Honey Bee as Alternative Medicine to Treat Eleven Multidrug-Resistant Bacteria Causing Urinary Tract Infection during Pregnancy
Abstract
:1. Introduction
2. Materials and Methods
2.1. Honey Bee Samples
2.2. Strains
2.3. Honey Samples Analysis
2.3.1. Sensory Analysis
2.3.2. pH Measurement
2.3.3. Color Intensity
2.4. Antibacterial Activity
2.4.1. Wells Assay
2.4.2. Spectrophotometric Assay for MIC Determination
2.4.3. Minimum Bactericidal Concentration Determination
2.4.4. Time-Kill Assay
2.5. Statistical Analysis
3. Results
3.1. Antibiotic Sensitivity
3.2. Honey Analysis
3.3. Inhibitory Diameters Determination
3.4. Determination of MICs and MBCs
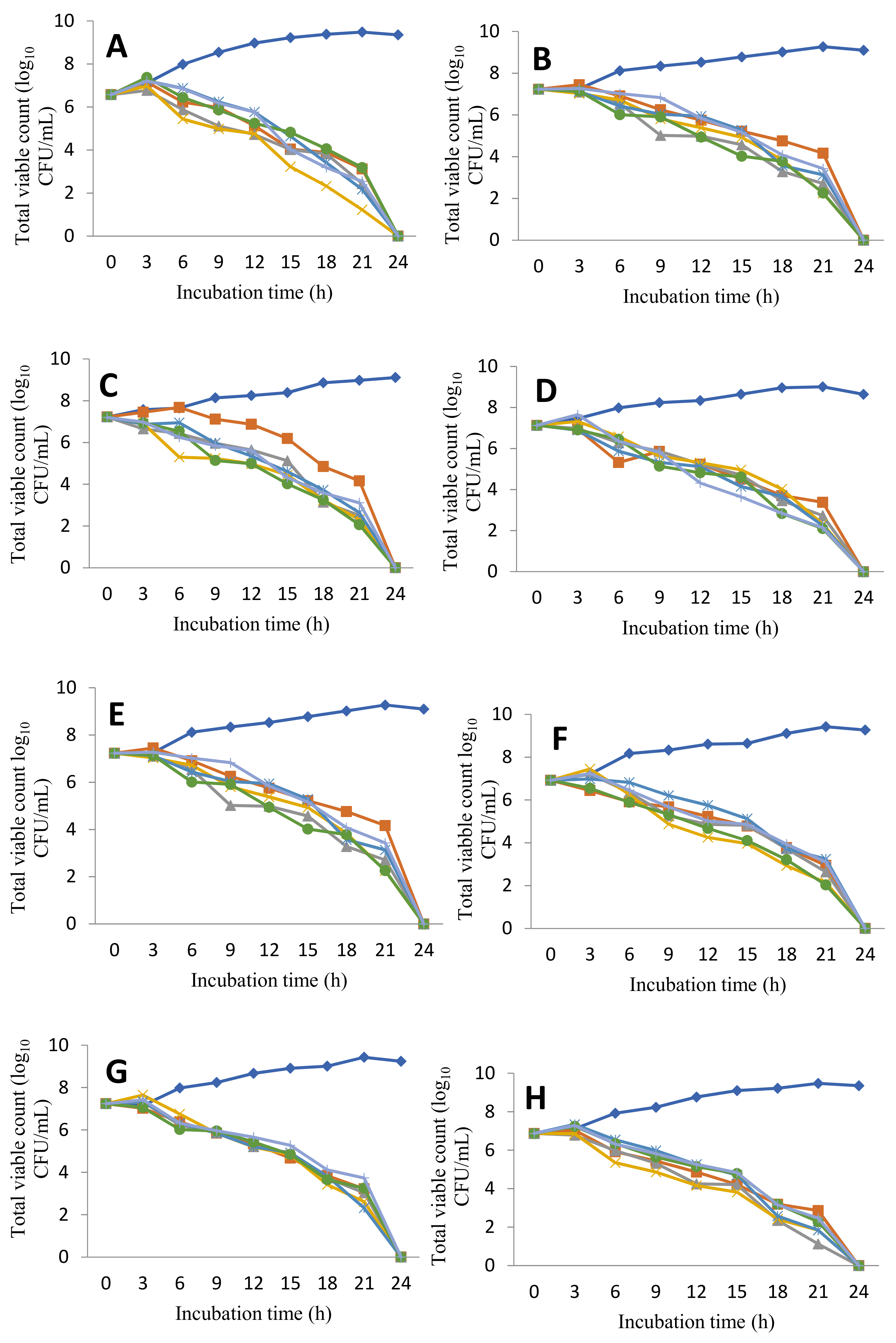
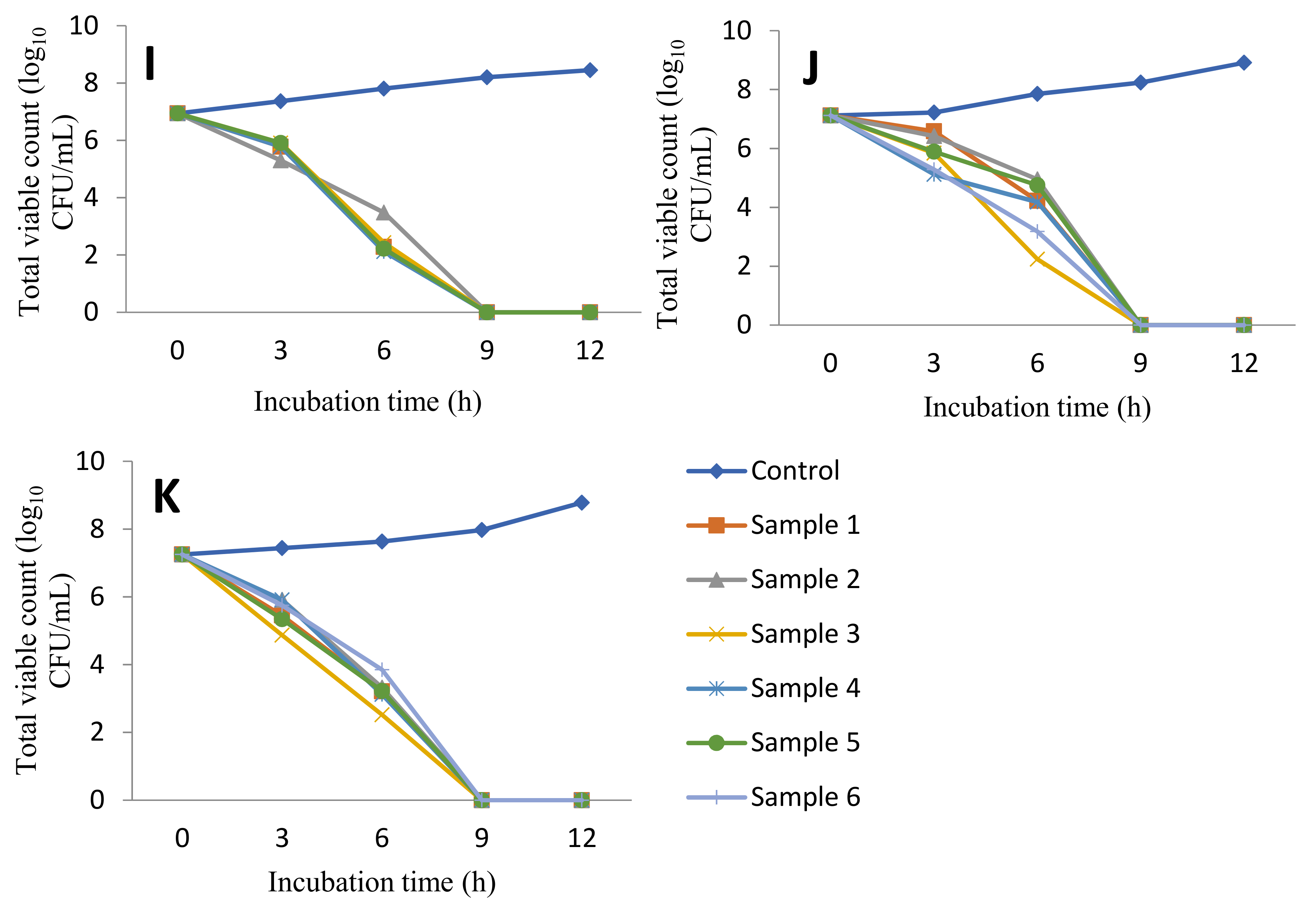
3.5. Time Kill Curve
4. Discussion
5. Conclusions
Author Contributions
Conflicts of Interest
References
- Curtiss, N.; Meththananda, I.; Duckett, J. Urinary tract infection in obstetrics and gynaecology. Obstet. Gynaecol. Reprod. Med. 2017, 27, 261–265. [Google Scholar] [CrossRef]
- Flores-Mireles, A.L.; Walker, J.N.; Caparon, M.; Hultgren, S.J. Urinary tract infections: Epidemiology, mechanisms of infection and treatment options. Nat. Rev. Microbiol. 2015, 13, 269–284. [Google Scholar] [CrossRef] [PubMed]
- Turpen, H.C. Frequent Urinary Tract Infection. Physician Assist. Clin. 2018, 3, 55–67. [Google Scholar] [CrossRef]
- Michelim, L.; Bosi, G.R.; Comparsi, E. Urinary Tract Infection in Pregnancy: Review of Clinical Management. J. Clin. Nephrol. Res. 2016, 3, 1030–1037. [Google Scholar]
- Souza, R.B.; Trevisol, D.J.; Schuelter-Trevisol, F. Bacterial sensitivity to fosfomycin in pregnant women with urinary infection. Braz. J. Infect. Dis. 2015, 19, 319–323. [Google Scholar] [CrossRef] [PubMed]
- Usta, T.A.; Dogan, O.; Ates, U.; Yucel, B.; Onar, Z.; Kaya, E. Comparison of single-dose and multiple-dose antibiotics for lower urinary tract infection in pregnancy. Int. J. Gynaecol. Obstet. 2011, 114, 229–233. [Google Scholar] [CrossRef] [PubMed]
- Glaser, A.P.; Schaeffer, A.J. Urinary tract infection and bacteriuria in pregnancy. Urol. Clin. N. Am. 2015, 42, 547–560. [Google Scholar] [CrossRef] [PubMed]
- Ronald, A.; Nicolle, L.E.; Harding, G. Single dose treatment failure in women with acute cystitis. Infection 1992, 20, S276–S279. [Google Scholar] [CrossRef] [PubMed]
- Sibi, G.; Pinki, K.; Kabungulundabungi, N. Antibiotic sensitivity pattern from pregnant women with urinary tract infection in Bangalore, India. Asian Pac. J. Trop. Med. 2014, 7, S116–S120. [Google Scholar] [CrossRef]
- Hecini-Hannachi, A.; Bentchouala, C.; Lezzar, A.; Laouar, H.; Benlabed, K.; Smati, F. Multidrug-resistant bacteria isolated from patients hospitalized in intensive care unit in university hospital of Constantine, Algeria (2011–2015). Afr. J. Microbiol. Res. 2016, 10, 1328–1336. [Google Scholar] [CrossRef]
- Laxminarayan, R.; Matsoso, P.; Pant, S.; Brower, C.; Rottingen, J.A.; Klugman, K.; Davies, S. Access to effective antimicrobials: A worldwide challenge. Lancet 2016, 387, 168–175. [Google Scholar] [CrossRef]
- Taylor, P.K.; Amy, T.Y.Y.; Robert, E.W.H. Antibiotic resistance in Pseudomonas aeruginosa biofilms: Towards the development of novel anti-biofilm therapies. J. Biotech. 2014, 12, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Manndal, M.D.; Mandal, S. Honey: Its medicinal property and antibacterial activity. Asian Pac. J. Trop. Biomed. 2011, 154–160. [Google Scholar] [CrossRef]
- Lusby, P.E.; Coombes, A.B.; Wilkinson, J.M. Honey: A Potent Agent for Wound Healing? J. Wound Ostomy Cont. Nurs. 2002, 29, 295–300. [Google Scholar] [CrossRef]
- Eteraf-Oskouei, T.; Najafi, M. Traditional and Modern Uses of Natural Honey in Human Diseases: A Review. Iran. J. Basic Med. Sci. 2013, 16, 731–742. [Google Scholar] [PubMed]
- Kateel, R.; Gopalakrishna, B.K.; Baliga, S.; Augustine, A.J.; Ullal, S.; Adhikari, P. Antibacterial action of Tropical honey on various bacteria obtained from diabetic foot ulcer. Complement. Ther. Clin. Pract. 2017, 30, 29–32. [Google Scholar] [CrossRef] [PubMed]
- Yaghoobi, R.; Kazerouni, A.; Kazerouni, O. Evidence for Clinical Use of Honey in Wound Healing as an Antibacterial, Anti-inflammatory Anti-oxidant and Anti-viral Agent: A Review. Jundishapur J. Nat. Pharm. Prod. 2013, 8, 100–104. [Google Scholar] [CrossRef] [PubMed]
- Pasias, I.N.; Kiriakou, I.K.; Kaitatzis, A.; Koutelidakis, A.E.; Proestos, C. Effect of late harvest and floral origin on honey antibacterial properties and quality parameters. Food Chem. 2018, 242, 513–518. [Google Scholar] [CrossRef] [PubMed]
- Molan, P.C. The role of honey in the management of wounds. J. Wound Care 1999, 8, 414–418. [Google Scholar] [CrossRef] [PubMed]
- Khan, I.U.; Dubey, W.; Gupta, V. Medicinal Properties of Honey: A Review. Int. J. Pure Appl. Biosci. 2014, 2, 149–156. [Google Scholar]
- Deng, J.; Liua, R.; Lua, Q.; Haoa, P.; Xua, A.; Zhanga, J.; Tan, J. Biochemical properties, antibacterial and cellular antioxidant activities of buckwheat honey in comparison to manuka honey. Food Chem. 2018, 252, 243–249. [Google Scholar] [CrossRef] [PubMed]
- Clinical and Laboratory Standards Institute (CLSI). Performance Standards for Antimicrobial Susceptibility Testing, M100, 27th ed.; Replaces M100-S26; CLSI: Wayne, PA, USA, 2017; Available online: http://www.facm.ucl.ac.be/intranet/CLSI/CLSI-2017-M100-S27.pdf (accessed on 3 February 2018).
- Piana, M.L.; Persano, O.L.; Bentabol, A.; Bruneau, E.; Bogdanov, S.; Guyot, D.C. Sensory analysis applied to honey: State of the art1. Apidologie 2004, 35, S26–S37. [Google Scholar] [CrossRef]
- Beretta, G.; Granata, P.; Ferrero, M.; Orioli, M.; Facino, R.M. Standardization of antioxidant properties of honey by a combination of spectrophotometric/fluorimetric assays and chemometrics. Anal. Chim. Acta 2005, 533, 185–191. [Google Scholar] [CrossRef]
- Patton, T.; Barett, J.; Brennan, J.; Moran, N. Use of a spectrophotometric bioassay for determination of microbial sensitivity to manuka honey. J. Microbiol. Methods 2005, 64, 84–95. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.; Hong, I.; Woo, S.; Jang, H.; Pak, S.; Han, S. Isolation of abscisic acid from Korean acacia honey with anti-Helicobacter pylori activity. Pharmacogn. Mag. 2017, 13 (Suppl. S2), 170–173. [Google Scholar] [CrossRef]
- Crider, K.S.; Cleves, M.A.; Reefhuis, J.; Berry, R.J.; Hobbs, C.A.; Hu, D.J. Antibacterial medication use during pregnancy and risk of birth defects. Arch. Pediatr. Adolesc. Med. 2009, 163, 978–985. [Google Scholar] [CrossRef] [PubMed]
- Hernandez-Diaz, S.; Werler, M.M.; Walker, A.M.; Mitchell, A.A. Neural tube defects in relation to use of folic acid antagonists during pregnancy. Am. J. Pidemiol. 2001, 153, 961–968. [Google Scholar] [CrossRef]
- Muanda, F.; Sheehy, O.; Bérard, A. Use of antibiotics during pregnancy and risk of spontaneous abortion. CMAG 2017, E625–E633. [Google Scholar] [CrossRef] [PubMed]
- Buchholtz, K.; Carsten, T.L.; Hassager, C.; Bruun, N.E. Severity of gentamicin’s nephrotoxic effect on patients with infective endocarditis: A prospective observational cohort study of 373 Patients. Clin. Infect. Dis. 2009, 48, 65–71. [Google Scholar] [CrossRef] [PubMed]
- Khalil, M.I.; Moniruzzaman, M.; Boukraâ, L.; Benhanifia, M.; Islam, M.A.; Islam, M.N.; Sulaiman, S.A.; Gan, S.H. Physicochemical and antioxidant properties of Algerian honey. Molecules 2012, 17, 11199–11215. [Google Scholar] [CrossRef] [PubMed]
- Mehdi, Y.; Mebrek, S.; Djebara, S.; Aissaoui, Y.; Benhamed, K.; Benali, A.I.; Benali, M.; Belbraout, S. Characterization of Algerian honey from Tiaret region and immunoassay study of its immunomodulatory effect in BALB/c Mice. J. Food Res. 2016, 5, 1–8. [Google Scholar] [CrossRef]
- Asiful, I.; Khalil, I.; Nazmul, I.; Moniruzzaman, M.; Mottali, A.; Sulaiman, S.A.; Gan, S.H. Physicochemical and antioxidant properties of Bangladeshi honeys stored for more than one year. BMC Complement. Altern. Med. 2012, 12, 177–187. [Google Scholar] [CrossRef]
- Saxena, S.; Gautam, S.; Sharma, A. Physical, biochemical and antioxidant properties of some Indian honeys. Food Chem. 2010, 118, 391–397. [Google Scholar] [CrossRef]
- Islem, M.R.; Pervin, T.; Hossain, H.; Saha, B.; Hossain, S.J. Physicochemical and antioxidant properties of honeys from the Sundarbans mangrove forests of Bangladesh. Prev. Nutr. Food Sci. 2017, 22, 335–344. [Google Scholar] [CrossRef] [PubMed]
- Al-Nahari, A.M.; Almasaudi, S.B.; El Sayed, A.M.; Barbour, E.; Al Jaouni, S.K.; Harakeh, S. Antimicrobial activities of Saudi honey against Pseudomonas aeruginosa. Saudi J. Biol. Sci. 2015, 22, 521–525. [Google Scholar] [CrossRef] [PubMed]
- Al-Namma, R.T. Evaluation of in vitro inhibitory effect of honey on some microbial isolate. J. Bacteriol. Res. 2009, 1, 64–67. [Google Scholar]
- Nazzaro, F.; Fratianni, F.; De Laura, M.; Coppola, R.; De Feo, V. Effect of essential oils on pathogenic bacteria. Pharmaceuticals 2013, 6, 1451–1474. [Google Scholar] [CrossRef] [PubMed]
- Nikaido, H. Prevention of drug access to bacterial targets: Permeability barriers and active efflux. Science 1994, 264, 382–388. [Google Scholar] [CrossRef] [PubMed]

| Strain | Susceptibility to Prescribed Antibiotics | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| β-Lactams | Cephalosporin | Fluoroquinolone | Aminglycosides | Others | |||||||||
| OX | AM | AMC | CN | CF | OF | NA | CIP | GM | TM | NIT | FOS | SXT | |
| E. coli | ND | R | R | R | R | S | S | S | S | R | R | S | R |
| E. aerogenes | ND | R | R | R | R | S | S | S | R | R | R | R | R |
| K. oxytoca | ND | R | R | R | R | S | S | S | S | R | R | R | R |
| K. pneumoniae | ND | R | R | R | R | R | R | R | R | R | R | R | R |
| P. mirabilis | ND | R | R | R | R | R | R | R | R | R | R | R | R |
| P. vulgaris | ND | R | R | R | R | R | R | R | R | R | R | R | R |
| C. koseri | ND | R | R | R | R | S | S | S | S | R | R | R | R |
| P. aeruginosa | ND | R | R | R | R | R | R | R | R | R | R | R | R |
| S. aureus | R | ND | ND | R | R | R | R | R | S | R | R | R | R |
| S. saprophyticus | S | ND | ND | R | R | S | S | S | S | R | R | S | R |
| E. faecalis | R | ND | ND | R | R | R | R | R | R | R | R | R | R |
| Susceptibility Percentage (%) | 33.3 | 0 | 0 | 0 | 0 | 45.4 | 45.4 | 45.4 | 45.4 | 0 | 0 | 18.2 | 0 |
| Honey Sample | Taste | pH | Color | Color Intensity (mAU) |
|---|---|---|---|---|
| Sample 1 | Sweet | 3.44 | Light brown | 742 |
| Sample 2 | Bitter | 4.54 | Dark brown | 1965 |
| Sample 3 | Sweet | 3.86 | Brown | 829 |
| Sample 4 | Sweet | 3.19 | Dark brown | 1654 |
| Sample 5 | Sweet | 3.54 | Brown | 982 |
| Sample 6 | Sweet | 4.02 | Light brown | 352 |
| Strain | Diameter of Inhibition (mm±SD) | |||||
|---|---|---|---|---|---|---|
| Sample 1 | Sample 2 | Sample 3 | Sample 4 | Sample 5 | Sample 6 | |
| E. coli | 30.33 ± 1.56 | 32 ± 0.67 | 36.67 ± 1.11 | 33.67 ± 0.44 | 30.00 ± 0.67 | 30.00 ± 0.67 |
| E. aerogenes | 30.33 ± 1.11 | 31.33 ± 0.89 | 37.00 ± 0.67 | 33.67 ± 0.89 | 31.33 ± 1.11 | 31.00 ± 0.67 |
| K. oxytoca | 28.33 ± 1.11 | 31.33 ± 0.44 | 36.00 ± 1.33 | 33.33 ± 0.44 | 29.00 ± 1.33 | 30.67 ± 1.56 |
| K. pneumoniae | 29.67 ± 1.11 | 32.33 ± 0.44 | 37.33 ± 0.89 | 35.00 ± 0.67 | 29.33 ± 0.44 | 29.33 ± 0.44 |
| P. mirabilis | 30.00 ± 0.67 | 31.33 ± 0.89 | 36.33 ± 1.11 | 33.33 ± 1.11 | 30.33 ± 0.44 | 28.67 ± 0.89 |
| P. vulgaris | 30.33 ± 1.11 | 32.33 ± 0.44 | 35.67 ± 1.56 | 31.00 ± 0.67 | 29.67 ± 0.44 | 29.67 ± 0.89 |
| C. koseri | 29.00 ± 1.33 | 30.33 ± 0.44 | 34.67 ± 0.89 | 31.67 ± 1.56 | 29.00 ± 0.67 | 29.00 ± 0.67 |
| P. aeruginosa | 19.67 ± 1.56 | 24.33 ± 0.89 | 27.33 ± 0.89 | 26.00 ± 0.67 | 20.33 ± 0.89 | 20.33 ± 0.44 |
| S. aureus | 47.667 ± 1.11 | 48.67 ± 0.89 | 50.67 ± 1.11 | 49.67 ± 0.44 | 47.67 ± 0.44 | 47.33 ± 1.56 |
| S. saprophyticus | 48.33 ± 0.89 | 50.00 ± 0.67 | 53.33 ± 0.44 | 50.33 ± 0.89 | 47.67 ± 0.44 | 47.67 ± 0.89 |
| E. faecalis | 43.67 ± 0.89 | 44.00 ± 0.67 | 46.67 ± 0.44 | 44.00 ± 0.67 | 43.33 ± 0.89 | 43.67 ± 0.44 |
| Strain | Sample 1 | Sample 2 | Sample 3 | Sample 4 | Sample 5 | Sample 6 | ||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| MIC | MBC | MIC | MBC | MIC | MBC | MIC | MBC | MIC | MBC | MIC | MBC | |
| E. coli | 10 | 10 | 10 | 10 | 5 | 10 | 10 | 10 | 10 | 10 | 10 | 10 |
| E. aerogenes | 10 | 10 | 10 | 10 | 5 | 10 | 10 | 10 | 10 | 10 | 10 | 10 |
| K. oxytoca | 20 | 20 | 10 | 20 | 5 | 20 | 10 | 20 | 20 | 20 | 10 | 20 |
| K. pneumoniae | 20 | 20 | 10 | 20 | 5 | 20 | 5 | 20 | 20 | 20 | 20 | 20 |
| P. mirabilis | 10 | 10 | 10 | 10 | 5 | 10 | 10 | 10 | 10 | 10 | 20 | 10 |
| P. vulgaris | 10 | 10 | 10 | 10 | 5 | 10 | 10 | 10 | 20 | 10 | 20 | 10 |
| C. koseri | 20 | 40 | 10 | 40 | 5 | 40 | 10 | 40 | 20 | 40 | 20 | 40 |
| P. aeruginosa | 40 | 80 | 20 | 40 | 20 | 40 | 20 | 20 | 40 | 80 | 40 | 80 |
| S. aureus | 2.5 | 2.5 | 2.5 | 2.5 | 2.5 | 2.5 | 2.5 | 2.5 | 2.5 | 2.5 | 2.5 | 2.5 |
| S. saprophyticus | 2.5 | 2.5 | 2.5 | 2.5 | 2.5 | 2.5 | 2.5 | 2.5 | 2.5 | 2.5 | 2.5 | 2.5 |
| E. faecalis | 5 | 5 | 5 | 5 | 2.5 | 5 | 5 | 5 | 5 | 5 | 5 | 5 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bouacha, M.; Ayed, H.; Grara, N. Honey Bee as Alternative Medicine to Treat Eleven Multidrug-Resistant Bacteria Causing Urinary Tract Infection during Pregnancy. Sci. Pharm. 2018, 86, 14. https://doi.org/10.3390/scipharm86020014
Bouacha M, Ayed H, Grara N. Honey Bee as Alternative Medicine to Treat Eleven Multidrug-Resistant Bacteria Causing Urinary Tract Infection during Pregnancy. Scientia Pharmaceutica. 2018; 86(2):14. https://doi.org/10.3390/scipharm86020014
Chicago/Turabian StyleBouacha, Mabrouka, Hayette Ayed, and Nedjoud Grara. 2018. "Honey Bee as Alternative Medicine to Treat Eleven Multidrug-Resistant Bacteria Causing Urinary Tract Infection during Pregnancy" Scientia Pharmaceutica 86, no. 2: 14. https://doi.org/10.3390/scipharm86020014
APA StyleBouacha, M., Ayed, H., & Grara, N. (2018). Honey Bee as Alternative Medicine to Treat Eleven Multidrug-Resistant Bacteria Causing Urinary Tract Infection during Pregnancy. Scientia Pharmaceutica, 86(2), 14. https://doi.org/10.3390/scipharm86020014





