In Vitro and In Silico Antistaphylococcal Activity of Indole Alkaloids Isolated from Tabernaemontana cymosa Jacq (Apocynaceae)
Abstract
1. Introduction
2. Materials and Methods
2.1. General Experimental Procedures
2.2. Isolation and Characterization of T. cymosa Alkaloids
2.3. Maintenance and Cultivation of Bacterial Strains
2.4. Minimum Inhibitory Concentration (MIC) of Indole Alkaloids
2.5. In Silico Study
Ligands and Receptors Preparation
2.6. Molecular Docking between Indole Alkaloids from T. cymosa and S. aureus Proteins
2.7. Statistical Analyses
3. Results
3.1. Isolation and Characterization of the Alkaloids Isolated from T. cymosa
3.2. Antibacterial Activity of the Alkaloids Isolated from T. cymosa
3.3. Validation of the Docking Method
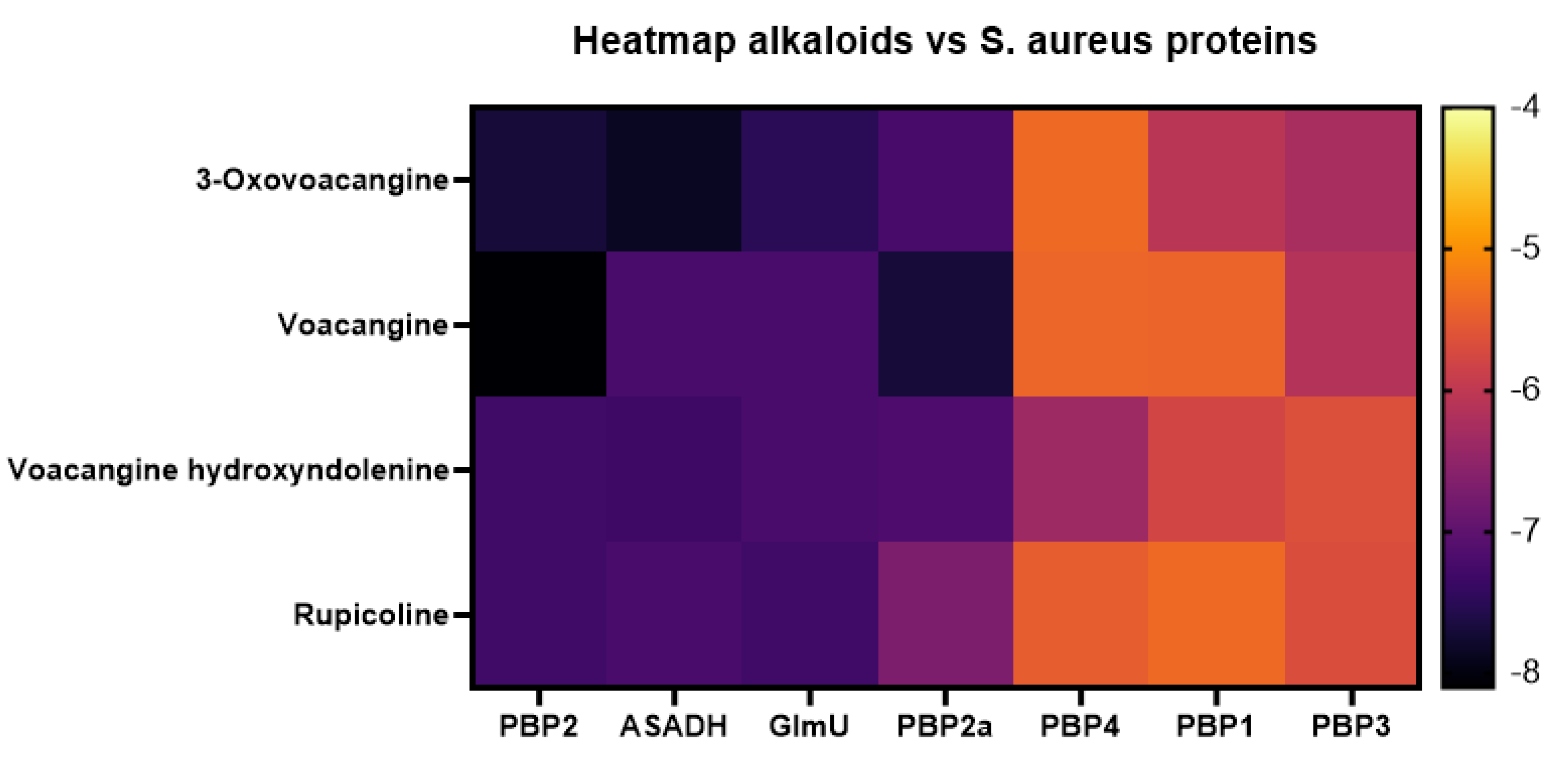
3.4. Molecular Docking Affinities of T. cymosa Alkaloids against S. aureus Proteins
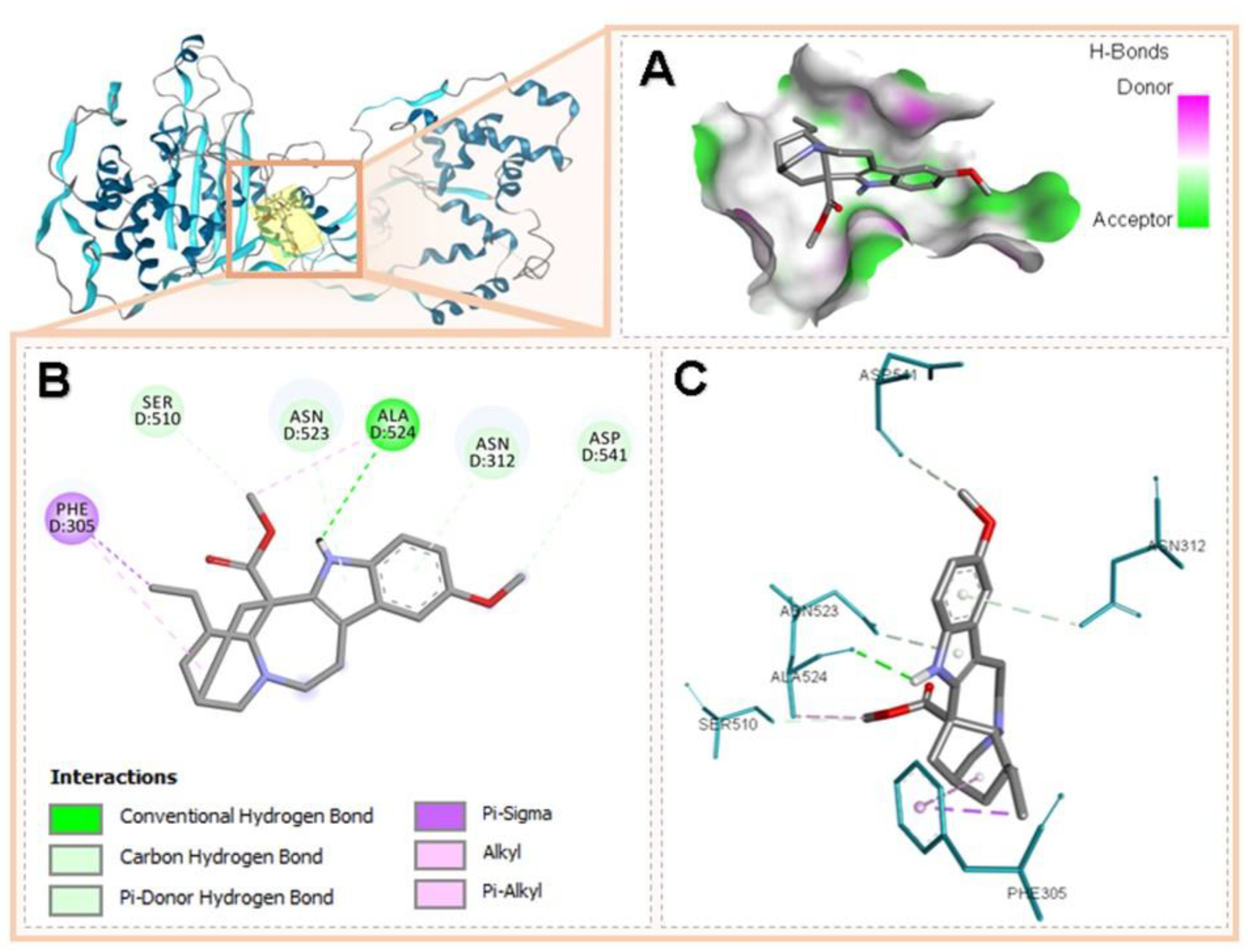
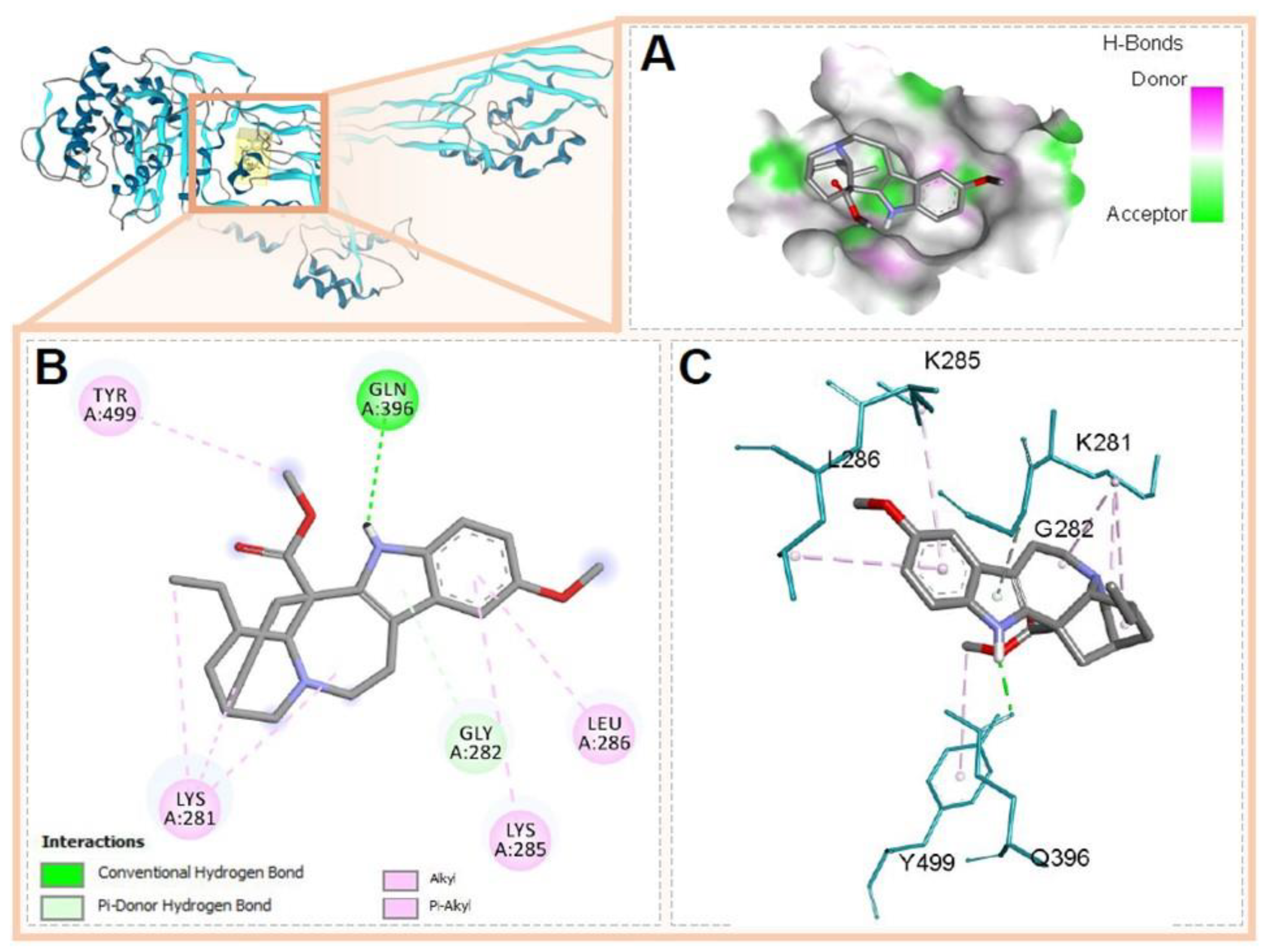
3.5. Molecular Docking of Voacangine and Proteins from S. aureus
3.6. ADMET Analysis of Voacangine Compound
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Silveira, D.; de Melo, A.M.M.F.; Magalhães, P.O.; Fonseca-Bazzo, Y.M. Tabernaemontana species: Promising sources of new useful drugs. In Studies in Natural Products Chemistry; Atta-ur-Rahman, Ed.; Elsevier: Amsterdam, The Netherlands, 2017; Volume 54, pp. 227–289. ISBN 9780444639295. [Google Scholar]
- Marinho, F.F.; Simões, A.O.; Barcellos, T.; Moura, S. Brazilian Tabernaemontana genus: Indole alkaloids and phytochemical activities. Fitoterapia 2016, 114, 127–137. [Google Scholar] [CrossRef] [PubMed]
- Van Beek, T.A.; Deelder, A.M.; Verpoorte, R.; Svendsen, A.B. Antimicrobial, antiamoebic and antiviral screening of some Tabernaemontana species. Planta Med. 1984, 50, 180–185. [Google Scholar] [CrossRef] [PubMed]
- Van Beek, T.A.; Verpoorte, R.; Svendsen, A.B.; Leeuwenberg, A.J.M.; Bisset, N.G. Tabernaemontana L. (Apocynaceae): A review of its taxonomy, phytochemistry, ethnobotany and pharmacology. J. Ethnopharmacol. 1984, 10, 1–156. [Google Scholar] [CrossRef]
- De Lourdes, F.D.; Passos, M.S.; Braz-Filho, R.; Vieira, I.J.C. Biological Activity and 13C NMR Spectral Data of Iboga-Type Skeleton Alkaloids. In Studies in Natural Products Chemistry; Atta-ur-Rahman, Ed.; Elsevier: Amsterdam, The Netherlands, 2022; Volume 72, pp. 287–369. ISBN 1572-5995. [Google Scholar]
- Danieli, B.; Palmisano, G. Chapter 1 Alkaloids from Tabernaemontana. In The Alkaloids: Chemistry and Pharmacology; Brossi, A., Ed.; Academic Press: Cambridge, MA, USA, 1986; Volume 27, pp. 1–130. ISBN 9780124695276. [Google Scholar]
- Casanova-Pérez, C.; Delgado-Caballero, C.E.; Cruz-Bautista, P.; Casanova-Pérez, L. Plantas Medicinales Usadas por los Tének en la Huasteca, México. CienciaUAT 2022, 16, 40–58. [Google Scholar] [CrossRef]
- Duraipandiyan, V.; Ayyanar, M.; Ignacimuthu, S. Antimicrobial Activity of Some Ethnomedicinal Plants Used by Paliyar Tribe from Tamil Nadu, India. BMC Complement. Altern. Med. 2006, 6, 35. [Google Scholar] [CrossRef]
- Shrikanth, V.M.; Janardhan, B.; Dhananjaya, D.L.; Muddapura, U.M.; More, S.S. Antimicrobial and Antioxidant Activity of Methanolic Root Extract of Tabernaemontana alternifolia L. Int. J. Pharm. Pharm. Sci. 2015, 7, 66–69. [Google Scholar]
- Sathishkumar, T.; Baskar, R.; Rajeshkumar, M. In Vitro Antibacterial and Antifungal Activities of Tabernaemontana heyneana Wall. Leaves. J. Appl. Pharm. Sci. 2012, 2, 107–111. [Google Scholar] [CrossRef]
- Marathe, N.P.; Rasane, M.H.; Kumar, H.; Patwardhan, A.A.; Shouche, Y.S.; Diwanay, S.S. In Vitro Antibacterial Activity of Tabernaemontana alternifolia (Roxb) Stem Bark Aqueous Extracts against Clinical Isolates of methicillin resistant Staphylococcus aureus. Ann. Clin. Microbiol. Antimicrob. 2013, 12, 26. [Google Scholar] [CrossRef]
- Suffredini, I.B.; Bacchi, E.M.; Sakuda, T.M.K.; Ohara, M.T.; Younes, R.N.; Varella, A.D. Antibacterial Activity of Apocynaceae Extracts and MIC of Tabernaemontana angulata Stem Organic Extract. Rev. Bras. Cienc. Farm. 2002, 38, 89–94. [Google Scholar] [CrossRef]
- Guida, A.; Battista, G.D.; Bargardi, S. Actividad antibacteriana de alcaloides de Tabernaemontana catharinensis A. DC. Ars Pharmaceutica 2003, 44, 167–173. [Google Scholar]
- Boligon, A.A.; Piana, M.; Kubiça, T.F.; Mario, D.N.; Dalmolin, T.V.; Bonez, P.C.; Weiblen, R.; Lovato, L.; Alves, S.H.; Campos, M.M.A.; et al. HPLC Analysis and Antimicrobial, Antimycobacterial and Antiviral Activities of Tabernaemontana catharinensis A. DC. J. Appl. Biomed. 2015, 13, 7–18. [Google Scholar] [CrossRef]
- Medeiros, M.R.F.; de Melo Prado, L.A.; Fernandes, V.C.; Figueiredo, S.S.; Coppede, J.; Martins, J.; Fiori, G.M.L.; Martinez-Rossi, N.M.; Beleboni, R.O.; Contini, S.H.T.; et al. Antimicrobial Activities of Indole Alkaloids from Tabernaemontana catharinensis. Nat. Prod. Commun. 2011, 6, 1934578X1100600209. [Google Scholar] [CrossRef]
- Mohamad, S.; Zin, N.M.; Wahab, H.A.; Ibrahim, P.; Sulaiman, S.F.; Zahariluddin, A.S.M.; Md Noor, S.S. Antituberculosis Potential of Some Ethnobotanically Selected Malaysian Plants. J. Ethnopharmacol. 2011, 133, 1021–1026. [Google Scholar] [CrossRef] [PubMed]
- Mohamad, S.; Ismail, N.N.; Parumasivam, T.; Ibrahim, P.; Osman, H.; Wahab, H.A. Antituberculosis Activity, Phytochemical Identification of Costus Speciosus (J. Koenig) Sm., Cymbopogon citratus (DC. Ex Nees) Stapf., and Tabernaemontana coronaria (L.) Willd. and Their Effects on the Growth Kinetics and Cellular Integrity of Mycobacterium tuberculosis H37Rv. BMC Complement. Altern. Med. 2018, 18, 5. [Google Scholar] [CrossRef]
- Marie-Magdeleine, C.; Mahieu, M.; D’Alexis, S.; Philibert, L.; Archimede, H. In Vitro Effects of Tabernaemontana citrifolia Extracts on Haemonchus contortus. Res. Vet. Sci. 2010, 89, 88–92. [Google Scholar] [CrossRef]
- Ashikur, R.M.; Hasanuzzaman, M.D.; Mofizur, R.; Zahan, S.I.; Muhuri, R.S. Evaluation of antibacterial activity of study of leaves of Tabernaemontana divaricata (L.). Int. Res. J. Pharm. 2011, 2, 123–127. [Google Scholar]
- Pallant, C.A.; Cromarty, A.D.; Steenkamp, V. Effect of an Alkaloidal Fraction of Tabernaemontana elegans (Stapf.) on Selected Micro-Organisms. J. Ethnopharmacol. 2012, 140, 398–404. [Google Scholar] [CrossRef]
- Rukunga, G.M.; Kofi-Tsekpo, M.W.; Kurokawa, M.; Kageyama, S.; Mungai, G.M.; Muli, J.M.; Tolo, F.M.; Kibaya, R.M.; Muthaura, C.N.; Kanyara, J.N. Evaluation of the HIV-1 Reverse Transcriptase Inhibitory Properties of Extracts from Some Medicinal Plants in Kenya. Afr. J. Health Sci. 2002, 9, 81–90. [Google Scholar] [CrossRef][Green Version]
- Lavaud, C.; Massiot, G. The Iboga Alkaloids. In Progress in the Chemistry of Organic Natural Products 105; Kinghorn, A.D., Falk, H., Gibbons, S., Kobayashi, J., Eds.; Springer International Publishing: Cham, Switzerland, 2017; pp. 89–136. ISBN 978-3-319-49712-9. [Google Scholar]
- Ding, C.-F.; Ma, H.-X.; Yang, J.; Qin, X.-J.; Njateng, G.S.S.; Yu, H.-F.; Wei, X.; Liu, Y.-P.; Huang, W.-Y.; Yang, Z.-F.; et al. Antibacterial Indole Alkaloids with Complex Heterocycles from Voacanga africana. Org. Lett. 2018, 20, 2702–2706. [Google Scholar] [CrossRef]
- Achenbach, H. Investigations on West African medicinal plants. Pure Appl. Chem. 1986, 58, 653–662. [Google Scholar] [CrossRef]
- Bernal, R.; Gradstein, R.; Celis, M. Catalogo de Plantas y Líquenes de Colombia; Primera Edicion; Universidad Nacional de Colombia: Bogotá, Colombia, 2016; Volume 1, ISBN 978-958-775-720-0. [Google Scholar]
- Achenbach, H.; Benirschke, M.; Torrenegra, R. Alkaloids and Other Compounds from Seeds of Tabernaemontana cymose. Phytochemistry 1997, 45, 325–335. [Google Scholar] [CrossRef]
- Rastogi, N.; Abaul, J.; Goh, K.S.; Devallois, A.; Philogène, E.; Bourgeois, P. Antimycobacterial Activity of Chemically Defined Natural Substances from the Caribbean Flora in Guadeloupe. FEMS Immunol. Med. Microbiol. 1998, 20, 267–273. [Google Scholar] [CrossRef] [PubMed]
- Díaz Castillo, F.; Morelos Cardona, S.M.; Carrascal Medina, M.; Pájaro González, Y.; Gómez Estrada, H. Actividad Larvicida de Extractos Etanólicos de Tabernaemontana cymosa y Trichilia hirta Sobre Larvas de Estadio III y IV de Aedes aegypti (Diptera: Culicidae). Rev. Cuba. Plantas Med. 2012, 17, 256–267. [Google Scholar]
- Monsalve-Escudero, L.M.; Loaiza-Cano, V.; Pájaro-González, Y.; Oliveros-Díaz, A.F.; Diaz-Castillo, F.; Quiñones, W.; Robledo, S.; Martinez-Gutierrez, M. Indole Alkaloids Inhibit Zika and Chikungunya Virus Infection in Different Cell Lines. BMC Complement. Med. Ther. 2021, 21, 216. [Google Scholar] [CrossRef] [PubMed]
- Monsalve-Escudero, L.M.; Loaiza-Cano, V.; Zapata-Cardona, M.I.; Quintero-Gil, D.C.; Hernández-Mira, E.; Pájaro-González, Y.; Oliveros-Díaz, A.F.; Diaz-Castillo, F.; Quiñones, W.; Robledo, S.; et al. The Antiviral and Virucidal Activities of Voacangine and Structural Analogs Extracted from Tabernaemontana cymosa Depend on the Dengue Virus Strain. Plants 2021, 10, 1280. [Google Scholar] [CrossRef] [PubMed]
- CLSI. Methods for Dilution Antimicrobial Susceptibility Tests f or Bacteria That Grow Aerobically, 9th ed.; CLSI Document M07-A9; Clinical and Laboratory Standards Institute: Wayne, PA, USA, 2012; Volume 32. [Google Scholar]
- Pájaro-González, Y.; Oliveros-Díaz, A.F.; Cabrera-Barraza, J.; Fernández-Daza, E.; Reyes, N.; Montes-Guevara, O.A.; Caro-Fuentes, D.; Franco-Ospina, L.; Quiñones-Fletcher, W.; Quave, C.L.; et al. Mammea B/BA Isolated from the Seeds of Mammea americana L. (Calophyllaceae) Is a Potent Inhibitor of Methicillin-Resistant Staphylococcus aureus. Front. Pharmacol. 2022, 13, 826404. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.; Thiessen, P.A.; Cheng, T.; Yu, B.; Shoemaker, B.A.; Wang, J.; Bolton, E.E.; Wang, Y.; Bryant, S.H. Literature Information in PubChem: Associations between PubChem Records and Scientific Articles. J. Cheminform. 2016, 8, 32. [Google Scholar] [CrossRef]
- Frisch, M.; Trucks, G.; Schlegel, H.; Scuseria, G.; Robb, M.; Cheeseman, J.; Scalmani, G.; Barone, V.; Mennucci, B.; Petersson, G. Gaussian 09 Citation; Gaussian Inc.: Wallingford, UK, 2016. [Google Scholar]
- Burley, S.K.; Berman, H.M.; Kleywegt, G.J.; Markley, J.L.; Nakamura, H.; Velankar, S. Protein Data Bank (PDB): The Single Global Macromolecular Structure Archive. Methods Mol. Biol. 2017, 1607, 627–641. [Google Scholar] [CrossRef] [PubMed]
- Figueira, L.; Li, W.Z.; McWilliam, H.; Lopez, R.; Xenarios, I.; Bougueleret, L.; Bridge, A.; Poux, S.; Redaschi, N.; Aimo, L.; et al. The UniProt Consortium UniProt: A Hub for Protein Information. Nucleic Acids Res. 2015, 43, D204–D212. [Google Scholar] [CrossRef]
- Waterhouse, A.; Bertoni, M.; Bienert, S.; Studer, G.; Tauriello, G.; Gumienny, R.; Heer, F.T.; de Beer, T.A.P.; Rempfer, C.; Bordoli, L.; et al. SWISS-MODEL: Homology Modelling of Protein Structures and Complexes. Nucleic Acids Res. 2018, 46, W296–W303. [Google Scholar] [CrossRef] [PubMed]
- Trott, O.; Olson, A.J. AutoDock Vina: Improving the Speed and Accuracy of Docking with a New Scoring Function, Efficient Optimization, and Multithreading. J. Comput. Chem. 2010, 31, 455–461. [Google Scholar] [CrossRef] [PubMed]
- Samal, D. In Silico Homology Modeling and Docking Study of Translationally Controlled Tumor Protein of Labeorohita. Int. J. For. Fish. Res. 2017, 1, 8–22. [Google Scholar] [CrossRef]
- Biovia, D.; Berman, H.; Westbrook, J.; Feng, Z.; Gilliland, G.; Bhat, T.; Richmond, T. Dassault Systèmes BIOVIA, Discovery Studio Visualizer, v. 17.2, San Diego: Dassault Systèmes, 2016. J. Chem. Phys. 2000, 10, 21–9991. [Google Scholar]
- Lagorce, D.; Bouslama, L.; Becot, J.; Miteva, M.A.; Villoutreix, B.O. FAF-Drugs4: Free ADME-Tox Filtering Computations for Chemical Biology and Early Stages Drug Discovery. Bioinformatics 2017, 33, 3658–3660. [Google Scholar] [CrossRef] [PubMed]
- Miteva, M.A.; Violas, S.; Montes, M.; Gomez, D.; Tuffery, P.; Villoutreix, B.O. FAF-Drugs: Free ADME/Tox Filtering of Compound Collections. Nucleic Acids Res. 2006, 34, W738–W744. [Google Scholar] [CrossRef] [PubMed]
- Gómez-Calderón, C.; Mesa-Castro, C.; Robledo, S.; Gómez, S.; Bolivar-Avila, S.; Diaz-Castillo, F.; Martínez-Gutierrez, M. Antiviral Effect of Compounds Derived from the Seeds of Mammea americana and Tabernaemontana cymosa on Dengue and Chikungunya Virus Infections. BMC Complement. Altern. Med. 2017, 17, 57. [Google Scholar] [CrossRef]
- Oliveros-Díaz, A.; Olivero-Verbel, J.; Pájaro-González, Y.; Díaz-Castillo, F. Molecular Human Targets of Bioactive Alkaloid-Type Compounds from Tabernaemontana cymosa Jacq. Molecules 2021, 26, 3765. [Google Scholar] [CrossRef]
- Orry, A.J.W.; Abagyan, R. (Eds.) Homology Modeling: Methods and Protocols, 1st ed.; Springer: New York, NY, USA, 2012; Volume 857. [Google Scholar]
- Hevener, K.E.; Zhao, W.; Ball, D.M.; Babaoglu, K.; Qi, J.; White, S.W.; Lee, R.E. Validation of Molecular Docking Programs for Virtual Screening against Dihydropteroate Synthase. J. Chem. Inf. Model. 2009, 49, 444–460. [Google Scholar] [CrossRef]
- Laksmiani, N.P.L.; Widiastari, M.I.; Reynaldi, K.R. The Inhibitory Activity of Peonidin Purple Sweet Potato in Human Epidermal Receptor-2 Receptor (Her-2) Expression by in Silico Study. J. Phys. Conf. Ser. 2018, 1040, 012010. [Google Scholar] [CrossRef]
- Siahaan, P.; Darmastuti, N.; Aisyafalah, S.; Sasongko, N.; Hudiyanti, D.; Asy’ari, M.; Prasasty, V. Probing the Interaction between EC1-EC2 Domain of E-Cadherin with Conformational Structure of Cyclic ADTC7 (Ac-CDTPDC-NH2) Peptide Using Molecular Docking Approach; IOP Publishing: Bristol, UK, 2020. [Google Scholar]
- Lovering, A.L.; De Castro, L.; Strynadka, N.C.J. Identification of Dynamic Structural Motifs Involved in Peptidoglycan Glycosyltransfer. J. Mol. Biol. 2008, 383, 167–177. [Google Scholar] [CrossRef]
- Bouley, R.; Kumarasiri, M.; Peng, Z.; Otero, L.H.; Song, W.; Suckow, M.A.; Schroeder, V.A.; Wolter, W.R.; Lastochkin, E.; Antunes, N.T.; et al. Discovery of Antibiotic (E)-3-(3-Carboxyphenyl)-2-(4-Cyanostyryl)Quinazolin-4(3H)-One. J. Am. Chem. Soc. 2015, 137, 1738–1741. [Google Scholar] [CrossRef] [PubMed]
- Chesnel, L.; Pernot, L.; Lemaire, D.; Champelovier, D.; Croizé, J.; Dideberg, O.; Vernet, T.; Zapun, A. The Structural Modifications induced by the M339F Substitution in PBP2x from Streptococcus Pneumoniae Further Decreases the Susceptibility to β-Lactams of Resistant Strains. J. Biol. Chem. 2003, 278, 44448–44456. [Google Scholar] [CrossRef] [PubMed]
- Yoshida, H.; Kawai, F.; Obayashi, E.; Akashi, S.; Roper, D.I.; Tame, J.R.H.; Park, S.-Y. Crystal Structures of Penicillin-Binding Protein 3 (PBP3) from methicillin-Resistant Staphylococcus aureus in the Apo and Cefotaxime-Bound Forms. J. Mol. Biol. 2012, 423, 351–364. [Google Scholar] [CrossRef] [PubMed]
- Alexander, J.A.N.; Chatterjee, S.S.; Hamilton, S.M.; Eltis, L.D.; Chambers, H.F.; Strynadka, N.C.J. Structural and Kinetic Analyses of Penicillin-Binding Protein 4 (PBP4)-Mediated Antibiotic Resistance in Staphylococcus aureus. J. Biol. Chem. 2018, 293, 19854–19865. [Google Scholar] [CrossRef] [PubMed]
- Wang, Q.; Lv, Y.; Pang, J.; Li, X.; Lu, X.; Wang, X.; Hu, X.; Nie, T.; Yang, X.; Xiong, Y.Q.; et al. In Vitro and In Vivo Activity of D-Serine in Combination with β-Lactam Antibiotics against methicillin-Resistant Staphylococcus aureus. Acta Pharm. Sin. B 2019, 9, 496–504. [Google Scholar] [CrossRef]
- Sulzenbacher, G.; Gal, L.; Peneff, C.; Fassy, F.; Bourne, Y. Crystal Structure of Streptococcus pneumoniae N-Acetylglucosamine-1-Phosphate Uridyltransferase Bound to Acetyl-Coenzyme A Reveals a Novel Active Site Architecture. J. Biol. Chem. 2001, 276, 11844–11851. [Google Scholar] [CrossRef]
- Kumar, R.; Garg, P. Molecular Modeling and Active Site Binding Mode Characterization of Aspartate β-Semialdehyde Dehydrogenase Family. Mol. Inform. 2013, 32, 377–383. [Google Scholar] [CrossRef]
- Santos, V.; Gonsalves, A.; Araújo, C. Abordagem didactica para o desenvolvimiento de moléculas bioativas: Regra dos cinco de Lipinski e preparacao de heterociclo1,3,4-oxadiazol em forno de micro-onda doméstico. Quim. Nova 2017, 41, 110–115. [Google Scholar] [CrossRef]
- Lipinski, C.A.; Lombardo, F.; Dominy, B.W.; Feeney, P.J. Experimental and Computational Approaches to Estimate Solubility and Permeability in Drug Discovery and Development Settings. Adv. Drug Deliv. Rev. 2001, 46, 3–26. [Google Scholar] [CrossRef]
- Zhang, M.-Q.; Wilkinson, B. Drug Discovery beyond the ‘Rule-of-Five.’. Curr. Opin. Biotechnol. 2007, 18, 478–488. [Google Scholar] [CrossRef]
- Veber, D.F.; Johnson, S.R.; Cheng, H.-Y.; Smith, B.R.; Ward, K.W.; Kopple, K.D. Molecular Properties That Influence the Oral Bioavailability of Drug Candidates. J. Med. Chem. 2002, 45, 2615–2623. [Google Scholar] [CrossRef]
- Hill, A.P.; Young, R.J. Getting Physical in Drug Discovery: A Contemporary Perspective on Solubility and Hydrophobicity. Drug Discov. Today 2010, 15, 648–655. [Google Scholar] [CrossRef] [PubMed]
- McGovern, S.L.; Caselli, E.; Grigorieff, N.; Shoichet, B.K. A Common Mechanism Underlying Promiscuous Inhibitors from Virtual and High-Throughput Screening. J. Med. Chem. 2002, 45, 1712–1722. [Google Scholar] [CrossRef] [PubMed]
- Roche, O.; Schneider, P.; Zuegge, J.; Guba, W.; Kansy, M.; Alanine, A.; Bleicher, K.; Danel, F.; Gutknecht, E.-M.; Rogers-Evans, M.; et al. Development of a Virtual Screening Method for Identification of “Frequent Hitters” in Compound Libraries. J. Med. Chem. 2002, 45, 137–142. [Google Scholar] [CrossRef] [PubMed]
- Lajiness, M.S.; Vieth, M.; Erickson, J. Molecular Properties That Influence Oral Drug-like Behavior. Curr. Opin. Drug Discov. Dev. 2004, 7, 470–477. [Google Scholar]
- Lim, D.; Strynadka, N.C.J. Structural Basis for the β Lactam Resistance of PBP2a from methicillin-Resistant Staphylococcus aureus. Nat. Struct. Biol. 2002, 9, 870–876. [Google Scholar] [CrossRef]
- Rani, N.; Vijayakumar, S.; Lakshmi, P.T.V.; Arunachalam, A. Allosteric Site-Mediated Active Site Inhibition of PBP2a Using Quercetin 3-O-Rutinoside and Its Combination. J. Biomol. Struct. Dyn. 2016, 34, 1778–1796. [Google Scholar] [CrossRef]
- Shalaby, M.-A.W.; Dokla, E.M.E.; Rabah, A.T.S.; Abouzid, K.A.M. Penicillin Binding Protein 2a: An Overview and a Medicinal Chemistry Perspective. Eur. J. Med. Chem. 2020, 199, 112312. [Google Scholar] [CrossRef]
- Otero, L.H.; Rojas-Altuve, A.; Llarrull, L.I.; Carrasco-López, C.; Kumarasiri, M.; Lastochkin, E.; Fishovitz, J.; Dawley, M.; Hesek, D.; Lee, M.; et al. How Allosteric Control of Staphylococcus aureus Penicillin Binding Protein 2a Enables methicillin Resistance and Physiological Function. Proc. Natl. Acad. Sci. USA 2013, 110, 16808–16813. [Google Scholar] [CrossRef]
- Mahasenan, K.V.; Molina, R.; Bouley, R.; Batuecas, M.T.; Fisher, J.F.; Hermoso, J.A.; Chang, M.; Mobashery, S. Conformational Dynamics in Penicillin-Binding Protein 2a of methicillin-Resistant Staphylococcus aureus, Allosteric Communication Network and Enablement of Catalysis. J. Am. Chem. Soc. 2017, 139, 2102–2110. [Google Scholar] [CrossRef]
- Chiang, Y.-C.; Wong, M.T.Y.; Essex, J.W. Molecular Dynamics Simulations of Antibiotic Ceftaroline at the Allosteric Site of Penicillin-Binding Protein 2a (PBP2a). Israel J. Chem. 2020, 60, 754–763. [Google Scholar] [CrossRef]
- Bouhss, A.; Trunkfield, A.E.; Bugg, T.D.H.; Mengin-Lecreulx, D. The Biosynthesis of Peptidoglycan Lipid-Linked Intermediates. FEMS Microbiol. Rev. 2008, 32, 208–233. [Google Scholar] [CrossRef] [PubMed]
- Iyer, R.N.; Favela, D.; Zhang, G.; Olson, D.E. The Iboga Enigma: The Chemistry and Neuropharmacology of Iboga Alkaloids and Related Analogs. Nat. Prod. Rep. 2021, 38, 307–329. [Google Scholar] [CrossRef] [PubMed]
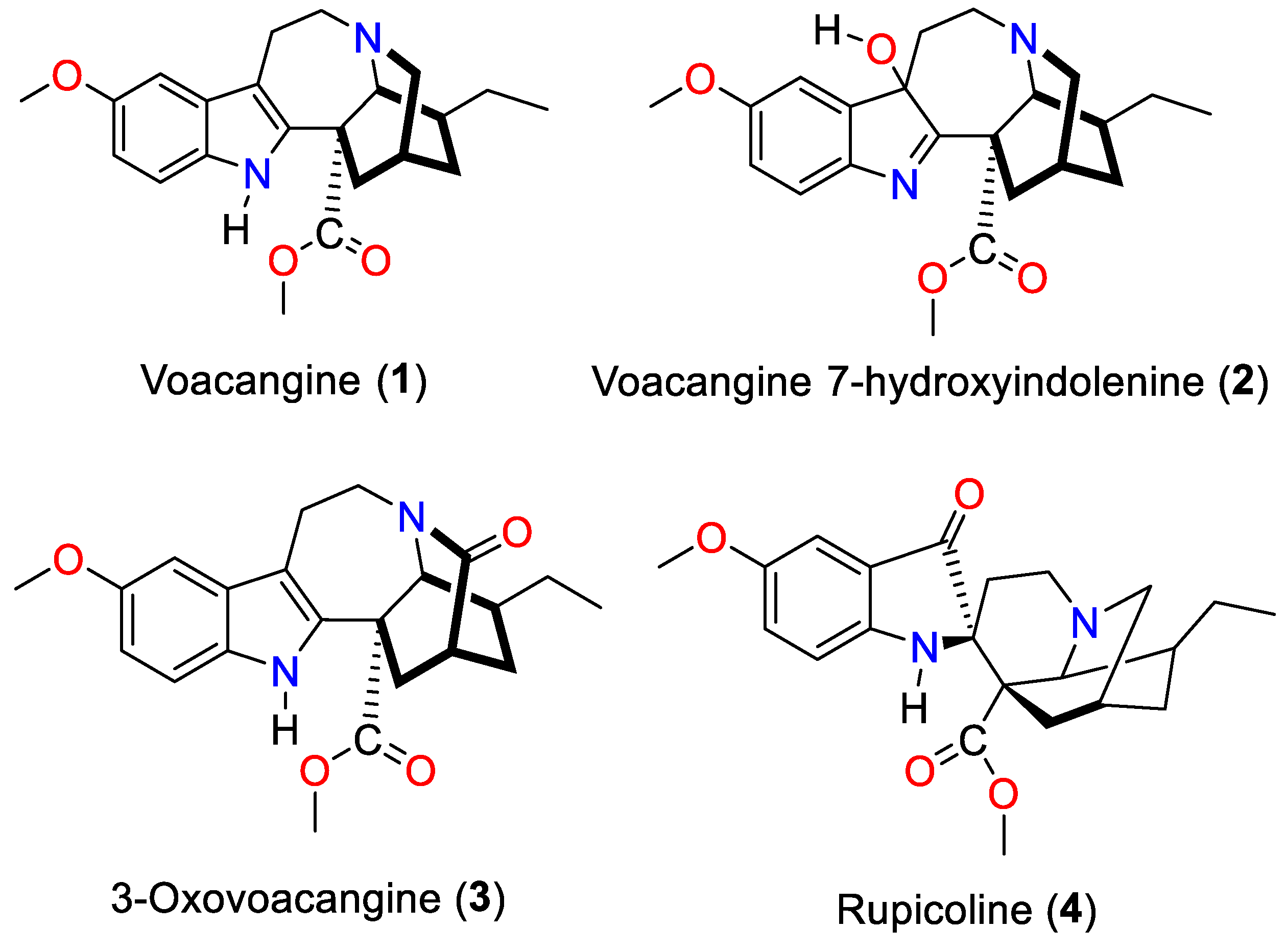
| Carbon | Voacangine | Voacangine-7-hydroxyindolenine | Rupicoline | 3-Oxo-voacangine | ||||
|---|---|---|---|---|---|---|---|---|
| 13C | 1H | 13C | 1H | 13C | 1H | 13C | 1H | |
| 2 | 137.65 | 186.97 | 68.39 | 134.65 | ||||
| 3 | 51.6 | 48.75 | 52.07 | 173.07 | ||||
| 5 | 53.24 | 49.22 | 47.67 | 42.72 | ||||
| 6 | 22.33 | 34.25 | 25.77 | 21.18 | ||||
| 7 | 100.84 | 88.43 | 202.9 | 109.22 | ||||
| 8 | 129.31 | 144.53 | 121.78 | 128.23 | ||||
| 9 | 110.24 | 6.94 | 108.09 | 6.91 | 104.63 | 7.02 | 100.48 | 6.95 |
| 10 | 154.11 | 159.23 | 153.82 | 154.21 | ||||
| 11 | 111.96 | 6.83 | 113.85 | 6.81 | 126.89 | 7.07 | 112.59 | 6.81 |
| 12 | 111.23 | 7.16 | 121.49 | 7.36 | 114.12 | 6.76 | 111.38 | 7.37 |
| 13 | 130.13 | 144.9 | 154.21 | 130.83 | ||||
| 14 | 27.44 | 27.08 | 31.1 | 35.97 | ||||
| 15 | 32.14 | 32.14 | 31.1 | 31.03 | ||||
| 16 | 55.25 | 55.88 | 52.12 | 56.11 | ||||
| 17 | 36.67 | 34.63 | 30.8 | 35.49 | ||||
| 18 | 11.82 | 0.91 | 11.71 | 0.86 | 12.14 | 0.91 | 11.41 | 0.86 |
| 19 | 26.86 | 26.62 | 28.69 | 27.67 | ||||
| 20 | 39.27 | 37.68 | 35.81 | 38.21 | ||||
| 21 | 57.69 | 3.56 | 58.68 | 52.05 | 3.95 | 56.02 | ||
| 22 | 176.03 | 174.05 | 174.57 | 175.6 | ||||
| CO2CH3 | 52.74 | 3.73 | 53.39 | 3.7 | 52.07 | 3.3 | 53.09 | 3.77 |
| OCH3 | 56.15 | 3.87 | 55.88 | 3.81 | 55.9 | 3.76 | 55.6 | 3.88 |
| Isolated Compounds | Bacterial Stains Tested | |
| MSSA (ATCC 29213) | MRSA (ATCC 33591) | |
| MIC90 (µg/mL) | ||
| Voacangine | 50 | 50 |
| Voacangine-7-hydroxyindolenine | >64 | >64 |
| 3-Oxo-voacangine | >64 | >64 |
| Rupicoline | >64 | >64 |
| Oxacillin | <0.5 | 256 |
| Vancomycin | 1 | 1 |
| Compound | Protein | Database ID | Specific Amino Acid Residues Involved | Type of Interaction |
|---|---|---|---|---|
| Voacangine | PBP1 | 5TRO | Asp257, Asn175, Phe176, Val250 | Conventional H-bridge, Pi-Anion, Alkyl, Pi-Alkyl, Pi-Alkyl |
| PBP2 | 3DWK Q8KHY3 | Phe305, Ser510, Asn523, Ala524, Asn312, Asp541 | Conventional H-Bridge, Hydrogen–Carbon Bridge, Pi-Donor H-Bridge, Pi-Sigma, Alkyl, Pi-Alkyl | |
| PBP2a | 4CJN A0A0J9X1X5 | Gln396, Tyr499, Lys281, Gly282, Leu285, Leu286 | Conventional H-Bridge, Pi-Donor H-Bridge, Alkyl, Pi-Alkyl | |
| PBP3 | 3VSL | Lys326, Lys375, Asp378, Glu329, Leu365 | Conventional H-Bridge, Pi-Anion, Alkyl | |
| PBP4 | 5TXI | Arg280, Ile216, Lys217, Tyr374 | Conventional H-bridge, Alkyl, Pi-Alkyl | |
| GlmU | Q6GJH2 | Lys108, Arg242, Tyr246, Val266 | Conventional H-bridge, Hydrogen–Carbon Bridge, Pi-Pi T-Shaped, Alkyl | |
| ASADH | A0A7U7EUW2 | Asn94, Cys126, Val13, Leu12, Gly159 | Conventional H-bridge, Hydrogen–Carbon Bridge, Non-Favorable Donor-Donor, Alkyl, Pi-Alkyl |
| Parameter Evaluated | Voacangine Compound |
|---|---|
| Molecular Weight | 368.47 |
| Log P | 3.51 |
| HBD | 1 |
| HBA | 5 |
| Rotating links | 4 |
| TPSA (Å2) | 55.76 |
| Toxic functional groups | 0 |
| Result | Accepted |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pájaro-González, Y.; Cabrera-Barraza, J.; Martelo-Ramírez, G.; Oliveros-Díaz, A.F.; Urrego-Álvarez, J.; Quiñones-Fletcher, W.; Díaz-Castillo, F. In Vitro and In Silico Antistaphylococcal Activity of Indole Alkaloids Isolated from Tabernaemontana cymosa Jacq (Apocynaceae). Sci. Pharm. 2022, 90, 38. https://doi.org/10.3390/scipharm90020038
Pájaro-González Y, Cabrera-Barraza J, Martelo-Ramírez G, Oliveros-Díaz AF, Urrego-Álvarez J, Quiñones-Fletcher W, Díaz-Castillo F. In Vitro and In Silico Antistaphylococcal Activity of Indole Alkaloids Isolated from Tabernaemontana cymosa Jacq (Apocynaceae). Scientia Pharmaceutica. 2022; 90(2):38. https://doi.org/10.3390/scipharm90020038
Chicago/Turabian StylePájaro-González, Yina, Julián Cabrera-Barraza, Geraldine Martelo-Ramírez, Andrés F. Oliveros-Díaz, Juan Urrego-Álvarez, Wiston Quiñones-Fletcher, and Fredyc Díaz-Castillo. 2022. "In Vitro and In Silico Antistaphylococcal Activity of Indole Alkaloids Isolated from Tabernaemontana cymosa Jacq (Apocynaceae)" Scientia Pharmaceutica 90, no. 2: 38. https://doi.org/10.3390/scipharm90020038
APA StylePájaro-González, Y., Cabrera-Barraza, J., Martelo-Ramírez, G., Oliveros-Díaz, A. F., Urrego-Álvarez, J., Quiñones-Fletcher, W., & Díaz-Castillo, F. (2022). In Vitro and In Silico Antistaphylococcal Activity of Indole Alkaloids Isolated from Tabernaemontana cymosa Jacq (Apocynaceae). Scientia Pharmaceutica, 90(2), 38. https://doi.org/10.3390/scipharm90020038







