Melt Fusion Techniques for Solubility Enhancement: A Comparison of Hot Melt Extrusion and KinetiSol® Technologies
Abstract
:1. Introduction
2. Mechanism
3. Formulation Aspects
3.1. Active Ingredients
3.2. Polymers
3.3. Additives and Processing Aids
4. Process Parameters Involved in HME and KinetiSol®
5. Physical Properties of ASDs Prepared Using HME and KinetiSol®
6. Scalability
7. Case Studies
8. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| GIT | Gastrointestinal tract |
| ASDs | Amorphous Solid Dispersions |
| HME | Hot Melt Extrusion |
| API | Active Pharmaceutical Ingredient |
| HMW | High-Molecular Weight |
| LMW | Low-Molecular Weight |
References
- Hua, S. Advances in oral drug delivery for regional targeting in the gastrointestinal tract-influence of physiological, pathophysiological and pharmaceutical factors. Front. Pharmacol. 2020, 11, 524. [Google Scholar] [CrossRef]
- Dokania, S.; Joshi, A.K. Self-microemulsifying drug delivery system (SMEDDS)–challenges and road ahead. Drug Deliv. 2015, 22, 675–690. [Google Scholar] [CrossRef] [Green Version]
- Blaabjerg, L.I.; Grohganz, H.; Lindenberg, E.; Löbmann, K.; Müllertz, A.; Rades, T. The influence of polymers on the supersaturation potential of poor and good glass formers. Pharmaceutics 2018, 10, 164. [Google Scholar] [CrossRef] [Green Version]
- Mishra, M. Concise Encyclopedia of Biomedical Polymers and Polymeric Biomaterials; CRC Press: Boca Raton, FL, USA, 2017; pp. 832–833. ISBN 978-14-3989-855-0. [Google Scholar]
- Graeser, K.A.; Patterson, J.E.; Zeitler, J.A.; Rades, T. The role of configurational entropy in amorphous systems. Pharmaceutics 2010, 2, 224–244. [Google Scholar] [CrossRef]
- Ilevbare, G.A.; Liu, H.; Edgar, K.J.; Taylor, L.S. Maintaining supersaturation in aqueous drug solutions: Impact of different polymers on induction times. Cryst. Growth Des. 2013, 13, 740–751. [Google Scholar] [CrossRef]
- Bhujbal, S.V.; Mitra, B.; Jain, U.; Gong, Y.; Agrawal, A.; Karki, S.; Taylor, L.S.; Kumar, S.; Zhou, Q.T. Pharmaceutical amorphous solid dispersion: A review of manufacturing strategies. Acta Pharm. Sin. B 2021, 11, 2505–2536. [Google Scholar] [CrossRef]
- Lang, B. Advanced Formulation and Processing Technologies in the Oral Delivery of Poorly Water-Soluble Drugs. Ph.D. Thesis, The University of Texas at Austin, Austin, TX, USA, 2013. [Google Scholar]
- Kallakunta, V.R.; Sarabu, S.; Bandari, S.; Tiwari, R.; Patil, H.; Repka, M.A. An update on the contribution of hot-melt extrusion technology to novel drug delivery in the twenty-first century: Part I. Expert Opin. Drug Deliv. 2019, 16, 539–550. [Google Scholar] [CrossRef]
- Tan, D.K.; Davis, D.A.; Miller, D.A.; Williams, R.O.; Nokhodchi, A. Innovations in thermal processing: Hot-melt extrusion and KinetiSol® dispersing. AAPS PharmSciTech 2020, 21, 312. [Google Scholar] [CrossRef]
- Shamsuddin, M.F.; Ansari, S.H.; Ali, J. Development and evaluation of solid dispersion of spironolactone using fusion method. Int. J. Pharm. Investig. 2016, 6, 63. [Google Scholar] [CrossRef] [Green Version]
- Jermain, V.S.; Miller, D.; Spangenberg, A.; Lu, X.; Moon, C.; Su, Y.; Williams, R.O. Homogeneity of amorphous solid dispersions–an example with KinetiSol®. Drug Dev. Ind. Pharm. 2019, 45, 724–735. [Google Scholar] [CrossRef]
- Jermain, S.V.; Lowinger, M.B.; Ellenberger, D.J.; Miller, D.A.; Su, Y.; Williams, R.O., III. In vitro and in vivo behaviors of KinetiSol® and spray-dried amorphous solid dispersions of a weakly basic drug and ionic polymer. Mol. Pharm. 2020, 17, 2789–2808. [Google Scholar] [CrossRef]
- Schenck, L.; Neri, C.; Jia, X.; Schafer, W.; Axnanda, S.; Canfield, N.; Li, F.; Shah, V. A Co-Processed API Approach for a Shear Sensitive Compound Affording Improved Chemical Stability and Streamlined Drug Product Processing. J. Pharm. Sci. 2021, 110, 3238–3245. [Google Scholar] [CrossRef]
- Polizzi, A.M.; Singhal, D.; Colvin, J. Mechanoradical-induced degradation in a pharmaceutical blend during high-shear processing. Pharm. Dev. Technol. 2008, 13, 457–462. [Google Scholar] [CrossRef]
- Sarat, M.; Samanta, S.; Kothari, K.; Mistry, P.; Suryanarayanan, R. Effect of polymer molecular weight on the crystallization behavior of indomethacin amorphous solid dispersions. Cryst. Growth Des. 2017, 17, 3142–3150. [Google Scholar]
- LaFountaine, S.J.; McGinity, J.W.; Williams, R.O. Challenges and strategies in thermal processing of amorphous solid dispersions: A review. AAPS PharmSciTech 2016, 17, 43–55. [Google Scholar] [CrossRef] [Green Version]
- Roberta, C.; Gigliobianco, M.R.; Casadidio, C.; Di Martino, P. Hot melt extrusion: Highlighting physicochemical factors to be investigated while designing and optimizing a hot melt extrusion process. Pharmaceutics 2018, 10, 89. [Google Scholar]
- Ellenberger, D.J. Processing Challenging Active Pharmaceutical Ingredients and Polymers by Kinetisol to Produce Amorphous Solid Dispersions with Improved In-Vitro and In-Vivo Performance. Ph.D. Thesis, The University of Texas at Austin, Austin, TX, USA, 2017. [Google Scholar]
- Kallakunta, V.R.; Sarabu, S.; Dudhipala, N.; Janga, K.Y.; Bandari, S.; Zhang, F.; Repka, M.A. Chrono modulated multiple unit particulate systems (MUPS) via a continuous hot melt double extrusion technique: Investigation of the formulation and process suitability. Eur. J. Pharm. Biopharm. 2021, 168, 184–194. [Google Scholar] [CrossRef]
- Marta, F.S.; Pinto, R.M.A.; Simões, S. Hot-melt extrusion in the pharmaceutical industry: Toward filing a new drug application. Drug Discov. Today 2019, 24, 1749–1768. [Google Scholar]
- Miller, D.A.; Dinunzio, J.C.; Hughey, J.R.; Williams, R.O.; Mcginity, J.W. KinetiSol: A new processing paradigm for amorphous solid dispersion systems. Drug Dev. Deliv 2012, 12, 1749–1768. [Google Scholar]
- DiNunzio, J.C.; Brough, C.; Miller, D.A.; Williams, R.O., III; McGinity, J.W. Applications of KinetiSol® Dispersing for the production of plasticizer free amorphous solid dispersions. Eur. J. Pharm. Sci. 2010, 40, 179–187. [Google Scholar] [CrossRef]
- Paulsen, K.; Leister, D.; Gryczke, A. Investigating Process Parameter Mechanism for Successful Scale-Up of a Hot-Melt Extrusion Process; AppNote LR-71; Thermo Fisher Scientific: Karlsruhe, Germany, 2013. [Google Scholar]
- Kostewicz, E.S.; Vertzoni, M.; Benson, H.A.E.; Roberts, M.S. Oral Drug Delivery for Modified Release Formulations; Wiley: Hoboken, NJ, USA, 2022; pp. 193–194. ISBN 978-11-1977-269-9. [Google Scholar]
- Ma, X.; Williams, R.O. Characterization of amorphous solid dispersions: An update. J. Drug Deliv. Sci. Technol. 2019, 50, 113–124. [Google Scholar] [CrossRef]
- Gurunath, S.; Kumar, S.P.; Basavaraj, N.K.; Patil, P.A. Amorphous solid dispersion method for improving oral bioavailability of poorly water-soluble drugs. J. Pharm. Res. 2013, 6, 476–480. [Google Scholar] [CrossRef]
- Stuart LGoldberg, M.D.; Patricia JGiardina, M.D.; Deborah Chirnomas, M.D.; Jason Esposito, M.S.H.S.; Carole Paley, M.D.; Elliott Vichinsky, M.D. The palatability and tolerability of deferasirox taken with different beverages or foods. Pediatr. Blood Cancer 2013, 60, 1507–1512. [Google Scholar] [CrossRef]
- Deborah, C.; Smith, A.L.; Braunstein, J.; Finkelstein, Y.; Pereira, L.; Bergmann, A.K.; Grant, F.D.; Paley, C.; Shannon, M.; Neufeld, E.J. Deferasirox pharmacokinetics in patients with adequate versus inadequate response. Blood. J. Am. Soc. Hematol. 2009, 114, 4009–4013. [Google Scholar]
- Available online: https://clinicaltrials.gov/ct2/show/NCT03637556 (accessed on 22 August 2022).
- Gala, U.; Miller, D.; Williams, R.O., III. Improved Dissolution and Pharmacokinetics of Abiraterone through KinetiSol® Enabled Amorphous Solid Dispersions. Pharmaceutics 2020, 12, 357. [Google Scholar] [CrossRef] [Green Version]
- Available online: https://clinicaltrials.gov/ct2/show/NCT04291664 (accessed on 13 July 2022).
- DiNunzio, C.J.; Brough, C.; Miller, D.A.; Williams, R.O., III; McGinity, J.W. Fusion processing of itraconazole solid dispersions by KinetiSol® dispersing: A comparative study to hot melt extrusion. J. Pharm. Sci. 2010, 99, 1239–1253. [Google Scholar] [CrossRef]
- Keen, M.J.; LaFountaine, J.S.; Hughey, J.R.; Miller, D.A.; McGinity, J.W. Development of itraconazole tablets containing viscous KinetiSol® solid dispersions: In vitro and in vivo analysis in dogs. AAPS PharmSciTech 2018, 19, 1998–2008. [Google Scholar] [CrossRef]
- Brough, C.; Miller, D.A.; Keen, J.M.; Kucera, S.A.; Lubda, D.; Williams, R.O., III. Use of polyvinyl alcohol as a solubility-enhancing polymer for poorly water-soluble drug delivery (part 1). AAPS PharmSciTech 2016, 17, 167–179. [Google Scholar] [CrossRef] [Green Version]
- Brough, C.; Miller, D.A.; Ellenberger, D.; Lubda, D.; Williams, R.O. Use of polyvinyl alcohol as a solubility enhancing polymer for poorly water-soluble drug delivery (part 2). AAPS PharmSciTech 2016, 17, 180–190. [Google Scholar] [CrossRef] [Green Version]
- Ellenberger, D.J.; Miller, D.A.; Kucera, S.U.; Williams, R.O., III. Improved Vemurafenib Dissolution and Pharmacokinetics as an Amorphous Solid Dispersion Produced by KinetiSol® Processing. AAPS PharmSciTech 2018, 19, 1957–1970. [Google Scholar] [CrossRef]
- Hughey, J.R.; Keen, J.M.; Brough, C.; Saeger, S.; McGinity, J.W. Thermal processing of a poorly water-soluble drug substance exhibiting a high melting point: The utility of KinetiSol® dispersing. Int. J. Pharm. 2011, 419, 222–230. [Google Scholar] [CrossRef] [PubMed]
- Ellenberger, D.J.; Miller, D.A.; Kucera, S.U.; Williams, R.O. Generation of a Weakly Acidic Amorphous Solid Dispersion of the Weak Base Ritonavir with Equivalent In Vitro and In Vivo Performance to Norvir Tablet. AAPS PharmSciTech 2018, 19, 1985–1997. [Google Scholar] [CrossRef] [PubMed]
- LaFountaine, J.S.; Jermain, S.V.; Prasad, L.K.; Brough, C.; Miller, D.A.; Lubda, D.; McGinity, J.W.; Williams, R.O., III. Enabling thermal processing of ritonavir–polyvinyl alcohol amorphous solid dispersions by KinetiSol® dispersing. Eur. J. Pharm. Biopharm. 2016, 101, 72–81. [Google Scholar] [CrossRef] [PubMed]
- DiNunzio, J.C.; Brough, C.; Hughey, J.R.; Miller, D.A.; Williams, R.O.; McGinity, J.W. Fusion production of solid dispersions containing a heat-sensitive active ingredient by hot melt extrusion and Kinetisol® dispersing. Eur. J. Pharm. Bio. Pharm. 2010, 74, 340–351. [Google Scholar] [CrossRef]
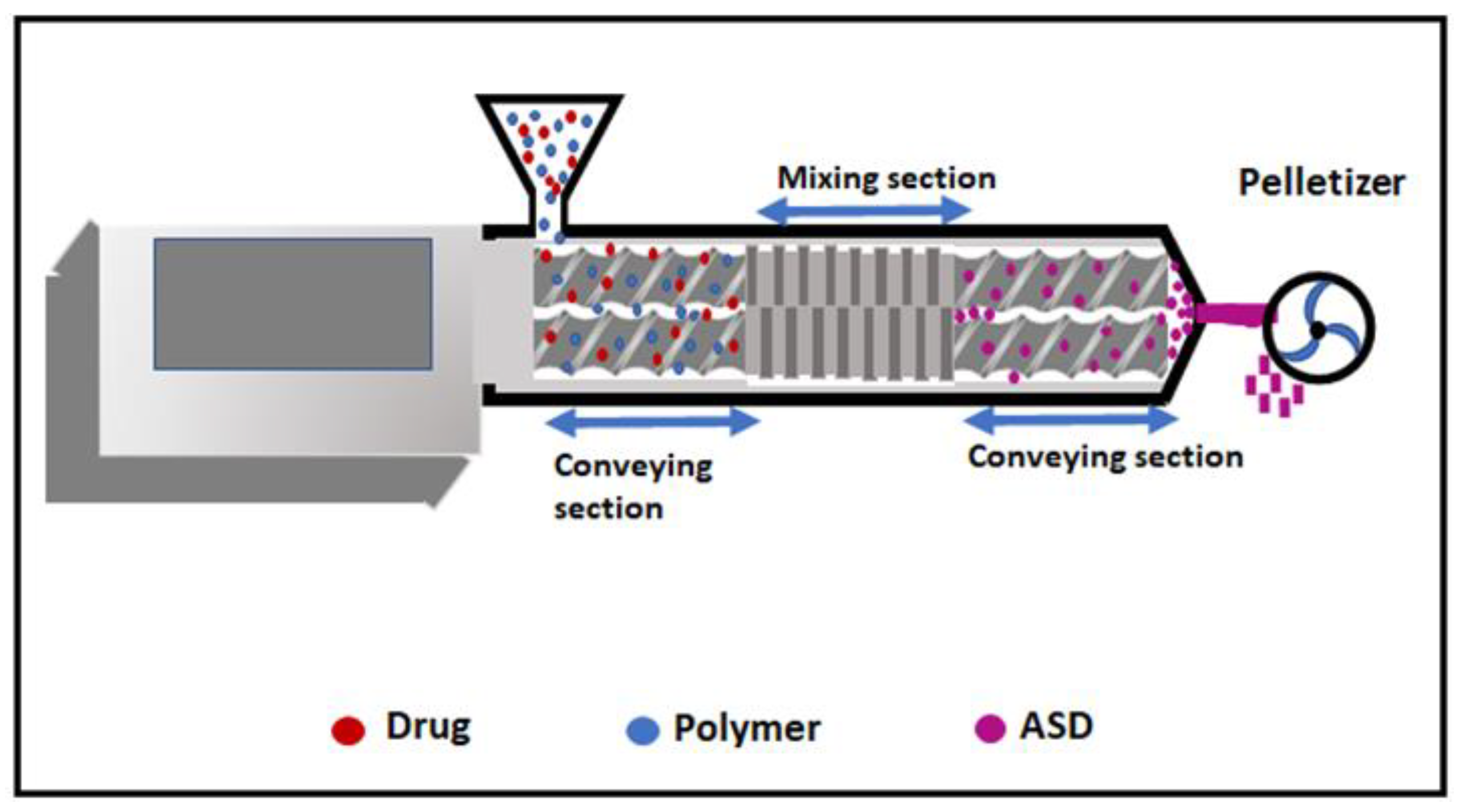
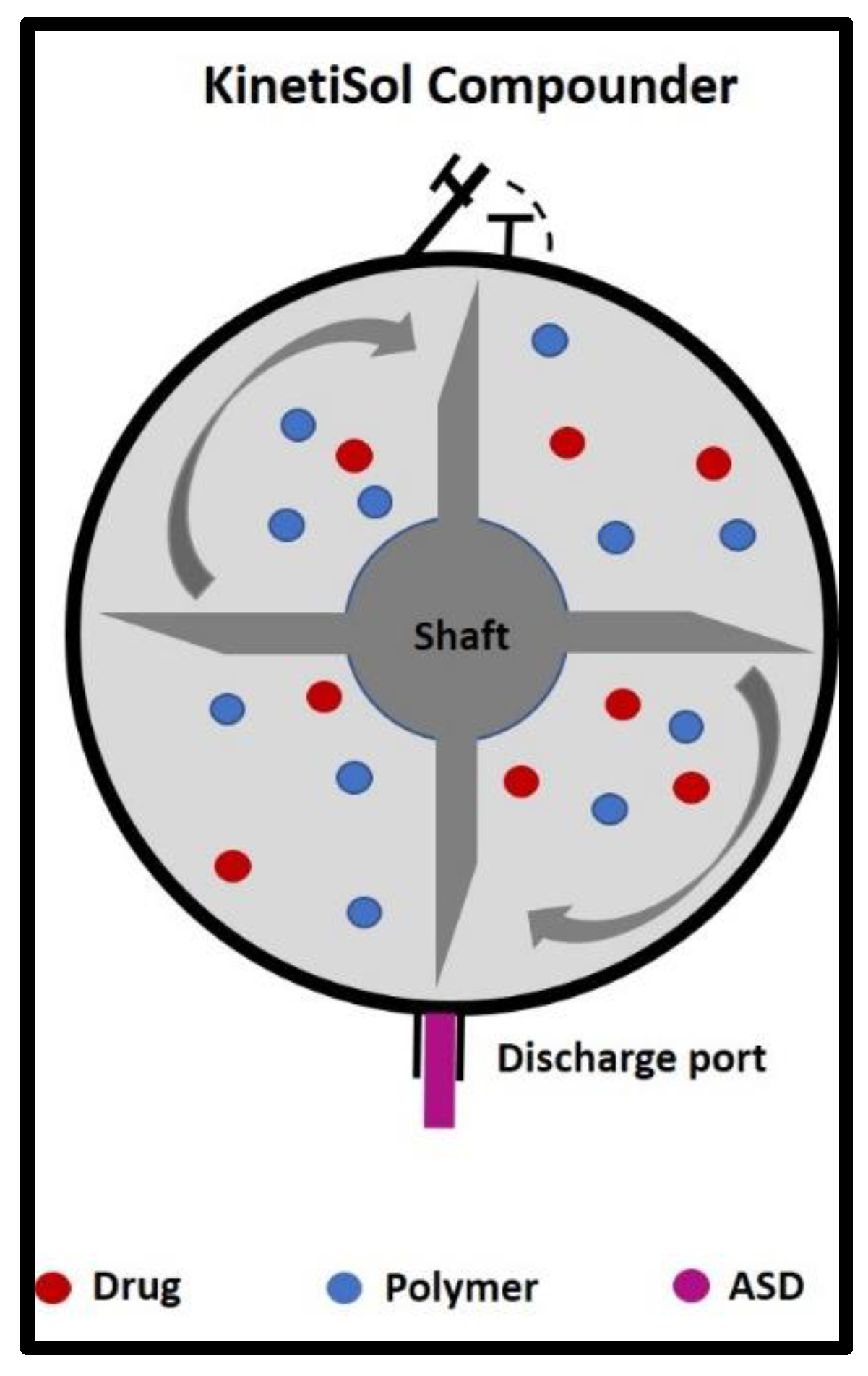
| Technique | Hot Melt Extrusion | KinetiSol® | References |
|---|---|---|---|
| Mechanism | Thermal fusion | Thermal fusion | [10] |
| API constraint | APIs sensitive to prolonged exposure to heat High melting point | APIs sensitive to shear | [12,13] |
| Polymers | Mostly hydrophilic | Mostly hydrophilic | [16] |
| Processing aid | Need for plasticizer in the case of high-melting-point APIs | Need for lubricant to improve processability | [17,18] |
| Processing time | Varies from minutes tohours | Seconds | [12,13] |
| Challenge | API/Polymer | Reference |
|---|---|---|
| Variable bioavailability | Deferasirox | [27,28,29] |
| High melting point and low solubility of API in organic solvents | Abiraterone | [31] |
| High viscous polymers | HPMC E50M, HPMC E4M, PVA | [34] |
| Drug solubility in neutral media | Investigational drug, BI 667 | [13] |
| Drug loading | Vemurafenib | [37] |
| Drug uniformity | Meloxicam | [38] |
| Drug stability | Ritonavir | [40] |
| Drug stability | Hydrocortisone | [41] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ajjarapu, S.; Banda, S.; Basim, P.; Dudhipala, N. Melt Fusion Techniques for Solubility Enhancement: A Comparison of Hot Melt Extrusion and KinetiSol® Technologies. Sci. Pharm. 2022, 90, 51. https://doi.org/10.3390/scipharm90030051
Ajjarapu S, Banda S, Basim P, Dudhipala N. Melt Fusion Techniques for Solubility Enhancement: A Comparison of Hot Melt Extrusion and KinetiSol® Technologies. Scientia Pharmaceutica. 2022; 90(3):51. https://doi.org/10.3390/scipharm90030051
Chicago/Turabian StyleAjjarapu, Srinivas, Srikanth Banda, Pratap Basim, and Narendar Dudhipala. 2022. "Melt Fusion Techniques for Solubility Enhancement: A Comparison of Hot Melt Extrusion and KinetiSol® Technologies" Scientia Pharmaceutica 90, no. 3: 51. https://doi.org/10.3390/scipharm90030051
APA StyleAjjarapu, S., Banda, S., Basim, P., & Dudhipala, N. (2022). Melt Fusion Techniques for Solubility Enhancement: A Comparison of Hot Melt Extrusion and KinetiSol® Technologies. Scientia Pharmaceutica, 90(3), 51. https://doi.org/10.3390/scipharm90030051







