Effect of Piperine on Skin Permeation of Curcumin from a Bacterially Derived Cellulose-Composite Double-Layer Membrane for Transdermal Curcumin Delivery
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
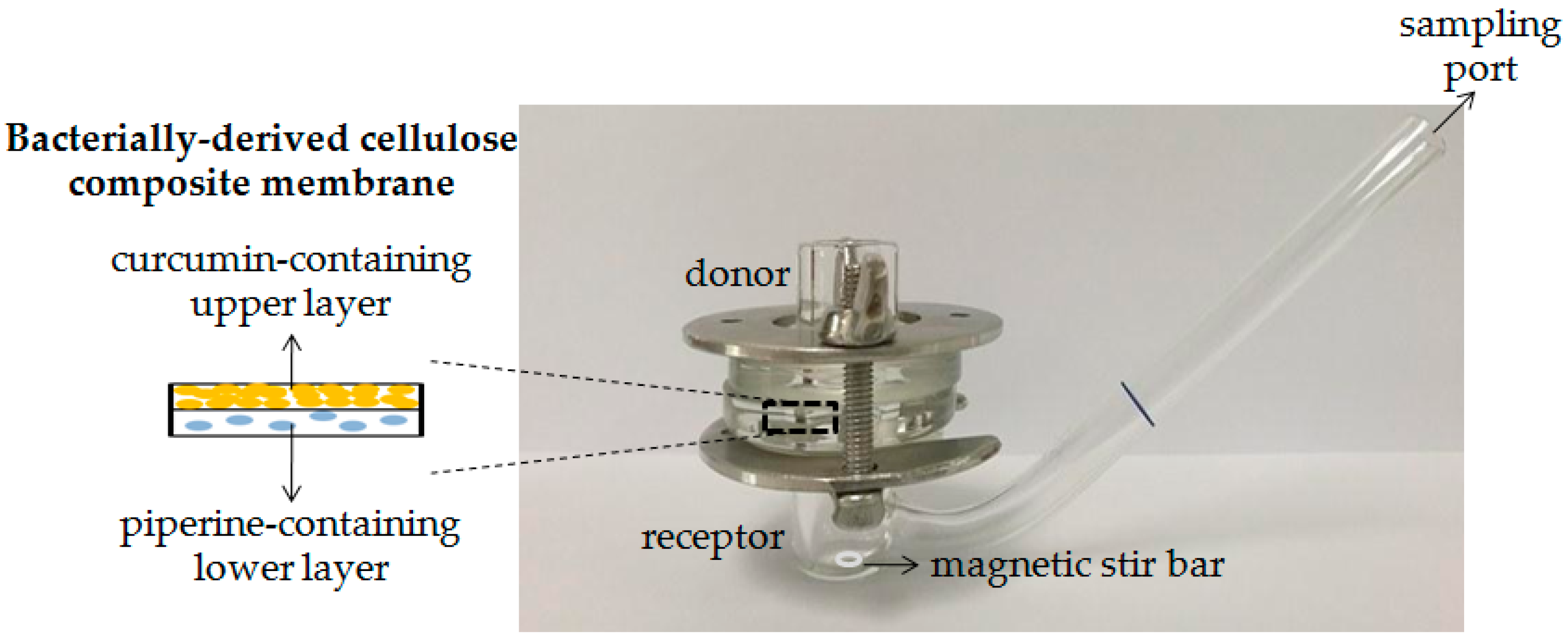
2.2. Preparation of a Bacterially Derived Cellulose-Composite Membrane
2.3. Physical Property Test
2.3.1. Swelling
2.3.2. Tensile Strength
2.3.3. Differential Scanning Calorimetry
2.4. Curcumin and Piperine Content
2.5. Stability of Curcumin in the Composite Membrane
2.6. Release Study
2.7. In Vitro Skin Permeation Study
3. Results
3.1. Preparation of Bacterially Derived Cellulose-Composite Membrane
3.2. Physical Properties
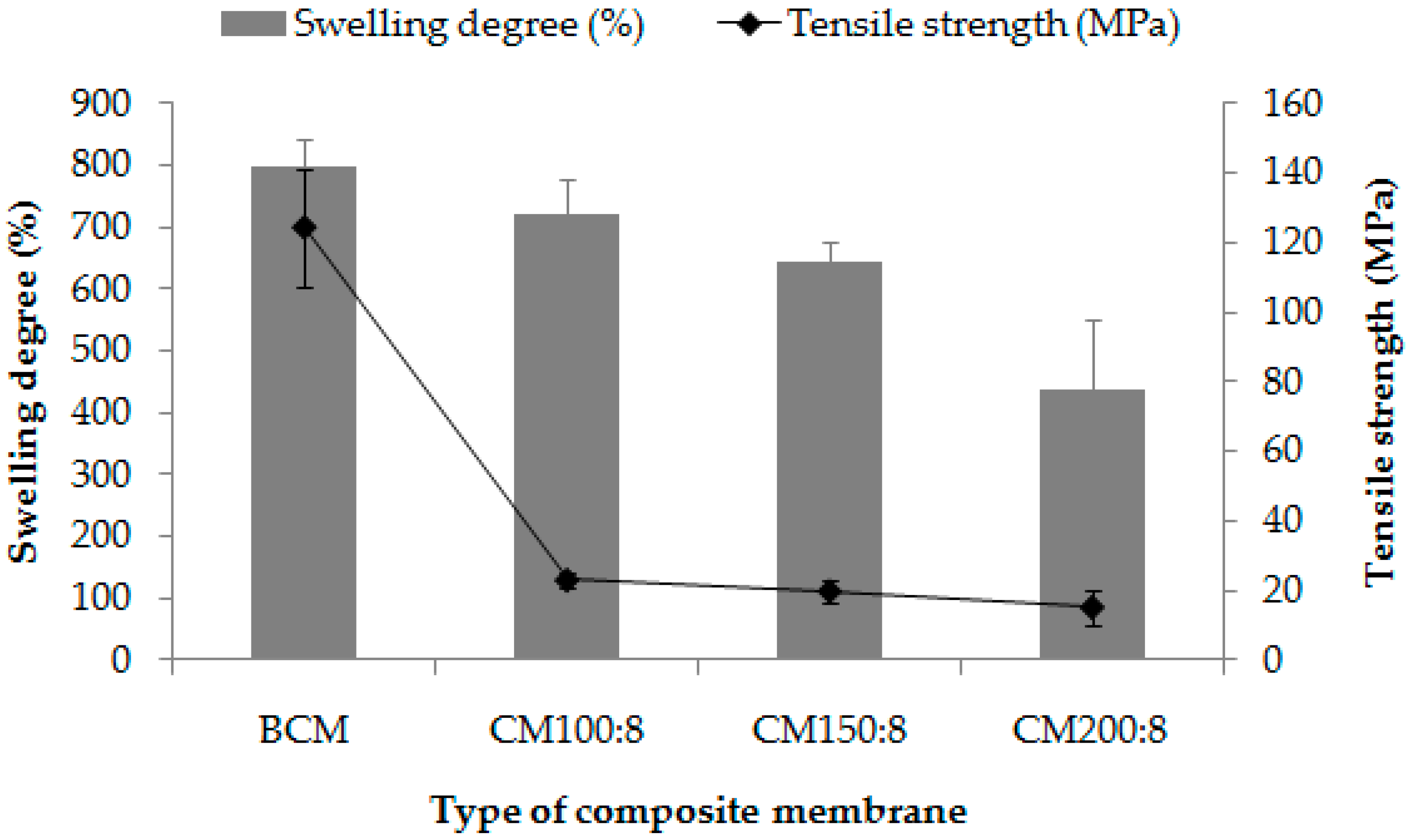
3.2.1. Swelling and Tensile Strength
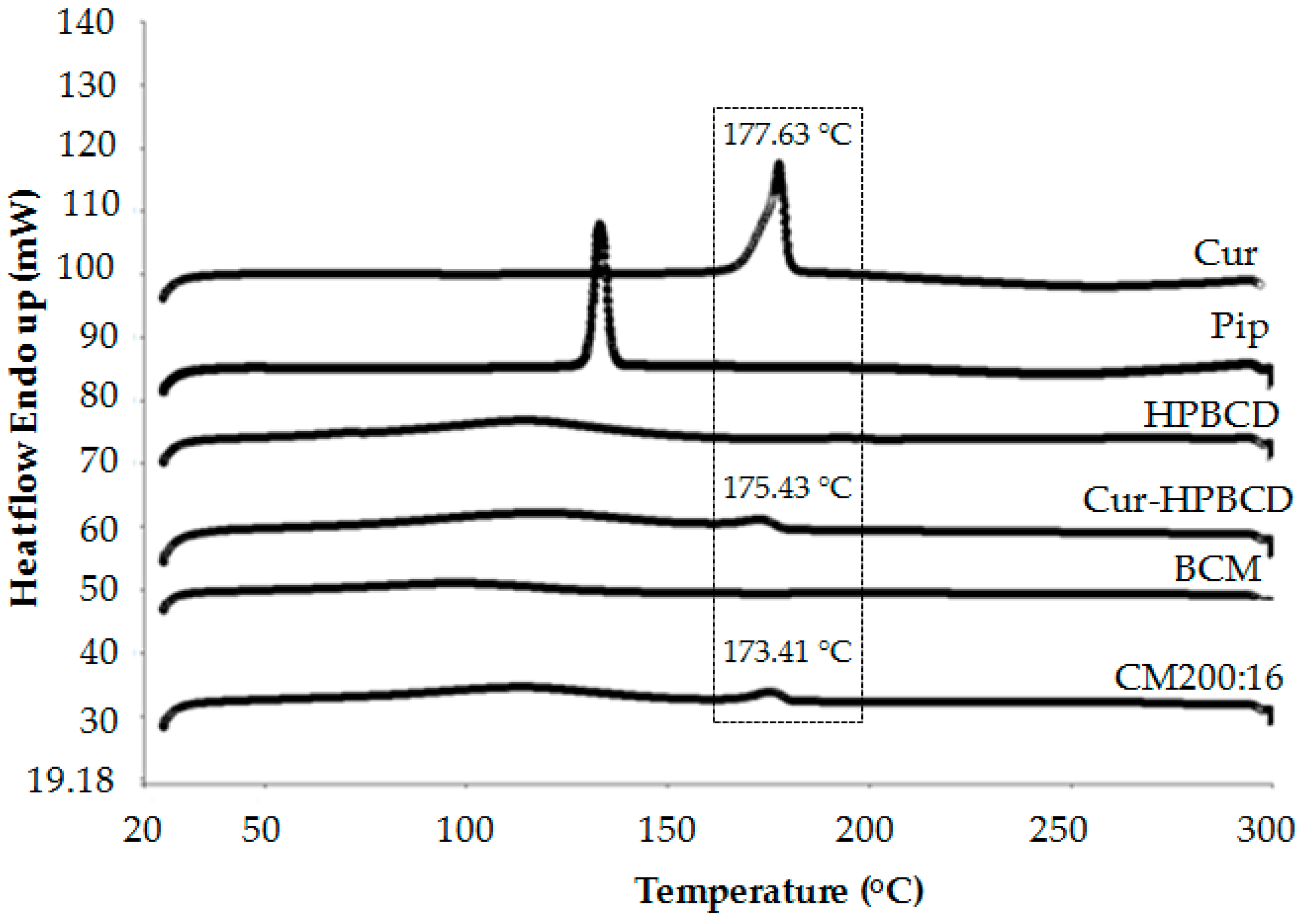
3.2.2. Differential Scanning Calorimetry
3.3. Curcumin and Piperine Content
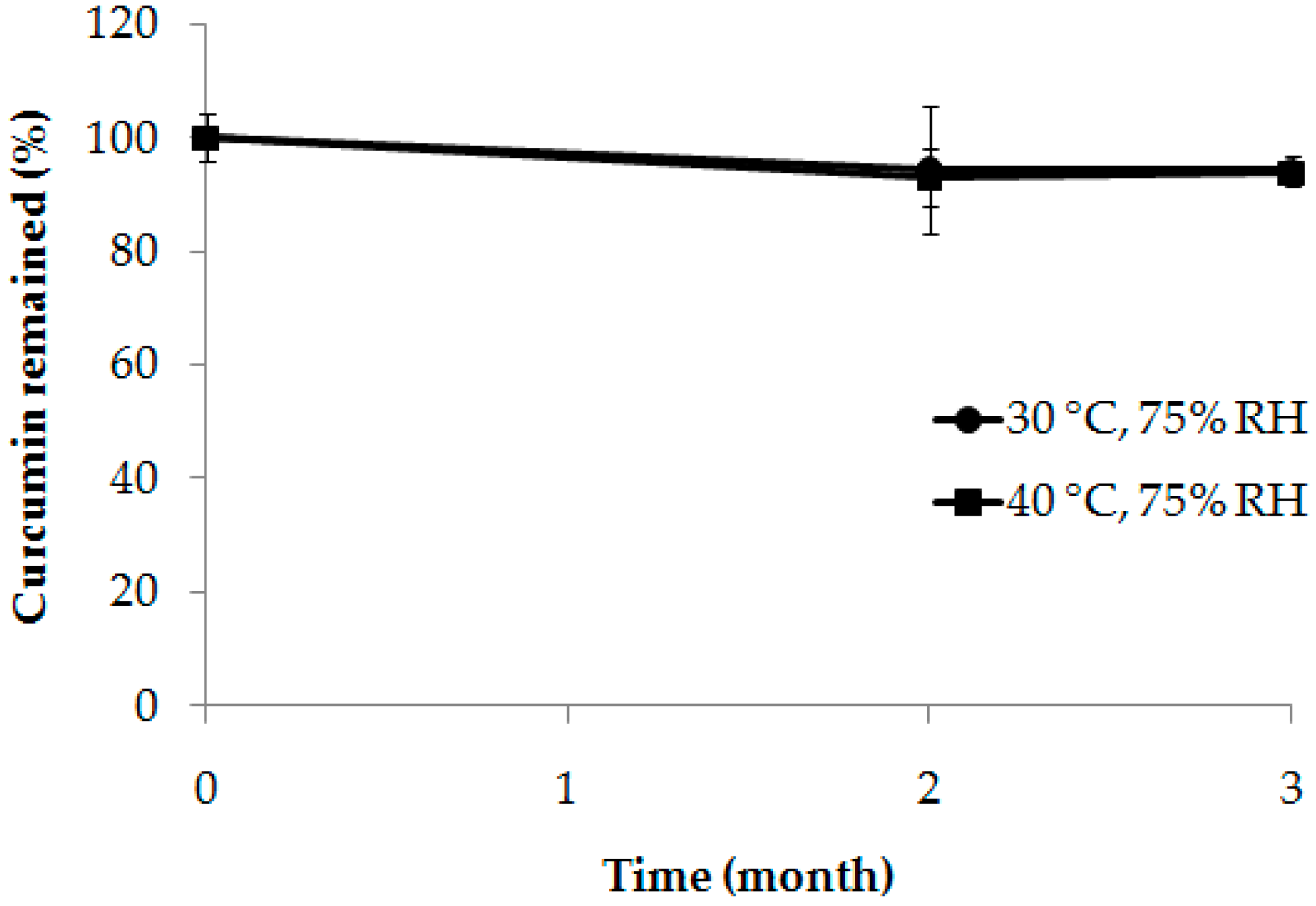
3.4. Stability of Curcumin in the Composite Membrane
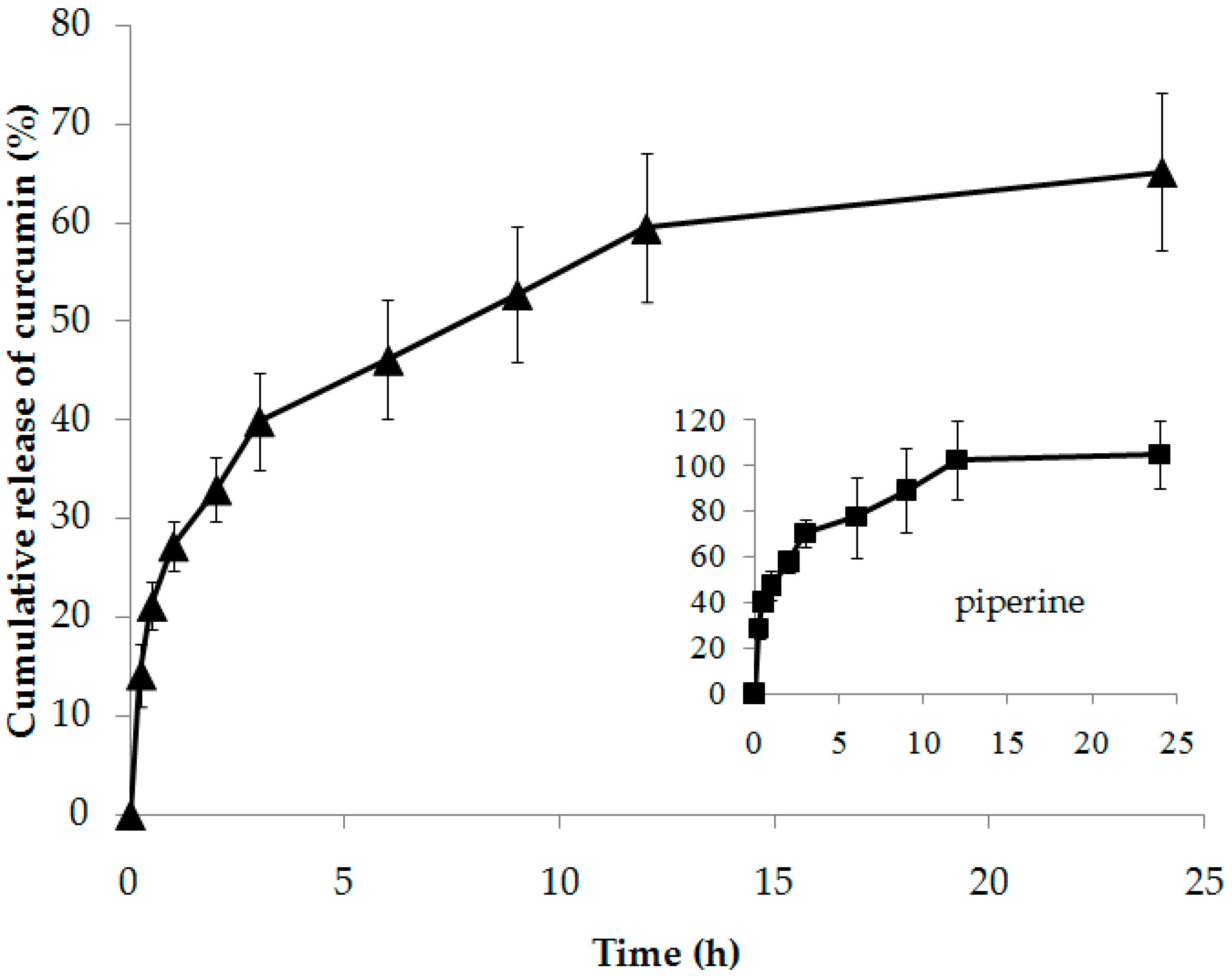
3.5. Release Study
3.6. In Vitro Skin Permeation Study
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- National List of Essential Medicines, 2018–Thailand. Available online: http://www.ratchakitcha.soc.go.th/DATA/PDF/2561/E/014/4.PDF (accessed on 8 July 2018).
- Priyadarsini, K.I. The Chemistry of Curcumin: From extraction to therapeutic agent. Molecules 2014, 19, 20091–20112. [Google Scholar] [CrossRef] [PubMed]
- Maheshwari, R.K.; Singh, A.K.; Gaddipati, J.; Srimal, R.C. Multiple biological activities of curcumin: A short review. Life Sci. 2006, 78, 2081–2087. [Google Scholar] [CrossRef] [PubMed]
- Gupta, S.C.; Patchva, S.; Koh, W.; Aggarwal, B. Discovery of curcumin, a component of the golden spice, and its miraculous biological activities. Clin. Exp. Pharmacol. Physiol. 2012, 39, 283–299. [Google Scholar] [CrossRef] [PubMed]
- Amalraj, A.; Pius, A.; Gopi, S.; Gopi, S. Biological activities of curcuminoids, other biomolecules from turmeric and their derivatives: A review. J. Tradit. Complement. Med. 2017, 7, 205–233. [Google Scholar] [CrossRef] [PubMed]
- Vareed, S.K.; Kakarala, M.; Ruffin, M.T.; Crowell, J.A.; Normolle, D.P.; Djuric, Z.; Brenner, D.E. Pharmacokinetics of curcumin conjugate metabolites in healthy human subjects. Cancer Epidemiol. Biomark. Prev. 2008, 17, 1411–1417. [Google Scholar] [CrossRef] [PubMed]
- Kumar, S.; Kesharwani, S.S.; Mathus, H.; Tyagi, M.; Bhat, G.J.; Tummala, H. Molecular complexation of curcumin with pH sensitive cationic copolymer enhances the aqueous solubility, stability and bioavailability of curcumin. Eur. J. Pharm. Sci. 2016, 82, 86–96. [Google Scholar] [CrossRef] [PubMed]
- Wahlang, B.; Pawar, Y.B.; Bansal, A.K. Identification of permeability-related hurdles in oral delivery of curcumin using the Caco-2 cell model. Eur. J. Pharm. Biopharm. 2011, 77, 275–282. [Google Scholar] [CrossRef] [PubMed]
- Sharma, R.A.; Steward, W.P.; Gescher, A.J. Pharmacokinetics and pharmacodynamics of curcumin. Adv. Exp. Med. Biol. 2007, 595, 453–470. [Google Scholar] [PubMed]
- Prasad, S.; Tyagi, A.; Aggarwal, B. Recent developments in delivery, bioavailability, absorption and metabolism of curcumin: The golden pigment from golden spice. Cancer Res. Treat. 2014, 46, 2–18. [Google Scholar] [CrossRef] [PubMed]
- Pathan, I.B.; Jaware, B.P.; Shelke, S.; Ambekar, W. Curcumin loaded ethosomes for transdermal application: Formulation, optimization, in-vitro and in-vivo study. J. Drug Deliv. Sci. Technol. 2018, 44, 49–57. [Google Scholar] [CrossRef]
- Angelova, A.; Garamus, V.M.; Angelov, B.; Tian, Z.; Li, Y.; Zou, A. Advances in structural design of lipid-based nanoparticle carriers for delivery of macromolecular drugs, phytochemicals and anti-tumor agents. Adv. Colloid Interface Sci. 2017, 249, 331–345. [Google Scholar] [CrossRef] [PubMed]
- Guerzoni, L.P.; Nicolas, V.; Angelova, A. In Vitro Modulation of TrkB Receptor Signaling upon Sequential Delivery of Curcumin-DHA Loaded Carriers Towards Promoting Neuronal Survival. Pharm. Res. 2017, 34, 492–505. [Google Scholar] [CrossRef] [PubMed]
- Kesarwani, K.; Gupta, R. Bioavailability enhancers of herbal origin: An overview. Asian Pac. J. Trop. Biomed. 2013, 3, 253–266. [Google Scholar] [CrossRef]
- Atal, C.K. A breakthrough in drug bioavailability: A clue from age old wisdom of Ayurveda. I.D.M.A. Bull. 1979, 10, 483–484. [Google Scholar]
- Shaikh, J.; Ankola, D.D.; Beniwal, V.; Singh, D.; Kumar, M.N. Nanoparticle encapsulation improves oral bioavailability of curcumin by at least 9-fold when compared to curcumin administered with piperine as absorption enhancer. Eur. J. Pharm. Sci. 2009, 37, 223–230. [Google Scholar] [CrossRef] [PubMed]
- Moorthi, C.; Krishnan, K.; Manavalan, R.; Kathiresan, K. Preparation and characterization of curcumin-piperine dual drug loaded nanoparticles. Asian Pac. J. Trop. Biomed. 2012, 2, 841–848. [Google Scholar] [CrossRef]
- Boddupalli, B.M.; Anisetti, R.N.; Mamani, R.; Malothu, N. Enhanced pharmacokinetics of omeprazole when formulated as gastroretentive microspheres along with piperine. Asian Pac. J. Trop. Dis. 2014, 1, S129–S133. [Google Scholar] [CrossRef]
- Di, X.; Wang, X.; Di, X.; Liu, Y. Effect of piperine on the bioavailability and pharmacokinetics of emodin in rats. J. Pharm. Biomed. Anal. 2015, 115, 144–149. [Google Scholar] [CrossRef] [PubMed]
- Cherniakov, I.; Izgelov, D.; Barasch, D.; Davidson, E.; Domb, A.J.; Hoffman, A. Piperine-pro-nanolipospheres as a novel oral delivery system of cannabinoids: Pharmacokinetic evaluation in healthy volunteers in comparison to buccal spray administration. J. Control. Release 2017, 266, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Baspinar, Y.; Ustundas, M.; Bayraktar, O.; Zezgin, C. Curcumin and piperine loaded zein-chitosan nanoparticles: Development and in-vitro characterization. Saudi Pharm. J. 2018, 26, 323–334. [Google Scholar] [CrossRef] [PubMed]
- Jin, Z.H.; Qiu, W.; Liu, H.; Jiang, X.H.; Wang, L. Enhancement of oral bioavailability and immune response of Ginsenoside Rh2 by co-administration with piperine. Chin. J. Nat. Med. 2018, 16, 143–149. [Google Scholar] [CrossRef]
- Khajuria, N.; Zutshi, U. Piperine modulates permeability characteristics of intestine by inducing alterations in membrane dynamics: Influence on brush border membrane fluidity, ultrastructure and enzyme kinetics. Phytomedicine 2002, 9, 224–231. [Google Scholar] [CrossRef] [PubMed]
- Atal, N.; Bedi, K.L. Bioenhancers: Revolutionary concept to market. J. Ayurveda Integr. Med. 2010, 1, 96–99. [Google Scholar] [CrossRef] [PubMed]
- Shoba, G.; Joy, D.; Joseph, T.; Majeed, M.; Rajendran, R.; Srinivas, P.S. Influence of piperine on the pharmacokinetics of curcumin in animals and human volunteers. Planta Med. 1998, 64, 353–356. [Google Scholar] [CrossRef] [PubMed]
- Kaushal, N.; Jain, S.; Kondaiah, P.; Tiwary, A.K. Influence of piperine on transcutaneous permeation of repaglinide in rats and on tight junction proteins in HaCaT cells: Unveiling the mechanisms for enhanced permeation. Sci. Pharm. 2009, 77, 877–897. [Google Scholar] [CrossRef]
- Shah, K.K.; Shiradkar, M.R.; Bindu, V.H. Transdermal delivery of aceclofenac: Effect of piperine and itsmechanism of action. Int. J. Pharma Bio Sci. 2011, 2, 10–18. [Google Scholar]
- Das, A.; Ahmed, A.B. Natural permeation enhancer for transdermal drug delivery system and permeation evaluation: A review. Asian J. Pharm. Clin. Res. 2017, 10, 5–7. [Google Scholar] [CrossRef]
- Patil, V.M.; Das, S.; Balasubramanian, K. Quantum chemical and docking insights into bioavailability enhancement of curcumin by piperine in pepper. J. Phys. Chem. A 2016, 120, 3643–3653. [Google Scholar] [CrossRef] [PubMed]
- Liu, C.H.; Chang, F.Y.; Hung, D.K. Terpene microemulsions for transdermal curcumin delivery: Effects of terpenes and cosurfactants. Colloids Surf. B Biointerfaces 2011, 82, 63–70. [Google Scholar] [CrossRef] [PubMed]
- Tsai, Y.H.; Yang, Y.N.; Ho, Y.C.; Tsai, M.L.; Mi, F.L. Drug release and antioxidant/antibacterial activities of silymarin-zein nanoparticle/bacterial cellulose nanofiber composite films. Carbohydr. Polym. 2018, 180, 286–296. [Google Scholar] [CrossRef] [PubMed]
- Qui, Y.; Qiu, L.; Cui, J.; Wei, Q. Bacterial cellulose and bacterial cellulose-vaccarin membranes for wound healing. Mater. Sci. Eng. C Mater. Biol. Appl. 2016, 59, 303–309. [Google Scholar]
- Yadav, V.R.; Suresh, S.; Devi, K.; Yadav, S. Effect of cyclodextrin complexation of curcumin on its solubility and antiangiogenic and anti-inflammatory activity in rat colitis model. AAPS PharmSciTech. 2009, 10, 752–762. [Google Scholar] [CrossRef] [PubMed]
- Jantarat, C.; Sirathanarun, P.; Ratanapongsai, S.; Watcharakan, P.; Sunyapong, S.; Wadu, A. Curcumin-hydroxypropyl-β-cyclodextrin inclusion complex preparation methods: Effect of common solvent evaporation, freeze drying, and pH shift on solubility and stability of curcumin. Trop. J. Pharm. Res. 2014, 13, 1215–1223. [Google Scholar] [CrossRef]
- Kharat, M.; Du, Z.; Zhang, G.; McClements, D.J. Physical and chemical stability of curcumin in aqueous solutions and emulsions: Impact of pH, temperature, and molecular environment. J. Agric. Food Chem. 2017, 65, 1525–1532. [Google Scholar] [CrossRef] [PubMed]
- Shin, G.H.; Li, J.; Cho, J.H.; Kim, J.T.; Park, H.J. Enhancement of curcumin solubility by phase change from crystalline to amorphous in cur-TPGS nanosuspension. J. Food Sci. 2016, 81, N494–N501. [Google Scholar] [CrossRef] [PubMed]
- Shao, B.; Cui, C.; Ji, H.; Tang, J.; Wang, Z.; Liu, H.; Qin, M.; Li, X.; Wu, L. Enhanced oral bioavailability of piperine by selfemulsifying drug delivery systems: In vitro, in vivo and in situ intestinal permeability studies. Drug Deliv. 2015, 22, 740–747. [Google Scholar] [CrossRef] [PubMed]
- Basak, S.C.; Mills, D.; Mumtaz, M.M. A quantitative structure-activity relationship (QSAR) study of dermal absorption using theoretical molecular descriptors. SAR QSAR Environ. Res. 2007, 18, 45–55. [Google Scholar] [CrossRef] [PubMed]


| Type of Composite Membrane | Composition (mg) | |
|---|---|---|
| Curcumin | Piperine | |
| Blank composite membrane (BCM) | - | - |
| Controlled composite membrane (CCM) | 200 | - |
| CM100:8 | 100 | 8 |
| CM150:8 | 150 | 8 |
| CM200:4 | 200 | 4 |
| CM200:8 | 200 | 8 |
| CM200:16 | 200 | 16 |
| Substance in CM200:8 | Lag Time (h) | D (mm/s) |
|---|---|---|
| Curcumin | 0.56 ± 0.46 | 0.27 ± 0.04 |
| Piperine | 0.47 ± 0.41 | 8.17 ± 0.87 |
| Type of Composite Membrane | Lag Time (h) | J (µg/cm2/h) | ||
|---|---|---|---|---|
| Curcumin | Piperine | Curcumin | Piperine | |
| CCM | 1.90 ± 0.63 | - | 4.29 ± 1.70 | - |
| CM200:4 | 2.62 ± 0.63 | 2.78 ± 1.98 | 5.51 ± 1.87 | 2.30 ± 1.37 |
| CM200:8 | 2.46 ± 0.36 | 2.54 ± 1.12 | 6.93 ± 0.76 | 7.03 ± 1.72 |
| CM200:16 | 1.91 ± 0.41 | 1.99 ± 0.99 | 8.10 ± 1.51 | 17.99 ± 8.87 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jantarat, C.; Sirathanarun, P.; Boonmee, S.; Meechoosin, W.; Wangpittaya, H. Effect of Piperine on Skin Permeation of Curcumin from a Bacterially Derived Cellulose-Composite Double-Layer Membrane for Transdermal Curcumin Delivery. Sci. Pharm. 2018, 86, 39. https://doi.org/10.3390/scipharm86030039
Jantarat C, Sirathanarun P, Boonmee S, Meechoosin W, Wangpittaya H. Effect of Piperine on Skin Permeation of Curcumin from a Bacterially Derived Cellulose-Composite Double-Layer Membrane for Transdermal Curcumin Delivery. Scientia Pharmaceutica. 2018; 86(3):39. https://doi.org/10.3390/scipharm86030039
Chicago/Turabian StyleJantarat, Chutima, Pornpak Sirathanarun, Somruedee Boonmee, Wanida Meechoosin, and Husna Wangpittaya. 2018. "Effect of Piperine on Skin Permeation of Curcumin from a Bacterially Derived Cellulose-Composite Double-Layer Membrane for Transdermal Curcumin Delivery" Scientia Pharmaceutica 86, no. 3: 39. https://doi.org/10.3390/scipharm86030039
APA StyleJantarat, C., Sirathanarun, P., Boonmee, S., Meechoosin, W., & Wangpittaya, H. (2018). Effect of Piperine on Skin Permeation of Curcumin from a Bacterially Derived Cellulose-Composite Double-Layer Membrane for Transdermal Curcumin Delivery. Scientia Pharmaceutica, 86(3), 39. https://doi.org/10.3390/scipharm86030039





