Abstract
Arabica coffee (Coffea arabica L.) ground nanoparticles contain phenolics compounds that have anti-inflammatory effects, so they can be used as sources of active compounds in anti-inflammatory gel preparations. This study aims to determine the optimum formulation of anti-inflammatory gel preparations using Arabica coffee ground nanoparticles as active compounds. Treatment optimization was performed using a Response Surface Methodology according to the Box-Behnken Design with a quadratic model in the Design Expert Version 10.0.3.0 software. In this study we used three factors (x): carbopol 940, triethanolamine (TEA), and nanoparticles, each of which consists of three levels, the response (y) observed including the acidity degree (pH), spreadability, viscosity and total phenolic content. ANOVA analysis results show that the quadratic model is very appropriate since it produces a high R2 value and a low PRESS value for all responses, as well as significant p-values (<0.0500) and an insignificant lack of Fit values (p-value> 5%). The optimum formulations for the gel preparations of the Arabica coffee ground nanoparticles obtained in this study are carbopol 940 (0.569%), TEA (0.468%), and nanoparticles (3.000%), which have values w/o an interval (0.994) and a desirable (0.981) response to acidity (5.212), spreadability (5.850 cm), viscosity (3734.244 cps) and total phenolic content (669.227 µgGAE/g).
1. Introduction
Nanotechnology is a new breakthrough in the world of technology, especially in the use of nano-sized materials [1] to produce extraordinary new properties. Nanotechnology has been widely developed in various fields including industry, agriculture [2], packaging [3], food and beverages [4], and health [5,6].
Nanotechnology is growing rapidly in the health sector, especially in the development of pharmaceutical preparations and medicines [7]. Nano technology has been used in recent years to produce nanoparticles as drug carriers [8], as well as active drug substances [9]. Nano-sized active drugs haves the ability to pass through cell walls and membranes to reach the target cell [5,10]. In addition, nano-sized particle also have the ability to dissolve and possess a high absorption efficiency, so materials at a nanoscale size have become very popular [6].
Arabica coffee ground nanoparticles are one example of the use of nano technology to produce materials used as active substances in pharmaceutical preparations. Arabica coffee grounds contain many bioactive compounds [11], such as alkaloids, phenolics [12] (flavonoids [13] and chlorogenic acid [14]), saponins, karatenoid [15], [16], and other active compounds [17], [18]. Arabica coffee grounds also contain polysaccharides [19] and oil [20]. Nurman et al. [21] presented GC-MS results from arabica coffee ground oil which showed the presence of compounds such as 1,2-benzenedicarboxylic acid, bis (2-etylhexyl) ester (18.09%), methylcyclopentane (14.93%), linoleic acid (9.00%), pentadecylic acid (8.81%), ethyl linoleate (6.36%), 2,3-dimethylbenzofuran (1.61%), cyclohexane (1.36%), and other compounds.
The secondary metabolite content of Arabica coffee ground nanoparticles can be used as an active ingredient in antioxidant and anti-inflammatory gel preparations [22,23]. Anti-inflammatory gel is a semi-solid preparation (for topical use), which contains active compounds that can provide anti-inflammatory effects [24,25]. To produce an optimal anti-inflammatory gel it is necessary to optimize the gel preparation formula. One of optimization methods used is the Response Surface Methodology.
The Response Surface Methodology is a set of mathematical methods and statistical techniques that are used to create a model and conduct an analysis of responses influenced by several factors to optimize the response [26]. One method for Response Surface is the Box-Behnken Design. The Box-Behnken Design is used in experiments with three levels of factorial design [27,28].
Based on the above description, we conducted this research to determine the optimum formulation of anti-inflammatory gel preparations using Arabica coffee ground nanoparticles as active compounds. Treatment optimization was performed using the Response Surface Methodology, according to the Box-Behnken Design, with a quadratic model in Design Expert Version 10.0.3.0 with three factors (x) and four responses (y).
2. Materials and Methods
2.1. Tools and Materials
The tools used in this study were analytical grade, baker glass, graduated cylinders, a volume pipette, and a mortar and pestle. The instruments used were a pH meter (pHep HI 98107 by HANNA Instrument, George Washington Hwy Smithfield, RI 02917, United States), Viscometer Brookfield (Dial Reading Viscometer Brookfield, Brookfield Engineering Labs., Inc.11 Commerce Boulevard, Middleboro, MA 02346, USA), particle size analyzer (DelsaTM Nano, Beckman Coulter, Inc. Indianapolis, IN 46268, USA), scanning electron microscope (QuantaTM 650, FEI, Hillsboro, OR, USA) and UV-Vis spectrophotometer (UV-1700 pharmaspec, Shimadzu Corporation, Kyoto, Japan).
The materials used in this study were carbopol 940, methylparaben, glyserin, triethanolamine (TEA) (Pharmaceutical Grade), aquadest and Arabica Coffee (Coffea arabica L.) ground nanoparticles.
2.2. Research Design
Optimization of the gel formulations was performed using the Box-Behnken Design because it can combine a 2k factorial with an incomplete blocking design [28], conducted with Software Design Expert Version 10.0.3.0, using three factors (x) and three levels (low, medium and high) as shown in Table 1. The Response surface quadratic model is used to interpret experimental data. Therefore, it can be obtained by the relationship between factors (x) and responses (y). The relationship between factors and responses is given by Equation (1):

Table 1.
Design level of Arabica coffee ground nanoparticles gel formulation with three factors (x) and three levels.
The quadratic relationship uses a higher polynominal degree approximation function called a second-order model, as given by Equation (2):
where x is the independent/factors variable, y is the responses variable, ε is the error responses, and β is the model variable.
2.3. Formulation of Gel Preparations
The carbopol 940 base was weighed and put into a beaker glass. Then, we added 100 mL of aquadest and stirred the solution using a magnetic stirrer at 80 °C for 30 min. The expanded carbopol base was put in a mortar, TEA, and 7.5 mL glycerin were added: they subtance was stirred evenly. Then, 0.1 g methylparaben was added and homogenized. Finally, Arabica coffee ground nanoparticles were added, crushed until homogeneous, and completely dispersed.
2.4. Characterization of Gel Preparations
The characterization and response (y) in the design of the experiments conducted on the gel preparations include an acidity degree (pH) test, a spreadability test, a viscosity test, and a total phenolic test.
2.4.1. Acidity Degree (pH) Test
We weighed a sample of 10 g, dissolved in aquadest to 100 mL, and stirred evenly. Then, the pH of the solution was measured using a pH meter.
2.4.2. Spreadability Test
The gel was weighed to be as high as 0.5 g and then placed on graph paper coated with glass. Then, we put another glass above the gel mass. The gel diameter was calculated by measuring the diameter length of several sides. Then we added an additional load of 150 g, allowed the mixture to stand for 1 min, and measured the diameter of the gel as before.
2.4.3. Viscosity Test
A maximum of 100 mL of gel was put into a container, and then placed on a viscometer with spindle no. 64 installed. Then, the spindle was lowered onto the gel to the specified limit. Next, we set the speed to 0.6 rpm and used the viscosity value shown on the tool.
2.4.4. Total Phenolic Test
Gallic acid was weighed to amaximum of 125 mg. Then, 96% ethanol was added up to 25 mL to obtain a 5000 mg/mL concentrated mother solution. From the main solution, 10 mL was pipetted and then diluted with 96% ethanol to 50 mL volume, so the second mother solution was obtained at a concentrate of 1000 mg/mL. The second mother solution was then pipetted 3, 4, 5, and 7 mL, and diluted with 96% ethanol to a volume of 10 mL. The resulting solution had concentrations of 300, 400, 500, and 700 mg/L. A total of 0.2 mL of each gallic acid solution at various concentrations was pipetted, and then we added 15.8 mL of distilled water and 1 mL of Folin-Ciocalteu reagent: the mixture was then shaken until homogeneous. The solution was allowed to stand for 8 min, and then we added 3 mL of 20% Na2CO3 solution, which was shaken homogeneously. The solution was then incubated for 2 h at room temperature. Uptake was measured by a UV-Vis spectrophotometer at a wavelength of 725 nm.
A total of 0.3 g of gel was weighed and then dissolved to 10 mL with 96% ethanol. A total of 0.2 mL of the solution was then pipetted, and we added 15.8 mL of distilled water and 1 mL of the Folin-Ciocalteu reagent; the solution was shaken until homogeneous. The solution was allowed to stand for 8 min, and then 3 mL of Na2CO3 20% of the solution was allowed to stand for 2 h at room temperature. Uptake was measured by a UV-Vis spectrophotometer at a wavelength of 725 nm.
3. Results and Discussion
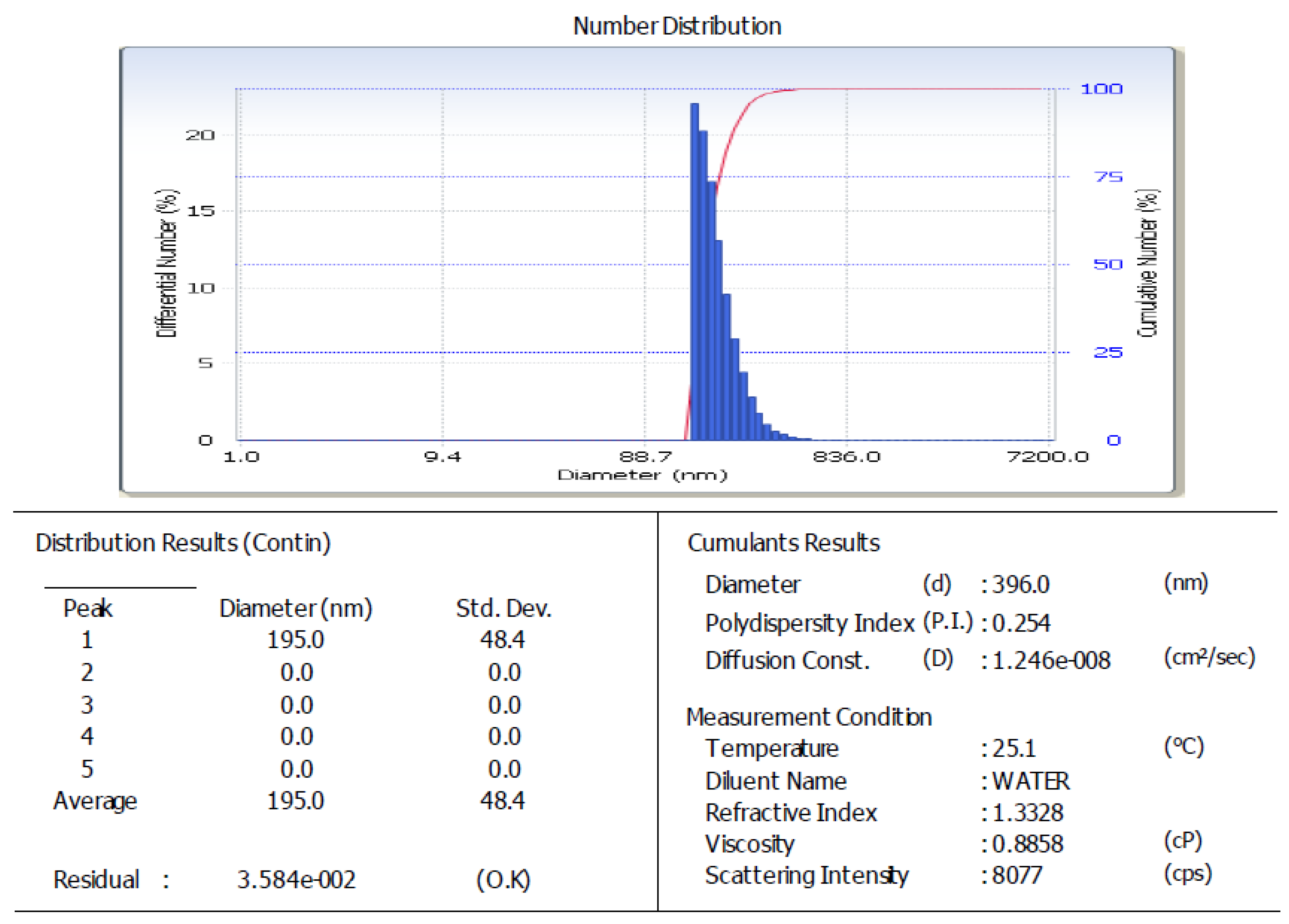
The arabica coffee ground nanoparticles used were residue from the processing of coffee drinks. The ultrasonication process, produced 396.0 nm nanoparticles (Figure 1), with 70.680% solubility and pH 5.33. The SEM analysis results for the arabica coffee ground nanoparticles can be seen in Figure 2. The secondary metabolite content of the arabica coffee ground nanoparticles are alkaloids, saponins, and phenolics, with a total phenolic content of 1246.897 µg GAE/g. Phenolic compounds are compounds providing antioxidant and anti-inflammatory effects [29,30,31,32,33,34]. A nano-sized active drug has the advantage of being able to pass through cell walls and membranes to reach the target cell. In addition, a nano-size also ensures high solubility and absorption efficiency [7,8,35].
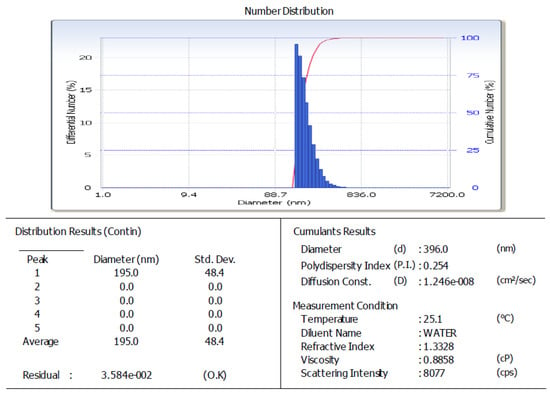
Figure 1.
Particle size analysis graphics of arabica coffee ground nanoparticles.

Figure 2.
Morphology SEM of Arabica coffee ground nanoparticles.
3.1. Prediction of Optimum Formulation
The optimum formulation of gel preparations using the Box-Behnken Design was performed using Design Expert Version 10.0.3.0 with three factors (x) on three levels and four responses. This analysis obtained 17 runs, as shown in Table 2. The design model used in this study was the qaudratic model because it obtained a high R2 value and a low PRESS value for all responses compared to the linear model, the 2FI model, and the cubic model. The value of R2 was expressed in %, which showed the contribution of the regression; The greater the value of R2, the greater the contribution or role of factor (x) to the response (y). An R2 value above 70% is considered sufficient [28]. Even though the cubic model has a high R2 value, it does not have a Pred-R2 value and a PRESS value, so the effect of each variable that has a signal difference is not necessary or aliased [36]. An analysis of the variance of the designed models in this study is shown in Table 3.

Table 2.
Formulation design and dan characterization results of arabica coffee ground nanoparticles gel formulation with three factors (x) and three levels.

Table 3.
Characterization for the statistical design model of arabica coffee ground nanoparticles gel formulation.
3.2. Organoleptic
Organoleptic gel preparations of Arabica coffee ground nanoparticles were carried out by visually observing their shape, color, odor, and homogeneity. Overall, the arabica coffee ground nanoparticles gel preparations have a semisolid (gel) shape, a brownish black color (typical of coffee grounds), and homogeneous. These physical properties were more dominant due to the active substance added: arabica coffee ground nanoparticles.
3.3. Acidity Degree
The acidity degree (pH) is a very important parameter in gel preparations since gel is a topical preparation used on the skin. Therefore, gel preparations must have a pH similar to that of human skin (4.5–6.5) [37] to avoid irritation or erythema on the skin. This test shows that the pH of the gel preparation ranges from 5.20 to 6.47, which is still in the human skin’s pH range (Table 2).
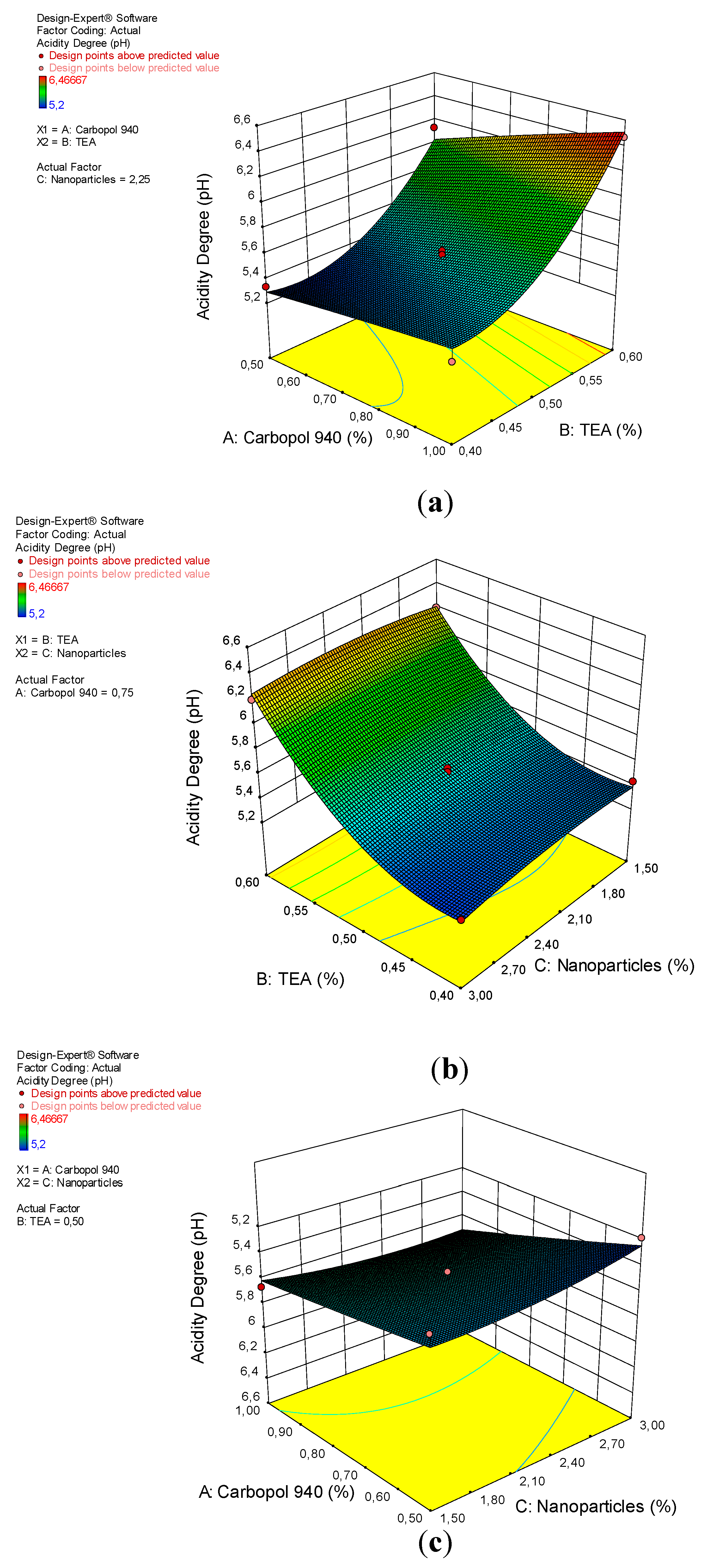
The quadratic design model has a significant effect on the acidity degree of gel preparations, with a p-value of 0.0001 (less than 0.0500), as shown in Table 4. Significant factors on acidity are A, B, and B2 because they have p-values, respectively, of 0.0016; <0.0001, and 0.0005. The insignificance of the “lack of fit” with an F-value of 6.41 and a p-value of 5.23%, indicates that the quadratic design model is appropriate for analyzing the acidity test data, but this model has a low probability because its p-value is less than 10%. The relationship between the degree of acidity of the gel preparation with a factor (x) based on the coefficient value can be seen in Equation (3).
y = 5.55 + 0.16A + 0.45B − 0.017C + 0.075AB +0.067AC + 0.033BC − 5.833E-003A 2 + 0.28B 2 − 0.048C 2

Table 4.
ANOVA analysis for the quadratic model of the acidity degree of arabica coffee ground nanoparticles gel preparations.
The degree of acidity increases with an increasing concentration of TEA used in accordance with a p-value of <0.0001, as shown in Figure 3. TEA is difficult to evaporate at room temperature, has an ammonia odor, and can form a solid or liquid depending on its temperature and the value of its purity. Its relatively basic nature means that a TEA with a pH of 10.5 can be used as a basic agent and also as an emulsifying agent [38]. However carbopol 940 has a pH of 7.7 and a nanoparticle has a pH of 5.33, which does not have a significant effect on the acidity level of the arabica coffee ground nanoparticles gel preparations.
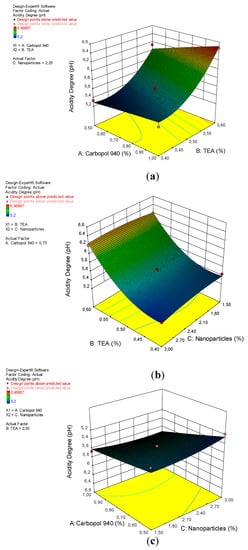
Figure 3.
A 3D plot of the relationship between (a) carbopol 940 and TEA and the acidity degree, (b) TEA and nanoparticles and the acidity degree, and (c) carbopol 940 and nanoparticles and the acidity degree.
3.4. Spreadibility
The spreadability of gel preparations is defined as the ability of the gel to be spread on the surface of the skin [39]. The greater the scatter diameter, the greater the surface area that can be reached by the gel. Good spreadability can guarantee the distribution of a gel when applied to the skin, good spreadability ranges from 5–7 cm [37]. The spread test results of the arabica coffee ground nanoparticles gel preparations reveal a value between 5.10–6.43 cm (Table 2), which indicates that the gel has a good spreadability.
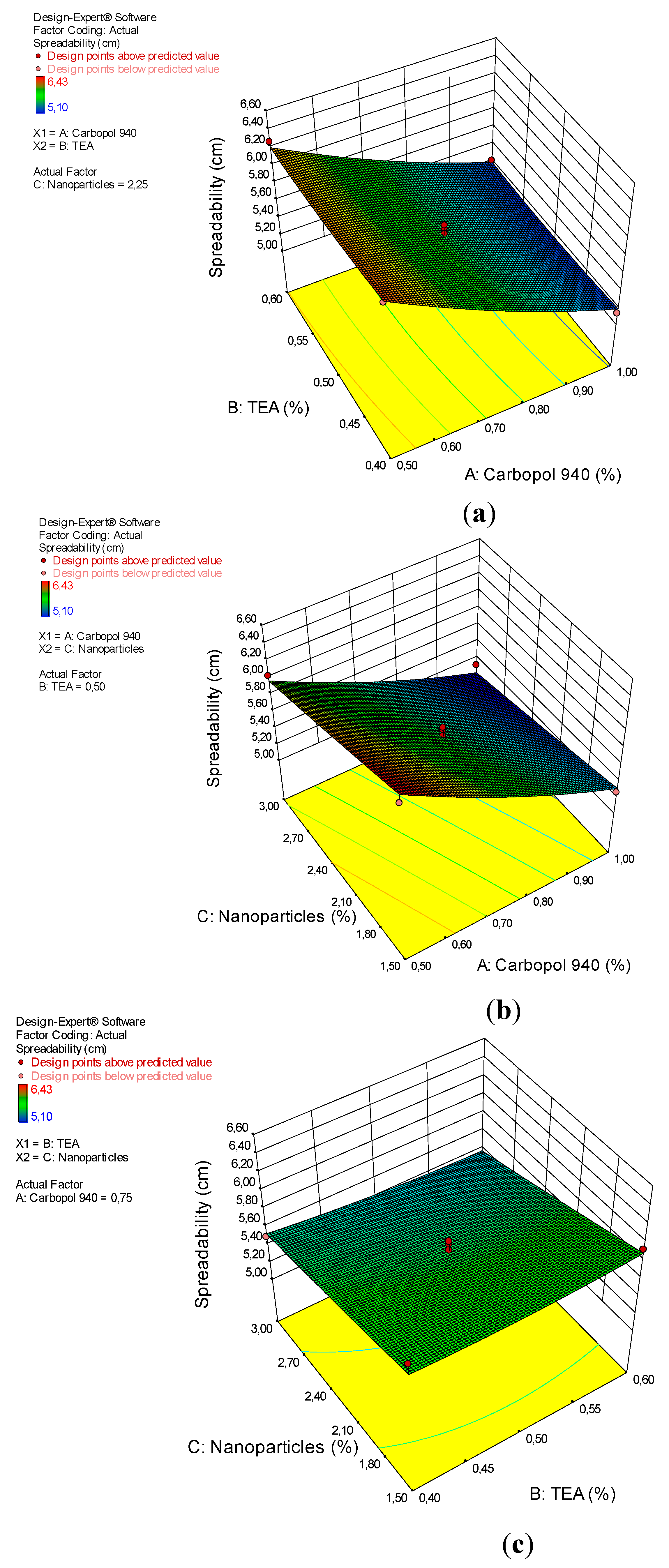
The results of the ANOVA analysis for the quadratic model of the spreadability of the arabica coffee ground nanoparticles gel preparations are shown in Table 5. This model shows significant results, with a p-value of 0.0002. Significant factors for spreadability are A and C, with p-values of <0.0001 and 0.0011, respectively. The “lack of fit” value is not significant, with an F-value of 2.39 and a p-value of 20.96%. This result shows that the quadratic design model is very appropriate for analyzing the spreadability test data. The interactions between the gel dispersion power and the factor (x) can be seen by the coefficent value, as shown in Equation (4).
y = 5.63 − 0.55A − 0.037B − 0.22C + 0.033AB + 0.058AC − 0.042BC + 0.067A 2 + 0.033B 2 + 8.333E-003C 2

Table 5.
ANOVA analysis for the quadratic model of the spreadability of the arabica coffee ground nanoparticles gel preparations.
Carbopol 940, used as a thickener, surfactant, stabilizer, or thickener [38], plays an important role in determining the spreadability of the gel preparations [37]. This effect is clearly seen in the results of the ANOVA analysis and the 3D plots in Figure 4. From the picture, it can be seen that carbopol 940 has an effect on spreadability. The greater the concentration of carbopol 940, the smaller the spreadability of the gel preparations, while the TEA and nanoparticles had no effect on spreadability.
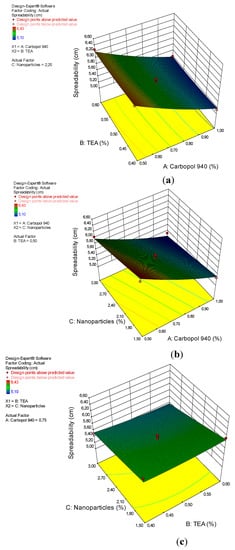
Figure 4.
3D plot of the relationship between (a) carbopol 940 and TEA and the spreadability, (b) carbopol 940 and nanoparticles and the spreadability, and (c) TEA and nanoparticles and the spreadability.
3.5. Viscosity
Viscosity is a measure of the thickness of a fluid; gel preparation refers to fluids that have high a viscosity of 2000–4000 cps [39]. The viscosity test results for the gel preparations ranged from 3405.97–4604.96 cps (Table 2). Viscosity is inversely proportional to spreadability; the greater the viscosity, the smaller the spreadability.
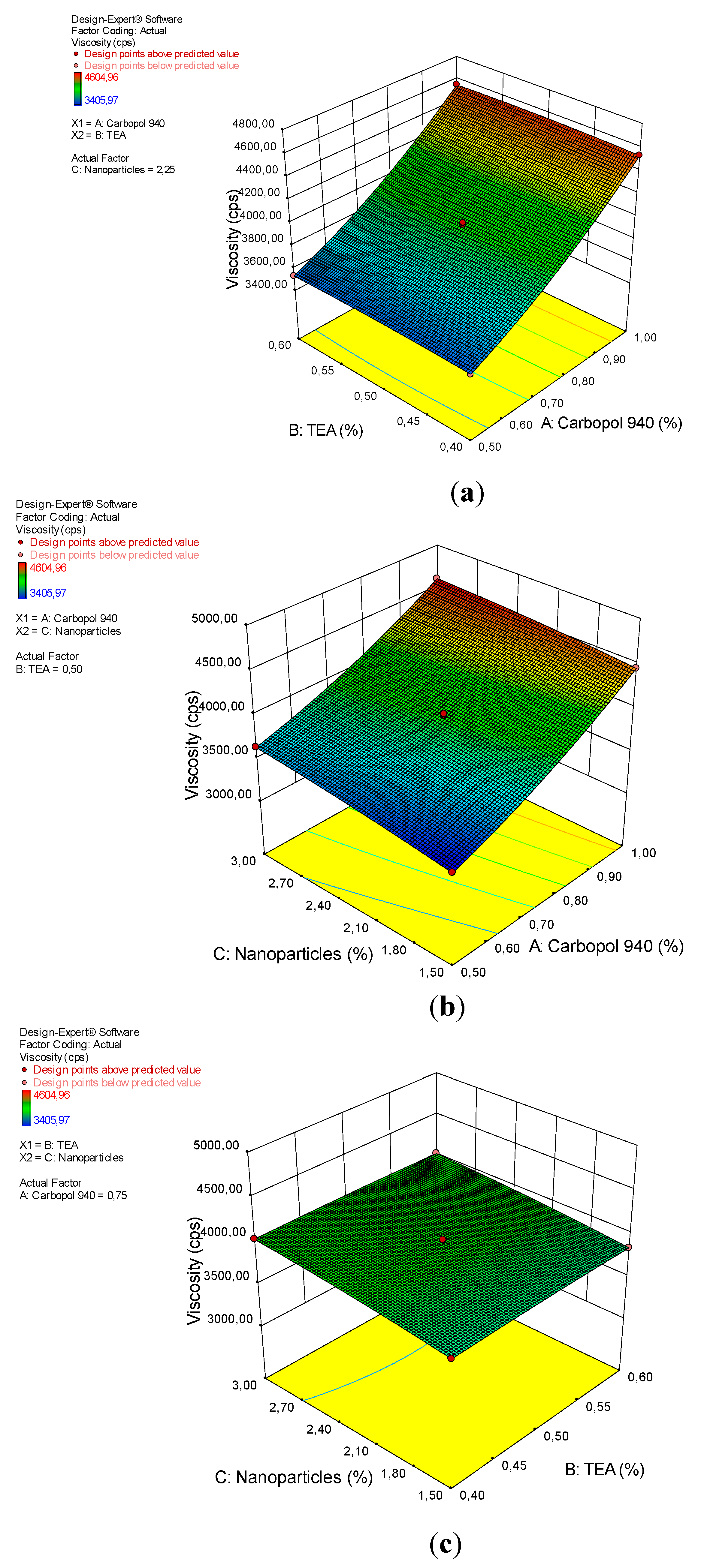
Table 6 shows the ANOVA analysis for the quadratic model of the viscosity of the arabica coffee ground nanoparticles gel preparations. The model shows significant results, with p-values <0.0001. The significant factors on viscosity are A, C, AC, A2, and C2, with p-values of <0.0001, <0.0001, 0.0113, <0.0001, and 0.0132. The “lack of fit” value shows insignificant results, with an F-value of 0.058 and p-value of 97.93%. These results demonstrate that the quadratic design model is very appropriate for the analysis of the viscosity test data. The interaction between the viscosity of the gel preparation and factor (x) can be seen from the coefficient value, as shown in Equation (5).
y = 3965.01 + 505.12A + 4.73B + 97.22C + 1.35AB − 21.48AC − 7.17BC + 82.16A 2 − 12.27B 2 − 20.23C 2

Table 6.
ANOVA analysis of quadratic model for the viscosity of the arabica coffee ground nanoparticles gel preparations.
Viscosity increases by increasing the concentrations of carbopol 940 and arabica coffee ground nanoparticles; However, increasing the TEA did not affect the viscosity of the gel preparations, as seen in the 3D plot (Figure 5). These results is consistent with the results of the ANOVA analysis, which showed that carbopol 940 and the nanoparticles had a significant effect on gel viscosity, with an indigo p-value <0.0001. Carbopol 940 is a synthetic polymer of acrylic acid, which is hygroscopic, slightly acidic and very easily ionized. Carbopol 940 used as a gelling agent plays an important role in regulating the viscosity of the gel preparations [38]. In acidic solutions (pH 3.5–4.0), the carbopol 940 dispersion shows low to medium viscosity, pH 5.0–10.0, and at temperatures above 75 °C, shows optimal viscosity.
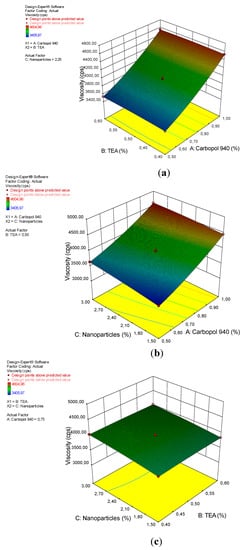
Figure 5.
A 3D plot of the relationship between (a) carbopol 940 and TEA and the viscosity, (b) carbopol 940 and nanoparticles and the viscosity, and (c) TEA and nanoparticles and the viscosity.
3.6. Total Phenolic
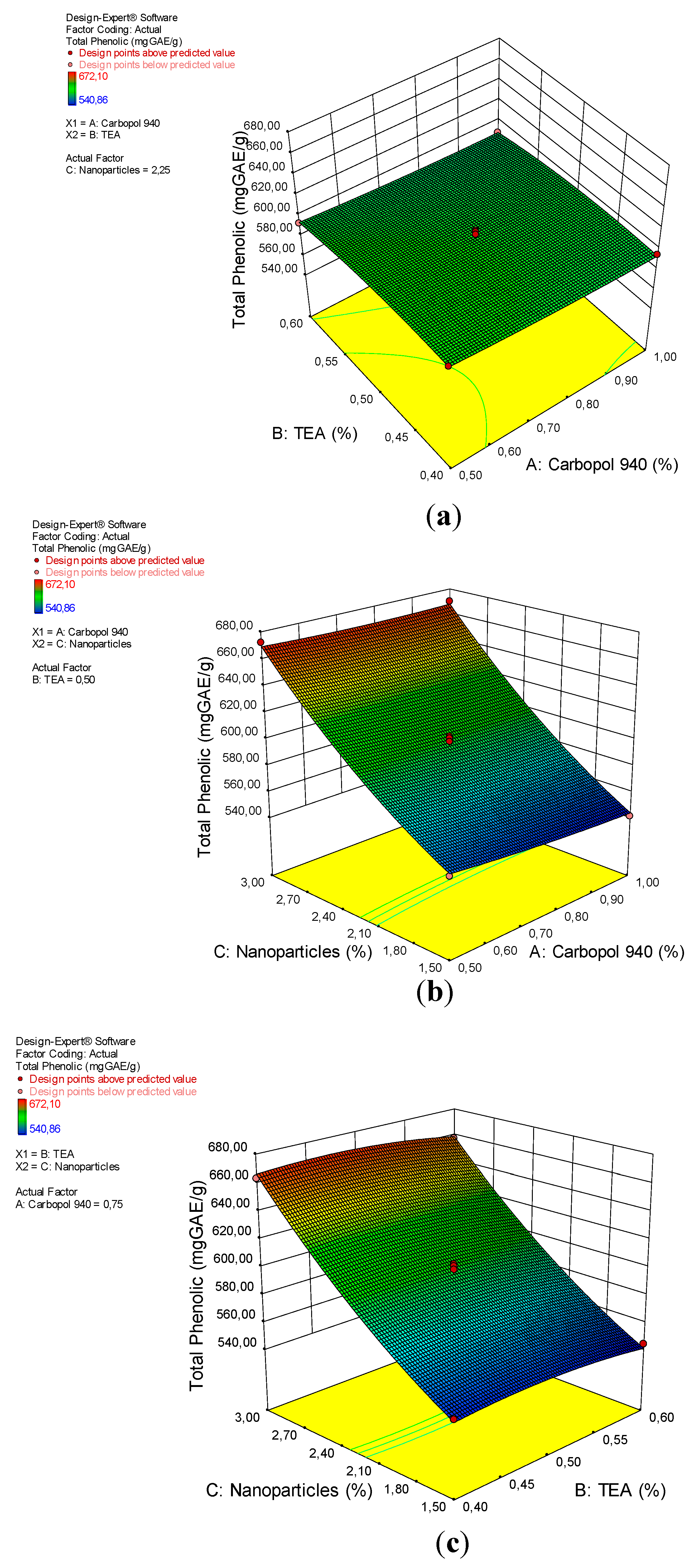
The total phenolic test used UV-Vis spectrophotometry at a wavelength of 725 nm. The total phenolic content was expressed as its gallic acid equivalent (GAE). The GAE is a reference for the total phenolic analysis contained in a phenolic substance [40]. Arabica coffee ground nanoparticles have a phenolic content of 1246.897 µg GAE/g. The phenolic compounds in coffee have some bioactivities, such as antioxidant, antibacterial and anti-inflammatory activities [29,30,32,33,41,42,43]. The total phenolic dosage of the coffee ground nanoparticles gel preparations ranged from 540.86 to 672.10 µg GAE/g (Table 2).
The ANOVA analysis for the quadratic model of the total phenolic content of the arabica coffee ground nanoparticles gel preparations is shown in Table 7. The analysis shows that the quadratic model is appropriate for the analysis of the total phenolic test data, with a p-value <0.0001 (significant). Significance factors for the total phenolic content were C and C2, with p-values <0.0001 and 0.0032. The “lack of fit” is not significant, with an F-value of 0.73 and a p-value of 58.62%. The interaction between the total phenolic gel preparation with a factor (x) can be seen from the coefficient value, as shown in Equation (6).

Table 7.
ANOVA analysis for the quadratic model of the total phenolic content of the arabica coffee ground nanoparticles gel preparations.
Figure 6 shows the relationship between the factors and the total phenolic content of arabica coffee ground nanoparticles gel preparations. The figure shows that an increase in the total phenolic content is influenced by the concentration of nanoparticles, whereas carbopol 940 and TEA have no effect on total phenolic content. This result is consistent with the p-value in the ANOVA analysis.
y = 597.96 − 3.40A − 3.28B + 59.43C + 0.52AB + 2.64AC − 0.46BC +1.54A 2 − 4.55B 2 + 9.62C 2

Figure 6.
A 3D plot of the relationship between (a) carbopol 940 and TEA and the total phenolic content, (b) carbopol 940 and nanoparticles and the total phenolic content, and (c) TEA and nanoparticles and the total phenolic content.
3.7. Optimization of Gel Preparations
The optimization results using the Box-Behnken Design provides 17 formulation solutions for arabica coffee ground nanoparticles gel formulations that can be selected to produce the optimal response shown in Table 8. Solution number 8 was chosen as the prediction for the optimum conditions because it has a high value w/o interval (0.994) and a high desirability (0.981). Other solutions, though they have the same w/o interval and desirability values, they produced lower total phenolic content. Therefore, the optimum arabica coffee ground nanoparticles gel formulations are carbopol 940 (0.569%), TEA (0.468%), and nanoparticles (3.000%) that produce a response in the acidity degree (5.212), spreadability (5.850 cm), viscosity (3734.244 cps), and total phenolic (669.227 µg GAE/g).

Table 8.
The solution of optimum formulation arabica coffee ground nanoparticles gel preparations.
4. Conclusions
Arabica coffee (Coffea arabica L.) ground nanoparticles can be used as a source of active compounds in anti-inflammatory gel preparations since it has a high total phenolic content. The quadratic model in the Box-Behnken Design was used to produce the optimum formulation of an arabica coffee ground nanoparticles gel, with a composition of: carbopol 940 (0.569%), TEA (0.468%), and nanoparticles (3.000%), which produce an acidity degree of (5.212), a spreadability of (5.850 cm), viscosity of (3734.244 cps), and total phenolic content of (669.227 µg GAE/g).
Author Contributions
Conceptualization, S.N.; Methodology, R.Y.; Project administration, I.; Supervision, E.N. and T.C.S.
Funding
This study has been financially supported by the Directorate of Research and Community Services Directorate General of Strengthening Research and Development, Ministry of Research, Technology and Higher Education, No. 235/SP2H/LT/DRPM/2019.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Mohanraj, V.J.; Chen, Y. Nanoparticles – A Review. Trop. J. Pharm. Res. 2006, 5, 561–573. [Google Scholar] [CrossRef]
- Ariningsih, E. Prospects of Nanotechnology Application in Agriculture and Food Processing in Indonesia. Forum Penelit. Agro Ekon. 2016, 34, 1–20. [Google Scholar] [CrossRef]
- Sudibyo, A.; Hutajulu, T.F. Potensi penerapan polimer nanokomposit dalam kemasan pangan. J. Kim. dan Kemasan 2013, 35, 6–19. [Google Scholar] [CrossRef]
- Berekaa, M.M. Review Article Nanotechnology in Food Industry ; Advances in Food processing, Packaging and Food Safety. Int. J. Curr. Microbiol. Appl. Sci. 2015, 4, 345–357. [Google Scholar]
- Liu, Y.; Niu, T.; Zhang, L.; Yang, J. Review on nano-drugs. Nat. Sci. 2010, 2, 41–48. [Google Scholar] [CrossRef]
- Astruc, D. Introduction to Nanomedicine. Molecule 2016, 21, 4. [Google Scholar] [CrossRef]
- Rizvi, S.A.A.; Saleh, A.M. Applications of Nanoparticle Systems in Drug Delivery Technology. Saudi Pharm. J. 2018, 26, 64–70. [Google Scholar] [CrossRef]
- Sawasdee, K.; Choksawad, P.; Pimcharoen, S.; Prapainop, K. Development of Size-Tunable Polymeric Nanoparticles for Drug Delivery Applications. Glob. Heal. Manag. J. 2017, 1, 31–36. [Google Scholar] [CrossRef]
- Prasetyo, Y.A.; Abdassah, M.; Rusdiana, T. Preparation and Characterization of Glucosamine Nanoparticle by Ionic Gelation Method Using Chitosan and Alginate. Indones. J. Pharm. 2019, 1, 1–10. [Google Scholar] [CrossRef]
- Hanutami, N.B.; Budiman, A. Review Artikel : Penggunaan Teknologi Nano pada Formulasi Obat Herbal. Farmaka 2017, 15, 29–41. [Google Scholar]
- Cruz, R.; Gomes, T.; Ferreie, A.; Mendes, E.; Baptista, P.; Cunha, S.; Pereira, J.A.; Ramalhosa, E.; Casal, S. Antioxidant activity and bioactive compounds of lettuce improved by espresso coffee residues. Food Chem. 2014, 145, 95–101. [Google Scholar] [CrossRef] [PubMed]
- Sant’Anna, V.; Biondo, E.; Kolchinski, E.M.; da Silva, L.F.S.; Correa, A.P.F.; Bach, E.; Brandelli, A. Total Polyphenols, Antioxidant, Antimicrobial and Allelopathic Activities of Spend Coffee Ground Aqueous Extract. Waste and Biomass Valorization 2017, 8, 439–442. [Google Scholar] [CrossRef]
- Yoo, D.E.; Jeong, K.M.; Han, S.Y.; Kim, E.M.; Jin, Y.; Lee, J. Deep eutectic solvent-based valorization of spent co ff ee grounds. Food Chem. 2018, 255, 357–364. [Google Scholar] [CrossRef] [PubMed]
- Jeon, J.; Kim, H.; Jeong, I.; Hong, S.; Oh, M.; Yoon, M. Contents of chlorogenic acids and caffeine in various coffee-related products. J. Adv. Res. 2019, 17, 85–94. [Google Scholar] [CrossRef] [PubMed]
- Petrik, S.; Obruča, S.; Benešová, P.; Márová, I. Bioconversion of spent coffee grounds into carotenoids and other valuable metabolites by selected red yeast strains. Biochem. Eng. J. 2014, 90, 307–315. [Google Scholar] [CrossRef]
- Obruca, S.; Benesova, P.; Kucera, D.; Petrik, S.; Marova, I. Biotechnological conversion of spent coffee grounds into polyhydroxyalkanoates and carotenoids. N. Biotechnol. 2015, 32, 569–574. [Google Scholar] [CrossRef]
- Page, J.C.; Arruda, N.P.; Freitas, S.P. Crude ethanolic extract from spent coffee grounds : Volatile and functional properties. Waste Manag. 2017, 69, 463–469. [Google Scholar] [CrossRef]
- Passos, C.P.; Moreira, A.S.P.; Domingues, M.R.M.; Evtuguin, D.V.; Coimbra, M.A. Sequential microwave superheated water extraction of mannans from spent coffee grounds. Carbohydr. Polym. 2014, 103, 333–338. [Google Scholar] [CrossRef]
- Passos, C.P.; Coimbra, M.A. Microwave superheated water extraction of polysaccharides from spent coffee grounds. Carbohydr. Polym. 2013, 94, 626–633. [Google Scholar] [CrossRef]
- Tuntiwiwattanapun, N.; Tongcumpou, C. Industrial Crops & Products Sequential extraction and reactive extraction processing of spent co ff ee grounds : An alternative approach for pretreatment of biodiesel feedstocks and biodiesel production. Ind. Crop. Prod. 2018, 117, 359–365. [Google Scholar]
- Nurman, S.; Lamona, A.; Nasir, M. Optimization and Characterization of N-Hexane Extracts of Arabica Coffee Ground ( Coffea arabica L.) from Gayo Plateau as Source of Natural Antioxidant. In Proceedings of the International Conference on Education, Science and Technology (ICON-EST 2018) at Serambi Mekkah University, Banda Aceh, Indonesia, 17–18th October 2018; 2019; pp. 1–9. [Google Scholar]
- Palupi, N.W.; Praptiningsih, Y. Oxidized Tapioca Starch As an Alginate Substitute for Encapsulation of Antioxidant from Coffee Residue. Agric. Agric. Sci. Procedia 2016, 9, 304–308. [Google Scholar] [CrossRef]
- Cheong, M.W.; Tong, K.H.; Ong, J.J.M.; Liu, S.Q.; Curran, P.; Yu, B. Volatile composition and antioxidant capacity of Arabica coffee. Food Res. Int. 2013, 51, 388–396. [Google Scholar] [CrossRef]
- Kasman, B.B.S.; Saputri, F.A. Artikel Review: Formulasi dan Evaluasi Sediaan Gel dari Ekstrak Seledri Apium graveolens Linn Sebagai Anti-Inflamasi. Farmaka 2018, 16, 63–71. [Google Scholar]
- Ulfa, M.; Hendrarti, W.; Muhram, P.N. Formulasi Gel Ekstrak Daun Kelor ( Moringa oleifera Lam.) Sebagai Anti Inflamasi Topikal Pada Tikus ( Rattus novergicus ). J. Pharm. Med. Sci. 2016, 1, 30–35. [Google Scholar]
- Oramahi, H. Optimasi dengan RSM dan Rancangan Percobaan (Aplikasi dengan SPSS dan SAS), 1st ed.; Gava Media: Yogyakarta, Indonesia, 2016. [Google Scholar]
- Getachew, A.T.; Cho, Y.J.; Chun, B.S. Effect of pretreatments on isolation of bioactive polysaccharides from spent coffee grounds using subcritical water. Int. J. Biol. Macromol. 2018, 109, 711–719. [Google Scholar] [CrossRef]
- Tekindal, M.A.; Bayrak, H.; Ozkaya, B.; Genc, Y. Box- Behnken Experimental Design in Factorial Experiments: The Importance of Bread For Nutrition and Health. Turkish J. F. Crop. 2012, 17, 115–123. [Google Scholar]
- Megha, M.A.; Unnma, U.; Rameshpathy, M.; Karikalan, K.; Vickram, S.; Kumar, S.V.; Sridharan, B. Formulation of nano-encapsulated poly-herbal ointment for anti- inflammation. Der Pharm. Lett. 2013, 5, 164–170. [Google Scholar]
- Ruiz-ruiz, J.C.; Matus-basto, A.J.; Acereto-escoffié, P.; Segura-Campos, M.R. Antioxidant and anti-inflammatory activities of phenolic compounds isolated from Melipona beecheii honey. Food Agric. Immunol. 2017, 28, 1424–1437. [Google Scholar] [CrossRef]
- Shahidi, F.; Yeo, J. Bioactivities of Phenolics by Focusing on Suppression of Chronic Diseases : A Review. Int. J. Mol. Sci. 2018, 19, 1573. [Google Scholar] [CrossRef]
- Lamona, A.; Nurman, S. Studi Awal Komponen Minyak Biji Kopi Arabika ( Coffea arabica L.) dari Kabupaten Bener Meriah, Provinsi Aeh Menggunakan GC-MS. Agriovet 2018, 1, 61–72. [Google Scholar]
- Nurman, S.; Yulia, R. The Potency of Gayo Arabica Coffee Powder Oil ( Coffea arabica L.) as a Source of Natural Antioxidants. J. Food, Agric. Environ. 2019, 17, 59–64. [Google Scholar]
- Złotek, U.; Szymanowska, U.; Pecio, L.; Kozachok, S.; Jakubczyk, A. Antioxidative and Potentially Anti-inflammatory Activity of Phenolics from Lovage Leaves Levisticum officinale Koch Elicited with Jasmonic Acid and Yeast Extract. Molecules 2019, 24, 2–12. [Google Scholar]
- Irianto, H.E.; Muljanah, I. Proses dan Aplikasi Nanopartikel Kitosan Sebagai Penghantar Obat. Squalen 2011, 6, 1–8. [Google Scholar]
- Yulia, R.; Meilina, H.; Adisalamun, A.; Darmadi, D. Aplikasi Metode Advance Oxidation Process (AOP) Fenton pada Pengolahan Limbah Cair Pabrik Kelapa Sawit Application of Advanced Oxidation Process (AOP) Fenton on Palm Oil Mill Effluent Treatment. J. Rekayasa Kim. dan Lingkung. 2016, 11, 1–9. [Google Scholar] [CrossRef][Green Version]
- Saryanti, D.; Zulfa, I.N. Optimization Carbopol And Glycerol As Basis Of Hand Gel Antiseptics Extract Ethanol Ceremai Leaf (Phyllantus Acidus (L.) Skeels) With Simplex Lattice Design. J. Pharm. Sci. Clin. Res. 2017, 02, 35–43. [Google Scholar] [CrossRef]
- Rowe, R.C.; Sheskey, P.J.; Quinn, M.E. Handbook of Pharmaceutical Excipients, 6th ed.; Pharmaceutical Press and American Pharmacists Association: London, UK; Washington, DC, USA, 2009. [Google Scholar]
- Danimayostu, A.A.; Shofiana, N.M.; Permatasari, D. Pengaruh Penggunaan Pati Kentang (Solanum tuberosum) Termodifikasi Asetilasi- Oksidasi sebagai Gelling agent terhadap Stabilitas Gel Natrium Diklofenak. Phamaceutical J. Indones. 2017, 3, 25–32. [Google Scholar] [CrossRef]
- Pristiana, D.Y.; Susanti, S.; Nurwantoro, N. Antioksidan dan Kadar Fenol Berbagai Ekstrak Daun Kopi (Coffea sp.): Potensi Aplikasi Bahan Alami untuk Fortifikasi Pangan. J. Apl. Teknol. Pangan 2017, 6, 89–92. [Google Scholar]
- Gómez-Ruiz, J.Á.; Ames, J.M.; Leake, D.S. Antioxidant activity and protective effects of green and dark coffee components against human low density lipoprotein oxidation. Eur. Food Res. Technol. 2008, 227, 1017–1024. [Google Scholar] [CrossRef]
- García, R.L.P.; Biasetto, C.R.; Araujo, A.R.; Luiz, V.; Bianchi, D. Enhanced Extraction of Phenolic Compounds from Coffee Industry’s Residues Through Solid State Fermentation by Penicillium Purpurogenum. Food Sci. Technol. 2015, 35, 704–711. [Google Scholar] [CrossRef]
- Shang, Y.F.; Xu, J.L.; Lee, W.J.; Um, B.H. Antioxidative Polyphenolics Obtained from Spent Coffee Grounds by Pressurized Liquid Extraction. South African J. Bot. 2017, 109, 75–80. [Google Scholar] [CrossRef]
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).