Polymyalgia Rheumatica (PMR) with Normal Values of Both Erythrocyte Sedimentation Rate (ESR) and C-Reactive Protein (CRP) Concentration at the Time of Diagnosis in a Centenarian Man: A Case Report
Abstract
1. Introduction
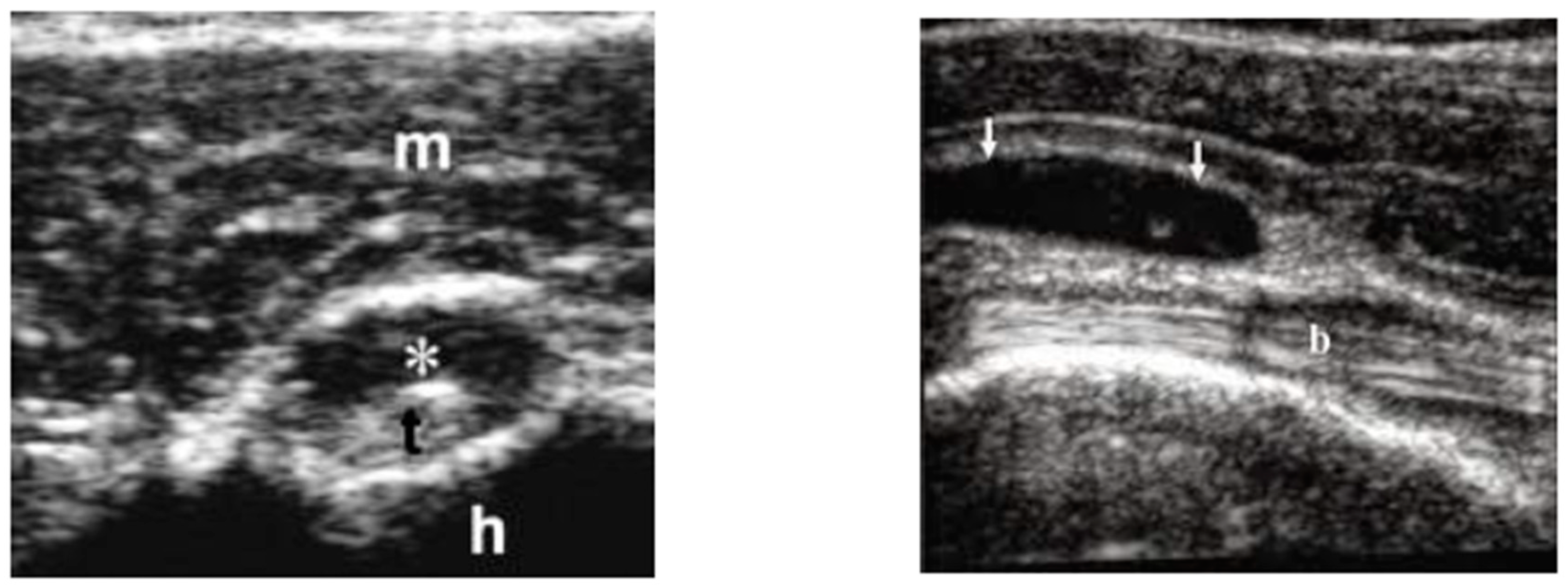
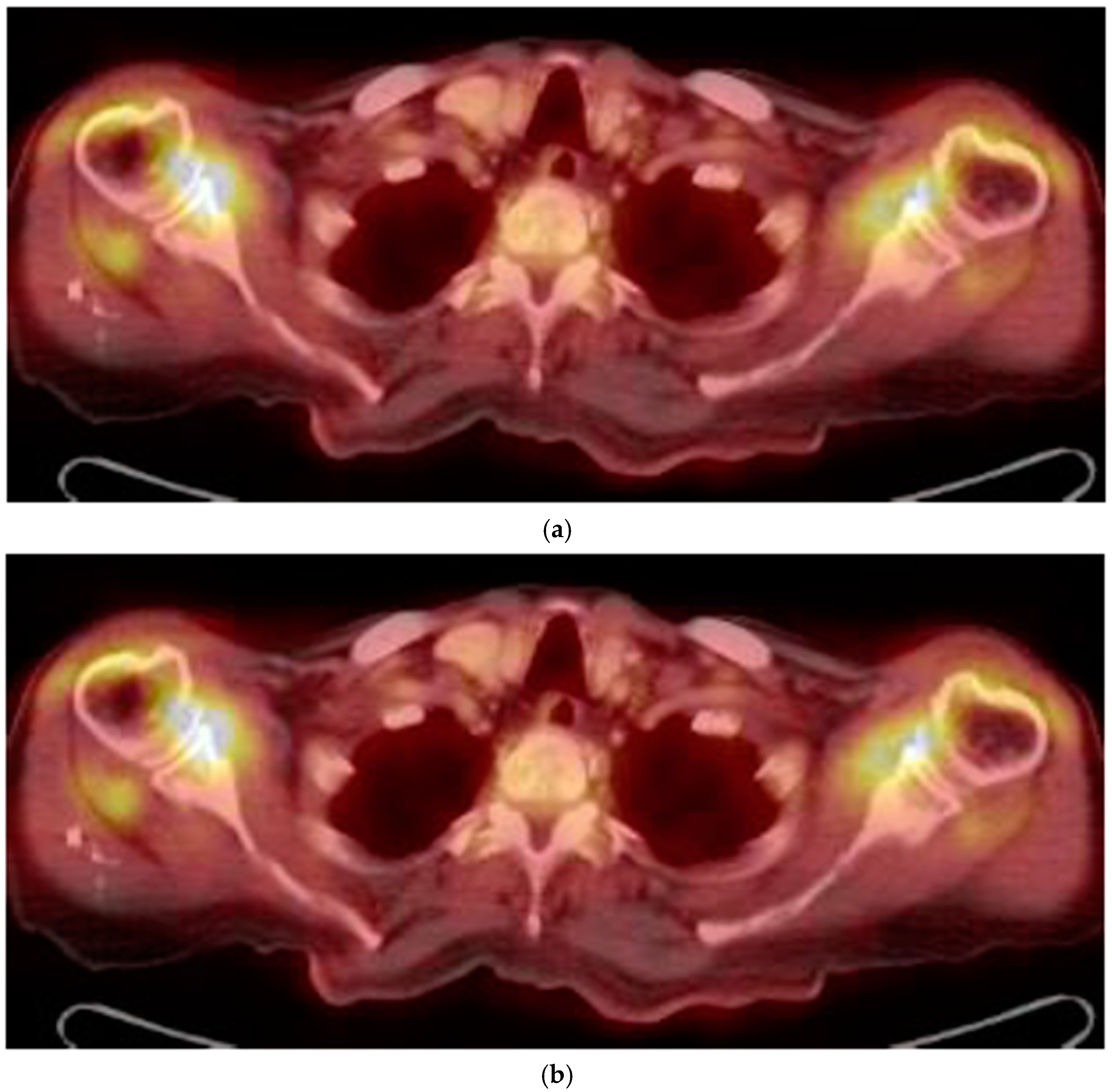
2. Case Report
3. Discussion
4. Conclusions
5. Take-Home Messages
- The diagnosis of PMR is possible, even if ESR and CRP have not increased.
- US and 18-FDG PET/CT evaluations might increase diagnostic rates of ESR and CRP negative PMR.
- A rigorous diagnostic work-up and a long follow-up (one year, as minimum) are mandatory to avoid mistakes.
- The onset of PMR in a centenarian is exceptional. In this age range, the genetic background seems to be important in conditioning laboratory data more than in other periods of life.
Funding
Conflicts of Interest
Consent for Publication
References
- Matteson, E.L.; Dejaco, C. Polymyalgia rheumatica. Ann. Intern. Med. 2017, 166, ITC65–ITC80. [Google Scholar] [CrossRef] [PubMed]
- Sobrero, A.; Manzo, C.; Stimamiglio, A. The role of the general practitioner and the out-of-hospital rheumatologist in the diagnosis and follow-up of patients with polymyalgia rheumatica. Reumatismo 2018, 70, 44–50. [Google Scholar] [CrossRef] [PubMed]
- Yates, M.; Graham, K.; Watts, R.A.; Mc Gregor, A.J. The prevalence of giant cell arteritis and polymyalgia rheumatica in a UK primary care population. BMC Musculoskelet. Dirord. 2016, 17, 285. [Google Scholar] [CrossRef] [PubMed]
- Milchert, M.; Brzosko, M. Diagnosis of polymyalgia rheumatica usually means a favourable outcome for your patient. Indian J. Med. Res. 2017, 145, 593–600. [Google Scholar] [PubMed]
- Bird, H.A.; Leeb, B.F.; Montecucco, C.M.; Misiuniene, N.; Nesher, G.; Pai, S.; Pease, C.; Rovensky, J.; Rozman, B. A comparison of the sensitivity of diagnostic criteria for polymyalgia rheumatica. Ann. Rheum. Dis. 2005, 64, 626–629. [Google Scholar] [CrossRef] [PubMed]
- Muratore, F.; Salvarani, C.; Macchioni, P. Contribution of the new 2012 EULAR/ACR classification criteria for the diagnosis of polymyalgia rheumatica. Reumatismo 2018, 70, 18–22. [Google Scholar] [CrossRef] [PubMed]
- Nesher, G. Polymyalgia rheumatica-diagnosis and classification. J. Autoimmun. 2014, 48–49, 76–78. [Google Scholar] [CrossRef] [PubMed]
- Gonzalez-Gay, M.A.; Matteson, E.L.; Castaneda, S. Polymyalgia reumatica. Lancet 2017, 390, 1700–1712. [Google Scholar] [CrossRef]
- Manzo, C.; Milchert, M. Polymyalgia rheumatica with normal values of both erythrocyte sedimentation rate and C-reactive protein concentration at the time of diagnosis: A four-point guidance. Reumatologia 2018, 56, 1–2. [Google Scholar] [CrossRef] [PubMed]
- Dejaco, C.; Singh, Y.P.; Perel, P.; Hutchings, A.; Camellino, D.; Mackie, S.; Abril, A.; Bachta, A.; Balint, P.; Barraclough, K.; et al. 2015 Recommendations for the management of polymyalgia rheumatica: A European League against Rheumatism/American College of Rheumatology collaborative initiative. Ann. Rheum. Dis. 2015, 74, 1799–1807. [Google Scholar] [CrossRef] [PubMed]
- Manzo, C.; Camellino, D. Polymyalgia Rheumatica (PMR): Diagnostic and therapeutic issues of an apparently straightforward disease. Recenti Prog. Med. 2017, 108, 221–231. [Google Scholar] [PubMed]
- Ceccato, F.; Uña, C.; Regidor, M.; Rillo, O.; Babini, S.; Paira, S. Conditions mimicking polymyalgia rheumatica. Reumatol. Clin. 2011, 7, 156–160. [Google Scholar] [CrossRef] [PubMed]
- Olivieri, I.; Garcia-Porrua, C.; Padula, A.; Cantini, F.; Salvarani, C.; Gonzalez-Gay, M.A. Late onset undifferentiated spondyloarthritis presenting with polymyalgia rheumatica features: Description of seven cases. Rheumatol. Int. 2007, 27, 927–933. [Google Scholar] [CrossRef] [PubMed]
- Dasgupta, B.; Cimmino, M.A.; Kremers, H.M.; Schmidt, W.A.; Schirmer, M.; Salvarani, C.; Bachta, A.; Dejaco, C.; Duftner, C.; Jensen, H.S.; et al. 2012 provisional classification criteria for polymyalgia rheumatica: A European League against Rheumatism/American College of Rheumatology collaborative initiative. Ann. Rheum. Dis. 2012, 71, 484–492. [Google Scholar] [CrossRef] [PubMed]
- Yuge, S.; Nakatani, K.; Yoshino, K.; Koyama, T. Diagnosing polymyalgia rheumatica on 18-F-FDG PET/CT: Typical uptake patterns. Ann. Nucl. Med. 2018. [Google Scholar] [CrossRef] [PubMed]
- Dejaco, C.; Ramiro, S.; Duftner, C.; Besson, F.L.; Bley, T.A.; Blockmans, D.; Brouwer, E.; Cimmino, M.A.; Clark, E.; Dasgupta, B.; et al. EULAR reccomendations for the use of imaging in large vessel vasculitis in clinical practice. Ann. Rheum. Dis. 2018, 77, 636–643. [Google Scholar] [CrossRef] [PubMed]
- Doria, A.; Zen, M.; Bettio, S.; Gatto, M.; Bassi, N.; Nalotto, L.; Ghirardello, A.; Iaccarino, L.; Punzi, L. Autoinflammation and autoimmunity: Brindging the divide. Autoimmun. Rev. 2012, 12, 22–30. [Google Scholar] [CrossRef] [PubMed]
- González-Gay, M.A.; Rodríiguez-Valverde, V.; Blanco, R.; Fernández-Sueiro, J.L.; Armona, J.; Figueroa, M.; Martínez-Taboada, V.M. Polymyalgia rheumatica without significantly increased erythrocyte sedimentation rate. A more benign syndrome. Arch. Intern. Med. 1997, 157, 317–320. [Google Scholar] [CrossRef] [PubMed]
- Bray, C.; Bell, L.N.; Liang, H.; Haykal, R.; Kaiksow, F.; Mazza, J.J.; Yale, S.H. Erythrocyte sedimentation rate and C-reactive protein measurements and their relevance in clinical medicine. WMJ 2016, 115, 317–321. [Google Scholar] [PubMed]
- Martínez-Taboada, V.M.; Blanco, R.; Rodríguez-Valverde, V. Polymyalgia rheumatica with normal erythrocyte sedimentation rate: Clinical aspects. Clin. Exp. Rheumatol. 2000, 18 (Suppl. 20), S34–S37. [Google Scholar] [PubMed]
- Bonafè, M.; Olivieri, F.; Cavallone, L.; Giovagnetti, S.; Marchegiani, F.; Cardelli, M.; Pieri, C.; Marra, M.; Antonicelli, R.; Lisa, R.; et al. A gender-dependent genetic predisposition to produce high levels of IL-6 is detrimental for longevity. Eur. J. Immunol. 2001, 31, 2357–2361. [Google Scholar] [CrossRef]
- Arai, Y.; Martin-Ruiz, C.M.; Takayama, M.; Abe, Y.; Takebayashi, T.; Koyasu, S.; Suematsu, M.; Hirose, N.; von Zglinicki, T. Inflammation, but not telomere length, predicts successful ageing at extreme old age: A longitudinal study of semi-supercentenarians. EBioMedicine 2015, 2, 1549–1558. [Google Scholar] [CrossRef] [PubMed]
- Salvioli, S.; Capri, M.; Bucci, L.; Lanni, C.; Racchi, M.; Uberti, D.; Memo, M.; Mari, D.; Govoni, S.; Franceschi, C. Why do centenarians escape or postpone cancer? The role of IGF-1, inflammation and p53. Cancer Immunol. Immunother. 2009, 58, 1909–1917. [Google Scholar] [CrossRef] [PubMed]
- Marzo-Ortega, H.; McGonagle, D.; O’Connor, P.; Pease, C.; Emery, P. Subclinical vasculitis in polymyalgia rheumatica. Ann. Rheum. Dis. 2001, 60, 1058–1059. [Google Scholar] [CrossRef] [PubMed]
- Kermani, T.A.; Schmidt, J.; Crowson, C.S.; Ytterberg, S.R.; Hunder, G.G.; Matteson, E.L.; Warrington, K.J. Utility of erythrocyte sedimentation rate and C-reactive protein for the diagnosis of giant cell arteritis. Semin. Arthritis Rheum. 2012, 41, 866–871. [Google Scholar] [CrossRef] [PubMed]
- Manzo, C.; Natale, M.; Traini, E. Diagnosis of polymyalgia rheumatica in primary health care: Favoring and confounding factors—A cohort study. Reumatologia 2018, 56, 131–139. [Google Scholar] [CrossRef] [PubMed]
© 2018 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Manzo, C. Polymyalgia Rheumatica (PMR) with Normal Values of Both Erythrocyte Sedimentation Rate (ESR) and C-Reactive Protein (CRP) Concentration at the Time of Diagnosis in a Centenarian Man: A Case Report. Diseases 2018, 6, 84. https://doi.org/10.3390/diseases6040084
Manzo C. Polymyalgia Rheumatica (PMR) with Normal Values of Both Erythrocyte Sedimentation Rate (ESR) and C-Reactive Protein (CRP) Concentration at the Time of Diagnosis in a Centenarian Man: A Case Report. Diseases. 2018; 6(4):84. https://doi.org/10.3390/diseases6040084
Chicago/Turabian StyleManzo, Ciro. 2018. "Polymyalgia Rheumatica (PMR) with Normal Values of Both Erythrocyte Sedimentation Rate (ESR) and C-Reactive Protein (CRP) Concentration at the Time of Diagnosis in a Centenarian Man: A Case Report" Diseases 6, no. 4: 84. https://doi.org/10.3390/diseases6040084
APA StyleManzo, C. (2018). Polymyalgia Rheumatica (PMR) with Normal Values of Both Erythrocyte Sedimentation Rate (ESR) and C-Reactive Protein (CRP) Concentration at the Time of Diagnosis in a Centenarian Man: A Case Report. Diseases, 6(4), 84. https://doi.org/10.3390/diseases6040084




