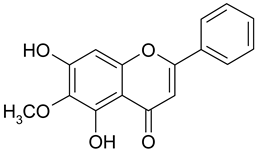
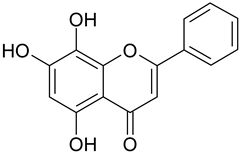
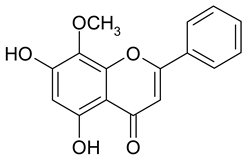
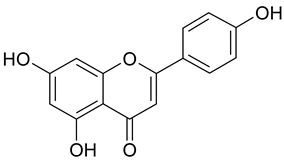
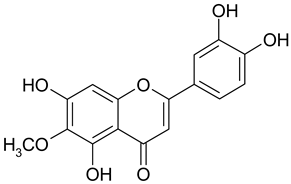
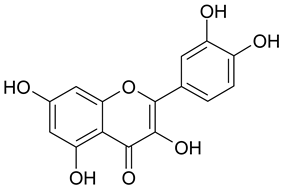
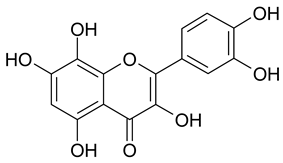
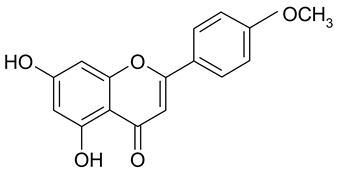
Abstract
We investigated the effect of 21 flavonoids in a three-dimensional in vitro system for their ability to inhibit gap formation by MCF-7 breast cancer spheroids in monolayers of lymphendothelial cells. Different representatives of the classes of flavones, flavonols, and flavanones were tested in the circular chemorepellent-induced defects (CCID)-assay. Bay11-7082, a known inhibitor of CCID formation served as the positive control. This study provides the first comparison of the potential of flavonoids to suppress features influencing the intravasation of MCF-7 breast cancer cells aggregates through the lymph endothelial barrier. The most significant effects were seen after incubation with the flavones luteolin, chrysin, and apigenin. Additional hydroxylation or methoxylation in positions 6 or 8, as expected, resulted in decreased activity. The tested flavanones remained without or low efficacy.
1. Introduction
Intravasation through the lymphatic border is an early- and rate-limiting step in the metastatic cascade of breast cancer [1,2,3]. Yet, hardly any anti-metastatic drugs are available. As from 1981 to 2014 approximately 83% of the approved small molecule anticancer drugs were natural products, based on natural compounds or mimicking them in different ways [4]. We tested traditional medicinal plants regarding their anti-intravasative properties [5,6,7,8]. In a study on the effects of different Scrophularia extracts on breast cancer cells´ proliferation, cell death, and intravasation through lymph endothelial cell barriers, a methanolic extract of Scrophularia lucida L. (Scrophulariaceae) proved particularly active in inhibiting intravasation in vitro [8]. An activity-guided fractionation of this extract revealed hispidulin, a member of the class of flavonoids, as significantly contributing to this activity [9].
Because of the multitude of structures and amounts of flavonoids in human nutrition, these compounds have gained a lot of interest for the prevention, deceleration of development, or even alleviation of different “Western Diseases” such as cardiovascular or neurodegenerative disorders as well as various cancers. For flavonoids in cancer chemoprevention numerous modes of action, modulation of a multiplicity of proteins and inhibition of many enzymes involved in cancer pathology have been shown [10]. Among those, different mechanisms involving many steps in the “metastatic cascade” influencing cell detachment, cell mobility, tissue barriers for intravasation and extravasation, angiogenesis etc., were studied in detail and many molecular targets involved in the anti-invasive and anti-metastatic properties of flavonoids such as luteolin, apigenin, kaempferol among others have been identified [11].
Nevertheless, for an activity-guided search for effective natural anticancer compounds (e.g., flavonoids) inhibiting mechanisms of metastasis, robust in vitro tools simulating the in vivo situation are necessary. For this purpose 3D multicellular tumor spheroids as used in the validated “circular chemorepellent-induced defect” (CCID) assay provide an excellent possibility [12]. Until now, data of the efficacy of flavonoids in this assay are scarce and only few flavonoids have been tested on breast cancer intravasation [1,3,9,13,14,15,16]. Yet, the IC50 of hispidulin was too high to be reached in vivo [9]. Therefore, to gain insight into structural features influencing the anti-intravasative potential of flavonoids, we compared hispidulin to 20 other structurally closely related flavonoids in this 3D assay consisting of MCF-7 cell spheroids and lymph endothelial cell (LEC) monolayers.
2. Materials and Methods
2.1. Materials
Apigenin, naringenin, hispidulin, scutellarein, baicalein, quercetin, wogonin, and diosmetin were purchased from Sigma-Aldrich (Vienna, Austria); nepetin, oroxylin A from Phytolab (Vestenbergsgreuth, Germany); kaempferol, acacetin, galangin, chrysin, homoeriodictyol from Carl Roth GmbH (Karlsruhe, Germany); herbacetin, pinocembrin, luteolin, eriodictyol, norwogonin from Extrasynthése (Genay, France); and gossypetin from Indofine Chemical Company (Hillsborough, UK). The I-κBα phosphorylation inhibitor (E)-3-[4-methylphenylsulfonyl]2-propenenitrile (Bay11-7082) was obtained from Biomol (Hamburg, Germany). Stock solutions were prepared in dimethylsulfoxide (DMSO; 99.7% cell culture grade, Sigma-Aldrich (Vienna, Austria)).
2.2. Cell Culture
Human microvessel endothelial cells were purchased from Clonetics (Basel, Switzerland), immortalized with human telomerase cDNA (TERT1) [17], and subsequently lymph endothelial cell population was isolated by single cell cloning [18]. For this work, LECs were authenticated by analyzing expression of the lymph endothelial cell markers PROX1, LYVE-1, and podoplanin by immunofluorescence and laser scanning microscopy (Supplemental Figure S1). LECs were cultivated in microvascular endothelial cell growth medium-2 (EGM2 MV) (Clonetics CC-4147; Lonza Group, Ltd., Basel, Switzerland) [1,8,18]. The human MCF-7 cell line was obtained from ATCC (Rockville, MD, USA). Cells were grown in minimum essential medium (MEM) supplemented with 10% fetal calf serum, 1% penicillin/streptomycin, and 1% non-essential amino acids (all from Life Technologies, Lofer, Austria). All cells were grown at 37 °C in a humidified atmosphere containing 5% CO2.
2.3. 3D Co-Cultivation of MCF-7 Cancer Cells with LECs
MCF-7 cells were transferred to 30 mL MEM medium containing 6 mL of 1.6% methylcellulose solution (0.3% final concentration; cat. no. M-512, 4000 centipoises; Sigma, Munich, Germany). A total of 150 μL of the cell suspension, each, were then transferred to the wells of a 96-well plate (Greiner Bio-one, Cellstar 650185, Kremsmünster, Austria) for spheroid formation within the following two days. The developed MCF-7 spheroids were washed in phosphate buffered saline and transferred to cytotracker-stained (Invitrogen; Thermo Fisher Scientific, Inc., Carlsbad, CA, USA) LEC monolayers that had been seeded into 24-well plates (Costar 3524, Sigma, Vienna, Austria) in 2 mL EGM2 MV medium (Kerjaschki et al., 2011; Vonach et al., 2011).
2.4. Circular Chemo-Repellent Induced Defect (CCID) Assay
After transfer of the MCF-7 cell spheroids (3000 cells/spheroid) to the 24-well plate containing LEC monolayers, four hours of incubation of the MCF-7 spheroids-LEC monolayer co-cultures followed. The CCID sizes in the LEC monolayer underneath the MCF-7 spheroids were photographed with an Axiovert (Zeiss, Jena, Germany) fluorescence microscope to visualize cytotracker (green)-stained LECs [1]. CCID areas were calculated with the Axiovision Re. 4.5 software (Carl Zeiss). MCF-7 spheroids were treated with solvent (DMSO) as negative control. The CCID sizes of at least 12 spheroids per experiment were measured.
2.5. Statistical Analyses
All experiments were performed in triplicate and analyzed by One-way ANOVA together with Tukeys post-hoc test with GraphPad Prism 5.0 program (San Diego, CA, USA).
3. Results
For the determination of the efficacy of flavonoids on the inhibition of lymph endothelial disintegration in the CCID assay in total 18 flavonoids were compared (Table 1).

Table 1.
Compounds tested in the circular chemorepellent-induced defects (CCID) assay (MCF-7 spheroids on lymph endothelial cell (LECs)), the respective IC50 (µM) and calculated logP (o/w) values.
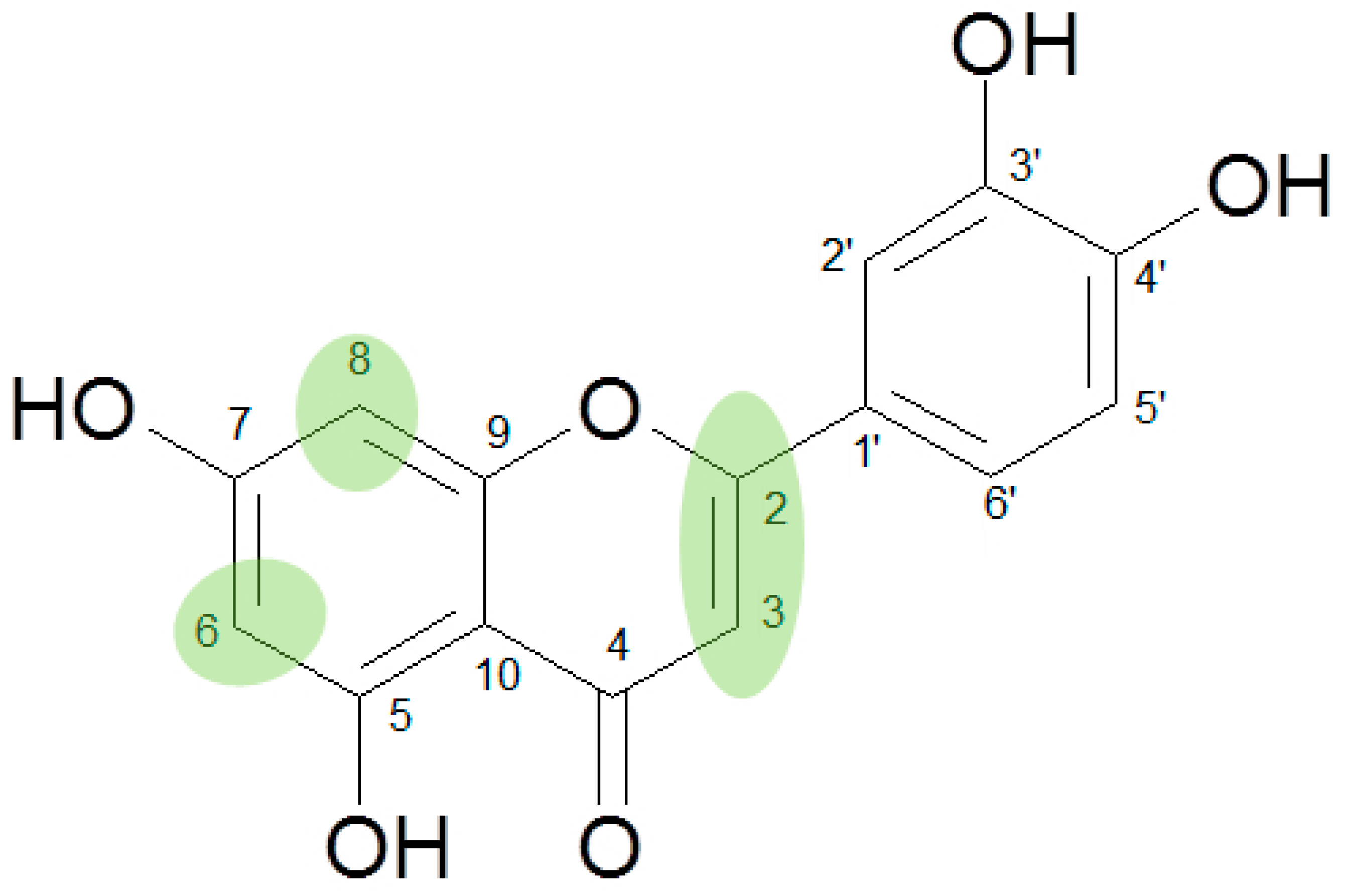
Among those were four flavanones and ten flavones and four flavonols with different degrees of hydroxylation and/or methoxylation. All compounds possessed the typical 5,7-dihydroxy- pattern. The differences applied to hydroxylation in positions 3′, 4′, 6, and/or 8. One compound each with a methoxy group in positions 3′, 4′, or 8, as well as two compounds with a methoxy group in position 6 were included into the series. Three concentrations (10, 25 and 50 or 75 µM) of each compound were tested (Supplemental Figure S2a) and the effects were compared to Bay11-7082 (Supplemental Figure S2b), an irreversible inhibitor of I-κBα phosphorylation and of CCID formation [2], for which an IC50 of 7.2 µM was determined.
The results showed that flavanones, lacking the double bond between positions 2 and 3, have obviously no or very little effect on the inhibition of CCID formation. This was independent of the degree of hydroxylation in ring B (no OH (pinocembrin, 1), 4′-OH (naringenin, 2) or 3′,4′-diOH (eriodictyol, 3)). Only homoeriodictyol (4) with a 3′-methoxy-4′-hydroxy pattern resulted in a very high IC50 value of 99.5 µM.
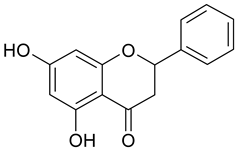
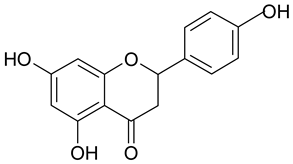
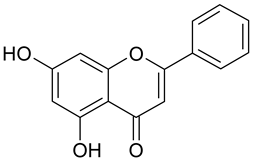
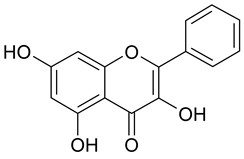
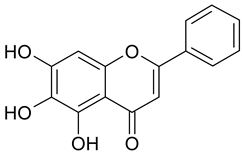
In contrast, the corresponding flavones chrysin (5, IC50 24.8 µM), apigenin (11, IC50 34.1 µM), and luteolin (14, IC50 19.3 µM) were the most active compounds. Unexpectedly, 14 with the lowest logP among these flavones showed the highest activity. Additional hydroxylation in position 3 in the flavonols galangin (6) and kaempferol (12) led to slightly higher IC50 values as compared to 5 and 11. Interestingly, quercetin (17), the 3′,4′-di-OH-flavonol corresponding to the 3′,4′-di-OH-flavone 14 remained without any activity. Gossypetin (18) with an additional 8-OH group as compared to 17 was inactive as well.
Methylation of the 4′-OH group in 14 to diosmetin (15) significantly decreased the activity to approximately one-tenth (IC50 183.2 µM).
Additional hydroxylation of the flavones in positions 6 or 8 also reduced the activity: only baicalein (7; 6-hydroxy-chrysin) showed a weak effect (IC50 130.2 µM) whereas norwogonin (9; 8-hydroxy-chrysin) was not active.
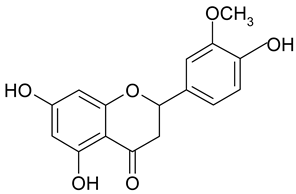
Methylation of the 6- or 8-OH groups in oroxylin A (8; 6-O-methyl-baicalein), wogonin (10; 8-O-methyl-norwogonin), and hispidulin (13; 6-O-methyl-scutellarein) resulted in an increase in the activity as compared to the respective unmethylated compounds with IC50 values of 74.8 µM, 113.9 µM, and 88.6 µM, respectively.
In comparison to 14, the most active compound, and its 4′-O-methylderivative 15, with only one-tenth of its activity, the 6-methoxyderivative nepetin (16) resulted in an IC50 value of 79 µM.
As proof of our hypothesis we included three further compounds, namely acacetin, scutellarein, and herbacetin (Table 2):

Table 2.
Compounds tested in the CCID assay (MCF-7 spheroids on LECs) as a proof of concept, the respective IC50 (µM) and calculated logP (o/w) values.
Acacetin (19), a 3′-OH methylated derivative of the highly active flavone apigenin (11 IC50 34.1 µM), resulted in almost equivalent activity (IC50 36.9 µM), underlining the impact of the 2,3-double bond and the free positions 6 and 8 on the effects of flavones in the CCID assay (Figure 1).
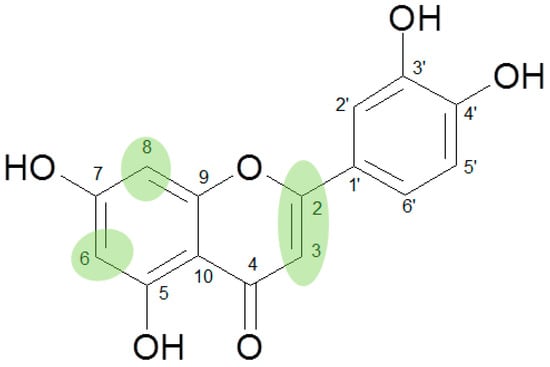
Figure 1.
Structural features of flavonoids indispensable for high activity in the CCID test.
The importance of free positions 6 and 8 was additionally shown by the inclusion of scutellarein (20; 6-OH-apigenin) and herbacetin (21; 8-OH-kaempferol). As expected from the IC50 values of baicalein (>100 µM) and norwogonin (not active) as compared to chrysin (24.74 µM), in comparison to 11 (IC50 34.1 µM) no activity was observed for 20 and in contrast to 12 (IC50 44.7 µM) 21 remained without effect as well.
The comparison of logP values did not allow an estimation of the anti-intravasative potential of the investigated flavonoids (Supplemental Figure S3). Regarding the effects of the three most active compounds luteolin (IC50 19.3 µM; logP 2.26), chrysin (IC50 24.8 µM; logP 2.84), and apigenin (IC50 34.1 µM; logP 2.53) in the CCID assay, no correlation between the IC50 and the logP values was seen. The only deducible trend was the lack of activity for compounds with logP values below 2.10. Compounds with similar logP values such as galangin (logP 2.61) and pinocembrin (logP 2.69) showed significant differences resulting in very good (IC50 39.6 µM) and no activity, respectively. The activity of the most potent flavonoid luteolin was in a comparable range with the positive control Bay11-7082.
In summary based on the results of 21 flavonoids, flavones showed better effects in our experimental set-up in the CCID assay than the corresponding flavonols, whereas flavanones remained without effects. An additional hydroxy group at C-6 or C-8 or an additional methoxy group at C-8 in ring A of the flavonoid scaffold resulted in a significant loss of activity. Further substitution of C-6 with a methoxy group decreased the activity to a lesser extent. In contrast, the impact of the hydroxylation/methoxylation pattern in ring B at C-4′ or C-3′ and C-4′ was much less and did not follow a deducible order (luteolin (3′,4′ OH) > chrysin (no OH) > apigenin (4′ OH) > acacetin (4′ OCH3)).
4. Discussion
The rationale to this study emerged from work on the anti-carcinogenic effects of a MeOH extract from the medicinal plant Scrophularia lucida which is endemic to the Eastern Mediterranean [8]. This extract strongly inhibited LEC barrier breaching because of the presence of hispidulin and unknown synergizing components [9]. Therefore, hispidulin was compared to other closely related flavonoids. Several flavonoids have been shown to disrupt the pathways and molecular mechanisms essential for cancer cell survival, also in breast cancer cells. Metastatic processes such as invasion and endothelial-to-mesenchymal-transition (EMT) were inhibited by such compounds as well [19,20]. Here, we did not focus on the tumor cells, but on the stabilizing effect of flavonoids on tumor-induced disintegration of the lymphendothelial barrier. In the clinical routine, the crossing of breast cancer cells into the lymphatic vasculature and subsequent colonization of the sentinel lymph node is a relevant prognostic marker. In our experimental set-up, anti-intravasative flavonoids with up to four-fold higher activity than hispidulin (IC50: 89 µM) were identified, namely luteolin (IC50: 19 µM) as the most potent one > chrysin (IC50: 25 µM) > apigenin (IC50: 34 µM) > acacetin (IC50: 37 µM) > galangin (IC50: 40 µM) > kaempferol (IC50: 45 µM) (see Table 1). The tested flavonoids are present in fruits and vegetables, plants, mushrooms, bee pollen and propolis [21,22]. Yet, the bio-available concentrations are too low to achieve LEC barrier stabilization by dietary consumption [23]. Nevertheless, a significant inverse correlation with colorectal cancer risk and the intake of flavonoid subclasses (flavonols, flavan-3-ols, anthocyanidins) was reported [24]. However, a lower risk of breast cancer associated with flavonoids was not observed [25]. The most active flavonoids, luteolin, chrysin, and apigenin, are commercially available and particular formulations of luteolin and apigenin reached plasma concentrations in the range of those exhibiting significant in vitro anti-intravasative effects in our study. In detail, a single oral dose of luteolin or luteolin together with a peanut hull extract (14.3 mg/kg each) resulted in peak concentrations of 1.9 µg/mL and 8.3 µg/mL (respectively) of luteolin in rat plasma [26]. This corresponds to luteolin concentrations of ~6.9 µM and ~29.1 µM, respectively. Similarly, a single oral dose of apigenin or apigenin delivered with a carbon nanopowder solid dispersion carrier (60 mg/kg each) gave peak concentrations of 1.3 µg/mL and 3.2 µg/mL (respectively) of apigenin in rat plasma [27]. This corresponds to apigenin concentrations of ~4.8 µM and ~11.8 µM, respectively. Chrysin at daily oral doses (50 mg/kg) in a mouse xenograft model with A549 non-small-cell lung cancer cells, reduced the tumor volume by 50% as compared to control. In breast cancer mouse models, oral administration of chrysin significantly reduced the growth of lung metastases of 4T1 breast cancer cells and tumor growth in a MDA-MB-231 model [28,29]. This proves the bio-availability of chrysin as well as its anti- carcinogenic potential [30]. However, it is still not known whether this effect was due to chrysin itself or due to a metabolite. For an anti-metastatic activity of chrysin, the inhibition of angiogenesis through reduced vascular endothelial growth factor (VEGF) expression might be responsible. Furthermore, chrysin induced E-cadherin, decreased vimentin, SNAIL and SLUG levels thereby reverting EMT. Chrysin inhibited AKT-dependent matrix metalloprotease (MMP)-10 expression in triple negative breast cancer, cell migration and invasion [31], and MMP9 through JNK—ERK—AP1 [32]. Importantly, a daily dose of 3 g chrysin did not exhibit toxicity in a pilot clinical trial [33].
Luteolin (25 µM) inhibited MMP9 and MMP2 in breast- and ovarian cancer cells [34,35] at concentrations which did not compromise the viability of MDA-MB231- and MCF-7/6 cells. Luteolin concentrations as low as 1 µM inhibited the invasion of MDA-MB231 cells through a basement membrane extract [36]. This supported the notion that luteolin specifically targets migratory/metastatic processes but not proliferation/survival mechanisms in cancer cells. MMP2 and MMP9 contribute to the intravasation in the MCF-7/LEC model [13] and this can explain the anti-intravasative properties of chrysin and luteolin. This is also in agreement with the observation that 20 µM luteolin inhibited the matrix metallo-protease 1 (MMP1) in MDA-MB231 cells [16]. MMP1, MMP2, and MMP9 are triggering the retraction of LECs [37] and the formation of CCIDs. Similar to chrysin, 10 µM luteolin induced the epithelial marker E-cadherin, down-regulated the mesenchymal markers vimentin, N-cadherin SNAIL, and SLUG, and attenuated the migration of MDA-MB231 cells in a wound healing assay [38]. This evidenced the inhibition of EMT and of cell mobility. Apigenin as well inhibited SNAIL, EMT, and metastasis of hepatocellular carcinoma [39]. Urokinase-plasminogen activator expression, migration, invasion and phorbol-12-myristate-13-acetate (PMA)-induced MMP-9 secretion were inhibited in MDA-MB231 cells [40] and in MCF-7 cells [41] upon treatment with apigenin.
In addition, apigenin, luteolin, and chrysin inhibit nuclear factor κB (NF-κB) activity [34,39,41,42,43]. MCF-7 cells secret 12-hydroxyeicosatetraenoic acid (12(S)-HETE), which triggers the activation of NF-κB in LECs as a prerequisite for CCID formation in the LEC monolayer [2,3]. Thus, luteolin, chrysin, and apigenin stabilize the endothelial barrier function by inhibiting NF-κB in LECs despite the de-stabilizing effects of 12(S)-HETE. Another mechanism of luteolin and apigenin is the inhibition of FAK phosphorylation at Tyr397 and thereby attachment/detachment oscillations at the matrix, which is necessary for LEC migration and CCID formation [16].
The methylation of the hydroxy group at C-4′ in apigenin leads to acacetin, which was almost as active as apigenin. In MDA-MB231 cells acacetin (25 µM) significantly inhibited PMA-induced MMP9 mRNA expression after 48 h [34]. The absorption of methylated flavonoids in intact organisms is much higher as compared to the respective non-methylated compounds (i.e., apigenin). Therefore, these compounds show improved metabolic stability and bio-availability [44]. Acacetin is also commercially available and deserves further investigations for a potential pharmaceutical use. Diosmetin, the 4′-O-methyl-luteolin, was inactive in our assay. In another investigation, diosmetin (~30 µM) inhibited ~50% invasion and migration of SK-HEP1- and MHcc97H hepatocellular carcinoma cells after 24 h. However, these results did not correlate with an inhibition of MMP2 and MMP9 [45]. We suppose that in our experimental set-up treatment with diosmetin for more than 4 h may have resulted in a similar inhibition of LEC migration. Consequently, the lesser effect of diosmetin, compared to luteolin, must have been the consequence of an un-favorable structural alteration because of the hydroxyl group at 3′ in combination with the 4′O-methylation.
Galangin and kaempferol exhibited almost similar anti-intravasative activities. They inhibited MMP2, MMP9, migration and invasion, and suppressed EMT in MDA-MB-231- and MCF-7 breast cancer cells [34,46,47,48], as well as NF-κB activity in various experimental models [49,50]. The bioavailability of kaempferol after oral administration (100 mg/kg) in Spargue-Dawley rats was 2% [51]. After a single oral dose of galangin (10 mg/kg) only 220 ng/mL of the unmetabolized compound were detected in rat plasma [52].
Similar to the flavonoids above, oroxylin A, wogonin, and baicalein were also reported to inhibit EMT marker expression in MCF-7, A549, and MDA-MB231 cell lines, respectively [53,54,55] and to inhibit MMP2/9, migration and invasiveness of MDA-MB231 cells [56,57,58]. In a xenograft nude mouse model, baicalein inhibited tumor metastasis [59]. However, the IC50 values of these flavonoids in our experiments were probably too high to achieve anti-intravasative effects in a patho-physiological setting.
Migration and invasion of MCF-7 cells were inhibited upon treatment with 80 µM quercetin and 20 µM blocked PMA-induced upregulation of MMP9 [60]. At a concentration of 50 µM this compound inhibited the migration of SAS oral squamous carcinoma cells which correlated with the suppression of MMP2 and MMP9 protein [61]. In contrast, suppression of MMP2 and MMP9 protein upon treatment with 25 µM quercetin did not show any correlation with the inhibition of cancer cell migration [62]. However, this concentration correlated with an inhibition of invasion of osteosarcoma cells in vitro and invasion of cancer cell xenografts into the lungs in a mouse model. This indicates that the anti-migratory property of quercetin was not necessarily connected to MMP2/9 expression in cancer cells. The migration of aorta blood endothelial cells was only weakly inhibited by 100 µM quercetin. This concentration had no effects on the tube formation of vascular endothelial cells, another cell migratory process [63]. In fact, the migration of blood endothelial cells during angiogenesis depends on VEGF/VEGFR2 instead of MMP2/9, and quercetin interferes with VEGFR2 expression [64,65]. However, in lymph endothelial cells, the VEGF/VEGFR2 signaling is not observed during intravasation of MCF-7 cells [13]. Also, the activation of NF-κB, on which the formation of CCIDs in the LEC monolayer is strictly dependent, was not inhibited by quercetin in the endothelial cell line ECV304 [66]. As CCID formation was not affected at 10–75 µM quercetin, this process might be devoid of targets addressed by quercetin.
The anti-intravasative activities of the other tested flavonoids were too low to consider targeting migratory and intravasation-supporting mechanisms in LECs, although herbacetin interferes with pro-metastatic HGF-cMET signaling in MDA-MB231 cells [67]. Hence, cMET obviously does not play a role in the migration of LECs.
The migration of LECs was dependent on the stimulation of NF-κB by MCF-7-secreted 12(S)-HETE and subsequent expression of ICAM-1 in LECs, which facilitates the adhesion of LECs to cancer cells as a prerequisite for CCID formation [3]. Furthermore, 12(S)-HETE induces LEC migration through the release of Ca2+ from intracellular stores, the induction of phospholipase C and generation of inositol 3-phosphate, as well as the activation of the RHO/ROCK pathway, both activating the mobility of protein MLC2 [37,68]. In breast cancer cells also matrix metalloproteases play a role in CCID formation such as MMP1 and MMP11 [37] and MMP2/9 and TIMP2 [13]. Whether MMP2/9 and TIMP2 activity is LEC- or cancer cell associated and whether and how these pathways are interconnected is still an open issue.
5. Conclusions
The comparison of 21 flavonoids for their potential to stabilize LEC barriers that were disintegrated by MCF-7 breast cancer spheroids clearly showed the highest activity for flavones with a moderate grade of hydroxylation. The vast majority of these compounds were studied in the CCID assay for the first time. Our results confirm studies which have shown the importance of the 2, 3 double bond in ring C of the flavonoid scaffold on different mechanisms in progression of cancer. Under consideration of the bio-availability of flavonoids, a sufficient provision of the most active compounds could be reached via special delivery systems.
Supplementary Materials
The following are available online at https://www.mdpi.com/2218-0532/87/3/23/s1, Figure S1: Lymph-endothelial marker expression in LEC. Figure S2: a Treatment of the MCF-7/LEC model with flavonoids; b Treatment of the MCF-7/LEC model with Bay11-7802. Figure S3: Correlation of logP values (o/w) and pIC50 values of 13 flavonoids which showed activity in the CCID assay.
Author Contributions
J.E., K.K., C.S. (Claudia Smöch), and C.S. (Christa Stadlbauer) performed the experiments, C.H.N. supervised the experiments, A.Ö. contributed to the initiation of the project, G.E. determined pK values, W.J., G.K., and L.K. planned the project and wrote the manuscript.
Funding
CHN was supported by a technology grant (TSA Doktorat) financed by the Austrian Federal Ministry of Science and Research (BMFW) in frame of Asea Uninet.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Madlener, S.; Saiko, P.; Vonach, C.; Viola, K.; Huttary, N.; Stark, N.; Popescu, R.; Gridling, M.; Vo, N.T.; Herbacek, I.; et al. Multifactorial anticancer effects of digalloyl-resveratrol encompass apoptosis, cell-cycle arrest, and inhibition of lymphendothelial gap formation in vitro. Br. J. Cancer 2010, 102, 1361–1370. [Google Scholar] [CrossRef]
- Vonach, C.; Viola, K.; Giessrigl, B.; Huttary, N.; Raab, I.; Kalt, R.; Krieger, S.; Vo, T.P.; Madlener, S.; Bauer, S.; et al. NF-kappaB mediates the 12(S)-HETE-induced endothelial to mesenchymal transition of lymphendothelial cells during the intravasation of breast carcinoma cells. Br. J. Cancer 2011, 105, 263–271. [Google Scholar] [CrossRef] [PubMed]
- Viola, K.; Kopf, S.; Huttary, N.; Vonach, C.; Kretschy, N.; Teichmann, M.; Giessrigl, B.; Raab, I.; Stary, S.; Krieger, S.; et al. Bay11-7082 inhibits the disintegration of the lymphendothelial barrier triggered by MCF-7 breast cancer spheroids; the role of ICAM-1 and adhesion. Br. J. Cancer 2013, 108, 564–569. [Google Scholar] [CrossRef] [PubMed]
- Newman, D.J.; Cragg, G.M. Natural Products as Sources of New Drugs from 1981 to 2014. J. Nat. Prod. 2016, 79, 629–661. [Google Scholar] [CrossRef]
- Özmen, A.; Bauer, S.; Gridling, M.; Singhuber, J.; Krasteva, S.; Madlener, S.; Vo, T.P.N.; Stark, N.; Saiko, P.; Fritzer-Szekeres, M.; et al. In vitro anti-neoplastic activity of the ethno-pharmaceutical plant Hypericum adenotrichum Spach endemic to Western Turkey. Oncol. Rep. 2009, 22, 845–852. [Google Scholar] [PubMed]
- Gridling, M.; Popescu, R.; Kopp, B.; Wagner, K.H.; Krenn, L.; Krupitza, G. Anti-leukaemic effects of two extract types of Lactuca sativa correlate with the activation of Chk2, induction of p21, downregulation of cyclin D1 and acetylation of α-tubulin. Oncol. Rep. 2010, 23, 1145–1151. [Google Scholar] [PubMed]
- Ozmen, A.; Madlener, S.; Bauer, S.; Krasteva, S.; Vonach, C.; Giessrigl, B.; Gridling, M.; Viola, K.; Stark, N.; Saiko, P.; et al. In vitro anti-leukemic activity of the ethno-pharmacological plant Scutellaria orientalis ssp. carica endemic to western Turkey. Phytomedicine 2010, 17, 55–62. [Google Scholar] [CrossRef] [PubMed]
- Giessrigl, B.; Yazici, G.; Teichmann, M.; Kopf, S.; Ghassemi, S.; Atanasov, A.G.; Dirsch, V.M.; Grusch, M.; Jäger, W.; Özmen, A.; et al. Effects of Scrophularia extracts on tumor cell proliferation, death and intravasation through lymphoendothelial cell barriers. Int. J. Oncol. 2012, 40, 2063–2074. [Google Scholar] [PubMed]
- Lewenhofer, V.; Schweighofer, L.; Ledermüller, T.; Eichsteininger, J.; Kählig, H.; Zehl, M.; Nguyen, C.H.; Krupitza, G.; Özmen, A.; Krenn, L. Chemical Composition of Scrophularia lucida and the Effects on Tumor Invasiveness in Vitro. Front Pharmacol. 2018, 9, 304. [Google Scholar] [CrossRef] [PubMed]
- Ravishankar, D.; Rajora, A.K.; Greco, F.; Osborn, H.M. Flavonoids as prospective compounds for anti-cancer therapy. Int. J. Biochem. Cell Biol. 2013, 45, 2821–2831. [Google Scholar] [CrossRef] [PubMed]
- Weng, C.J.; Yen, G.C. Flavonoids, a ubiquitous dietary phenolic subclass, exert extensive in vitro anti-invasive and in vivo anti-metastatic activities. Cancer Metastasis. Rev. 2012, 31, 323–351. [Google Scholar] [CrossRef] [PubMed]
- Stadler, M.; Scherzer, M.; Walter, S.; Holzner, S.; Pudelko, K.; Riedl, A.; Unger, C.; Kramer, N.; Weil, B.; Neesen, J.; et al. Exclusion from spheroid formation identifies loss of essential cell-cell adhesion molecules in colon cancer cells. Sci. Rep. 2018, 8, 1151. [Google Scholar] [CrossRef] [PubMed]
- Kerjaschki, D.; Bago-Horvath, Z.; Rudas, M.; Sexl, V.; Schneckenleithner, C.; Wolbank, S.; Bartel, G.; Krieger, S.; Kalt, R.; Hantusch, B.; et al. Lipoxygenase mediates invasion of intrametastatic lymphatic vessels and propagates lymph node metastasis of human mammary carcinoma xenografts in mouse. J. Clin. Invest. 2011, 121, 2000–2012. [Google Scholar] [CrossRef] [PubMed]
- Holzner, S.; Brenner, S.; Atanasov, A.G.; Senfter, D.; Stadler, S.; Nguyen, C.H.; Fristiohady, A.; Milovanovic, D.; Huttary, N.; Krieger, S.; et al. Intravasation of SW620 colon cancer cell spheroids through the blood endothelial barrier is inhibited by clinical drugs and flavonoids in vitro. Food Chem. Toxicol. 2017, 111, 114–124. [Google Scholar] [CrossRef] [PubMed]
- Stadler, S.; Nguyen, C.H.; Schachner, H.; Milovanovic, D.; Holzner, S.; Brenner, S.; Eichsteininger, J.; Stadler, M.; Senfter, D.; Krenn, L.; et al. Colon cancer cell-derived 12(S)-HETE induces the retraction of cancer-associated fibroblast via MLC2, RHO/ROCK and Ca(2+) signalling. Cell Mol. Life Sci. 2017, 74, 1907–1921. [Google Scholar] [CrossRef] [PubMed]
- Hong, J.; Fristiohady, A.; Nguyen, C.H.; Milovanovic, D.; Huttary, N.; Krieger, S.; Hong, J.; Geleff, S.; Birner, P.; Jager, W.; et al. Apigenin and Luteolin Attenuate the Breaching of MDA-MB231 Breast Cancer Spheroids Through the Lymph Endothelial Barrier in Vitro. Front. Pharmacol. 2018, 9, 220. [Google Scholar] [CrossRef] [PubMed]
- Yang, J.; Chang, E.; Cherry, A.M.; Bangs, C.D.; Oei, Y.; Bodnar, A.; Bronstein, A.; Chiu, C.P.; Herron, G.S. Human endothelial cell life extension by telomerase expression. J. Biol. Chem. 1999, 274, 26141–26148. [Google Scholar] [CrossRef]
- Schoppmann, S.F.; Kalt, R.; Okubo, Y.; Benisch, C.; Nagavarapu, U.; Herron, G.S.; Geleff, S. Telomerase-immortalized lymphatic and blood vessel endothelial cells are functionally stable and retain their lineage specificity. Microcirculation 2004, 11, 261–269. [Google Scholar] [CrossRef] [PubMed]
- Ci, Y.; Qiao, J.; Han, M. Molecular Mechanisms and Metabolomics of Natural Polyphenols Interfering with Breast Cancer Metastasis. Molecules 2016, 21, 1634. [Google Scholar] [CrossRef] [PubMed]
- Lee, G.A.; Hwang, K.A.; Choi, K.C. Roles of Dietary Phytoestrogens on the Regulation of Epithelial-Mesenchymal Transition in Diverse Cancer Metastasis. Toxins 2016, 8, 162. [Google Scholar] [CrossRef] [PubMed]
- Badolato, M.; Carullo, G.; Cione, E.; Aiello, F.; Caroleo, M.C. From the hive: Honey, a novel weapon against cancer. Eur. J. Med. Chem. 2017, 142, 290–299. [Google Scholar] [CrossRef] [PubMed]
- Aiello, F.; Armentano, B.; Polera, N.; Carullo, G.; Loizzo, M.R.; Bonesi, M.; Cappello, M.S.; Capobianco, L.; Tundis, R. From Vegetable Waste to New Agents for Potential Health Applications: Antioxidant Properties and Effects of Extracts, Fractions and Pinocembrin from Glycyrrhiza glabra L. Aerial Parts on Viability of Five Human Cancer Cell Lines. J. Agric. Food Chem. 2017, 65, 7944–7954. [Google Scholar] [CrossRef] [PubMed]
- Vogiatzoglou, A.; Mulligan, A.A.; Lentjes, M.A.; Luben, R.N.; Spencer, J.P.; Schroeter, H.; Khaw, K.T.; Kuhnle, G.G. Flavonoid intake in European adults (18 to 64 years). PLoS ONE 2015, 10, e0128132. [Google Scholar] [CrossRef] [PubMed]
- Woo, H.D.; Kim, J. Dietary flavonoid intake and risk of stomach and colorectal cancer. World J. Gastroenterol. 2013, 19, 1011–1019. [Google Scholar] [CrossRef] [PubMed]
- Hui, C.; Qi, X.; Qianyong, Z.; Xiaoli, P.; Jundong, Z.; Mantian, M. Flavonoids, flavonoid subclasses and breast cancer risk: a meta-analysis of epidemiologic studies. PLoS ONE 2013, 8, e54318. [Google Scholar] [CrossRef] [PubMed]
- Zhou, P.; Li, L.P.; Luo, S.Q.; Jiang, H.D.; Zeng, S. Intestinal absorption of luteolin from peanut hull extract is more efficient than that from individual pure luteolin. J. Agric Food Chem. 2008, 56, 296–300. [Google Scholar] [CrossRef]
- Ding, S.M.; Zhang, Z.H.; Song, J.; Cheng, X.D.; Jiang, J.; Jia, X.B. Enhanced bioavailability of apigenin via preparation of a carbon nanopowder solid dispersion. Int. J. Nanomed. 2014, 9, 2327–2333. [Google Scholar] [CrossRef] [PubMed]
- Sun, L.P.; Chen, A.L.; Hung, H.C.; Chien, Y.H.; Huang, J.S.; Huang, C.Y.; Chen, Y.W.; Chen, C.N. Chrysin: A histone deacetylase 8 inhibitor with anticancer activity and a suitable candidate for the standardization of Chinese propolis. J. Agric. Food Chem. 2012, 60, 11748–11758. [Google Scholar] [CrossRef] [PubMed]
- Lirdprapamongkol, K.; Sakurai, H.; Abdelhamed, S.; Yokoyama, S.; Maruyama, T.; Athikomkulchai, S.; Viriyaroj, A.; Awale, S.; Yagita, H.; Ruchirawat, S.; et al. A flavonoid chrysin suppresses hypoxic survival and metastatic growth of mouse breast cancer cells. Oncol. Rep. 2013, 30, 2357–2364. [Google Scholar] [CrossRef] [PubMed]
- Kim, K.M.; Lim, H.K.; Shim, S.H.; Jung, J. Improved chemotherapeutic efficacy of injectable chrysin encapsulated by copolymer nanoparticles. Int. J. Nanomed. 2017, 12, 1917–1925. [Google Scholar] [CrossRef] [PubMed]
- Yang, B.; Huang, J.; Xiang, T.; Yin, X.; Luo, X.; Huang, J.; Luo, F.; Li, H.; Li, H.; Ren, G. Chrysin inhibits metastatic potential of human triple-negative breast cancer cells by modulating matrix metalloproteinase-10, epithelial to mesenchymal transition, and PI3K/Akt signaling pathway. J. Appl. Toxicol. 2014, 34, 105–112. [Google Scholar] [CrossRef] [PubMed]
- Xia, Y.; Lian, S.; Khoi, P.N.; Yoon, H.J.; Joo, Y.E.; Chay, K.O.; Kim, K.K.; Do Jung, Y. Chrysin inhibits tumor promoter-induced MMP-9 expression by blocking AP-1 via suppression of ERK and JNK pathways in gastric cancer cells. PLoS ONE 2015, 10, e0124007. [Google Scholar] [CrossRef] [PubMed]
- Tobin, P.J.; Beale, P.; Noney, L.; Liddell, S.; Rivory, L.P.; Clarke, S. A pilot study on the safety of combining chrysin, a non-absorbable inducer of UGT1A1, and irinotecan (CPT-11) to treat metastatic colorectal cancer. Cancer Chemother. Pharmacol. 2006, 57, 309–316. [Google Scholar] [CrossRef] [PubMed]
- Amrutha, K.; Nanjan, P.; Shaji, S.K.; Sunilkumar, D.; Subhalakshmi, K.; Rajakrishna, L.; Banerji, A. Discovery of lesser known flavones as inhibitors of NF-kappaB signaling in MDA-MB-231 breast cancer cells--A SAR study. Bioorg. Med. Chem. Lett. 2014, 24, 4735–4742. [Google Scholar] [CrossRef] [PubMed]
- Liu, H.; Zeng, Z.; Wang, S.; Li, T.; Mastriani, E.; Li, Q.H.; Bao, H.X.; Zhou, Y.J.; Wang, X.; Liu, Y.; et al. Main components of pomegranate, ellagic acid and luteolin, inhibit metastasis of ovarian cancer by down-regulating MMP2 and MMP9. Cancer Biol. Ther. 2017, 18, 990–999. [Google Scholar] [CrossRef] [PubMed]
- Attoub, S.; Hassan, A.H.; Vanhoecke, B.; Iratni, R.; Takahashi, T.; Gaben, A.M.; Bracke, M.; Awad, S.; John, A.; Kamalboor, H.A.; et al. Inhibition of cell survival, invasion, tumor growth and histone deacetylase activity by the dietary flavonoid luteolin in human epithelioid cancer cells. Eur. J. Pharmacol. 2011, 651, 18–25. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, C.H.; Senfter, D.; Basilio, J.; Holzner, S.; Stadler, S.; Krieger, S.; Huttary, N.; Milovanovic, D.; Viola, K.; Simonitsch-Klupp, I.; et al. NF-κB contributes to MMP1 expression in breast cancer spheroids causing paracrine PAR1 activation and disintegrations in the lymph endothelial barrier in vitro. Oncotarget 2015, 6, 39262–39275. [Google Scholar] [CrossRef]
- Lin, D.; Kuang, G.; Wan, J.; Zhang, X.; Li, H.; Gong, X.; Li, H. Luteolin suppresses the metastasis of triple-negative breast cancer by reversing epithelial-to-mesenchymal transition via downregulation of beta-catenin expression. Oncol. Rep. 2017, 37, 895–902. [Google Scholar] [CrossRef]
- Qin, Y.; Zhao, D.; Zhou, H.G.; Wang, X.H.; Zhong, W.L.; Chen, S.; Gu, W.G.; Wang, W.; Zhang, C.H.; Liu, Y.R.; et al. Apigenin inhibits NF-κB and snail signaling, EMT and metastasis in human hepatocellular carcinoma. Oncotarget 2016, 7, 41421–41431. [Google Scholar] [CrossRef]
- Lindenmeyer, F.; Li, H.; Menashi, S.; Soria, C.; Lu, H. Apigenin acts on the tumor cell invasion process and regulates protease production. Nutr. Cancer 2001, 39, 139–147. [Google Scholar] [CrossRef]
- Liao, Y.F.; Rao, Y.K.; Tzeng, Y.M. Aqueous extract of Anisomeles indica and its purified compound exerts anti-metastatic activity through inhibition of NF-kappaB/AP-1-dependent MMP-9 activation in human breast cancer MCF-7 cells. Food Chem. Toxicol. 2012, 50, 2930–2936. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Huang, Q.; Ong, C.N.; Yang, X.F.; Shen, H.M. Chrysin sensitizes tumor necrosis factor-α-induced apoptosis in human tumor cells via suppression of nuclear factor-kappaB. Cancer Lett. 2010, 293, 109–116. [Google Scholar] [CrossRef]
- Guo, Y.F.; Xu, N.N.; Sun, W.; Zhao, Y.; Li, C.Y.; Guo, M.Y. Luteolin reduces inflammation in Staphylococcus aureus-induced mastitis by inhibiting NF-kB activation and MMPs expression. Oncotarget 2017, 8, 28481–28493. [Google Scholar] [CrossRef] [PubMed]
- Walle, T. Methylation of dietary flavones increases their metabolic stability and chemopreventive effects. Int. J. Mol. Sci. 2009, 10, 5002–5019. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Wen, X.; Liu, B.; Zhang, Q.; Zhang, J.; Miao, H.; Zhu, R. Diosmetin inhibits the metastasis of hepatocellular carcinoma cells by downregulating the expression levels of MMP-2 and MMP-9. Mol. Med. Rep. 2016, 13, 2401–2408. [Google Scholar] [CrossRef] [PubMed]
- Lee, G.A.; Choi, K.C.; Hwang, K.A. Kaempferol, a phytoestrogen, suppressed triclosan-induced epithelial-mesenchymal transition and metastatic-related behaviors of MCF-7 breast cancer cells. Environ. Toxicol. Pharmacol. 2017, 49, 48–57. [Google Scholar] [CrossRef] [PubMed]
- Li, C.; Zhao, Y.; Yang, D.; Yu, Y.; Guo, H.; Zhao, Z.; Zhang, B.; Yin, X. Inhibitory effects of kaempferol on the invasion of human breast carcinoma cells by downregulating the expression and activity of matrix metalloproteinase-9. Biochem. Cell Biol. 2015, 93, 16–27. [Google Scholar] [CrossRef] [PubMed]
- Xu, Y.F.; Yao, X. Inhibitory effect and mechanism of galangin on breast cancer metastasis. Chin. Tradit. Herb. Drugs 2016, 47, 1731–1739. [Google Scholar]
- Jung, Y.C.; Kim, M.E.; Yoon, J.H.; Park, P.R.; Youn, H.Y.; Lee, H.W.; Lee, J.S. Anti-inflammatory effects of galangin on lipopolysaccharide-activated macrophages via ERK and NF-κB pathway regulation. Immunopharmacol. Immunotoxicol. 2014, 36, 426–432. [Google Scholar] [CrossRef]
- Kadioglu, O.; Nass, J.; Saeed, M.E.M.; Schuler, B.; Efferth, T. Kaempferol is an anti-inflammatory compound with activity towards NF-ℵB pathway proteins. Anticancer Res. 2015, 35, 2645–2650. [Google Scholar]
- Barve, A.; Chen, C.; Hebbar, V.; Desiderio, J.; Saw, C.L.; Kong, A.N. Metabolism, oral bioavailability and pharmacokinetics of chemopreventive kaempferol in rats. Biopharm. Drug Dispos. 2009, 30, 356–365. [Google Scholar] [CrossRef] [PubMed]
- Chen, F.; Tan, Y.F.; Li, H.L.; Qin, Z.M.; Cai, H.D.; Lai, W.Y.; Zhang, X.P.; Li, Y.H.; Guan, W.W.; Li, Y.B.; et al. Differential systemic exposure to galangin after oral and intravenous administration to rats. Chem. Cent. J. 2015, 9, 14. [Google Scholar] [CrossRef] [PubMed]
- Biteau, F.; Nisse, E.; Hehn, A.; Miguel, S.; Hannewald, P.; Bourgaud, F. A rapid and efficient method for isolating high quality DNA from leaves of carnivorous plants from the Drosera genus. Mol. Biotechnol. 2012, 51, 247–253. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Y.; Yao, J.; Wu, X.P.; Zhao, L.; Zhou, Y.X.; Zhang, Y.; You, Q.D.; Guo, Q.L.; Lu, N. Wogonin suppresses human alveolar adenocarcinoma cell A549 migration in inflammatory microenvironment by modulating the IL-6/STAT3 signaling pathway. Mol. Carcinog. 2015, 54, E81–E93. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, L.T.; Song, Y.W.; Cho, S.K. Baicalein Inhibits Epithelial to Mesenchymal Transition via Downregulation of Cyr61 and LOXL-2 in MDA-MB231 Breast Cancer Cells. Mol. Cells 2016, 39, 909–914. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Ling, Y.; Chen, Y.; Li, C.L.; Feng, F.; You, Q.D.; Lu, N.; Guo, Q.L. Flavonoid baicalein suppresses adhesion, migration and invasion of MDA-MB-231 human breast cancer cells. Cancer Lett. 2010, 297, 42–48. [Google Scholar] [CrossRef] [PubMed]
- Chen, P.; Lu, N.; Ling, Y.; Chen, Y.; Hui, H.; Lu, Z.; Song, X.; Li, Z.; You, Q.; Guo, Q. Inhibitory effects of wogonin on the invasion of human breast carcinoma cells by downregulating the expression and activity of matrix metalloproteinase-9. Toxicology 2011, 282, 122–128. [Google Scholar] [CrossRef]
- Lu, Z.; Lu, N.; Li, C.; Li, F.; Zhao, K.; Lin, B.; Guo, Q. Oroxylin A inhibits matrix metalloproteinase-2/9 expression and activation by up-regulating tissue inhibitor of metalloproteinase-2 and suppressing the ERK1/2 signaling pathway. Toxicol Lett. 2012, 209, 211–220. [Google Scholar] [CrossRef]
- Ma, X.; Yan, W.; Dai, Z.; Gao, X.; Ma, Y.; Xu, Q.; Jiang, J.; Zhang, S. Baicalein suppresses metastasis of breast cancer cells by inhibiting EMT via downregulation of SATB1 and Wnt/beta-catenin pathway. Drug. Des. Devel. Ther. 2016, 10, 1419–1441. [Google Scholar] [CrossRef]
- Lin, C.W.; Hou, W.C.; Shen, S.C.; Juan, S.H.; Ko, C.H.; Wang, L.M.; Chen, Y.C. Quercetin inhibition of tumor invasion via suppressing PKC delta/ERK/AP-1-dependent matrix metalloproteinase-9 activation in breast carcinoma cells. Carcinogenesis 2008, 29, 1807–1815. [Google Scholar] [CrossRef]
- Lai, W.W.; Hsu, S.C.; Chueh, F.S.; Chen, Y.Y.; Yang, J.S.; Lin, J.P.; Lien, J.C.; Tsai, C.H.; Chung, J.G. Quercetin inhibits migration and invasion of SAS human oral cancer cells through inhibition of NF-κB and matrix metalloproteinase-2/-9 signaling pathways. Anticancer Res. 2013, 33, 1941–1950. [Google Scholar] [PubMed]
- Lan, H.; Hong, W.; Fan, P.; Qian, D.; Zhu, J.; Bai, B. Quercetin Inhibits Cell Migration and Invasion in Human Osteosarcoma Cells. Cell. Physiol. Biochem. 2017, 43, 553–567. [Google Scholar] [CrossRef] [PubMed]
- Igura, K.; Ohta, T.; Kuroda, Y.; Kaji, K. Resveratrol and quercetin inhibit angiogenesis in vitro. Cancer Lett. 2001, 171, 11–16. [Google Scholar] [CrossRef]
- Pratheeshkumar, P.; Budhraja, A.; Son, Y.O.; Wang, X.; Zhang, Z.; Ding, S.; Wang, L.; Hitron, A.; Lee, J.C.; Xu, M.; et al. Quercetin inhibits angiogenesis mediated human prostate tumor growth by targeting VEGFR- 2 regulated AKT/mTOR/P70S6K signaling pathways. PLoS ONE 2012, 7, e47516. [Google Scholar] [CrossRef] [PubMed]
- Ravishankar, D.; Watson, K.A.; Boateng, S.Y.; Green, R.J.; Greco, F.; Osborn, H.M.I. Exploring quercetin and luteolin derivatives as antiangiogenic agents. Eur. J. Med. Chem. 2015, 97, 259–274. [Google Scholar] [CrossRef] [PubMed]
- Kobuchi, H.; Roy, S.; Sen, C.K.; Nguyen, H.G.; Packer, L. Quercetin inhibits inducible ICAM-1 expression in human endothelial cells through the JNK pathway. Am. J. Physiol.- Cell Physiol. 1999, 277, C403–C411. [Google Scholar] [CrossRef] [PubMed]
- Hyuga, S.; Hyuga, M.; Yoshimura, M.; Amakura, Y.; Goda, Y.; Hanawa, T. Herbacetin, a constituent of ephedrae herba, suppresses the HGF-induced motility of human breast cancer MDA-MB-231 cells by inhibiting c-Met and Akt phosphorylation. Planta Med. 2013, 79, 1525–1530. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, C.H.; Brenner, S.; Huttary, N.; Li, Y.; Atanasov, A.G.; Dirsch, V.M.; Holzner, S.; Stadler, S.; Riha, J.; Krieger, S.; et al. 12(S)-HETE increases intracellular Ca(2+) in lymph-endothelial cells disrupting their barrier function in vitro; stabilization by clinical drugs impairing calcium supply. Cancer Lett. 2016, 380, 174–183. [Google Scholar] [CrossRef] [PubMed]
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).