A PCR-Based Molecular Detection of Strongyloides stercoralis in Human Stool Samples from Tabriz City, Iran
Abstract
:1. Introduction
2. Materials and Methods
2.1. Subjects and Microscopic Diagnosis
2.2. DNA Extraction
2.3. Polymerase Chain Reaction
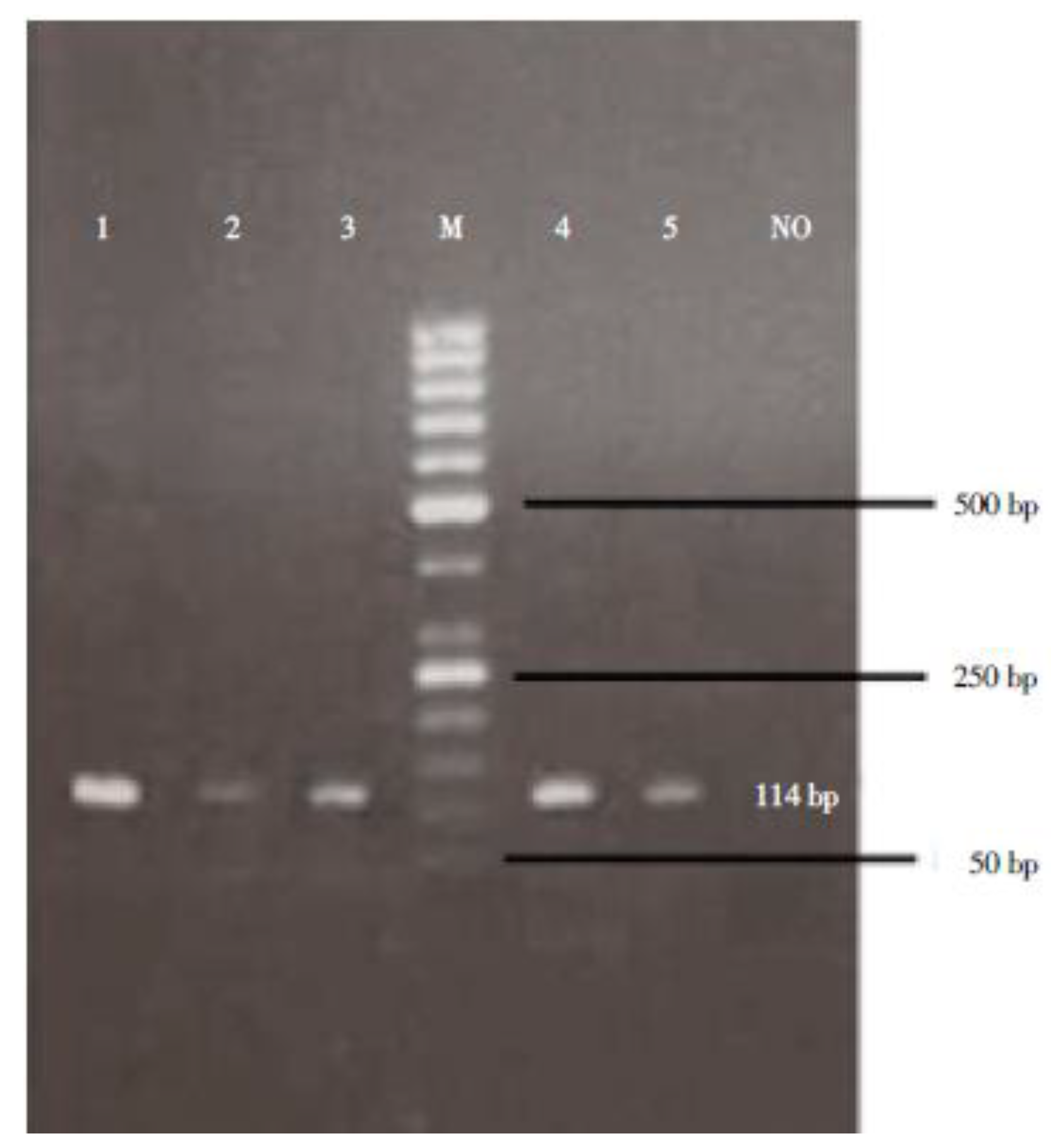
2.4. Electrophoresis
2.5. Statistical Analyses
3. Results
3.1. Microscopic Diagnosis
3.2. DNA Extraction
3.3. PCR and Electrophoresis Results
4. Discussion
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Grove, D.I. Clinical manifestations. In Strongyloidiasis A Major Roundworm infection of Man; Grove, D.I., Ed.; Taylor and Francis: London, UK, 1989; pp. 155–174. [Google Scholar]
- Viney, M.E.; Lok, J.B. Strongyloides Spp.; WormBook, Ed.; The C. elegans Research Community, WormBook; 2015; pp. 1–15. [Google Scholar] [CrossRef]
- Tabei, S.Z.; Asadian, F.; Fakhar, M.; Safaei, A. Gastrointestinal hyperinfection due to Strongyloides stercoralis in a patient with Behcet’ssyndrome. Comp. Clin. Pathol. 2009, 18, 89–91. [Google Scholar] [CrossRef]
- Steinmann, P.; Zhou, X.N.; Du, Z.W.; Jiang, J.Y.; Wang, L.B.; Wang, X.Z.; Li, L.H.; Marti, H.; Utzinger, J. Occurrence of Strongyloides stercoralis in Yunnan Province, China, and comparison of diagnostic methods. PLoS Negl. Trop. Dis. 2007, 1, e75. [Google Scholar] [CrossRef] [PubMed]
- Chaves, N.J.; Gibney, K.B.; Leder, K.; O’Brien, D.P.; Marshall, C.; Biggs, B. Screening practices for infectious diseases among Burmeserefugees in Australia. Emerg. Infect. Dis. 2009, 15, 1769–1772. [Google Scholar] [CrossRef] [PubMed]
- Glinz, D.; N’Guessan, N.A.; Utzinger, J.; N’Goran, E.K. Highprevalence of Strongyloides stercoralis among school children inrural Côte d’Ivoire. J. Parasitol. 2010, 96, 431–433. [Google Scholar] [CrossRef] [PubMed]
- Knopp, S.; Mohammed, K.A.; Stothard, J.R.; Khamis, I.S.; Rollinson, D.; Marti, H.; Utzinger, J. Patterns and risk factors of helminthiasis andanemia in a rural and a peri-urban community in Zanzibar, in the context of helminth control programs. PLoS Negl. Trop. Dis. 2010, 4, e681. [Google Scholar] [CrossRef] [PubMed]
- Repetto, S.A.; Durán, P.A.; Lasala, M.B.; González-Cappa, S.M. Highrate of strongyloidosis infection, out of endemic area, in patientswith eosinophilia and without risk of exogenous reinfections. Am. J. Trop. Med. Hyg. 2010, 82, 1088–1093. [Google Scholar] [CrossRef] [PubMed]
- Keiser, P.B.; Nutman, T.B. Strongyloides stercoralis in theimmunocompromised population. Clin. Microb. Rev. 2004, 17, 208–217. [Google Scholar] [CrossRef]
- Krolewiecki, A.J.; Ramanathan, R.; Fink, V.; McAuliffe, I.; Cajal, S.P.; Won, K.; Juarez, M.; Di Paolo, A.; Tapia, L.; Acosta, N.; et al. Improved diagnosis of Strongyloides stercoralis using recombinant antigen-based serologies in a community-wide studyin northern Argentina. Clin. Vaccine Immunol. 2010, 17, 1624–1630. [Google Scholar] [CrossRef] [PubMed]
- Marcos, L.A.; Terashima, A.; Dupont, H.L.; Gotuzzo, E. Strongyloides Hyperinfection syndrome: An emerging global infectious disease. Trans. R. Soc. Trop. Med. Hyg. 2008, 102, 314–318. [Google Scholar] [CrossRef] [PubMed]
- Meamar, A.R.; Rezaian, M.; Mohraz, M.; Hadighi, R.; Kia, E.B. Strongyloides stercoralis hyper-infection syndrome in HIV+/AIDSpatients in Iran. Parasitol. Res. 2007, 101, 663–665. [Google Scholar] [CrossRef] [PubMed]
- Kia, E.B.; Rahimi, H.R.; Mirhendi, H.; Nilforoushan, M.R.; Talebi, A.; Zahabiun, F.; Kazemzadeh, H.; Meamar, A.R. A case of fatal strongyloidiasis in a patient withchronic lymphocytic leukemia and molecular characterization ofthe isolate. Korean J. Parasitol. 2008, 46, 261–263. [Google Scholar] [CrossRef] [PubMed]
- Pays, J.F. Combined infection with HTLV-1 and Strongyloides stercoralis. Bull. Soc. Pathol. Exot. 2011, 104, 188–199. [Google Scholar] [CrossRef] [PubMed]
- Azira, N.M.; Zeehaida, M. Strongyloides stercoralis hyperinfectionin a diabetic patient: Case report. Trop. Biomed. 2010, 27, 115–119. [Google Scholar] [PubMed]
- Ghasemikhah, R.; Sharbatkhori, M.; Mobedi, I.; Kia, E.; Harandi, M.F.; Mirhendi, H. Sequence Analysis of the Second Internal Transcribed Spacer (ITS2) Region of rDNA for Species Identification of Trichostrongylus Nematodes Isolated From Domestic Livestock in Iran. Iran J. Parasitol. 2012, 7, 40–46. [Google Scholar] [PubMed]
- Liu, Z.; Tu, K.; Liu, Q. Effects of microRNA-30a on migration, invasion and prognosis of hepatocellular carcinoma. FEBS Lett. 2014, 588, 3089–3097. [Google Scholar] [CrossRef] [PubMed]
- Ouzounova, M.; Vuong, T.; Ancey, P.-B.; Ferrand, M.; Durand, G.; Kelm, F.L.-C.; Croce, C.; Matar, C.; Herceg, Z.; Hernandez-Vargas, H. MicroRNA miR-30 family regulates non-attachment growth of breast cancer cells. BMC Genom. 2013, 14, 1. [Google Scholar] [CrossRef] [PubMed]
- Budhu, A.; Jia, H.L.; Forgues, M.; Liu, C.G.; Goldstein, D.; Lam, A.; Zanetti, K.A.; Ye, Q.H.; Qin, L.X.; Croce, C.M.; et al. Identification of metastasis-related microRNAs in hepatocellular carcinoma. Hepatology 2008, 47, 897–907. [Google Scholar] [CrossRef] [PubMed]
- Mowlavi, G.R.; Mir Ahmadi, H.; Rezaeian, M.; EbrahimiDaryani, N.; Rokni, M.B.; Golestan, B.; Shafiei, R.; Fereshtehnejad, S.M.; Keramati, M.R. Prevalence of intestinal parasites intribal parts of Khuzestan Province 2005–2007. Govaresh 2008, 12, 219–229. [Google Scholar]
- Rahimi-Esboei, B.; Gholami, S.H.; Ghorbani Pasha Kolaei, A.; PourHaji, B.M.; Hasannia, H.; Shaban, R. The prevalenceof intestinal parasitic infections among the people living in thecentral areas of Mazandaran Province (2009–2010). MLJ 2013, 7, 43–48. [Google Scholar]
- Hanan, Z.R.; Rasha, H.S.; Nahla, M.G. Detection of Strongyloides stercoralis in fecal samples using conventional parasitological techniques and real-time PCR: A comparative study. PUJ 2012, 5, 27–34. [Google Scholar]
- Getaneh, A.; Medhin, G.; Shimelis, T. Cryptosporidium and Strongyloides stercoralis infections among people with andwithout HIV infection and efficiency of diagnostic methods for Strongyloides in Yirgalem Hospital, southern Ethiopia. BMC Res. Notes 2010, 3, 90. [Google Scholar] [CrossRef] [PubMed]
- Verweij, J.J.; Canales, M.; Polman, K.; Ziem, J.; Brienen, E.A.; Polderman, A.M.; van Lieshout, L. Molecular diagnosis of Strongyloides stercoralis in faecal samples using real-time PCR. R. Soc. Trop. Med. Hyg. 2009, 103, 342–346. [Google Scholar] [CrossRef] [PubMed]
- Moghaddassani, H.; Mirhendi, H.; Hosseini, M.; Rokni, M.B.; Mowlavi, G.H.; Kia, E.B. Molecular Diagnosis of Strongyloides stercoralis Infection by PCR Detection of Specific DNA in Human Stool Samples. Iran. J. Parasitol. 2011, 6, 23–30. [Google Scholar] [PubMed]
| S. stercoralis | Diagnosis Method | |
|---|---|---|
| Negative (%) | Positive (%) | |
| 1798 (99.9) | 2 (0.1) | Direct exam |
| 1795 (99.7) | 5 (0.3) | Formalin ether |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ghasemikhah, R.; Tabatabaiefar, M.A.; Shariatzadeh, S.A.; Shahbazi, A.; Hazratian, T. A PCR-Based Molecular Detection of Strongyloides stercoralis in Human Stool Samples from Tabriz City, Iran. Sci. Pharm. 2017, 85, 17. https://doi.org/10.3390/scipharm85020017
Ghasemikhah R, Tabatabaiefar MA, Shariatzadeh SA, Shahbazi A, Hazratian T. A PCR-Based Molecular Detection of Strongyloides stercoralis in Human Stool Samples from Tabriz City, Iran. Scientia Pharmaceutica. 2017; 85(2):17. https://doi.org/10.3390/scipharm85020017
Chicago/Turabian StyleGhasemikhah, Reza, Mohammad Amin Tabatabaiefar, Seyed Ali Shariatzadeh, Abbas Shahbazi, and Teymour Hazratian. 2017. "A PCR-Based Molecular Detection of Strongyloides stercoralis in Human Stool Samples from Tabriz City, Iran" Scientia Pharmaceutica 85, no. 2: 17. https://doi.org/10.3390/scipharm85020017
APA StyleGhasemikhah, R., Tabatabaiefar, M. A., Shariatzadeh, S. A., Shahbazi, A., & Hazratian, T. (2017). A PCR-Based Molecular Detection of Strongyloides stercoralis in Human Stool Samples from Tabriz City, Iran. Scientia Pharmaceutica, 85(2), 17. https://doi.org/10.3390/scipharm85020017





