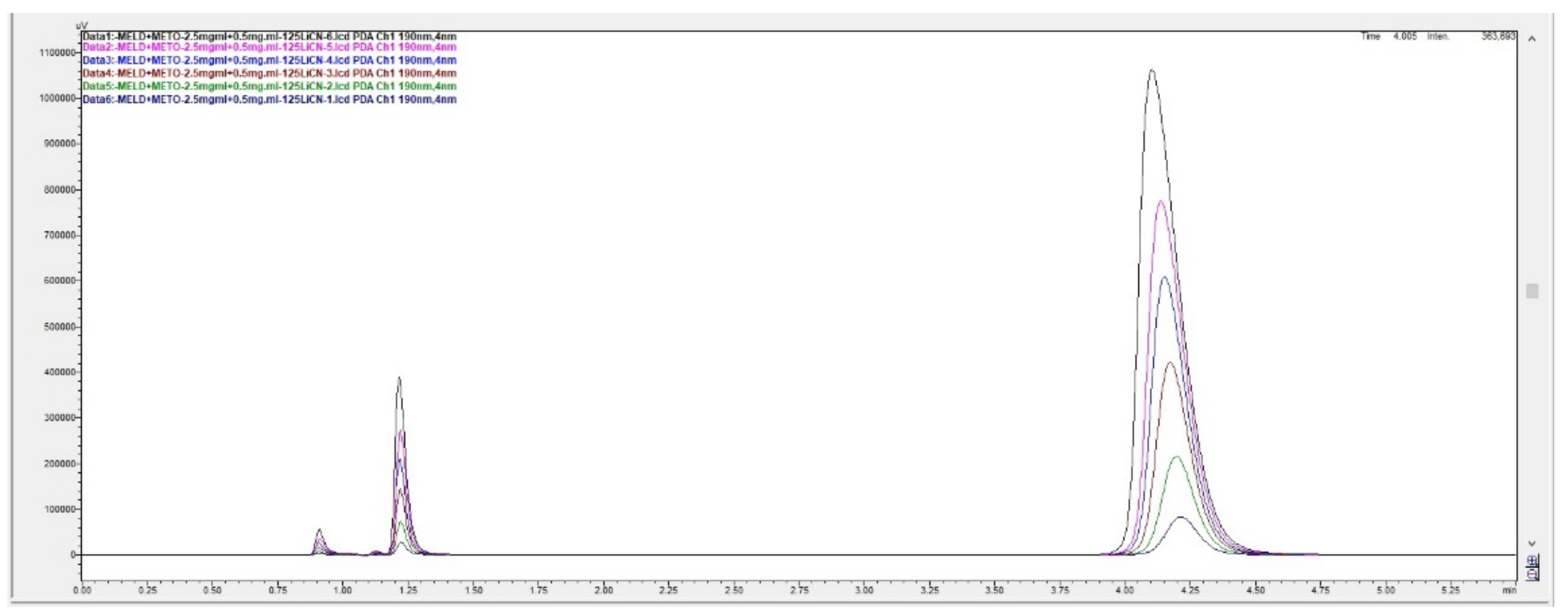
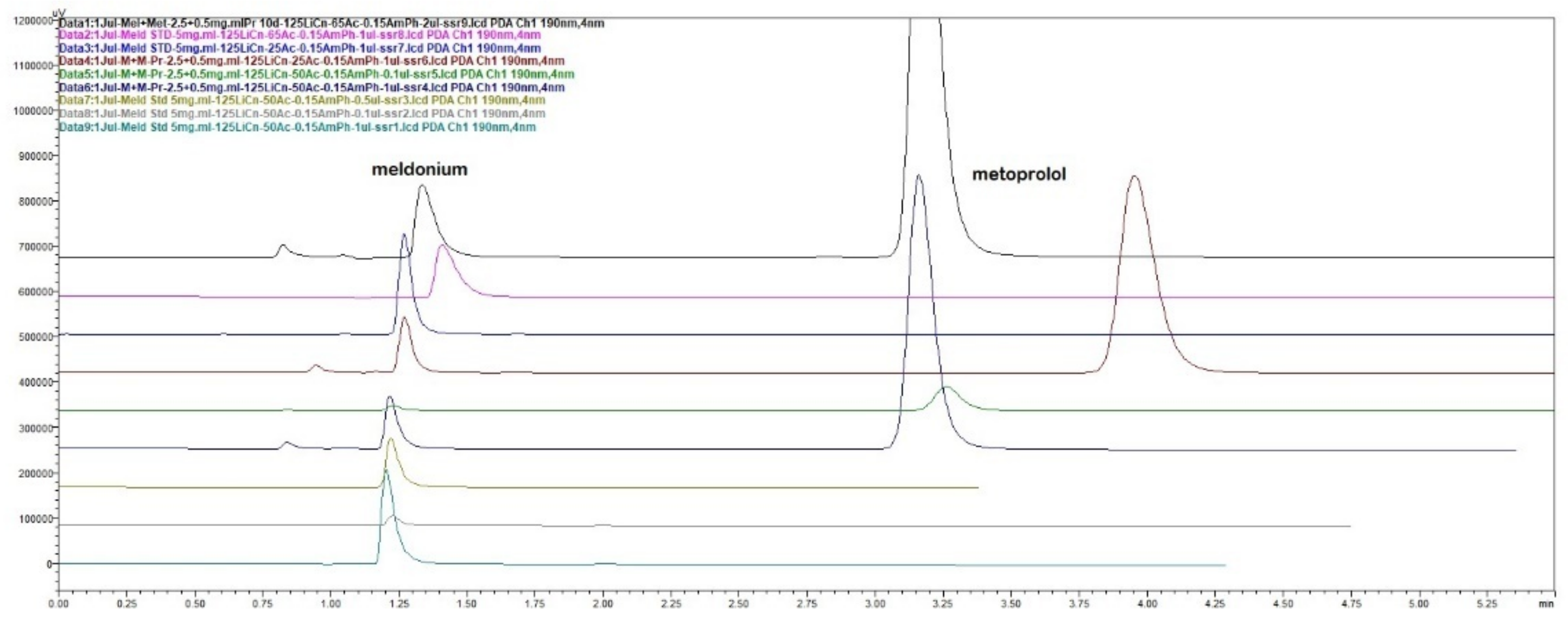
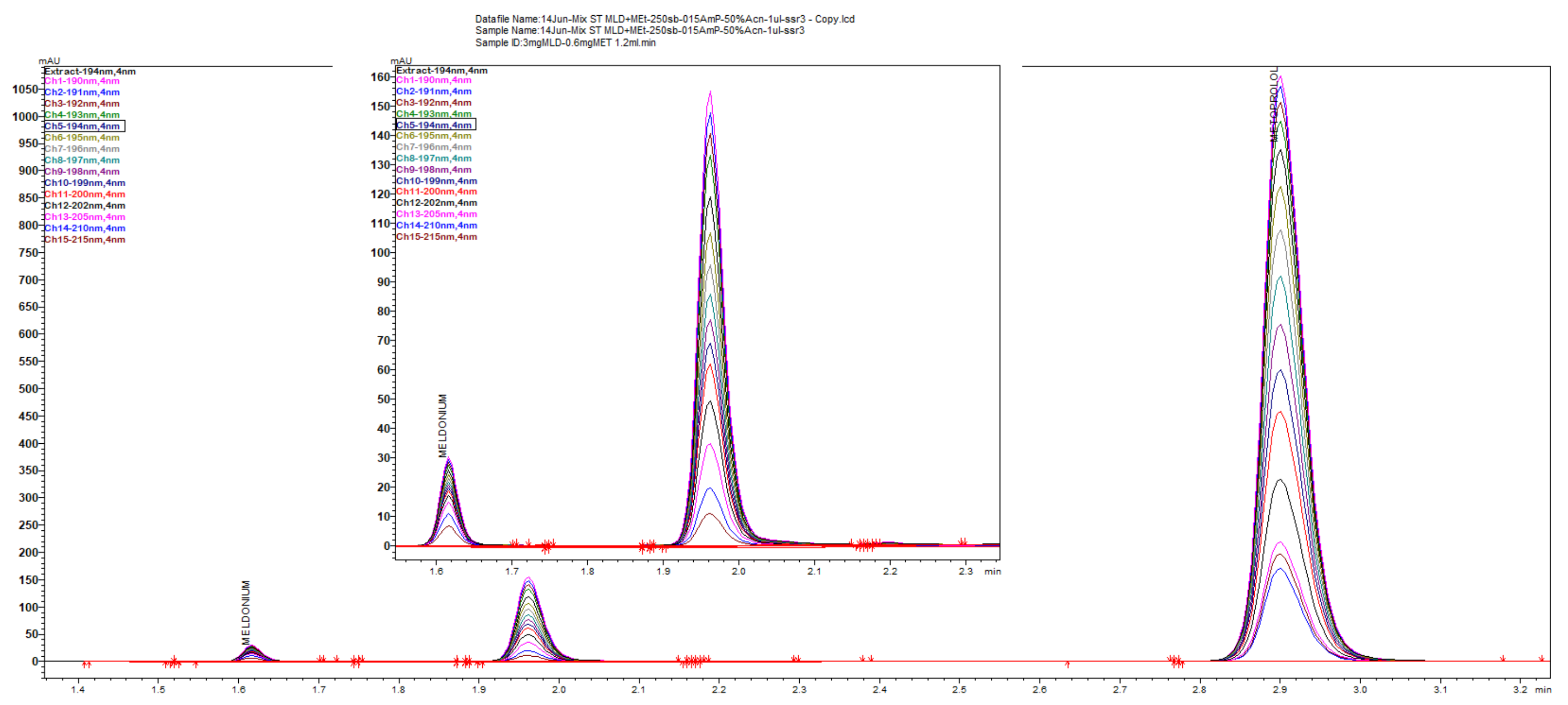
Figure 3.
Overlaid chromatograms for linearity of peak responses to concentrations for meldonium and metoprolol with the short 125 × 4 mm LiChrospher column.
Figure 3.
Overlaid chromatograms for linearity of peak responses to concentrations for meldonium and metoprolol with the short 125 × 4 mm LiChrospher column.
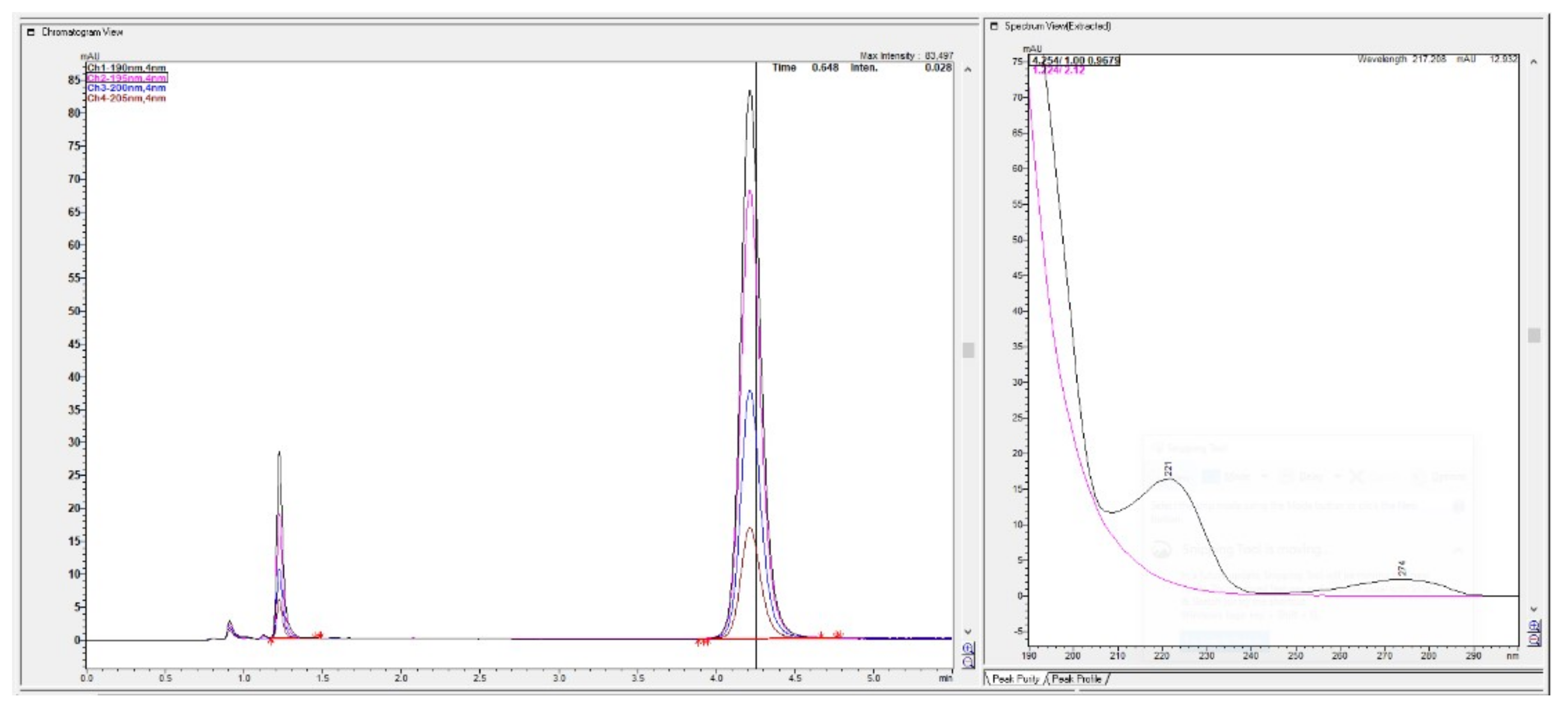
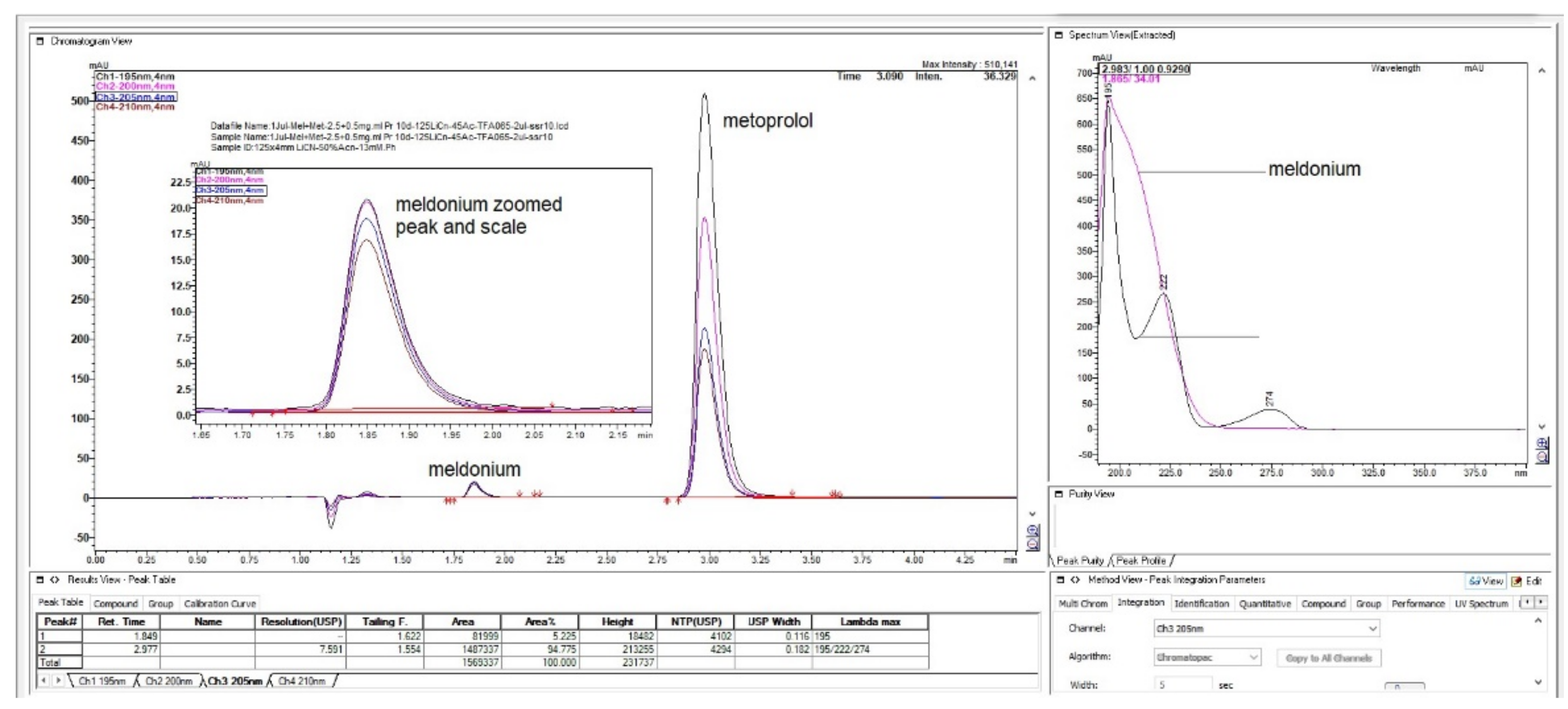
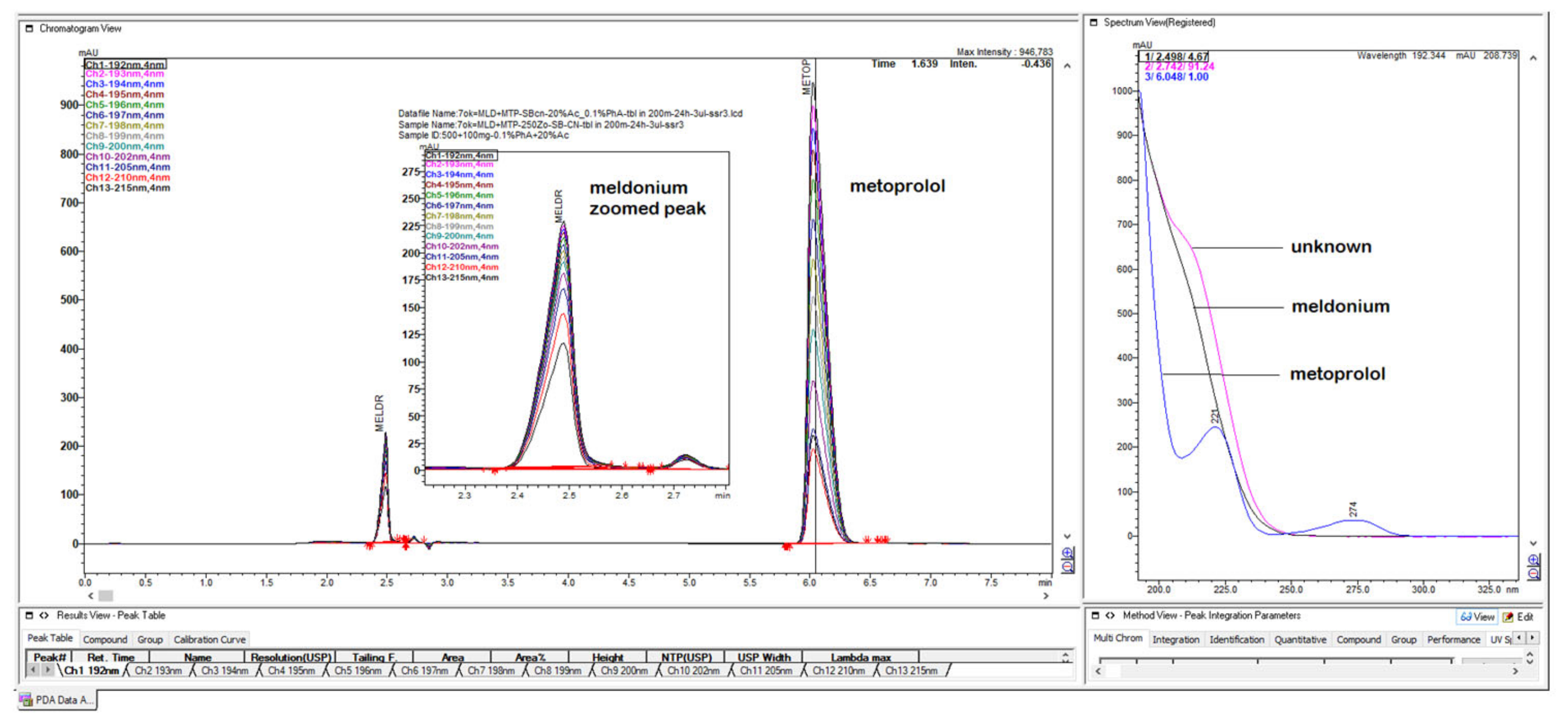
Figure 4.
Chromatograms with first peak with 1.25 min of meldonium with its concave UV spectrum was overlaid with metoprolol spectrum, from the bigger peak eluted second at 4 min.
Figure 4.
Chromatograms with first peak with 1.25 min of meldonium with its concave UV spectrum was overlaid with metoprolol spectrum, from the bigger peak eluted second at 4 min.
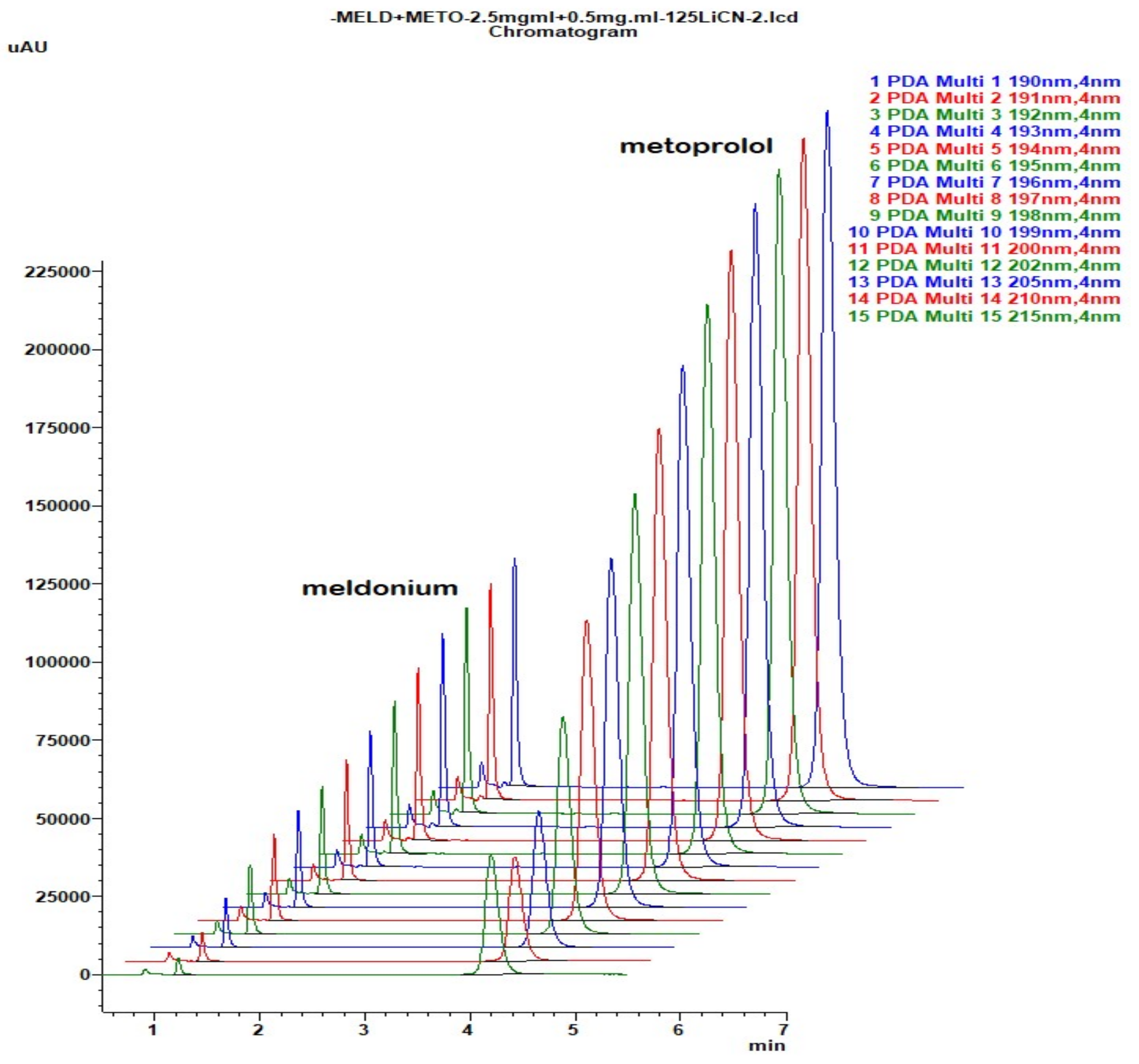
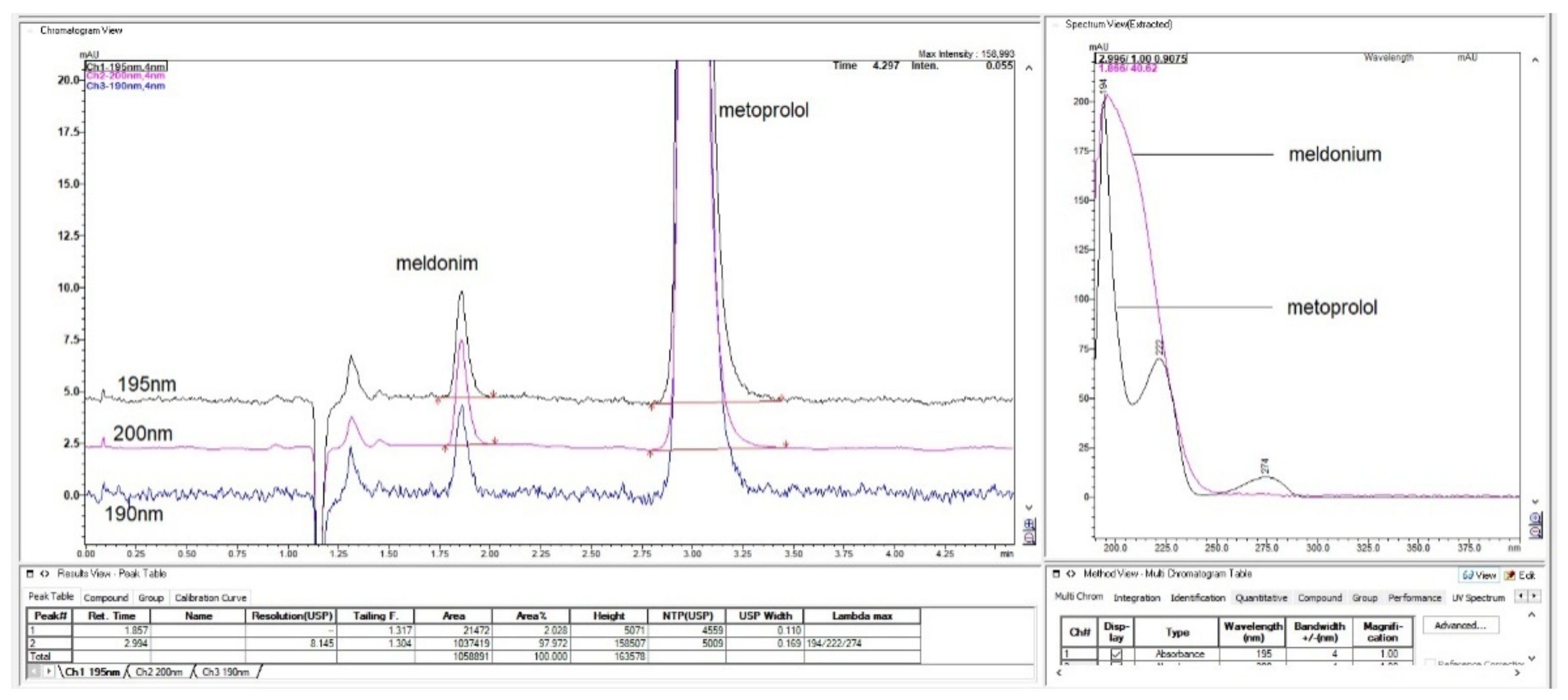
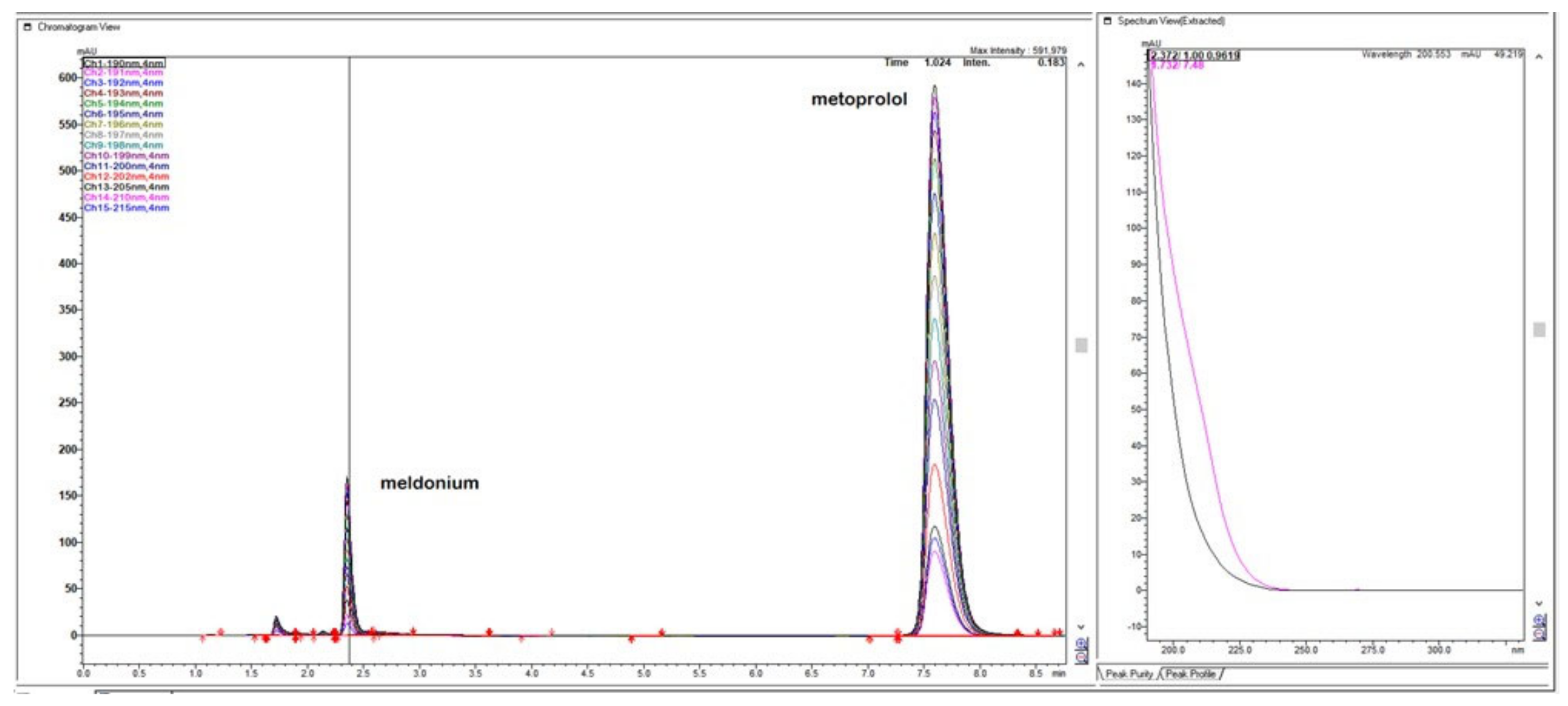
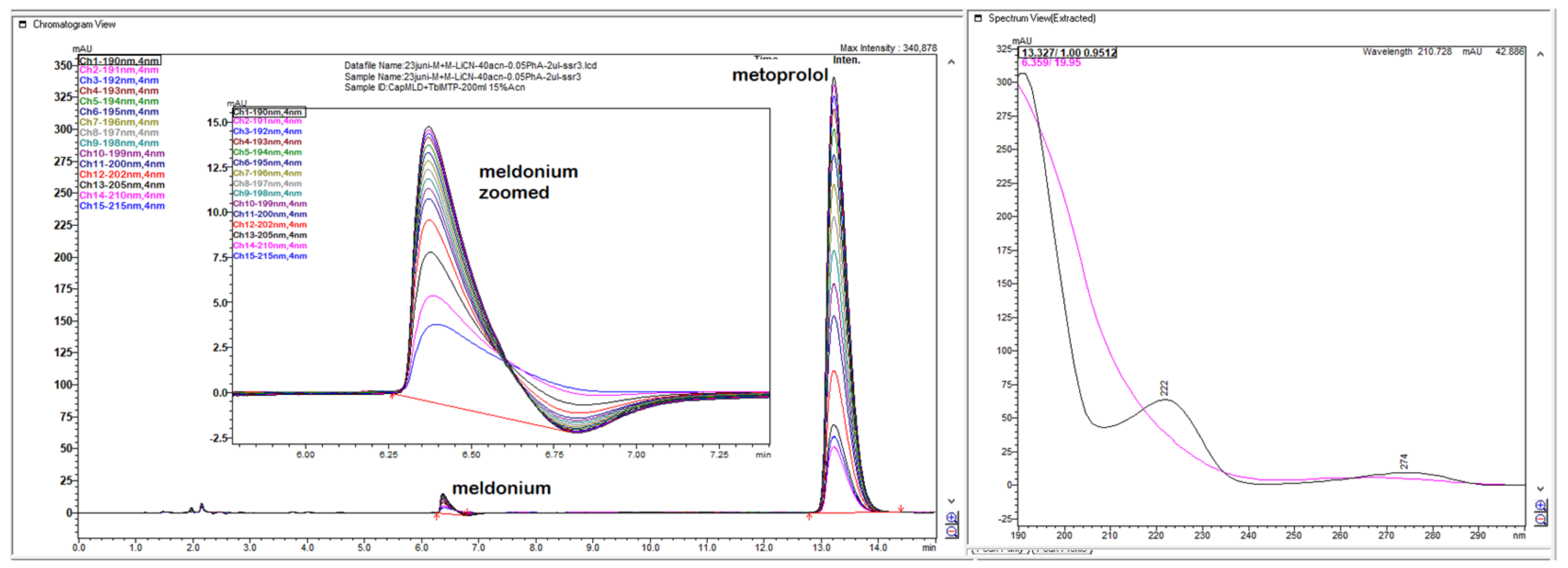
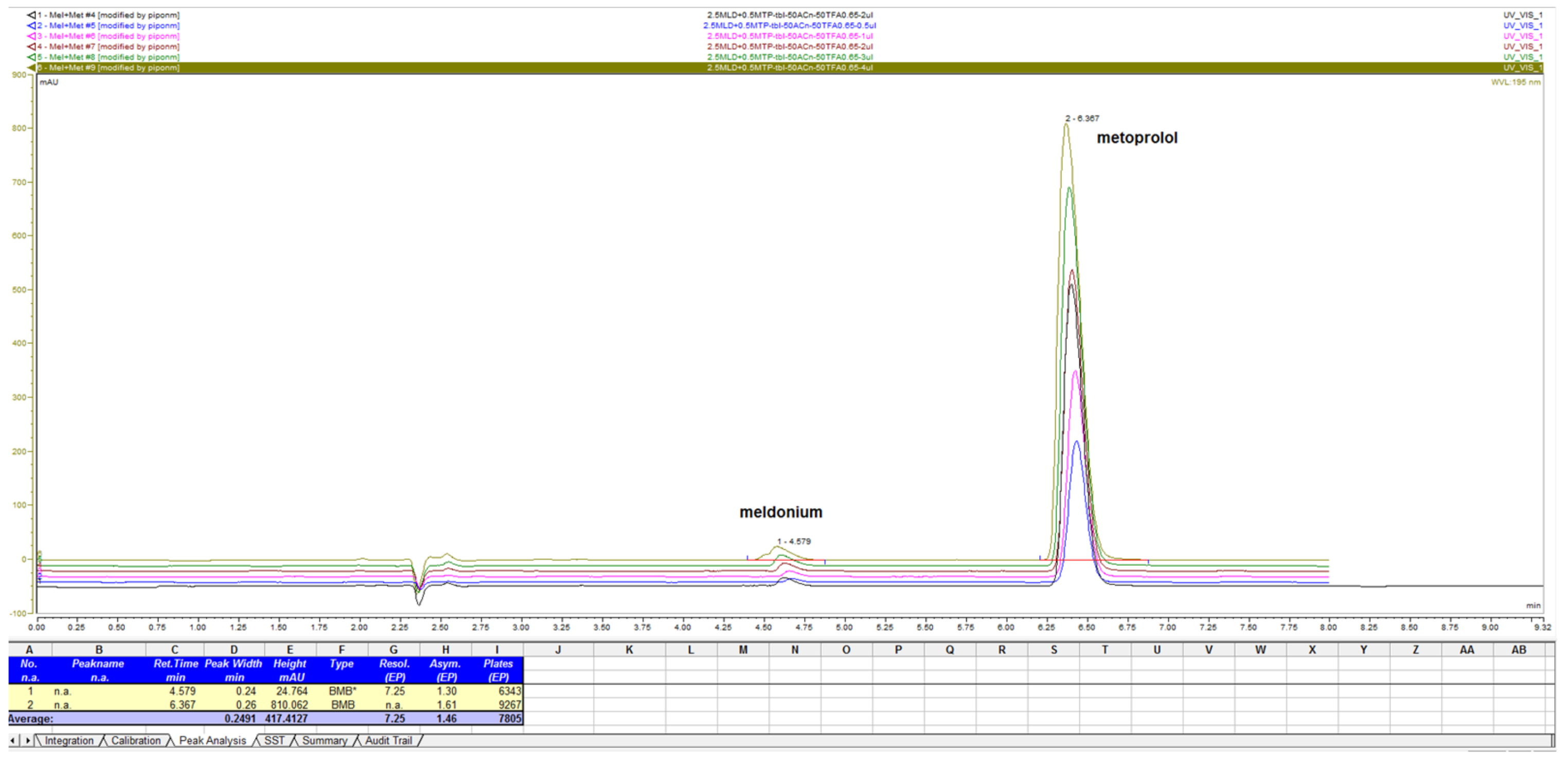
Figure 5.
Chromatogram with LiChrospher CN 125 × 4 mm, mobile phase 20% ACN and 80% 13 mM NH4H2PO4, monitored and overlaid from 15-channels overlaid, depending on peaks sizes with increments of 1 nm in UV-wavelength. Both analyte peaks were highly sensitive to the change of the wavelength, significantly, thus directly influencing the method of sensitivity for both API.
Figure 5.
Chromatogram with LiChrospher CN 125 × 4 mm, mobile phase 20% ACN and 80% 13 mM NH4H2PO4, monitored and overlaid from 15-channels overlaid, depending on peaks sizes with increments of 1 nm in UV-wavelength. Both analyte peaks were highly sensitive to the change of the wavelength, significantly, thus directly influencing the method of sensitivity for both API.
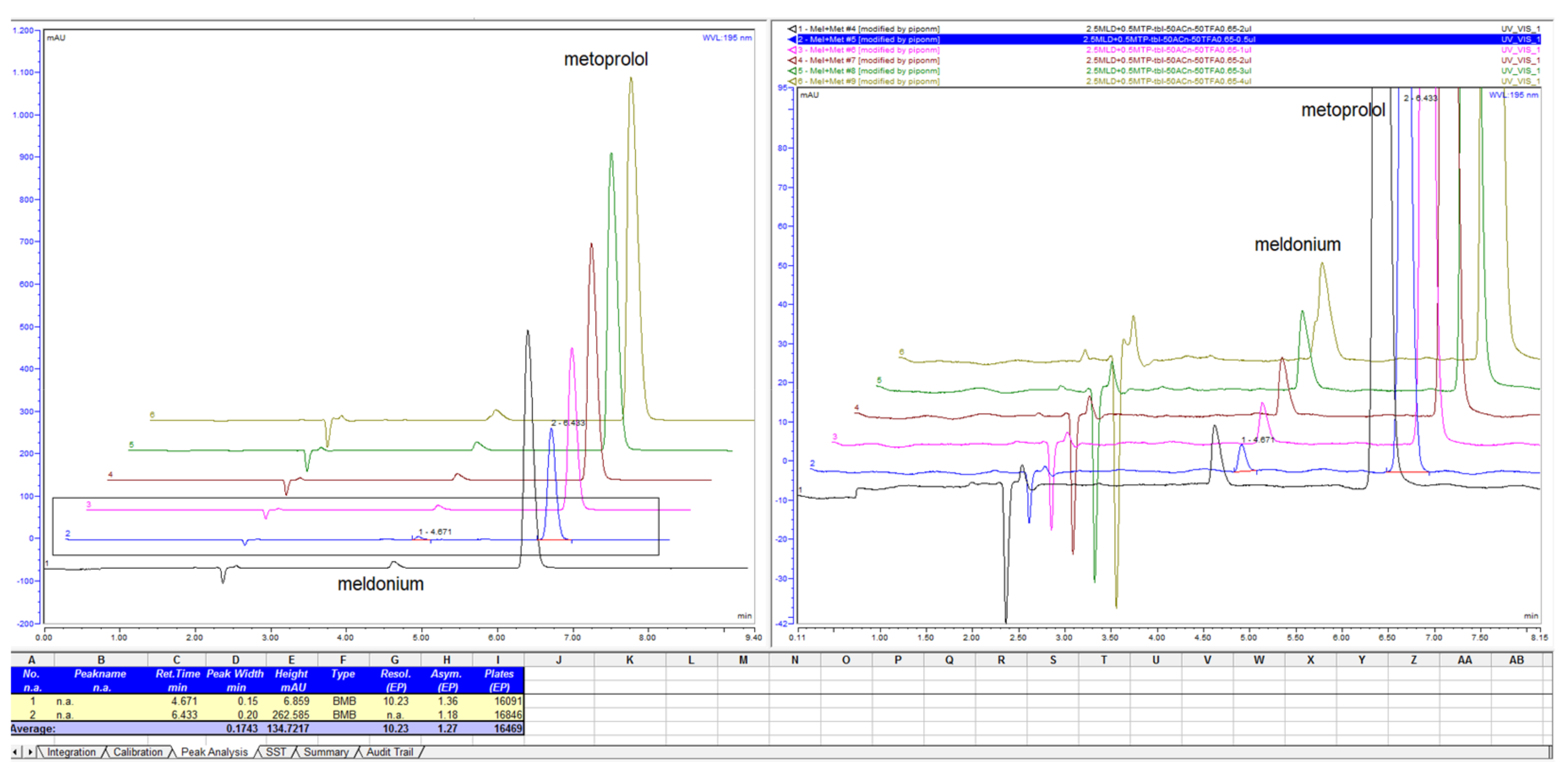
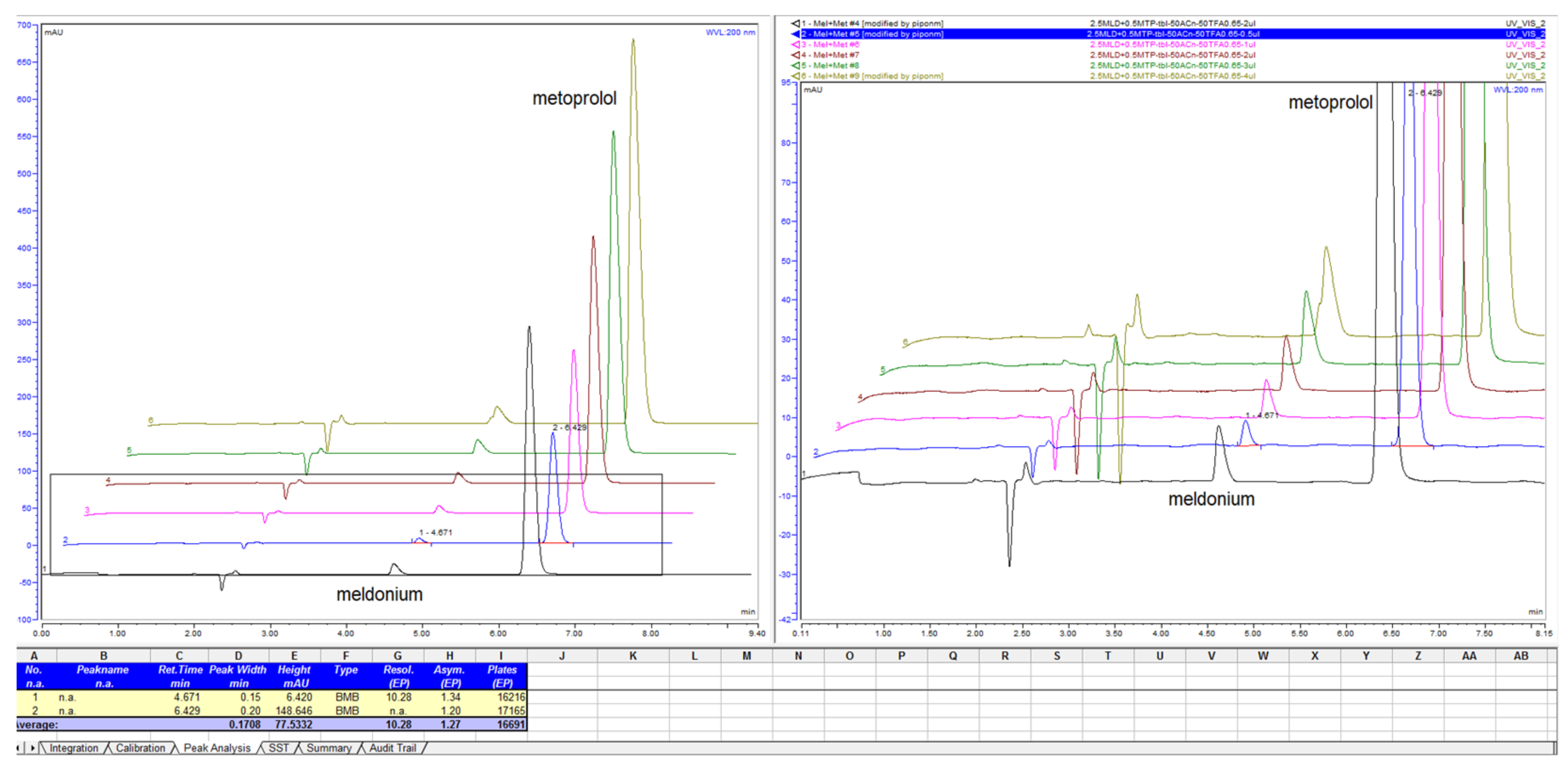
Figure 6.
Retention time of meldonium and metoprolol mapping with variations of ACN percentage in composition of mobile phase with 13 mM NH4H2PO4 with column LiChrospher CN 125 × 4 mm.
Figure 6.
Retention time of meldonium and metoprolol mapping with variations of ACN percentage in composition of mobile phase with 13 mM NH4H2PO4 with column LiChrospher CN 125 × 4 mm.
Figure 7.
Chromatogram with LiChrospher CN125 × 4 mm with 45% ACN and 55% TFA 0.065 %, visible good retentions of meldonium, quite small peak and quite smaller peak height increasing with the decreasing wavelength in the lowest UV-range. While the baseline was remarkably different, changes in meldonium peak size were quite small, and very big to the peak of metoprolol. Other visible change was meldonium UV-abs spectrum shape, while the metoprolol UV-spectrum was unchanged.
Figure 7.
Chromatogram with LiChrospher CN125 × 4 mm with 45% ACN and 55% TFA 0.065 %, visible good retentions of meldonium, quite small peak and quite smaller peak height increasing with the decreasing wavelength in the lowest UV-range. While the baseline was remarkably different, changes in meldonium peak size were quite small, and very big to the peak of metoprolol. Other visible change was meldonium UV-abs spectrum shape, while the metoprolol UV-spectrum was unchanged.
Figure 8.
Column LiChrospher CN 125 × 4 mm baseline noise dependence of wavelength with mobile phase ingredient diluted TFA. While the baseline was remarkably different, changes in meldonium peak were quite small, and no more concave shaped as with ammonium phosphate used in mobile phases.
Figure 8.
Column LiChrospher CN 125 × 4 mm baseline noise dependence of wavelength with mobile phase ingredient diluted TFA. While the baseline was remarkably different, changes in meldonium peak were quite small, and no more concave shaped as with ammonium phosphate used in mobile phases.
Figure 9.
Multi 15-channel chromatogram 250 × 4 mm LiCnrospher 100 CN with 20% ACN -13 mM NH4H2PO4. The two extracted peaks were from first eluted not-integrated small peak with tR about 1.7, with the characteristic meldonium concave Abs spectrum.
Figure 9.
Multi 15-channel chromatogram 250 × 4 mm LiCnrospher 100 CN with 20% ACN -13 mM NH4H2PO4. The two extracted peaks were from first eluted not-integrated small peak with tR about 1.7, with the characteristic meldonium concave Abs spectrum.
Figure 10.
Chromatogram of 250 × 4 mm LiChrospher CN column with 40% ACN and 60% 0.05% NH4H2PO4, with extracted spectrums of meldonium and metoprolol.
Figure 10.
Chromatogram of 250 × 4 mm LiChrospher CN column with 40% ACN and 60% 0.05% NH4H2PO4, with extracted spectrums of meldonium and metoprolol.
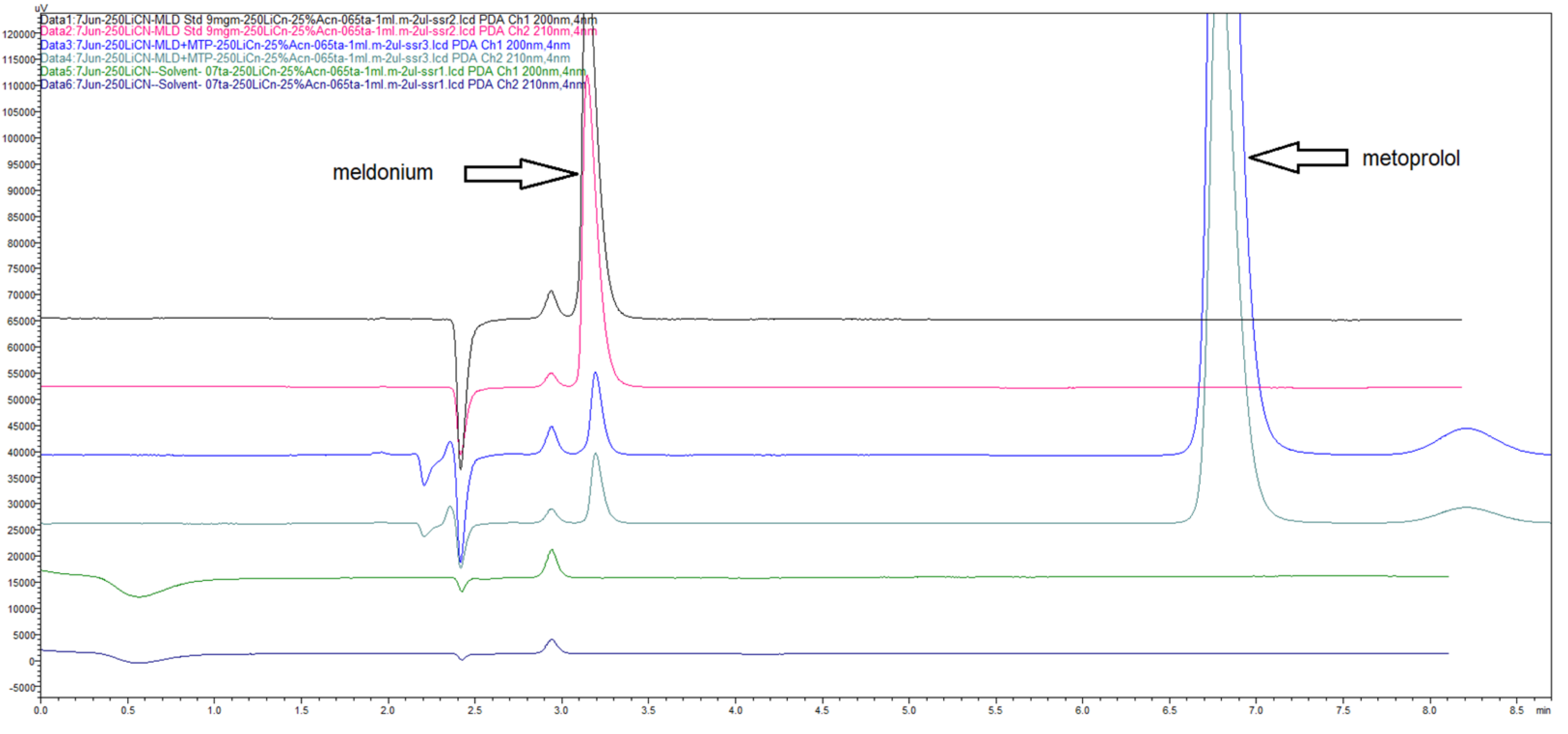
Figure 11.
Chromatograms confirming the selectivity of 250 × 4 mm LiChrospher CN column when 25% ACN with 75% diluted 0.065% v/v TFA was used at mobile phase, simultaneously compared at 2 wavelengths 200 nm and 210 nm.
Figure 11.
Chromatograms confirming the selectivity of 250 × 4 mm LiChrospher CN column when 25% ACN with 75% diluted 0.065% v/v TFA was used at mobile phase, simultaneously compared at 2 wavelengths 200 nm and 210 nm.
Figure 12.
Chromatogram of LiChrospher 250 × 4 mm, with 55% diluted TFA and 45 %ACN at 2 comparison wavelengths, 195 nm and 210 nm. Operational and monitoring parameters presented on the table on the right.
Figure 12.
Chromatogram of LiChrospher 250 × 4 mm, with 55% diluted TFA and 45 %ACN at 2 comparison wavelengths, 195 nm and 210 nm. Operational and monitoring parameters presented on the table on the right.
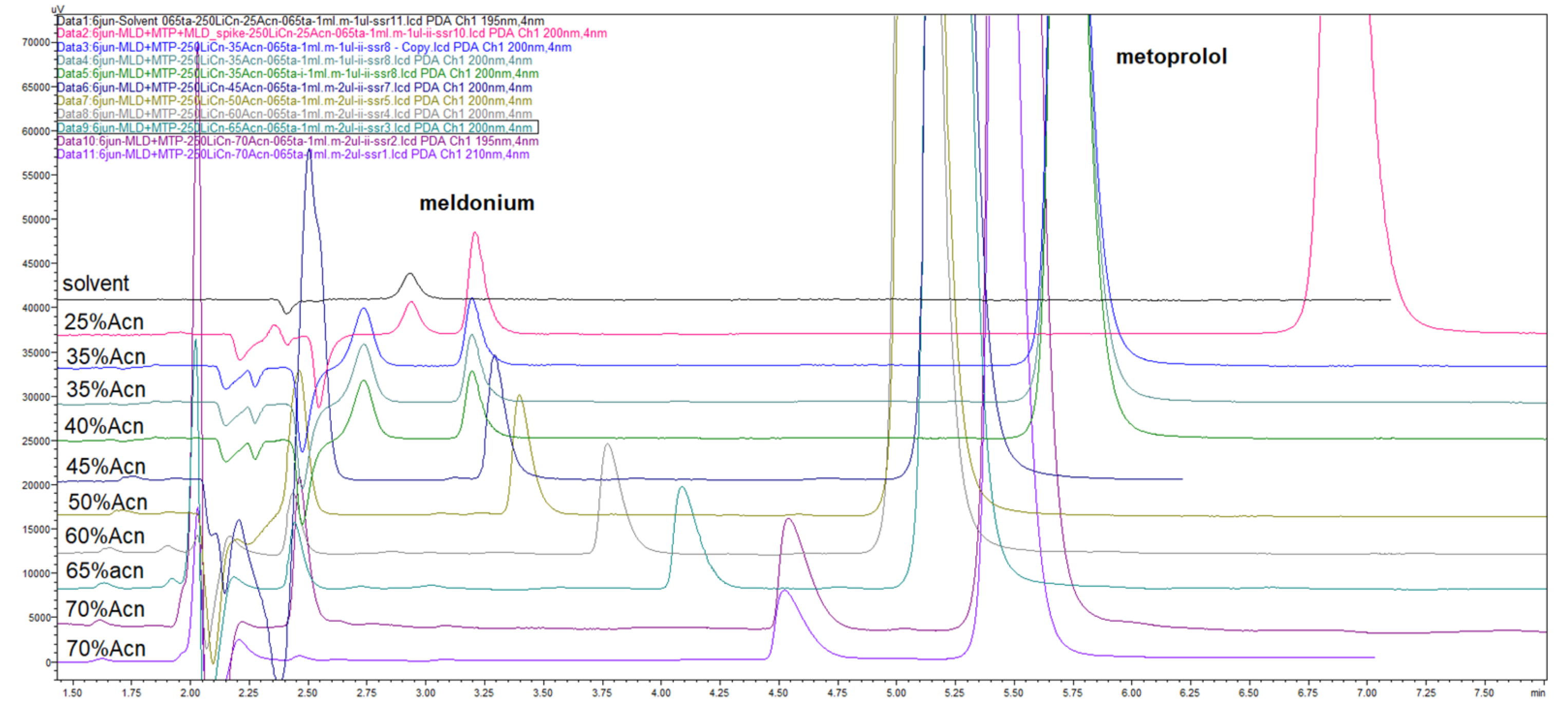
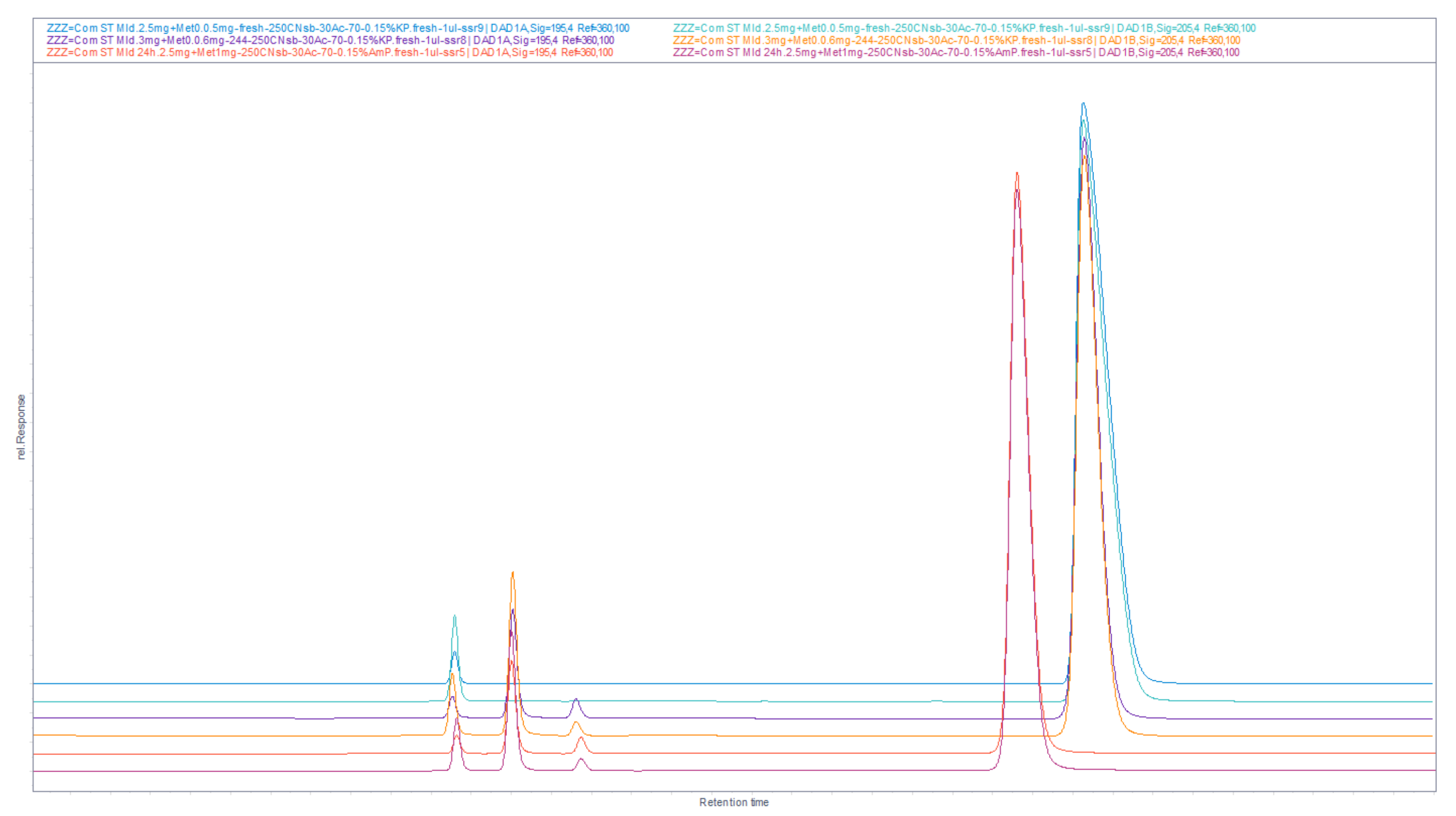
Figure 13.
Overlaid chromatograms of LiChrospher CN 250 × 4 mm column experiments, that were useful for rough and fine tuning of analyte peaks according to presence of other compounds. Peaks mapping as a function of acetonitrile percentage in eluents, in the range of 25–70% ACN.
Figure 13.
Overlaid chromatograms of LiChrospher CN 250 × 4 mm column experiments, that were useful for rough and fine tuning of analyte peaks according to presence of other compounds. Peaks mapping as a function of acetonitrile percentage in eluents, in the range of 25–70% ACN.
Figure 14.
Chromatogram using Waters Spherisob CNRP 250 × 4.6 mm 5 µm column with 50% ACN and 0.15% NH4H2PO4 in 50:50 ratio.
Figure 14.
Chromatogram using Waters Spherisob CNRP 250 × 4.6 mm 5 µm column with 50% ACN and 0.15% NH4H2PO4 in 50:50 ratio.
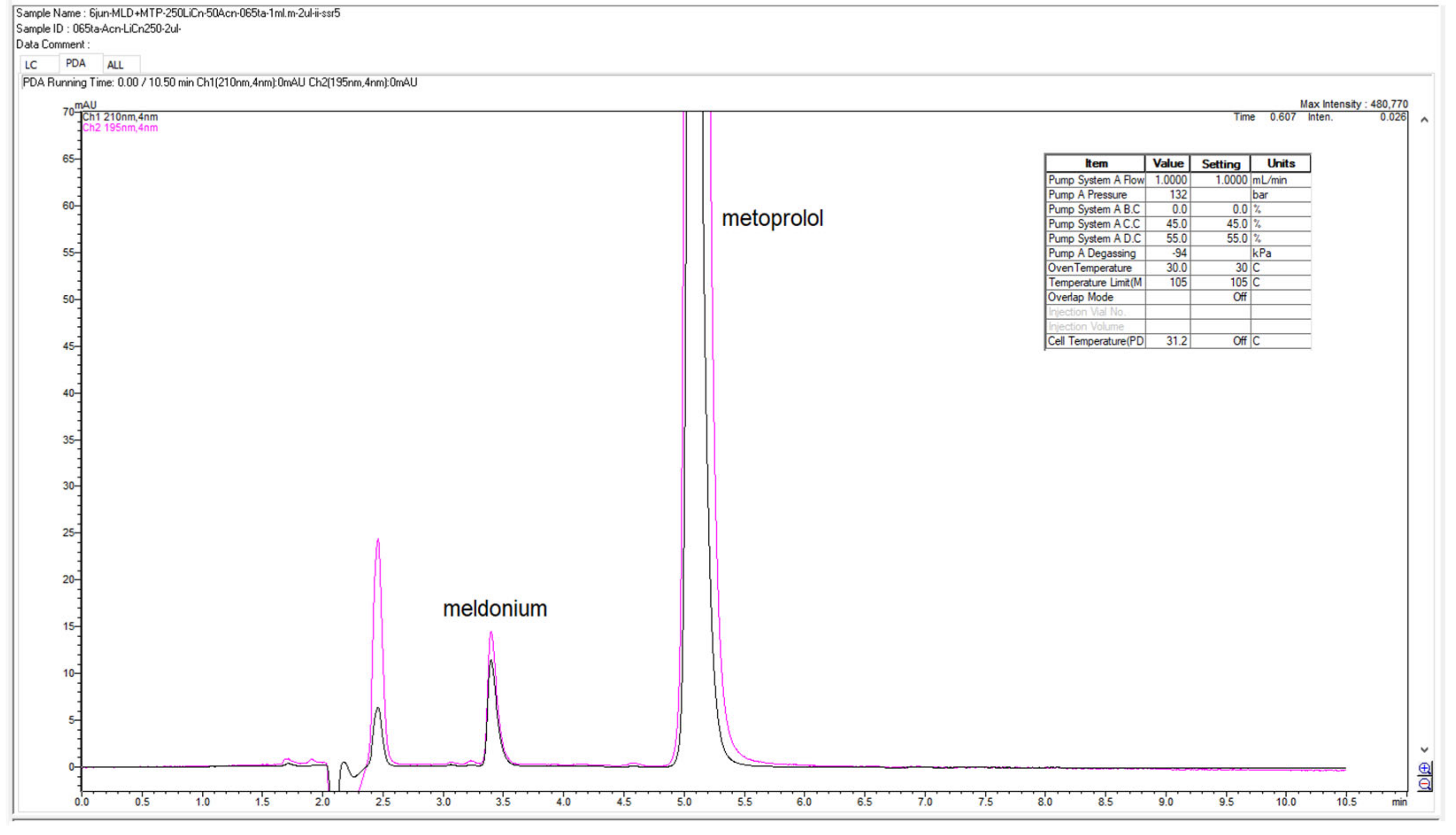
Figure 15.
Chromatograms present expanded view of meldonium peaks with system suitability parameters below in the table, monitored and recorded at 5 wavelengths.
Figure 15.
Chromatograms present expanded view of meldonium peaks with system suitability parameters below in the table, monitored and recorded at 5 wavelengths.
Figure 16.
Chromatogram of sample 2.5 mg/mL meldonium and 0.5 mg/mL metoprolol with Water Spherisorb CNRP-250 × 4.6 mm eluted 0.15% NH4H2PO4 + 50% ACN meldonium expended view.
Figure 16.
Chromatogram of sample 2.5 mg/mL meldonium and 0.5 mg/mL metoprolol with Water Spherisorb CNRP-250 × 4.6 mm eluted 0.15% NH4H2PO4 + 50% ACN meldonium expended view.
Figure 17.
Full view-chromatogram of sample 2.5 mg/mL meldonium and 0.5 mg/mL metoprolol with Waters Spherisorb CNRP-250 × 4.6 mm eluted 0.15% NH4H2PO4 + 50% ACN full chromatogram view.
Figure 17.
Full view-chromatogram of sample 2.5 mg/mL meldonium and 0.5 mg/mL metoprolol with Waters Spherisorb CNRP-250 × 4.6 mm eluted 0.15% NH4H2PO4 + 50% ACN full chromatogram view.
Figure 18.
Chromatogram of sample 2.5 mg/mL meldonium and 0.5 mg/mL metoprolol with Waters Spherisorb CNRP-250 × 4.6 mm eluted 0.15% NH4H2PO4 + 70% ACN full view.
Figure 18.
Chromatogram of sample 2.5 mg/mL meldonium and 0.5 mg/mL metoprolol with Waters Spherisorb CNRP-250 × 4.6 mm eluted 0.15% NH4H2PO4 + 70% ACN full view.
Figure 19.
Chromatogram with 50% ACN and 50% diluted TFA with full size chromatogram below, monitored and overlaid at 4-channels with 4 different wavelengths.
Figure 19.
Chromatogram with 50% ACN and 50% diluted TFA with full size chromatogram below, monitored and overlaid at 4-channels with 4 different wavelengths.
Figure 20.
Chromatogram with 50% ACN and 50% diluted TFA, with full size chromatogram below and inserted expanded view of meldonium peak in zoomed scale above, monitored and overlaid at 4-channels with 4 different wavelengths. The right side of figure contains extracted UV spectrums of meldonium and metoprolol. The system suitability of chromatogram can be seen in the table below. The bottom figure illustrates mobile phase noises from upper chromatograms.
Figure 20.
Chromatogram with 50% ACN and 50% diluted TFA, with full size chromatogram below and inserted expanded view of meldonium peak in zoomed scale above, monitored and overlaid at 4-channels with 4 different wavelengths. The right side of figure contains extracted UV spectrums of meldonium and metoprolol. The system suitability of chromatogram can be seen in the table below. The bottom figure illustrates mobile phase noises from upper chromatograms.
Figure 21.
4-channel multichannel view of full chromatograms with 50% ACN and 50% diluted TFA of samples with Waters Spherisorb CNRP-250 × 4.6 mm.
Figure 21.
4-channel multichannel view of full chromatograms with 50% ACN and 50% diluted TFA of samples with Waters Spherisorb CNRP-250 × 4.6 mm.
Figure 22.
Dionex-Full scale overlaid chromatograms linearity of method with Waters Spherisorb CNRP-250 × 4.6 mm and 50% ACN and 50% diluted TFA at 195 nm, with system suitability parameters in table below.
Figure 22.
Dionex-Full scale overlaid chromatograms linearity of method with Waters Spherisorb CNRP-250 × 4.6 mm and 50% ACN and 50% diluted TFA at 195 nm, with system suitability parameters in table below.
Figure 23.
Dionex-full scale overlaid and expanded zoomed Y-axis of overlaid chromatograms for linearity of method with Waters Spherisorb CNRP-250 × 4.6 mm and 50% ACN and 50% diluted TFA at 195 nm, with system suitability parameters in table below.
Figure 23.
Dionex-full scale overlaid and expanded zoomed Y-axis of overlaid chromatograms for linearity of method with Waters Spherisorb CNRP-250 × 4.6 mm and 50% ACN and 50% diluted TFA at 195 nm, with system suitability parameters in table below.
Figure 24.
Dionex full-scale overlaid and expanded zoomed Y-axis of overlaid chromatograms for the linearity of the method with Waters Spherisorb CNRP-250 × 4.6 mm and 50%ACN and 50% diluted TFA at 200 nm, with system suitability parameters in the table.
Figure 24.
Dionex full-scale overlaid and expanded zoomed Y-axis of overlaid chromatograms for the linearity of the method with Waters Spherisorb CNRP-250 × 4.6 mm and 50%ACN and 50% diluted TFA at 200 nm, with system suitability parameters in the table.
Figure 25.
Overview of the retention map, testing Waters Spherisorb CNRP-250 × 4.6 mm, a final orientational overview of the behavior of these columns with different mobile phases, with the same organic solvent ACN but with different modifiers, can be seen and analyzed on the overlaid chromatograms.
Figure 25.
Overview of the retention map, testing Waters Spherisorb CNRP-250 × 4.6 mm, a final orientational overview of the behavior of these columns with different mobile phases, with the same organic solvent ACN but with different modifiers, can be seen and analyzed on the overlaid chromatograms.
Figure 26.
Chromatograms testing of column Zorbax CN-SB-250 × 4.6 mm with 20% ACN and 80% 13 mM (0.15% w/v) NH4H2PO4 and overlaid 15-channels of different UV-absorbing wavelengths on the left, extracted spectrum on right.
Figure 26.
Chromatograms testing of column Zorbax CN-SB-250 × 4.6 mm with 20% ACN and 80% 13 mM (0.15% w/v) NH4H2PO4 and overlaid 15-channels of different UV-absorbing wavelengths on the left, extracted spectrum on right.
Figure 27.
Chromatograms with column Zorbax CN-SB-250 × 4.6 mm with 20% ACN and 80% 13 mM (0.15% w/v) NH4H2PO4 and overlaid 15-channels of different UV-absorbing wavelengths, with an expanded, zoomed peak of meldonium for better visibility in comparison with metoprolol.
Figure 27.
Chromatograms with column Zorbax CN-SB-250 × 4.6 mm with 20% ACN and 80% 13 mM (0.15% w/v) NH4H2PO4 and overlaid 15-channels of different UV-absorbing wavelengths, with an expanded, zoomed peak of meldonium for better visibility in comparison with metoprolol.
Figure 28.
Chromatograms with Zorbax CN-SB-250 × 4.6 mm with 50% ACN and 50% 13 mM (0.15% w/v) NH4H2PO4 and overlaid 15-channels of different UV-absorbing wavelengths.
Figure 28.
Chromatograms with Zorbax CN-SB-250 × 4.6 mm with 50% ACN and 50% 13 mM (0.15% w/v) NH4H2PO4 and overlaid 15-channels of different UV-absorbing wavelengths.
Figure 29.
Chromatograms with Zorbax CN-SB-250 × 4.6 mm with 50% ACN and 50% 13 mM (0.15% w/v) NH4H2PO4 and overlaid 15-channels of different UV-absorbing wavelengths, with expanded zoomed peak of meldonium inserted for better comparison with metoprolol peak.
Figure 29.
Chromatograms with Zorbax CN-SB-250 × 4.6 mm with 50% ACN and 50% 13 mM (0.15% w/v) NH4H2PO4 and overlaid 15-channels of different UV-absorbing wavelengths, with expanded zoomed peak of meldonium inserted for better comparison with metoprolol peak.
Figure 30.
Chromatograms with Zorbax CN-SB-250 × 4.6 mm, gained with organic solvent ACN in mobile phase to 60% with 40% 13 mM NH4H2PO4.
Figure 30.
Chromatograms with Zorbax CN-SB-250 × 4.6 mm, gained with organic solvent ACN in mobile phase to 60% with 40% 13 mM NH4H2PO4.
Figure 31.
Chromatograms with Zorbax CN-SB-250 × 4.6 mm retention mapping of analytes as a function of eluate composition and pH, the overlay of mobile phase changes experiments for chromatogram shaping results, in trials with mobile phase composed of ACN,13 mM NH4H2PO4, and 0.08% v/v o-H3PO4.
Figure 31.
Chromatograms with Zorbax CN-SB-250 × 4.6 mm retention mapping of analytes as a function of eluate composition and pH, the overlay of mobile phase changes experiments for chromatogram shaping results, in trials with mobile phase composed of ACN,13 mM NH4H2PO4, and 0.08% v/v o-H3PO4.
Figure 32.
Full 3-D chromatogram of meldonium, extracted line at 197 nm and UV-spectrum of analytes with 50% ACN and 50% 13 mM NH4H2PO4 using column Zorbax CN-SB-250 × 4.6 mm.
Figure 32.
Full 3-D chromatogram of meldonium, extracted line at 197 nm and UV-spectrum of analytes with 50% ACN and 50% 13 mM NH4H2PO4 using column Zorbax CN-SB-250 × 4.6 mm.
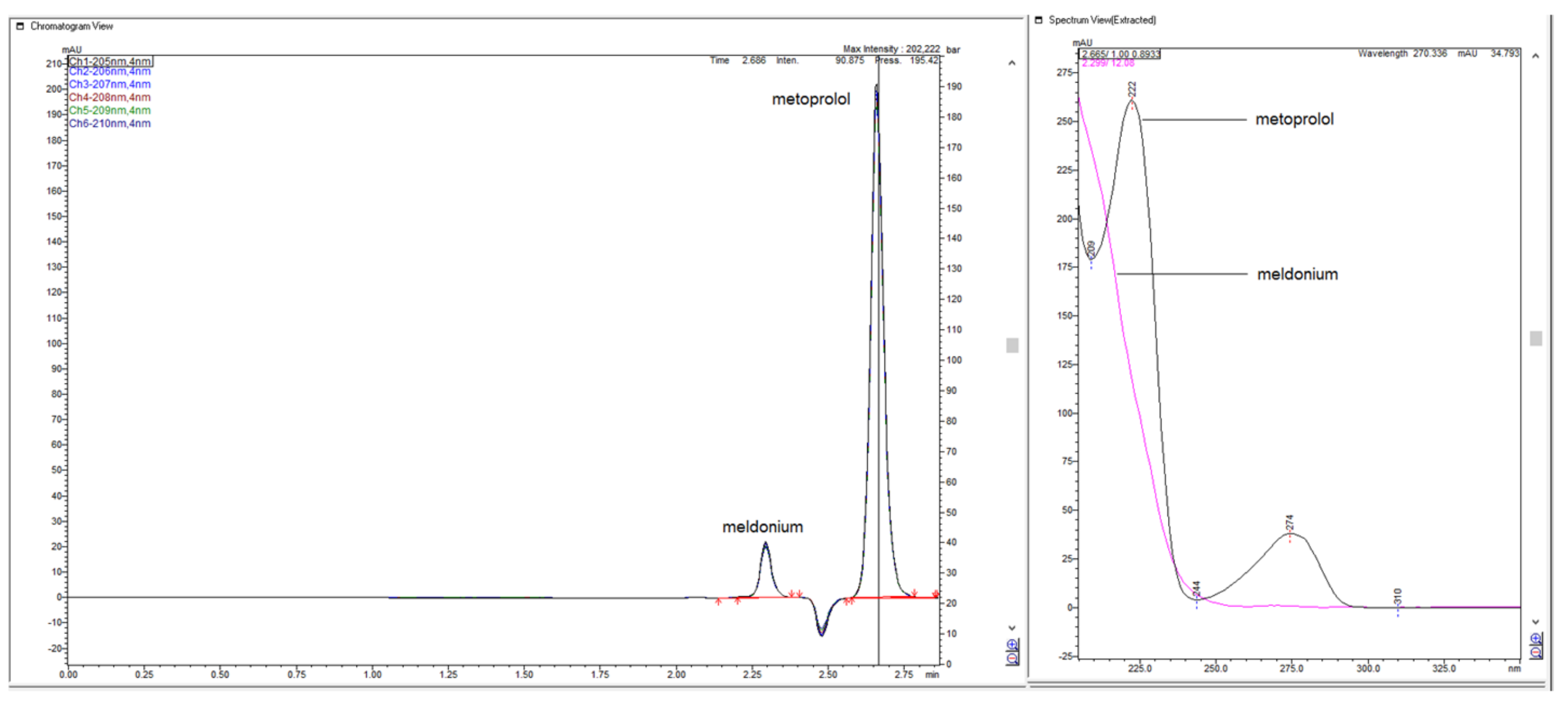
Figure 33.
Chromatograms obtained with Zorbax CN-SB-250 × 4.6 mm in the term of confirmation of identities of meldonium and metoprolol with standard 50% ACN and 50% NH4H2PO4.
Figure 33.
Chromatograms obtained with Zorbax CN-SB-250 × 4.6 mm in the term of confirmation of identities of meldonium and metoprolol with standard 50% ACN and 50% NH4H2PO4.
Figure 34.
Overlaid chromatograms of meldonium standard pure, standard meldonium contaminated with trace of metoprolol and sample with 2.5 mg/mL meldonium and 0.5 mg/mL metoprolol with ACN and 13 mM NH4H2PO4 using column Zorbax CN-SB-250 × 4.6 mm.
Figure 34.
Overlaid chromatograms of meldonium standard pure, standard meldonium contaminated with trace of metoprolol and sample with 2.5 mg/mL meldonium and 0.5 mg/mL metoprolol with ACN and 13 mM NH4H2PO4 using column Zorbax CN-SB-250 × 4.6 mm.
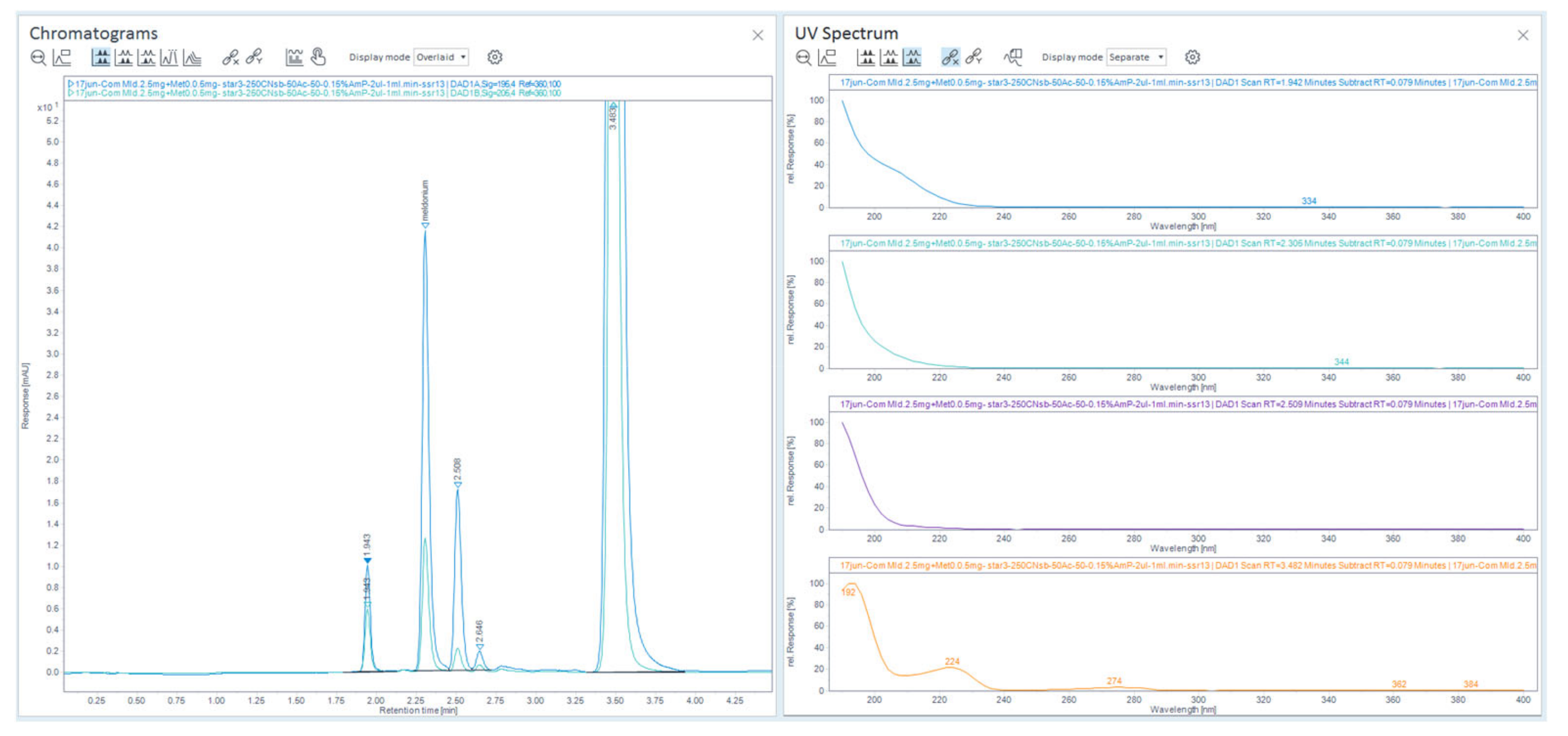
Figure 35.
Chromatograms of retention time mapping with significant changes of organic percentage of ACN with 13 mM NH4H2PO4 in mobile phase, comparison of overlaid retentions of analytes on chromatograms using column Zorbax CN-SB-250 × 4.6 mm.
Figure 35.
Chromatograms of retention time mapping with significant changes of organic percentage of ACN with 13 mM NH4H2PO4 in mobile phase, comparison of overlaid retentions of analytes on chromatograms using column Zorbax CN-SB-250 × 4.6 mm.
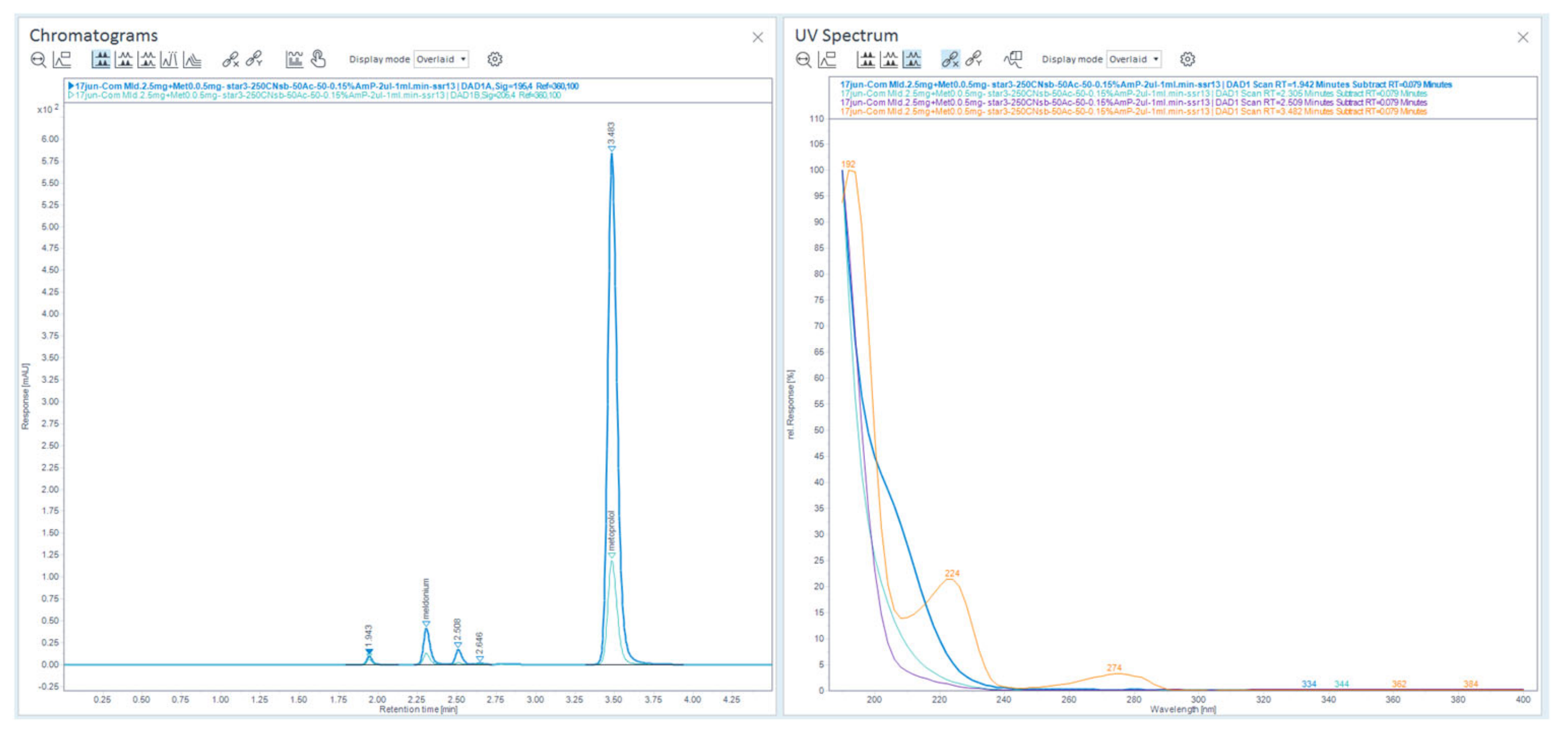
Figure 36.
Chromatograms, eluted with 50% ACN and 50 % 13 mM NH4H2PO4, were recorded and overlaid at 2-channels of 195 nm and 205 nm overlaid are on the left and their characteristic UV-spectrums on the right. The second peak is meldonium on chromatogram, and the last highest peak was metoprolol, third and fourth peaks were new appeared. From the 4 presented UV-Abs spectrums, the second from the top was the meldonium peak extracted and fourth at the bottom was of metoprolol.
Figure 36.
Chromatograms, eluted with 50% ACN and 50 % 13 mM NH4H2PO4, were recorded and overlaid at 2-channels of 195 nm and 205 nm overlaid are on the left and their characteristic UV-spectrums on the right. The second peak is meldonium on chromatogram, and the last highest peak was metoprolol, third and fourth peaks were new appeared. From the 4 presented UV-Abs spectrums, the second from the top was the meldonium peak extracted and fourth at the bottom was of metoprolol.
Figure 37.
Chromatograms, obtained by usage mobile phase composed of 50% ACN and 50% 13 mM NH4H2PO4, the most effective separation was achieved in less than a 4 min run time. The second peak was meldonium on chromatogram, and the last highest peak was metoprolol, the third and fourth peaks were new.
Figure 37.
Chromatograms, obtained by usage mobile phase composed of 50% ACN and 50% 13 mM NH4H2PO4, the most effective separation was achieved in less than a 4 min run time. The second peak was meldonium on chromatogram, and the last highest peak was metoprolol, the third and fourth peaks were new.
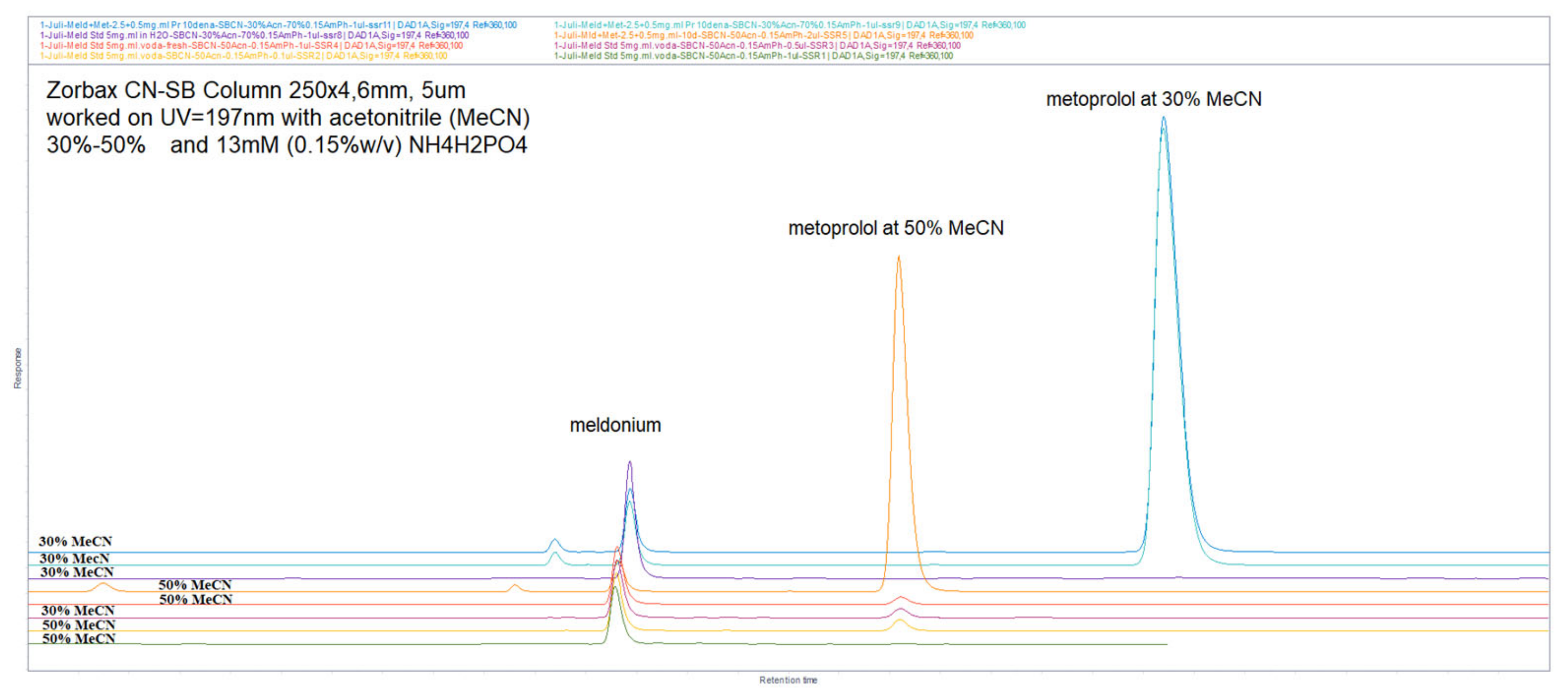
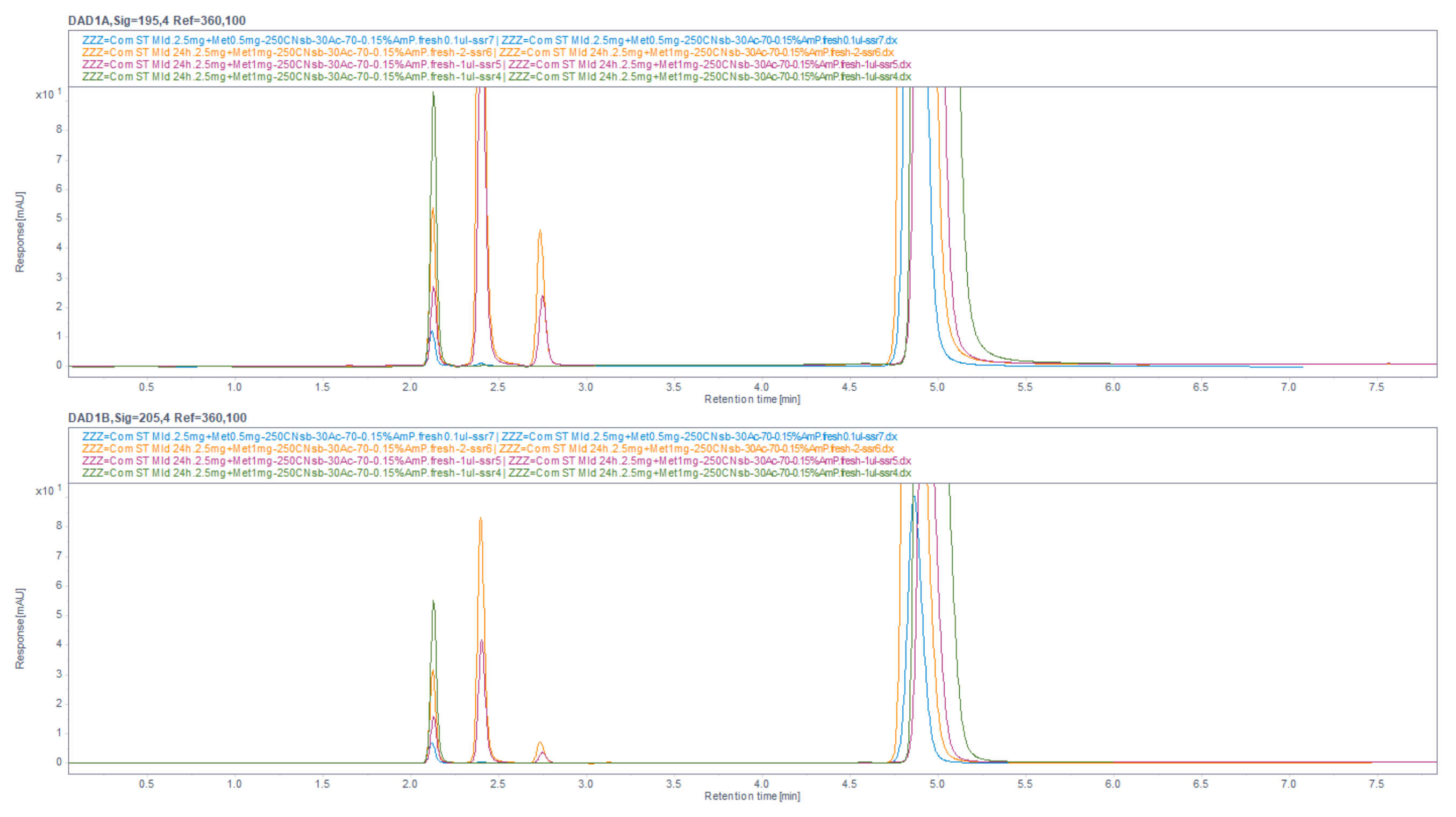
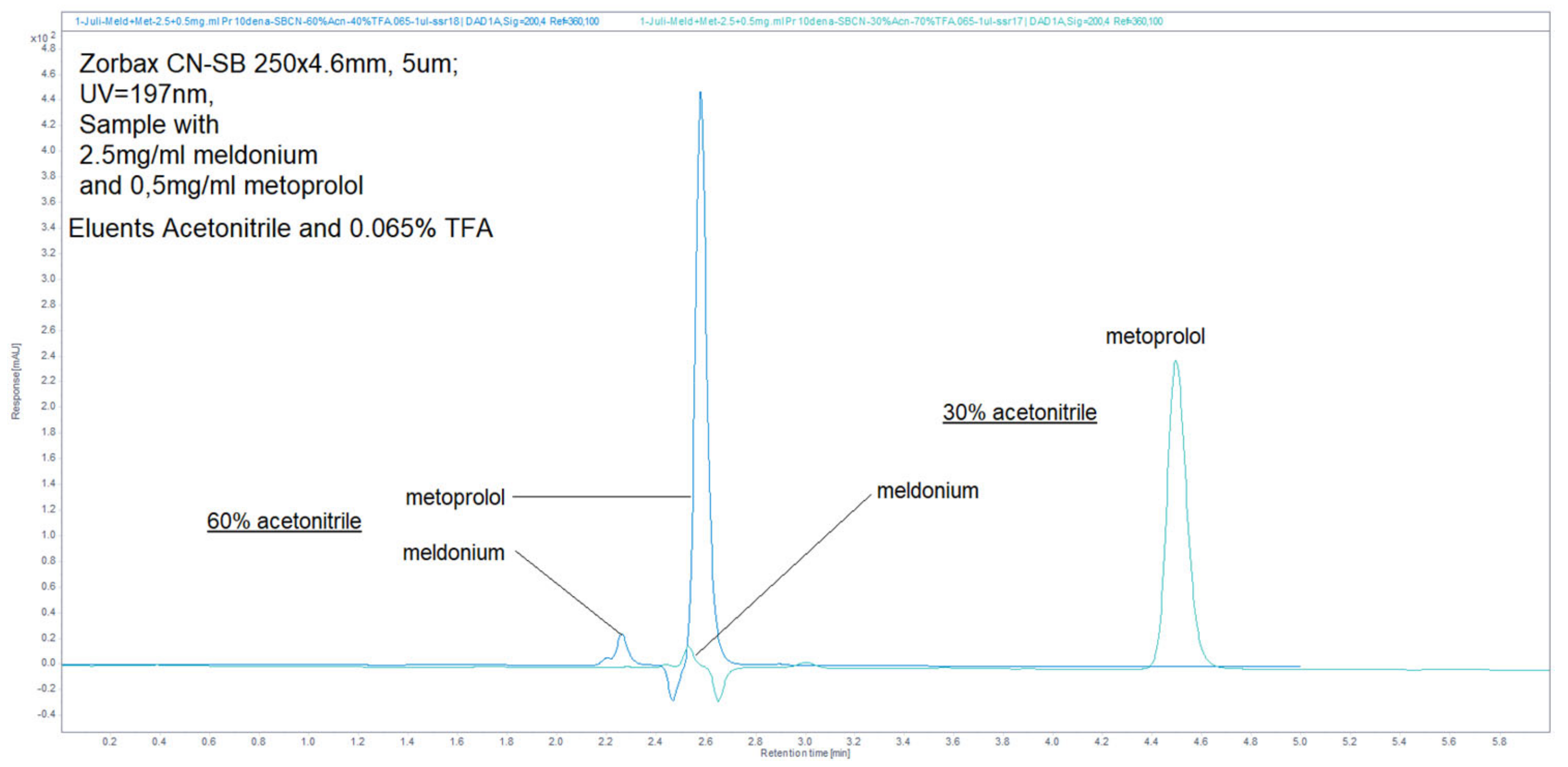
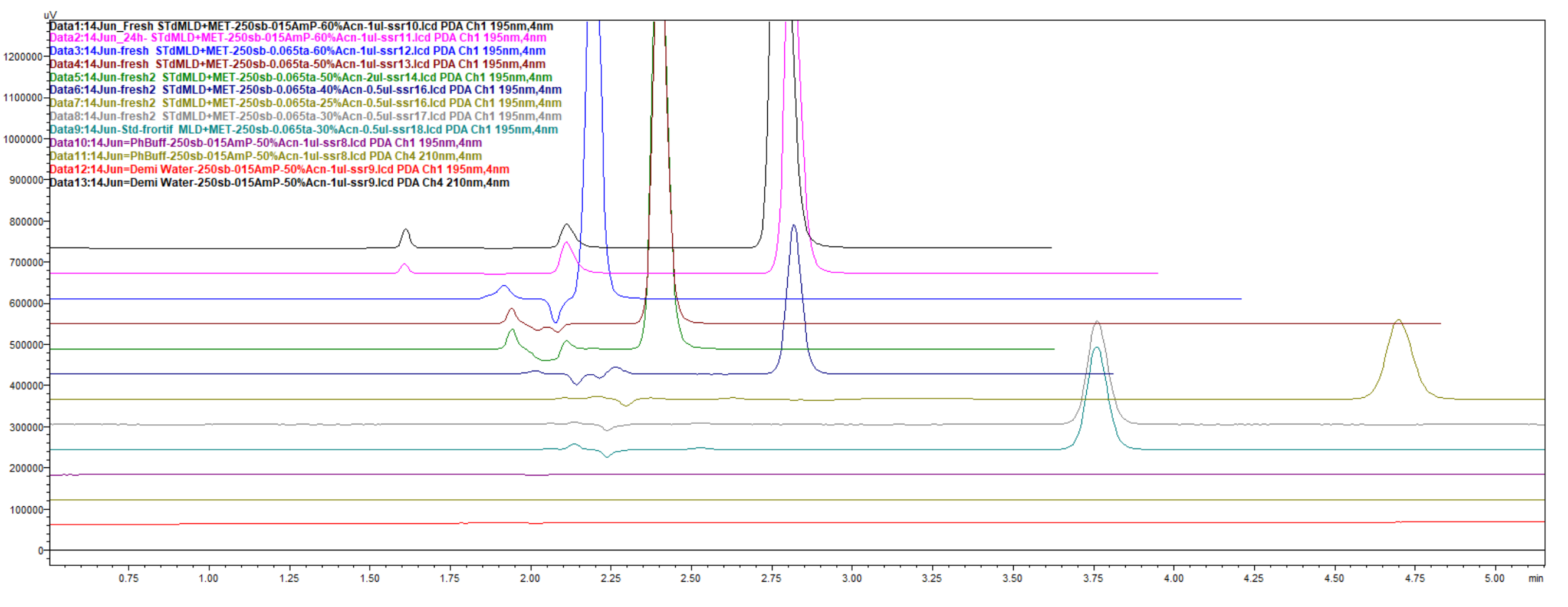
Figure 38.
Chromatograms, obtained by usage mobile phase composed of 30% ACN and 70% 13 mM NH4H2PO4, and achieved better resolutions function of increased retention, and overall run time of about 5 min, but significant peaks increased resolution. Overlaid 2 different situations of samples, freshly prepared and incubated for 3 days at room conditions. The first peak was meldonium, the second and third were degradation products, and the last one was metoprolol. The sizes of the peaks were different due to increasing injection volumes.
Figure 38.
Chromatograms, obtained by usage mobile phase composed of 30% ACN and 70% 13 mM NH4H2PO4, and achieved better resolutions function of increased retention, and overall run time of about 5 min, but significant peaks increased resolution. Overlaid 2 different situations of samples, freshly prepared and incubated for 3 days at room conditions. The first peak was meldonium, the second and third were degradation products, and the last one was metoprolol. The sizes of the peaks were different due to increasing injection volumes.
Figure 39.
Chromatograms, obtained by usage ACN content to 50% (upper chromatogram) and with 30% ACN (bottom chromatogram) with of 13 mM NH4H2PO4. Chromatograms were presented in full and expanded overlaid sizes, with UV-abs spectrum extracted at right.
Figure 39.
Chromatograms, obtained by usage ACN content to 50% (upper chromatogram) and with 30% ACN (bottom chromatogram) with of 13 mM NH4H2PO4. Chromatograms were presented in full and expanded overlaid sizes, with UV-abs spectrum extracted at right.
Figure 40.
Chromatograms, obtained by usage of the ACN content to 30% with 70% 13 mM NH4H2PO4 the last at bottom two chromatograms, and replaced with 13 mM KH2PO4 above them. The last top chromatograms were made from a fresh sample prepared with 13 mM KH2PO4 70% and 30% ACN, with noticeable width spread and small retention shift of metoprolol peak.
Figure 40.
Chromatograms, obtained by usage of the ACN content to 30% with 70% 13 mM NH4H2PO4 the last at bottom two chromatograms, and replaced with 13 mM KH2PO4 above them. The last top chromatograms were made from a fresh sample prepared with 13 mM KH2PO4 70% and 30% ACN, with noticeable width spread and small retention shift of metoprolol peak.
Figure 41.
Chromatograms, performed with diluted acids, without use of phosphate salts. The first choice was 0.1% v/v o-H3PO4 80% and 20% ACN, recorded and overlaid at 13-different UV-wavelengths N- 20%ACN-80% 0.1% o-H3PO4. Distorted peaks with leading of meldonium and tailing of metoprolol were, for the first time, clearly visible.
Figure 41.
Chromatograms, performed with diluted acids, without use of phosphate salts. The first choice was 0.1% v/v o-H3PO4 80% and 20% ACN, recorded and overlaid at 13-different UV-wavelengths N- 20%ACN-80% 0.1% o-H3PO4. Distorted peaks with leading of meldonium and tailing of metoprolol were, for the first time, clearly visible.
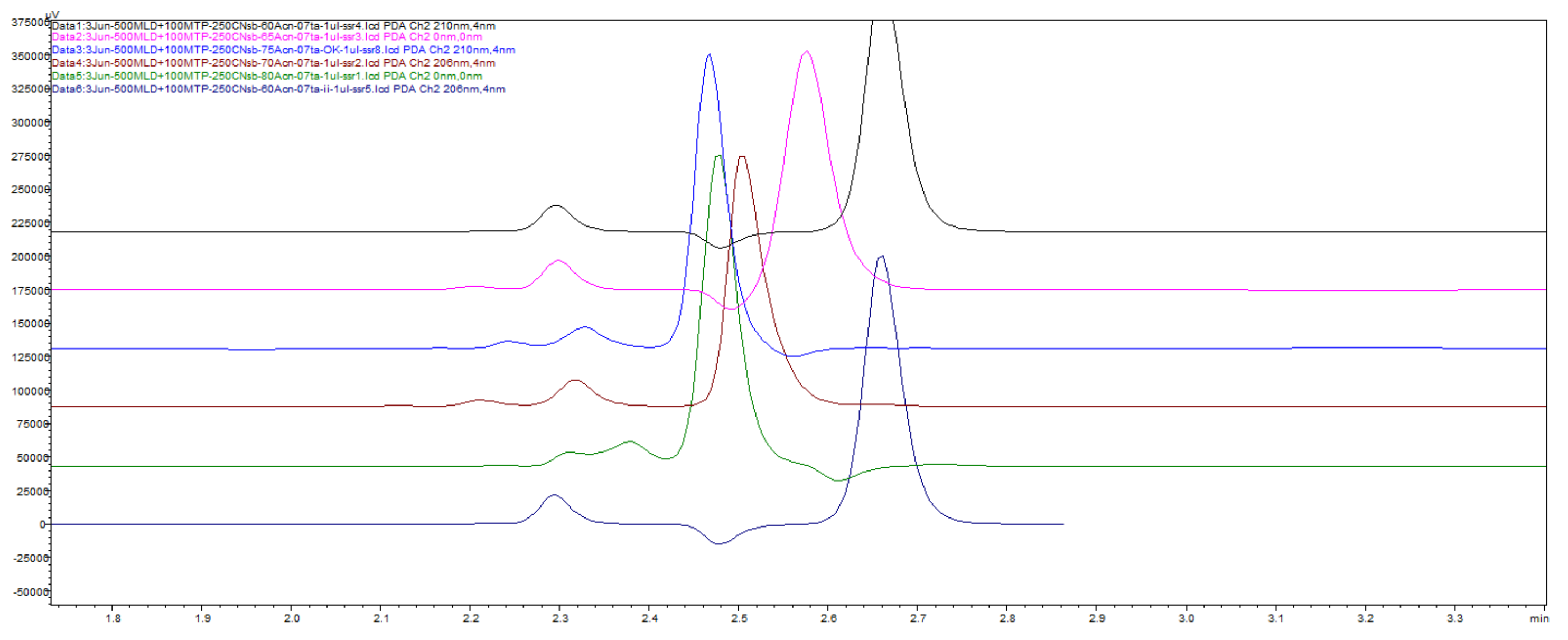
Figure 42.
Chromatograms with TFA, a function of changes of percentage of ACN in 60–65–70–75–80% ACN, creating map of meldonium and metoprolol peak elution movement across the X-scale 60–80% increment of ACN.
Figure 42.
Chromatograms with TFA, a function of changes of percentage of ACN in 60–65–70–75–80% ACN, creating map of meldonium and metoprolol peak elution movement across the X-scale 60–80% increment of ACN.
Figure 43.
Chromatograms, obtained by usage mobile phase composed of 70% ACN and 30% 0.07% v/v TFA, with exceptionally short run times lasting less than 3 min, with perfect shaped all peaks, but with no impressible resolution between them. UV spectrum extracted peaks on the right.
Figure 43.
Chromatograms, obtained by usage mobile phase composed of 70% ACN and 30% 0.07% v/v TFA, with exceptionally short run times lasting less than 3 min, with perfect shaped all peaks, but with no impressible resolution between them. UV spectrum extracted peaks on the right.
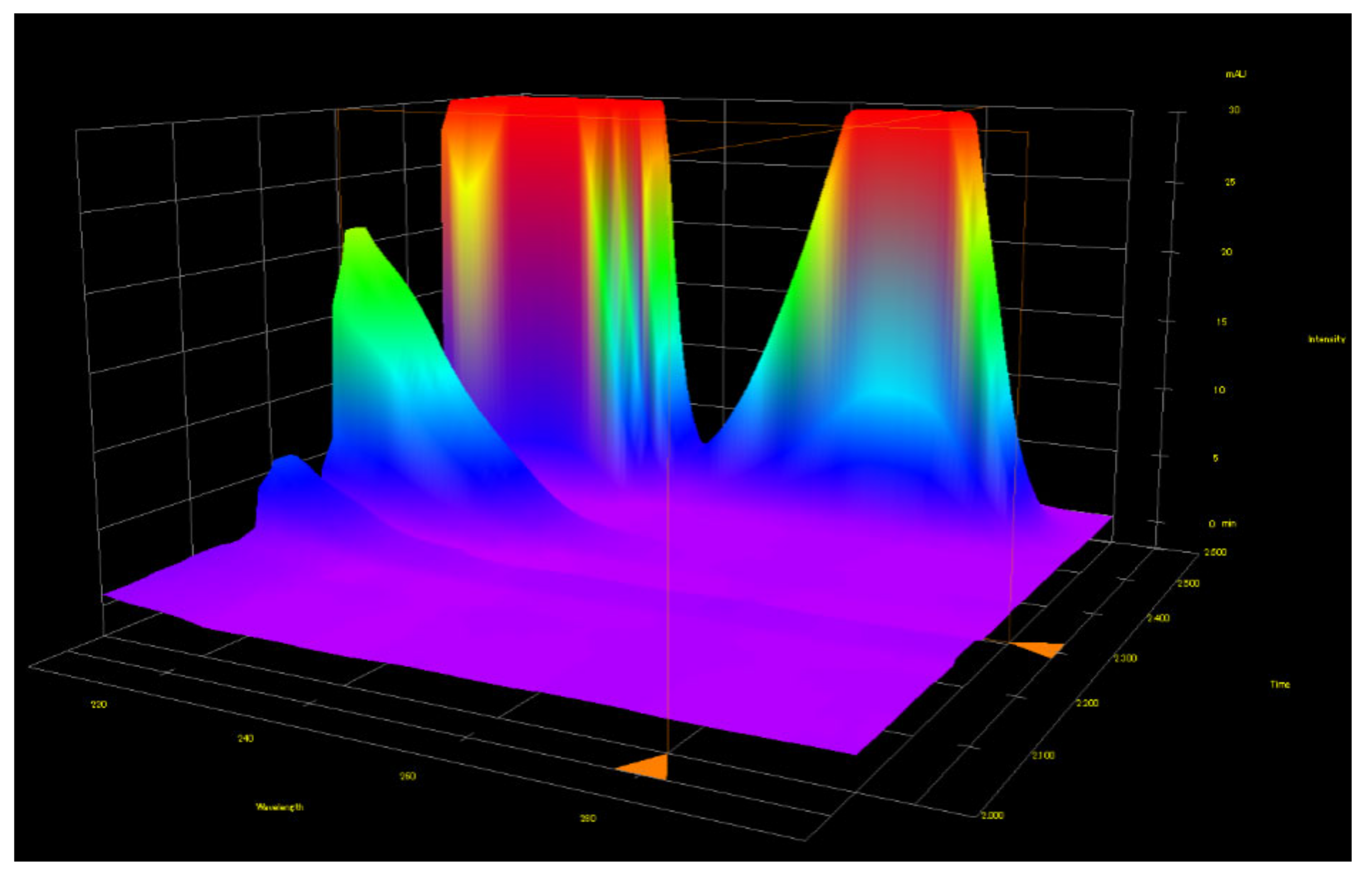
Figure 44.
High resolution PDA detector full 3-D chromatogram confirms the identity of peaks with implying certain extent of confidence in peak recognition and measuring, with easily recognized characteristic UV-Abs spectrum of meldonium, annotated with marking triangle located at the axis presenting retention time.
Figure 44.
High resolution PDA detector full 3-D chromatogram confirms the identity of peaks with implying certain extent of confidence in peak recognition and measuring, with easily recognized characteristic UV-Abs spectrum of meldonium, annotated with marking triangle located at the axis presenting retention time.
Figure 45.
Chromatograms with application of percentage of ACN with 60%, showed a very slight increased retention for both analytes with very good shaped peaks, but with disappearing small peaks and appearing negative peaks between meldonium and metoprolol, with column Zorbax CN-SB 250 × 4.6 mm.
Figure 45.
Chromatograms with application of percentage of ACN with 60%, showed a very slight increased retention for both analytes with very good shaped peaks, but with disappearing small peaks and appearing negative peaks between meldonium and metoprolol, with column Zorbax CN-SB 250 × 4.6 mm.
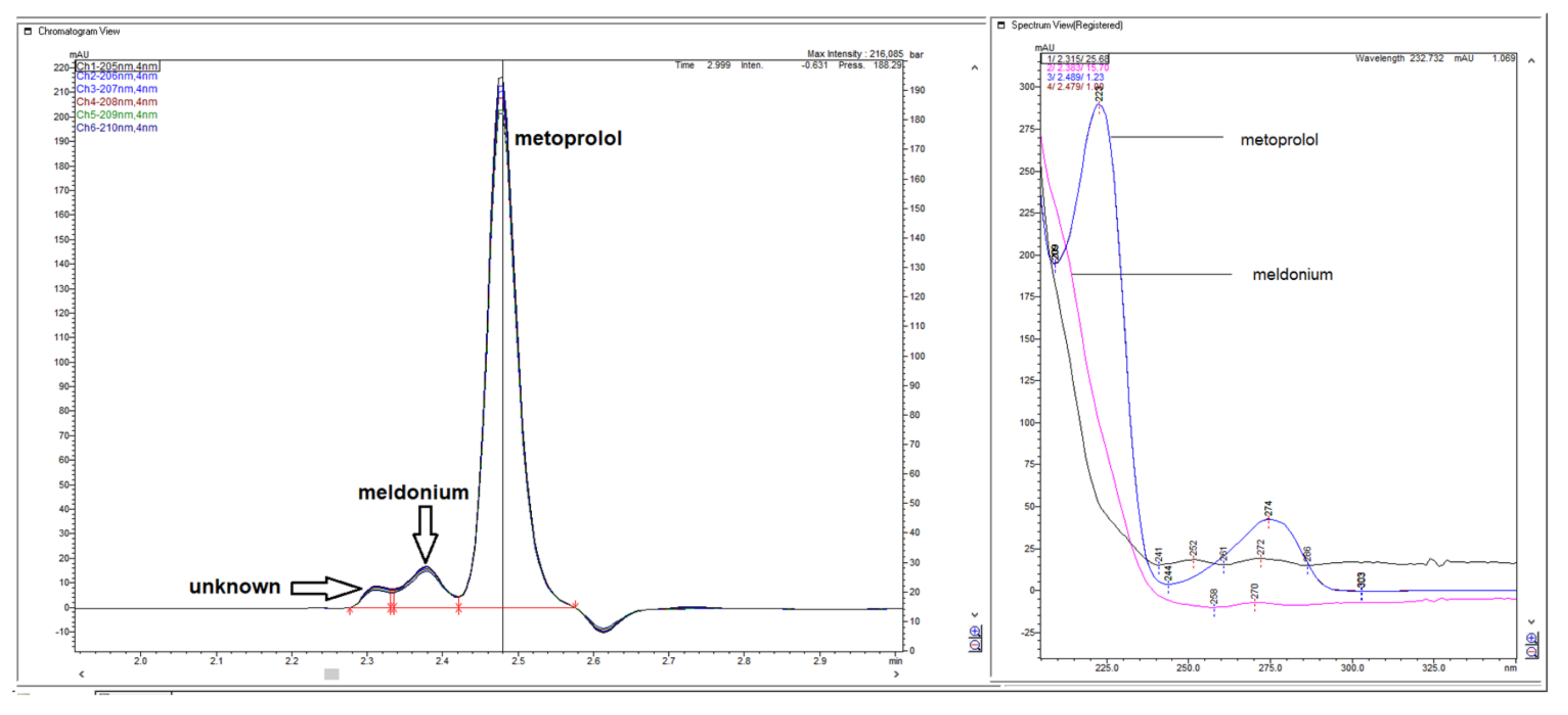
Figure 46.
Chromatograms, obtained by increasing ACN to 80%, guided the appearance of chromatogram with a slightly increased retention, appearing on new non separated peaks of unknown and meldonium, separated metoprolol peak, and with a negative peak on the baseline eluting right behind metoprolol, jeopardizing this peak accuracy definition.
Figure 46.
Chromatograms, obtained by increasing ACN to 80%, guided the appearance of chromatogram with a slightly increased retention, appearing on new non separated peaks of unknown and meldonium, separated metoprolol peak, and with a negative peak on the baseline eluting right behind metoprolol, jeopardizing this peak accuracy definition.
Figure 47.
Two chromatograms with Zorbax CN-SB column of sample with doubled percentage of CAN in eluent. TFA enables enormous an approach of peak of metoprolol to meldonium, reducing their resolution to very low values approaching to 2.
Figure 47.
Two chromatograms with Zorbax CN-SB column of sample with doubled percentage of CAN in eluent. TFA enables enormous an approach of peak of metoprolol to meldonium, reducing their resolution to very low values approaching to 2.
Figure 48.
Chromatograms created with 20% ACN and 80% o-H3PO4 (the top 2 chromatograms), with acidified 13 mM NH4H2PO4 to pH = 2.5 (medial 4 chromatograms) and with untreated 13.3 mM NH4H2PO4. High acidity of eluents showed higher peaks asymmetries of some peak.
Figure 48.
Chromatograms created with 20% ACN and 80% o-H3PO4 (the top 2 chromatograms), with acidified 13 mM NH4H2PO4 to pH = 2.5 (medial 4 chromatograms) and with untreated 13.3 mM NH4H2PO4. High acidity of eluents showed higher peaks asymmetries of some peak.
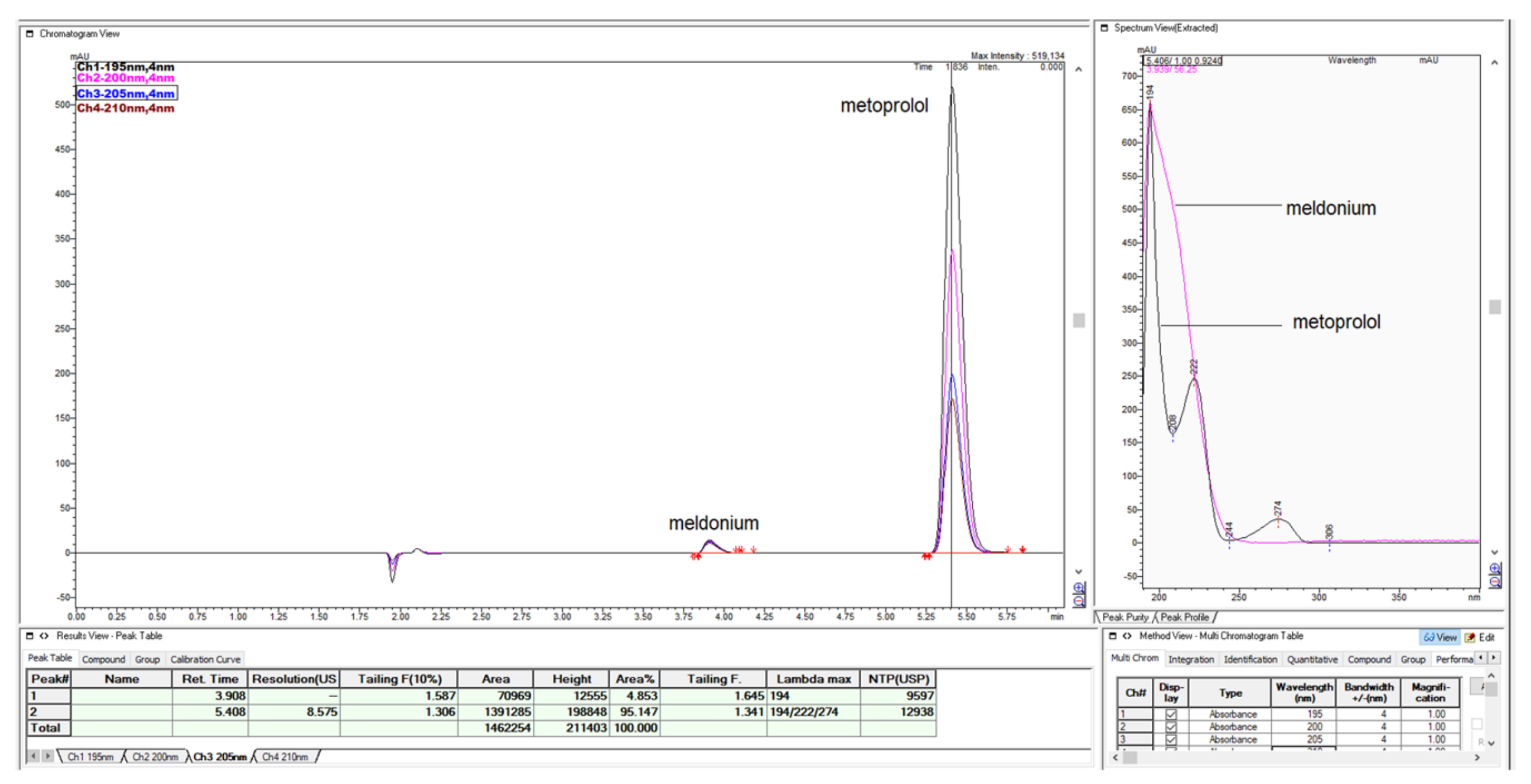
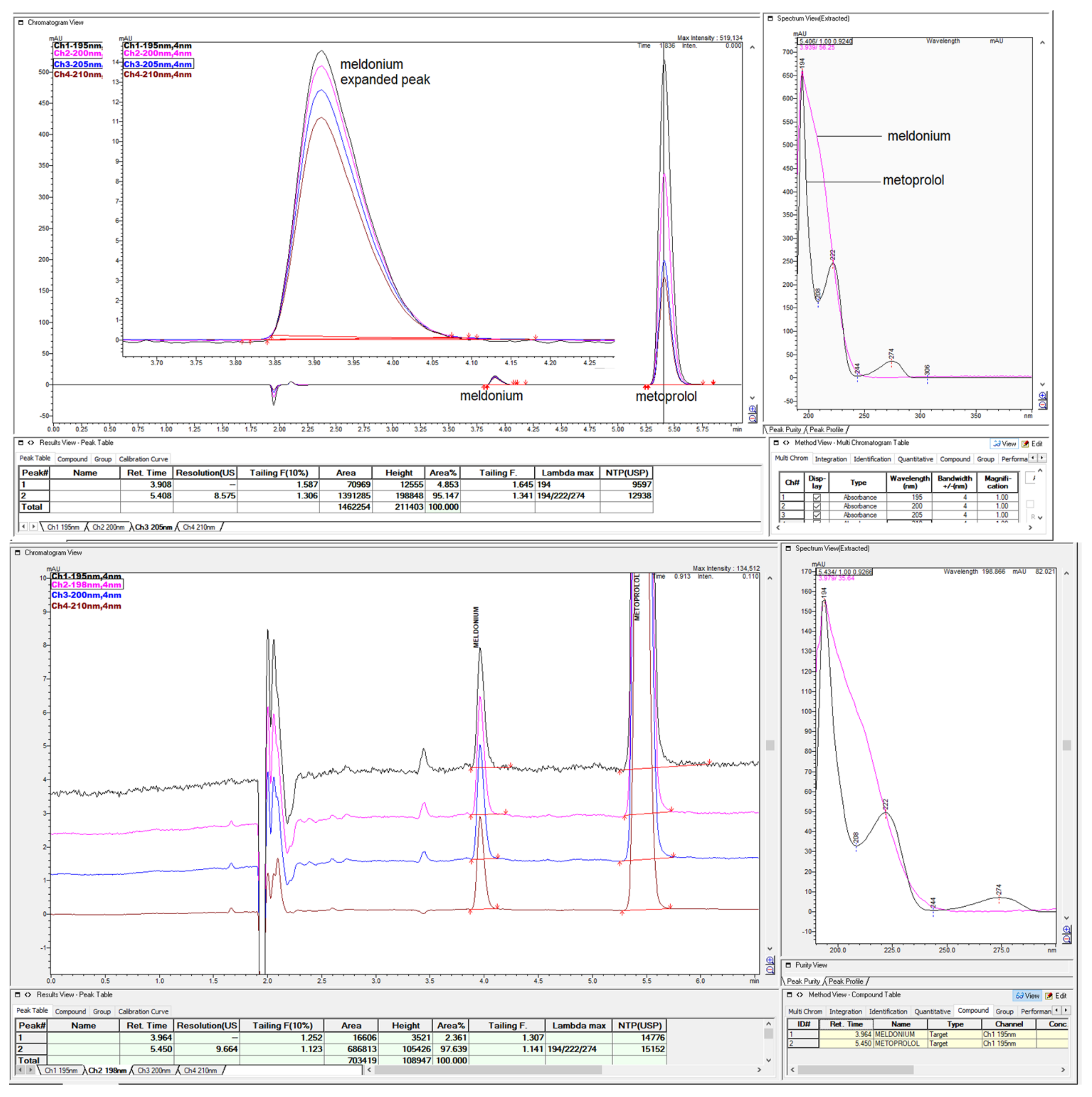
Figure 49.
The short summary overview of created chromatograms of sample with 2.5 mg/mL meldonium and 0.5 mg/mL metoprolol, with different percentages of ACN and different inorganic part composed of 0.065% TFA or 15 mM NH4H2PO4 were presented for comparison in overlaid mode, performed on Zorbax CN-SB 250 × 4.6 mm.
Figure 49.
The short summary overview of created chromatograms of sample with 2.5 mg/mL meldonium and 0.5 mg/mL metoprolol, with different percentages of ACN and different inorganic part composed of 0.065% TFA or 15 mM NH4H2PO4 were presented for comparison in overlaid mode, performed on Zorbax CN-SB 250 × 4.6 mm.
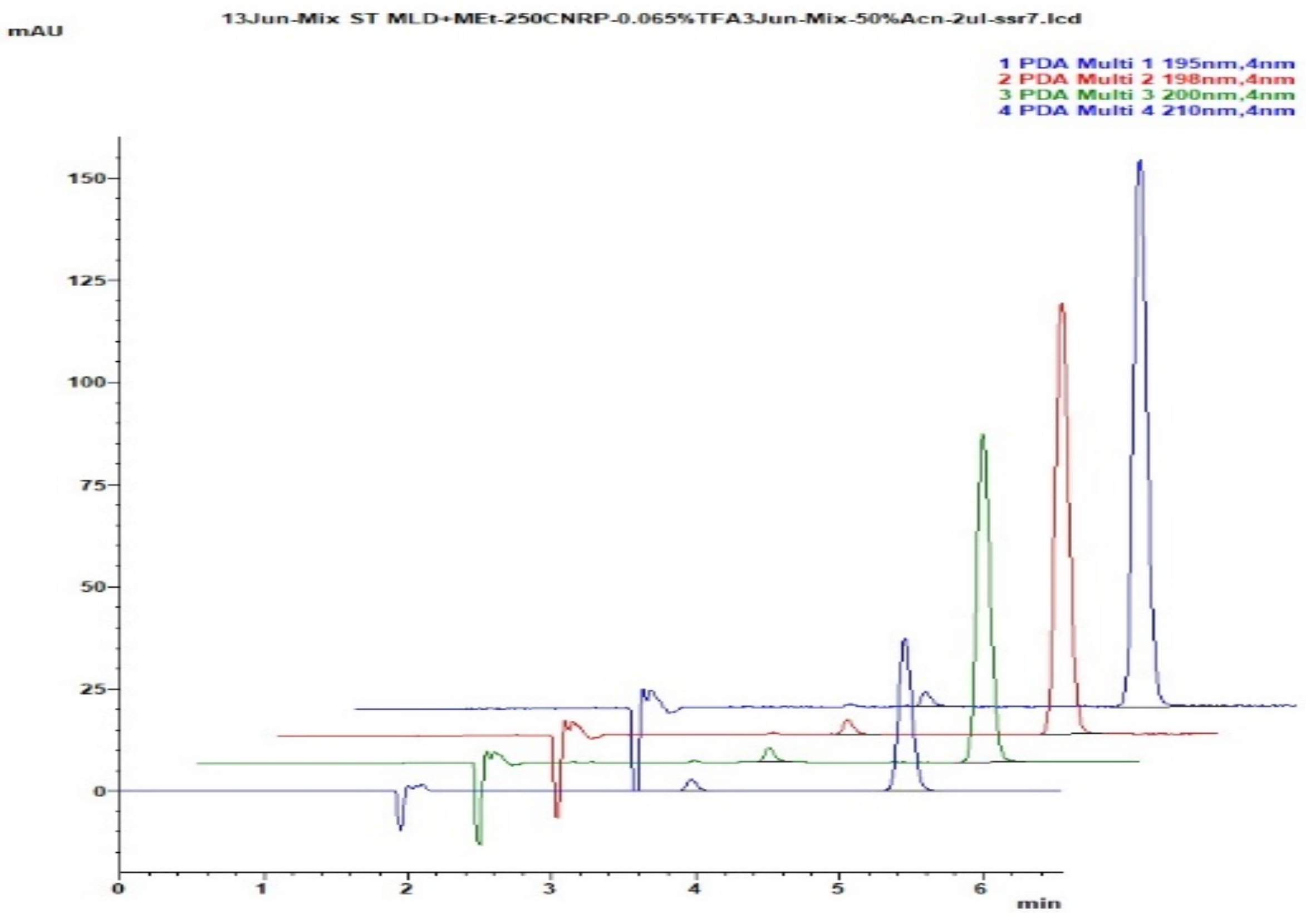
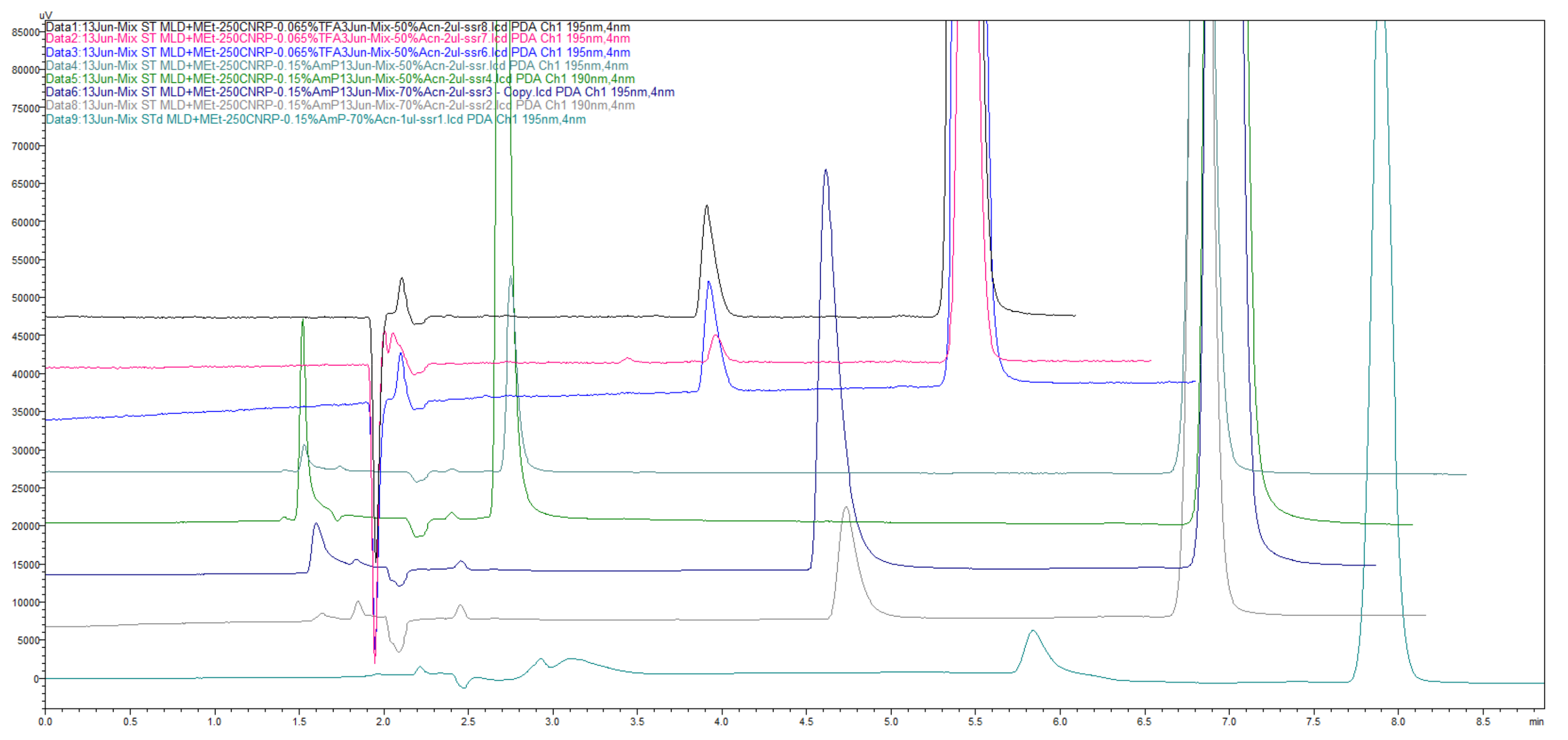
Figure 50.
Chromatograms, obtained for use in CN-cyano column, Zorbax CN-SB 250 × 4.6 mm for simultaneous determination of meldonium with metoprolol. Numerous combinations of ACN with 0.15% NH4H2PO4 have been presented overlaid at 195 nm and 200 nm, compared with the last two chromatograms on the bottom, which are made of on 100 × 4 mm C18 Xterra column with 10% ACN and not eluting metoprolol even to 20 min.
Figure 50.
Chromatograms, obtained for use in CN-cyano column, Zorbax CN-SB 250 × 4.6 mm for simultaneous determination of meldonium with metoprolol. Numerous combinations of ACN with 0.15% NH4H2PO4 have been presented overlaid at 195 nm and 200 nm, compared with the last two chromatograms on the bottom, which are made of on 100 × 4 mm C18 Xterra column with 10% ACN and not eluting metoprolol even to 20 min.
Table 1.
Comparison between the proposed HPLC concepts.
Table 1.
Comparison between the proposed HPLC concepts.
| Concept | I | II | III | IV | V | VI | VII | VIII | IX | X | XI | XII |
|---|
| Linearity range, mg/mL | 0.2–1.0—metoprolol 1–5—meldonium | |
| Mobile phase | ACN—0.15% NH4H2PO4(20:80, v/v) | ACN—0.065% TFA (45:55, v/v) | ACN—0.15% NH4H2PO4 (20:80, v/v) | ACN—0.065% TFA (25:75, v/v) | ACN—0.065% TFA (45:55, v/v) | ACN—0.15% NH4H2PO4 (50:50, v/v) | ACN—0.065% TFA (50:50, v/v) | ACN—0.15% NH4H2PO4 (20:80, v/v) | ACN—0.15% NH4H2PO4 (50:50, v/v) | ACN—0.15% NH4H2PO4 (60:40, v/v) | ACN—0.07% TFA (40:60, v/v) | ACN—0.07% TFA (30:70, v/v) |
| Stationary phase | LiChrospher®® 125 CN (5 µm) (4 mm i.d. × 125 mm, 5 μm) | LiChrospher®® 125 CN (5 µm) (4 mm i.d. × 125 mm, 5 μm) | LiChrospher®® 250 CN (5 µm) (4 mm i.d. × 250 mm, 5 μm) | LiChrospher®® 250 CN (5 µm) (4 mm i.d. × 250 mm, 5 μm) | LiChrospher®® 250 CN (5 µm) (4 mm i.d. × 250 mm, 5 μm) | Waters Spherisorb CNRP (4.6 mm i.d. × 250 mm, 5 μm) | Waters Spherisorb CNRP (4.6 mm i.d. × 250 mm, 5 μm) | Zorbax CN SB (4.6 mm i.d. × 250 mm, 5 μm) | Zorbax CN SB (4.6 mm i.d. × 250 mm, 5 μm) | Zorbax CN SB (4.6 mm i.d. × 250 mm, 5 μm) | Zorbax CN SB (4.6 mm i.d. × 250 mm, 5 μm) | Zorbax CN SB (4.6 mm i.d. × 250 mm, 5 μm) |
| Run time, min | 5.5 | 4 | 9 | 8.5 | 7 | 8 | 6 | 10 | 3.5 | 3.5 | 5 | 3 |
| Flow rate, mL min−1 | 1 | 1 | 1 | 1 | 1 | 1.2 | 1.2 | 1 | 1 | 1 | 1 | 1 |
| Wavelength, nm | 200 | 200 | 200 | 200 | 200 | 200 and 210 | 200 and 210 | 200 | 200 | 210 | 210 | 210 |
| LOD/LOQ, mg/mL | 0.011/0.035—meldonium0.00022/0.00075—metoprolol | 0.032/0.011—meldonium0.00041/0.00175—metoprolol | 0.011/0.035—meldonium0.00022/0.00075—metoprolol | 0.043/0.133—meldonium0.00038/0.00157—metoprolol | 0.039/0.0.180—meldonium0.00039/0.00143—metoprolol | 0.015/0.047—meldonium0.00022/0.0008—metoprolol | 0.051/0.153—meldonium0.00043/0.00164—metoprolol | 0.017/0.056—meldonium0.00016/0.00051—metoprolol | 0.021/0.059—meldonium0.00016/0.00052—metoprolol | 0.017/0.056—meldonium0.00019/0.00066—metoprolol | 0.045/0.161—meldonium0.0045/0.0015—metoprolol | 0.052/0.173—meldonium0.0053/0.0018—metoprolol |
| Linearity regression, R2 | y-16700× +9814.120.9998 | y-15963× +5987.530.9999 | y-13753× +5492.891.0000 | y-19784× +8905.750.9999 | y-17906× +7859.940.9999 | y-18945× +7896.570.9997 | y-17834× +7894.381.0000 | y-18563× +9473.840.9997 | y-15978x+6739.970.9999 | y-17890× +6491.230.9997 | y-18856× +7819.781.0000 | y-17829× +6743.190.9998 |
| Analytical GREEnness (AGREE) | 0.76 | 0.69 | 0.74 | 0.67 | 0.68 | 0.75 | 0.68 | 0.74 | 0.77 | 0.77 | 0.68 | 0.69 |
Table 2.
Intra-day accuracy and precision results.
Table 2.
Intra-day accuracy and precision results.
| Concept | I | II | III | IV | V | VI | VII | VIII | IX | X | XI | XII |
|---|
| Analyte | Mean, % (RSD, %) |
| Meldonium | 99.86 (0.32) | 99.67 (0.39) | 100.48 (0.21) | 100.87 (0.40) | 99.56 (0.30) | 100.09 (0.20) | 100.36 (0.31) | 100.03 (0.34) | 100.15 (0.28) | 100.09 (0.29) | 99.89 (0.41) | 100.14 (0.40) |
| 99.84 (0.38) | 99.42 (0.42) | 99.47 (0.46) | 100.67 (0.31) | 100.05 (0.24) | 100.17 (0.41) | 100.51 (0.27) | 99.84 (0.44) | 100.22 (0.34) | 100.17 (0.21) | 99.75 (0.23) | 100.09 (0.35) |
| 100.12 (0.28) | 99.53 (0.45) | 100.32 (0.35) | 100.76 (0.34) | 100.18 (0.29) | 100.15 (0.34) | 100.60 (0.20) | 100.01 (0.42) | 100.11 (0.40) | 99.81 (0.33) | 99.69 (0.34) | 100.29 (0.38) |
| Metoprolol | 99.97 (0.22) | 100.35 (0.27) | 99.47 (0.25) | 100.13 (0.29) | 99.55 (0.35) | 100.78 (0.45) | 99.90 (0.18) | 99.45 (0.44) | 99.56 (0.31) | 100.22 (0.37) | 100.05 (0.22) | 100.01 (0.27) |
| 99.84 (0.41) | 99.95 (0.39) | 99.67 (0.30) | 100.35 (0.40) | 99.78 (0.39) | 100.66 (0.37) | 100.17 (0.33) | 99.48 (0.20) | 99.77 (0.37) | 100.45 (0.38) | 100.01 (0.30) | 100.18 (0.29) |
| 100.53 (0.36) | 99.56 (0.27) | 99.70 (0.27) | 100.22 (0.28) | 99.70 (0.23) | 100.56 (0.29) | 100.12 (0.28) | 99.67 (0.31) | 99.65 (0.40) | 100.30 (0.31) | 100.10 (0.27) | 100.15 (0.35) |
Table 3.
Inter-day accuracy and precision results.
Table 3.
Inter-day accuracy and precision results.
| Concept | I | II | III | IV | V | VI | VII | VIII | IX | X | XI | XII |
|---|
| Analyte | Mean, % (RSD, %) |
| Meldonium | 99.56 (0.21) | 100.05 (0.30) | 100.10 (0.26) | 100.01 (0.22) | 100.15 (0.20) | 99.61 (0.48) | 99.93 (0.28) | 100.06 (0.32) | 100.15 (0.16) | 100.05 (0.18) | 100.14 (0.30) | 100.18 (0.26) |
| 99.51 (0.30) | 100.02 (0.36) | 99.93 (0.19) | 99.64 (0.36) | 100.01 (0.29) | 99.82 (0.45) | 99.84 (0.20) | 100.27 (0.40) | 100.22 (0.40) | 100.22 (0.41) | 99.95 (0.13) | 100.12 (0.25) |
| 99.89 (0.34) | 99.67 (0.24) | 100.18 (0.26) | 100.05 (0.42) | 100.24 (0.36) | 99.89 (0.38) | 100.06 (0.25) | 100.20 (0.23) | 100.11 (0.35) | 99.86 (0.20) | 99.80 (0.23) | 100.34 (0.44) |
| Metoprolol | 100.18 (0.28) | 100.06 (0.25) | 99.83 (0.22) | 99.91 (0.17) | 99.70 (0.30) | 100.03 (0.26) | 99.95 (0.37) | 99.97 (0.40) | 99.56 (0.24) | 100.12 (0.27) | 100.17 (0.12) | 99.69 (0.42) |
| 100.24 (0.22) | 99.78 (0.28) | 99.50 (0.29) | 99.83 (0.32) | 99.91 (0.26) | 100.14 (0.17) | 100.08(0.31) | 99.51 (0.44) | 99.77 (0.31) | 100.09 (0.18) | 100.12 (0.28) | 99.93 (0.15) |
| 100.13 (0.36) | 99.85 (0.20) | 99.89 (0.24) | 99.63 (0.38) | 99.95 (0.29) | 100.25 (0.20) | 100.17 (0.44) | 99. (0.37) | 99.65 (0.33) | 100.01 (0.22) | 100.03 (0.35) | 99.74 (0.31) |