A Narrative Review of the Potential Roles of Lipid-Based Vesicles (Vesiculosomes) in Burn Management
Abstract
1. Introduction
1.1. Burn Degrees
1.2. Skin Substitutes
1.3. Current Local Treatment of Burns
1.4. Vesicular Drug Delivery Systems
2. Methodology
3. Vesiculosomes in Burn Management
3.1. Liposomes
3.2. Cubosomes
3.3. Niosomes
3.4. Ethosomes
3.5. Transfersomes
3.6. Phytosomes
4. Natural Products
5. Current Status and Future Concepts
6. Discussion and Expert Opinion
7. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Jeschke, M.G.; van Baar, M.E.; Choudhry, M.A.; Chung, K.K.; Gibran, N.S.; Logsetty, S. Burn injury. Nat. Rev. Dis. Prim. 2020, 6, 1–25. [Google Scholar] [CrossRef] [PubMed]
- Hao, D.; Qu, M.; Nourbakhsh, M. Experimental study of burn damage progression in a human composite tissue model. Biology 2021, 10, 40. [Google Scholar] [CrossRef]
- Neha Pathak, M. Treating Pain Caused by Burn: 1st, 2nd, and 3rd Degree. WebMed. Published. 2020. Available online: https://www.webmd.com/pain-management/guide/pain-caused-by-burn (accessed on 1 June 2022).
- Halim, A.S.; Khoo, T.L.; Mohd Yussof, S.J. Biologic and synthetic skin substitutes: An overview. Indian J. Plast. Surg. 2010, 43, S23–S28. [Google Scholar] [CrossRef] [PubMed]
- Ogundipe, K.O.; Adigun, I.A.; Solagberu, B.A. Economic burden of drug use in patients with acute burns: Experience in a developing country. J. Trop. Med. 2009, 2009, 734712. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Leppert, W.; Malec-Milewska, M.; Zajaczkowska, R.; Wordliczek, J. Transdermal and topical drug administration in the treatment f pain. Molecules 2018, 17, 681. [Google Scholar] [CrossRef]
- Norman, G.; Christie, J.; Liu, Z.; Westby, M.J.; Jefferies, J.M.; Hudson, T.; Edwards, J.; Mohapatra, D.P.; Hassan, I.A.; Dumville, J.C. Antiseptics for burns. Cochrane Database Syst. Rev. 2017, 12, CD011821. [Google Scholar] [CrossRef]
- Cancio, L.C. Topical antimicrobial agents for burn wound care: History and current status. Surg. Infect. 2021, 22, 3–11. [Google Scholar] [CrossRef]
- Abdul Rasool, B.K.; Hussain, F.N.; Bahrainwala, I.M.; Akbar, N.; Umar, S.; Kalady, S.P.; Shamsheer, Z. Advances in vaccine delivery strategies to promote effective immunization. J. Appl. Pharm. Sci. 2022, 12, 001–026. [Google Scholar] [CrossRef]
- Agarwal, S.; Muniyandi, P.; Maekawa, T.; Kumar, D.S. Vesicular systems employing natural substances as promising drug candidates for MMP inhibition in glioblastoma: A nanotechnological approach. Int. J. Pharm. 2018, 551, 339–361. [Google Scholar] [CrossRef]
- Rommasi, F.; Esfandiari, N. Liposomal Nanomedicine: Applications for drug delivery in cancer therapy. Nanoscale Res. Lett. 2021, 16, 95. [Google Scholar] [CrossRef]
- Lamichhane, N.; Udayakumar, T.S.; D’Souza, W.D.; Simone, C.B., 2nd; Raghavan, S.R.; Polf, J.; Mahmood, J. Liposomes: Clinical Applications and Potential for Image-Guided Drug Delivery. Molecules 2018, 23, 288. [Google Scholar] [CrossRef] [PubMed]
- He, H.; Lu, Y.; Qi, J.; Zhu, Q.; Chen, Z.; Wu, W. Adapting liposomes for oral drug delivery. Acta Pharm. Sin. B 2019, 9, 36–48. [Google Scholar] [CrossRef] [PubMed]
- Dubald, M.; Bourgeois, S.; Andrieu, V.; Fessi, H. Ophthalmic drug delivery systems for antibiotherapy—A review. Pharmaceutics 2018, 13, 10. [Google Scholar] [CrossRef] [PubMed]
- Hong, S.S.; Oh, K.T.; Choi, H.G.; Lim, S.J. Liposomal formulations for nose-to-brain delivery: Recent advances and future perspectives. Pharmaceutics 2019, 17, 540. [Google Scholar] [CrossRef]
- Dai, T.; Huang, Y.Y.; Sharma, S.K.; Hashmi, J.T.; Kurup, D.B.; Hamblin, M.R. Topical antimicrobials for burn wound infections. Recent Pat. Antiinfect. Drug Discov. 2010, 5, 124–151. [Google Scholar] [CrossRef]
- Matei, A.-M.; Caruntu, C.; Tampa, M.; Georgescu, S.R.; Matei, C.; Constantin, M.M.; Constantin, T.V.; Calina, D.; Ciubotaru, D.A.; Badarau, I.A.; et al. Applications of nanosized-lipid-based drug delivery systems in wound care. Appl. Sci. 2021, 11, 4915. [Google Scholar] [CrossRef]
- Ghasemiyeh, P.; Mohammadi-Samani, S. Potential of nanoparticles as permeation enhancers and targeted delivery options for skin: Advantages and disadvantages. Drug Des. Devel. Ther. 2020, 14, 3271–3289. [Google Scholar] [CrossRef]
- Lin, D.M.; Koskella, B.; Lin, H.C. Phage therapy: An alternative to antibiotics in the age of multi-drug resistance. World J. Gastrointest. Pharm. Ther. 2017, 8, 162–173. [Google Scholar] [CrossRef]
- Duplessis, C.A.; Biswas, B. A review of topical phage therapy for chronically infected wounds and preparations for a randomized adaptive clinical trial evaluating topical phage therapy in chronically infected diabetic foot ulcers. Antibiotics 2020, 9, 377. [Google Scholar] [CrossRef]
- Chhibber, S.; Kaur, J.; Kaur, S. Liposome entrapment of bacteriophages improves wound healing in a diabetic mouse MRSA infection. Front. Microbiol. 2018, 9, 561. [Google Scholar] [CrossRef]
- Chadha, P.; Katare, O.P.; Chhibber, S. Liposome loaded phage cocktail: Enhanced therapeutic potential in resolving Klebsiella Pneumoniae mediated burn wound infections. Burn 2017, 43, 1532–1543. [Google Scholar] [CrossRef] [PubMed]
- Li, Z.; Liu, M.; Wang, H.; Du, S. Increased cutaneous wound healing effect of biodegradable liposomes containing madecassoside: Preparation optimization, in vitro dermal permeation, and in vivo bioevaluation. Int. J. Nanomed. 2016, 11, 2995–3007. [Google Scholar] [CrossRef] [PubMed]
- Parchen, G.P.; Jacumazo, J.; Koop, H.S.; Biscaia, S.M.P.; Trindade, E.S.; Silveira, J.L.M.; de Freitas, R.A. Modulation of epidermal growth factor release by biopolymer-coated liposomes. J. Pharm. Sci. 2020, 109, 2294–2301. [Google Scholar] [CrossRef]
- Choi, J.U.; Lee, S.W.; Pangeni, R.; Byun, Y.; Yoon, I.S.; Park, J.W. Preparation and in vivo evaluation of cationic elastic liposomes comprising highly skin-permeable growth factors combined with hyaluronic acid for enhanced diabetic wound-healing therapy. Acta Biomater. 2017, 57, 197–215. [Google Scholar] [CrossRef] [PubMed]
- Hurler, J.; Berg, O.A.; Skar, M.; Conradi, A.H.; Johnsen, P.J.; Skalko-Basnet, N. Improved burn therapy: Liposomes-in-hydrogel delivery system for mupirocin. J. Pharm. Sci. 2012, 101, 3906–3915. [Google Scholar] [CrossRef]
- Wadhwa, S.; Singh, B.; Sharma, G.; Raza, K.; Katare, O.P. Liposomal fusidic acid as a potential delivery system: A new paradigm in the treatment of chronic plaque psoriasis. Drug Deliv. 2016, 23, 1204–1213. [Google Scholar] [CrossRef]
- Nicolosi, D.; Cupri, S.; Genovese, C.; Tempera, G.; Mattina, R.; Pignatello, R. Nanotechnology approaches for antibacterial drug delivery: Preparation and microbiological evaluation of fusogenic liposomes carrying fusidic acid. Int. J. Antimicrob. Agents 2015, 45, 622–626. [Google Scholar] [CrossRef]
- Lichtenstein, A.; Margalit, R. Liposome-encapsulated silver sulfadiazine (SSD) for the topical treatment of infected burn: Thermodynamics of drug encapsulation and kinetics of drug release. J. Inorg. Biochem. 1995, 60, 187–198. [Google Scholar] [CrossRef]
- Patel, D.; Patel, N. Fabrication and characterization of sterically stabilized liposomes of topotecan. Futur J. Pharm. Sci. 2020, 6, 1–18. [Google Scholar] [CrossRef]
- Guan, J.; Shen, Q.; Zhang, Z.; Jiang, Z.Y.M.; Qian, J.; Lu, W.; Zhan, C. Enhanced immunocompatibility of ligand-targeted liposomes by attenuating natural IgM absorption. Nat. Commun. 2018, 9, 1–11. [Google Scholar] [CrossRef]
- Homann, H.H.; Rosbach, O.; Moll, W.; Vogt, P.M.; Germann, G.; Hopp, M.; Langer-Brauburger, B.; Reimer, K.; Steinau, H.U. A liposome hydrogel with polyvinylpyrrolidone iodine in the local treatment of partial-thickness burn wounds. Ann. Plast. Surg. 2007, 59, 423–427. [Google Scholar] [CrossRef] [PubMed]
- Peng, S.; Zou, L.; Liu, W.; Li, Z.; Liu, W.; Hu, X.; Chen, X.; Liu, C. Hybrid liposomes composed of amphiphilic chitosan and phospholipid: Preparation, stability and bioavailability as a carrier for curcumin. Carbohydr. Polym. 2017, 156, 322–332. [Google Scholar] [CrossRef] [PubMed]
- Liu, W.; Liu, J.; Liu, W.; Li, T.; Liu, C. Improved physical and in vitro digestion stability of a polyelectrolyte delivery system based on layer-by-layer self-assembly alginate-chitosan-coated nanoliposomes. J. Agric. Food Chem. 2013, 61, 4133–4144. [Google Scholar] [CrossRef] [PubMed]
- Kozhikhova, K.V.; Ivantsova, M.N.; Tokareva, M.I.; Shulepov, I.D.; Tretiyakov, A.V.; Shaidarov, L.V.; Rusinov, V.L.; Mironov, M.A. Preparation of chitosan-coated liposomes as a novel carrier system for the antiviral drug Triazavirin. Pharm. Dev. Technol. 2018, 23, 334–342. [Google Scholar] [CrossRef]
- Abdul Rasool, B.K.; Shehab, N.G.; Khan, S.A.; Bayoumi, F.A. A new natural gel of Fagonia indica Burm f. extract for the treatment of burn on rats. Pak. J. Pharm. Sci. 2014, 27, 73–81. [Google Scholar]
- Zadeh, B.S.M.; Zamin, B.K. The effect of chitosan coating on mafenide acetate-loaded liposome characterization and delivery through burned rat skin. Asian J. Pharm. Clin. Res. 2019, 12, 212–217. [Google Scholar] [CrossRef]
- Hao, J.; Guo, B.; Yu, S.; Zhang, W.; Zhang, D.; Wang, J.; Wang, Y. Encapsulation of the flavonoid quercetin with chitosan-coated nano-liposomes. Food Sci. Technol. 2017, 85, 37–44. [Google Scholar] [CrossRef]
- Price, C.I.; Horton, J.W.; Baxter, C.R. Topical liposomal delivery of antibiotics in soft tissue infection. J. Surg. Res. 1990, 49, 174–178. [Google Scholar] [CrossRef]
- Nunes, P.S.; Rabelo, A.S.; Souza, J.C.; Santana, B.V.; da Silva, T.M.; Serafini, M.R.; Dos Passos Menezes, P.; Dos Santos Lima, B.; Cardoso, J.C.; Alves, J.C.; et al. Gelatin-based membrane containing usnic acid-loaded liposome improves dermal burn healing in a porcine model. Int. J. Pharm. 2016, 513, 473–482. [Google Scholar] [CrossRef]
- Laurent, A.; Pantet, O.; Laurent, L.; Laurent, L.; Hirt-Burri, N.; Roessingh, A.B.; Raffoul, W.; Laurent, P.; Monod, M.; Applegate, L.A. Potency and stability of liposomal Amphotericin B formulated for topical management of Aspergillus spp. infections in burn patients. Burn Open 2020, 4, 110–116. [Google Scholar] [CrossRef]
- Değim, Z.; Çelebi, N.; Alemdaroğlu, C.; Deveci, M.; Öztürk, S.; Özoğul, C. Evaluation of chitosan gel containing liposome-loaded epidermal growth factor on burn wound healing. Int. Wound J. 2011, 8, 343–354. [Google Scholar] [CrossRef] [PubMed]
- Boyd, A.N.; Blair, M.E.; Degenkolb, K.E.; Foster, D.R.; Hartman, B.C.; Sood, R.; Walroth, T.A. A prospective analysis describing the innovative use of liposomal bupivacaine in burn patients. Burn 2020, 46, 370–376. [Google Scholar] [CrossRef] [PubMed]
- Karami, Z.; Hamidi, M. Cubosomes: Remarkable drug delivery potential. Drug. Discov. Today 2016, 21, 789–801. [Google Scholar] [CrossRef]
- Rarokar, N.R.; Khedekar, P.B. Cubosomes: A vehicle for delivery of various therapeutic agents. MOJ Toxicol. 2018, 4, 19–21. [Google Scholar] [CrossRef]
- Garg, G.; Saraf, S.; Saraf, S. Cubosomes: An overview. Biol Pharm. Bull. 2007, 30, 350–353. [Google Scholar] [CrossRef] [PubMed]
- Yang, Z.; Tan, Y.; Chen, M.; Dian, L.; Shan, Z.; Peng, X.; Wu, C. Development of amphotericin B-loaded cubosomes through the SolEmuls technology for enhancing the oral bioavailability. AAPS PharmSciTech 2012, 13, 1483–1491. [Google Scholar] [CrossRef]
- Yang, Z.; Peng, X.; Tan, Y.; Chen, M.; Zhu, X.; Feng, M.; Xu, Y.; Wu, C. optimization of the preparation process for an oral phytantriol-based Amphotericin B cubosomes. J. Nanomater. 2011, 2011, 308016. [Google Scholar] [CrossRef]
- Nasr, M.; Younes, H.; Abdel-Rashid, R.S. Formulation and evaluation of cubosomes containing colchicine for transdermal delivery. Drug Deliv. Transl. Res. 2020, 10, 1302–1313. [Google Scholar] [CrossRef]
- Han, S.; Shen Jq Gan, Y.; Geng, H.; Zhang, X.; Zhu, C.; Gan, L. Novel vehicle based on cubosomes for ophthalmic delivery of flurbiprofen with low irritancy and high bioavailability. Acta Pharm. Sin. 2010, 31, 990–998. [Google Scholar] [CrossRef]
- Cytryniak, A.; Nazaruk, E.; Bilewicz, R.; Górzyńska, E.; Żelechowska-Matysiak, K.; Walczak, R.; Mames, A.; Bilewicz, A.; Majkowska-Pilip, A. Lipidic cubic-phase nanoparticles (cubosomes) loaded with doxorubicin and labeled with 177Lu as a potential tool for combined chemo and internal radiotherapy for cancers. Nanomaterials 2020, 10, 2272. [Google Scholar] [CrossRef]
- Beena, P.; Varghese, S.A.; Alexander, J.A.; Raju, S.P.; Koshy, S.; Mathew, F.E.; Abraham, E. Formulation and evaluation of cubosomal gel of an anti-inflammatory agent. Res. J. Pharm. Tech. 2021, 14, 857–862. [Google Scholar] [CrossRef]
- Morsi, N.M.; Abdelbary, G.A.; Ahmed, M.A. Silver sulfadiazine-based cubosome hydrogels for topical treatment of burn: Development and in vitro/in vivo characterization. Eur. J. Pharm. Biopharm. 2014, 86, 178–189. [Google Scholar] [CrossRef] [PubMed]
- Mahlapuu, M.; Björn, C.; Ekblom, J. Antimicrobial peptides as therapeutic agents: Opportunities and challenges. Crit. Rev. Biotechnol. 2020, 40, 978–992. [Google Scholar] [CrossRef] [PubMed]
- Boge, L.; Browning, K.L.; Nordström, R.; Campana, M.; Damgaard, L.S.E.; Seth Caous, J.; Hellsing, M.; Ringstad, L.; Andersson, M. Peptide-loaded cubosomes functioning as an antimicrobial unit against Escherichia coli. ACS Appl. Mater. Interfaces 2019, 11, 21314–21322. [Google Scholar] [CrossRef] [PubMed]
- Boge, L.; Umerska, A.; Matougui, N.; Bysell, H.; Ringstad, L.; Davoudi, M.; Eriksson, J.; Edwards, K.; Andersson, M. Cubosomes post-loaded with antimicrobial peptides: Characterization, bactericidal effect and proteolytic stability. Int. J. Pharm. 2017, 526, 400–412. [Google Scholar] [CrossRef]
- Boge, L.; Hallstensson, K.; Ringstad, L.; Johansson, J.; Andersson, T.; Davoudi, M.; Larsson, P.T.; Mahlapuu, M.; Håkansson, J.; Andersson, M. Cubosomes for topical delivery of the antimicrobial peptide LL-37. Eur. J. Pharm. Biopharm. 2019, 134, 60–67. [Google Scholar] [CrossRef]
- Rahman, H.S.; Othman, H.H.; Hammadi, N.I.; Yeap, S.K.; Amin, K.M.; Abdul Samad, N.; Alitheen, N.B. Novel drug delivery systems for loading of natural plant extracts and their biomedical applications. Int. J. Nanomed. 2020, 15, 2439–2483. [Google Scholar] [CrossRef]
- Khalifa, A.M.; Abdul Rasool, B.K. Optimized mucoadhesive coated niosomes as a sustained oral delivery system of famotidine. AAPS PharmSciTech 2017, 18, 3064–3075. [Google Scholar] [CrossRef]
- Aparajay, P.; Dev, A. Functionalized niosomes as a smart delivery device in cancer and fungal infection. Eur. J. Pharm. Sci. 2022, 168, 106052. [Google Scholar] [CrossRef]
- Abdul Rasool, B.K.; Azeez, O.S.; Lootah, H.A.; Abusharbain, I.M.; Abu-Alhaj, H.A.; Nessa, F. Extended-release niosomal hydrogel for ocular targeting of piroxicam: In vitro and ex vivo evaluation. Br. J. Pharm. Res. 2014, 4, 2494–2510. [Google Scholar] [CrossRef]
- Durga, B.G.; Veera, L.P. Recent advances of nonionic surfactant-based nano-vesicles (niosomes and proniosomes): A brief review of these in enhancing transdermal delivery of drug. Future J. Pharm. Sci. 2020, 6, 100. [Google Scholar] [CrossRef]
- Muzzalupo, R.; Tavano, L. Niosomal drug delivery for transdermal targeting: Recent advances. Res. Rep. Transdermal. Drug Deliv. 2015, 4, 23–33. [Google Scholar] [CrossRef]
- Drugbank Online. Silver Sulfadiazine. Available online: https://go.drugbank.com/drugs/DB05245 (accessed on 18 October 2021).
- Dharashivkar, S.S.; Sahasrabuddhe, S.H.; Saoji, A.N. Niosomally encapsulated silver sulfadiazine gel for burn treatment. J. Microencapsul. 2015, 32, 137–142. [Google Scholar] [CrossRef] [PubMed]
- Sohrabi, S.; Haeri, A.; Mahboubi, A.; Mortazavi, A.; Dadashzadeh, S. Chitosan gel-embedded moxifloxacin niosomes: An efficient antimicrobial hybrid system for burn infection. Int. J. Biol. Macromol. 2016, 85, 625–633. [Google Scholar] [CrossRef]
- Yilmaz Atay, H. Antibacterial Activity of Chitosan-Based Systems. Funct. Chitosan 2020, 6, 457–489. [Google Scholar] [CrossRef]
- Farmoudeh, A.; Akbari, J.; Saeedi, M.; Ghasemi, M.; Asemi, N.; Nokhodchi, A. Methylene blue-loaded niosome: Preparation, physicochemical characterization, and in vivo wound healing assessment. Drug Deliv. Transl. Res. 2020, 10, 1428–1441. [Google Scholar] [CrossRef]
- Yucel, B.; Coruh, A.; Deniz, K. Salvaging the zone of stasis in burn by pentoxifylline: An experimental study in rats. J. Burn Care Res. 2019, 40, 211–219. [Google Scholar] [CrossRef]
- Aghajani, A.; Kazemi, T.; Enayatifard, R.; Amiri, F.T.; Narenji, M. Investigating the skin penetration and wound healing properties of niosomal pentoxifylline cream. Eur. J. Pharm. Sci. 2020, 151, 105434. [Google Scholar] [CrossRef]
- Muzzalupo, R.; Tavano, L.; Lai, F.; Picci, N. Niosomes containing hydroxyl additives as percutaneous penetration enhancers: Effect on the transdermal delivery of sulfadiazine sodium salt. Colloids Surf. B Biointerfaces 2014, 123, 207–212. [Google Scholar] [CrossRef]
- Nur Un, R.; Barlas, B.F.; Yavuz, M.; Seleci, D.A.; Seleci, M.; Gumus, Z.P.; Guler, E.; Demir, B.; Can, M.; Coskunol, H.; et al. Phyto-niosomes: In vitro assessment of the novel nanovesicles containing marigold extract. Int. J. Polym. Mater. Polym. Biomater. 2015, 64, 927–937. [Google Scholar] [CrossRef]
- Dharashivkar, S.; Sahasrabuddhe, S.; Saoji, A.N. Silver sulfadiazine niosomes: A novel sustained-release once-a-day formulation for burn treatment. Int. J. Pharm. Pharm. Sci. 2014, 6, 611–616. [Google Scholar]
- Bolatchiev, A.; Baturin, V.; Bazikov, I.; Maltsev, A.; Kunitsina, E. Effect of antimicrobial peptides HNP-1 and hBD-1 on Staphylococcus aureus strains in vitro and in vivo. Fundam Clin. Pharmacol. 2020, 34, 102–108. [Google Scholar] [CrossRef]
- Heidari, F.; Akbarzadeh, I.; Nourouzian, D.; Mirzaie, A.; Bakhshandeh, H. Optimization and characterization of tannic acid-loaded niosomes for enhanced antibacterial and anti-biofilm activities. Adv. Powder Technol. 2020, 31, 4768–4781. [Google Scholar] [CrossRef]
- Ali, A.; Sarhan, H.A.; Magdy, T. Preparation and characterization of phenytoin sodium niosomes for enhanced closure of skin injuries. Int. J. Pharm. Pharm. Sci. 2014, 6, 5426. [Google Scholar]
- Ali, M.; Abdel Motaal, A.; Ahmed, M.A.; Alsayari, A.; El-Gazayerly, O.N. An in vivo study of Hypericum perforatum in a niosomal topical drug delivery system. Drug Deliv. 2018, 25, 417–425. [Google Scholar] [CrossRef]
- Hallan, S.S.; Sguizzato, M.; Mariani, P.; Cortesi, R.; Huang, N.; Simelière, F.; Marchetti, N.; Drechsler, M.; Ruzgas, T.; Esposito, E. Design and characterization of ethosomes for transdermal delivery of caffeic acid. Pharmaceutics 2020, 12, 740. [Google Scholar] [CrossRef]
- Ascenso, A.; Raposo, S.; Batista, C.; Cardoso, P.; Mendes, T.; Praça, F.G.; Bentley, M.V.; Simões, S. Development, characterization, and skin delivery studies of related ultradeformable vesicles: Transfersomes, ethosomes, and transethosomes. Int. J. Nanomed. 2015, 10, 5837–5851. [Google Scholar] [CrossRef]
- Razavi, S.; Partoazar, A.; Takzaree, N.; Fasihi-Ramandi, M.; Bahador, A.; Darvishi, M.H. Silver sulfadiazine nanoethogel for burn healing: Characterization and investigation of its in vivo effects. Nanomedicine 2018, 13, 1319–1331. [Google Scholar] [CrossRef]
- Kumar, L.; Verma, S.; Singh, M.; Chalotra, T.; Utreja, P. Advanced drug delivery systems for transdermal delivery of nonsteroidal anti-inflammatory drugs: A review. Curr. Drug Deliv. 2018, 15, 1087–1099. [Google Scholar] [CrossRef]
- Paliwal, S.; Tilak, A.; Sharma, J.; Dave, V.; Sharma, S.; Yadav, R.; Patel, S.; Verma, K.; Tak, K. Flurbiprofen loaded ethosomes—Transdermal delivery of anti-inflammatory effect in rat model. Lipids Heal. Dis. 2019, 18, 1–15. [Google Scholar] [CrossRef]
- Anjum, F.; Zakir, F.; Verma, D.; Aqil, M.; Singh, M.; Jain, P.; Mirza, M.A.; Anwer, M.K.; Iqbal, Z. Exploration of nanoethosomal transgel of naproxen sodium for the treatment of arthritis. Curr. Drug Deliv. 2020, 17, 885–897. [Google Scholar] [CrossRef]
- Ghanbarzadeh, S.; Arami, S. Enhanced transdermal delivery of diclofenac sodium via conventional liposomes, ethosomes, and transfersomes. BioMed Res. Int. 2013, 2013, 616810. [Google Scholar] [CrossRef]
- Chourasia, M.K.; Kang, L.; Chan, S.Y. Nanosized ethosomes bearing ketoprofen for improved transdermal delivery. Results Pharma Sci. 2011, 1, 60–67. [Google Scholar] [CrossRef]
- Barupal, A.K.; Gupta, V.; Ramteke, S. Preparation and characterization of ethosomes for topical delivery of aceclofenac. Indian J. Pharm. Sci. 2010, 72, 582–586. [Google Scholar] [CrossRef]
- Qadir, A.; Ahmad, U.; Ali, A.; Shahid, A.; Aqil, M.; Khan, N.; Ali, A.; Almalki, W.H.; Alghamdi, S.; Abul Barkat, M.; et al. Lipid Engineered Nanoparticle Therapy for Burn Wound Treatment; Bentham Science Publishers: Sharjah, United Arab Emirates, 2021. [Google Scholar] [CrossRef]
- Fu, X.; Shi, Y.; Wang, H.; Zhao, X.; Sun, Q.; Huang, Y.; Qi, T.; Lin, G. Ethosomal gel for improving transdermal delivery of Thymosin β-4. Int. J. Nanomed. 2019, 14, 9275–9284. [Google Scholar] [CrossRef]
- Sarwa, K.K.; Suresh, P.K.; Rudrapal, M.; Verma, V.K. Penetration of tamoxifen citrate loaded ethosomes and liposomes across human skin: A comparative study with confocal laser scanning microscopy. Curr. Drug Deliv. 2014, 11, 332–337. [Google Scholar] [CrossRef]
- Kehinde, E.O.; Akrutiben, S.; Patel, J. Revolutionary approach towards transdermal drug delivery: Ethosomal gels. J. Pharm. Res. Int. 2021, 33, 35–43. [Google Scholar] [CrossRef]
- Rai, S.; Pandey, V.; Rai, G. Transfersomes as versatile and flexible nano-vesicular carriers in skin cancer therapy: The state of the art. Nano Rev. Exp. 2017, 8, 1325708. [Google Scholar] [CrossRef]
- Opatha, S.A.T.; Titapiwatanakun, V.; Chutoprapat, R. Transfersomes: A promising nanoencapsulation technique for transdermal drug delivery. Pharmaceutics 2020, 12, 855. [Google Scholar] [CrossRef]
- Shamshiri, M.K.; Momtazi-Borojeni, A.A.; Shahraky, M.K.; Rahimi, F. Lecithin soybean phospholipid nano-transfersomes as potential carriers for transdermal delivery of the human growth hormone. J. Cell. Biochem. 2019, 120, 9023–9033. [Google Scholar] [CrossRef]
- Al Shuwaili, A.H.; Rasool, B.K.; Abdulrasool, A.A. Optimization of elastic transfersomes formulations for transdermal delivery of pentoxifylline. Eur. J. Pharm. Biopharm. 2016, 102, 101–114. [Google Scholar] [CrossRef]
- Omar, M.M.; Hasan, O.A.; Sisi, A.M.E.l. Preparation and optimization of lidocaine transferosomal gel containing permeation enhancers: A promising approach for enhancement of skin permeation. Int. J. Nanomed. 2019, 14, 1551. [Google Scholar] [CrossRef]
- Chaudhari, Y.; Dharashivkar, S.S.; Palkar, P.; Chaudhari, M.A.; Ruhatiya, G.; Patil, M.; Gaikwad, M. Formulation and evaluation of transfersomal cream of acriflavine. Int. Res. J. Pharm. 2016, 7, 75–78. [Google Scholar] [CrossRef]
- Caddeo, C.; Manca, M.L.; Peris, J.E.; Usach, I.; Sales, O.D.; Matos, M.; Busquets, X.F.; Fadda, A.M.; Manconi, M. Tocopherol-loaded transfersomes: In vitro antioxidant activity and efficacy in skin regeneration. Int. J. Pharm. 2018, 551, 34–41. [Google Scholar] [CrossRef]
- Di Lonardo, A.; De Rosa, M.; Graziano, A.; Pascone, C.; Lucattelli, E. Effectiveness of topical α-Tocopherol Acetate in burn infection treatment. Ann. Burn Fire Disasters 2019, 32, 282. [Google Scholar]
- Leonyza, A.; Surini, S. Optimization of sodium deoxycholate-based transfersomes for percutaneous delivery of peptides and proteins. Int. J. Appl. Pharm. 2019, 11, 329–332. [Google Scholar] [CrossRef]
- Allaw, M.; Pleguezuelos-Villa, M.; Manca, M.L.; Caddeo, C.; Aroffu, M.; Nacher, A.; Diez-Sales, O.; Saurí, A.R.; Ferrer, E.E.; Fadda, A.M.; et al. Innovative strategies to treat skin wounds with mangiferin: Fabrication of transfersomes modified with glycols and mucin. Nanomedicine 2020, 15, 1671–1685. [Google Scholar] [CrossRef]
- Lu, M.; Qiu, Q.; Luo, X.; Liu, X.; Sun, J.; Wang, C.; Lin, X.; Deng, Y.; Song, Y. Phyto-phospholipid complexes (phytosomes): A novel strategy to improve the bioavailability of active constituents. Asian J. Pharm. Sci. 2019, 14, 265–274. [Google Scholar] [CrossRef]
- Alharbi, W.S.; Almughem, F.A.; Almehmady, A.M.; Jarallah, S.J.; Alsharif, W.K.; Alzahrani, N.M.; Alshehri, A.A. Phytosomes as an emerging nanotechnology platform for the topical delivery of bioactive phytochemicals. Pharmaceutics 2021, 13, 1475. [Google Scholar] [CrossRef]
- Kumar, A.B.; Habbu, P.; Thimmasetty, L.; Hullatti, P.; Kumar, S.R. Phytosomes as novel drug delivery system for herbal medicine—A review. Syst. Rev. Pharm. 2016, 8, 5–7. [Google Scholar] [CrossRef]
- Barani, M.; Sangiovanni, E.; Angarano, M.; Rajizadeh, M.A.; Mehrabani, M.; Piazza, S.; Gangadharappa, H.V.; Pardakhty, A.; Mehrbani, M.; Dell’Agli, M.; et al. Phytosomes as innovative delivery systems for phytochemicals: A comprehensive review of literature. Int. J. Nanomed. 2021, 16, 6983–7022. [Google Scholar] [CrossRef] [PubMed]
- Rafiee, M.H.; Abdul Rasool, B.K. An overview of microparticulate drug delivery system and its extensive therapeutic applications in diabetes. Adv. Pharm. Bull. 2022. [Google Scholar] [CrossRef]
- Sravanthi, M.; Krishna, J.S. Phytosomes: A novel drug delivery for herbal extracts. Int. J. Pharm. Sci. Res. 2017, 4, 949–959. [Google Scholar] [CrossRef]
- Singh, D.; Upadhyay, P.; Upadhyay, S. Phytosomes: An advanced drug delivery system for herbal drug. Glob. J. Pharmaceu. Sci. 2018, 6, 1. [Google Scholar] [CrossRef]
- Safta, D.A.; Bogdan, C.; Moldovan, M.L. Vesicular nanocarriers for phytocompounds in wound care: Preparation and characterization. Pharmaceutics 2022, 14, 991. [Google Scholar] [CrossRef]
- Shahzad, M.N.; Ahmed, N. Effectiveness of Aloe Vera gel compared with 1% silver sulphadiazine cream as burns wound dressing in second-degree burns. J. Pak. Med. Assoc. 2013, 63, 225–230. [Google Scholar]
- Jain, P.; Taleuzzaman, M.; Kala, C.; Kumar Gupta, D.; Ali, A.; Aslam, M. Quality by design (Qbd) assisted development of phytosomal gel of aloe vera extract for topical delivery. J. Liposome Res. 2021, 31, 381–388. [Google Scholar] [CrossRef]
- Darvishi, B.; Dinarvand, R.; Mohammadpour, H.; Kamarul, T.; Sharifi, A.M. Dual l-Carnosine/Aloe vera Nanophytosomes with Synergistically Enhanced Protective Effects against Methylglyoxal-Induced Angiogenesis Impairment. Mol. Pharm. 2021, 18, 3302–3325. [Google Scholar] [CrossRef]
- Mazumder, A.; Dwivedi, A.; Fox, L.T.; Brümmer, A.; du Preez, J.L.; Gerber, M.; du Plessis, J. In vitro skin permeation of sinigrin from its phytosome complex. J. Pharm. Pharmacol. 2016, 68, 1577–1583. [Google Scholar] [CrossRef]
- Agrawal, S.; Yallatikar, T.; Gurjar, P. Reversed-phase high-performance liquid chromatographic method development and validation for allyl isothiocyanate estimation in phytosomes of Brassica nigra extract. J. Adv. Pharm. Technol. Res. 2019, 10, 126. [Google Scholar] [CrossRef]
- Allawi, H.M.; Al-bayati, M. Formulation of Camellia sinensis phytosome encapsulated diclofenac and effect on analgesia and inflammation in mice. Cancer Nanotechnol. 2020, 24, 175–190. [Google Scholar]
- Demir, B.; Barlas, F.B.; Guler, E.; Gumus, P.Z.; Can, M.; Yavuz, M.; Coskunol, H.; Timur, S. Gold nanoparticle loaded phytosomal systems: Synthesis, characterization and in vitro investigations. RSC Adv. 2014, 4, 34687–34695. [Google Scholar] [CrossRef]
- Lim, A.W.; Ng, P.Y.; Chieng, N.; Ng, S.F. Moringa oleifera leaf extract–loaded phytophospholipid complex for potential application as wound dressing. J. Drug. Deliv. Sci. Technol. 2019, 54, 101329. [Google Scholar] [CrossRef]
- Varadkar, M.; Gadgoli, C. Preparation and evaluation of wound healing activity of phytosomes of crocetin from Nyctanthes arbor-tristis in rats. J. Tradit. Complement. Med. 2021, 12, 354–360. [Google Scholar] [CrossRef]
- Pananchery, J.; Gadgoli, C. Phytosomes of naphthoquinone enriched extract of root bark of Onosma echioides exhibit wound healing activity in rats. Indones. J. Pharm. 2021, 32, 474–483. [Google Scholar] [CrossRef]
- Rajashekar, K.; Sundari, P.J.; Srinivas, P.; Venkateshwara, S. Development of a topical phytosomal gel of Woodford fruticosa. WJPPS 2015, 4, 919–932. [Google Scholar]
- Sarheed, O.; Rasool, B.K.; Abu-Gharbieh, E.; Aziz, U.S. An investigation and characterization of alginate hydrogel dressing loaded with metronidazole prepared by combined inotropic gelation and freeze-thawing cycles for controlled release. AAPS PharmSciTech 2015, 16, 601–609. [Google Scholar] [CrossRef]
- Sofowora, A.; Ogunbodede, E.; Onayade, A. The role and place of medicinal plants in the strategies for disease prevention. Afr. J. Tradit. Complement. Altern. Med. 2013, 10, 210–229. [Google Scholar] [CrossRef]
- Liang, J.; Cui, L.; Li, J.; Guan, S.; Zhang, K.; Li, J. Aloe vera: A Medicinal Plant Used in Skin Wound Healing. Tissue Eng. Part B Rev. 2021, 27, 455–474. [Google Scholar] [CrossRef]
- Miastkowska, M.; Kulawik-Pióro, A.; Szczurek, M. Nanoemulsion Gel Formulation Optimization for Burn Wounds: Analysis of Rheological and Sensory Properties. Processes 2020, 8, 1416. [Google Scholar] [CrossRef]
- Hekmatpou, D.; Mehrabi, F.; Rahzani, K.; Aminiyan, A. The Effect of Aloe Vera Clinical Trials on Prevention and Healing of Skin Wound: A Systematic Review. Iran. J. Med. Sci. 2019, 44, 1. [Google Scholar] [PubMed]
- Rösch, D.; Krumbein, A.; Mügge, C.; Kroh, L.W. Structural investigations of flavonol glycosides from sea buckthorn (Hippophaë rhamnoides) pomace by NMR spectroscopy and HPLC-ESI-MS n. J. Agric. Food Chem. 2004, 52, 4039–4046. [Google Scholar] [CrossRef] [PubMed]
- Sadowska, B.; Budzyńska, A.; Stochmal, A.; Żuchowski, J.; Różalska, B. Novel properties of Hippophae rhamnoides L. twig and leaf extracts—Anti-virulence action and synergy with antifungals studied in vitro on Candida spp. model. Microb. Pathog. 2017, 107, 372–379. [Google Scholar] [CrossRef] [PubMed]
- Xiang, L.; Wang, J.; Zhang, G.; Rong, L.; Wu, H.; Sun, S.; Guo, Y.; Yang, Y.; Lu, L.; Qu, L. Analysis and identification of two similar traditional Chinese medicines by using a three-stage infrared spectroscopy: Ligusticum chuanxiong, Angelica sinensis and their different extracts. J. Mol. Struct. 2016, 1124, 164–172. [Google Scholar] [CrossRef]
- Zhong, L.J.; Hua, Y.L.; Ji, P.; Yao, W.L.; Zhang, W.Q.; Li, J.; Wei, Y.M. Evaluation of the anti-inflammatory effects of volatile oils from processed products of Angelica sinensis radix by GC–MS-based metabolomics. J. Ethnopharmacol. 2016, 191, 195–205. [Google Scholar] [CrossRef]
- Kulkarni, R.N.; Baskaran, K.; Jhang, T. Breeding medicinal plant, periwinkle [Catharanthus roseus (L) G. Don]: A review. Plant. Genet. Resour. Characterisation Util. 2016, 14, 283–302. [Google Scholar] [CrossRef]
- Das, S.; Krishi Viswavidyalaya, C.; Sharangi, A.B. Madagascar periwinkle (Catharanthus roseus L.): Diverse medicinal and therapeutic benefits to humankind. J. Pharm. Phytochem. 2017, 6, 1695–1701. [Google Scholar]
- Givol, O.; Kornhaber, R.; Visentin, D.; Cleary, M.; Haik, J.; Harats, M. A systematic review of Calendula officinalis extract for wound healing. Wound Repair Regen. 2019, 27, 548–561. [Google Scholar] [CrossRef]
- Nicolaus, C.; Junghanns, S.; Hartmann, A.; Murillo, R.; Ganzera, M.; Merfort, I. In vitro studies to evaluate the wound healing properties of Calendula officinalis extracts. J. Ethnopharmacol. 2017, 196, 94–103. [Google Scholar] [CrossRef]
- Thakur, V.; Vishwavidyalya, R.D.; Paroha, S. Climatic impact on physical properties, oil and protein content of two sesame (Sesamum indicum L.) varieties. Int. J. Chem. Stud. 2018, 6, 2098–2104. [Google Scholar]
- Afroz, M.; Zihad, S.M.N.K.; Uddin, S.J.; Rouf, R.; Rahman, M.S.; Islam, M.T.; Khan, I.N.; Ali, E.S.; Aziz, S.; Shilpi, J.A.; et al. A systematic review on antioxidant and antiinflammatory activity of sesame (Sesamum indicum L.) oil and further confirmation of antiinflammatory activity by chemical profiling and molecular docking. Phyther. Res. 2019, 33, 2585–2608. [Google Scholar] [CrossRef]
- Almeida, É.S.; de Oliveira, D.; Hotza, D. Properties and Applications of Morinda citrifolia (Noni): A Review. Compr. Rev. Food Sci. Food Saf. 2019, 18, 883–909. [Google Scholar] [CrossRef]
- Jin, M.; Wang, Y.; Yang, X.; Yin, H.; Nie, S.; Wu, X. Structure characterization of a polysaccharide extracted from noni (Morinda citrifolia L.) and its protective effect against DSS-induced bowel disease in mice. Food Hydrocoll. 2019, 90, 189–197. [Google Scholar] [CrossRef]
- Ramadon, D.; Anwar, E.; Diazputri Utami, T. Transfersomal gel containing green tea (Camellia sinensis L. kuntze) leaves extract: Increasing in vitro penetration. Artic Asian J. Pharm. Clin. Res. 2017, 10, 294–298. [Google Scholar] [CrossRef]
- Farooqui, A.; Khan, A.; Borghetto, I.; Kazmi, S.U.; Rubino, S.; Paglietti, B. Synergistic antimicrobial activity of Camellia sinensis and Juglans regia against multidrug-resistant bacteria. PLoS ONE 2015, 10, e0118431. [Google Scholar] [CrossRef]
- Nutrizio, M.; Gajdoš Kljusurić, J.; Marijanović, Z.; Dubrović, I.; Viskić, M.; Mikolaj, E.; Chemat, F.; Režek Jambrak, A. The potential of high voltage discharges for green solvent extraction of bioactive compounds and aromas from rosemary (Rosmarinus officinalis L.)—Computational simulation and experimental methods. Molecules 2020, 25, 3711. [Google Scholar] [CrossRef]
- Andrade, J.M.; Faustino, C.; Garcia, C.; Ladeiras, D.; Reis, C.P.; Rijo, P. Rosmarinus officinalis L.: An update review of its phytochemistry and biological activity. Future Sci. OA 2018, 4, FSO283. [Google Scholar] [CrossRef]
- Borges, R.S.; Ortiz, B.L.S.; Pereira, A.C.M.; Keita, H.; Carvalho, J.C.T. Rosmarinus officinalis essential oil: A review of its phytochemistry, anti-inflammatory activity, and mechanisms of action involved. J. Ethnopharmacol. 2019, 229, 29–45. [Google Scholar] [CrossRef]
- Hızır-Kadı, İ.; Gültekin-Özgüven, M.; Altin, G.; Demircan, E.; Özçelik, B. Liposomal nanodelivery systems generated from proliposomes for pollen extract with improved solubility and in vitro bioaccessibility. Heliyon 2020, 6, e05030. [Google Scholar] [CrossRef]
- Raemdonck, K.; Braeckmans, K.; Demeester, J.; De Smedt, S.C. Merging the best of both worlds: Hybrid lipid-enveloped matrix nanocomposites in drug delivery. Chem. Soc. Rev. 2014, 43, 444–472. [Google Scholar] [CrossRef]
- Aytekin, A.A.; Tuncay Tanrıverdi, S.; Aydın Köse, F.; Kart, D.; Eroğlu, İ.; Özer, Ö. Propolis-loaded liposomes: Evaluation of antimicrobial and antioxidant activities. J. Liposome Res. 2020, 30, 107–116. [Google Scholar] [CrossRef]
- El-Kased, R.F.; Amer, R.I.; Attia, D.; Elmazar, M.M. Honey-based hydrogel: In vitro and comparative In vivo evaluation for burn wound healing. Sci. Rep. 2017, 7, 9692. [Google Scholar] [CrossRef]
- Perminaite, K.; Maria, F.A.; Sinico, C.; Ramanauskiene, K. Formulation of liposomes containing royal jelly and their quality assessment. J. Nanosci. Nanotechnol. 2021, 21, 2841–2846. [Google Scholar] [CrossRef]
- Sogut, O.; Aydemir, S.U.; Sezer, S. Liposomal delivery systems for herbal extracts. J. Drug Deliv. Sci. Technol. 2021, 61, 102147. [Google Scholar] [CrossRef]
- Ghatnur, S.M.; Sonale, R.S.; Balaraman, M.; Kadimi, U.S. Engineering liposomes of leaf extract of seabuckthorn (SBT) by supercritical carbon dioxide (SCCO2)-mediated process. J. Liposome Res. 2012, 22, 215–223. [Google Scholar] [CrossRef]
- Arora, D.; Rani, A.; Sharma, A. A review on phytochemistry and ethnopharmacological aspects of genus Calendula. Pharmacogn. Rev. 2013, 7, 179–187. [Google Scholar] [CrossRef]
- Dosio, F.; Milla, P. EC-145, a folate-targeted Vinca alkaloid conjugate for the potential treatment of folate receptor-expressing cancers drug profile. Curr. Opin. Investig. Drugs 2010, 11, 1424–1433. [Google Scholar]
- Cui, M.; Di Pan, Z.H.; Pan, L.Q. Danggui Buxue extract-loaded liposomes in thermosensitive gel enhance in vivo dermal wound healing via activation of the VEGF/PI3K/Akt and TGF-β/Smads signaling pathway. Evid.-Based Complement. Altern. Med. 2017, 2017, 8407249. [Google Scholar] [CrossRef]
- Abdelfattah, S.; Nasr, M.; Ali, A.; Genedy, A.S. Vesicular systems used for wound healing. Arch. Pharm. Sci. Ain. Shams. Univ. 2021, 5, 184–203. [Google Scholar] [CrossRef]
- Rohilla, S.; Bhatt, D.C.; Gupta, A. Therapeutic potential of phytomedicines and novel polymeric strategies for significant management of candidiasis. Curr. Pharm. Des. 2018, 24, 1748–1765. [Google Scholar] [CrossRef]
- García-Díaz, M.; Patiño, B.; Vázquez, C.; Gil-Serna, J. A novel niosome-encapsulated essential oil formulation to prevent aspergillus flavus growth and aflatoxin contamination of maize grains during storage. Toxins 2019, 11, 646. [Google Scholar] [CrossRef]
- Liu, F.; Wang, Y.; Ding, Z.; Zhao, L.; Xiao, J.; Wang, L.; Ding, S. Transcriptomic analysis of flower development in tea (Camellia sinensis L.). Gene 2017, 631, 39–51. [Google Scholar] [CrossRef]
- Filipczak, N.; Pan, J.; Yalamarty, S.S.K.; Torchilin, V.P. Recent advancements in liposome technology. Adv. Drug Deliv. Rev. 2020, 156, 4–22. [Google Scholar] [CrossRef]
- Ferrari, G.; Pang, L.Y.; De Moliner, F.; Vendrell, M.; Reardon, R.J.M.; Higgins, A.J.; Chopra, S.; Argyle, D.J. Effective penetration of a liposomal formulation of bleomycin through ex-vivo skin explants from two different species. Cancers 2022, 14, 1083. [Google Scholar] [CrossRef]
- HISTX—Liposomal Vitamin C and Quercetin—LipoLife. Lipolife. Available online: https://www.lipolife.co.za/histx-liposomal-vitamin-c-quercetin/ (accessed on 12 March 2022).
- Mi, Y.; Zhong, L.; Lu, S.; Hu, P.; Pan, Y.; Ma, X.; Yan, B.; Wei, Z.; Yang, G. Quercetin promotes cutaneous wound healing in mice through Wnt/β-catenin signaling pathway. J. Ethnopharmacol. 2022, 290, 115066. [Google Scholar] [CrossRef]
- Khalili, A.; Alipour, S.; Fathalipour, M.; Purkhosrow, A.; Mashghoolozekr, E.; Bayat, G.; Nekooeian, A.A. Liposomal and Non-Liposomal Formulations of Vitamin C: Comparison of the Antihypertensive and Vascular Modifying Activity in Renovascular Hypertensive Rats. Iran. J. Med. Sci. 2020, 45, 41–49. [Google Scholar] [CrossRef]
- What Is Liposomal Vitamin C? Health Benefits, Usage & Dosage. Available online: https://vitaminc.co.uk/what-is-liposomal-vitamin-c-health-benefits-usage-and-dosage/ (accessed on 14 March 2022).
- Concept Crucial Values for The Brand. Decorté. Available online: https://www.decortecosmetics.com/ourstory/my/crucialvalues.html (accessed on 14 March 2022).
- Dematte, M.F.; Gemperli, R.; Salles, A.G.; Dolhnikoff, M.; Lanças, T.; Saldiva, P.H.; Ferreira, M.C. Mechanical evaluation of the resistance and elastance of post-burn scars after topical treatment with tretinoin. Clinics 2011, 66, 1949–1954. [Google Scholar] [CrossRef]
- Lu, K.J.; Wang, W.; Xu, X.L.; Jin, F.Y.; Qi, J.; Wang, X.J.; Kang, X.Q.; Zhu, M.L.; Huang, Q.L.; Yu, C.H.; et al. A dual deformable liposomal ointment functionalized with retinoic acid and epidermal growth factor for enhanced burn wound healing therapy. Biomater. Sci. 2019, 7, 2372–2382. [Google Scholar] [CrossRef]
- Huaqing, L.; Chuqin, Y.; Wei, L.; Taxi, L. Tacrolimus Transfersome Solution and Preparation Method Thereof. CN102949341B, 4 April 2014. Available online: https://patents.google.com/patent/CN102949341B/en?oq=CN102949341B (accessed on 5 February 2022).
- Patel, P.M. High-Concentration Lidocaine Compositions and Methods for Their Preparation. US7476400B2, 13 January 2009. Available online: https://patents.google.com/patent/US7476400B2/en?oq=US7476400B2 (accessed on 5 February 2022).
- Milián, H.J.S.; Rull, L.V.; Díaz, E.M.; Acosta, J.A.B.; Puig, I.C.; Miró, J.V. Vesicles Which Include Epidermal Growth Factor and Compositions that Contain Same. EP2915541B1, 9 November 2016. Available online: https://patents.google.com/patent/EP2915541B1/en?oq=EP2915541B1 (accessed on 5 February 2022).
- Touitou, E. Composition for Applying Active Substances to or through the Skin. US5716638A, 10 February 1998. Available online: https://patents.google.com/patent/US5716638A/en?oq=US5716638 (accessed on 5 February 2022).
- Pang, N.S.; Bohnen, M.A. Liposomal Antibiotic Formulation. CA2117046C, 3 October 2000. Available online: https://patents.google.com/patent/CA2117046C/en?oq=CA2117046C (accessed on 5 February 2022).
- Alexander, L.; Wu, H.G. Enhanced Antivirulent for Antibiotic-Resistant Bacteria. US20200276231A1, 3 September 2020. Available online: https://patents.google.com/patent/US20200276231A1/en?oq=US20200276231A1 (accessed on 5 February 2022).
- Thorel, G.N. Topical Cosmetic Composition Containing an Improved Pro-Penetrating System. WO2012153075A2, 24 January 2013. Available online: https://patents.google.com/patent/WO2012153075A2/en?oq=wo2012153075A2 (accessed on 5 February 2022).
- Ray, J.R.; Hodge, C.D. Composition and Method for Compounded Therapy. US9468599B2, 18 October 2016. Available online: https://patents.google.com/patent/US9468599B2/en?oq=US9468599B2 (accessed on 5 February 2022).
- Edelson, J.; Timothy, K.; Zhang, B. Transdermal Delivery. JP2018009026A, 18 January 2018. Available online: https://patents.google.com/patent/JP2018009026A/en?oq=JP2018009026A (accessed on 6 February 2022).
- Kim, Y.M.; Jang, G.H.; Park, Y.J.; Oh, G.H. Method of Preparing Bioactive Substance-Encapsulated Ethosome, Ethosome Composition, and Cosmetic Composition Including Ethosome Composition. WO2019004563A1, 3 January 2019. Available online: https://patents.google.com/patent/WO2019004563A1/en?oq=WO2019004563A1 (accessed on 6 February 2022).
- Chegregor, C. Liquid Droplets for the Manufacture of a Preparation for the Non-Invasive Application or for the Non-Invasive Transport of Active Ingredients through Barriers. KR100446832B1, 4 September 2004. Available online: https://patents.google.com/patent/KR100446832B1/en?oq=KR100446832B1 (accessed on 6 February 2022).
- Azevedo, M.M.; Pina-Vaz, C.; Rodrigues, A.G. The Role of Phage Therapy in Burn Wound Infections Management: Advantages and Pitfalls. J. Burn Care Res. 2022, 43, 336–342. [Google Scholar] [CrossRef]
- PlumX- Novel Microbe-Resistant Clay Dressing for Healing Burn Wounds. Available online: https://plu.mx/plum/a/?repo_url=https://stars.library.ucf.edu/honorstheses/1193&theme=plum-bigben-theme (accessed on 19 June 2022).
- Khan, A.D.; Rastogi, V.; Lavhale, P.M.; Jain, J. Novel approaches for herbal drug delivery in wound healing: A review. Indian J. Pharm. Sci. 2022, 84, 247–260. [Google Scholar] [CrossRef]
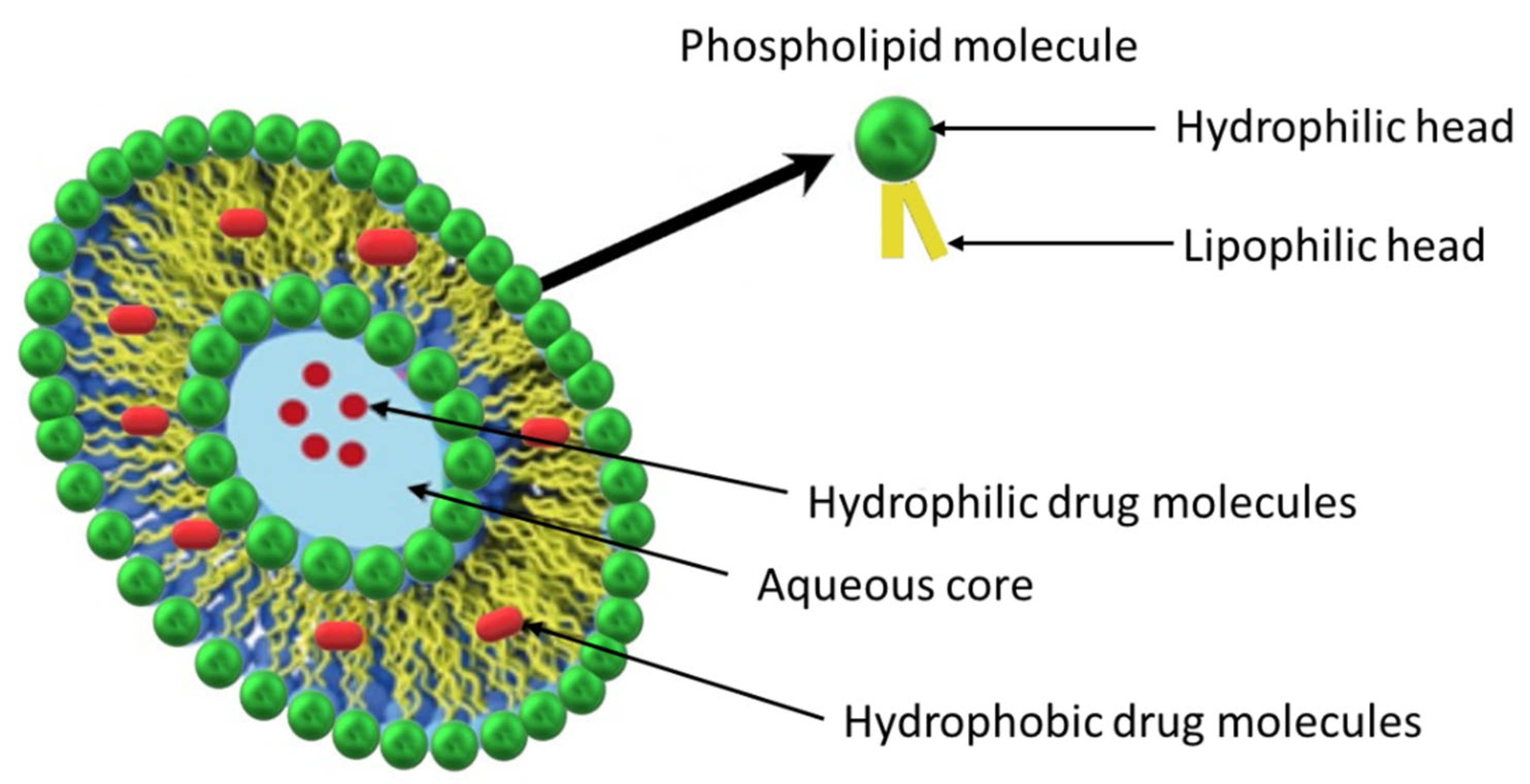
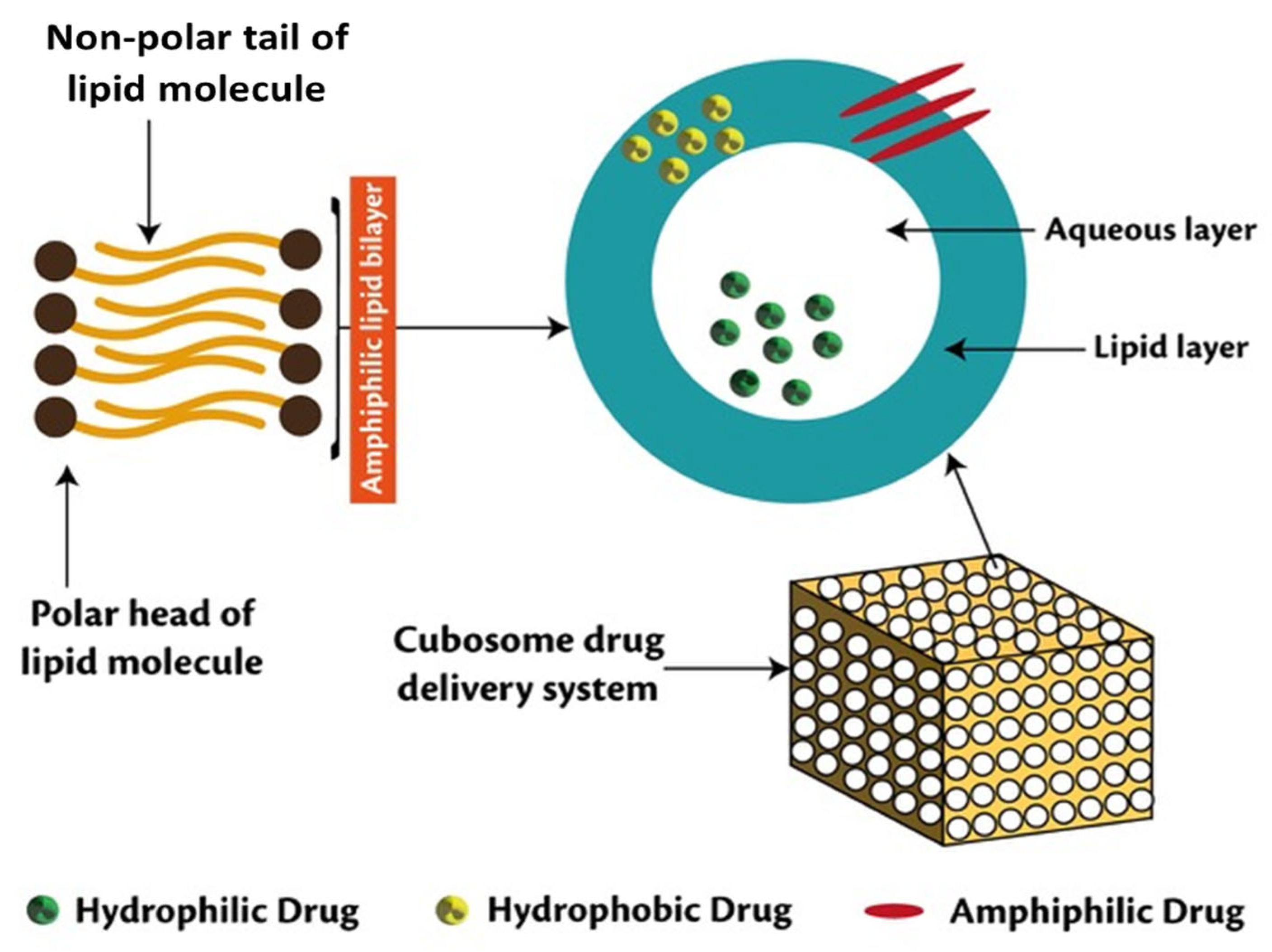
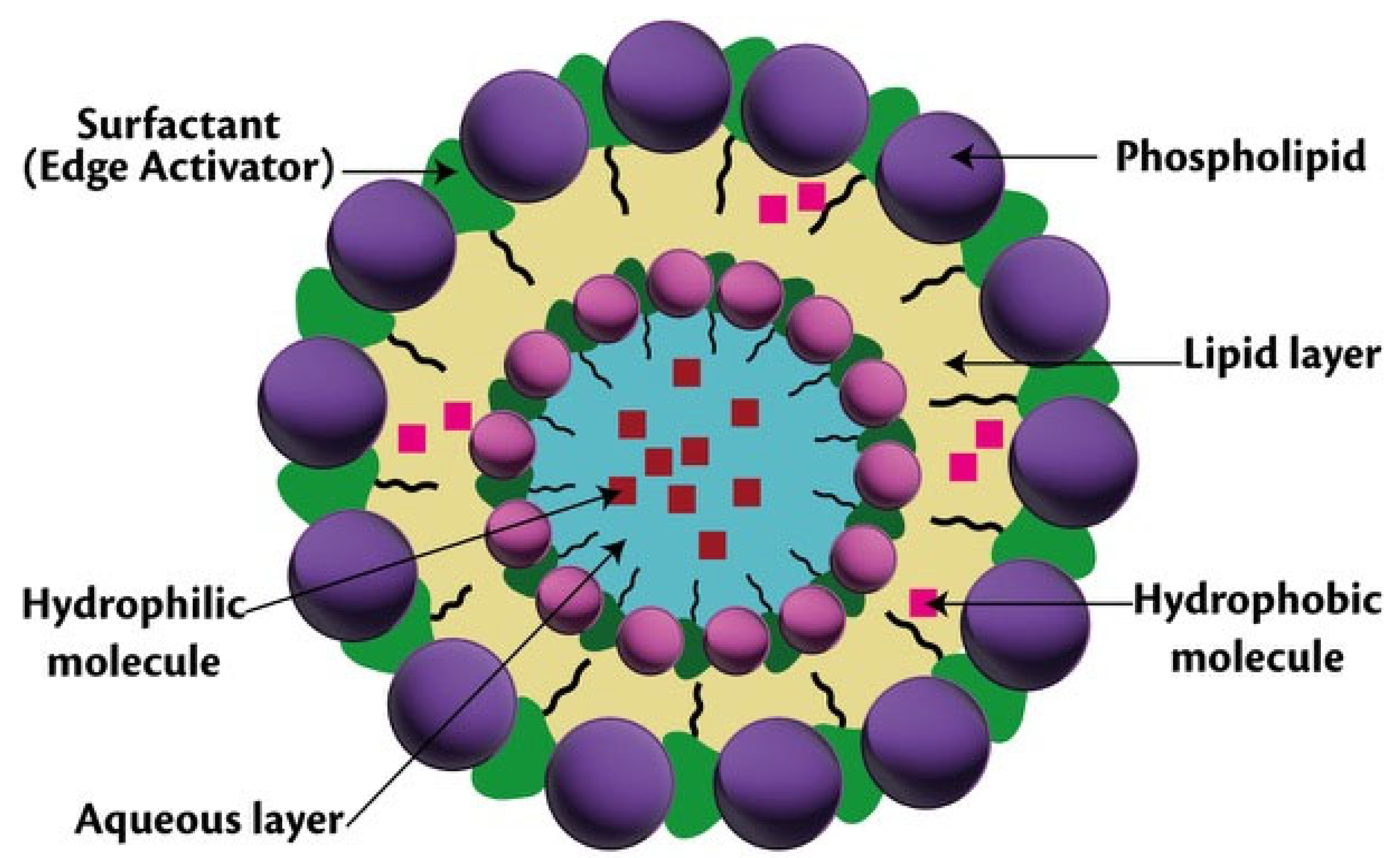
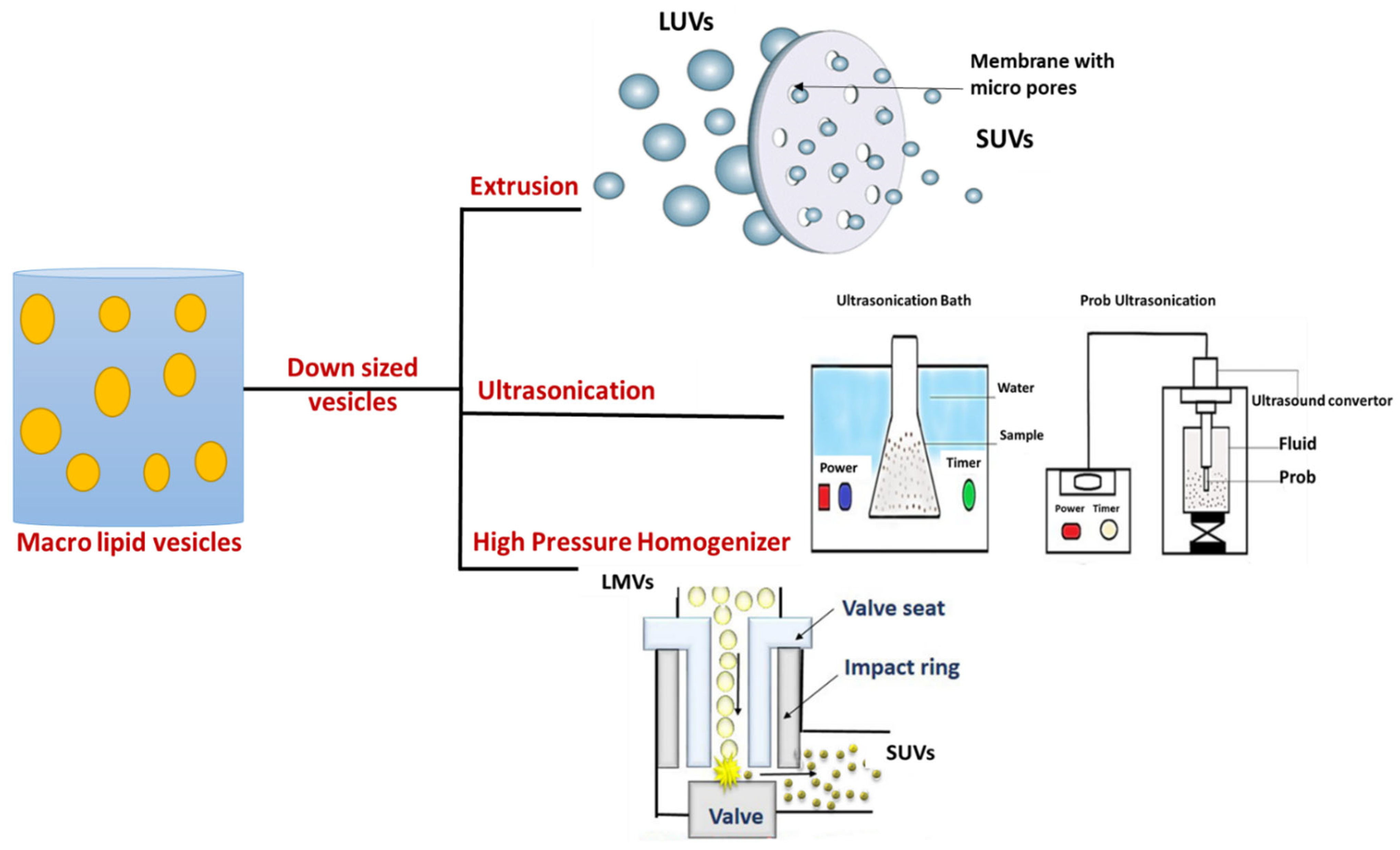
| Burn Class | Features | Symptoms | Management |
|---|---|---|---|
| First degree |
|
|
|
| Second degree |
|
|
|
| Third degree |
|
|
|
| Class | Characteristics | Types | Composition | Example |
|---|---|---|---|---|
| Class I | Materials for impermeable dressings and short-term dressings. | (a) Single-layer substances |
|
|
|
| |||
| (b) Double-layer substances |
|
| ||
| Durable single-layer skin replacements. | Epidermal replacements |
|
| |
| Class II | Skin substitutes |
|
| |
| Class III | Alternatives to composite skin. | Grafted skin |
|
|
| Skin tissue engineering |
|
|
| Antimicrobial Agents | Dosage Form | Clinical Indications | Side Effects | Contraindications |
|---|---|---|---|---|
| Silver sulfadiazine (1% cream) with or without cerium | Cream (1%) | All types of burns (small, medium, and large wound surface areas). | Skin allergies, neutropenia, and leukopenia. | Pregnancy, lactating, infants, and allergies to sulfonamides. |
| Nanocrystalline silver (Acticoat®, Aquacel Ag®) | Dressings | All sizes of burns (small, medium, and large wound surface areas). | Systemic silver absorption, and skin staining. | Burn near eyes, Pregnancy, and allergic to silver. |
| Bacitracin | Ointment 500 IU/g | All sizes of burn wounds, including face, perineum, graft sites. An alternative if allergic to sulfonamides. | Yeast growth, skin allergies. | Bacterial resistance, allergic reaction, and signs of reepithelialization. |
| Mupirocin | Ointment Cream (2%) | Face, perineum, and small and medium surface area burns. An alternative if allergic to sulfonamides. | Yeast growth, skin hypersensitivity. | Bacterial resistance, allergic reaction. |
| Mafenide | Cream (8.5%) Solution (5%) | Small wound surface area, ears, and nose burn. | Pain, epithelial regeneration inhibition, and metabolic acidosis. | Large burn area of burn (>40% of the total BSA); allergic to sulfonamides. |
| Chlorhexidine | Skin cleanser | Only superficial burns. | Skin hypersensitivity. | Deep burns. |
| Povidone-iodine | Liposomal ointment | Small, and medium surface area burns | Pain, skin hypersensitivity, iodine toxicity, renal failure. | Infants, pregnancy, lactation, and thyroid diseases. |
| Acetic acid | Diluted solution | Antiseptic, Pseudomonas aeruginosa infections | Inhibits epithelialization at high concentrations. | Superficial burns. |
| Bismuth-impregnated petroleum gauze | Dressings | Preferred dressing for skin graft donor sites and use in children. | No adverse effects are reported. | Allergic reactions. |
| Sodium hypochlorite Dakin’s solution® | Solution (0.025%) | Difficult burn wounds and other antibiotic-resistant bacteria. | Pain. | Allergic reactions. |
| Advantages | Limitations |
|---|---|
|
|
| Drug | Therapeutic Activity | Prepared Formulation | Model Used | Outcomes | Reference |
|---|---|---|---|---|---|
| Silver Sulfadiazine | Prevents and treats wound infection in patients with second- and third-degree burns. | Topical wound dressing containing liposome-entrapped silver sulfadiazine. | Sacrificed rats: colony-forming units of P. aeruginosa were injected into the muscle tissue of the rats. | One application of liposomal-entrapped antibiotics resulted in a therapeutic effect that would normally require multiple administrations of conventional therapy. | [39] |
| Usnic acid | Antimicrobial, anti-inflammatory antiviral, and antitumor activities. | Gelatin-based dressings containing usnic acid-loaded liposomes. | Male pigs | According to the data, topical wound dressing for second-degree burn treatment promotes faster-wound healing than silver sulfadiazine ointment. | [40] |
| Amphotericin B | Broad-spectrum antifungal agent of choice for burn patients. | Experimental liposomal amphotericin B in commercial products. | In vitro | Liposomal amphotericin B exhibited superior in vitro antimicrobial activity against Aspergillus fumigatus compared to the free drug as well as superior physical stability. | [41] |
| Epidermal growth factor (EGF) | EGF enhances wound healing by stimulating epidermal and dermal regeneration and promoting cell growth. | EGF-loaded liposomes in chitosan gel formulations. | Rats | An experiment was conducted on rats to investigate the effect of formulations on healing second-degree burn wounds. The histochemical results showed that the EGF–liposomal gel formulations had the highest epithelialization rate. | [42] |
| Bupivacaine | Postoperative analgesia | Bupivacaine-loaded liposomes. | A clinical study in burn patients. | The administration on the surgical site (autograft harvesting) produced postsurgical analgesia in burn patients. | [43] |
| Drug | Therapeutic Activity | Preparation Technique | Formulation Concepts | Study Outcomes | Study Design | References |
|---|---|---|---|---|---|---|
| Sulfadiazine sodium | Antibiotic | Film hydration method | The vesicular size increased with the amount of alcohol. Large-sized vesicles were obtained with ethanol, followed by propylene glycol and glycerol. | The drug permeation increased with an alcohol concentration. | Ex vivo permeation through rabbit ear skin. | [71] |
| C. officinalis | Antioxidant, antimicrobial, and promotes wound healing. | Thin-film hydration method | Anthocyanins-loaded niosomes were incorporated into a mucoadhesive gel formulation. | The bioactivity of C. officinalis methanolic extract significantly increased after loading into the phytoniosome formulation. | In vitro cell lines | [72] |
| Silver sulfadiazine | Topical antibacterial | Solvent injection method | The niosomes with Span 60 gave the highest entrapment efficiency and exhibited considerable retardation in vitro drug release. | A sustained release once-a-day niosomal formulation to improve patient compliance was developed. | In vitro antimicrobial study | [73] |
| hBD-1 and HNP-1 | Antimicrobial peptides (AMPs) | Mechanical shaking/ homogenization | The AMP niosomal topical gels were prepared, and the antimicrobial activity was investigated against Staphylococcus aureus clinical strains in vitro and in vivo. | The niosomal gel containing recombinant hBD-1 (1 μg/mL) increased the healing rate of infected wounds in rats. | Animal studies (rats) | [74] |
| Tannic acid | Antibacterial and antibiofilm activities | Optimized formulation using response surface methodology | Niosomes were found as an ideal candidate for designing a new delivery method for the antibacterial agent. | In vitro antimicrobial study | [75] | |
| Phenytoin sodium | Wound-healing activity | Solvent evaporation–film hydration method | The phenytoin-loaded niosomes were incorporated in sodium alginate gel. | In contrast to the vehicle-treated control group, the niosome-treated lesions were fully healed by the ninth day of the in vivo study. | Guinea pigs with induced wounds | [76] |
| Hypericum perforatum | Wound-healing and anti-inflammatory activities | Film hydration technique | Optimized formulation using response surface methodology. | By the seventh day of the in vivo study, the niosomal gel treatment group showed increased angiogenesis and fibroblastic proliferation. | Adult mongrel dogs | [77] |
| Drug | Vesicles Formers | Therapeutic Activity | Dosage Form | References |
|---|---|---|---|---|
| Human Growth Hormone | Lecithin soybean phospholipid | Reduction in wound healing time. | Transdermal suspension | [93] |
| Tocopherol Acetate | Soy phosphatidylcholine (Lipoid S75) | Antioxidant activity and wound healing properties. A simple and low-cost technique for speeding up the healing of infected tough wounds. | Topical suspension | [97,98] |
| Recombinant Human Epidermal Growth Factor | Phosphatidylcholine and sodium deoxycholate | Accelerates chronic wound healing. | Transdermal suspension | [99] |
| Mangiferin | Soy lecithin and mucine | Antioxidant and anti-inflammatory. | Bioadhesive topical preparation | [100] |
| Scientific Name | Dosage Form | Model Used | Magnitude of Results | Reference |
|---|---|---|---|---|
| Aloe vera | Mucilage (98%) | Clinical studies (patients with second-degree burns) | Aloe therapy proved effective for partial burn injuries (total burn surface area <25%). Burn wound healing and pain relief in the tested patient (who received aloe treatment) were faster than in the control group patients (who received silver sulfadiazine cream 1%). Pain relief was effective by the 5th–12th day of treatment, and 24 out of 25 total patients healed completely by the 40th day of treatment. | [109] |
| Phytosomal gel | Ex vivo permeation study using rat skin | The formulation was optimized by a factorial design experiment. The skin permeation and flux profile of the phytosomal gel were superior compared to aloe vera extract mucilage. The phytosomal gel showed stability for three months. | [110] | |
| l-Carnosine/Aloe vera-loaded dual nanophytosomes | In vitro model using human umbilical vein endothelial cells (HUVECs) | The nanophytosomal preparation was effective in protecting the HUVECs against methylglyoxal (MGO)-induced toxicity for 72 h of incubation. Moreover, the formula showed efficient free radical scavenging potency. Thus, it could be used in the treatment of microvascular dysfunction in diabetics with uncontrolled hyperglycemia. | [111] | |
| Sinigrin glucosinolate (seeds of Brassica nigra) | Sinigrin–phytosome complexes | Ex vivo permeation study using the abdominal skin of Caucasian female patients after plastic surgery | The phytosomes delivered a statistically significantly higher concentration of sinigrin into the stratum corneum–epidermis compared to the control (free sinigrin). | [112] |
| Brassica oleracea | Leaf aqueous extract-loaded phytosomes | In vitro assessment | HPLC was accurate, fast, and cost-effective for the estimation of allyl isothiocyanate in black mustard extract-loaded phytosomes. | [113] |
| Camellia sinensis | Leaf ethanolic extract-loaded phytosomes | Male Balb-c mice | Analgesia and the anti-inflammatory activity of green tea phytosomes that contained diclofenac were enhanced and prolonged in a dose-dependent manner when compared to diclofenac alone. | [114] |
| Calendula officinalis | Gold nanoparticle-loaded phytosomes | In vitro cell culture studies | Fluorescence imaging showed that gold nanoparticle-loaded phytosomes penetrated deep into the cells and accumulated around the nucleus. They also exhibited antioxidant and wound healing properties. | [115] |
| Moringa oleifera | Leaf aqueous extract-loaded phytosomes | In vitro normal human dermal fibroblast cell migration and cytotoxicity tests | Comparatively to the controls, the phytosomes exhibited significantly higher cell migration and proliferation rates. Moreover, the phytosomes had no cytotoxic effects at concentrations up to 1.5 mg/mL, exhibiting significant potential as a therapeutic wound dressing. | [116] |
| Crocetin from Nyctanthes arbor-tristis | Crocetin-loaded phytosomal gel | Incision and excision wounds inflicted on Wister albino rats | With the phytosome-loaded gel, both wound models showed good wound healing potential, as the epithelization period was significantly reduced from 26 to 9 days when compared to the control group. Additionally, the entrapment of crocetin into phytosomes increased its stability. | [117] |
| Onosma echioides (Root bark) | Naphthoquinone-enriched extract-loaded phytosomes | Incision and excision wounds inflicted on Wister albino rats | The formulation-treated animals showed improved wound healing effects for wounds concerning both wound contraction and tensile strength compared with the control group. A reduction in lipid peroxidation and an increase in catalase activity were also indicative of reduced oxidative stress in the granulation tissue. | [118] |
| Woodfordia fruticosa (Flower extract) | Phytosomal gels | In vitro antioxidant activity assay | The phytosomal gel outperformed crude extract in terms of solubility, antioxidant activity, and in vitro drug release. | [119] |
| Herbs | Main Constituents | Dosage Forms | Administration Routes | Clinical Evidence | References |
|---|---|---|---|---|---|
| Aloe vera | Soluble sugars, polysaccharides, lignin, glycoproteins, and antiseptic agents. | Solutions, creams, mucilage, gels, and dressings. | Topical, oral | It has anti-inflammatory, anti-bacterial, and wound-contracting properties, as well as contributing to cell proliferation, collagen formation, and angiogenesis. | [122,123,124] |
| Hippophae rhamnoides | Flavonoids, tannins, vitamins (C, E, K), organic acids, glycerides of palmitic, triterpenes, stearic acid, oleic acid, and amino acids. | Aqueous leaf extract, seed oil | Topical, oral | It has antioxidant and anti-inflammatory properties, as well as the ability to promote wound contraction and epithelization and increase wound hydroxyproline and the content of protein. | [125,126] |
| Angelica Sinensis | Ferulic acid is the major active component, in addition to the essential oils, and water-soluble ingredients. | Ethanol extracts | In vitro tests | Stimulates the proliferation of human skin fibroblasts, collagen synthesis, and TGF-b production in vitro. | [127,128] |
| Catharanthus roseus (Vinca rosea) | Alkaloids, and tannins. | Leaf ethanol extract | Topical | Stimulates wound healing and wound contraction, and has antimicrobial activity against Pseudomonas aeruginosa and Staphylococcus aureus. | [129,130] |
| Calendula officinalis (Marigold) | Triterpenoids, and Flavonoids. | Gels, aqueous and alcoholic extracts | Topical | Proliferates and migrates fibroblasts in vitro; stimulates collagen synthesis and angiogenesis. Moreover, it has antimicrobial activity. | [131,132] |
| Sesamum indicum | Sesamolin, Sesaminol, and antioxidants. | Solution | Intraperitoneal, intramuscular injections | Enhances wound tensile strength, wound contraction, and hydroxyproline levels in rats using various wound models. | [133,134] |
| Morinda citrifolia (noni) | Phenols, esters, acids, alcohols, anthraquinones, flavonoids, triterpenoids, saccharides, carotenoid esters, ketone and lactone molecules, lignans, and nucleosides. | Ethanol extract of plant leaves mixed with water | Oral | Reduces wound area and epithelialization time and improves hydroxyproline content in rat excision wounds. | [135,136] |
| Camellia sinensis | Polyphenols, flavonoids, tannins, caffeine, and amino acids | Ethanolic plant extract in pure Vaseline ointment | Topical | Incision wounds in Wistar rats were reduced in healing time and wound area. | [137,138] |
| Rosmarinus officinalis L. (rosemary) | Most bioactive constituents include terpenoids and polyphenols, e.g., carnosol, rosmarinic, and carnosic acids. | Aqueous and essential oil extracts | Topical, intraperitoneal injection | In diabetic mice with full-thickness wounds, reduces inflammation and promotes wound contraction, re-epithelialization, angiogenesis, and collagen production. | [139,140,141] |
| Vesicular Carrier | Herb | Clinical Evidence | References |
|---|---|---|---|
| Liposomes | Aloe vera | Anti-inflammatory and antimicrobial activities. | [147,148,149,150,151] |
| Hippophae rhamnoides | Anti-inflammatory/antioxidant properties. | ||
| Angelica Sinensis | Stimulates collagen production, and human skin fibroblast proliferation. | ||
| Catharanthus roseus (Vinca rosea) | Antimicrobial activity. | ||
| Calendula officinalis (marigold) | Antibacterial and anti-inflammatory properties. | ||
| Noisomes | Sesamum indicum | Improves wound tensile strength, and wound contraction. | [152,153,154] |
| Morinda citrifolia (Noni) | Improves the hydroxyproline content and decreases the wound. | ||
| Rosmarinus officinalis L. (Rosemary) | Reduces inflammation and enhances wound contraction, re-epithelialization, angiogenesis, and collagen deposition in diabetic mice with full-thickness wounds. | ||
| Phytochromes | Camellia sinensis | Reduces the healing duration and wound length. | [155] |
| Patent Number | Source | Patent Title | Vesicles | Publication Date | Reference |
|---|---|---|---|---|---|
| CN102949341B | China | Tacrolimus transfersome solution and preparation method thereof | Transferosomes | 9 April 2014 | [165] |
| US7476400B2 | United States | High-concentration lidocaine compositions and methods for their preparation | Liposomes | 13 January 2009 | [166] |
| EP2915541B1 | European Patent Office | Vesicles which include epidermal growth factors and compositions that contain the same | Liposomes | 9 November 2016 | [167] |
| US5716638A | United States | Composition for applying active substances to or through the skin | Ethosomes | 10 February 1998 | [168] |
| CA2117046C | Canada | Liposomal antibiotic formulation | Liposomes | 3 October 2000 | [169] |
| US20200276231A1 | United States | Enhanced antivirulent for antibiotic-resistant bacteria | Liposomes | 3 September 2020 | [170] |
| WO2012153075A2 | WIPO | Topical cosmetic composition containing an improved pro-penetrating system | 15 November 2012 | [171] | |
| US9468599B2 | United States | Composition and method for compounded therapy | Transferosomes | 18 October 2016 | [172] |
| JP2018009026A | Japan | Transdermal delivery | Transferosomes | 18 January 2018 | [173] |
| WO2019004563A1 | WIPO | Method of preparing bioactive substance-encapsulated ethosome, ethosome composition, and cosmetic composition including ethosome composition | Ethosomes Liposomes | 3 January 2019 | [174] |
| KR100446832B1 | South Korea | Liquid droplets for the manufacture of preparation for the non-invasive application or the non-invasive transport of active ingredients through barriers | Liposomes | 4 September 2004 | [175] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Abdul Rasool, B.K.; Al Mahri, N.; Alburaimi, N.; Abdallah, F.; Shamma, A.S.B. A Narrative Review of the Potential Roles of Lipid-Based Vesicles (Vesiculosomes) in Burn Management. Sci. Pharm. 2022, 90, 39. https://doi.org/10.3390/scipharm90030039
Abdul Rasool BK, Al Mahri N, Alburaimi N, Abdallah F, Shamma ASB. A Narrative Review of the Potential Roles of Lipid-Based Vesicles (Vesiculosomes) in Burn Management. Scientia Pharmaceutica. 2022; 90(3):39. https://doi.org/10.3390/scipharm90030039
Chicago/Turabian StyleAbdul Rasool, Bazigha K., Nema Al Mahri, Nora Alburaimi, Fatima Abdallah, and Anfal Saeed Bin Shamma. 2022. "A Narrative Review of the Potential Roles of Lipid-Based Vesicles (Vesiculosomes) in Burn Management" Scientia Pharmaceutica 90, no. 3: 39. https://doi.org/10.3390/scipharm90030039
APA StyleAbdul Rasool, B. K., Al Mahri, N., Alburaimi, N., Abdallah, F., & Shamma, A. S. B. (2022). A Narrative Review of the Potential Roles of Lipid-Based Vesicles (Vesiculosomes) in Burn Management. Scientia Pharmaceutica, 90(3), 39. https://doi.org/10.3390/scipharm90030039









