Timely Interventions for Children with ADHD through Web-Based Monitoring Algorithms
Abstract
1. Introduction
2. Materials and Methods
2.1. Overall Design
2.2. Participants and Setting
2.3. TriVox Health Platform
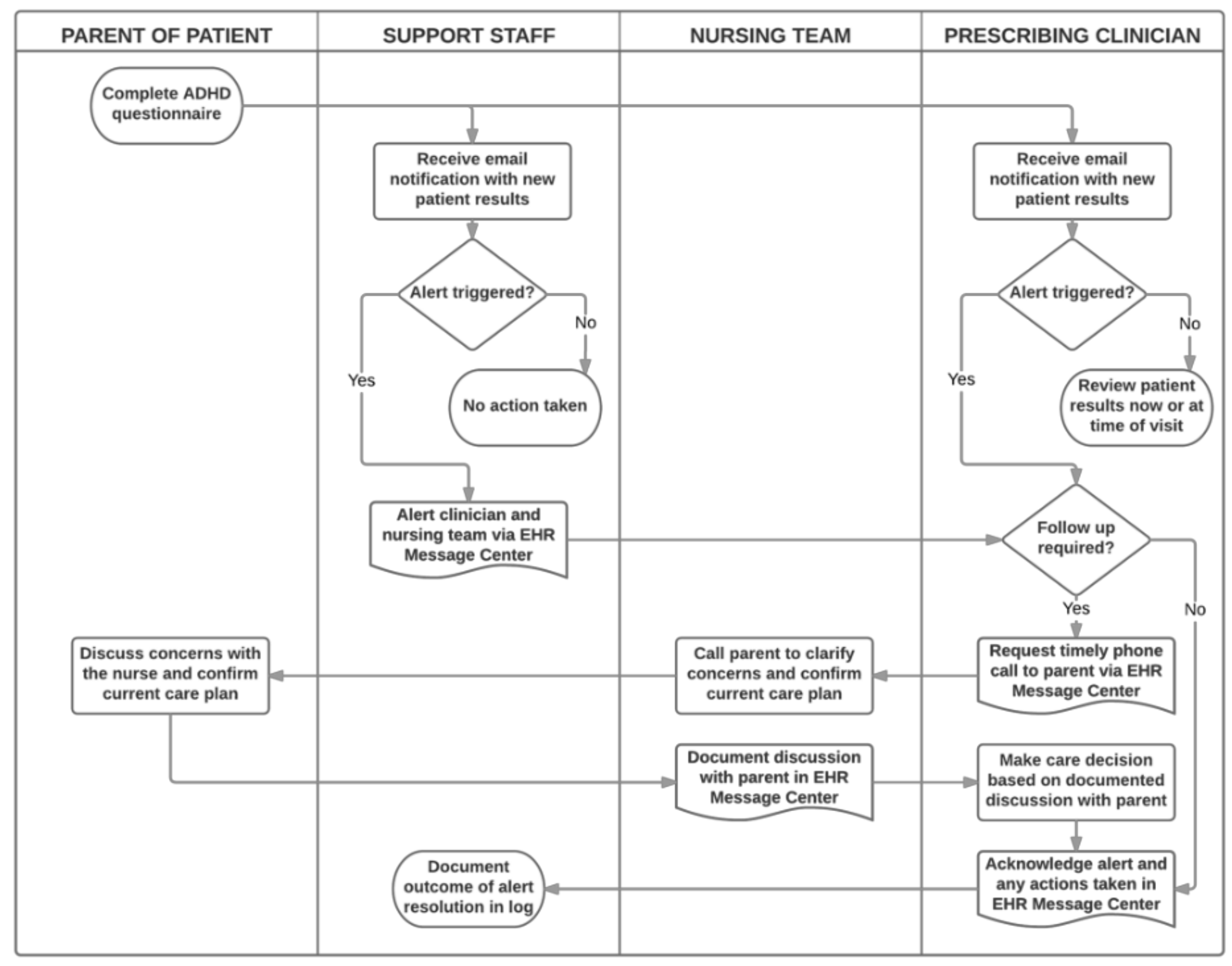
2.4. Interventions: Trigger Algorithm and Alert Resolution Process
2.5. Outcome Measures
2.6. Process Measure
2.7. Balancing Measure
2.8. Analysis
3. Results
3.1. Patient Characteristics
3.2. Primary Outcome
3.3. Secondary Outcomes
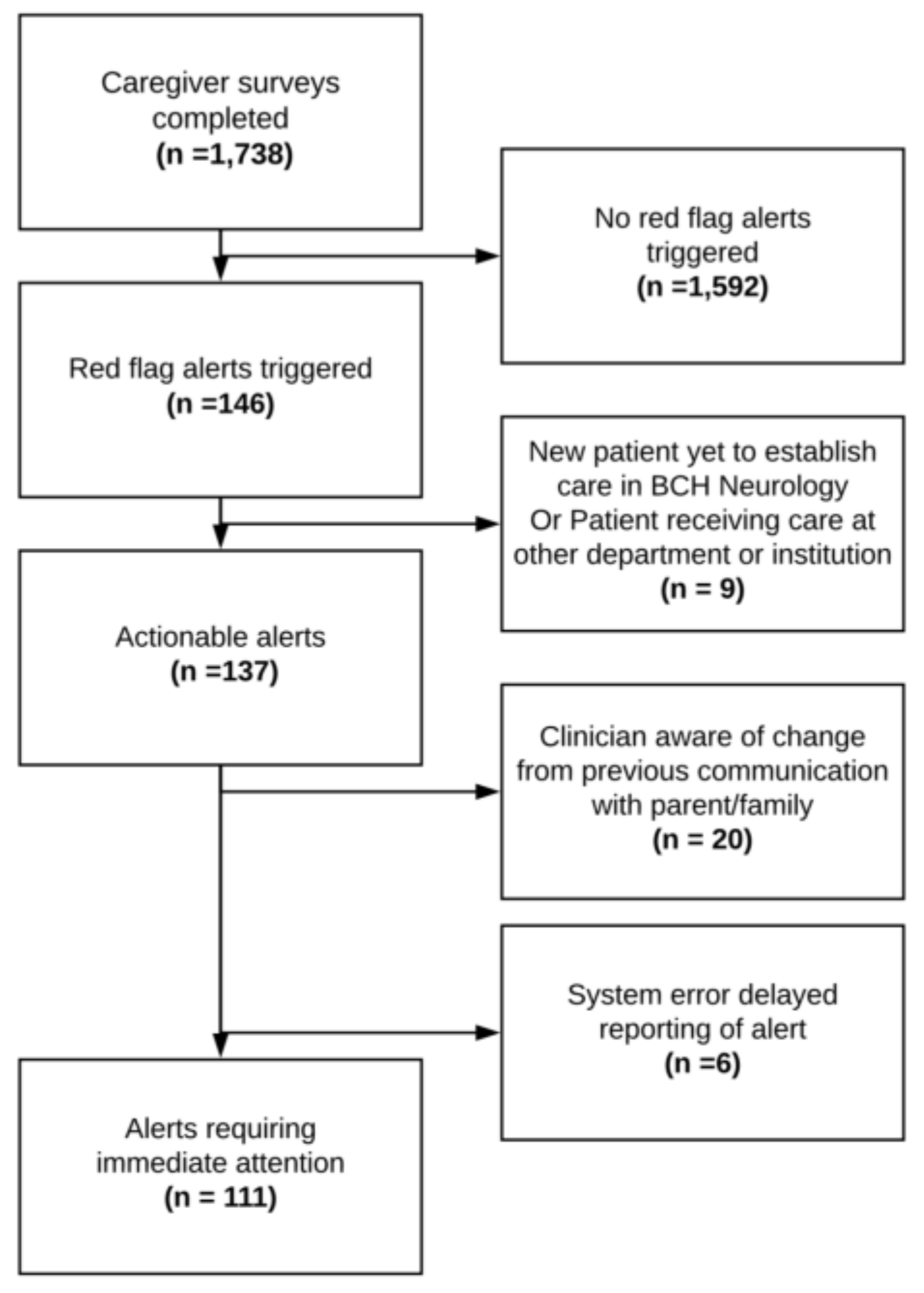
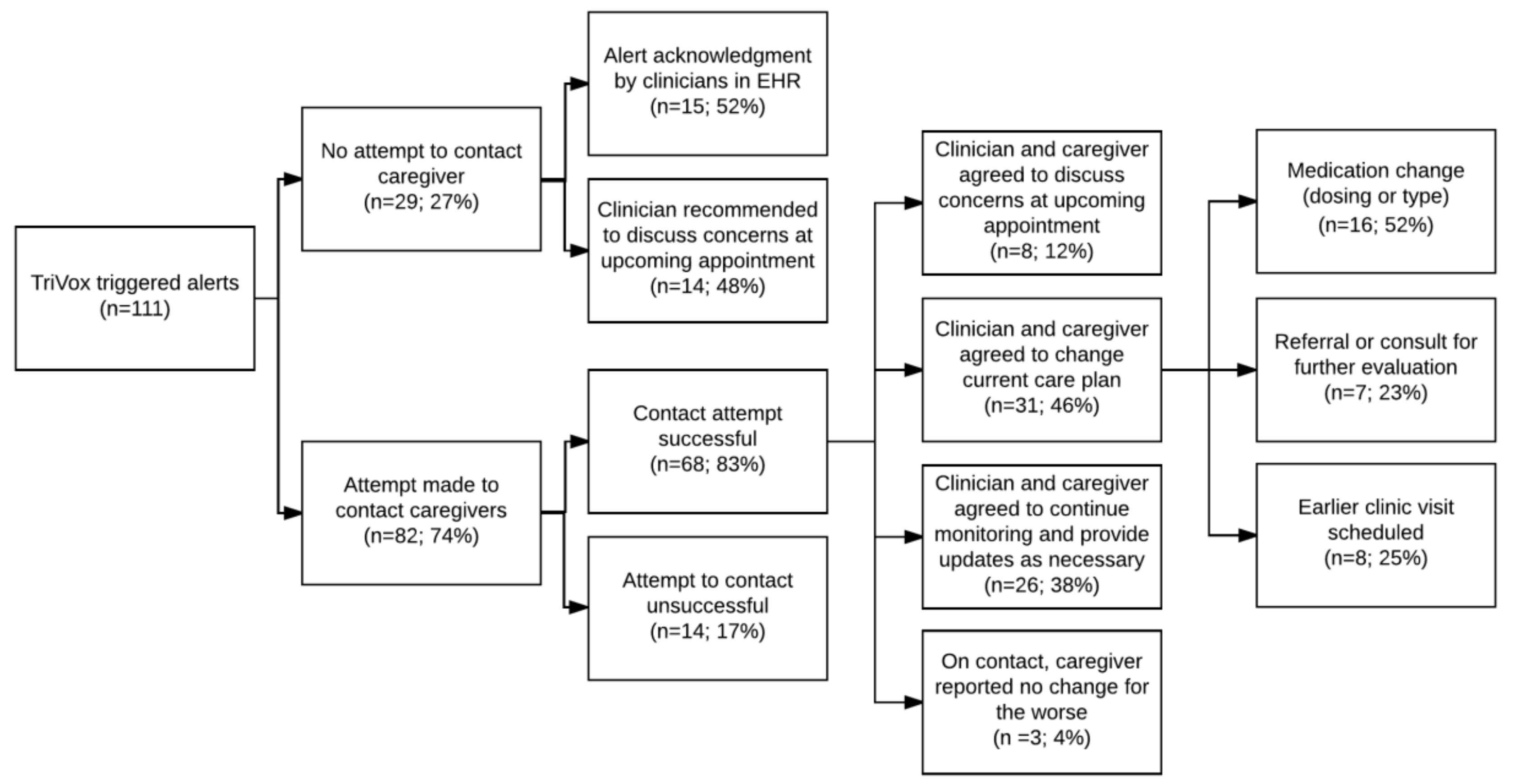
3.4. Process Measure
3.5. Balancing Measure
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Institute of medicine committee on quality of health care in A. In Crossing the Quality Chasm: A New Health System for the 21st Century; National Academies Press: Washington, DC, USA, 2001.
- Gentles, S.J.; Lokker, C.; McKibbon, K.A. Health information technology to facilitate communication involving health care providers, caregivers, and pediatric patients: A scoping review. J. Med. Int. Res. 2010, 12, e22. [Google Scholar] [CrossRef] [PubMed]
- Davis Giardina, T.; King, B.J.; Ignaczak, A.P.; Paull, D.E.; Hoeksema, L.; Mills, P.D.; Neily, J.; Hemphill, R.R.; Singh, H. Root cause analysis reports help identify common factors in delayed diagnosis and treatment of outpatients. Health Affair. 2013, 32, 1368–1375. [Google Scholar] [CrossRef] [PubMed]
- Sarkar, U.; Handley, M.A.; Gupta, R.; Tang, A.; Murphy, E.; Seligman, H.K.; Shojania, K.G.; Schillinger, D. What What happens between visits? Adverse and potential adverse events among a low-income, urban, ambulatory population with diabetes. Quality Safety Health Care. 2010, 19, 223–228. [Google Scholar] [CrossRef] [PubMed]
- Subcommittee on Attention-Deficit/Hyperactivity Disorder; Steering Committee on Quality Improvement and Management. ADHD: Clinical practice guideline for the diagnosis, evaluation, and treatment of attention-defi cit/hyperactivity disorder in children and adolescents. Pediatrics 2011, 128, 1007–1022. [Google Scholar] [CrossRef] [PubMed]
- Baum, R.A.; Epstein, J.N.; Kelleher, K. Healthcare reform, quality, and technology: ADHD as a case study. Curr. Psychiatry Rep. 2013, 15, 369. [Google Scholar] [CrossRef] [PubMed]
- Epstein, J.N.; Kelleher, K.J.; Baum, R.; Brinkman, W.B.; Peugh, J.; Gardner, W.; Lichtenstein, P.; Langberg, J.M. Variability in ADHD care in community-based pediatrics. Pediatrics 2014, 134, 1136–1143. [Google Scholar] [CrossRef] [PubMed]
- Epstein, J.N.; Kelleher, K.J.; Baum, R.; Brinkman, W.B.; Peugh, J.; Gardner, W.; Lichtenstein, P.; Langberg, J.M. Impact of a web-portal intervention on community ADHD care and outcomes. Pediatrics 2016, 138, e20154240. [Google Scholar] [CrossRef]
- Simons, L.; Valentine, A.Z.; Falconer, C.J.; Groom, M.; Daley, D.; Craven, M.P.; Young, Z.; Hall, C.; Hollis, C. Developing mHealth remote monitoring technology for attention deficit hyperactivity disorder: A qualitative study eliciting user priorities and needs. JMIR mHealth and uHealth. 2016, 4, e31. [Google Scholar] [CrossRef]
- Fleegler, E.; Weas, S.; Zhang, X.; Rhodes, E.; Kahn, J.; Harris, S.; Huntington, N.; Borus, J.; Loddenkemper, T.; Chan, E. Implementation of an internet-based patient monitoring system improves families’ perception of quality of communication and experience of care; Pediatric Academic Societies: Baltimore, MD, USA, 2016. [Google Scholar]
- Goldzweig, C.L.; Towfigh, A.; Maglione, M.; Shekelle, P.G. Costs and benefits of health information technology: New trends from the literature. Health Affair. 2009, 28, w282–w293. [Google Scholar] [CrossRef]
- The hazards of alarm overload. Keeping excessive physiologic monitoring alarms from impeding care. Health Devices 2007, 36, 73–83. [Google Scholar]
- Baysari, M.T.; Westbrook, J.I.; Richardson, K.; Day, R.O. Optimising computerised alerts within electronic medication management systems: A synthesis of four years of research. Studies Health Technol. Infor. 2014, 204, 1–6. [Google Scholar]
- Lee, E.K.; Wu, T.L.; Senior, T.; Jose, J. Medical alert management: A real-time adaptive decision support tool to reduce alert fatigue. AMIA Annu. Symp. Proc. 2014, 2014, 845–854. [Google Scholar] [PubMed]
- McDaniel, R.B.; Burlison, J.D.; Baker, D.K.; Hasan, M.; Robertson, J.; Hartford, C.; Howard, S.C.; Sablauer, A.; Hoffman, J.M. Alert dwell time: Introduction of a measure to evaluate interruptive clinical decision support alerts. JAMIA 2016, 23, e138–e141. [Google Scholar] [CrossRef]
- De Wet, C.; Bowie, P. The preliminary development and testing of a global trigger tool to detect error and patient harm in primary-care records. Postgrad. Med. J. 2009, 85, 176–180. [Google Scholar] [CrossRef] [PubMed]
- Singh, H.; Arora, H.S.; Vij, M.S.; Rao, R.; Khan, M.M.; Petersen, L.A. Communication outcomes of critical imaging results in a computerized notification system. JAMIA 2007, 14, 0459–0466. [Google Scholar] [CrossRef] [PubMed]
- Committee on Quality of Health Care in America IoM. To Err Is Human: Building a Safer Health System; National Academy of Sciences: Washington, DC, USA, 2000. [Google Scholar]
- Guidi, J.L.; Clark, K.; Upton, M.T.; Faust, H.; Umscheid, C.A.; Lane-Fall, M.B.; Mikkelsen, M.E.; Schweickert, W.D.; Vanzandbergen, C.A.; Betesh, J.; Tait, G. Clinician perception of the effectiveness of an automated early warning and response system for sepsis in an academic medical center. Ann. Am. Thorac. Soc. 2015, 12, 1514–1519. [Google Scholar] [CrossRef] [PubMed]
- Murphy, D.R.; Wu, L.; Thomas, E.J.; Forjuoh, S.N.; Meyer, A.N.D.; Singh, H. Electronic trigger-based intervention to reduce delays in diagnostic evaluation for cancer: A cluster randomized controlled trial. J. Clin. Oncol. 2015, 33, 3560–3567. [Google Scholar] [CrossRef]
- Shiloh, A.L.; Lominadze, G.; Gong, M.N.; Savel, R.H. Early warning/track-and-trigger systems to detect deterioration and improve outcomes in hospitalized patients. Semin. Respir. Crit. Care Med. 2016, 37, 88–95. [Google Scholar]
- Terrell, K.M.; Perkins, A.J.; Dexter, P.R.; Hui, S.L.; Callahan, C.M.; Miller, D.K. Computerized decision support to reduce potentially inappropriate prescribing to older emergency department patients: A randomized, controlled trial. J. Am. Geriatr. Soc. 2009, 57, 1388–1394. [Google Scholar] [CrossRef]
- Bates, D.W.; Evans, R.S.; Murff, H.; Stetson, P.D.; Pizziferri, L.G. Detecting adverse events using information technology. JAMIA 2003, 10, 115–128. [Google Scholar] [CrossRef]
- Murphy, D.R.; Thomas, E.J.; Meyer, A.N.; Singh, H. Development and validation of electronic health record-based triggers to detect delays in follow-up of abnormal lung imaging findings. Radiology. 2015, 277, 81–87. [Google Scholar] [CrossRef] [PubMed]
- Bard, D.E.; Wolraich, M.L.; Neas, B.; Doffing, M.; Beck, L. The psychometric properties of the Vanderbilt attention-deficit hyperactivity disorder diagnostic parent rating scale in a community population. JDBP 2013, 34, 72–82. [Google Scholar] [CrossRef] [PubMed]
- Wolraich, M.L.; Lambert, W.; Doffing, M.A.; Bickman, L.; Simmons, T.; Worley, K. Psychometric properties of the Vanderbilt ADHD diagnostic parent rating scale in a referred population. J. Pediatr. Psychol. 2003, 28, 559–567. [Google Scholar] [CrossRef]
- Green, B.; Shirk, S.; Hanze, D.; Wanstrath, J. The Children’s Global Assessment Scale in clinical practice: An empirical evaluation. J. Am. Acad. Child Adolesc. Psychiatry 1994, 33, 1158–1164. [Google Scholar] [CrossRef] [PubMed]
- Park, J.H.; Lee, S.I.; Schachar, R.J. Reliability and validity of the child and adolescent functioning impairment scale in children with attention-deficit/hyperactivity disorder. Psychiatry Investig. 2011, 8, 113–122. [Google Scholar] [CrossRef] [PubMed]
- Shaffer, D.; Gould, M.S.; Brasic, J.; Ambrosini, P.; Fisher, P.; Bird, H.; Aluwahlia, S. A children’s global assessment scale (CGAS). Arch. Gen. Psychiatry 1983, 40, 1228–1231. [Google Scholar] [CrossRef] [PubMed]
- Dekarske, B.M.; Zimmerman, C.R.; Chang, R.; Grant, P.J.; Chaffee, B.W. Increased appropriateness of customized alert acknowledgement reasons for overridden medication alerts in a computerized provider order entry system. Int. J. Med. Infor. 2015, 84, 1085–1093. [Google Scholar] [CrossRef]
- Parke, C.; Santiago, E.; Zussy, B.; Klipa, D. Reduction of clinical support warnings through recategorization of severity levels. AJHP 2015, 72, 144–148. [Google Scholar] [CrossRef]
- Simpao, A.F.; Ahumada, L.M.; Desai, B.R.; Bonafide, C.P.; Gálvez, J.A.; Rehman, M.A.; Jawad, A.F.; Palma, K.L.; Shelov, E.D. Optimization of drug-drug interaction alert rules in a pediatric hospital’s electronic health record system using a visual analytics dashboard. JAMIA 2015, 22, 361–369. [Google Scholar] [CrossRef]
- Alert Fatigue. PSNet: Agency for Healthcare Research and Quality; Updated Jan 2019; Wachter, R.M., Gupta, K., Ranji, S., Sarkar, U., Sharpe, B., Shojania, K., Lyndon, A., Franck, L.S., Pon, T., Murray, S, Eds.; U.S. Department of Health and Human Services: Rockville, MD, USA, 2016. [Google Scholar]
- Shojania, K.G.; Jennings, A.; Mayhew, A.; Ramsay, C.; Eccles, M.; Grimshaw, J. Effect of point-of-care computer reminders on physician behaviour: A systematic review. CMAJ 2010, 182, E216–E225. [Google Scholar] [CrossRef]
- Liang, E.F.; Lim, S.Z.; Tam, W.W.; Ho, C.S.; Zhang, M.W.; McIntyre, R.S.; Ho, R. The effect of methylphenidate and atomoxetine on heart rate and systolic blood pressure in young people and adults with attention-deficit hyperactivity disorder (ADHD): Systematic review, meta-analysis, and meta-regression. Int. J. Environ. Res. Public Health 2018, 15, E1789. [Google Scholar] [CrossRef] [PubMed]
- Blumberg, S.J.; Luke, J.V. Wireless substitution: Early release of estimates from the national health interview survey, July–December 2016. National Center for Health Statistics 2017.
- Barbaresi, W.J. Improving care for children with ADHD: The information is just a rating scale away. Pediatrics. 2016, 137. [Google Scholar] [CrossRef] [PubMed]
- Lai, J.S.; Yount, S.; Beaumont, J.L.; Cella, D.; Toia, J.; Goldman, S. A patient-centered symptom monitoring and reporting system for children and young adults with cancer (SyMon-SAYS). Pediatr. Blood Cancer. 2015, 62, 1813–1818. [Google Scholar] [CrossRef] [PubMed]
- Gee, P.M.; Paterniti, D.A.; Ward, D.; Soederberg Miller, L.M. e-Patients perceptions of using personal health records for self-management support of chronic illness. CIN 2015, 33, 229–237. [Google Scholar] [CrossRef] [PubMed]
- Newman, E.D.; Lerch, V.; Billet, J.; Berger, A.; Kirchner, H.L. Improving the quality of care of patients with rheumatic disease using patient-centric electronic redesign software. Arthrit. Care Res. 2015, 67, 546–553. [Google Scholar] [CrossRef] [PubMed]
- Coye, M.J.; Haselkorn, A.; DeMello, S. Remote patient management: Technology-enabled innovation and evolving business models for chronic disease care. Health Affair. 2009, 28, 126–135. [Google Scholar] [CrossRef]
- Jonassaint, C.R.; Shah, N.; Jonassaint, J.; De Castro, L. Usability and feasibility of an mHealth intervention for monitoring and managing pain symptoms in sickle cell disease: The sickle cell disease mobile application to record symptoms via technology (SMART). Hemoglobin 2015, 39, 162–168. [Google Scholar] [CrossRef]
- Hale, T.M.; Jethwani, K.; Kandola, M.S.; Saldana, F.; Kvedar, J.C. A remote medication monitoring system for chronic heart failure patients to reduce readmissions: A two-arm randomized pilot study. J. Med. Int. Res. 2016, 18, e91. [Google Scholar] [CrossRef]
| Question Item | Survey Source | Response Triggering Alert |
|---|---|---|
| Increased hostility or aggression | Symptoms and side effects severity rating | “Increased hostility and aggression” (mild, moderate, or severe) AND “change for the worse” 1,2 |
| At risk of hurting self or others | Symptoms and side effects severity rating | “Expresses thoughts of hurting self or others (mild, moderate, or severe)” AND “change for the worse” 1,2 |
| Clinical improvement since start of treatment or start of the most recent change in treatment | CGI-I | Clinical improvement rated as “much worse” OR “very much worse” 2 |
| Alert Group (N = 98) | Non-Alert Group (N = 420) | ||||
|---|---|---|---|---|---|
| N | mean (SD) | mean (SD) | Δ (95% CI) | p | |
| Age (years) | 518 | 9.85 (3.21) | 11.09 (3.24) | 1.2 (0.5, 1.9) | <0.001 * |
| N | N (%) | N (%) | OR (95% CI) | p | |
| Sex | 0.6 (0.34, 1.06) | 0.08 | |||
| Female | 126 | 17 (17.4%) | 109 (26.0%) | ||
| Male | 392 | 81 (82.7%) | 311 (74.1%) | ||
| Race (56 missing) | 1.32 (0.60, 2.91) | 0.49 | |||
| White | 406 | 72 (90.0%) | 334 (87.2%) | ||
| Non-White | 57 | 8 (10.0%) | 49 (12.8%) | ||
| Ethnicity (114 missing) | 1.21 (0.48, 3.08) | 0.69 | |||
| Hispanic | 30 | 6 (8.6%) | 24 (7.2%) | ||
| Non-Hispanic | 374 | 64 (91.4%) | 310 (9.8%) | ||
| Insurance (7 missing) | 1.79 (1.14, 2.81) | 0.01 * | |||
| Any public insurance | 164 | 42 (42.9%) | 122 (29.6%) | ||
| Private | 347 | 56 (57.1%) | 291 (70.5%) |
| Vanderbilt Scores (Range: 0 [Least Severe] to 54 [Most Severe]) | ||||
| Alert Group (N= 62) | Non-Alert Group (N = 202) | |||
| mean | mean | Δ (95% CI) | p * | |
| Time 1 ** | 24.8 | 22.1 | ||
| Time 2 | 28.2 | 20.2 | ||
| Diff | 3.4 (SD: ±9.5) | −1.9 (SD: ±7.4) | 5.8 (3.5, 8.1) *** | 0.001 |
| CGI-S scores (Range: 0 [Most Impaired] to 9 [Least Impaired]) | ||||
| Alert group (N = 61) | Non-Alert group (N = 202) | |||
| mean | mean | Δ (95% CI) | p * | |
| Time 1 ** | 4.8 | 5.3 | ||
| Time 2 | 4.6 | 5.4 | ||
| Diff | −0.2 (SD: ±0.9) | 0.1 (SD: ±0.8) | −0.3 (−0.5, −0.1) *** | 0.015 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Oppenheimer, J.; Ojo, O.; Antonetty, A.; Chiujdea, M.; Garcia, S.; Weas, S.; Loddenkemper, T.; Fleegler, E.; Chan, E. Timely Interventions for Children with ADHD through Web-Based Monitoring Algorithms. Diseases 2019, 7, 20. https://doi.org/10.3390/diseases7010020
Oppenheimer J, Ojo O, Antonetty A, Chiujdea M, Garcia S, Weas S, Loddenkemper T, Fleegler E, Chan E. Timely Interventions for Children with ADHD through Web-Based Monitoring Algorithms. Diseases. 2019; 7(1):20. https://doi.org/10.3390/diseases7010020
Chicago/Turabian StyleOppenheimer, Julia, Oluwafemi Ojo, Annalee Antonetty, Madeline Chiujdea, Stephanie Garcia, Sarah Weas, Tobias Loddenkemper, Eric Fleegler, and Eugenia Chan. 2019. "Timely Interventions for Children with ADHD through Web-Based Monitoring Algorithms" Diseases 7, no. 1: 20. https://doi.org/10.3390/diseases7010020
APA StyleOppenheimer, J., Ojo, O., Antonetty, A., Chiujdea, M., Garcia, S., Weas, S., Loddenkemper, T., Fleegler, E., & Chan, E. (2019). Timely Interventions for Children with ADHD through Web-Based Monitoring Algorithms. Diseases, 7(1), 20. https://doi.org/10.3390/diseases7010020




