Antibacterial, Antibiofilm, and Antischistosomal Activity of Montrichardia linifera (Arruda) Schott (Araceae) Leaf Extracts
Abstract
1. Introduction
2. Materials and Methods
2.1. Plant Material and Preparation of Extract
2.2. High-Performance Liquid Chromatography (HPLC) Analysis
2.3. Antibacterial Assay
2.3.1. Bacterial Strains and Culture Conditions
2.3.2. Determination of Minimum Inhibitory Concentration (MIC)
2.3.3. Agar Diffusion Method (ADM)
2.3.4. Inhibition of S. Aureus Biofilm Adherence Assay
2.3.5. AFM Biofilm Analysis
2.4. In Vitro Antischistosomal Activity
2.4.1. Maintenance of the Schistosoma Mansoni Life Cycle
2.4.2. In Vitro Antischistosomal Assay
2.4.3. AFM Schistosoma Analysis
3. Results
3.1. HPLC Analysis
3.2. Antibacterial Assay
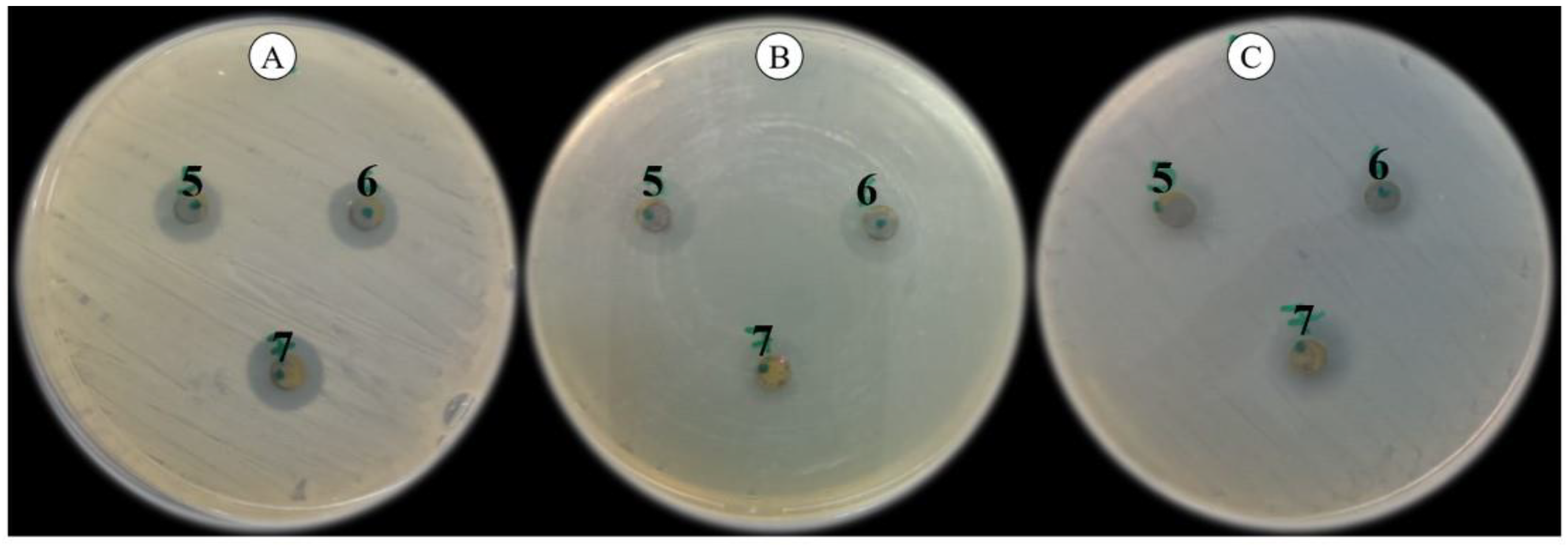
3.3. Agar Diffusion Method (ADM)
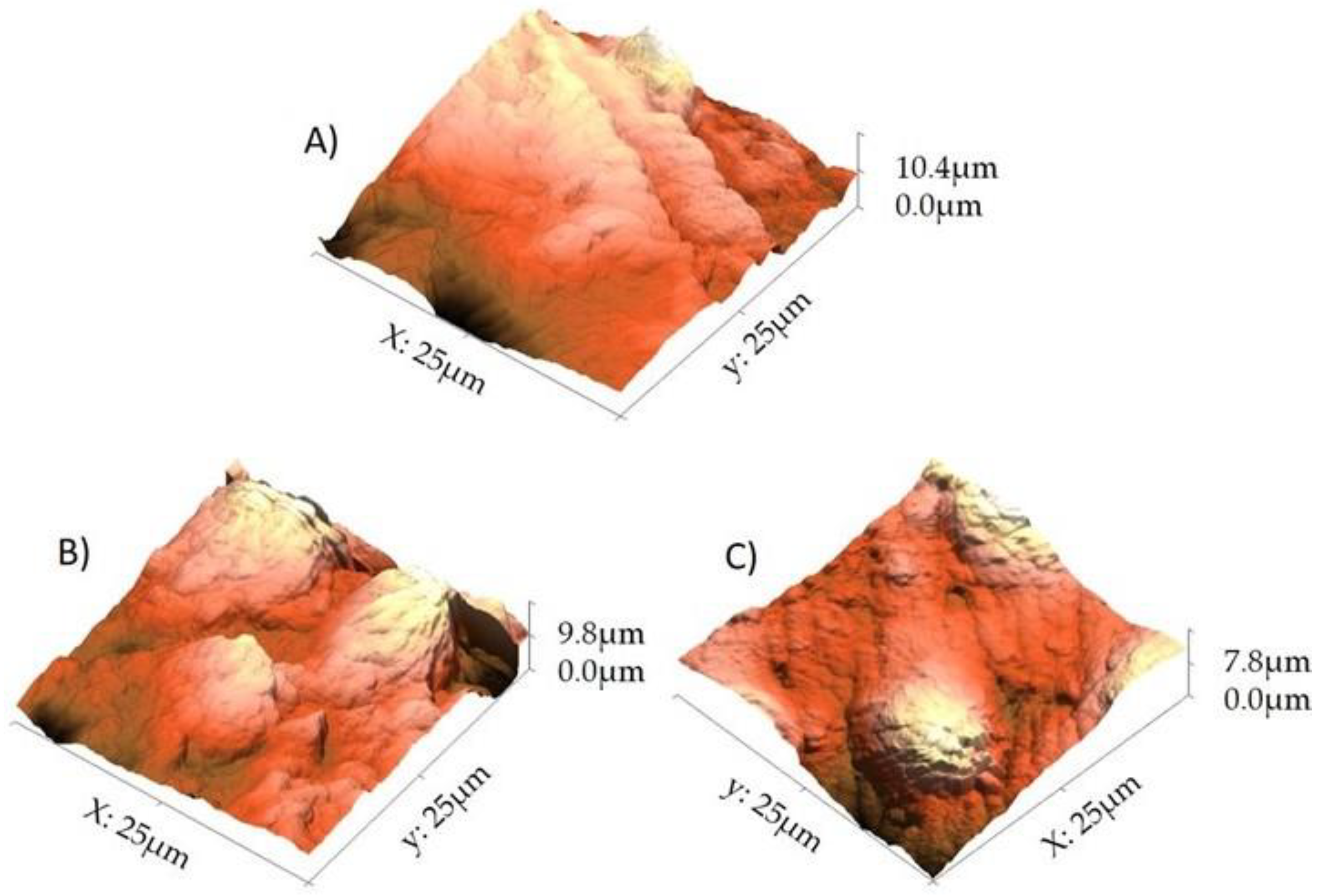
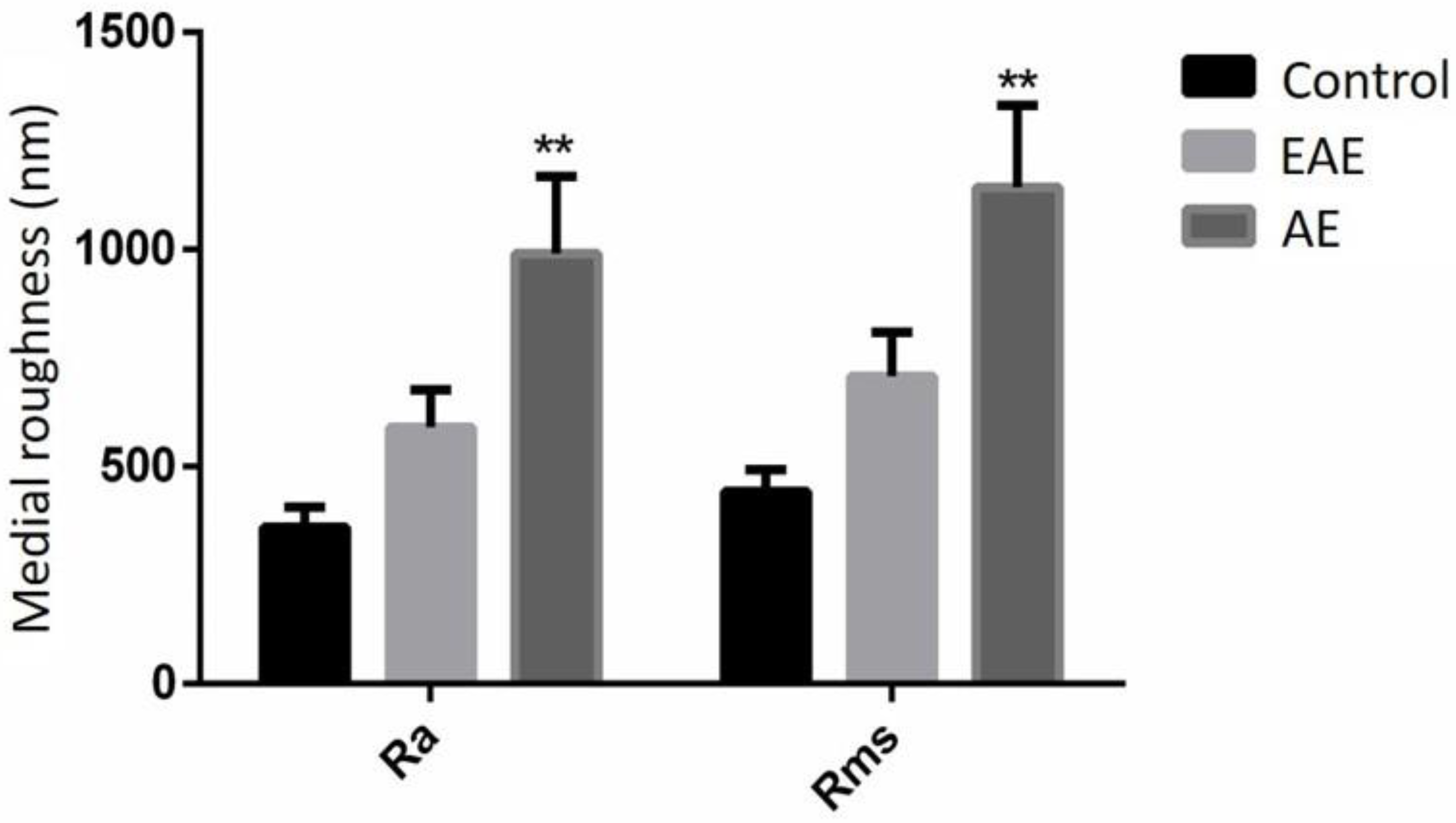
3.4. Antibiofilm Assay
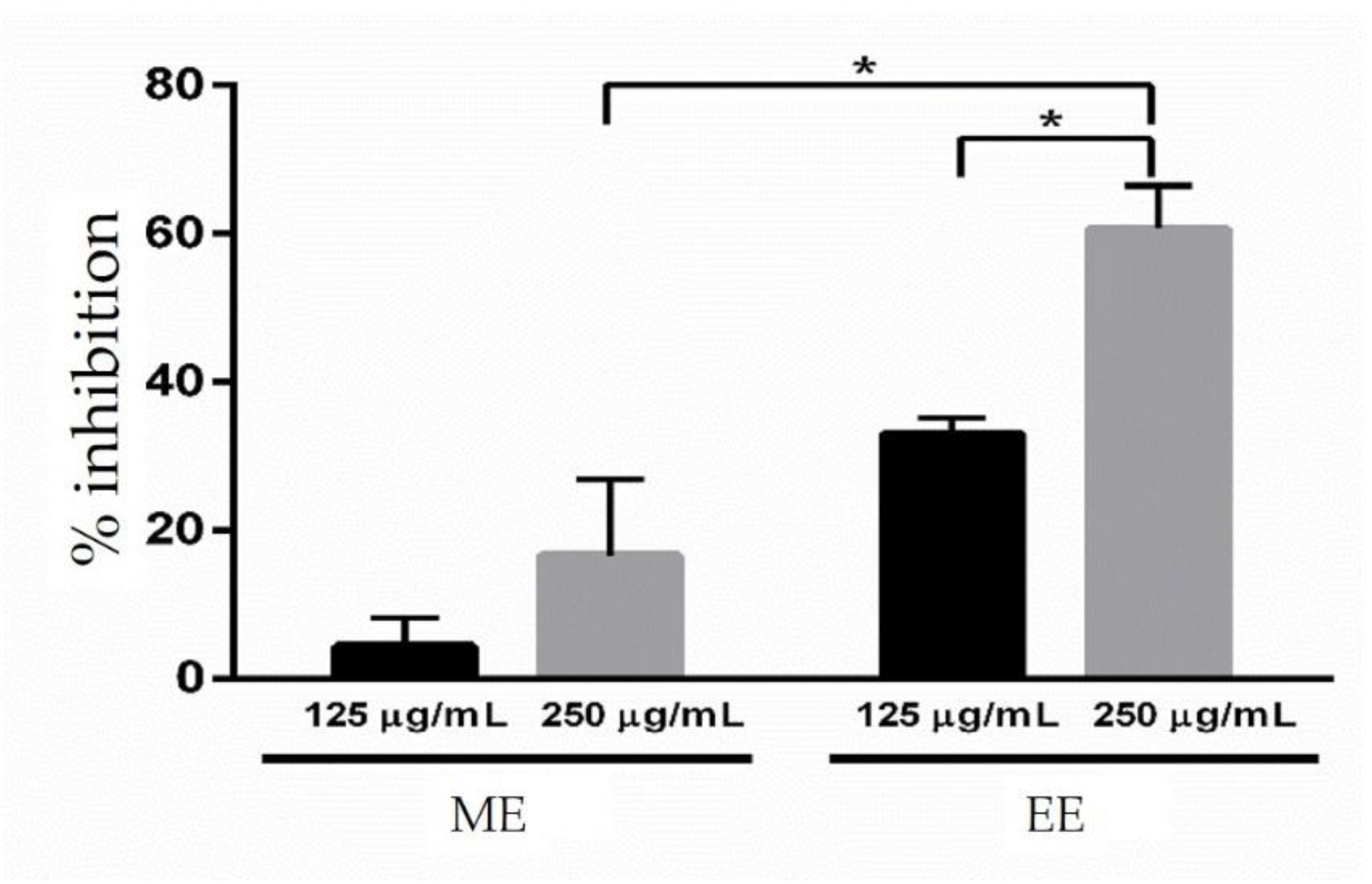
3.5. In Vitro Antischistosomal Assay
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Miranda, J.; Rocha, J.; Araújo, K.; Quelemes, P.; Mayo, S.; Andrade, I. Atividade antibacteriana de extratos de folhas de Montrichardia linifera (Arruda) Schott (Araceae). Rev. Bras. Plantas Med. 2015, 17, 1142–1149. [Google Scholar] [CrossRef]
- Silva, M.F.S.; Andrade, I.M.; Mayo, S.J. Geometric morphometrics of leaf blade shape in Montrichardia linifera (Araceae) pop-ulations from the Rio Parnaíba Delta, north-east Brazil. Bot. J. Linn. Soc. 2012, 170, 554–572. [Google Scholar] [CrossRef]
- Amarante, C.B.D.; Müller, R.C.S.; Dantas, K.D.G.F.; Alves, C.N.; Müller, A.H.; Palheta, D.D.C. Composição química e valor nutricional para grandes herbívoros das folhas e frutos de aninga (Montrichardia linifera, Araceae). Acta Amaz. 2010, 40, 729–736. [Google Scholar] [CrossRef]
- Ferreira, L.B.; Rodrigues, M.O.; Costa, J.M. Etnobotânica das Plantas Medicinais Cultivadas nos Quintais do Bairro de Algodoal em Abaetetuba/PA. Rev. Fitos 2016, 10, 220–372. [Google Scholar] [CrossRef][Green Version]
- Amarante, C.; Solano, F.; Lins, A.; Müller, A.; Müller, R. Caracterização física, química e nutricional dos frutos da Aninga. Planta Daninha 2011, 29, 295–303. [Google Scholar] [CrossRef]
- Gois, M.; Lucas, F.; Costa, J.; De Moura, P.; Lobato, G.D.J. Etnobotânica de espécies vegetais medicinais no tratamento de transtornos do sistema gastrointestinal. Rev. Bras. Plantas Med. 2016, 18, 547–557. [Google Scholar] [CrossRef]
- Costa, E.D.S.D.S.; Dolabela, M.F.; Povoa, M.M.; Oliveira, D.D.J.; Müller, A.H. Estudos farmacognósticos, fitoquímicos, atividade antiplasmódica e toxicidade em Artemia salina de extrato etanólico de folhas de Montrichardia linifera (Arruda) Schott, Araceae. Rev. Bras. Farm. 2009, 19, 834–838. [Google Scholar] [CrossRef]
- Hincapié, G.M.; Valange, S.; Barrault, J.; Moreno, J.A.; López, D.P. Effect of microwave-assisted system on transesterification of castor oil with ethanol. Univ. Sci. 2014, 19, 193–200. [Google Scholar] [CrossRef]
- McManus, D.P.; Dunne, D.W.; Sacko, M.; Utzinger, J.; Vennervald, B.J.; Zhou, X.-N. Schistosomiasis. Nat. Rev. Dis. Prim. 2018, 4, 13. [Google Scholar] [CrossRef] [PubMed]
- Mafud, A.C.; Silva, M.P.; Monteiro, D.C.; Oliveira, M.F.; Resende, J.G.; Coelho, M.L.; De Sousa, D.P.; Mendonça, R.Z.; Pinto, P.L.; Freitas, R.M.; et al. Structural parameters, molecular properties, and biological evaluation of some terpenes targeting Schistosoma mansoni parasite. Chem. Interact. 2016, 244, 129–139. [Google Scholar] [CrossRef] [PubMed]
- Lago, E.M.; Xavier, R.P.; Teixeira, T.R.; Silva, L.M.; Filho, A.A.D.S.; De Moraes, J. Antischistosomal agents: State of art and perspectives. Future Med. Chem. 2018, 10, 89–120. [Google Scholar] [CrossRef]
- Sohail, M.; Latif, Z. Molecular analysis, biofilm formation, and susceptibility of methicillin-resistant Staphylococcus aureus strains causing community- and health care-associated infections in central venous catheters. Rev. Soc. Bras. Med. Trop. 2018, 51, 603–609. [Google Scholar] [CrossRef]
- Rezaei, M.; Moniri, R.; Mousavi, S.G.A.; Shiade, M.J. Prevalence of Biofilm Formation Among Methicillin Resistance Staphylococcus aureus Isolated From Nasal Carriers. Jundishapur J. Microbiol. 2013, 6, 9601. [Google Scholar] [CrossRef]
- Piechota, M.; Kot, B.; Frankowska-Maciejewska, A.; Grużewska, A.; Woźniak-Kosek, A. Biofilm Formation by Methicillin-Resistant and Methicillin-Sensitive Staphylococcus aureus Strains from Hospitalized Patients in Poland. Biomed Res. Int. 2018, 2018, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Neopane, P.; Nepal, H.P.; Shrestha, R.; Uehara, O.; Abiko, Y. In vitro biofilm formation by Staphylococcus aureus isolated from wounds of hospital-admitted patients and their association with antimicrobial resistance. Int. J. Gen. Med. 2018, 11, 25–32. [Google Scholar] [CrossRef]
- Manandhar, S.; Singh, A.; Varma, A.; Pandey, S.; Shrivastava, N. Biofilm Producing Clinical Staphylococcus aureus Isolates Augmented Prevalence of Antibiotic Resistant Cases in Tertiary Care Hospitals of Nepal. Front. Microbiol. 2018, 9, 2749. [Google Scholar] [CrossRef] [PubMed]
- Mudzengi, C.P.; Murwira, A.; Tivapasi, M.; Murungweni, C.; Burumu, J.V.; Halimani, T. Antibacterial activity of aqueous and methanol extracts of selected species used in livestock health management. Pharm. Biol. 2017, 55, 1054–1060. [Google Scholar] [CrossRef]
- De Araújo, K.M.; De Lima, A.; Silva, J.D.N.; Rodrigues, L.L.; Amorim, A.G.N.; Quelemes, P.V.; Dos Santos, R.C.; Rocha, J.A.; De Andrades, É.O.; Leite, J.R.S.A.; et al. Identification of Phenolic Compounds and Evaluation of Antioxidant and Antimicrobial Properties of Euphorbia Tirucalli L. Antioxidants 2014, 3, 159–175. [Google Scholar] [CrossRef]
- Marinho, A.T.; Oliveira, M.G.; Menezes-Filho, A.C.P.; Castro, C.F.S.; Oliveira, I.M.M.; Borges, L.L.; Melo-Reis, P.R.; Silva, N.J., Jr. Phytochemical characterization, and antioxidant and antibacterial activities of the hydroethanolic extract of Anadenanthera peregrina stem bark. Braz. J. Biol. 2021, 82, e234476. [Google Scholar] [CrossRef]
- Matos, F.J.A. Introdução à Fitoquímica Experimental, 3rd ed.; UFC: Fortaleza, Ceará, Brazil, 2009; p. 141. [Google Scholar]
- Clinical and Laboratory Standards Institute. Methods for Dilution Antimicrobial Susceptibility Tests for Bacteria that Grow Aerobically; Approved Standard. CLSI Document M07-A9, 9th ed.; Clinical and Laboratory Standards Institute: Wayne, PA, USA, 2012. [Google Scholar]
- Sufian, A.S.; Ramasamy, K.; Ahmat, N.; Zakaria, Z.A.; Yusof, M.I.M. Isolation and identification of antibacterial and cytotoxic compounds from the leaves of Muntingia calabura L. J. Ethnopharmacol. 2013, 146, 198–204. [Google Scholar] [CrossRef]
- Niu, Y.; Wang, K.; Zheng, S.; Wang, Y.; Ren, Q.; Li, H.; Ding, L.; Li, W.; Zhang, L. Antibacterial Effect of Caffeic Acid Phenethyl Ester on Cariogenic Bacteria and Streptococcus mutans Biofilms. Antimicrob. Agents Chemother. 2020, 64, e00251-20. [Google Scholar] [CrossRef] [PubMed]
- Matilla-Cuenca, L.; Gil, C.; Cuesta, S.; Rapún-Araiz, B.; Žiemytė, M.; Mira, A.; Lasa, I.; Valle, J. Antibiofilm activity of flavonoids on staphylococcal biofilms through targeting BAP amyloids. Sci. Rep. 2020, 10, 1–12. [Google Scholar] [CrossRef]
- Hochbaum, A.I.; Kolodkin-Gal, I.; Foulston, L.; Kolter, R.; Aizenberg, J.; Losick, R. Inhibitory Effects of D-Amino Acids on Staphylococcus aureus Biofilm Development. J. Bacteriol. 2011, 193, 5616–5622. [Google Scholar] [CrossRef]
- Silva, M.P.; de Oliveira, R.N.; Mengarda, A.C.; Roquini, D.B.; Allegretti, S.M.; Salvadori, M.C.; Teixeira, F.S.; de Sousa, D.P.; Pinto, P.L.; Filho, A.A.D.S.; et al. Antiparasitic activity of nerolidol in a mouse model of schistosomiasis. Int. J. Antimicrob. Agents 2017, 50, 467–472. [Google Scholar] [CrossRef] [PubMed]
- De Lima, L.I.; Py-Daniel, K.R.; Guimarães, M.A.; Muehlmann, L.A.; Mafud, A.C.; Mascarenhas, Y.P.; De Moraes, J.; Leite, J.R.D.S.D.A.; Jiang, C.-S.; Azevedo, R.B.; et al. Self-nanoemulsifying drug-delivery systems improve oral absorption and antischistosomal activity of epiisopiloturine. Nanomedicine 2018, 13, 689–702. [Google Scholar] [CrossRef]
- De Castro, C.C.; Costa, P.S.; Laktin, G.T.; de Carvalho, P.H.; Geraldo, R.B.; de Moraes, J.; Pinto, P.L.; Couri, M.R.; Pinto, P.D.F.; Filho, A.A.D.S. Cardamonin, a schistosomicidal chalcone from Piper aduncum L. (Piperaceae) that inhibits Schistosoma mansoni ATP diphosphohydrolase. Phytomedicine 2015, 22, 921–928. [Google Scholar] [CrossRef] [PubMed]
- Mafud, A.C.; Silva, M.P.; Nunes, G.B.; de Oliveira, M.A.; Batista, L.F.; Rubio, T.I.; Mengarda, A.C.; Lago, E.M.; Xavier, R.P.; Gutierrez, S.J.; et al. Antiparasitic, structural, pharmacokinetic, and toxicological properties of riparin derivatives. Toxicol. Vitr. 2018, 50, 1–10. [Google Scholar] [CrossRef]
- Filho, A.C.P.D.M.; Castro, C.F.D.S. Identificação das classes de metabólitos secundários em extratos etanólicos foliares de Campomanesia adamantium, Dimorphandra mollis, Hymenaea stigonocarpa, Kielmeyera lathrophytum e Solanum lycocarpum. Estação Científica (Unifap) 2019, 9, 89–101. [Google Scholar] [CrossRef]
- Silva, J.B.; Costa, K.M.; Coelho, W.A.; Paiva, K.A.; Freitas, C.I.; Batista, J.S.; Costa, G.A.; Salatino, A. Quantificação de fenóis, flavonoides totais e atividades farmacológicas de geoprópolis de Plebeia aff. Flavocincta do Rio Grande do Norte. Pesqui. Veterinária Bras. 2016, 36, 874–880. [Google Scholar] [CrossRef]
- Lôbo, G.B.S.; Silva, A.V.; Menezes, G.B.L. Polifenóis dietéticos e função endotelial em adultos sem diagnóstico de doenças: Uma revisão sistemática de ensaios randomizados. Braz. J. Develop. 2020, 6, 85320–85346. [Google Scholar] [CrossRef]
- Simões, C.M.O.; Schenkel, E.P.; Mello, J.C.P.; Mentz, L.A.; Petrovick, P.R. Farmacognosia: Do Produto Natural ao Medicamento; Artmed: Porto Alegre, Brazil, 2017; p. 486. [Google Scholar]
- Canto, G.S.; Treter, J.; Yang, S.; Borré, G.L.; Peixoto, M.P.G.; Ortega, G.G. Avaliação das propriedades da espuma da saponina de Ilex paraguariensis. Bras. Ciênc. Farmac. 2010, 46, 237–243. [Google Scholar] [CrossRef][Green Version]
- Kubde, M.S.; Khadabadi, S.S.; Saboo, S.S.; Ghorpade, D.S.; Modi, A.J. In vitro antimicrobial activity of the crude extracts of Colocasia esculenta leaves (Araceae). Int. J. Pharm. Sci. Res. 2010, 1, 88–91. [Google Scholar] [CrossRef]
- Zeni, A.L.B.; Parisotto, A.V.; Mattos, G.; Helena, E.T.D.S. Utilização de plantas medicinais como remédio caseiro na Atenção Primária em Blumenau, Santa Catarina, Brasil. Ciência Saúde Coletiva 2017, 22, 2703–2712. [Google Scholar] [CrossRef] [PubMed]
- Da Silva, R.E.; Ribeiro, F.D.O.S.; De Carvalho, A.M.A.; Daboit, T.C.; Marinho-Filho, J.D.B.; Matos, T.S.; Pessoa, O.D.L.; Leite, J.R.D.S.D.A.; De Araújo, A.R.; Soares, M.J.D.S. Antimicrobial and antibiofilm activity of the benzoquinone oncocalyxone A. Microb. Pathog. 2020, 149, 104513. [Google Scholar] [CrossRef]
- Høiby, N.; Bjarnsholt, T.; Givskov, M.; Molin, S.; Ciofu, O. Antibiotic resistance of bacterial biofilms. Int. J. Antimicrob. Agents 2010, 35, 322–332. [Google Scholar] [CrossRef]
- Tang, Y.-W.; Stratton, C.W. Staphylococcus aureus: An Old Pathogen with New Weapons. Clin. Lab. Med. 2010, 30, 179–208. [Google Scholar] [CrossRef] [PubMed]
- Merino, N.; Toledo-Arana, A.; Vergara-Irigaray, M.; Valle, J.; Solano, C.; Calvo, E.; Lopez, J.A.; Foster, T.J.; Penadés, J.R.; Lasa, I. Protein A-Mediated Multicellular Behavior in Staphylococcus aureus. J. Bacteriol. 2008, 191, 832–843. [Google Scholar] [CrossRef] [PubMed]
- Haaber, J.; Cohn, M.T.; Frees, D.; Andersen, T.J.; Ingmer, H. Planktonic Aggregates of Staphylococcus aureus Protect against Common Antibiotics. PLoS ONE 2012, 7, e41075. [Google Scholar] [CrossRef]
- Peeters, G. The evaluative face of a descriptive model: Communion and agency in Peabody’s tetradic model of trait organization. Eur. J. Soc. Psychol. 2008, 38, 1066–1072. [Google Scholar] [CrossRef]
- Amorim, C.R.; Pavani, T.F.A.; Lopes, A.F.S.; Duque, M.D.; Mengarda, A.C.A.; Silva, M.P.; Rando, D.G.G. Bases de Schiff de 4-Fenil-2-Aminotiazóis como sucessos para novos anti-esquistossomóticos: Estudos de síntese, in vitro, in vivo e in silico. Eur. J. Pharm. Sci. 2020, 150, 105371. [Google Scholar] [CrossRef]
- Roquini, D.B.; Cogo, R.M.; Mengarda, A.C.; Mazloum, S.F.; Morais, C.S.; Xavier, R.P.; Moraes, J.A. Prometazina exibe pro-priedades antiparasitárias in vitro e reduz a carga de vermes, produção de ovos, hepato—e esplenomegalia em um modelo animal de esquistossomose. Antimicrob. Agents. Chemother. 2020, 63, 01208–01219. [Google Scholar] [CrossRef]
- Abdulla, M.-H.; Ruelas, D.S.; Wolff, B.; Snedecor, J.; Lim, K.-C.; Xu, F.; Renslo, A.R.; Williams, J.; McKerrow, J.H.; Caffrey, C.R. Drug Discovery for Schistosomiasis: Hit and Lead Compounds Identified in a Library of Known Drugs by Medium-Throughput Phenotypic Screening. Plos Negl. Trop. Dis. 2009, 3, e478. [Google Scholar] [CrossRef] [PubMed]
- Xiao, S.-H.; Mei, J.-Y.; Jiao, P.-Y. The in vitro effect of mefloquine and praziquantel against juvenile and adult Schistosoma japonicum. Parasitol. Res. 2009, 106, 237–246. [Google Scholar] [CrossRef]
- Manneck, T.; Haggenmüller, Y.; Keiser, J. Morphological effects and tegumental alterations induced by mefloquine on schistosomula and adult flukes ofSchistosoma mansoni. Parasitology 2009, 137, 85–98. [Google Scholar] [CrossRef] [PubMed]
- Braguine, C.G.; Costa, E.S.; Magalhães, L.G.; Rodrigues, V.; Filho, A.A.D.S.; Bastos, J.K.; Silva, M.L.A.; Cunha, W.R.; Januário, A.H.; Pauletti, P.M. Schistosomicidal Evaluation of Zanthoxylum naranjillo and its Isolated Compounds against Schistosoma mansoni Adult Worms. Z. Nat. C 2009, 64, 793–797. [Google Scholar] [CrossRef] [PubMed]
- Magalhães, L.G.; Machado, C.B.; Morais, E.R.; Moreira, É.B.D.C.; Soares, C.S.; Da Silva, S.H.; Filho, A.A.D.S.; Rodrigues, V. In vitro schistosomicidal activity of curcumin against Schistosoma mansoni adult worms. Parasitol. Res. 2008, 104, 1197–1201. [Google Scholar] [CrossRef]
- Parreira, N.A.; Magalhães, L.G.; Morais, D.R.; Caixeta, S.C.; De Sousa, J.P.B.; Bastos, J.K.; Cunha, W.R.; Silva, M.L.A.; Nanayakkara, N.P.D.; Rodrigues, V.; et al. Antiprotozoal, Schistosomicidal, and Antimicrobial Activities of the Essential Oil from the Leaves ofBaccharis dracunculifolia. Chem. Biodivers. 2010, 7, 993–1001. [Google Scholar] [CrossRef]
- Queiroz, L.S.; Ferreira, E.A.; Mengarda, A.C.; Almeida, A.D.C.; Pinto, P.D.F.; Coimbra, E.S.; de Moraes, J.; Denadai, ÂM.L.; Filho, A.A.D.S. In vitro and in vivo evaluation of cnicin from blessed thistle (Centaurea benedicta) and its inclusion complexes with cyclodextrins against Schistosoma mansoni. Parasitol. Res. 2021, 120, 1321–1333. [Google Scholar] [CrossRef]
- De, J. Antischistosomal Natural Compounds: Present Challenges for New Drug Screens. In Current Topics in Tropical Medicine; IntechOpen: Rijeka, Croatia, 2012; pp. 333–358. [Google Scholar]
- Veras, L.M.; Guimaraes, M.A.; Campelo, Y.D.; Vieira, M.M.; Nascimento, C.; Lima, D.F.; Vasconcelos, L.; Nakano, E.; Kuckelhaus, S.S.; Batista, M.C.; et al. Activity of epiisopiloturine against Schistosoma mansoni. Curr. Med. Chem. 2012, 19, 2051–2058. [Google Scholar] [CrossRef]
- Rocha, J.A.; Andrade, I.M.; Véras, L.M.; Quelemes, P.V.; Lima, D.F.; Soares, M.J.; Pinto, P.L.; Mayo, S.J.; Ivanova, G.; Rangel, M.; et al. Anthelmintic, Antibacterial and Cytotoxicity Activity of Imidazole Alkaloids from Pilocarpus microphyllus Leaves. Phytother. Res. 2017, 31, 624–630. [Google Scholar] [CrossRef]
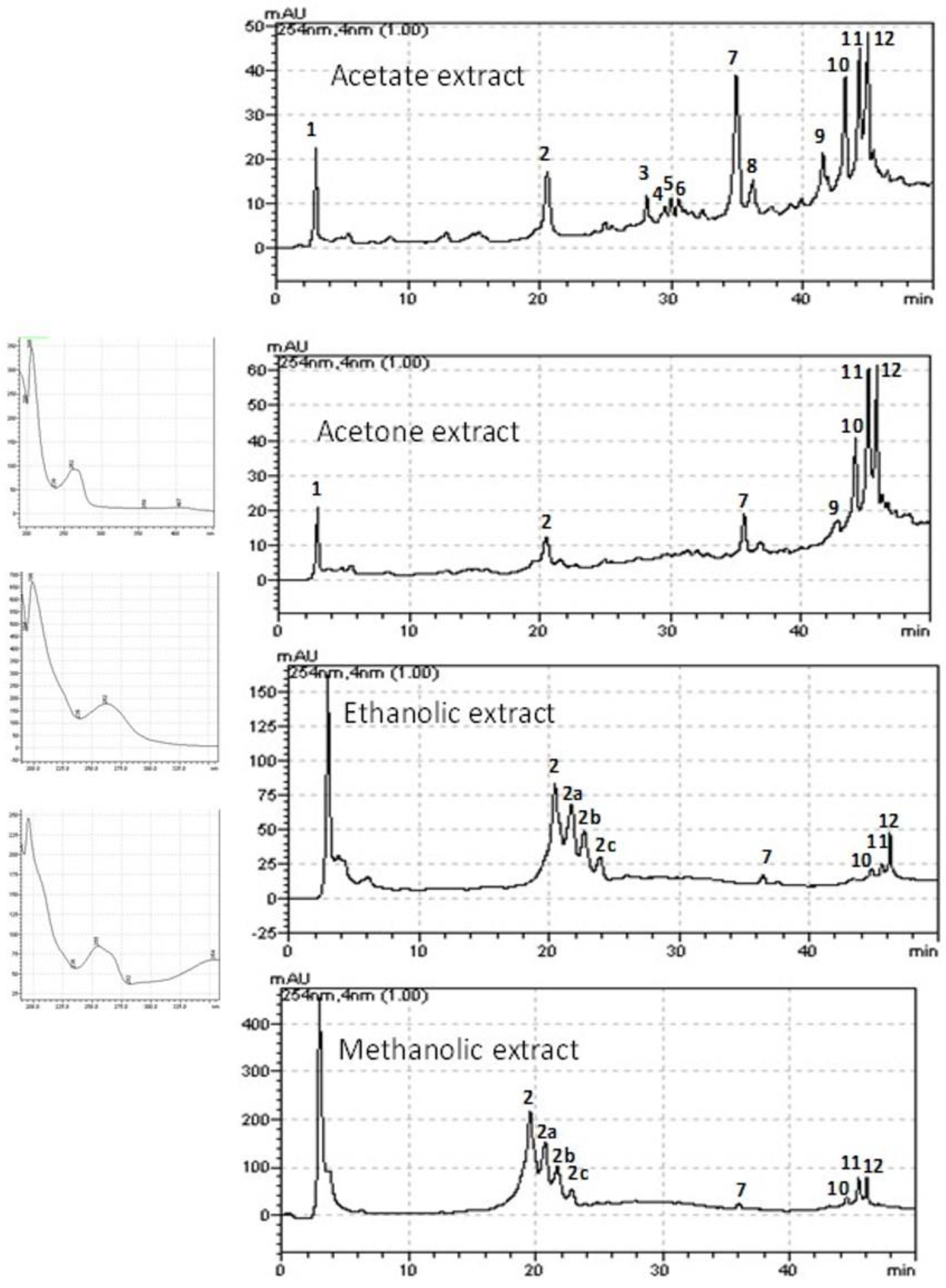

| ID | Time Retention (min) | UV max (nm) |
|---|---|---|
| 1 | 2.1 | 225; 280 |
| 2 | 20.1 | 299; 327 |
| 2a | 20.6 | 299; 327 |
| 2b | 21.0 | 262 |
| 2c | 22.2 | 262 |
| 3 | 28.1 | 255; 364 |
| 4 | 28.9 | 255; 364 |
| 5 | 30.0 | 256; 365 |
| 6 | 30.2 | 256; 365 |
| 7 | 35.0 | 255; 270 |
| 8 | 36.2 | 252; 265 |
| 9 | 42.0 | 253; 266 |
| 10 | 43.8 | 254; 270 |
| 11 | 44.0 | 285 |
| 12 | 45.1 | 287 |
| Microorganisms | EE (µg/mL) | EM (µg/mL) | EA (µg/mL) | EAE (µg/mL) | EH (µg/mL) | ED (µg/mL) | EC (µg/mL) | Control (µg/mL) |
|---|---|---|---|---|---|---|---|---|
| E. coli | 1000 | 1000 | 1000 | 1000 | 1000 | 1000 | 1000 | <0.5 * |
| S. enterica | 1000 | 1000 | 1000 | 1000 | 1000 | 1000 | 1000 | <0.5 * |
| S. maltophilia | 500 | 500 | 500 | 1000 | 500 | 1000 | 1000 | <2 ** |
| A. baumanni | 1000 | 500 | 500 | 1000 | 1000 | 1000 | 1000 | <2 ** |
| S. aureus | 1000 | 250 | 1000 | 2000 | 2000 | 1000 | 1000 | 1 *** |
| E. faecalis | 125 | 125 | 125 | 125 | 125 | 125 | 125 | 1 *** |
| S. epidermidis | 1000 | 1000 | 1000 | 1000 | 1000 | 1000 | 1000 | 1 *** |
| Group | Period of Incubation (h) | Dead Worms (%) a | Motor Activity Reduction (%) a | ||||
|---|---|---|---|---|---|---|---|
| Slight | Significant | ||||||
| M | F | M | F | M | F | ||
| Control | Immediate | 0 | 0 | 0 | 0 | 0 | 0 |
| 24 | 0 | 0 | 0 | 0 | 0 | 0 | |
| 48 | 0 | 0 | 0 | 0 | 0 | 0 | |
| 72 | 0 | 0 | 0 | 0 | 0 | 0 | |
| 0.5% DMSO | Immediate | 0 | 0 | 0 | 0 | 0 | 0 |
| 24 | 0 | 0 | 0 | 0 | 0 | 0 | |
| 48 | 0 | 0 | 0 | 0 | 0 | 0 | |
| 72 | 0 | 0 | 0 | 0 | 0 | 0 | |
| Praziquantel (1 µg/mL) | Immediate | 100 | 100 | 0 | 0 | 100 | 100 |
| 24 | 100 | 100 | 0 | 0 | 100 | 100 | |
| 48 | 100 | 100 | 0 | 0 | 100 | 100 | |
| 72 | 100 | 100 | 0 | 0 | 100 | 100 | |
| EAE (50 µg/mL) | Immediate | 0 | 0 | 0 | 0 | 0 | 0 |
| 24 | 0 | 0 | 0 | 0 | 0 | 0 | |
| 48 | 60 | 60 | 0 | 0 | 60 | 60 | |
| 72 | 100 | 100 | 0 | 0 | 100 | 100 | |
| AE (50 µg/mL) | Immediate | 0 | 0 | 0 | 0 | 0 | 0 |
| 24 | 0 | 0 | 0 | 0 | 0 | 0 | |
| 48 | 60 | 60 | 0 | 0 | 60 | 60 | |
| 72 | 100 | 100 | 0 | 0 | 100 | 100 | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lima, C.; Andrade, D.; Moreira, G.; Sousa, Â.; Leal, A.; Figuerêdo, J.; Furtado, P.; Feitosa, C.; Araujo, A.; Andrade, I.; et al. Antibacterial, Antibiofilm, and Antischistosomal Activity of Montrichardia linifera (Arruda) Schott (Araceae) Leaf Extracts. Sci. Pharm. 2021, 89, 31. https://doi.org/10.3390/scipharm89030031
Lima C, Andrade D, Moreira G, Sousa Â, Leal A, Figuerêdo J, Furtado P, Feitosa C, Araujo A, Andrade I, et al. Antibacterial, Antibiofilm, and Antischistosomal Activity of Montrichardia linifera (Arruda) Schott (Araceae) Leaf Extracts. Scientia Pharmaceutica. 2021; 89(3):31. https://doi.org/10.3390/scipharm89030031
Chicago/Turabian StyleLima, Cleiane, Dallyla Andrade, Gleidiany Moreira, Ângelo Sousa, Antonia Leal, Juliana Figuerêdo, Pedro Furtado, Chistiane Feitosa, Alyne Araujo, Ivanilza Andrade, and et al. 2021. "Antibacterial, Antibiofilm, and Antischistosomal Activity of Montrichardia linifera (Arruda) Schott (Araceae) Leaf Extracts" Scientia Pharmaceutica 89, no. 3: 31. https://doi.org/10.3390/scipharm89030031
APA StyleLima, C., Andrade, D., Moreira, G., Sousa, Â., Leal, A., Figuerêdo, J., Furtado, P., Feitosa, C., Araujo, A., Andrade, I., Miranda, J., Lima, A., Rocha, C., Silva, T., Mengarda, A. C., de Moraes, J., & Rocha, J. (2021). Antibacterial, Antibiofilm, and Antischistosomal Activity of Montrichardia linifera (Arruda) Schott (Araceae) Leaf Extracts. Scientia Pharmaceutica, 89(3), 31. https://doi.org/10.3390/scipharm89030031










