P-Glycoprotein/ABCB1 Might Contribute to Morphine/Cisplatin-Induced Hepatotoxicity in Rats
Abstract
1. Introduction
2. Methods
2.1. Drugs and Chemicals
2.2. Experimental Animal Protocol
2.3. Blood and Tissue Sample Preparation
2.4. Determination of Serum Liver Function Tests and Liver Tissue Oxidant/Antioxidant Markers
2.5. Liver Histopathological Examination
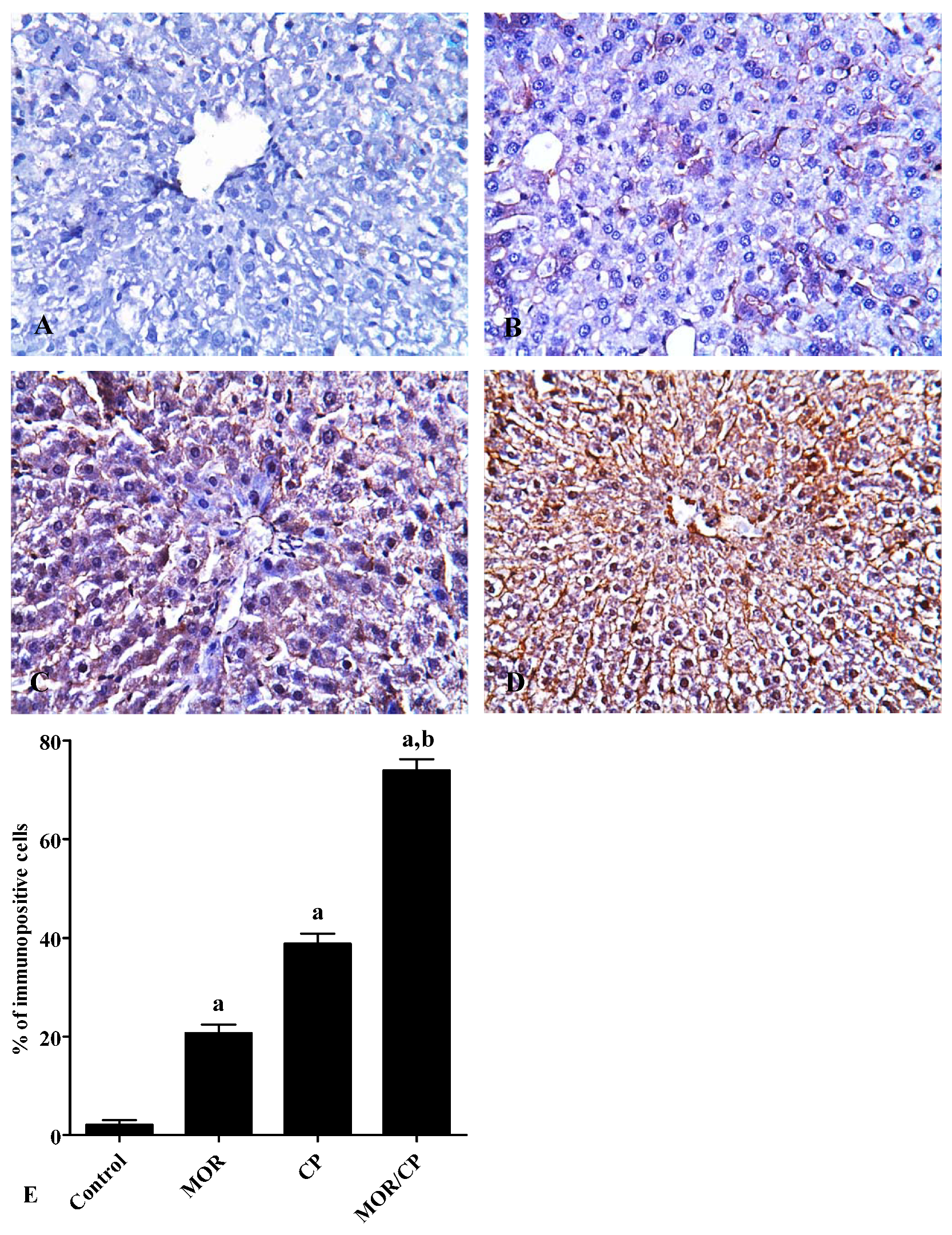
2.6. Caspase 3 Immunohistochemical Staining of Liver Sections
2.7. Determination of TNF-α, IL-6, and P-gp in Liver Tissue Homogenate
2.8. Statistical Analysis
3. Results
3.1. Effect of MOR on Liver Index and Hepatic Function in CP-Induced Toxicity
3.2. Effect of MOR on Hepatic Oxidant/Antioxidant Markers in CP-Induced Toxicity
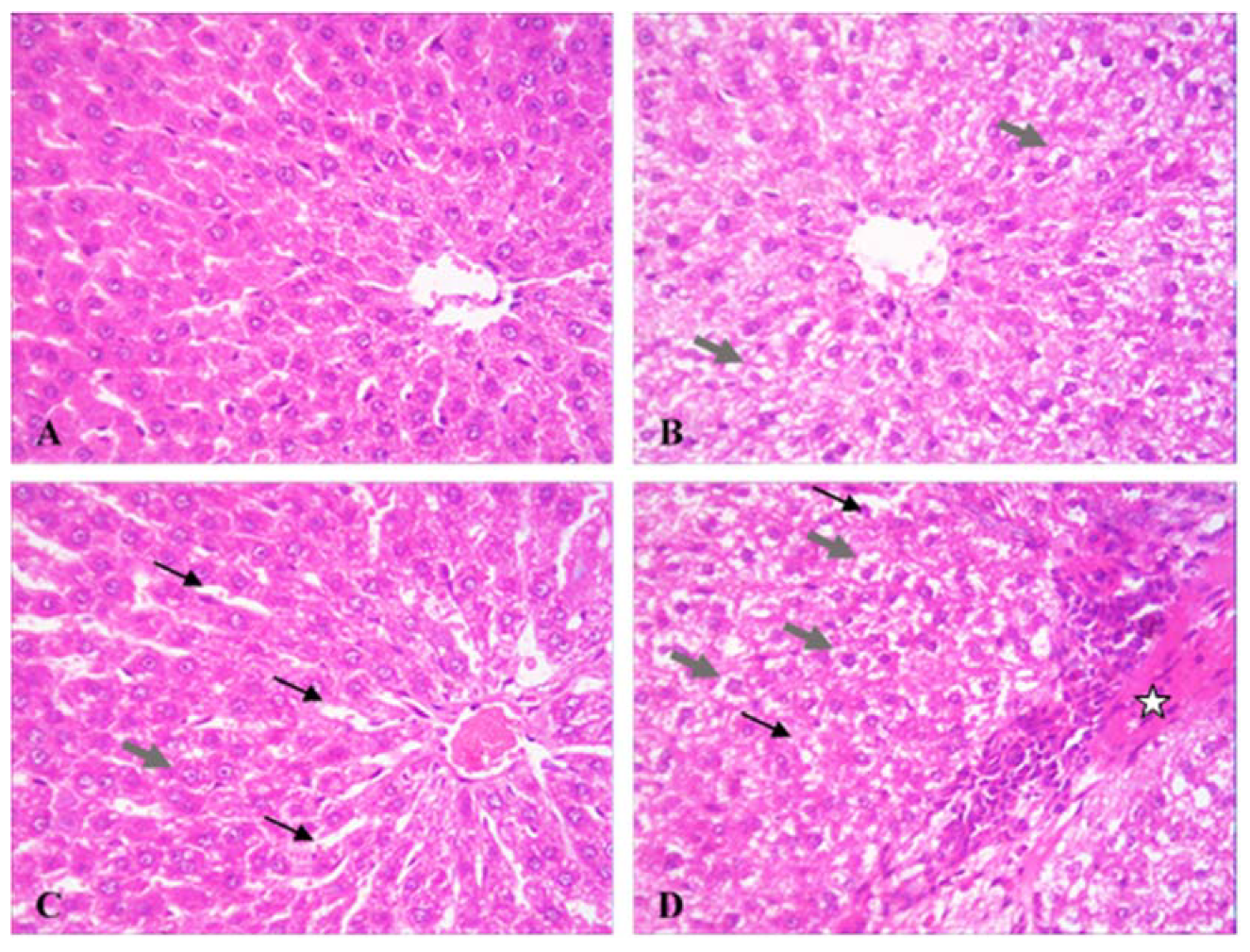
3.3. Effect of MOR on Liver Microscopic Features and Caspase 3 Regulation in CP-Induced Toxicity
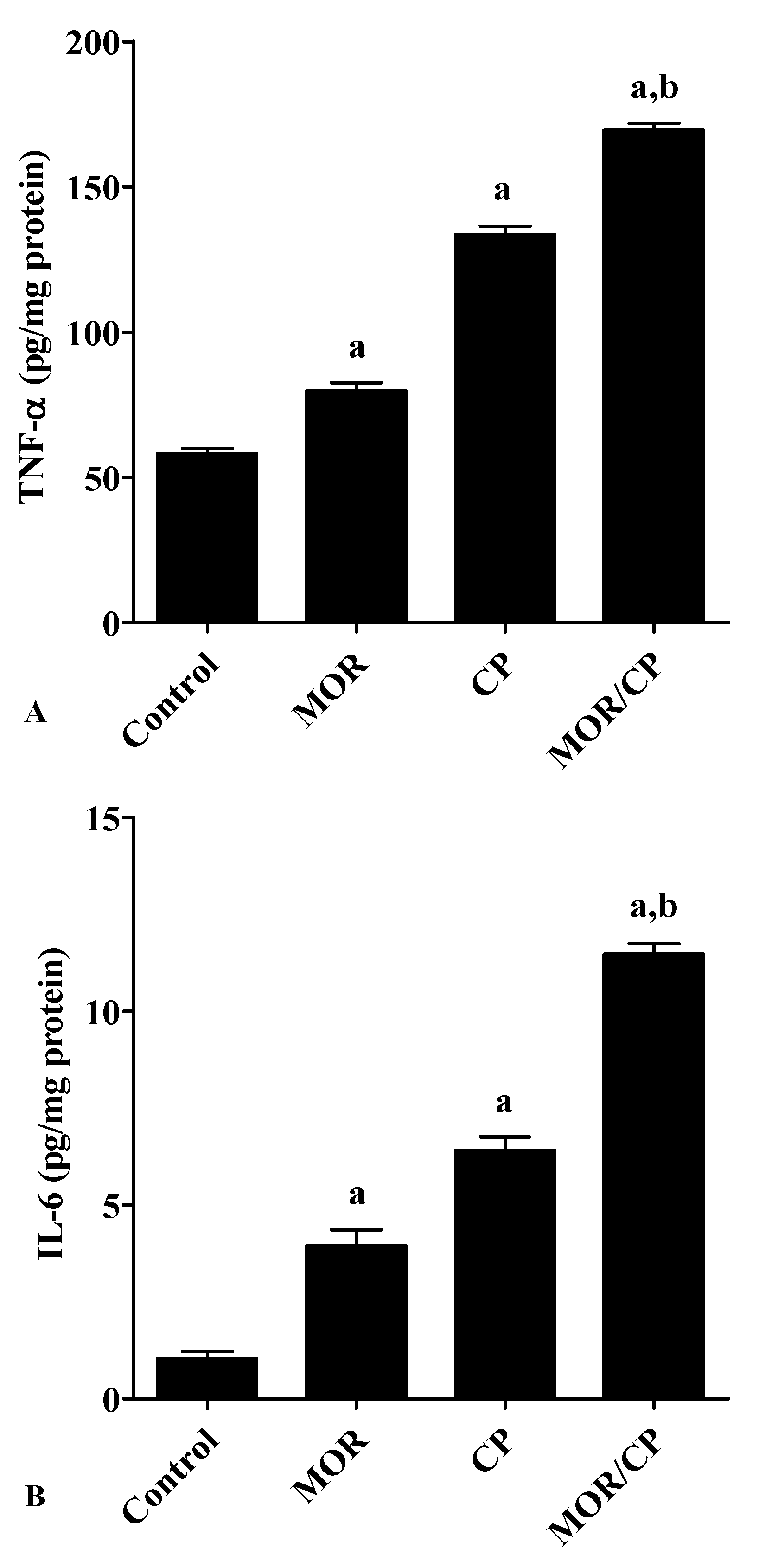
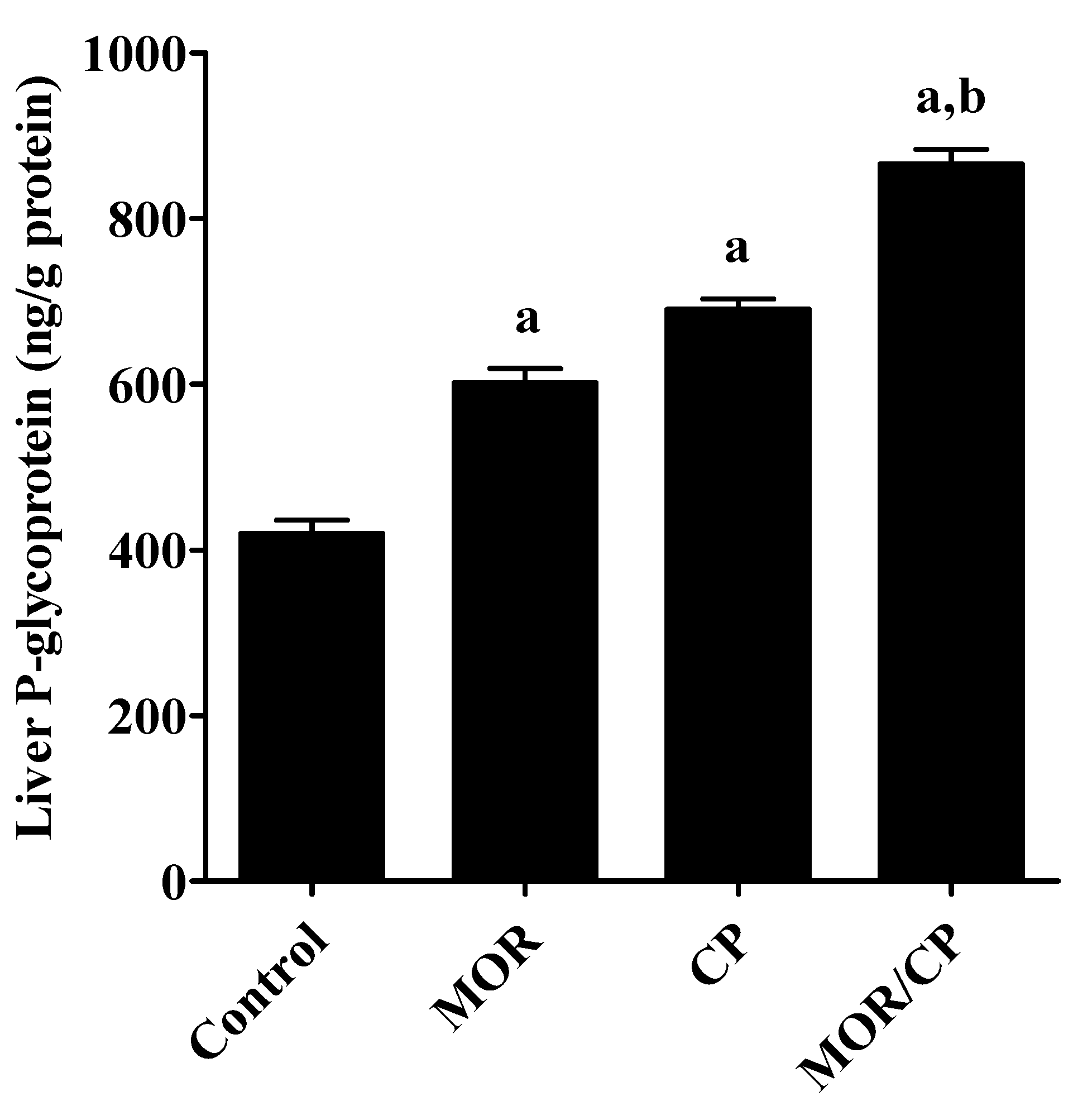
3.4. Effect of MOR on Liver TNF-α, IL-6 and P-gp Levels in CP-Induced Toxicity
4. Discussion
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| ALP | alkaline phosphatase |
| ALT | alanine aminotransferase |
| AST | aspartate aminotransferase |
| CP | Cisplatin |
| ELISA | enzyme-linked immunosorbent assay |
| GSH | reduced glutathione |
| IL-6 | Interleukin-6 |
| MDA | Malondialdehyde |
| MOR | Morphine |
| NO | nitric oxide |
| P-gp | P-glycoprotein |
| SOD | superoxide dismutase |
| TNF-α | tumor necrosis factor-α |
References
- Oun, R.; Moussa, Y.E.; Wheate, N.J. The side effects of platinum-based chemotherapy drugs: A review for chemists. Dalton Trans. 2018, 47, 6645–6653. [Google Scholar] [CrossRef] [PubMed]
- Hwang, D.B.; Won, D.H.; Shin, Y.S.; Kim, S.Y.; Kang, B.C.; Lim, K.M.; Che, J.H.; Nam, K.T.; Yun, J.W. Ccrn4l as a pre-dose marker for prediction of cisplatin-induced hepatotoxicity susceptibility. Free Radic. Biol. Med. 2020, 148, 128–139. [Google Scholar] [CrossRef] [PubMed]
- Hagar, H.; Husain, S.; Fadda, L.M.; Attia, N.M.; Attia, M.M.A.; Ali, H.M. Inhibition of NF-kappaB and the oxidative stress-dependent caspase-3 apoptotic pathway by betaine supplementation attenuates hepatic injury mediated by cisplatin in rats. Pharmacol. Rep. 2019, 71, 1025–1033. [Google Scholar] [CrossRef] [PubMed]
- Yay, A.; Onses, M.S.; Sahmetlioglu, E.; Ceyhan, A.; Pekdemir, S.; Onder, G.O.; Sezer, G.; Sarica, Z.S.; Aydin, F. Raman spectroscopy: A novel experimental approach to evaluating cisplatin induced tissue damage. Talanta 2020, 207, 120343. [Google Scholar] [CrossRef]
- Neamatallah, T.; El-Shitany, N.A.; Abbas, A.T.; Ali, S.S.; Eid, B.G. Honey protects against cisplatin-induced hepatic and renal toxicity through inhibition of NF-kappaB-mediated COX-2 expression and the oxidative stress dependent BAX/Bcl-2/caspase-3 apoptotic pathway. Food Funct. 2018, 9, 3743–3754. [Google Scholar] [CrossRef]
- Pınar, N.; Çakırca, G.; Hakverdi, S.; Kaplan, M. Protective effect of alpha lipoic acid on cisplatin induced hepatotoxicity in rats. Biotech. Histochem. 2019, 1–6. [Google Scholar] [CrossRef]
- Calls, A.; Carozzi, V.; Navarro, X.; Monza, L.; Bruna, J. Pathogenesis of platinum-induced peripheral neurotoxicity: Insights from preclinical studies. Exp. Neurol. 2019, 325, 113141. [Google Scholar] [CrossRef]
- Donertas, B.; Unel, C.C.; Erol, K. Cannabinoids and agmatine as potential therapeutic alternatives for cisplatin-induced peripheral neuropathy. J. Exp. Pharmacol. 2018, 10, 19–28. [Google Scholar] [CrossRef]
- Li, W.; Sparidans, R.; El-Lari, M.; Wang, Y.; Lebre, M.C.; Beijnen, J.H.; Schinkel, A.H. P-glycoprotein (ABCB1/MDR1) limits brain accumulation and Cytochrome P450-3A (CYP3A) restricts oral availability of the novel FGFR4 inhibitor fisogatinib (BLU-554). Int. J. Pharm. 2020, 573, 118842. [Google Scholar] [CrossRef]
- Hernández, L.I.; Bauer, M.; Wulkersdorfer, B.; Traxl, A.; Philippe, C.; Weber, M.; Häusler, S.; Stieger, B.; Jäger, W.; Mairinger, S.; et al. Measurement of Hepatic ABCB1 and ABCG2 Transport Activity with [(11)C]Tariquidar and PET in Humans and Mice. Mol. Pharm. 2020, 17, 316–326. [Google Scholar] [CrossRef]
- Chaves, C.; Remiao, F.; Cisternino, S.; Decleves, X. Opioids and the Blood-Brain Barrier: A Dynamic Interaction with Consequences on Drug Disposition in Brain. Curr. Neuropharmacol. 2017, 15, 1156–1173. [Google Scholar] [CrossRef] [PubMed]
- Hole, L.D.; Larsen, T.H.; Fossan, K.O.; Limé, F.; Schjøtt, J. Morphine enhances doxorubicin-induced cardiotoxicity in the rat. Cardiovasc. Toxicol. 2014, 14, 251–259. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Afsar, T.; Razak, S.; Almajwal, A.; Shabbir, M.; Khan, M.R. Evaluating the protective potency of Acacia hydaspica R. Parker on histological and biochemical changes induced by Cisplatin in the cardiac tissue of rats. BMC Complement. Altern. Med. 2019, 19, 182. [Google Scholar] [CrossRef] [PubMed]
- Buege, J.A.; Aust, S.D. Microsomal lipid peroxidation. Methods Enzymol. 1978, 52, 302–310. [Google Scholar] [PubMed]
- Sastry, K.V.; Moudgal, R.P.; Mohan, J.; Tyagi, J.S.; Rao, G.S. Spectrophotometric determination of serum nitrite and nitrate by copper-cadmium alloy. Anal. Biochem. 2002, 306, 79–82. [Google Scholar] [CrossRef]
- El-Sheikh, A.A.; Rifaai, R.A. Peroxisome Proliferator Activator Receptor (PPAR)-gamma Ligand, but Not PPAR-alpha, Ameliorates Cyclophosphamide-Induced Oxidative Stress and Inflammation in Rat Liver. PPAR Res. 2014, 2014, 626319. [Google Scholar] [CrossRef]
- Samarghandian, S.; Afshari, R.; Farkhondeh, T. Effect of long-term treatment of morphine on enzymes, oxidative stress indices and antioxidant status in male rat liver. Int. J. Clin. Exp. Med. 2014, 7, 1449–1453. [Google Scholar]
- Shahid, M.; Subhan, F.; Ullah, I.; Ali, G.; Alam, J.; Shah, R. Beneficial effects of Bacopa monnieri extract on opioid induced toxicity. Heliyon 2016, 2, e00068. [Google Scholar] [CrossRef]
- Malekshah, R.E.; Khaleghian, A. Influence of Silybum Marianum on Morphine Addicted Rats, Biochemical Parameters and Molecular Simulation Studies on micro-Opioid Receptor. Drug Res. 2019, 69, 630–638. [Google Scholar]
- Aminian, A.; Javadi, S.; Rahimian, R.; Dehpour, A.R.; Asadi, A.F.; Moghaddas, P.; Ejtemaei, M.S. Enhancement of Cisplatin Nephrotoxicity by Morphine and Its Attenuation by the Opioid Antagonist Naltrexone. Acta Med. Iran. 2016, 54, 422–429. [Google Scholar]
- Madera-Salcedo, I.K.; Cruz, S.L.; Gonzalez-Espinosa, C. Morphine prevents lipopolysaccharide-induced TNF secretion in mast cells blocking IkappaB kinase activation and SNAP-23 phosphorylation: Correlation with the formation of a beta-arrestin/TRAF6 complex. J. Immunol. 2013, 191, 3400–3409. [Google Scholar] [CrossRef] [PubMed]
- El-Hage, N.; Dever, S.M.; Fitting, S.; Ahmed, T.; Hauser, K.F. HIV-1 coinfection and morphine coexposure severely dysregulate hepatitis C virus-induced hepatic proinflammatory cytokine release and free radical production: Increased pathogenesis coincides with uncoordinated host defenses. J. Virol. 2011, 85, 11601–11614. [Google Scholar] [CrossRef] [PubMed]
- Bishr, A.; Sallam, N.; Nour El-Din, M.; Awad, A.S.; Kenawy, S.A. Ambroxol attenuates cisplatin-induced hepatotoxicity and nephrotoxicity via inhibition of p-JNK/p-ERK. Can. J. Physiol. Pharmacol. 2019, 97, 55–64. [Google Scholar] [CrossRef] [PubMed]
- Barrère-Lemaire, S.; Combes, N.; Sportouch-Dukhan, C.; Richard, S.; Nargeot, J.; Piot, C. Morphine mimics the antiapoptotic effect of preconditioning via an Ins(1,4,5)P3 signaling pathway in rat ventricular myocytes. Am. J. Physiol Heart Circ. Physiol. 2005, 288, H83–H88. [Google Scholar] [CrossRef]
- Luo, F.C.; Zhao, L.; Deng, J.; Liang, M.; Zeng, X.S.; Liu, H.; Bai, J. Geranylgeranylacetone protects against morphine-induced hepatic and renal damage in mice. Mol. Med. Rep. 2013, 7, 694–700. [Google Scholar] [CrossRef]
- Demeule, M.; Brossard, M.; Béliveau, R. Cisplatin induces renal expression of P-glycoprotein and canalicular multispecific organic anion transporter. Am. J. Physiol. 1999, 277, F832–F840. [Google Scholar] [CrossRef]
- He, C.; Sun, Z.; Hoffman, R.; Yang, Z.; Jiang, Y.; Wang, L.; Hao, Y. P-Glycoprotein Overexpression Is Associated With Cisplatin Resistance in Human Osteosarcoma. Anticancer Res. 2019, 39, 1711–1718. [Google Scholar] [CrossRef]
- Cheng, Q.; Liao, M.; Hu, H.; Li, H.; Wu, L. Asiatic Acid (AA) Sensitizes Multidrug-Resistant Human Lung Adenocarcinoma A549/DDP Cells to Cisplatin (DDP) via Downregulation of P-Glycoprotein (MDR1) and Its Targets. Cell Physiol. Biochem. 2018, 47, 279–292. [Google Scholar] [CrossRef]
- Wang, J.; Wang, H.; Zhao, L.; Fan, S.; Yang, Z.; Gao, F.; Chen, L.; Xiao, G.G.; Molnar, J.; Wang, Q. Down-regulation of P-glycoprotein is associated with resistance to cisplatin and VP-16 in human lung cancer cell lines. Anticancer Res. 2010, 30, 3593–3598. [Google Scholar]
- Gibalova, L.; Seres, M.; Rusnak, A.; Ditte, P.; Labudova, M.; Uhrik, B.; Pastorek, J.; Sedlak, J.; Breier, A.; Sulova, Z. P-glycoprotein depresses cisplatin sensitivity in L1210 cells by inhibiting cisplatin-induced caspase-3 activation. Toxicol. In Vitro 2012, 26, 435–444. [Google Scholar] [CrossRef]
- Breier, A.; Gibalova, L.; Seres, M.; Barancik, M.; Sulova, Z. New insight into p-glycoprotein as a drug target. Anticancer Agents Med. Chem. 2013, 13, 159–170. [Google Scholar] [CrossRef] [PubMed]
- Hano, M.; Tomášová, L.; Šereš, M.; Pavlíková, L.; Breier, A.; Sulová, Z. Interplay between P-Glycoprotein Expression and Resistance to Endoplasmic Reticulum Stressors. Molecules 2018, 23, 337. [Google Scholar] [CrossRef] [PubMed]
- Lu, C.; Shan, Z.; Li, C.; Yang, L. MiR-129 regulates cisplatin-resistance in human gastric cancer cells by targeting P-gp. Biomed. Pharmacother. 2017, 86, 450–456. [Google Scholar] [CrossRef] [PubMed]
- Ochiai, W.; Kaneta, M.; Nagae, M.; Yuzuhara, A.; Li, X.; Suzuki, H.; Hanagata, M.; Kitaoka, S.; Suto, W.; Kusunoki, Y.; et al. Mice with neuropathic pain exhibit morphine tolerance due to a decrease in the morphine concentration in the brain. Eur. J. Pharm. Sci. 2016, 92, 298–304. [Google Scholar] [CrossRef] [PubMed]
- Kaneta, M.; Ochiai, W.; Nagae, M.; Suto, W.; Hanagata, M.; Suzuki, H.; Kitaoka, S.; Hatogai, J.; Ikarashi, N.; Sugiyama, K. Mechanism for Increased Expression of UGT2B in the Liver of Mice with Neuropathic Pain. Biol. Pharm. Bull. 2016, 39, 1809–1814. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Kimura, Y.; Shibata, M.; Tamada, M.; Ozaki, N.; Arai, K. Pharmacokinetics of Morphine in Rats with Adjuvant-induced Arthritis. In Vivo 2017, 31, 811–817. [Google Scholar]
- Balayssac, D.; Cayre, A.; Ling, B.; Maublant, J.; Penault-Llorca, F.; Eschalier, A.; Coudoré, F.; Authier, N. Increase in morphine antinociceptive activity by a P-glycoprotein inhibitor in cisplatin-induced neuropathy. Neurosci. Lett. 2009, 465, 108–112. [Google Scholar] [CrossRef]
- Schaefer, C.P.; Arkwright, N.B.; Jacobs, L.M.; Jarvis, C.K.; Hunn, K.C.; Largent-Milnes, T.M.; Tome, M.E.; Davis, T.P. Chronic morphine exposure potentiates p-glycoprotein trafficking from nuclear reservoirs in cortical rat brain microvessels. PLoS ONE 2018, 13, e0192340. [Google Scholar] [CrossRef]
- Leibrand, C.R.; Paris, J.J.; Jones, A.M.; Masuda, Q.N.; Halquist, M.S.; Kim, W.K.; Knapp, P.E.; Kashuba, A.D.M.; Hauser, K.F.; McRae, M. HIV-1 Tat and opioids act independently to limit antiretroviral brain concentrations and reduce blood-brain barrier integrity. J. Neurovirol. 2019, 25, 560–577. [Google Scholar] [CrossRef]
- Amawi, H.; Sim, H.M.; Tiwari, A.K.; Ambudkar, S.V.; Shukla, S. ABC Transporter-Mediated Multidrug-Resistant Cancer. Adv. Exp. Med. Biol. 2019, 1141, 549–580. [Google Scholar]
- Machado, S.P.; Cunha, V.; Reis-Henriques, M.A.; Ferreira, M. Histopathological lesions, P-glycoprotein and PCNA expression in zebrafish (Danio rerio) liver after a single exposure to diethylnitrosamine. Environ. Toxicol. Pharmacol. 2014, 38, 720–732. [Google Scholar] [CrossRef] [PubMed]
- Ren, T.; Fu, G.H.; Liu, T.F.; Hu, K.; Li, H.R.; Fang, W.H.; Yang, X.L. Toxicity and accumulation of zinc pyrithione in the liver and kidneys of Carassius auratus gibelio: Association with P-glycoprotein expression. Fish. Physiol Biochem. 2017, 43, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Santana, M.T.; Poitevin, S.; Paul, P.; McKay, N.; Jourde-Chiche, N.; Legris, T.; Mouly-Bandini, A.; Dignat-George, F.; Brunet, P.; Masereeuw, R.; et al. Indoxyl Sulfate Upregulates Liver P-Glycoprotein Expression and Activity through Aryl Hydrocarbon Receptor Signaling. J. Am. Soc. Nephrol. 2018, 29, 906–918. [Google Scholar]
- Duflou, J.; Darke, S.; Easson, J. Morphine concentrations in stomach contents of intravenous opioid overdose deaths. J. Forensic Sci. 2009, 54, 1181–1184. [Google Scholar] [CrossRef]
| Control | MOR | CP | MOR/CP | |
|---|---|---|---|---|
| Liver index | 21.3 ± 0.6 | 22.6 ± 0.7 | 27.6 ± 0.3 a | 30.6 ± 0.5 a,b |
| ALT (U/dL) | 34 ± 1 | 46 ± 3 a | 59 ± 3 a | 70 ± 2 a,b |
| AST (U/dL) | 122 ± 3 | 142 ± 3 a | 172 ±5 a | 189 ± 2 a,b |
| ALP (U/L) | 109 ± 8 | 149 ± 10 a | 222 ± 11 a | 265 ± 7 a,b |
| Total bilirubin (mg/dL) | 0.15 ± 0.02 | 0.25 ± 0.01 a | 0.31 ± 0.02 a | 0.42 ± 0.03 a,b |
| Albumin (g/dL) | 4.5 ± 0.2 | 3.2 ± 0.3 a | 2.8 ± 0.3 a | 1.9 ± 0.2 a,b |
| Control | MOR | CP | MOR/CP | |
|---|---|---|---|---|
| MDA (μmol/g protein) | 2.9 ± 0.8 | 5.4 ± 0.6 a | 6.7 ± 1.8 a | 11.6 ± 0.7 a,b |
| NO (μmol/mg protein) | 24 ± 3 | 35 ± 4 a | 41 ± 5 a | 74 ± 6 a,b |
| GSH (μg/g protein) | 39 ± 5 | 29 ± 4 a | 18 ± 3 a | 8 ± 4 a,b |
| Catalase (U/mg protein) | 22 ± 3 | 15 ± 2 a | 13 ± 1 a | 8 ± 2 a,b |
| SOD (U/mg protein) | 8.5 ± 1.1 | 6.2 ± 1.2 a | 5.2 ± 0.8 a | 4.0 ± 0.5 a,b |
| Control | MOR | CP | MOR/CP | |
|---|---|---|---|---|
| Degeneration and necrosis | 0 | 0 | + | ++ |
| Sinusoidal dilatation and biliary hyperplasia | 0 | + | ++ | +++ |
| Hepatocyte vacuolation | 0 | + | ++ | +++ |
| Cellular infiltration | 0 | 0 | + | ++ |
© 2020 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
El-Sheikh, A.A.K. P-Glycoprotein/ABCB1 Might Contribute to Morphine/Cisplatin-Induced Hepatotoxicity in Rats. Sci. Pharm. 2020, 88, 14. https://doi.org/10.3390/scipharm88010014
El-Sheikh AAK. P-Glycoprotein/ABCB1 Might Contribute to Morphine/Cisplatin-Induced Hepatotoxicity in Rats. Scientia Pharmaceutica. 2020; 88(1):14. https://doi.org/10.3390/scipharm88010014
Chicago/Turabian StyleEl-Sheikh, Azza A.K. 2020. "P-Glycoprotein/ABCB1 Might Contribute to Morphine/Cisplatin-Induced Hepatotoxicity in Rats" Scientia Pharmaceutica 88, no. 1: 14. https://doi.org/10.3390/scipharm88010014
APA StyleEl-Sheikh, A. A. K. (2020). P-Glycoprotein/ABCB1 Might Contribute to Morphine/Cisplatin-Induced Hepatotoxicity in Rats. Scientia Pharmaceutica, 88(1), 14. https://doi.org/10.3390/scipharm88010014




