Comparison of the Purity and Impurity of Glucagon-for-Injection Products under Various Stability Conditions
Abstract
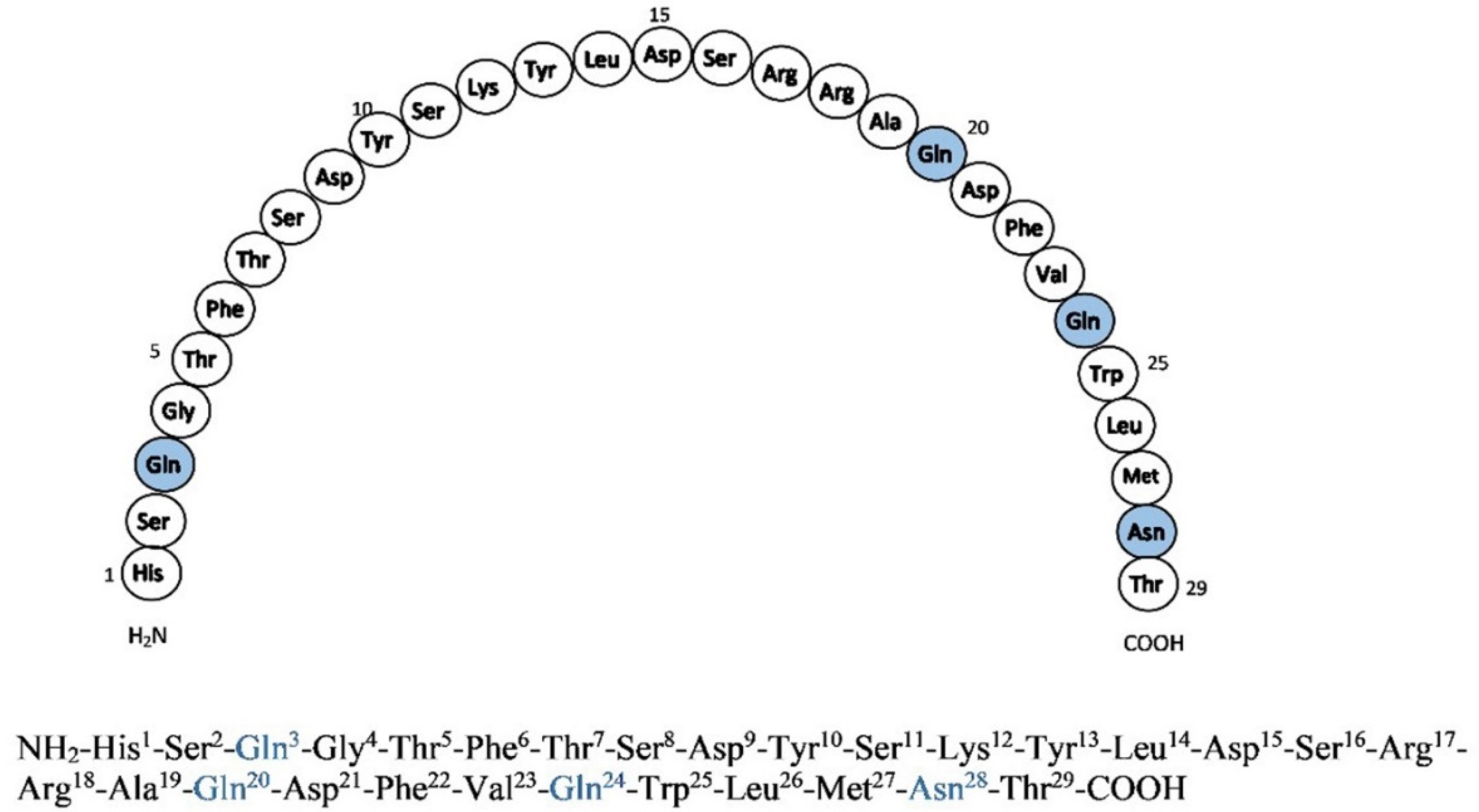
1. Introduction
2. Materials and Methods
2.1. Sample Preparation
2.2. UPLC Method
2.3. Data Analysis
2.4. Equivalence Evaluation Criteria
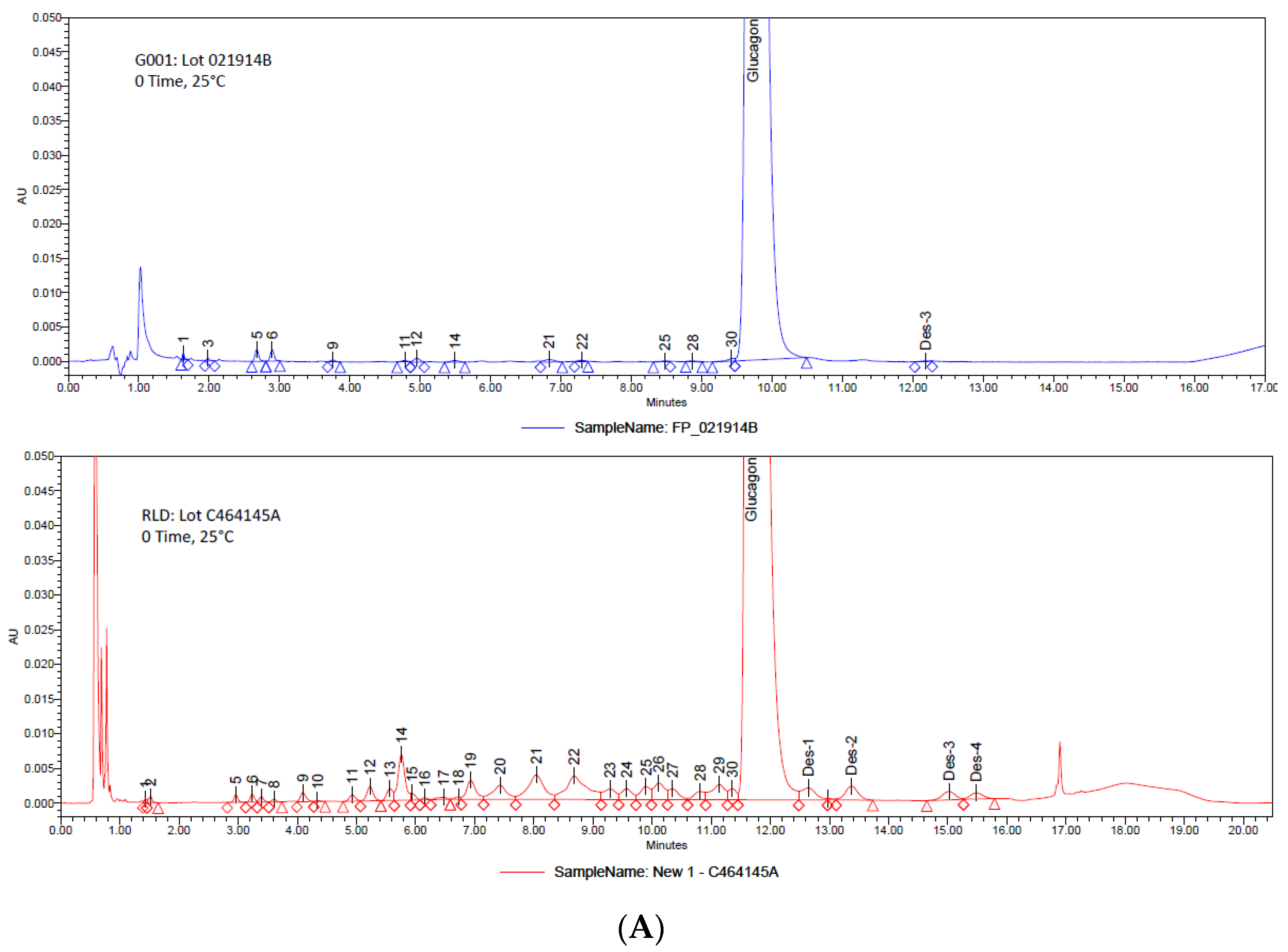
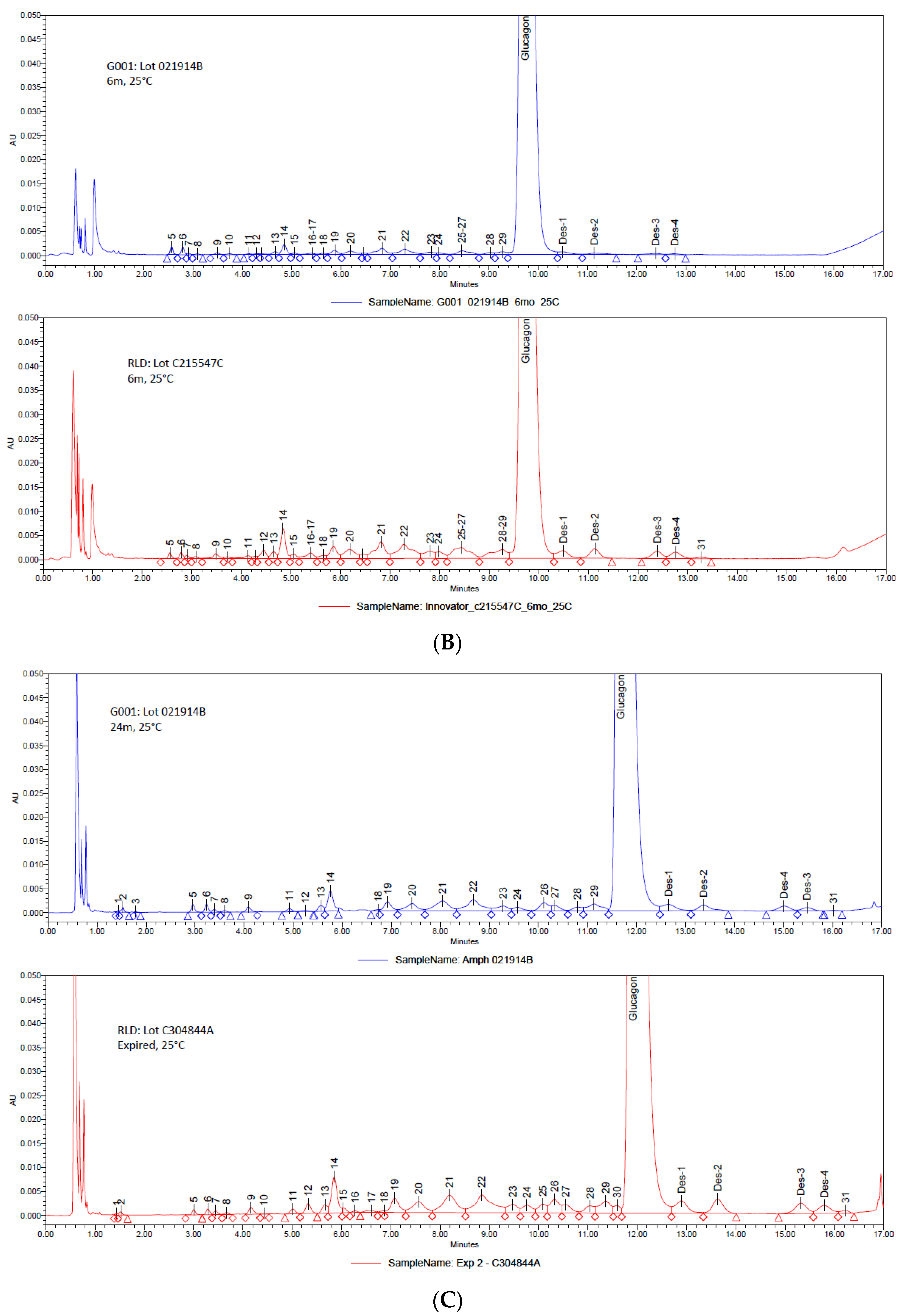
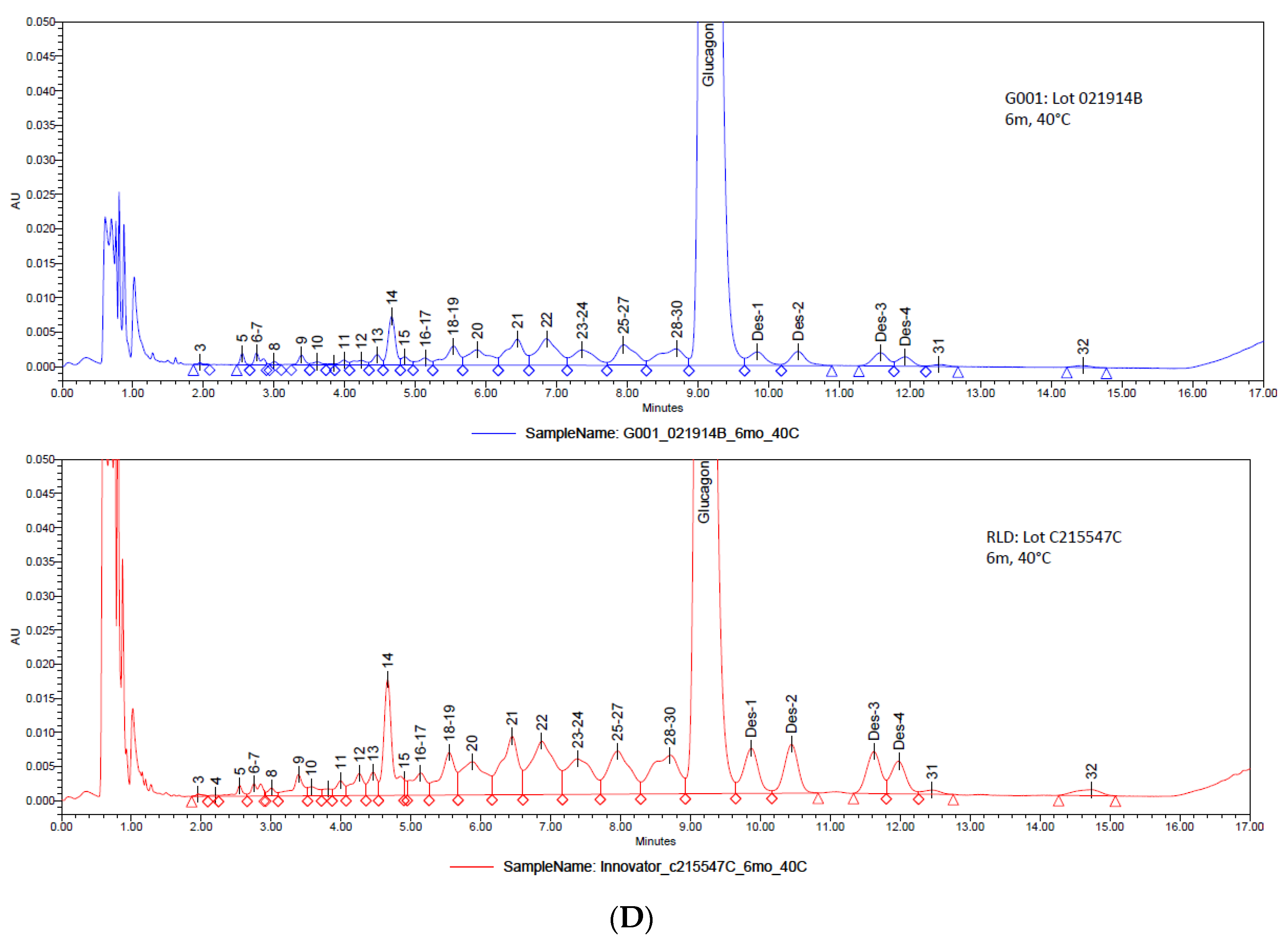
3. Results and Discussion
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Hall-Boyer, K.; Zaloga, G.P.; Chernow, B. Glucagon: Hormone or therapeutic agent? Crit. Care Med. 1984, 12, 584–589. [Google Scholar] [CrossRef] [PubMed]
- Caputo, N.; Castle, J.R.; Bergstrom, C.P.; Carroll, J.M.; Bakhtiani, P.A.; Jackson, M.A.; Ward, W.K. Mechanisms of glucagon degradation at alkaline pH. Peptides 2013, 45, 40–47. [Google Scholar] [CrossRef] [PubMed]
- Wilson, L.M.; Castle, J.R. Stable Liquid Glucagon: Beyond Emergency Hypoglycemia Rescue. J. Diabetes Sci. Technol. 2018, 12, 847–853. [Google Scholar] [CrossRef] [PubMed]
- FDA. FDA Approves First Generic of Drug Used to Treat Severe Hypoglycemia; FDA: Silver Spring, MD, USA, 2020. Available online: https://www.fda.gov/news-events/press-announcements/fda-approves-first-generic-drug-used-treat-severe-hypoglycemia (accessed on 16 June 2021).
- Pang, E.; Chong, W.; Luke, M.C. Scientific and Regulatory Considerations for the Approval of the First Generic Glucagon. J. Endocr. Soc. 2021, 5 (Suppl. 1), A324–A325. [Google Scholar] [CrossRef]
- Lodish, H.; Berk, A.; Zipursky, S.L.; Matsudaira, P.; Baltimore, D.; Darnell, J. Molecular Cell Biology, 4th ed.; Section 7.1, DNA Cloning with Plasmid Vectors; W.H. Freeman: New York, NY, USA, 2000. Available online: https://www.ncbi.nlm.nih.gov/books/NBK21498/ (accessed on 27 October 2021).
- Mäde, V.; Els-Heindl, S.; Beck-Sickinger, A.G. Automated solid-phase peptide synthesis to obtain therapeutic peptides. Beilstein J. Org. Chem. 2014, 10, 1197–1212. [Google Scholar] [CrossRef] [PubMed]
- Manning, M.C.; Chou, D.K.; Murphy, B.M.; Payne, R.W.; Katayama, D.S. Stability of protein pharmaceuticals: An update. Pharm. Res. 2010, 27, 544–575. [Google Scholar] [CrossRef] [PubMed]
- Joshi, A.B.; Kirsch, L.E. The estimation of glutaminyl deamidation and aspartyl cleavage rates in glucagon. Int. J. Pharm. 2004, 273, 213–219. [Google Scholar] [CrossRef]
- Joshi, A.B.; Sawai, M.; Kearney, W.R.; Kirsch, L.E. Studies on the mechanism of aspartic acid cleavage and glutamine deamidation in the acidic degradation of glucagon. J. Pharm. Sci. 2005, 94, 1912–1927. [Google Scholar] [CrossRef] [PubMed]
- Jackson, M.A.; Caputo, N.; Castle, J.R.; David, L.L.; Roberts, C.T., Jr.; Ward, W.K. Stable liquid glucagon formulations for rescue treatment and bi-hormonal closed-loop pancreas. Curr. Diab. Rep. 2012, 12, 705–710. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Doyle, H.A.; Gee, R.J.; Mamula, M.J. Altered immunogenicity of isoaspartate containing proteins. Autoimmunity 2007, 40, 131–137. [Google Scholar] [CrossRef] [PubMed]
- Doyle, H.A.; Zhou, J.; Wolff, M.J.; Harvey, B.P.; Roman, R.M.; Gee, R.J.; Mamula, M.J. Isoaspartyl post-translational modification triggers anti-tumor T and B lymphocyte immunity. J. Biol. Chem. 2006, 281, 32676–32683. [Google Scholar] [CrossRef] [PubMed]
- Fernandez, L.; Bustos, R.H.; Zapata, C.; Garcia, J.; Jauregui, E.; Ashraf, G.M. Immunogenicity in Protein and Peptide Based-Therapeutics: An Overview. Curr. Protein Pept. Sci. 2018, 19, 958–971. [Google Scholar] [CrossRef] [PubMed]
- Hermeling, S.; Crommelin, D.J.; Schellekens, H.; Jiskoot, W. Structure-immunogenicity relationships of therapeutic proteins. Pharm. Res. 2004, 21, 897–903. [Google Scholar] [CrossRef] [PubMed]
- ICH Harmonized Triplicate Guidelines. Validation of analytical procedures: Text and methodology Q2(R1). In International Conference on Harmonization of Technical Requirements for Registration of Pharmaceuticals for Human Use; ICH: Geneva, Switzerland, 2005.
- USP. Glucagon for Injection. In USP 43-NF 38; United States Pharmacopeia: Rockville, MD, USA, 2020. [Google Scholar]
- FDA. FDA Guidance for Industry: ANDAs for Certain Highly Purified Synthetic Peptide Drug Products That Refer to Listed Drugs of rDNA Origin; FDA: Silver Spring, MD, USA, 2021. Available online: https://www.fda.gov/media/107622/download (accessed on 20 August 2021).
- Li, M.; Josephs, R.D.; Daireaux, A.; Choteau, T.; Westwood, S.; Martos, G.; Wielgosz, R.I.; Li, H. Structurally related peptide impurity identification and accurate quantification for synthetic oxytocin by liquid chromatography-high-resolution mass spectrometry. Anal. Bioanal. Chem. 2021, 413, 1861–1870. [Google Scholar] [CrossRef] [PubMed]
| Storage Condition | AMP-Glucagon | ELI-Glucagon | |||
|---|---|---|---|---|---|
| Lot # | Age at Testing | Lot # | Age at Testing | ||
| Initial | - | 021914 021914A 021914B | 0 | C464145A * | 10 |
| 6 Months | 25 °C 40 °C | 021914 021914A 021914B | 6 | C215547C ** | 13 |
| 24 Months | 25 °C † | 021914 021914A 021914B | 24 | C304844A *** C304844C *** | 24 |
| Item | Description |
|---|---|
| Instrumentation | Waters Acquity UPLC system equipped with PDA detector |
| Column | Waters Acquity UPLC BEH300 C18, 1.7 µm, 2.1 mm × 100 mm, PN 186003686 |
| Mobile Phase Composition | A: 65%; B: 35% |
| Flow Rate | 0.4 mL/min |
| Column Temperature | 45 °C |
| Injection volume | 4 µL |
| Run time | 30 min |
| Detector Wavelength | 214 nm |
| Sample Temperature | 6 °C |
| Weak Wash (0.1% H3PO4 in 10% ACN) | 1200 µL |
| Strong Wash (MPB) | 400 µL |
| Peak | Peak Name | Initial Time, 25 °C | 6 Months, 25 °C | 24 Months, 25 °C | 6 Months, 40 °C | |||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| AMP-Glucagon Lots | RLD * | EEC Upper Limit † | AMP-Glucagon Lots | RLD * | EEC Upper Limit † | AMP-Glucagon Lots | RLD* | EEC Upper Limit † | AMP-Glucagon Lots | RLD * | EEC Upper Limit † | |||||||||||
| 021914 | 021914A | 021914B | C464145A | 021914 | 021914A | 021914B | C215547C | 021914 | 021914A | 021914B | C304844A | C304844C | 021914 | 021914A | 021914B | C215547C | ||||||
| 1 | Imp-1 | <D.L. | <D.L. | <D.L. | <D.L. | 0.15 | - | - | - | - | 0.15 | <D.L. | - | <D.L. | <D.L. | <D.L. | 0.15 | - | - | - | - | 0.15 |
| 2 | Imp-2 | <D.L. | <D.L. | - | 0.05 | 0.20 | - | - | - | - | 0.20 | 0.07 | 0.05 | 0.06 | <D.L. | <D.L. | 0.20 | - | - | - | - | 0.20 |
| 3 | Imp-3 | <D.L. | <D.L. | <D.L. | - | 0.15 | - | - | - | - | 0.15 | <D.L. | <D.L. | <D.L. | - | - | 0.15 | <D.L. | - | <D.L. | <D.L. | 0.15 |
| 4 | Imp-4 | - | - | - | - | 0.15 | - | - | - | - | 0.15 | - | - | - | - | <D.L. | 0.15 | - | - | - | <D.L. | 0.15 |
| 5 | Imp-5 | 0.14 | 0.13 | 0.12 | 0.07 | 0.22 | 0.15 | 0.14 | 0.16 | 0.12 | 0.27 | 0.14 | 0.12 | 0.11 | 0.07 | 0.08 | 0.27 | 0.15 | 0.15 | 0.12 | 0.17 | 0.32 |
| 6 | Imp-6 | 0.13 | 0.14 | 0.12 | 0.08 | 0.23 | 0.17 | 0.16 | 0.17 | 0.13 | 0.28 | 0.15 | 0.12 | 0.12 | 0.08 | 0.10 | 0.28 | 0.21 | 0.19 | 0.19 | 0.27 | 0.42 |
| 7 | Imp-7 | - | - | - | 0.06 | 0.21 | <D.L. | - | <D.L. | 0.07 | 0.22 | <D.L. | <D.L. | 0.05 | 0.06 | 0.08 | 0.23 | - | - | - | - | 0.23 |
| 8 | Imp-8 | - | - | - | <D.L. | 0.15 | - | - | <D.L. | 0.06 | 0.21 | <D.L. | - | <D.L. | <D.L. | <D.L. | 0.21 | <D.L. | <D.L. | <D.L. | 0.13 | 0.28 |
| 9 | Imp-9 | <D.L. | <D.L. | <D.L. | 0.12 | 0.27 | <D.L. | <D.L. | 0.06 | 0.20 | 0.35 | 0.09 | <D.L. | 0.11 | 0.14 | 0.16 | 0.35 | 0.09 | <D.L. | 0.13 | 0.52 | 0.67 |
| 10 | Imp-10 | - | - | - | <D.L. | 0.15 | <D.L. | - | <D.L. | 0.05 | 0.20 | - | - | - | <D.L. | <D.L. | 0.20 | 0.05 | <D.L. | 0.05 | 0.35 | 0.50 |
| 11 | Imp-11 | <D.L. | - | <D.L. | 0.09 | 0.24 | <D.L. | - | <D.L. | 0.14 | 0.29 | 0.06 | <D.L. | 0.07 | 0.10 | 0.12 | 0.29 | 0.05 | - | 0.09 | 0.30 | 0.45 |
| 12 | Imp-12 | 0.05 | - | 0.06 | 0.21 | 0.36 | 0.05 | <D.L. | 0.06 | 0.35 | 0.50 | <D.L. | - | <D.L. | 0.22 | 0.22 | 0.50 | 0.10 | <D.L. | 0.13 | 0.58 | 0.73 |
| 13 | Imp-13 | - | - | - | 0.17 | 0.32 | 0.08 | <D.L. | 0.10 | 0.22 | 0.37 | 0.11 | <D.L. | 0.14 | 0.17 | 0.20 | 0.37 | 0.15 | 0.06 | 0.19 | 0.45 | 0.60 |
| 14 | Imp-14 | 0.06 | <D.L. | <D.L. | 0.71 | 0.89 | 0.24 | 0.08 | 0.33 | 0.90 | 1.13 | 0.36 | 0.11 | 0.50 | 0.86 | 0.98 | 1.23 | 0.57 | 0.22 | 0.88 | 2.28 | 2.85 |
| 15 | Imp-15 | - | - | - | 0.07 | 0.22 | <D.L. | - | 0.05 | 0.15 | 0.30 | <D.L. | - | - | 0.08 | 0.10 | 0.30 | 0.10 | <D.L. | 0.16 | 0.10 | 0.30 |
| 16 | Imp-16 | - | - | - | <D.L. | 0.15 | 0.06 | 0.05 | 0.07 | 0.29 | 0.44 | <D.L. | - | - | <D.L. | <D.L. | 0.44 | 0.16 | 0.06 | 0.19 | 0.73 | 0.91 |
| 17 | Imp-17 | - | - | - | 0.05 | 0.20 | - | - | - | - | 0.20 | 0.08 | 0.05 | - | 0.07 | 0.07 | 0.22 | - | - | - | - | 0.22 |
| 18 | Imp-18 | - | - | - | <D.L. | 0.15 | <D.L. | <D.L. | 0.06 | 0.12 | 0.27 | <D.L. | - | <D.L. | <D.L. | <D.L. | 0.27 | 0.44 | <D.L. | 0.63 | 1.41 | 1.76 |
| 19 | Imp-19 | <D.L. | - | - | 0.39 | 0.54 | 0.11 | <D.L. | 0.19 | 0.53 | 0.68 | 0.22 | 0.07 | 0.31 | 0.48 | 0.50 | 0.68 | - | 0.12 | - | - | 0.68 |
| 20 | Imp-20 | - | - | - | 0.46 | 0.61 | 0.15 | <D.L. | 0.29 | 0.78 | 0.98 | 0.27 | 0.08 | 0.39 | 0.55 | 0.61 | 0.98 | 0.55 | 0.20 | 0.81 | 1.77 | 2.21 |
| 21 | Imp-21 | 0.05 | <D.L. | 0.07 | 0.90 | 1.13 | 0.25 | 0.05 | 0.45 | 1.07 | 1.34 | 0.43 | 0.11 | 0.65 | 1.02 | 1.17 | 1.46 | 0.76 | 0.28 | 1.07 | 2.27 | 2.84 |
| 22 | Imp-22 | - | - | <D.L. | 1.12 | 1.40 | 0.24 | 0.06 | 0.46 | 1.34 | 1.68 | 0.48 | 0.08 | 0.74 | 1.28 | 1.47 | 1.84 | 0.92 | 0.29 | 1.35 | 2.91 | 3.64 |
| 23 | Imp-23 | - | - | - | 0.29 | 0.44 | <D.L. | <D.L. | 0.16 | 0.48 | 0.63 | 0.15 | - | 0.27 | 0.35 | 0.43 | 0.63 | 0.54 | 0.13 | 0.82 | 2.07 | 2.59 |
| 24 | Imp-24 | - | - | - | 0.26 | 0.41 | - | - | 0.08 | 0.32 | 0.47 | 0.09 | - | 0.16 | 0.30 | 0.36 | 0.51 | - | - | - | - | 0.51 |
| 25 | Imp-25 | - | - | - | 0.29 | 0.44 | 0.21 | 0.10 | 0.31 | 1.14 | 1.43 | - | - | - | 0.28 | 0.28 | 1.43 | 0.73 | 0.28 | 0.98 | 2.23 | 2.79 |
| 26 | Imp-26 | - | - | <D.L. | 0.41 | 0.56 | - | - | - | - | 0.56 | 0.21 | 0.07 | 0.34 | 0.52 | 0.63 | 0.79 | - | - | - | - | 0.79 |
| 27 | Imp-27 | - | - | - | 0.25 | 0.40 | - | - | - | - | 0.40 | 0.13 | 0.08 | 0.21 | 0.30 | 0.34 | 0.49 | - | - | - | - | 0.49 |
| 28 | Imp-28 | 0.05 | <D.L. | <D.L. | 0.20 | 0.35 | - | - | 0.12 | 0.88 | 1.10 | 0.12 | 0.06 | 0.16 | 0.27 | 0.30 | 1.10 | 0.27 | <D.L. | 1.03 | 2.44 | 3.05 |
| 29 | Imp-29 | - | - | - | 0.46 | 0.61 | 0.11 | 0.07 | 0.17 | - | 0.61 | 0.21 | - | 0.44 | 0.56 | 0.69 | 0.86 | 0.42 | - | - | - | 0.86 |
| 30 | Imp-30 | 0.05 | 0.05 | 0.05 | 0.19 | 0.34 | 0.10 | - | - | - | 0.34 | 0.09 | 0.18 | - | 0.19 | 0.19 | 0.34 | - | - | - | - | 0.34 |
| 31 | Glucagon | 99.2 | 99.4 | 99.3 | 91.7 | 83.7 | 97.1 | 98.8 | 95.8 | 88.1 | 83.7 | 95.6 | 98.2 | 93.9 | 89.6 | 88.4 | 83.7 | 92.1 | 97.3 | 89.1 | 70.3 | 83.7 |
| 32 | Des-1 | - | - | - | 0.42 | 0.57 | 0.23 | 0.13 | 0.22 | 0.55 | 0.70 | 0.32 | 0.24 | 0.41 | 0.64 | 0.63 | 0.80 | 0.39 | - | 0.57 | 1.55 | 1.94 |
| 33 | Des-2 | 0.07 | <D.L. | - | 0.40 | 0.55 | 0.18 | 0.05 | 0.16 | 0.56 | 0.71 | 0.13 | 0.14 | 0.32 | 0.62 | 0.63 | 0.79 | 0.33 | 0.06 | 0.54 | 1.56 | 1.95 |
| 34 | Des-3 | - | - | <D.L. | 0.25 | 0.40 | 0.08 | <D.L. | 0.11 | 0.46 | 0.61 | 0.19 | 0.10 | 0.28 | 0.52 | 0.54 | 0.69 | 0.27 | 0.13 | 0.50 | 1.37 | 1.71 |
| 35 | Des-4 | - | - | - | 0.18 | 0.33 | 0.05 | <D.L. | 0.07 | 0.43 | 0.58 | 0.13 | 0.05 | 0.17 | 0.40 | 0.43 | 0.58 | 0.18 | 0.10 | 0.34 | 1.13 | 1.41 |
| 36 | Imp-31 | - | - | - | - | 0.15 | - | - | - | 0.08 | 0.23 | - | - | <D.L. | 0.09 | 0.09 | 0.24 | - | - | 0.06 | 0.16 | 0.31 |
| 37 | Imp-32 | - | - | - | - | 0.15 | - | - | - | - | 0.15 | - | - | - | - | - | 0.15 | - | - | 0.06 | 0.37 | 0.52 |
| NOI **, Observed | 8 | 3 | 5 | 27 | N/A | 17 | 10 | 22 | 26 | N/A | 23 | 17 | 22 | 27 | 27 | N/A | 22 | 14 | 23 | 24 | N/A | |
| NOI **, ≥0.5% | 0 | 0 | 0 | 3 | N/A | 0 | 0 | 0 | 9 | N/A | 0 | 0 | 3 | 9 | 10 | N/A | 6 | 0 | 11 | 15 | N/A | |
| All Imp. > 0.5% | 0.0 | 0.0 | 0.0 | 3.7 | 4.6 | 0.0 | 0.0 | 0.9 | 8.7 | 10.9 | 0.5 | 0.0 | 1.9 | 7.1 | 7.8 | 10.9 | 4.1 | 0.0 | 9.2 | 25.3 | 31.6 | |
| Highest single | 0.14 | 0.14 | 0.12 | 1.12 | 1.4 | 0.25 | 0.16 | 0.46 | 1.34 | 1.7 | 0.48 | 0.24 | 0.74 | 1.28 | 1.47 | 1.8 | 0.92 | 0.29 | 1.35 | 2.91 | 3.6 | |
| All Desamino | 0.07 | 0.00 | 0.00 | 1.25 | 14 | 0.54 | 0.18 | 0.56 | 2 | 14 | 0.77 | 0.53 | 1.18 | 2.18 | 2.23 | 14 | 1.17 | 0.29 | 1.95 | 5.61 | 14 | |
| All Other Imp. | 0.5 | 0.3 | 0.4 | 6.9 | 8.6 | 1.9 | 0.7 | 3.3 | 9.4 | 11.8 | 3.5 | 1.2 | 4.8 | 8.0 | 9.2 | 11.8 | 6.3 | 2.0 | 8.9 | 21.5 | 26.9 | |
| Total Impurities | 0.6 | 0.3 | 0.4 | 8.2 | 31 | 2.5 | 0.9 | 3.9 | 11.4 | 31 | 4.2 | 1.7 | 6.0 | 10.2 | 11.4 | 31 | 7.4 | 2.3 | 10.9 | 27.1 | 31 | |
| Purity (Glucagon) | 99.2 | 99.4 | 99.3 | 91.7 | 83.7 | 97.1 | 98.8 | 95.8 | 88.1 | 83.7 | 95.6 | 98.2 | 93.9 | 89.6 | 88.4 | 83.7 | 92.1 | 97.3 | 89.1 | 70.3 | 83.7 | |
| Type of Purity/Impurity Analysis | Products | Time and Temperature, (as Mean ± S.D.) * | |||
|---|---|---|---|---|---|
| Initial Time | 6 Months, 25 °C | 24 Months, 25 °C ** | 6 Months, 40 °C | ||
| Purity | AMP-glucagon | 99.3 ± 0.1 | 97.2 ± 1.5 | 95.9 ± 2.2 | 92.8 ± 4.1 |
| ELI-glucagon | 91.7 | 88.1 | 89.0 ± 0.8 | 70.3 | |
| Total Impurities | AMP-glucagon | 0.4 ± 0.1 | 2.4 ± 1.5 | 4.0 ± 2.2 | 6.9 ± 4.3 |
| ELI-glucagon | 8.2 | 11.4 | 10.8 ± 0.83 | 27.1 | |
| Impurities with Area > 0.5% | AMP-glucagon | 0.0 ± 0.0 | 0.3 ± 0.5 | 0.8 ± 1.0 | 4.4 ± 4.6 |
| ELI-glucagon | 3.7 | 8.7 | 7.5 ± 0.6 | 25.3 | |
| Highest single Impurity | AMP-glucagon | 0.13 ± 0.01 | 0.29 ± 0.15 | 0.49 ± 0.25 | 0.85 ± 0.53 |
| ELI-glucagon | 1.12 | 1.34 | 1.38 ± 0.13 | 2.91 | |
| All Desamido Impurities | AMP-glucagon | 0.02 ± 0.04 | 0.4 ± 0.2 | 0.8 ± 0.3 | 1.1 ± 0.8 |
| ELI-glucagon | 1.3 | 2.0 | 2.2 ± 0.04 | 5.6 | |
| All Other Impurities | AMP-glucagon | 0.4 ± 0.1 | 2.0 ± 1.29 | 3.2 ± 1.8 | 5.7 ± 3.5 |
| ELI-glucagon | 6.9 | 9.4 | 8.6 ± 0.8 | 21.5 | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bao, Z.; Cheng, Y.-C.; Luo, M.Z.; Zhang, J.Y. Comparison of the Purity and Impurity of Glucagon-for-Injection Products under Various Stability Conditions. Sci. Pharm. 2022, 90, 32. https://doi.org/10.3390/scipharm90020032
Bao Z, Cheng Y-C, Luo MZ, Zhang JY. Comparison of the Purity and Impurity of Glucagon-for-Injection Products under Various Stability Conditions. Scientia Pharmaceutica. 2022; 90(2):32. https://doi.org/10.3390/scipharm90020032
Chicago/Turabian StyleBao, Zhongli, Ya-Chi Cheng, Mary Ziping Luo, and Jack Yongfeng Zhang. 2022. "Comparison of the Purity and Impurity of Glucagon-for-Injection Products under Various Stability Conditions" Scientia Pharmaceutica 90, no. 2: 32. https://doi.org/10.3390/scipharm90020032
APA StyleBao, Z., Cheng, Y.-C., Luo, M. Z., & Zhang, J. Y. (2022). Comparison of the Purity and Impurity of Glucagon-for-Injection Products under Various Stability Conditions. Scientia Pharmaceutica, 90(2), 32. https://doi.org/10.3390/scipharm90020032






