Development and Evaluation of Curcumin Liquid Crystal Systems for Cervical Cancer
Abstract
1. Introduction
2. Materials and Methods
2.1. Curcumin-Loaded LC Preparation
2.2. Physiochemical Evaluation of the LC System
2.2.1. Organoleptic Characteristic
2.2.2. Determination of pH
2.2.3. Encapsulation Efficiency (EE)
2.2.4. Particle Size Determination
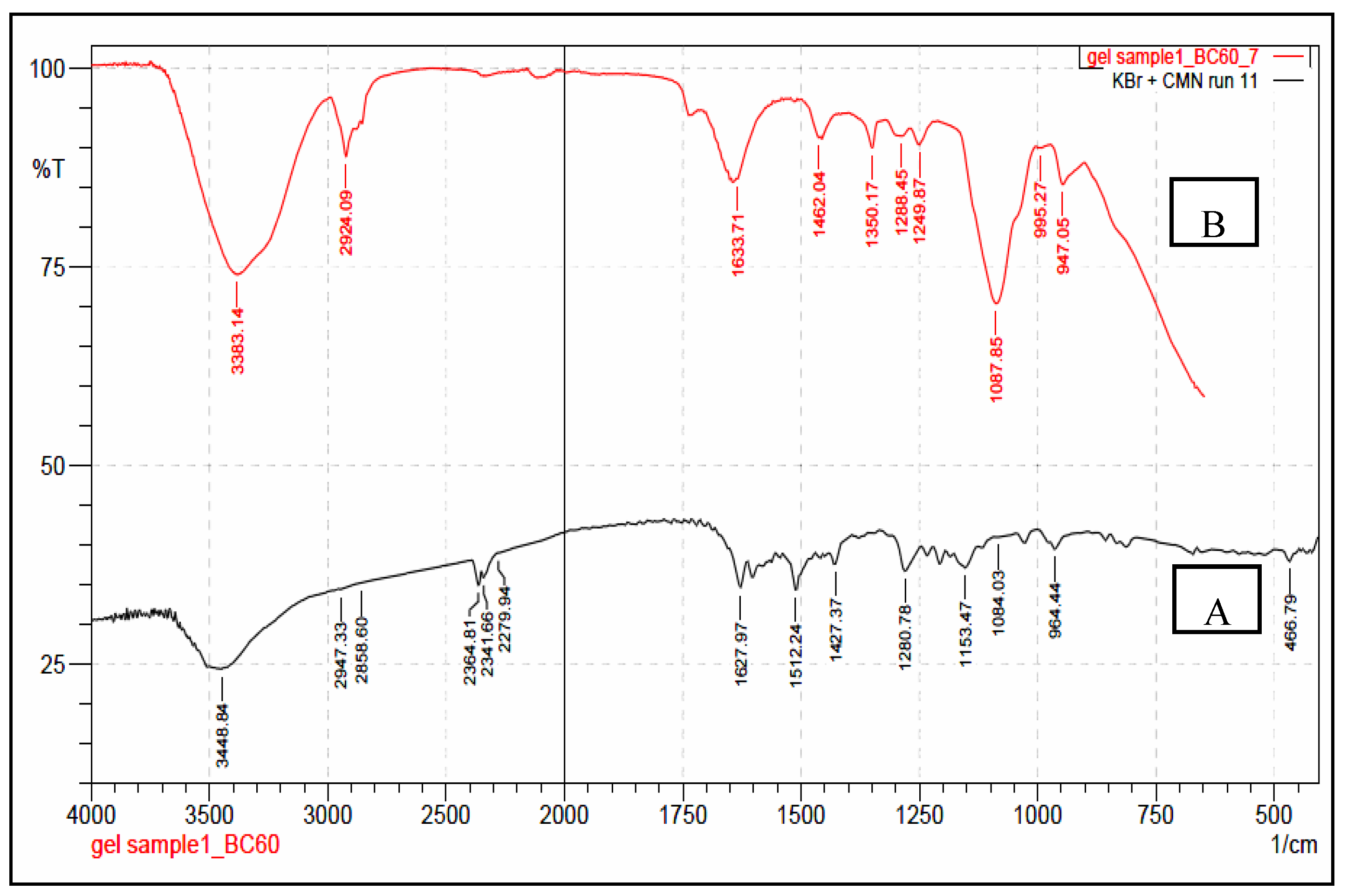
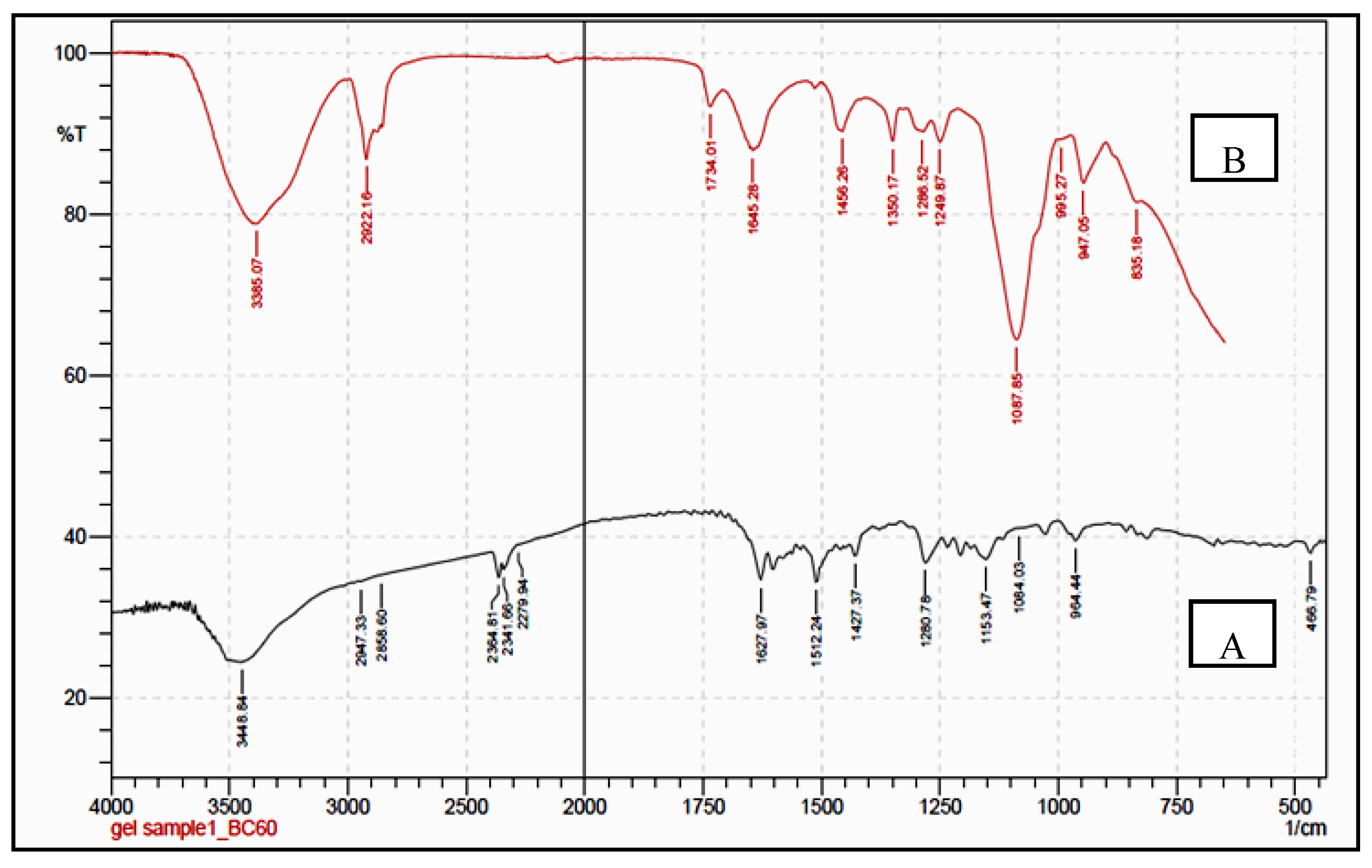
2.3. FTIR and ATR–FTIR Analysis
2.4. Environmental Scanning Electron Microscopy (ESEM)
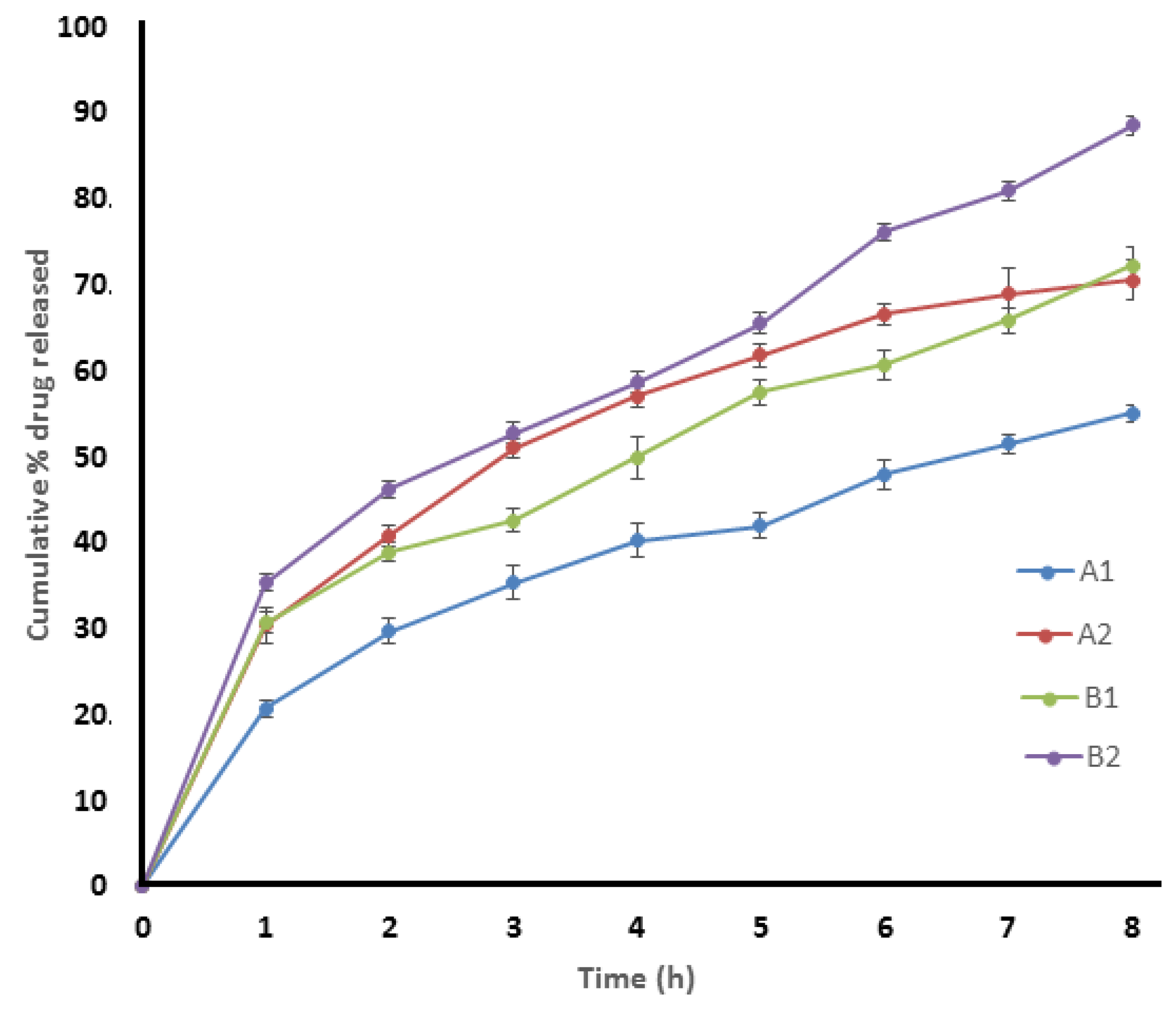
2.5. Dissolution Study
2.6. Stability Studies
2.7. Cytotoxicity of Curcumin, Liquid Crystal System, and Curcumin-Loaded Liquid Crystal System in HeLa Cells
2.7.1. MTT Assay
2.7.2. Determination of Half-Maximal Inhibitory Concentrations (IC50) of Curcumin in HeLa Cells
2.7.3. Determination of the Safety Profile of the LC Preparations
2.7.4. Determination of the Efficacy of the Curcumin Liquid Crystal Preparations
2.8. Statistical Analysis
3. Results
3.1. Selection of a Suitable Surfactant for the Liquid Crystal System
3.2. Physiochemical evaluation of the LC system
3.3. FTIR/ATR–FTIR Analysis
3.4. ESEM
3.5. Dissolution Study
3.6. Stability Studies
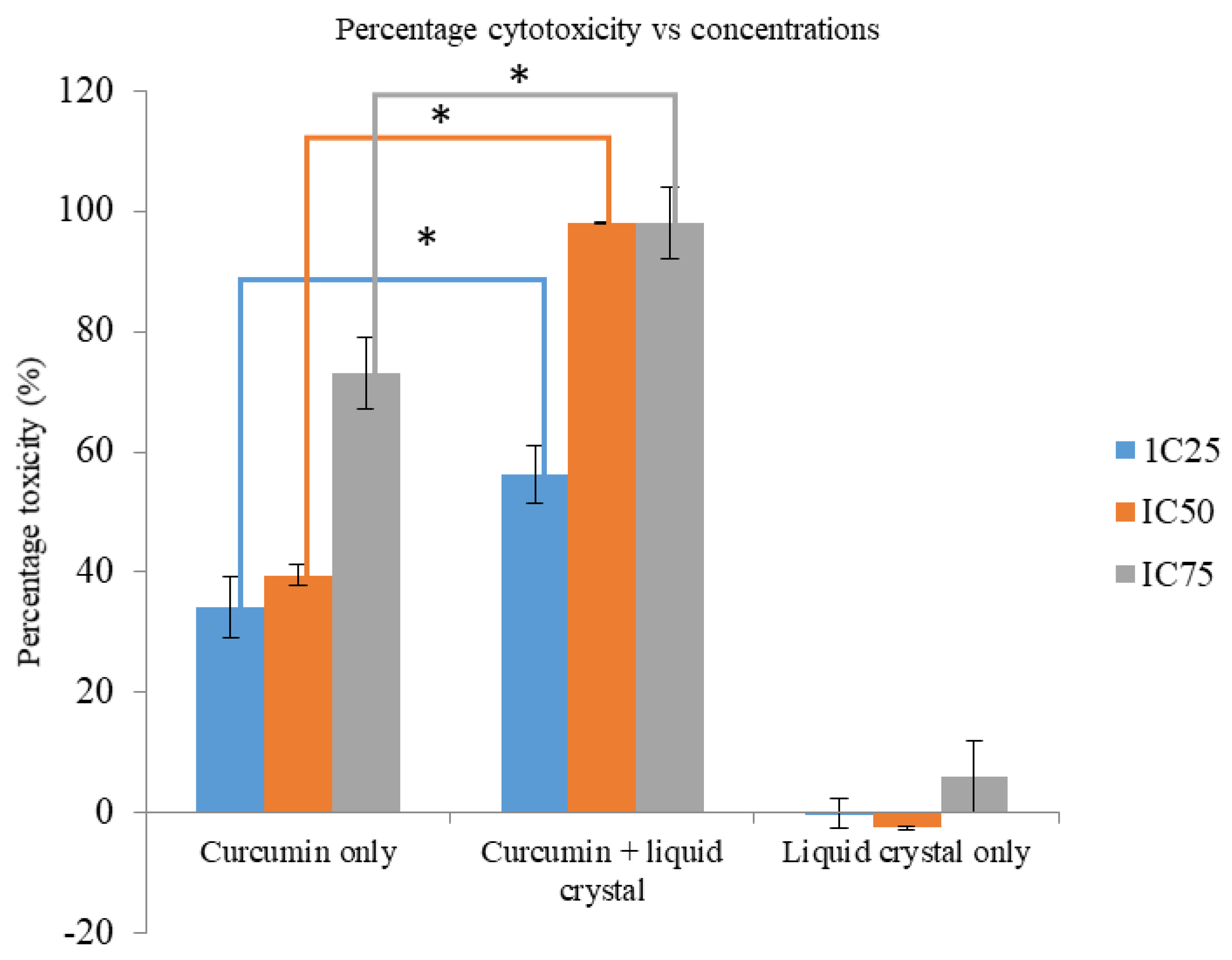
3.7. Cytotoxicity of Curcumin, Liquid Crystal System, and Curcumin-Loaded Liquid Crystal System
4. Discussion
4.1. Organoleptic Properties of Liquid Crystal Formulations
4.2. pH Measurement
4.3. Encapsulation Efficiency
4.4. Particle Size, zeta Potential, and Polydispersity Index
4.5. FTIR
4.6. ESEM
4.7. Dissolution Study
4.8. Kinetic Analysis of In Vitro Dissolution Study
4.9. Stability Studies
4.10. Determination of IC50 of Curcumin in HeLa Cells
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Franco, E.L.; Rohan, T.E.; Villa, L.L. Epidemiologic evidence and human papillomavirus infection as a necessary cause of cervical cancer. JNCI J. Natl. Cancer Inst. 1999, 91, 506–511. [Google Scholar] [CrossRef]
- Walboomers, J.M.; Jacobs, M.V.; Manos, M.M.; Bosch, F.X.; Kummer, J.A.; Shah, K.V.; Snijders, P.J.; Peto, J.; Meijer, C.J.; Muñoz, N. Human papillomavirus is a necessary cause of invasive cervical cancer worldwide. J. Pathol. 1999, 189, 12–19. [Google Scholar] [CrossRef]
- Boda, D.; Docea, A.O.; Calina, D.; Ilie, M.A.; Caruntu, C.; Zurac, S.; Neagu, M.; Constantin, C.; Branisteanu, D.E.; Voiculescu, V.; et al. Human papilloma virus: Apprehending the link with carcinogenesis and unveiling new research avenues (Review). Int. J. Oncol. 2018, 52, 637–655. [Google Scholar] [CrossRef] [PubMed]
- Zhao, J.; Zhao, Y. Effects of curcumin on proliferation and apoptosis of human cervical carcinoma HeLa cells in vitro. Chin. J. Cancer Res. 2004, 16, 225–228. [Google Scholar] [CrossRef]
- Zhao, J.; Zhao, Y.; Zhang, Y.; Chen, W. Anti-tumor effect of curcumin on human cervical carcinoma HeLa cells in vitro and in vivo. Chin. J. Cancer Res. 2007, 19, 32–36. [Google Scholar] [CrossRef]
- Jie, H.; Liu, L.; Shuangying, G.; Xingqi, W.; Rongfeng, H.; Yong, Z.; Chunling, T.; Mengqiu, X.; Xiaoqin, C. A Novel Phytantriol-Based In Situ Liquid Crystal Gel for Vaginal Delivery. AAPS PharmSciTech 2019, 20, 185. [Google Scholar] [CrossRef]
- Varan, C.; Wickström, H.; Sandler, N.; Aktaş, Y.; Bilensoy, E. Inkjet printing of antiviral PCL nanoparticles and anticancer cyclodextrin inclusion complexes on bioadhesive film for cervical administration. Int. J. Pharm. 2017, 531, 701–713. [Google Scholar] [CrossRef]
- Muhamad, N.; Plengsuriyakarn, T.; Na-Bangchang, K. Application of active targeting nanoparticle delivery system for chemotherapeutic drugs and traditional/herbal medicines in cancer therapy: A systematic review. Int. J. Nanomed. 2018, 13, 3921–3935. [Google Scholar] [CrossRef]
- Saini, J.; Bansal, V.; Chandra, A.; Madan, J.; Jain, U.K.; Chandra, R.; Jain, S.M. Bleomycin sulphate loaded nanostructured lipid particles augment oral bioavailability, cytotoxicity and apoptosis in cervical cancer cells. Colloids Surf. B Biointerfaces 2014, 118, 101–110. [Google Scholar] [CrossRef]
- Rodero, C.F.; Fioramonti Calixto, G.M.; Cristina dos Santos, K.; Sato, M.R.; Aparecido dos Santos Ramos, M.; Miró, M.S.; Rodríguez, E.; Vigezzi, C.; Bauab, T.M.; Sotomayor, C.E.; et al. Curcumin-loaded liquid crystalline systems for controlled drug release and improved treatment of vulvovaginal candidiasis. Mol. Pharm. 2018, 15, 4491–4504. [Google Scholar] [CrossRef]
- Sajomsang, W.; Gonil, P.; Saesoo, S.; Ruktanonchai, U.R.; Srinuanchai, W.; Puttipipatkhachorn, S. Synthesis and anticervical cancer activity of novel pH responsive micelles for oral curcumin delivery. Int. J. Pharm. 2014, 477, 261–272. [Google Scholar] [CrossRef] [PubMed]
- Rajabalaya, R.; Musa, M.N.; Kifli, N.; David, S.R. Oral and transdermal drug delivery systems: Role of lipid-based lyotropic liquid crystals. Drug Des. Devel. Ther. 2017, 11, 393–406. [Google Scholar] [CrossRef] [PubMed]
- Guo, C.; Wang, J.; Cao, F.; Lee, R.J.; Zhai, G. Lyotropic liquid crystal systems in drug delivery. Drug Discov. Today 2010, 15, 1032–1040. [Google Scholar] [CrossRef] [PubMed]
- Rajabalaya, R.; David, S.R.; Chellian, J.; Xin Yun, G.; Chakravarthi, S. Transdermal delivery of oxybutynin chloride proniosomal gels for the treatment of overactive bladder. Drug Deliv. 2016, 23, 1578–1587. [Google Scholar] [CrossRef]
- Vallamkondu, J.; Corgiat, E.; Buchaiah, G.; Kandimalla, R.; Reddy, P. Liquid crystals: A novel approach for cancer detection and treatment. Cancers 2018, 10, 462. [Google Scholar] [CrossRef]
- Boyd, B.J.; Whittaker, D.V.; Khoo, S.-M.; Davey, G. Lyotropic liquid crystalline phases formed from glycerate surfactants as sustained release drug delivery systems. Int. J. Pharm. 2006, 309, 218–226. [Google Scholar] [CrossRef]
- Kanai, M. Therapeutic applications of curcumin for patients with pancreatic cancer. World J. Gastroenterol. 2014, 20, 9384–9391. [Google Scholar]
- Hsu, C.-H.; Cheng, A.-L. Clinical studies with curcumin. Adv. Exp. Med. Biol. 2007, 595, 471–480. [Google Scholar]
- Salmazi, R.; Calixto, G.; Bernegossi, J.; dos Santos Ramos, M.A.; Bauab, T.; Chorilli, M. A curcumin-loaded liquid crystal precursor mucoadhesive system for the treatment of vaginal candidiasis. Int. J. Nanomed. 2015, 10, 4815–4824. [Google Scholar]
- Wang, Z.; Fu, L.; Liu, X.; Zhang, L.; Guo, F.; Zhao, X. Phase and rheological properties of a curcumin-encapsulated cubic liquid crystal. J. Surfactants Deterg. 2017, 20, 673–679. [Google Scholar] [CrossRef]
- Rajabalaya, R.; Leen, G.; Chellian, J.; Chakravarthi, S.; David, S. Tolterodine tartrate proniosomal gel transdermal delivery for overactive bladder. Pharmaceutics 2016, 8, 27. [Google Scholar] [CrossRef] [PubMed]
- David, S.R.N.; Hui, M.S.; Pin, C.F.; Ci, F.Y.; Rajabalaya, R. Formulation and in vitro evaluation of ethosomes as vesicular carrier for enhanced topical delivery of isotretinoin. Int. J. Drug Deliv. 2013, 5, 28. [Google Scholar]
- Mukhopadhyay, H.K.; Das, S.K.; Bhowmik, M.; Ray, S.; Rajabalaya, R.; Ghosh, L.; Gupta, B.K. Preparation and characterization of polymethacrylate-based matrix microspheres of carbamazepine using solvent evaporation method. Farmacia 2013, 62, 137–158. [Google Scholar]
- Ghosal, K.; Chandra, A.; Rajabalaya, R.; Chakraborty, S.; Nanda, A. Mathematical modeling of drug release profiles for modified hydrophobic HPMC based gels. Pharmazie 2012, 67, 147–155. [Google Scholar] [PubMed]
- David, S.R.; Abd Malek, N.; Mahadi, A.H.; Chakravarthi, S.; Rajabalaya, R. Development of controlled release silicone adhesive-based mupirocin patch demonstrates antibacterial activity on live rat skin against Staphylococcus aureus. Drug Des. Devel. Ther. 2018, 12, 481–494. [Google Scholar] [CrossRef]
- Ding, L.; Ma, S.; Lou, H.; Sun, L.; Ji, M. Synthesis and biological evaluation of curcumin derivatives with water-soluble groups as potential antitumor agents: An in vitro investigation using tumor cell lines. Molecules 2015, 20, 21501–21514. [Google Scholar] [CrossRef]
- Ujhelyi, Z.; Kalantari, A.; Vecsernyés, M.; Róka, E.; Fenyvesi, F.; Póka, R.; Kozma, B.; Bácskay, I. The enhanced inhibitory effect of different antitumor agents in self-microemulsifying drug delivery systems on human cervical cancer hela cells. Molecules 2015, 20, 13226–13239. [Google Scholar] [CrossRef]
- Sharma, C.; Vas, A.J.; Goala, P.; Gheewala, T.M.; Rizvi, T.A.; Hussain, A. Ethanolic neem (azadirachta indica) leaf extract prevents growth of mcf-7 and hela cells and potentiates the therapeutic index of cisplatin. J. Oncol. 2014, 2014, 321754. [Google Scholar] [CrossRef]
- Hussain, A.; Sharma, C.; Khan, S.; Shah, K.; Haque, S. Aloe vera inhibits proliferation of human breast and cervical cancer cells and acts synergistically with cisplatin. Asian Pac. J. Cancer Prev. 2015, 16, 2939–2946. [Google Scholar] [CrossRef]
- Rajabalaya, R.; Xian, T.W.; David, S.R.N. Preparation and evaluation of transdermal drug delivery of ondansetron hydrochloride: Effect of vegetable oils as permeation enhancer. Lat. Am. J. Pharm. 2012, 31, 1005–1012. [Google Scholar]
- De Mattos, C.B.; Argenta, D.F.; de Melchiades, G.L.; Cordeiro, M.N.S.; Tonini, M.L.; Moraes, M.H.; Weber, T.B.; Roman, S.S.; Nunes, R.J.; Teixeira, H.F.; et al. Nanoemulsions containing a synthetic chalcone as an alternative for treating cutaneous leshmaniasis: Optimization using a full factorial design. Int. J. Nanomed. 2015, 10, 5529–5542. [Google Scholar]
- Bonferoni, M.; Rossi, S.; Sandri, G.; Ferrari, F.; Gavini, E.; Rassu, G.; Giunchedi, P. Nanoemulsions for “Nose-to-Brain” drug delivery. Pharmaceutics 2019, 11, 84. [Google Scholar] [CrossRef] [PubMed]
- Lucero, M.J.; Vigo, J.; León, M.J. A study of shear and compression deformations on hydrophilic gels of tretinoin. Int. J. Pharm. 1994, 106, 125–133. [Google Scholar] [CrossRef]
- Du Plessis, J.; Ramachandran, C.; Weiner, N.; Müller, D.G. The influence of particle size of liposomes on the deposition of drug into skin. Int. J. Pharm. 1994, 103, 277–282. [Google Scholar] [CrossRef]
- El-Samaligy, M.S.; Afifi, N.N.; Mahmoud, E.A. Increasing bioavailability of silymarin using a buccal liposomal delivery system: Preparation and experimental design investigation. Int. J. Pharm. 2006, 308, 140–148. [Google Scholar] [CrossRef]
- Jukanti, R.; Sheela, S.; Bandari, S.; Veerareddy, P.R. Enhanced bioavailability of exemestane via proliposomes based transdermal delivery. J. Pharm. Sci. 2011, 100, 3208–3222. [Google Scholar] [CrossRef]
- Scholes, P.D.; Coombes, A.G.A.; Illum, L.; Davis, S.S.; Watts, J.F.; Ustariz, C.; Vert, M.; Davies, M.C. Detection and determination of surface levels of poloxamer and PVA surfactant on biodegradable nanospheres using SSIMS and XPS. J. Control. Release 1999, 59, 261–278. [Google Scholar] [CrossRef]
- Zhang, Y.; Tang, L.; Sun, L.; Bao, J.; Song, C.; Huang, L.; Liu, K.; Tian, Y.; Tian, G.; Li, Z.; et al. A novel paclitaxel-loaded poly(ε-caprolactone)/Poloxamer 188 blend nanoparticle overcoming multidrug resistance for cancer treatment. Acta Biomater. 2010, 6, 2045–2052. [Google Scholar] [CrossRef]
- Kronberg, B.; Krister Holmberg, B.L.; Kronberg, B.; Holmberg, K.; Lindman, B. Polymers in Solution. In Surface Chemistry of Surfactants and Polymers; John Wiley & Sons, Ltd.: Chichester, UK, 2014; pp. 175–195. ISBN 9781118695968. [Google Scholar]
- Shustova, N.B.; McCarthy, B.D.; Dincǎ, M. Turn-on fluorescence in tetraphenylethylene-based metal-organic frameworks: An alternative to aggregation-induced emission. J. Am. Chem. Soc. 2011, 133, 20126–20129. [Google Scholar] [CrossRef]
- Ishikawa, J.; Takahashi, Y.; Hazawa, M.; Fukushi, Y.; Yoshizawa, A.; Kashiwakura, I. Suppressive effects of liquid crystal compounds on the growth of U937 human leukemic monocyte lymphoma cells. Cancer Cell Int. 2012, 12, 3. [Google Scholar] [CrossRef]

| Formulation Code | Curcumin: Tween 80 (%) | Colour | Odour | pH | Encapsulation Efficiency (%) ± SD | Vesicle Size (nm) ± SD | Polydispersity (PDI) Index | Zeta Potential (mV) |
|---|---|---|---|---|---|---|---|---|
| A1 | 0.5:40 | Brown | Oil-like | 3.91 | 91.02 ± 1.69 | 8.01 ± 0.85 | 0.158 ± 0.07 | −28.2 ± 1.42 |
| A2 | 1.0:40 | Brown | Oil-like | 4.13 | 92.31 ± 2.36 | 10.31± 1.81 | 0.102 ± 0.04 | −23.4 ± 2.27 |
| A3 | 5.0:40 | Yellowish brown | Oil-like | 4.24 | 81.58 ± 1.57 | 11.01 ± 1.30 | 0.441 ± 0.12 | −18.1 ± 1.47 |
| B1 | 0.5:60 | Brown | Oil-like | 4.18 | 88.01 ± 2.80 | 6.12 ± 0.67 | 0.461 ± 0.17 | −19.5 ± 1.89 |
| B2 | 1.0:60 | Brown | Oil-like | 4.07 | 93.26 ± 3.05 | 7.56 ± 1.21 | 0.254 ± 0.09 | −22.5 ± 1.43 |
| B3 | 5.0:60 | Yellowish brown | Oil-like | 4.39 | 80.36 ± 2.46 | 12.97 ± 1.12 | 0.458 ± 0.13 | −16.8 ± 1.22 |
| Formulation Code | Zero Order | First Order | Higuchi Model | Korsmeyer–Peppas | |||||
|---|---|---|---|---|---|---|---|---|---|
| Ko (h−1) | r2 | K1 (h−1) | r2 | KH (h−1/2) | r2 | KKP (h−n) | r2 | n | |
| A1 | 4.6318 | 0.9734 | −0.0335 | 0.9897 | 30.7499 | 0.9448 | 0.2087 | 0.9950 | 0.46 |
| A2 | 5.6309 | 0.9275 | −0.0540 | 0.9734 | 28.3905 | 0.9323 | 0.3088 | 0.9902 | 0.42 |
| B1 | 5.7982 | 0.9923 | −0.0545 | 0.9867 | 32.7567 | 0.9528 | 0.2913 | 0.9793 | 0.41 |
| B2 | 7.4150 | 0.9943 | −0.1002 | 0.9485 | 32.6238 | 0.9547 | 0.3379 | 0.9819 | 0.44 |
| Formulation Code | Period | Drug Content (%) | ||
|---|---|---|---|---|
| 5 ± 2 °C | 25 ± 0.5 °C | 45 ± 0.5 °C | ||
| A1 | Initial | 91.02 ± 1.69 | 91.02 ± 1.69 | 91.02 ± 1.69 |
| After 3 months | 88.01 ± 1.11 | 87.57 ± 1.87 | 85.02 ± 2.35 | |
| A2 | Initial | 92.31 ± 2.36 | 92.31 ± 2.36 | 92.31 ± 2.36 |
| After 3 months | 89.41 ± 2.06 | 87.23 ± 1.36 | 85.87 ± 1.57 | |
| B1 | Initial | 88.01 ± 2.80 | 88.01 ± 2.80 | 88.01 ± 2.80 |
| After 3 months | 86.98 ± 1.40 | 84.08 ± 2.94 | 82.14 ± 13.57 | |
| B2 | Initial | 93.26 ± 3.05 | 93.26 ± 3.05 | 93.26 ± 3.05 |
| After 3 months | 91.23 ± 2.10 | 89.02 ±2.23 | 85.52 ± 3.67 | |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
R David, S.; Akmar Binti Anwar, N.; Yian, K.R.; Mai, C.-W.; Das, S.K.; Rajabalaya, R. Development and Evaluation of Curcumin Liquid Crystal Systems for Cervical Cancer. Sci. Pharm. 2020, 88, 15. https://doi.org/10.3390/scipharm88010015
R David S, Akmar Binti Anwar N, Yian KR, Mai C-W, Das SK, Rajabalaya R. Development and Evaluation of Curcumin Liquid Crystal Systems for Cervical Cancer. Scientia Pharmaceutica. 2020; 88(1):15. https://doi.org/10.3390/scipharm88010015
Chicago/Turabian StyleR David, Sheba, Nurul Akmar Binti Anwar, Koh Rhun Yian, Chun-Wai Mai, Sanjoy Kumar Das, and Rajan Rajabalaya. 2020. "Development and Evaluation of Curcumin Liquid Crystal Systems for Cervical Cancer" Scientia Pharmaceutica 88, no. 1: 15. https://doi.org/10.3390/scipharm88010015
APA StyleR David, S., Akmar Binti Anwar, N., Yian, K. R., Mai, C.-W., Das, S. K., & Rajabalaya, R. (2020). Development and Evaluation of Curcumin Liquid Crystal Systems for Cervical Cancer. Scientia Pharmaceutica, 88(1), 15. https://doi.org/10.3390/scipharm88010015





