In Vitro Antimicrobial Activity of Essential Oils from Sardinian Flora against Cutibacterium (Formerly Propionibacterium) acnes and Its Enhancement by Chitosan
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Plant Material and Isolation of Essential Oil
2.3. Oil Analyses and Quantification
2.4. Antimicrobial Activity of Essential Oils and Chitosan
2.5. Antibacterial Activity of M. communis, S. insularis and J. oxycedrus Essential Oils in the Presence of Sub-Inhibitory Concentrations of Chitosan
3. Results
3.1. Essential Oils Characterization
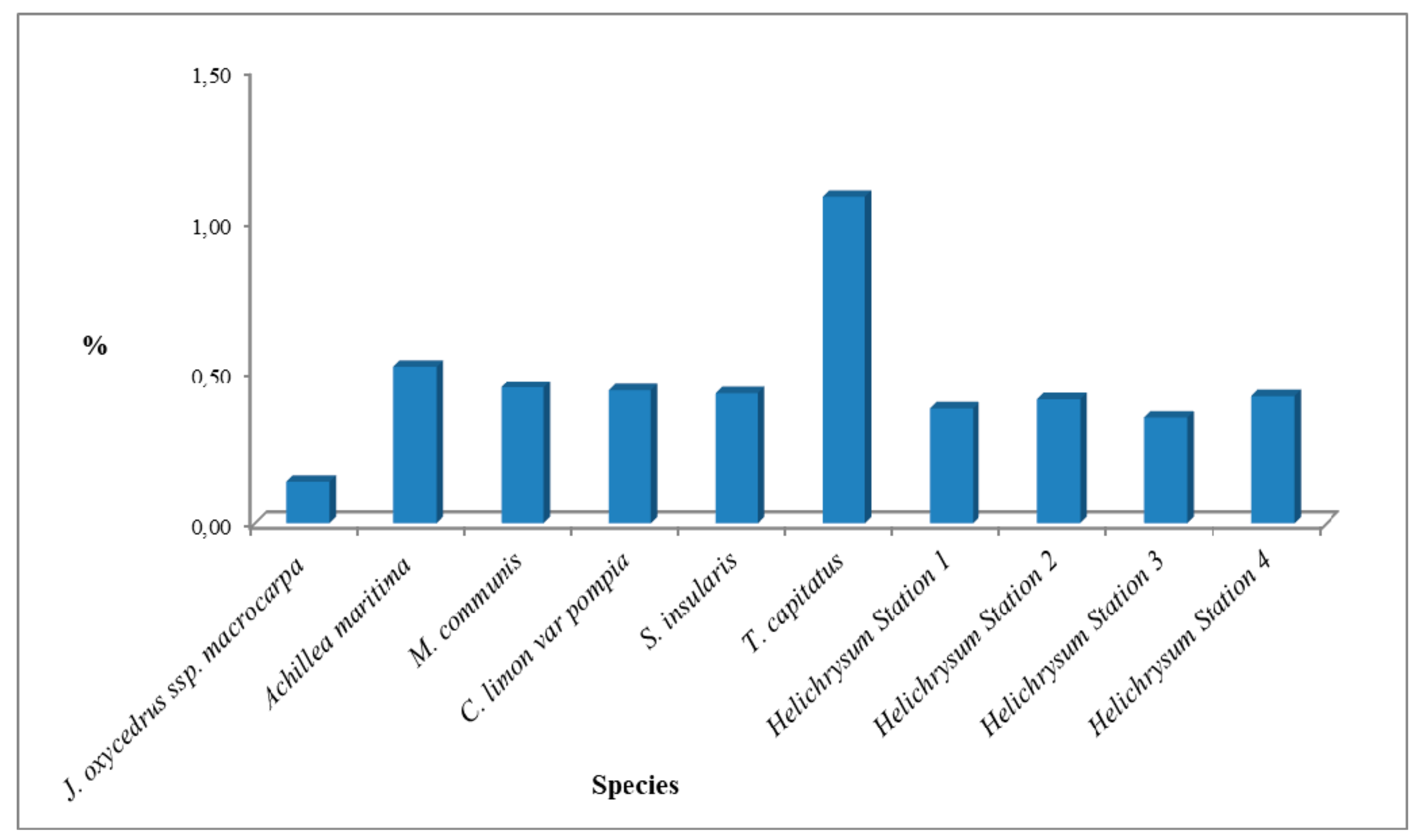
3.2. Antimicrobial Activity of the Essential Oils against C. acnes
3.3. Antibacterial Activity of M. communis, S. insularis and J. oxycedrus Essential Oils in the Presence of Sub-Inhibitory Concentrations of Chitosan
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- White, G.M. Recent findings in the epidemiologic evidence, classification and subtypes of acne vulgaris. J. Am. Acad. Dermatol. 1998, 39, S34–S37. [Google Scholar] [CrossRef]
- Melnik, B.C. Acne vulgaris: The metabolic syndrome of the pilosebaceous follicle. Clin. Dermatol. 2018, 36, 29–40. [Google Scholar] [CrossRef] [PubMed]
- Monfrecola, G.; Lembo, S.; Caiazzo, G.; de Vita, V.; Di Caprio, R.; Balato, A.; Fabbrocini, G. Mechanistic target of rapamycin (mTOR) expression is increased in acne patients’ skin. Exp. Dermatol. 2016, 25, 153–155. [Google Scholar] [CrossRef] [PubMed]
- Bergler-Czop, B. The aetiopathogenesis of acne vulgaris—What’s new? Int. J. Cosmet. Sci. 2014, 36, 187–194. [Google Scholar] [CrossRef] [PubMed]
- Zouboulis, C.C.; Eady, A.; Philpott, M.; Goldsmith, L.A.; Orfanos, C.; Cunliffe, W.C.; Rosenfield, R. What is the pathogenesis of acne? Exp. Dermatol. 2005, 14, 143–152. [Google Scholar] [CrossRef] [PubMed]
- Kumar, B.; Pathak, R.; Mary, P.B.; Jha, D.; Sardana, K.; Gautam, H.K. New insights into acne pathogenesis: Exploring the role of acne-associated microbial population. Dermatol. Sin. 2016, 34, 67–73. [Google Scholar] [CrossRef]
- Dessiniotis, C.; Katsambas, A.D. The role of Propionibacterium acnes in acne pathogenesis: Facts and controversies. Clin. Dermatol. 2010, 28, 2–7. [Google Scholar] [CrossRef] [PubMed]
- Omer, H.; McDowell, A.; Alexeyev, O.A. Understanding the role of Propionibacterium acnes in acne vulgaris: The critical importance of skin sampling methodologies. Clin. Dermatol. 2017, 35, 118–129. [Google Scholar] [CrossRef] [PubMed]
- Fitz-Gibbon, S.; Tomida, S.; Chiu, B.-H.; Nguyen, L.; Du, C.; Liu, M.; Elashoff, D.; Erfe, M.C.; Loncaric, A.; Kim, J.; et al. Propionibacterium acnes strain populations in the human skin microbiome associated with acne. J. Investig. Dermatol. 2013, 133, 2152–2160. [Google Scholar] [CrossRef] [PubMed]
- Harder, J.; Tsuruta, D.; Murakami, M.; Kurokawa, I. What is the role of antimicrobial peptides (AMP) in acne vulgaris? Exp. Dermatol. 2013, 22, 386–391. [Google Scholar] [CrossRef] [PubMed]
- Hosthota, A.; Bondade, S.; Basvaraja, V. Impact of acne vulgaris on quality of life and self-esteem. Cutis 2016, 98, 121–124. [Google Scholar] [PubMed]
- Kraft, J.; Freiman, A. Management of acne. Can. Med. Assoc. J. 2011, 183, E430–E435. [Google Scholar] [CrossRef] [PubMed]
- Harper, J.C. Use of oral contraceptives for management of acne vulgaris: Practical considerations in real world practice. Dermatol. Clin. 2016, 34, 159–165. [Google Scholar] [CrossRef] [PubMed]
- Harris, V.R.; Cooper, A.J. Modern management of acne. Med. J. Aust. 2017, 206, 41–45. [Google Scholar] [CrossRef]
- Azimi, H.; Fallah-Tafti, M.; Khakshur, A.A.; Abdollai, M. A review of phytotherapy of acne vulgaris: Perspective of new pharmacological treatments. Fitoterapia 2012, 83, 1306–1317. [Google Scholar] [CrossRef] [PubMed]
- Sinha, P.; Srivastava, S.; Mishra, N.; Prasad Yadav, N. New perspectives on antiacne plant drugs: Contribution to modern therapeutics. BioMed Res. Int. 2014, 2014, 301304. [Google Scholar] [CrossRef] [PubMed]
- Julianti, E.; Rajah, K.K.; Fidrianny, I. Antibacterial activity of ethanolic extract of cinnamon bark, honey, and their combination effects against acne-causing bacteria. Sci. Pharm. 2017, 85, 19. [Google Scholar] [CrossRef] [PubMed]
- Jantarat, C.; Sirathanarun, P.; Chuchue, T.; Konpian, A.; Sukkua, G.; Wongprasert, P. In vitro antimicrobial activity of gel containing the herbal ball extract against Propionibacterium acnes. Sci. Pharm. 2018, 86, 8. [Google Scholar] [CrossRef] [PubMed]
- Lertsatitthanakorn, P.; Taweechaisupapong, S.; Aromdee, C.; Khunkitti, W. In vitro bioactivities of essential oils used for acne control. Int. J. Aromather. 2006, 16, 43–49. [Google Scholar] [CrossRef]
- Zu, Y.; Yu, H.; Liang, L.; Fu, Y.; Efferth, T.; Liu, X.; Wu, N. Activities of ten essential oils towards Propionibacterium acnes and PC-3, A-549 and MCF-7 cancer cells. Molecules 2010, 15, 3200–3210. [Google Scholar] [CrossRef] [PubMed]
- Hammer, K.A. Treatment of acne with tea tree oil (melaleuca) product: A review of efficacy, tolerability and potential modes of action. Int. J. Antimicrob. Agents 2015, 45, 106–110. [Google Scholar] [CrossRef] [PubMed]
- NIST 2011 Library of Mass Spectra; Agilent Technologies Co.: Palo Alto, CA, USA, 2011.
- Adams, R.P. Identification of Essential Oil Components by Gas Chromatography/Mass Spectrometry; Allured Publishing Corporation: Carol Stream, IL, USA, 2007. [Google Scholar]
- Bonferoni, M.C.; Cerri, G.; de’ Gennaro, M.; Juliano, C.; Caramella, C. Zn++-exchanged clinoptilolite-rich rock as active carrier for antibiotics in anti-acne topical therapy. In vitro characterization and preliminary formulation studies. Appl. Clay Sci. 2007, 36, 95–102. [Google Scholar] [CrossRef]
- Oh, T.-H.; Kim, S.-S.; Yoon, W.-J.; Kim, J.-Y.; Yang, E.-J.; Lee, N.H.; Hyun, C.-G. Chemical composition and biological activities of Jeju Thymus quinquecostatus essential oils against Propionibacterium species inducing acne. J. Gen. Appl. Microbiol. 2009, 55, 63–68. [Google Scholar] [CrossRef] [PubMed]
- Du, E.; Gan, L.; Li, Z.; Wang, W.; Liu, D.; Guo, Y. In vitro antibacterial activity of thymol and carvacrol and their effects on broiler chickens challenged with Clostridium perfringens. J. Anim. Sci. Biotechnol. 2015, 6, 58. [Google Scholar] [CrossRef] [PubMed]
- Peana, A.T.; D’Aquila, A.T.; Panin, F.; Serra, G.; Pippia, P.; Moretti, M.D. Anti-inflammatory activity of linalool and linalyl acetate constituents of essential oils. Phytomedicine 2002, 9, 721–726. [Google Scholar] [CrossRef] [PubMed]
- Djihane, B.; Wafa, N.; Elkhamassa, S.; Pedro, H.J.; Maria, A.E.; Mohamed Mihoub, Z. Chemical constituents of Helichrysum italicum (Roth) G. Don essential oil and their antimicrobial activity against Gram-positive and Gram-negative bacteria, filamentous fungi and Candida albicans. Saudi Pharm. J. 2017, 25, 780–787. [Google Scholar] [CrossRef] [PubMed]
- Rossi, P.-G.; Berti, L.; Panighi, J.; Luciani, A.; Maury, J.; Muselli, A.; De Rocca Serra, D.; Gonny, M.; Bolla, J.-M. Antibacterial action of essential oils from Corsica. J. Essent. Oil Res. 2007, 19, 176–182. [Google Scholar] [CrossRef]
- Yesil Celiktas, O.; Hames Kocabas, E.E.; Bedir, E.; Vardar Sukan, F.; Ozek, T.; Baser, K.H.C. Antimicrobial activities of methanol extracts and essential oils of Rosmarinus officinalis, depending on location and seasonal variations. Food Chem. 2007, 100, 553–559. [Google Scholar] [CrossRef]
- Radulović, N.S.; Randjelović, P.J.; Stojanović, N.M.; Blagojević, P.D.; Stojanović-Radić, Z.Z.; Ilić, I.R.; Djordjević, V.B. Toxic essential oils. Part II: Chemical, toxicological, pharmacological and microbiological profiles of Artemisia annua L. volatiles. Food Chem. Toxicol. 2013, 58, 37–49. [Google Scholar] [CrossRef] [PubMed]
- Mneimne, M.; Baydoun, S.; Nemer, N.; Apostolides, N.A. Chemical Composition and Antimicrobial Activity of Essential Oils Isolated from Aerial Parts of Prangos asperula Boiss. (Apiaceae) Growing Wild in Lebanon. Med. Aromat. Plants 2016, 5, 242–246. [Google Scholar] [CrossRef]
- Kong, M.; Chen, X.G.; Xing, K.; Park, H.J. Antimicrobial properties of chitosan and mode of action: A state of the art review. Int. J. Food Microbiol. 2010, 144, 51–63. [Google Scholar] [CrossRef] [PubMed]
- Champer, J.; Patel, J.; Fernando, N.; Salehi, E.; Wong, V.; Kim, J. Chitosan against cutaneous pathogens. AMB Express 2013, 3, 37. [Google Scholar] [CrossRef] [PubMed]
- Juliano, C.; Demurtas, C.; Piu, L. In vitro study of the anticandidal activity of Melaleuca alternifolia (tea tree) essential oil combined with chitosan. Flavour Fragr. J. 2008, 23, 227–231. [Google Scholar] [CrossRef]
- Wang, L.; Liu, F.; Jiang, Y.; Chai, Z.; Li, P.; Cheng, Y.; Jing, H.; Leng, X. Synergistic antimicrobial activities of natural essential oils with chitosan films. J. Agric. Food Chem. 2011, 59, 12411–12419. [Google Scholar] [CrossRef] [PubMed]
- De Oliveira, K.Á.R.; Berger, L.R.R.; De Araújo, S.A.; Câmara, M.P.S.; De Souza, E.L. Synergistic mixtures of chitosan and Mentha piperita L. essential oil to inhibit Colletotrichum species and anthracnose development in mango cultivar Tommy Atkins. Food Microbiol. 2017, 66, 96–103. [Google Scholar] [CrossRef] [PubMed]
- Mimica-Dukić, N.; Bugarin, D.; Grbović, S.; Mitić-Ćulafić, D.; Vuković-Gačić, B.; Orčić, D.; Jovin, E.; Couladis, M. Essential oil of Myrtus communis L. as a potential antioxidant and antimutagenic agent. Molecules 2010, 15, 2759–2770. [Google Scholar] [CrossRef] [PubMed]
- Zheljazkov, V.D.; Semerdjieva, I.B.; Dincheva, I.; Kacaniova, M.; Astatkie, T.; Radoukova, T.; Schlegel, V. Antimicrobial and antioxidant activity of Juniper galbuli essential oil constituents eluted at different times. Ind. Crops Prod. 2017, 109, 529–537. [Google Scholar] [CrossRef]
- Höferl, M.; Stoilova, I.; Schmidt, E.; Wanner, J.; Jirovetz, L.; Trifonova, D.; Krastev, L.; Krastanov, A. Chemical composition and antioxidant properties of Juniper berry (Juniperus communis L.) essential oil. Action of the essential oil on the antioxidant protection of Saccharomyces cerevisiae model organism. Antioxidants 2014, 3, 81–98. [Google Scholar] [CrossRef] [PubMed]
- Briganti, S.; Picardo, M. Antioxidant activity, lipid peroxidation and skin disease. What’s new? J. Eur. Acad. Dermatol. Venereol. 2003, 17, 663–669. [Google Scholar] [CrossRef] [PubMed]
| e.o. M. communis | e.o. S. insularis | e.o. J. oxycedrus | |
|---|---|---|---|
| MIC of essential oil | >4 mg/mL | 1 mg/mL | 1 mg/mL |
| MIC of essential oil + 0.031 mg/mL chitosan | 1.5 mg/mL | 0.063 mg/mL | 0.094 mg/mL |
| Essential Oil | M.I.C. | M.B.C. |
|---|---|---|
| Thymus capitatus | 0.25 mg/mL | 0.5 mg/mL |
| Santolina insularis | 1 mg/mL | 2 mg/mL |
| Helichrysum microphyllum subsp. tyrrhenicum (Station 1) | 1 mg/mL | 2 mg/mL |
| Helichrysum microphyllum subsp. tyrrhenicum (Station 2) | 2 mg/mL | 4 mg/mL |
| Helichrysum microphyllum subsp. tyrrhenicum (Station 3) | 1 mg/mL | n.d. |
| Achillea maritima | 2 mg/mL | 4 mg/mL |
| Juniperus oxycedrus | 1 mg/mL | 2 mg/mL |
| Citrus limon var. pompia | 4 mg/mL | >4 mg/mL |
| Lavandula officinalis | 4 mg/mL | n.d. |
| Myrtus communis | >4 mg/mL | >4 mg/mL |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Juliano, C.; Marchetti, M.; Pisu, M.L.; Usai, M. In Vitro Antimicrobial Activity of Essential Oils from Sardinian Flora against Cutibacterium (Formerly Propionibacterium) acnes and Its Enhancement by Chitosan. Sci. Pharm. 2018, 86, 40. https://doi.org/10.3390/scipharm86030040
Juliano C, Marchetti M, Pisu ML, Usai M. In Vitro Antimicrobial Activity of Essential Oils from Sardinian Flora against Cutibacterium (Formerly Propionibacterium) acnes and Its Enhancement by Chitosan. Scientia Pharmaceutica. 2018; 86(3):40. https://doi.org/10.3390/scipharm86030040
Chicago/Turabian StyleJuliano, Claudia, Mauro Marchetti, Maria Luisa Pisu, and Marianna Usai. 2018. "In Vitro Antimicrobial Activity of Essential Oils from Sardinian Flora against Cutibacterium (Formerly Propionibacterium) acnes and Its Enhancement by Chitosan" Scientia Pharmaceutica 86, no. 3: 40. https://doi.org/10.3390/scipharm86030040
APA StyleJuliano, C., Marchetti, M., Pisu, M. L., & Usai, M. (2018). In Vitro Antimicrobial Activity of Essential Oils from Sardinian Flora against Cutibacterium (Formerly Propionibacterium) acnes and Its Enhancement by Chitosan. Scientia Pharmaceutica, 86(3), 40. https://doi.org/10.3390/scipharm86030040





