Quality and In Vivo Assessment of a Fulvic Acid Complex: A Validation Study
Abstract
1. Introduction
2. Materials and Methods
2.1. Phase Solubility Study
2.2. Preparation of Complex
2.3. High-Performance Liquid Chromatography (HPLC) Analysis of CBZ
2.4. Determination of Aqueous Solubility of Solid Complex
2.5. Differential Scanning Calorimetry (DSC)
2.6. Fourier Transform Infrared Spectroscopy (FTIR)
2.7. X-ray Diffraction (XRD)
2.8. Drug Release Study
2.9. Animals
2.9.1. Ex Vivo Everted Intestinal Sac Permeation Study
2.9.2. Pharmacokinetic Study
- Group I received pure CBZ at a dose of 80 mg/kg p.o.
- Group II received CBZ-FA complex at a dose equivalent to 80 mg/kg p.o.
- Group III received normal saline, p.o.
2.9.3. Pharmacodynamic Study/MES (Maximal Electroshock)-Induced Convulsion
3. Results
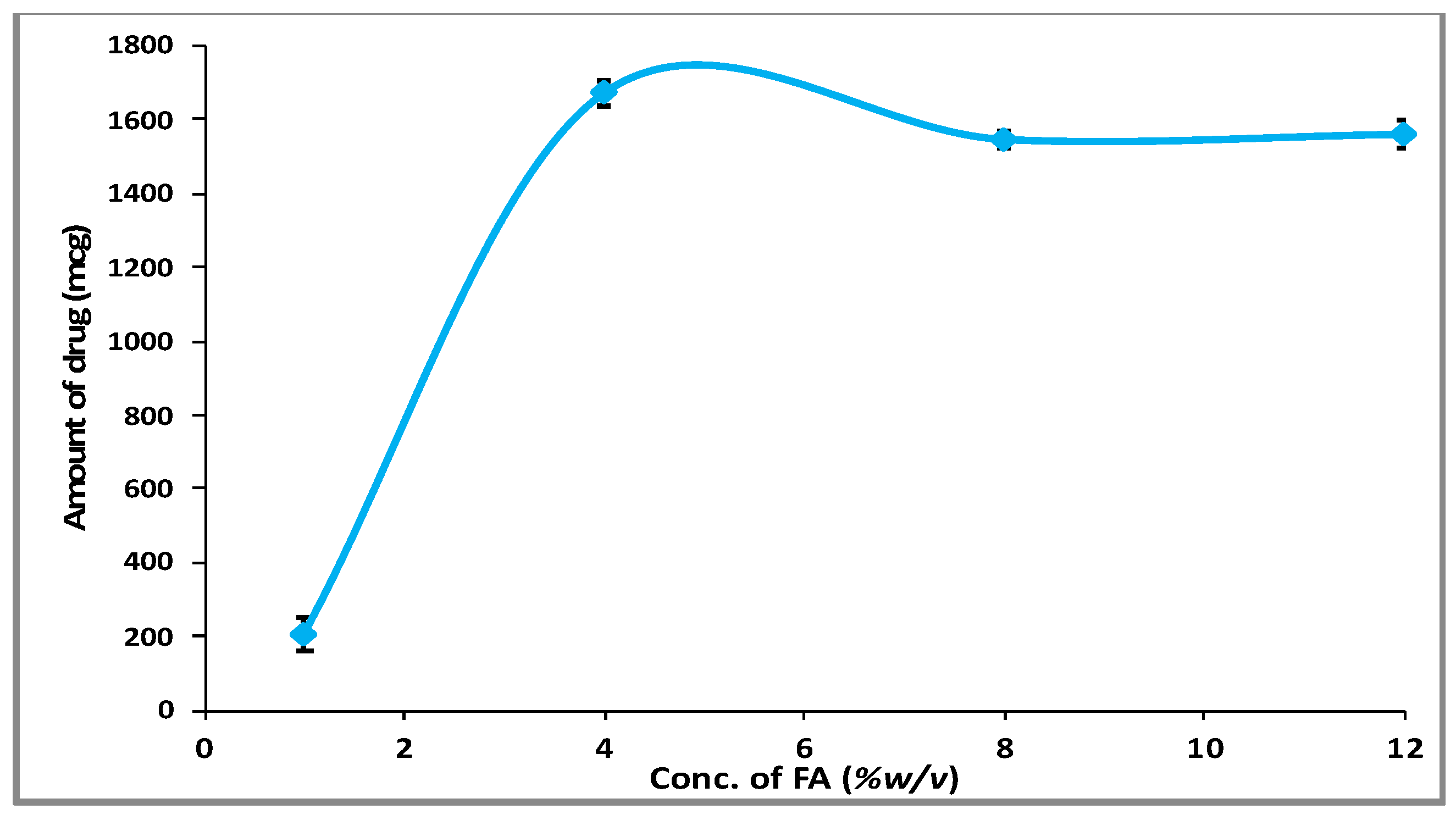
3.1. Phase Solubility Behavior
3.2. Characterization of the Complex
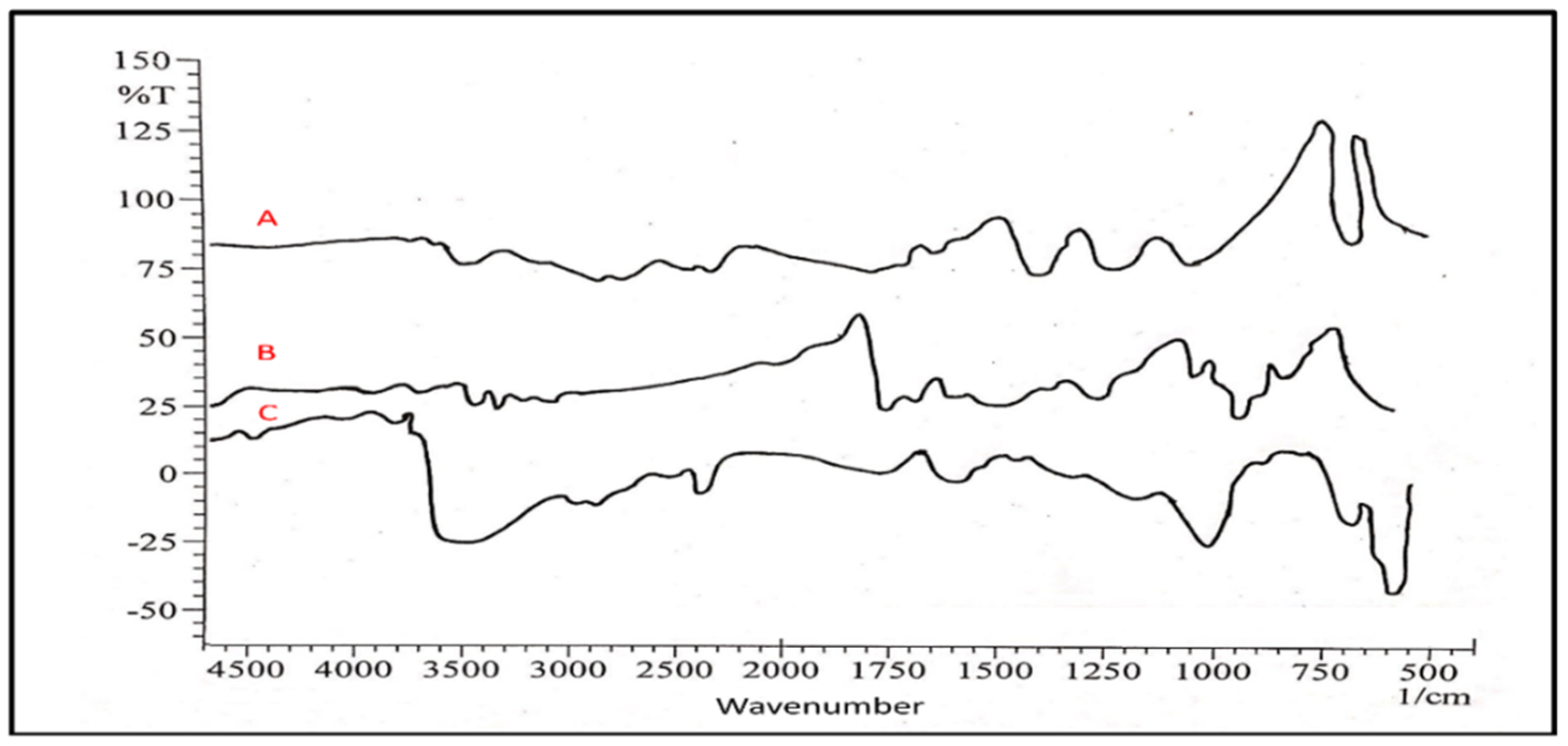
3.2.1. Fourier Transform Infrared Spectroscopy (FTIR)
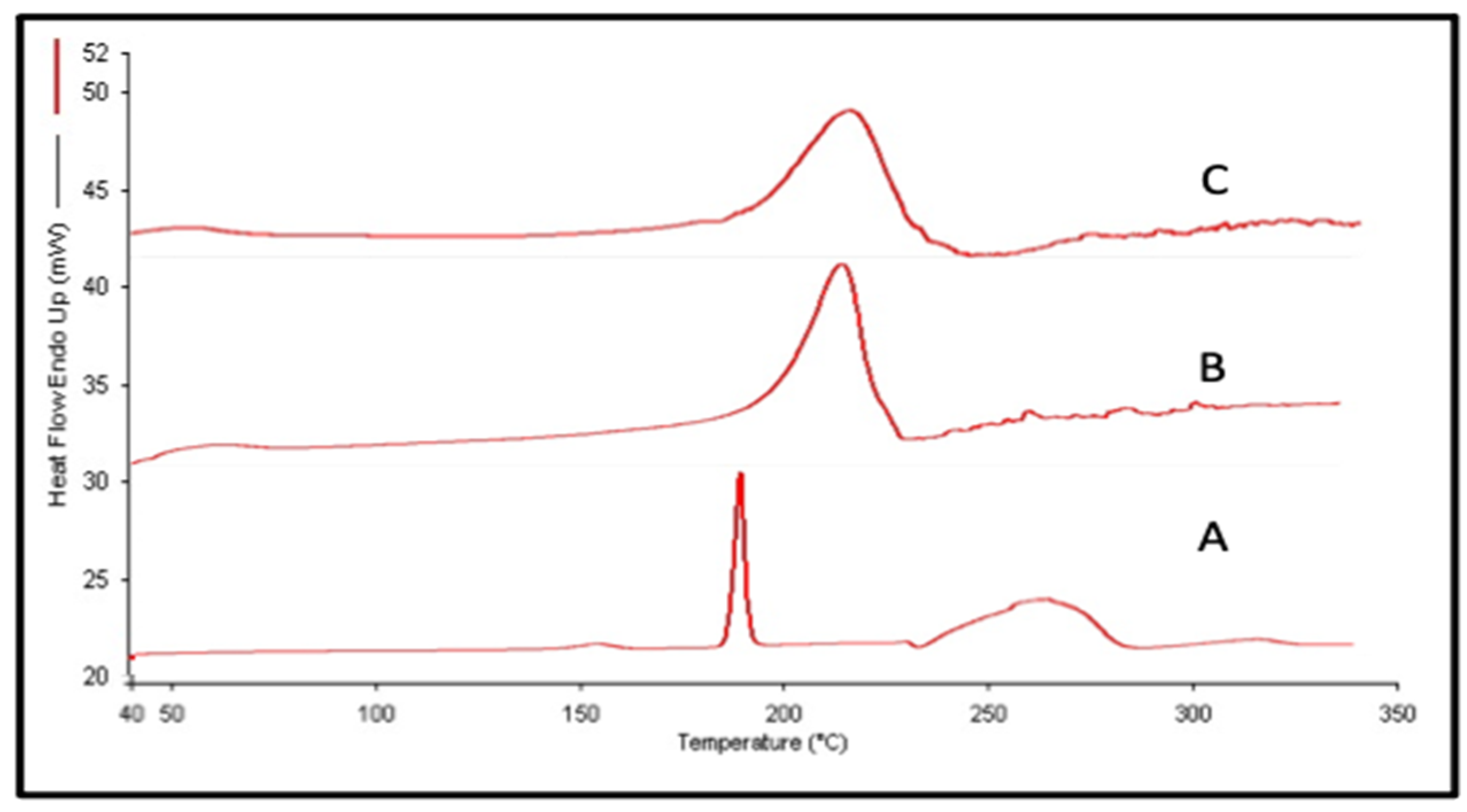
3.2.2. Differential Scanning Colorimetry (DSC)
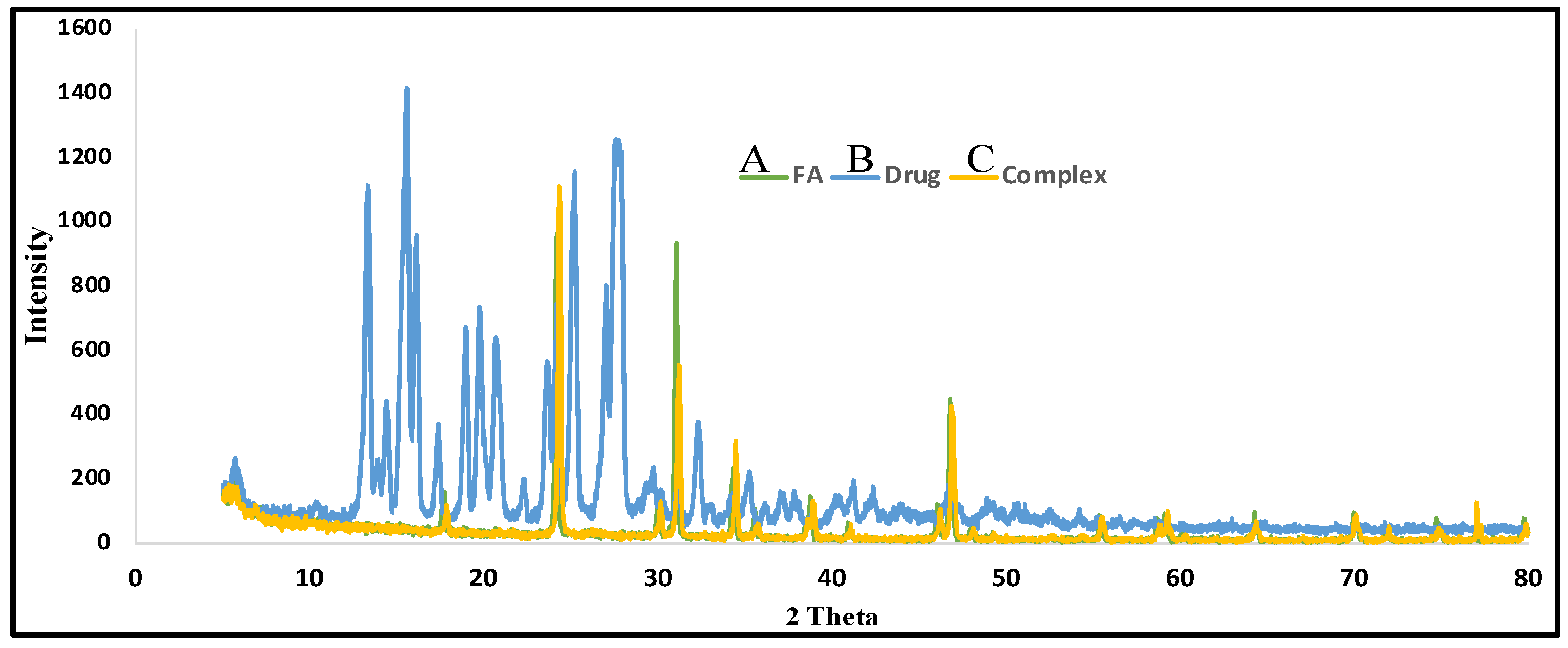
3.2.3. X-ray Diffraction (XRD)
3.2.4. Determination of Saturation Aqueous Solubility of Solid Complex
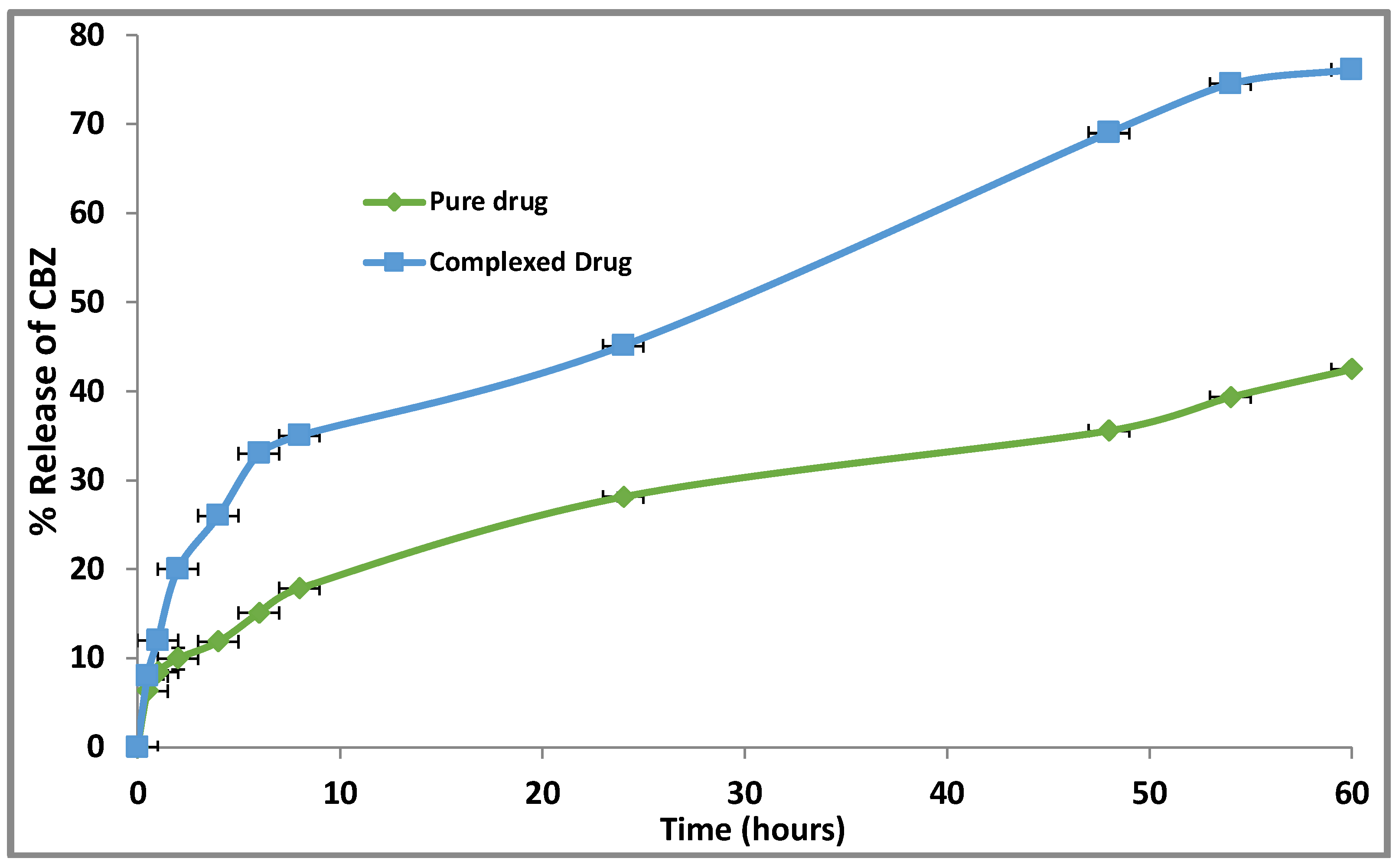
3.2.5. CBZ Release Profile from the Developed Complex
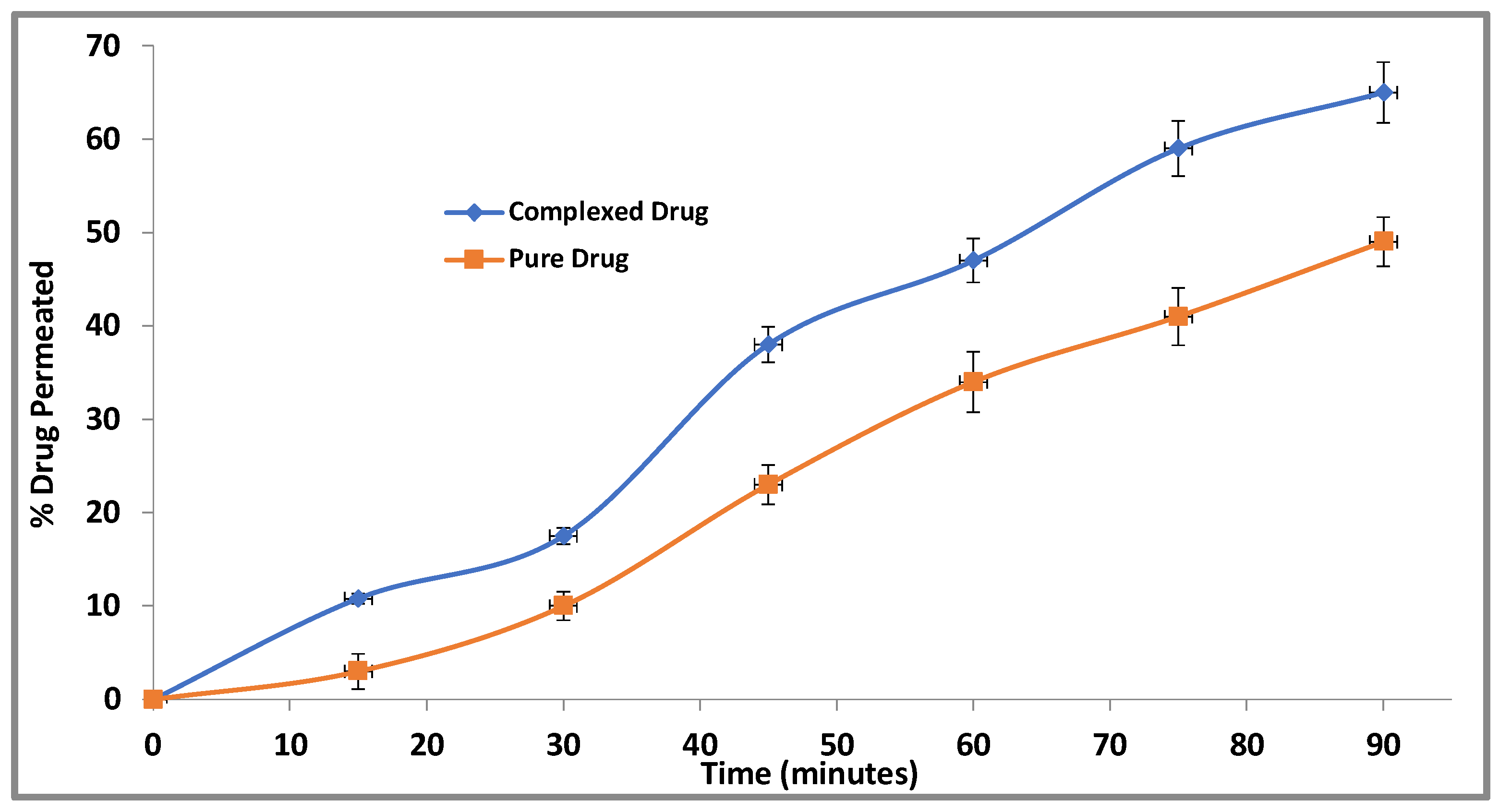
3.2.6. Ex Vivo Everted Intestinal Gut Sac Permeation Study for CBZ-FA Complex
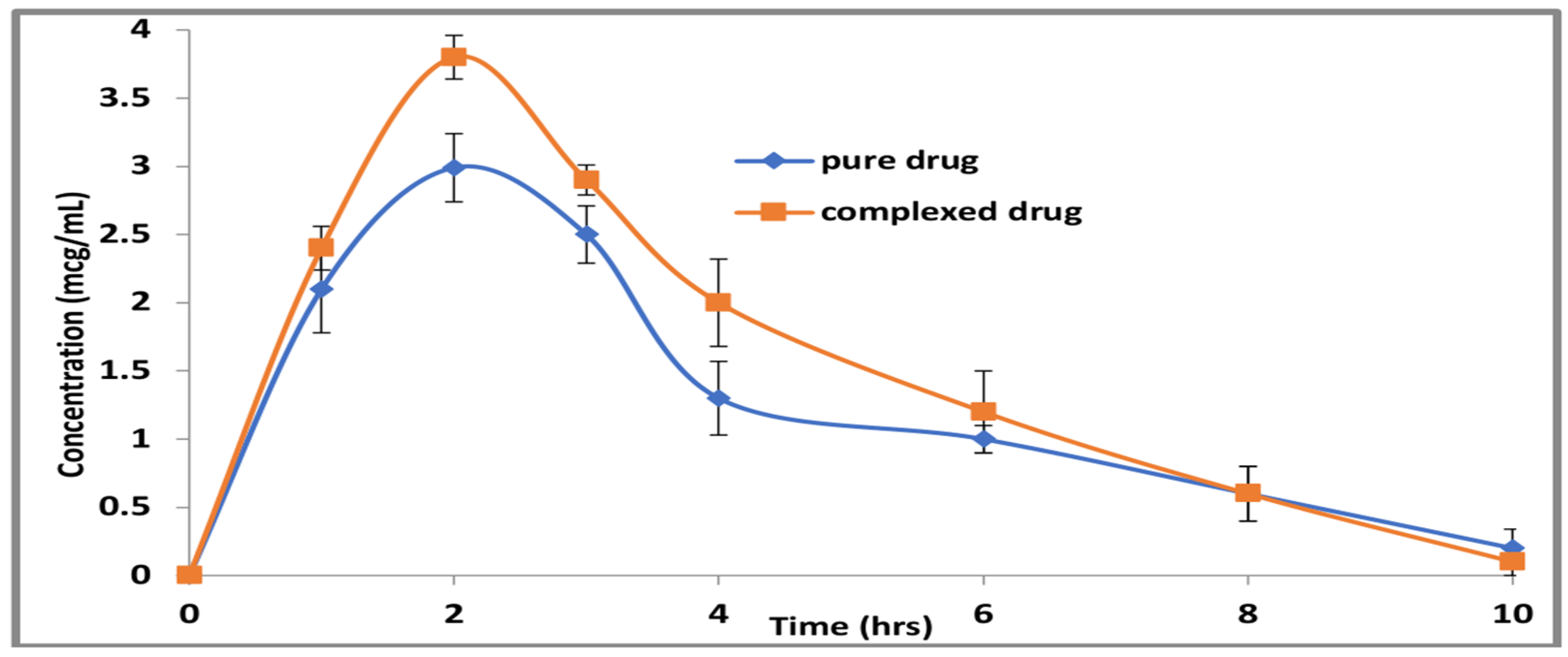
3.2.7. Pharmacokinetics of Carbamazepine
3.2.8. MES (Maximal Electroshock Seizure)-Induced Convulsion
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Steffens, K.E.; Wagner, K.G. Dissolution enhancement of carbamazepine using twin-screw melt granulation. Eur. J. Pharm. Biopharm. 2020, 148, 77–87. [Google Scholar] [CrossRef] [PubMed]
- Zerrouk, N.; Chemtob, C.; Arnaud, P.; Toscani, S.; Dugue, J. In vitro and in vivo evaluation of carbamazepine-PEG6000 solid dispersions. Int. J. Pharm. 2001, 225, 49–62. [Google Scholar] [CrossRef]
- Klučáková, M. Size and charge evaluation of standard humic and fulvic acids as crucial factors to determine their environmental behavior and impact. Front. Chem. 2018, 6, 235. [Google Scholar] [CrossRef]
- Jadhav, A.; Acharya, R.; Chandola, H.M.; Harisha, C.R. Development and Validation of Uv Spectrophotophotometric Method for Analysis of Fulvic Acid—A Decomposition Product in Shilajith. Int. J. Ayurveda Allied Sci. 2013, 2, 327–331. [Google Scholar]
- Mirza, M.A. Future of Humic substances as Pharmaceutical Excipient. Pharm. Sci. Anal. Res. J. Mini Rev. 2018, 2018, 180004. Available online: http://chembioepub.com/PSARJ/ (accessed on 13 June 2021).
- Raish, M.; Ahmad, A.; Alkharfy, K.M.; Jan, B.L.; Mohsin, K.; Ahad, A.; Al-Jenoobi, F.I.; Al-Mohizea, A.M. Effects of Paeonia emodi on hepatic cytochrome P450 (CYP3A2 and CYP2C11) expression and pharmacokinetics of carbamazepine in rats. Biomed. Pharmacother. 2017, 90, 694–698. [Google Scholar] [CrossRef]
- Emad, N.A.; Ahmed, B.; Alhalmi, A.; Alzobaidi, N.; Al-Kubati, S.S. Recent progress in nanocarriers for direct nose to brain drug delivery. J. Drug Deliv. Sci. Technol. 2021, 64, 102642. [Google Scholar] [CrossRef]
- Zhao, Y.; Paderu, P.; Delmas, G.; Dolgov, E.; Lee, M.H.; Senter, M.; Park, S.; Leivers, S.; Perlin, D.S. Carbohydrate-derived fulvic acid is a highly promising topical agent to enhance healing of wounds infected with drug-resistant pathogens. J. Trauma Acute Care Surg. 2015, 79, S121–S129. [Google Scholar] [CrossRef]
- Swift, R.S.; Posner, A.M. Gel Chromatography of Humic Acid. J. Soil Sci. 1971, 22, 237–249. [Google Scholar] [CrossRef]
- Sabi, R.; Very, P.; van Rensburg, C.E.J. Carbohydrate-derived fulvic acid (CHD-FA) inhibits carrageenan-induced inflammation and enhances wound healing: Efficacy and toxicity study in rats. Drug Dev. Res. 2012, 73, 18–23. [Google Scholar] [CrossRef]
- Hart, R.M.; Jones, H.L.; Jones, V.L.; Malik, S.; Kenny, M.A.; Love, B.; Harnisch, J.P. United States Patent E 2001 1. Available online: https://patents.google.com/patent/US6267962B1/en (accessed on 7 April 2022).
- Schnitzer, M.; Kodama, H. An electron microscopic examination of fulvic acid. Geoderma 1975, 13, 279–287. [Google Scholar] [CrossRef]
- Leppard, G.G.; Buffle, J.; Baudat, R. A description of the aggregation properties of aquatic pedogenic fulvic acids. Combining physico-chemical data and microscopical observations. Water Res. 1986, 20, 185–196. [Google Scholar] [CrossRef]
- Mirza, M.A.; Agarwal, S.P.; Rahman, M.A.; Rauf, A.; Ahmad, N.; Alam, A.; Iqbal, Z. Role of humic acid on oral drug delivery of an antiepileptic drug. Drug Dev. Ind. Pharm. 2011, 37, 310–319. [Google Scholar] [CrossRef] [PubMed]
- Gandy, J.J.; Snyman, J.R.; van Rensburg, C.E. Randomized, parallel-group, double-blind, controlled study to evaluate the efficacy and safety of carbohydrate-derived fulvic acid in topical treatment of eczema. Clin. Cosmet. Investig. Dermatol. 2011, 4, 145–148. [Google Scholar] [CrossRef]
- Khan, R.; Jain, P.; Aqil, M.; Agarwal, S.P.; Mirza, M.A.; Iqbal, Z. Pharmacokinetic evaluation of fulvic acid-ketoconazole complexes: A validation and line extension study. J. Drug Deliv. Sci. Technol. 2020, 55, 101469. [Google Scholar] [CrossRef]
- Agarwal, S.P.; Ansari, S.H.; Karmarkar, R.R. Enhancement of the dissolution rate of ketoconazole through a novel complexation with humic acid extracted from shilajit. Asian J. Chem. 2008, 20, 380–388. [Google Scholar]
- Liu, H.; Nie, J.; Chan, H.C.S.; Zhang, H.; Li, L.; Lin, H.; Tong, H.H.Y.; Ma, A.; Zhou, Z. Phase solubility diagrams and energy surface calculations support the solubility enhancement with low hygroscopicity of Bergenin: 4-Aminobenzamide (1:1) cocrystal. Int. J. Pharm. 2021, 601, 120537. [Google Scholar] [CrossRef]
- Prashanth, S.; Kumar, A.; Madhu, B.; Rama, N.; Sagar, J.V. Pharmacokinetic and pharmacodynamic drug interactions of carbamazepine and glibenclamide in healthy albino Wistar rats. J. Pharmacol. Pharmacother. 2011, 2, 7–10. [Google Scholar] [CrossRef]
- Kouhail, Y.Z.; Benedetti, M.F.; Reiller, P.E. Formation of mixed Eu(III)-CO3-fulvic acid complex: Spectroscopic evidence and NICA-Donnan modeling. Chem. Geol. 2019, 522, 175–185. [Google Scholar] [CrossRef]
- Mowafy, H.A.; Alanazi, F.K.; el Maghraby, G.M. Development and validation of an HPLC-UV method for the quantification of carbamazepine in rabbit plasma. Saudi Pharm. J. 2012, 20, 29–34. [Google Scholar] [CrossRef]
- Gan, S.H.; Ismail, R.; Adnan, W.A.W.; Wan, Z. Method development and validation of a high-performance liquid chromatographic method for tramadol in human plasma using liquid-liquid extraction. J. Chromatogr. B Anal. Technol. Biomed. Life Sci. 2002, 772, 123–129. [Google Scholar] [CrossRef]
- Mirza, M.A.; Agarwal, S.P.; Iqbal, Z. Effect of fulvic acid on oral delivery of carbamazepine. Sci. Adv. Mater. 2011, 3, 223–232. [Google Scholar] [CrossRef]
- Verma, D.; Thakur, P.S.; Padhi, S.; Khuroo, T.; Talegaonkar, S.; Iqbal, Z. Design expert assisted nanoformulation design for co-delivery of topotecan and thymoquinone: Optimization, in vitro characterization and stability assessment. J. Mol. Liq. 2017, 242, 382–394. [Google Scholar] [CrossRef]
- Anzar, N.; Mirza, M.A.; Anwer, K.; Khuroo, T.; Alshetaili, A.S.; Alshahrani, S.M.; Meena, J.; Hasan, N.; Talegaonkar, S.; Panda, A.K.; et al. Preparation, evaluation and pharmacokinetic studies of spray dried PLGA polymeric submicron particles of simvastatin for the effective treatment of breast cancer. J. Mol. Liq. 2018, 249, 609–616. [Google Scholar] [CrossRef]
- Gnananath, K.; Nataraj, K.S.; Rao, B.G.; Kumar, K.P.; Mahnashi, M.H.; Anwer, M.K.; Umar, A.; Iqbal, Z.; Mirza, M.A. Exploration of fulvic acid as a functional excipient in line with the regulatory requirement. Environ. Res. 2020, 187, 109642. [Google Scholar] [CrossRef] [PubMed]
- Rahman, M.; Almalki, W.H.; Afzal, O.; Altamimi, A.S.A.; Kazmi, I.; Al-Abbasi, F.A.; Choudhry, H.; Alenezi, S.K.; Barkat, M.A.; Beg, S.; et al. Cationic solid lipid nanoparticles of resveratrol for hepatocellular carcinoma treatment: Systematic optimization, in vitro characterization and preclinical investigation. Int. J. Nanomed. 2020, 15, 9283–9299. [Google Scholar] [CrossRef]
- Fatima, S.; Iqbal, Z.; Panda, A.K.; Samim, M.; Talegaonkar, S.; Ahmad, F.J. Polymeric nanoparticles as a platform for permeability enhancement of class III drug amikacin. Colloids Surf. B Biointerfaces 2018, 169, 206–213. [Google Scholar] [CrossRef]
- Tambe, A.; Mokashi, P.; Pandita, N. Ex-vivo intestinal absorption study of boswellic acid, cyclodextrin complexes and poloxamer solid dispersions using everted gut sac technique. J. Pharm. Biomed. Anal. 2019, 167, 66–73. [Google Scholar] [CrossRef]
- Mirza, M.A.; Ahmad, N.; Agarwal, S.P.; Mahmood, D.; Anwer, M.K.; Iqbal, Z. Comparative evaluation of humic substances in oral drug delivery. Results Pharma Sci. 2011, 1, 16–26. [Google Scholar] [CrossRef]
- Wollina, U. Peat: A natural source for dermatocosmetics and dermatotherapeutics. J. Cutan. Aesthet. Surg. 2009, 2, 17–20. [Google Scholar] [CrossRef]
| Models | R2 Value |
|---|---|
| Zero order | 0.883 |
| First order | 0.874 |
| Korsmeyer–Peppas | 0.932 |
| Higuchi | 0.984 |
| Parameters | Group I (Pure Drug) | Group II (Complexed Drug) |
|---|---|---|
| Cmax (μg/mL) | 2.9935 ± 0.006 | 3.80 ± 0.1 |
| Tmax (h) | 2.0 ± 0.0 | 2 ± 0.0 |
| AUC0–t (μg/mL ∗ h) | 12.94 ± 0.06 | 15.8 ± 0.04 |
| AUC0–∞ (μg/mL ∗ h) | 13.66 ± 0.081 | 16.08 ± 0.05 |
| Parameter | Group I (Pure Drug) | Group II (Complexed Drug) | Group III (Control) |
|---|---|---|---|
| Duration of seizures (n = 6) | 49.4 ± 2 s | 25 ± 4 s | 2.48 ± 0.3 min |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Khan, R.; Jain, P.; Zakir, F.; Aqil, M.; Alshehri, S.; Mirza, M.A.; Iqbal, Z. Quality and In Vivo Assessment of a Fulvic Acid Complex: A Validation Study. Sci. Pharm. 2022, 90, 33. https://doi.org/10.3390/scipharm90020033
Khan R, Jain P, Zakir F, Aqil M, Alshehri S, Mirza MA, Iqbal Z. Quality and In Vivo Assessment of a Fulvic Acid Complex: A Validation Study. Scientia Pharmaceutica. 2022; 90(2):33. https://doi.org/10.3390/scipharm90020033
Chicago/Turabian StyleKhan, Rahmuddin, Pooja Jain, Foziyah Zakir, Mohd Aqil, Sameer Alshehri, Mohd Aamir Mirza, and Zeenat Iqbal. 2022. "Quality and In Vivo Assessment of a Fulvic Acid Complex: A Validation Study" Scientia Pharmaceutica 90, no. 2: 33. https://doi.org/10.3390/scipharm90020033
APA StyleKhan, R., Jain, P., Zakir, F., Aqil, M., Alshehri, S., Mirza, M. A., & Iqbal, Z. (2022). Quality and In Vivo Assessment of a Fulvic Acid Complex: A Validation Study. Scientia Pharmaceutica, 90(2), 33. https://doi.org/10.3390/scipharm90020033






