1. Introduction
Origanum vulgare L., or oregano, is a perennial herbaceous plant of the Lamiaceae family.
Origanum herb (lat.
Herba Origani vulgaris) is used as a medicinal raw material and is cut during mass flowering.
O. vulgare L. is referred to in the USSR/Russian
Pharmacopoeia and is among the popular wild plants of the Russian
Pharmacopoeia that were traditionally used as food [
1,
2,
3].
O. vulgare is one of the most famous and economically important culinary herbs due to its essential oil, which is also used for medicinal purposes [
4,
5]. However, it must be kept in mind that plant species of the genus
Origanum have an intraspecific polymorphism and contain different amounts of biologically active substances; therefore, investigations and studies of new varieties and forms of processing of these plants are still ongoing [
6]. Pezzani et al. [
7] reviewed the biological activities of
O. vulgare and its main bioactive phytochemicals, considering both preclinical (in vitro and in vivo) and human studies focused on the plant’s anticancer, anti-inflammatory, antioxidant and antimicrobial activities. The contents of the active substances in the essential oils varies depending on the growing conditions, and consequently on the storage conditions. Moreover, the amounts of these substances change significantly when passing through the gastrointestinal tract, and their effective concentrations can be significantly reduced at the site of action (the small intestine and colon) [
8].
The popularity and importance of
O. vulgare are associated with its content of essential oil, which, in the dry aerial part of the plant, is 0.3–1.2%. To obtain the essential oil, the aerial mass is processed by hydrodistillation immediately after harvesting, and for the preparation of raw materials, it is dried as a spice in well-ventilated rooms or in a dryer at a temperature of no more than 30–40 °C. It must be taken into account that the chemical composition of oregano is determined by no single drying method that can be effectively used for the dehydration of all herbs. Heat treatment can lead to biochemical changes, which increases the plant’s antibacterial activity [
9,
10]. The main identifying substances in oregano essential oils are thymol—44–50%, carvacrol, bicyclic and tricyclic sesquiterpenes—12.5%, and geranyl acetate—2.6–5.0%. All species of
Origanum contain carvacrol as the main compound, and other compounds (thymol, gamma-terpinene, p-cymene, linalool, terpinen-4-ol, germacrene-D, beta-caryophyllene, myrcene, etc.) are found in most species as well [
11,
12].
Origanum vulgare is described as one of the sources of vegetable tannins [
13], which probably explains the widespread use of its essential oil in treating skin diseases [
14]. It is known that plants rich in polyphenols are used to prevent thrombosis, and the antiplatelet activity of
O. vulgare is associated with the presence of caffeic acid, salvigenin and umbelliferone [
15].
A possible dose-dependent effect of the biomolecules present in
O. vulgare on the secretion of male steroids that play a role in the regulation of testicular spermatogenesis was also identified by Tvrdá et al. [
16]. In an in vitro experiment, testicular fragments from mature mice were incubated with different concentrations of oregano extract for 24 h. The results showed that the oregano extract significantly reduced the mice’s cholesterol and androstenedione (but not dehydroepiandrosterone and testosterone) [
16].
Extracts of the
Origanum genus species exhibit antibacterial, antifungal and microorganism-inhibiting effects. Essential oils with a high content of carvacrol among their bactericidal and antihistamine properties can reflect the activity of some antibiotics. It is with carvacrol and thymol that the antimicrobial and antioxidant activity is associated, which is achieved by increasing the permeability of the cell membrane due to these substances’ impregnation in the hydrophobic domains, and this effect is more pronounced in relation to gram-positive bacteria [
10,
17,
18,
19]. Marchese et al. [
20] studied the mechanism of the antimicrobial action of carvacrol and its synergy with antibiotics and established its activity against the formation of biofilms, which is an important aspect of the fight against fungi and gram-positive and gram-negative bacteria.
Waller et al. [
21] described estragole, 1,8-cineole, terpineol-4 and gamma-terpinene isolated from plant species of the Lamiaceae family that exhibit a pronounced fungicidal activity against the pathogenic fungi
Aspergillus spp.,
Candida spp.,
Malassezia spp.,
Cryptococcus spp.,
Sporothrix spp.,
Microsporum spp.,
Trichophyton spp. and
Epidermophyton spp., and highlighted their potential use as antifungal agents for mycoses. In addition to being antimicrobial, oregano essential oils have both antidiabetic and anticancer activities [
22,
23]. Mahmoudian-Sani et al. [
24] found that
O. vulgare can stimulate the function of the T
reg cells (defined as CD4+CD25+Foxp3+), which play an important role in maintaining self-tolerance and preventing autoimmune diseases. The anticancer properties of carvacrol through its effects on proapoptotic processes have been reported in preclinical models of breast, liver and lung carcinomas [
18].
The genus
Scutellaria includes about 350 plant species, which contain over 295 different compounds. Among them, flavonoids and diterpenes are the most interesting for pharmacology [
25,
26,
27].
Scutellaria baicalensis Georgi (Baikal skullcap or Chinese skullcap) is a perennial species of the Lamiaceae family, growing up to 60 cm high. The species is distributed across China, Korea, Mongolia and the Russian Far East and Siberia [
28]. The phytochemistry and pharmacology of plants of the genus
Scutellaria, particularly the species
S. baicalensis,
S. barbata and
S. lateriflora, have been attracted the interest of many researchers in recent decades. However, so far, many of the results of traditional and clinical research on these plants have not been systematized and summarized [
29,
30].
Scutellaria baicalensis is referred to in the
American Herbal Pharmacopoeia and regarded as an adaptogen [
31].
The root of
S. baicalensis has long been widely used in traditional Chinese medicine and contains several major biologically active compounds, including wogonin, scutellarein, baicalein and baicalin [
32,
33,
34]. Qing Fei Pai Du Tang (QFPDT), a Chinese medical formula composed of 21 herb from five classical formulae, was effective against COVID-19 in 10 provinces of mainland China [
35]. According to the report, QFPDT prevented the progression of mild cases and decreased the average duration of symptoms and hospital stays. Therefore, it is recommended in the 6th and 7th editions of the Clinical Practice Guidelines for the Treatment of COVID-19 in China. QFPDT and its constituent herbs, including
S. baicalensis, were reviewed by Zhong et al. [
35]. The roots of
S. baicalensis are used to prevent and treat type-2 diabetes, atherosclerosis, hypertension, hyperlipidemia, dysentery and respiratory diseases [
36].
The review of Li et al. [
37] provides an overview of the different separation, detection and identification methods employed for the quantitative and qualitative determination of the active components of
S. baicalensis.
Scutellaria baicalensis contains around 53 polyphenols in total, including 50 flavonoids and 3 stilbenes. As the active chemicals of the
S. baicalensis root, 18 polyphenols showed apparent anticancer activities [
38,
39,
40].
The main bioactive flavonoids of
S. baicalensis are baicalein and baicalin. Due to its anti-inflammatory and immunomodulatory effects, baicalin has been successfully used in the treatment of cerebrovascular and neurological diseases, which has been confirmed by numerous in vivo and in vitro studies [
41]. Baicalin can penetrate the blood–brain barrier and be distributed in the nuclei of the brain [
42,
43]. In recent years, a number of researchers have shown that baicalin has powerful neuroprotective effects in various models of neuronal injuries [
44,
45].
The neuroprotective effects of baicalin and baicalein have been confirmed by a number of studies on many models of ischemic neuronal injuries in vitro and in vivo [
44,
45]. Pharmacological mechanisms have shown that these substances exhibit several biological activities, including antioxidant, anti-apoptotic, anti-inflammatory and anti-excitotoxicity activities, the protection of the mitochondria, and the promotion of the expression of neuronal protective factors as well as the effects of adult neurogenesis, etc. [
42]. Zhang et al. [
46] described the potential biochemical mechanisms of action of skullcap flavonoids in spinal cord injuries. The therapeutic potential of baicalein has been demonstrated in the cases of Parkinson’s and Alzheimer’s diseases due to its key pharmacological properties— the reduction of oxidative stress, anti-inflammatory action, inhibition of the aggregation of disease-specific amyloid proteins, the stimulation of neurogenesis and differentiation and its anti-apoptotic activity [
47]. Hussain et al. [
48] reviewed the active components of different
Scutellaria species and their mechanisms of action to prevent chronic neurological disorders that involve neuroinflammation and neurodegeneration. Ashbaugh & McGrew [
49] examined the most recent evidence regarding
S. baicalensis and the supplements and vitamins used for the treatment and prevention of concussions. The neuroprotective activity of the plant itself and its isolated flavonoids in various neurodegenerative diseases modelled in vitro and in vivo were also indicated by Gaire et al. [
50].
The non-toxicity of different doses of flavones isolated from the skullcap root, when used as neuroprotective substances for the brain, was demonstrated in studies by Gasiorowski et al. [
51]. Their neuroprotective properties have been illustrated in the cases of oxidative stress-induced as well as amyloid-beta- and alpha-synuclein-induced neuronal death models. Baicalein inhibits the aggregation of neuronal amyloidogenic proteins and causes the dissolution of the formed amyloid. Baicalein also exhibits strong antioxidant and anti-inflammatory effects and possesses anticonvulsant, anxiolytic and mild sedative actions, while significantly enhancing the cognitive and mnestic functions in animal models of ageing brains and neurodegeneration [
52]. The toxicity of
S. baicalensis mainly manifested in liver fibrosis and allergic reactions and was mainly caused by baicalin [
31].
The anti-inflammatory, anti-cancer and anti-viral effects of baicalin are associated with its regulatory effect on the immune system [
52]. Baicalin improves chronic inflammation, immune imbalance, lipid metabolism disorders, apoptosis and oxidative stress, thus playing an important role in preventing the onset and progression of cardiovascular system diseases (atherosclerosis, hypertension, myocardial infarction and heart failure) [
53]. Chien et al. [
54] compiled and discussed the biological analytical methods of using herbal medicines containing
S. baicalensis and other plant species for the protection of the liver.
Numerous collected data show that
S. baicalensis exhibits a powerful anticancer activity. A review by Li-Weber [
55] summarized the results obtained over the past 20 years and highlighted the main molecular mechanisms of its action.
Park et al. [
56] attributed the relief of symptoms of cancer cachexia through
S. baicalensis to the plant’s anti-inflammatory effects, regulation of the neuroendocrine pathway and modulation of the ubiquitin-proteasome system, or protein synthesis. Zhang et al. [
57] associated the antitumor effect of
S. baicalensis with the mechanism of angiogenesis inhibition through the inhibition of cytokines (vascular endothelial growth factor), which initiate this process. The extracts and basic flavonoids of
S. baicalensis have shown anti-cancer activities in several cancer cell lines in vitro and in vivo [
58]. In a review, Bie et al. [
59] summarized the various anti-cancer effects of baicalein on hepatocellular carcinoma (HCC) and their underlying molecular mechanisms based on the in vitro and in vivo experimental evidence discovered thus far. In light of this research, baicalein may be developed as a potential, novel anticancer drug for HCC treatment. Liu et al. [
60] also associated the antitumor activity of baicalein with its ability to inhibit cyclin complexes in the regulation of the cell cycle and scavenging free radicals, to attenuate enzyme activity (mitogen-activated protein kinase (MAPK), protein kinase B (Akt), or the mammalian target of rapamycin (mTOR)) to induce apoptosis by activating caspase-9/-3, and to inhibit tumor invasion and metastasis by reducing the expression of matrix metalloproteinase-2/-9 (MMP-2/-9) [
60]. Baicalein induces apoptosis in cancer cells and induces cell cycle arrest, as indicated by Gao et al. [
61]. Chen et al. [
62] considered the anticancer activities of baicalin and its aglycone baicalein through their effects on signaling pathways and their potential biological mechanisms in different types of hematologic malignancies, as well as their combined effects as chemotherapeutic adjuvants in the treatment of these diseases.
Although the chemopreventive properties of baicalin and baicalein are significant and associated with the induction of apoptosis, triggering autophagy, cell cycle arrest, the suppression of metastasis and the inhibition of 12-lipoxygenase, they are characterized by poor solubility and low oral bioavailability; thus, their clinical use is limited [
63]. Restrictions on the use and continued research on the mechanisms of action of
S. baicalensis flavonoids are required due to the fact that they cause an increase in the activity of the immune system [
64,
65,
66]. At the same time, it is known that many diseases, including tumors and neurodegenerative diseases (Alzheimer’s disease), are accompanied by inflammatory reactions, and the inflammation of the neurons is often the result of excessive immune activity [
67,
68,
69].
Scutellaria baicalensis root flavonoids can only aggravate the course of these diseases [
70].
Despite numerous reports on the pharmacological effects of baicalin and baicalein, some reports have indicated that baicalin, due to its form, is unlikely to be absorbed in the intestine, and that it must be pre-hydrolyzed to baicalein for absorption to take place [
71,
72]. Most likely, the pharmacokinetic characteristics of baicalin and baicalein are influenced by the intestinal microflora [
73,
74].
Wogonin is a widely studied product obtained from the roots of the plant
S. baicalensis. Its popularity stems from its antioxidant, antiviral, anti-inflammatory, anti-allergic and anti-thrombotic properties, leading to its applications in the treatment of cardiovascular disease [
75,
76,
77]. Experimental and clinical data prove that wogonin exhibits a variety of biological activities, such as anti-cancer and anti-inflammatory activities, and it is used to treat bacterial and viral diseases [
78]. Wogonin is safe, exhibits negligible organ toxicity at high doses and is non-toxic with long-term intravenous use, facilitating apoptosis and endoplasmic reticulum (ER) stress in the cells, suggesting its potential for use as a therapeutic agent in the treatment of diabetic cardiomyopathy [
75,
76,
77,
78].
The objective of this study was to determine the general effects of S. baicalensis and O. vulgare on the growth, changes in the organ mass index, biochemical and morphological blood parameters, and functional state of the nervous system of white outbred laboratory rats fed on a high-fat content diet.
2. Materials and Methods
The choice of animals for the experiment, the research protocols, and the withdrawal of animals from the experiment were approved by the local ethical committee of Dnipro State Agrarian and Economic University (Dnipro, Ukraine). The maintenance, nutrition care of the animals and their withdrawal from the experiment were carried out in accordance with the principles set forth in the “European Convention for the Protection of Vertebrate Animals used for Experimental or other Scientific Purposes” (Strasbourg, France, 18 March 1986, ETS No. 123) and in Law of Ukraine “On protection of animals from cruel treatment" (Kyiv, Ukraine, 21 February 2006, No. 3447-IV).
A total of 24 outbred male laboratory rats of 1.5 months of age were randomly divided into three groups (8 rats/group), so that the average body weight was the same for each group. The body weight range did not exceed ±20% of the average body weight at the beginning of the experiment. The rats were kept in polycarbonate cages with steel lattice lids and a feeding recess, with 4 individuals per cage. The rats were kept in a room at a temperature of 20–22 °C and relative humidity of 50–65%. The light regimen was 12 h of light and 12 h of darkness. According to this regime, ventilation was provided. The animals received water
ad libitum [
79].
From the first day of the experiment, the diet of all animals had an excess fat content. The high-fat diet was based on a standard diet (75% grain mixture (corn, sunflower grain, wheat, barley), 8% root crops (potatoes, carrots), 2% meat and bone meal and 2% mineral-vitamin complex)) with the introduction of 15% sunflower oils [
80,
81]. The control group animals received a high-fat diet, and the experimental group received a high-fat diet with the addition of medicinal plants. The high-fat diet of the first experimental group was supplemented with 5% dry
O. vulgare leaves, and the second experimental group was supplemented with 5% crushed
S. baicalensis roots.
Origanum vulgare plants were collected from the Botanical Garden of the Oles Honchar Dnipro National University (48.435509 °N, 35.043132 °E) on 15 June 2021. The plants were dried under a tent with no exposure to direct sunlight. The above-ground part was fragmented and all rough parts were removed, leaving only the leaves and soft parts of the inflorescences.
The roots of S. baicalensis were dug out on 20 November 2021 in the Botanical Garden of the Oles Honchar National University. The roots were cleaned of soil, rinsed, dried and fragmented with a knife into small pieces no larger than 2–3 mm. The obtained plant material was dried to an air-dried condition for two days in a dark, dry room at the temperature of 25 °C. After drying, fragments of the roots of S. baicalensis were 1.2–2.0 mm and were not fragmented further.
The main ingredients of the rats’ diet were ground in a mill (grain, meat and bone meal, mineral and vitamin complex, dry shoots of medicinal plants) and mixed, then sunflower oil was added and the granules were produced at the rate of 4200 g for each group for the entire period of the experiment (30 days). Root crops, in the appropriate amount and in fresh form, were additionally given every day. The animals had free access to food. The rats were observed daily and their body weight, food intake and water intake were recorded. Morphometric parameters (live body weight, abdominal volume) were determined on the first day of the experiment and on the 30th day [
82]. The total body weight gain of the animals and the daily gain in live weight were calculated. The average body weight per group was calculated weekly based on the individual animals.
The orientation-physical activity and emotional status of the experimental animals were studied in an “open field” test. We used an installation consisting of a square area of 1 m
2, divided into 16 squares and limited by an opaque wall that was 20 cm high. The experiment was conducted in complete silence, with intense lighting of the field itself. The experimental animal, taken from a cage in a previously darkened room, was placed in the center of the field. The exposure time was 2 min. The animals were tested for four days (days 1–4) at the beginning of the experiment and four days at the end (days 26–30). The number of crossed squares was counted. The parameters included: the peripheral and central floorspace—for assessing the rats’ physical activity; the peripheral (leaning against the wall) and central (without leaning against the wall) racks—for the orientation activity; and the number of acts of grooming, defecation and urination—for assessing the animals’ emotional status [
83].
The euthanasia of the animals was performed on the 30th day of the experiment under anesthesia (80 mg/kg ketamine and 12 mg/kg xylazine, intraperitoneally) by total bloodletting from the heart. After autopsy, the condition of the internal organs was visually assessed for the presence of pathological changes [
84]. Selection of organs and tissues (heart, liver, lungs, thymus, spleen, stomach, small and large intestines, kidneys) was performed with surgical instruments. The mass of the internal organs was determined with an accuracy of ±10 mg.
Blood samples taken during euthanasia were used for biochemical and hematological analyses. Biochemical parameters were determined using an automatic analyzer, Miura 200 (I.S.E. Srl, Rome, Italy), and High Technology reagent kits (High Technology Inc, North Attleborough, MA, USA), PZ Cormay S.A. (Cormay Diagnostics, Lublin, Poland) and Spinreact S.A. (Spinreact, Girona, Spain). The numbers of erythrocytes and leukocytes in the stabilized blood of the mice were determined using an automatic hematology analyzer, BC-2800Vet (Mindray, Shenzhen, China). Pappenheim blood smears were prepared for the leukogram, followed by Romanovsky–Gimza staining. The numbers of erythrocytes and leukocytes in the stabilized blood of the mice were determined using an automatic hematology analyzer, BC-2800Vet, and Mindray [
81].
The data were analyzed using the Statistica 8.0 program (StatSoft Inc., Tulsa, OK, USA). The tables demonstrate the results as x ± SD (mean ± standard deviation). Differences between the values of the control and experimental groups were determined using Tukey’s test (with consideration of Bonferroni’s correction), where the differences were considered significant at p < 0.05.
3. Results
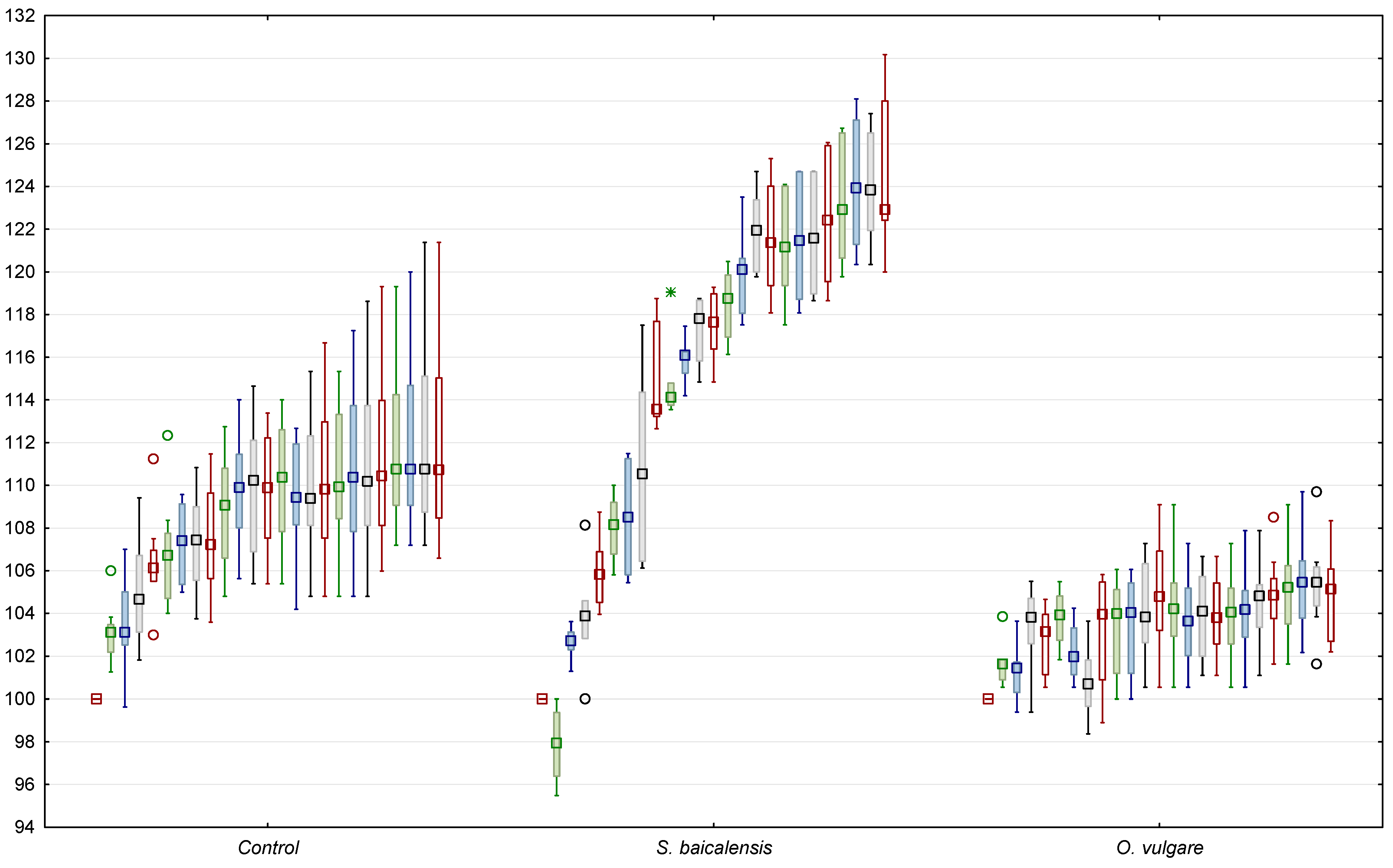
In the control group, the body weight increased to 111.5% from baseline, and in the group of rats fed on the dry shoots of
O. vulgare, it increased to only 105.5% (
Figure 1). Contradictory changes in body weight, compared to the control group, were observed in the animals fed with crushed roots of
S. baicalensis. Their body weight increased to 124.0% relative to their initial weight, i.e., more than in the control group (
Figure 1).
When the dry grass of
O. vulgare was added to the diet of the animals, their feed intake decreased to 80.0% compared to the control group (Table 3). The addition of the dry roots of
S. baicalensis to the feed did not affect the rate of feed consumption but increased the amount of daily water intake to 116.0% compared to the rats in the control group. The daily weight gain in the rats fed with the
O. vulgare supplementation decreased to 205 mg/day, while rats fed with the supplementation of
S. baicalensis, on the contrary, exhibited a daily weight gain increased to 1417 mg/day compared to the 700 mg/day in the control group (
Table 1). Significant changes in the abdominal volumes of the experimental groups of animals were not observed.
The addition of the
O. vulgare dry grass to the diet of animals did not cause significant changes in the relative mass of their organs (
Table 2). The addition of
S. baicalensis roots to the diet over the period of the increase in the total body weight significantly reduced the relative weight of the thymus (to 50.0%) and the brain (to 74.6% of the control group level,
Table 2).
Under the influence of
O. vulgare, added to the diet of the rats, their blood biochemical parameters changed (
Table 3,
Table 4 and
Table 5). In the rats’ blood plasma, the activity of alkaline phosphatase significantly increased (223.8% compared to the control group) and gamma-glutamyl transferase decreased (to a level of 72.3% compared to the control group). In rats consuming
O. vulgare dry grass, the normalization of the lipid metabolism was observed: the atherogenic index decreased to 33.7% of the control group value, due to an increase in the concentration of high-density lipoprotein cholesterol (increased to 165.8% compared to the value of the control group). The concentration of triglycerides in the blood plasma also sharply decreased (to 38.3% compared to the control). The normalization of the metabolism was also evidenced by a significant decrease in the concentration of glucose in the blood of the animals (ranging from 7.4 to 5.0 mmol/L on average) under the influence the herb
O. vulgare.
The roots of
S. baicalensis caused similar changes in the blood biochemical parameters of the male rats. Compared to the changes induced by oregano (
Table 5), the activity of alkaline phosphatase increased even more strongly (to 444.1% of the control group); however, changes in the activity of gamma-glutamyl transferase were insignificant (enzyme activity tended to decrease). In addition, the concentration of urea and blood urea nitrogen significantly decreased (to 71.4% and 71.5% compared with the control group, respectively). The atherogenic index significantly decreased (to 35.5% of the control) and the concentration of high-density lipoprotein cholesterol increased (to 164.2% of the control group value). Moreover, as with the use of oregano, under the influence of the dry crushed roots of
S. baicalensis, the concentration of triglycerides in the blood plasma decreased by more than twice (46.2% of the control group), and the concentration of glucose decreased (to 58.2% compared to the control group, or to 4.3 mmol/L,
Table 3).
When added to the diet,
O. vulgare was not observed in the general analysis of the blood and leukogram of the male rats (
Table 6). The roots of
S. baicalensis contributed to a significant decrease in the number of platelets in the blood of the animals (to 64.7% of the control group) and a sharp increase in the number of leukocytes (to 170.4% of the control group,
Table 4).
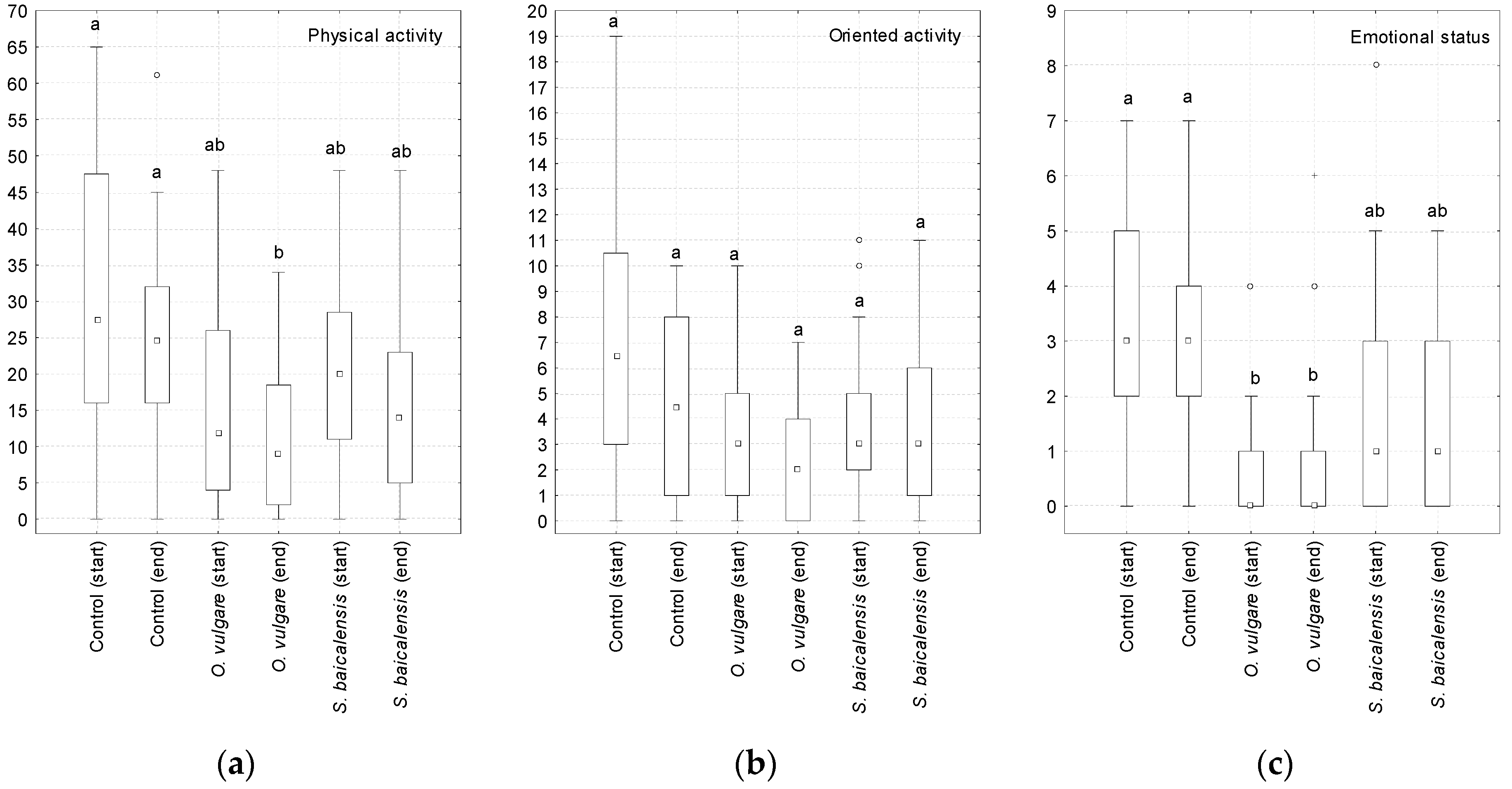
The physical activity (
Figure 2a) of the rats showed a slight tendency to decrease when
O. vulgare grass and
S. baicalensis roots were added to their diet. No significant changes in the orientation activity were observed (
Figure 2b). Both plants studied contributed to a decrease in the emotional status of the animals, which was most pronounced when oregano grass was added to the feed (
Figure 2c).
There were no statistically reliable differences between animal groups (
Table 7). Nevertheless, it should be mentioned that there was a prominent decrease in the average fecal boluses (almost 10 times less) and the frequency of urination (more than 3 times lower compared to the control group) when supplementing the rats’ diet with the
O. vulgare herb (
Table 7).
4. Discussion
In our study, we determined the effects of
O. vulgare and
S. baicalensis on the growth and development of laboratory animals in the context of their consumption of a high-fat diet. The lack of unified methods for extracting biologically active compounds from medicinal herbs or medicinal raw materials, and the use of various methods for their extraction, makes it very difficult to carry out the procedure for identifying and determining the composition of plant objects [
85,
86,
87,
88,
89]. Therefore, in our study, we selected the strategy of mixing dry crushed plant parts into granulated animal feed.
The research results indicate that the studied medicinal plants significantly affected the body weight gain of the laboratory animals in different ways. Thus, the addition of
O. vulgare grass to the diet significantly slowed down the weight gain, probably due to the fact that the amount of food consumed was reduced to 80.0% compared with the control group. The average daily weight gain of the rats was lower than in the control group and significantly lower than in the group of rats that consumed the roots of
S. baicalensis. The addition of
S. baicalensis dry roots to the high-fat diet of the rats, on the contrary, led to more active growth, and their average daily weight gain was at the level of 1417 mg/day, which was higher than that in the control group of animals. The decrease in the rats’ body weight and the average daily feed intake when dry chopped grass
O. vulgare was added to the diet may be associated with the high content of essential oils in the plant and, as a result, its unpleasant taste. However, preliminary palatability studies of rats showed that the maximum dose that animals consumed, when added to neutral gelatin, was 200 mg/kg of pure
O. vulgare essential oil [
89]. In the same study, it was found that different doses (50, 100 and 200 mg/kg per day) of this essential oil did not lead to significant differences in the final weight, body weight gain or total food intake between groups.
At the end of the experiment, no pathological changes in the internal organs of the studied animals were found. The absence of pathomorphological changes in the organs of animals under the influence of various doses (up to 200 mg/kg of body weight) of
O. vulgare essential oils in their food was also observed in the studies of Llana-Ruiz-Cabello et al. [
89] and Horky et al. [
8].
It was determined that the addition of
O. vulgare to the rats’ high-fat diet across the 30-day experiment did not cause significant changes in the relative mass of the individual internal organs. At the same time, the addition of
S. baicalensis roots over the period of the general increase in body weight caused a decrease in the relative mass of the thymus, which can be explained by the active delymphotization of this organ, which is central to immunogenesis. The regulatory and stimulating effects on the immune system of the main active flavonoid, baicalin, has been observed in many studies [
36,
52,
53]. The significant neuroprotective effects of the
S. baicalensis active substances, which are actively used in the treatment of neurological and cerebrovascular diseases, has long been known. Possibly, this is the reason for the decrease in the relative mass of the brains in the rats that received
S. baicalensis roots in their diet, but more thorough and in-depth studies are required to confirm this. When analyzing the blood biochemical parameters of the animals that were consuming
O. vulgare, it was found that the lipid metabolism was normalized in comparison with the control group (on the high-fat diet). Thus, the atherogenic index decreased to 33.7% of the value of the control group, due to an increase in the concentration of high-density lipoprotein cholesterol, and the concentration of triglycerides decreased. Similar changes in the lipid metabolism were also observed in the rats which were fed
S. baicalensis. In the blood, the atherogenic index also decreased, and the level of high-density lipoproteins increased in the context of a decrease in the triglycerides. According to Abd Rashed et al. [
90], the attenuating effects of essential oils, including
O. vulgare, on obesity can be explained by several possible mechanisms, which include anti-lipase activity, an increased plasma glycerol concentration, and the inhibition of fat accumulation. Both plants have also been found to lower blood glucose levels.
Origanum vulgare is among the medical species that have been used in Russia for the management of diabetes, especially when this is accompanied by obesity [
3]. The hypoglycemic effects of the active substances of
O. vulgare across a 90-day experiment were also demonstrated in the studies of Horky et al. [
8].
Attention is drawn to the significant increase in the activity of the enzymes in the blood, while the AST and ALT were increased in the blood of animals of all groups, including the control, which can be explained by the significant load on the liver hepatocytes due to excess fat in the rats’ diet and the increased work required for its metabolism [
80,
81,
83]. At the same time, the activity of alkaline phosphatase was significantly increased in the blood of the animals that received the tested plants in addition to the diet. It should be noted that the indicators of AP activity did not transgress the physiological limits for this animal species.
When added to the diet,
O. vulgare was not observed in the complete blood count and leukogram of the male rats. At the same time, the addition of the dry roots of
S. baicalensis to the feed of the rats led to a significant decrease in the number of platelets and an increase in the number of leukocytes compared with animals in the control group. The antithrombotic effect of wogonin, the active substance of
S. baicalensis, has been indicated in some publications, which has enabled the use of this medicinal plant in the treatment of cardiovascular diseases [
75].
Baicalein has the most pronounced neuroprotective activity [
91,
92]. It was shown in a meta-analysis to significantly improve the neurobehavioral functions in animal models of Parkinson’s disease, in experiments including the spontaneous locomotor test, the pole test, the spinning pole test, the apomorphine-induced rotation test, the grid test and the tremor test [
93]. In our study, based on an open field test of healthy rats who received a high-fat diet, the locomotor activity in the group of animals consuming the additional crushed roots of
S. baicalensis did not significantly differ from the control group and the animals consuming
O. vulgare grass.