Correlation between Cyclosporine Blood Levels and Area under Blood Concentration Time Curve in Iraqi Bone Marrow Transplant Patients Treated with Neoral® Oral Solution
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Design
2.2. Cyclosporine Assay
2.3. Pharmacokinetic and Statistical Analysis
3. Results and Discussion
4. Conclusions
Author Contributions
Acknowledgments
Conflicts of Interest
References and Notes
- Neoral® Prescribing Information; Novartis Pharmaceuticals Corporation: East Hanover, NJ, USA, 2015.
- Azzi, J.; Sayegh, M.; Mallat, S. Calcineurin Inhibitors: 40 Years Later, Can’t Live Without. J. Immunol. 2013, 191, 5785–5791. [Google Scholar] [CrossRef] [PubMed]
- Lindholm, A. Factors influencing the pharmacokinetics of cyclosporine in man. Ther. Drug Monit. 1991, 13, 465–477. [Google Scholar] [CrossRef] [PubMed]
- Golubovic, B.; Prostran, M.; Miljkovic, B.; Vucicevic, K.; Radivojevic, D.; Grabnar, I. Population pharmacokinetic approach of immunosuppressive therapy in kidney transplant patients. Curr. Med. Chem. 2016, 23, 1998–2011. [Google Scholar] [CrossRef]
- Xue, L.; Zhang, W.W.; Ding, X.L.; Zhang, J.J.; Bao, J.A.; Miao, L.Y. Population pharmacokinetics and individualized dosage prediction of cyclosporine in allogeneic hematopoietic stem cell transplant patients. Am. J. Med. Sci. 2014, 348, 448–454. [Google Scholar] [CrossRef] [PubMed]
- Schiff, J.; Cole, E.; Cantarovich, M. Therapeutic monitoring of calcineurin inhibitors for the nephrologist. Clin. J. Am. Soc. Nephrol. 2007, 2, 374–384. [Google Scholar] [CrossRef]
- Jacobson, P.A.; Ng, J.; Green, K.G.; Rogosheske, J.; Brundage, R. Post-transplant day significantly influences pharmacokinetics of cyclosporine after hematopoietic stem cell transplantation. Biol. Blood Marrow Transplant. 2003, 9, 304–311. [Google Scholar] [CrossRef]
- Guidance for Industry, FDA. Bioavailability and Bioequivalence Studies for Orally Administered Drug Products. General Considerations; 2003.
- Guidance for Industry, FDA. Bioequivalence Studies with Pharmacokinetic Endpoints for Drugs Submitted under an ANDA. Draft Guidance; 2013.
- European Medicines Agency (EMEA). Guidelines on the Investigation of Bioequivalence; 2010.
- Shargel, L.; Andrew, Y. Applied Biopharmaceutics and Pharmacokinetics, 6th ed.; McGraw-Hill Education: New York, NY, USA, 2012. [Google Scholar]
- Malcolm, R.; Thomas, N.T. Clinical Pharmacokinetics and Pharmacodynamics: Concepts and Applications, 4th ed.; LWW: Philadelphia, PA, USA, 2011. [Google Scholar]
- Sara, Z.; Molouk, H.; Asieh, A.; Amir, S.; Seyed, H.K.; Sara, M.; Mania, R.; Ardeshir, G. Assessment of cyclosporine serum concentrations on the incidence of acute graft versus host disease post-hematopoietic stem cell transplantation. Iran. J. Pharm. Res. 2014, 13, 305–312. [Google Scholar]
- García, C.I.; Valcarcel, D.; Martino, R.; Piñana, J.L.; Barba, P.; Novelli, S.; Esquirol, A.; Garrido, A.; Saavedra, S.; Granell, M.; et al. Impact of Cyclosporine Levels on the Development of Acute Graft versus Host Disease after Reduced Intensity Conditioning Allogeneic Stem Cell Transplantation. Mediat. Inflamm. 2014, 2014, 620682. [Google Scholar]
- Gerull, S.; Arber, C.; Bucher, C.; Gratwohl, A.; Halter, J.; Heim, D.; Tichelli, A.; Stern, M. Cyclosporine levels and rate of graft rejection following non-myeloablative conditioning for allogeneic hematopoietic SCT. Bone Marrow Transplant. 2010, 46, 740–746. [Google Scholar] [CrossRef]
- Sibbald, C.; Seto, W.; Taylor, T.; Saunders, E.F.; Doyle, J.; Dupuis, L.L. Determination of area under the whole blood concentration versus time curve after first intravenous cyclosporine dose in children undergoing hematopoietic stem cell transplant: Limited sampling strategies. Ther. Drug Monit. 2008, 30, 434–438. [Google Scholar] [CrossRef]
- Shibata, N.; Hoshino, N.; Minouchi, T.; Yamaji, A.; Park, K.; Tomoyoshi, T.; Abe, H.; Kodama, M. Relationship between area under the concentration versus time curve of cyclosporine A, creatinine clearance, hematocrit value, and other clinical factors in Japanese renal transplant patients. Int. J. Clin. Pharmacol. Ther. 1998, 36, 202–209. [Google Scholar] [PubMed]
- Sarem, S.; Nekka, F.; Barrière, O.; Bittencourt, H.; Duval, M.; Teira, P.; Haddad, E.; Théorêt, Y.; Lapeyraque, A.L.; Litalien, C. Limited sampling strategies for estimating intravenous and oral cyclosporine area under the curve in pediatric hematopoietic stem cell transplantation. Ther. Drug Monit. 2015, 37, 198–205. [Google Scholar] [CrossRef] [PubMed]
- Srinivas, N.R. Therapeutic drug monitoring of cyclosporine and area under the curve prediction using a single time point strategy: Appraisal using peak concentration data. Biopharm. Drug Dispos. 2015, 36, 575–586. [Google Scholar] [CrossRef] [PubMed]
- Frassetto, L.A.; Tan-Tam, C.C.; Barin, B.; Browne, M.; Wolfe, A.R.; Stock, P.G.; Roland, M.; Benet, L.Z. Best single time point correlations with AUC for cyclosporine and tacrolimus in HIV-infected kidney and liver transplant recipients. Transplantation 2014, 97, 702–707. [Google Scholar] [CrossRef] [PubMed]
- Yixin, J.I.A.; Xu, M.; Yan, L.; Chunlei, X.U.; Wen, Z.; Yuqing, J.; WEI, H. Optimal sampling time-point for cyclosporine A concentration monitoring in heart transplant recipients. Exp. Ther. Med. 2018, 16, 4265–4270. [Google Scholar]
- Jaksch, P.; Kocher, A.; Neuhauser, P.; Sarahrudi, K.; Seweryn, J.; Wisser, W.; Klepetko, W. Monitoring C2 level predicts exposure in maintenance lung transplant patients receiving the microemulsion formulation of cyclosporine (Neoral). J. Heart Lung Transplant. 2005, 24, 1076–1080. [Google Scholar] [CrossRef]
- Morris, R.G. Cyclosporin therapeutic drug monitoring -an established service revisited. Clin. Biochem. Rev. 2003, 24, 33–46. [Google Scholar]
- Cantarovich, M.; Barkun, J.S.; Tchervenkov, J.I.; Besner, J.G.; Aspeslet, L.; Metrakos, P. Comparison of neoral dose monitoring with cyclosporine through levels versus 2-hr postdose levels in stable liver transplant patients. Transplantation 1998, 66, 1621–1627. [Google Scholar] [CrossRef]
- Cantarovich, M.; Besner, J.G.; Barkun, J.S.; Elstein, E.; Loertscher, R. Two-hour cyclosporine level determination is the appropriate tool to monitor Neoral therapy. Clin. Transplant. 1998, 12, 243–249. [Google Scholar]
- Kong, D.C.M.; Shuttleworth, P.; Bailey, M.; Grigg, A. CsA 2-h concentration correlates best with area under the concentration–time curve after allo-SCT compared with trough CsA. Bone Marrow Transplant. 2012, 47, 54–59. [Google Scholar] [CrossRef][Green Version]
- Henriques, L.S.; Matos, F.M.; Vaisbich, M.H. Pharmacokinetics of cyclosporin—A microemulsion in children with idiopathic nephrotic syndrome. Clinics 2012, 67, 1197–1202. [Google Scholar] [CrossRef]
- Jorga, A.; Holt, D.W.; Johnston, A. Therapeutic drug monitoring of cyclosporine. Transplant. Proc. 2004, 36 (Suppl. 2), S396–S403. [Google Scholar] [CrossRef] [PubMed]
- Wada, Y.; Kotani, T.; Takeuchi, T.; Wakura, R.; Wakura, D.; Makino, S.; Hanafusa, T. Therapeutic drug monitoring of cyclosporine microemulsion in patients with corticosteroid-resistant systemic lupus erythematosus. Mod. Rheumatol. 2015, 25, 708–713. [Google Scholar] [CrossRef] [PubMed]
- Furukawa, T.; Kurasaki-Ida, T.; Masuko, M.; Tsukada, N.; Okazuka, K.; Sato, N.; Yano, T.; Abe, T.; Momoi, A.; Shibasaki, Y.; et al. Pharmacokinetic and pharmacodynamic analysis of cyclosporine A (CsA) to find the best single time point for the monitoring and adjusting of CsA dose using twice-daily 3-h intravenous infusions in allogeneic hematopoietic stem cell transplantation. Int. J. Hematol. 2010, 92, 144–151. [Google Scholar] [CrossRef] [PubMed]
- Duncan, N.; Arrazi, J.; Nagra, S.; Cook, M.; Thomson, A.H.; Craddock, C. Prediction of intravenous cyclosporine area under the concentration-time curve after allogeneicstem cell transplantation. Ther. Drug Monit. 2010, 32, 353–358. [Google Scholar] [CrossRef] [PubMed]
- Eljebari, H.; Ben, F.N.; Salouage, I.; Gaies, E.; Trabelsi, S.; Jebabli, N.; Lakhal, M.; Ben Othman, T.; Kouz, A. Estimation of abbreviated cyclosporine A area under the concentration-time curve in allogenic stem cell transplantation after oral administration. J. Transplant. 2012, 2012, 342701. [Google Scholar] [CrossRef] [PubMed]
- Dupuis, L.L.; Seto, W.; Teuffel, O.; Gibson, P.; Schultz, K.R.; Doyle, J.D.; Gassas, A.; Egeler, R.M.; Sung, L.; Schechter, T. Prediction of area under the cyclosporine concentration versus time curve in children undergoing hematopoietic stem cell transplantation. Biol. Blood Marrow Transplant. 2013, 19, 418–423. [Google Scholar] [CrossRef][Green Version]
- Schrauder, A.; Saleh, S.; Sykora, K.W.; Hoy, H.; Welte, K.; Boos, J.; Hempel, G.; Grigull, L. Pharmacokinetic monitoring of intravenous cyclosporine A in pediatric stem-cell transplant recipients. The trough level is not enough. Pediatr. Transplant. 2009, 13, 444–450. [Google Scholar] [CrossRef]
- Nashan, B.; Cole, E.; Levy, G.; Thervet, E. Clinical validation studies of neoral C2 monitoring: A review. Transplantation 2002, 73 (Suppl. 9), S3–S11. [Google Scholar] [CrossRef]
- Pescovitz, M.D.; Barbeito, R. Two-hour post-dose cyclosporine level is a better predictor than trough level of acute rejection of renal allografts. Clin. Transplant. 2002, 16, 378–382. [Google Scholar] [CrossRef]
- Duncan, N.; Craddock, C. Optimizing the use of cyclosporine in allogeneic stem cell transplantation. Bone Marrow Transplant. 2006, 38, 169–174. [Google Scholar] [CrossRef] [PubMed]
- Levy, G.; Thervet, E.; Lake, J.; Uchida, K. Patient management by Neoral C(2) monitoring: An international consensus statement. Transplantation 2002, 73 (Suppl. 9), S8–S12. [Google Scholar] [CrossRef] [PubMed]
- ICH Guideline for Good Clinical Practice (GCP), E6; 1996.
- Latest WMA Declaration of Helsinki. Ethical Principles for Medical Research Involving Human Subjects; 2013.
- Sanquer, S.; Schwarzinger, M.; Maury, S.; Yakouben, K.; Rafi, H.; Pautas, C.; Kuentz, M.; Barouki, R.; Cordonnier, C. Calcineurin activity as a functional index of immunosuppression after allogeneic stem-cell transplantation. Transplantation 2004, 77, 854–858. [Google Scholar] [CrossRef] [PubMed]
- Sommerer, C.; Giese, T.; Schmidt, J.; Meuer, S.; Zeier, M. Ciclosporin A tapering monitored by NFAT-regulated gene expression: A new concept of individual immunosuppression. Transplantation 2008, 85, 15–21. [Google Scholar] [CrossRef]
- Al-Tamimi, J.J.; Al-Khames, Q.A.; Abass, H.M. CD4 T-Helper cell count is as an alternative promising marker for dosing cyclosporine in kidney transplant patient. IJPPS 2016, 8, 85–92. [Google Scholar]
- Al-Khames, Q.A.; Al-Tamimi, J.J.; Abass, H.M.; Alawi, A.A.; Al-Tamimi, K.F. Adverse effects and biochemical changes, in Iraqis kidney transplanted patients using cyclosporine. Pharm. Glob. 2015, 1, 1–11. [Google Scholar]
- Al-Temimi, H.M.; Ghareeb, M.M.; Dyab, A.A.; Al-Tamimi, K.F. Therapeutic monitoring of cyclosporine—A in Iraqi patients kidney transplanted. WJPP 2014, 10, 50–59. [Google Scholar]
- Einollahi, B.; Nemati, E.; Rostami, Z.; Teimoori, M.; Ghadian, A.R. Electrolytes disturbance and cyclosporine blood levels among kidney transplant recipients. Int. J. Org. Transplant. Med. 2012, 3, 166–175. [Google Scholar]
- Chavers, B.M.; Hårdstedt, M.; Gillingham, K.J. Hyperlipidemia in pediatric kidney transplant recipients treated with cyclosporine. Pediatr Nephrol. 2003, 18, 565–569. [Google Scholar] [CrossRef]
- Morales, J.M.; Wramner, L.; Kreis, H.; Durand, D.; Campistol, J.M.; Andres, A.; Arenas, J.; Nègre, E.; Burke, J.T.; Growth, C.G. Sirolimus does not exhibit nephrotoxicity compared to cyclosporine in renal transplant recipients. Am. J. Transplant. 2002, 2, 436–442. [Google Scholar] [CrossRef]
- Lucey, M.R.; Abdelmalek, M.F.; Gagliardi, R.; Granger, D.; Holt, C.; Kam, I.; Klintmalm, G.; Langnas, A.; Shetty, K.; Tzakis, A.; et al. A comparison of tacrolimus and cyclosporine in liver transplantation: Effects on renal function and cardiovascular risk status. Am. J. Transplant. 2005, 5, 1111–1119. [Google Scholar] [CrossRef] [PubMed]
- Marienhagen, K.; Lehner, F.; Klempnauer, J.; Hecker, H.; Borlak, J. Treatment of cyclosporine induced hypertension: Results from long-term observational study using different antihypertensive medications. Vascul. Pharmacol. 2019, 115, 69–83. [Google Scholar] [CrossRef] [PubMed]
- Robert, N.; Wong, G.W.; Wright, J.M. Effect of cyclosporine on blood pressure. Cochrane Database Syst. Rev. 2010, CD007893. [Google Scholar] [CrossRef] [PubMed]
| Patients Characteristics | Mean ± SD | Range |
|---|---|---|
| Gender | 20 males, 34 females | |
| Age (years) | 31.2 ± 10.3 | 18–56 |
| Body weight (kg) | 69.1 ± 10.2 | 42–83 |
| Height (cm) | 164.5 ± 5.4 | 150–172 |
| Body Mass Index (kg/m2) | 25.4 ± 2.8 | 18.7–29.8 |
| Systolic blood pressure (mm Hg) | 120.3 ± 1.4 | 116–124 |
| Diastolic blood pressure (mmHg) | 80.5 ± 1.6 | 76–87 |
| Serum Aspartate transaminase (U/L) | 25.4 ± 7.9 | 10–40 |
| Serum Alanine transaminase (U/L) | 28.0 ± 10.9 | 10–50 |
| Serum bilirubin (mg/dL) | 0.92 ± 0.18 | 0.6–1.2 |
| Blood K (mEq/L) | 4.1 ± 0.66 | 3–5 |
| Blood Mg (mg/dL) | 2.1 ± 0.34 | 1.6–2.8 |
| Serum creatinine (mg/dL) | 1.0 ± 0.17 | 0.8–1.3 |
| Blood urea (mg/dL) | 28.8 ± 6.4 | 20–45 |
| Blood uric acid (mg/dL) | 5.1 ± 0.90 | 3–7 |
| Total cholesterol (mg/dL) | 191.1 ± 20.6 | 127–230 |
| Triglyceride (mg/dL) | 134.9 ± 9.3 | 121–150 |
| Cyclosporine Concentration | Mean ± SD | Range |
|---|---|---|
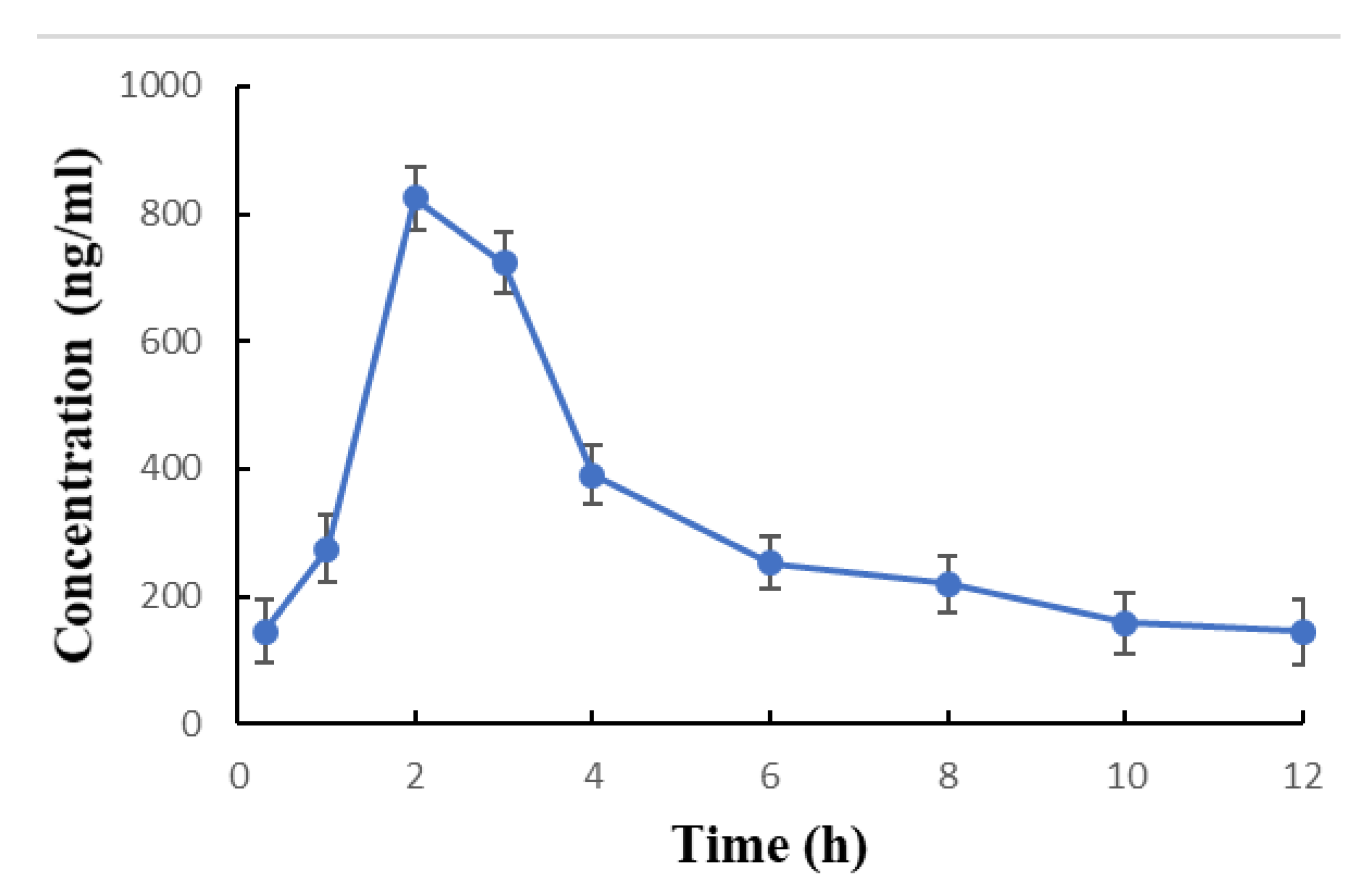
| C0 (0.3 h pre-dosing) | 145.0 ± 48.3 | 030–244 |
| C1 (1h after dosing) | 273.7 ± 52.7 | 100–400 |
| C2 (2 h after dosing) | 824.4 ± 48.7 | 700–952 |
| C3 (3 h after dosing) | 723.4 ± 47.6 | 598–850 |
| C4 (4 h after dosing) | 390.9 ± 45.9 | 286–520 |
| C6 (6 h after dosing) | 251.9 ± 41.4 | 180–380 |
| C8 (8 h after dosing) | 219.0 ± 43.4 | 130–350 |
| C10 (10 h after dosing) | 158.0 ± 48.4 | 052–271 |
| C12 (12 h after dosing) | 144.0 ± 50.4 | 025–199 |
| Parameter | Mean ± SD | Range |
|---|---|---|
| AUC0–12h (ng∙h/mL) | 3732.5 ± 451.6 | 2700.4–5096.0 |
| AUC0–4h (ng∙h/mL) | 2590.1 ± 225.5 | 1950.0–3162.0 |
| Cmax (ng/mL) | 824.4 ± 48.7 | 700.0–952.0 |
| C0 (ng/mL) | 145.0 ± 48.3 | 30.0–244.0 |
| C12 (ng/mL) | 144.0 ± 50.4 | 25.0–199.0 |
| * Tmax (h) | 2.0 * | 2.0 * |
| Conc. | AUC0–4h | AUC0–12h |
|---|---|---|
| C0 | 0.43 | 0.54 |
| C1 | 0.85 | 0.86 |
| C2 | 0.94 | 0.90 |
| C3 | 0.94 | 0.88 |
| C4 | 0.93 | 0.86 |
| C6 | 0.85 | 0.81 |
| C8 | 0.48 | 0.50 |
| C10 | 0.49 | 0.68 |
| C12 | 0.42 | 0.62 |
| Parameter | Before Treatment (baseline) | After Treatment | Elevation (%) | Parameter | Before Treatment (baseline) | After Treatment | Elevation (%) |
|---|---|---|---|---|---|---|---|
| SBP (mm Hg) | 120.3 ± 1.3 | 140.3 ± 1.2 | 16.6 | Bilirubin (mg/dL) | 0.92 ± 0.18 | 1.6 ± 0.18 | 73% |
| DBP (mm Hg) | 80.5 ± 1.7 | 95.5 ± 1.1 | 18.7 | Creatinine (mg/dL) | 0.96 ± 0.10 | 1.6 ± 0.16 | 66% |
| TC (mg/dL) | 179 ± 21.7 | 254.9 ± 8.7 | 42.3 | Urea (mg/dL) | 34.4 ± 4.5 | 50.1 ± 5.4 | 45% |
| TG (mg/dL) | 135.4 ± 9.6 | 175.6 ± 9.8 | 29.7 | Uric acid (mg/dL) | 5.07 ± 0.95 | 8.5 ± 1.18 | 67% |
| AST (U/L) | 25 ± 8.1 | 36 ± 7.8 | 44 | K (mEq/L) | 4.1 ± 0.66 | 6.4 ± 0.55 | 56% |
| ALT (U/L) | 28 ± 11 | 44 ± 10 | 57 | Mg * (mg/dL) | 2.2 ± 0.30 | 0.9 ± 0.51 | 59%* |
| Adverse Effects | No. of Patients | % | Adverse Effects | No. of Patients | % |
|---|---|---|---|---|---|
| Nausea, bloating, loose stools, abdominal cramps | 21 | 35 | Tender or swollen gums | 45 | 75 |
| Slight trembling of the hands | 6 | 10 | Acne, oily skin | 14 | 23 |
| Tingling in the fingers, toes, lips | 3 | 5 | Fatigue | 28 | 46 |
| Muscle or joint discomfort, cramping | 9 | 15 | Edema in the legs or ankles | 5 | 8 |
| Sensitivity to heat and cold | 2 | 3 | Mild depression or mood swings | 44 | 73 |
| Mild headaches | 24 | 40 | Increased growth of fine hairs on the body | 33 | 55 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Abass, H.M.; Al-Tamimi, K.F.; Al-Tamimi, D.J.; Ibraheem, J.J. Correlation between Cyclosporine Blood Levels and Area under Blood Concentration Time Curve in Iraqi Bone Marrow Transplant Patients Treated with Neoral® Oral Solution. Sci. Pharm. 2020, 88, 12. https://doi.org/10.3390/scipharm88010012
Abass HM, Al-Tamimi KF, Al-Tamimi DJ, Ibraheem JJ. Correlation between Cyclosporine Blood Levels and Area under Blood Concentration Time Curve in Iraqi Bone Marrow Transplant Patients Treated with Neoral® Oral Solution. Scientia Pharmaceutica. 2020; 88(1):12. https://doi.org/10.3390/scipharm88010012
Chicago/Turabian StyleAbass, Hassan M., Kawther F. Al-Tamimi, Duaa J. Al-Tamimi, and Jaafar J. Ibraheem. 2020. "Correlation between Cyclosporine Blood Levels and Area under Blood Concentration Time Curve in Iraqi Bone Marrow Transplant Patients Treated with Neoral® Oral Solution" Scientia Pharmaceutica 88, no. 1: 12. https://doi.org/10.3390/scipharm88010012
APA StyleAbass, H. M., Al-Tamimi, K. F., Al-Tamimi, D. J., & Ibraheem, J. J. (2020). Correlation between Cyclosporine Blood Levels and Area under Blood Concentration Time Curve in Iraqi Bone Marrow Transplant Patients Treated with Neoral® Oral Solution. Scientia Pharmaceutica, 88(1), 12. https://doi.org/10.3390/scipharm88010012




