Action Observation in People with Parkinson’s Disease. A Motor–Cognitive Combined Approach for Motor Rehabilitation. A Preliminary Report
Abstract
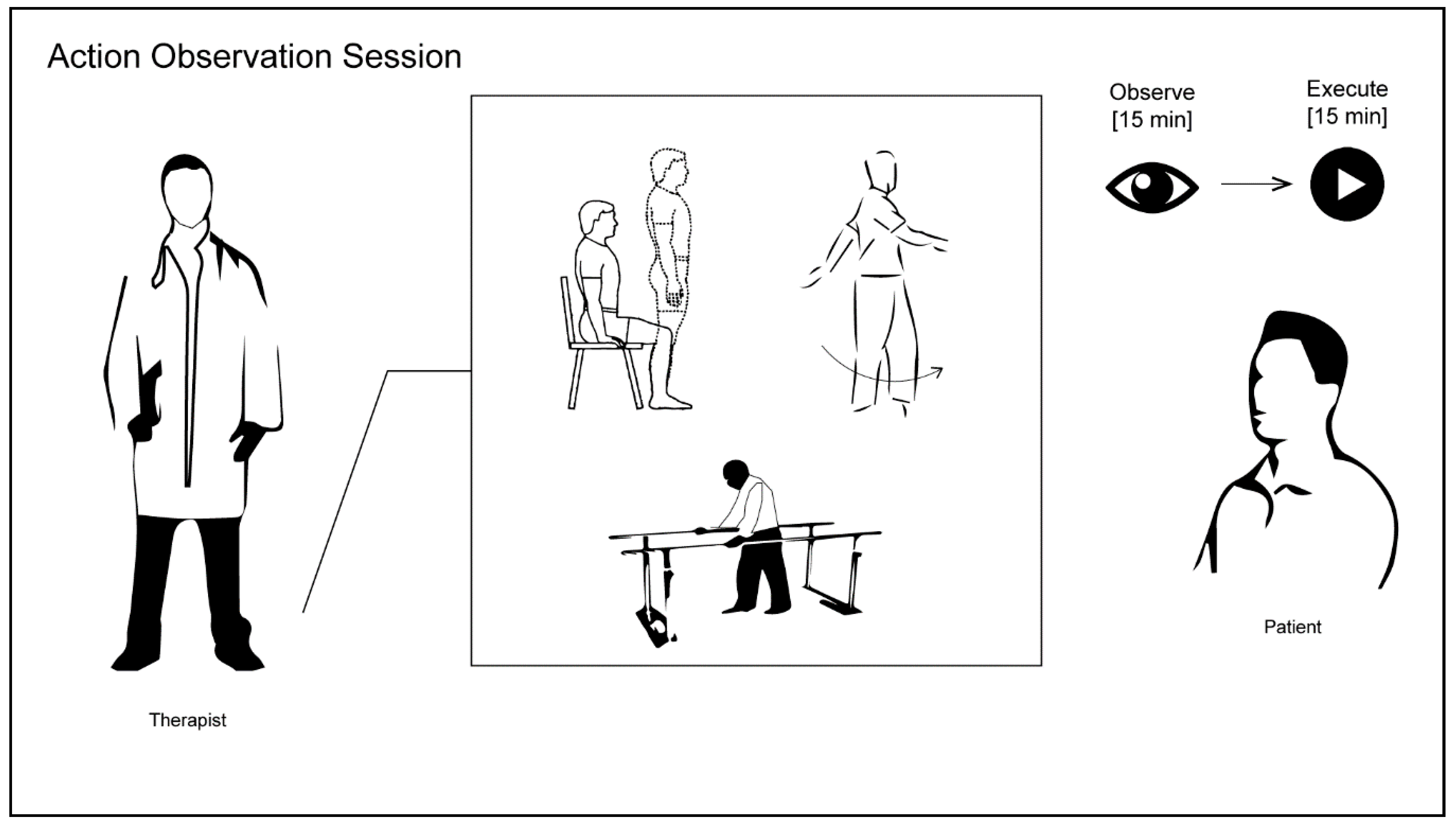
- Rising from a chair without the help of the upper limbs.
- Alternating monopodial support for 2–3 s per side.
- Going through a parallel walking bar (approximately 3 m) and overriding a small wood obstacle, about 15 cm in height.
- Going out from parallel walking bars.
- Change of direction while walking: 90° turn to the right.
- Change of direction while walking: 90° turn to the left.
- Walking through a narrow space consisting of two beds closely displaced (about 2 m).
- Going back to the starting point (chair).
Author Contributions
Funding
Conflicts of Interest
References
- Sale, P.; De Pandis, M.F.; Le Pera, D.; Sova, I.; Cimolin, V.; Ancillao, A.; Albertini, G.; Galli, M.; Stocchi, F.; Franceschini, M. Robot-assisted walking training for individuals with Parkinson’s disease: A pilot randomized controlled trial. BMC Neurol. 2013, 13, 50. [Google Scholar] [CrossRef] [PubMed]
- Sale, P.; Franceschini, M. Action Observation and Mirror Neuron Network: A Tool for Motor Stroke Rehabilitation. Eur. J. Phys. Rehabil. Med. 2012, 48, 313–318. Available online: http://www.ncbi.nlm.nih.gov/pubmed/22522432 (accessed on 14 June 2018). [PubMed]
- Abbruzzese, G.; Avanzino, L.; Marchese, R.; Pelosin, E. Action Observation and Motor Imagery: Innovative Cognitive Tools in the Rehabilitation of Parkinson’s Disease. Parkinson’s Dis. 2015, 2015, 1–9. Available online: http://www.ncbi.nlm.nih.gov/pubmed/26495150 (accessed on 17 May 2018). [CrossRef] [PubMed]
- Sale, P.; Ceravolo, M.G.; Franceschini, M. Action Observation Therapy in the Subacute Phase Promotes Dexterity Recovery in Right-Hemisphere Stroke Patients. Biomed. Res. Int. 2014, 2014, 1–7. Available online: http://www.hindawi.com/journals/bmri/2014/457538/ (accessed on 14 June 2018). [CrossRef] [PubMed]
- Franceschini, M.; Ceravolo, M.G.; Agosti, M.; Cavallini, P.; Bonassi, S.; Dall’Armi, V.; Massucci, M.; Schifini, F.; Sale, P. Clinical Relevance of Action Observation in Upper-Limb Stroke Rehabilitation: A Possible Role in Recovery of Functional Dexterity. A Randomized Clinical Trial. Neurorehabil. Neural Repair 2012, 26, 456–462. Available online: http://nnr.sagepub.com/cgi/doi/10.1177/1545968311427406 (accessed on 14 June 2018). [CrossRef] [PubMed]
- Franceschini, M.; Agosti, M.; Cantagallo, A.; Sale, P.; Mancuso, M.; Buccino, G. Mirror Neurons: Action Observation Treatment as a Tool in Stroke Rehabilitation. Eur. J. Phys. Rehabil. Med. 2010, 46, 517–523. [Google Scholar] [PubMed]
- Rocca, M.A.; Fumagalli, S.; Pagani, E.; Gatti, R.; Riccitelli, G.C.; Preziosa, P.; Comi, G.; Falini, A.; Filippi, M. Action Observation Training Modifies Brain Gray Matter Structure in Healthy Adult Individuals. Brain Imaging Behav. 2017, 11, 1343–1352. Available online: http://www.ncbi.nlm.nih.gov/pubmed/27730478 (accessed on 14 June 2018). [CrossRef] [PubMed]
- Mizuguchi, N.; Kanosue, K. Changes in Brain Activity during Action Observation and Motor Imagery: Their Relationship with Motor Learning. Prog. Brain Res. 2017, 234, 189–204. Available online: http://www.ncbi.nlm.nih.gov/pubmed/29031463 (accessed on 14 June 2018). [PubMed]
- Mouthon, A.; Ruffieux, J.; Mouthon, M.; Hoogewoud, H.-M.; Annoni, J.-M.; Taube, W. Age-Related Differences in Cortical and Subcortical Activities during Observation and Motor Imagery of Dynamic Postural Tasks: An fMRI Study. Neural Plast. 2018, 2018, 1–12. Available online: http://www.ncbi.nlm.nih.gov/pubmed/29675037 (accessed on 14 June 2018). [CrossRef] [PubMed]
- Bähr, F.; Ritter, A.; Seidel, G.; Puta, C.; Gabriel, H.H.W.; Hamzei, F. Boosting the Motor Outcome of the Untrained Hand by Action Observation: Mirror Visual Feedback, Video Therapy, or both Combined—What Is More Effective? Neural Plast. 2018, 2018, 1–10. Available online: http://www.ncbi.nlm.nih.gov/pubmed/29849570 (accessed on 14 June 2018). [CrossRef] [PubMed]
- Zhang, J.J.Q.; Fong, K.N.K.; Welage, N.; Liu, K.P.Y. The Activation of the Mirror Neuron System during Action Observation and Action Execution with Mirror Visual Feedback in Stroke: A Systematic Review. Neural Plast. 2018, 2018, 1–14. Available online: http://www.ncbi.nlm.nih.gov/pubmed/29853839 (accessed on 14 June 2018). [CrossRef] [PubMed]
- Özmüş, G.; Yerlikaya, D.; Gökçeoğlu, A.; Savaş, D.D.; Cakmur, R.; Çolakoğlu, B.D.; Yener, G.G. Demonstration of Early Cognitive Impairment in Parkinson’s Disease with Visual P300 Responses. Arch. Neuropsychiatry 2017, 54, 21–27. Available online: http://www.ncbi.nlm.nih.gov/pubmed/28566954 (accessed on 17 May 2018). [CrossRef] [PubMed]
- Polich, J. Updating P300: An Integrative Theory of P3a and P3b. Clin. Neurophysiol. 2007, 118, 2128–2148. Available online: http://www.ncbi.nlm.nih.gov/pubmed/17573239 (accessed on 17 May 2018). [CrossRef] [PubMed]
- Goodin, D.S. Cognitive Event-Related Potentials. J. Clin. Neurophysiol. 1999, 52, 91–95. Available online: https://insights.ovid.com/crossref?an=00004691-199801000-00002 (accessed on 17 May 2018). [CrossRef]
- Mecklinger, A.; Kramer, A.F.; Strayer, D.L. Event Related Potentials and EEG Components in a Semantic Memory Search Task. Psychophysiology 1992, 29, 104–119. Available online: http://doi.wiley.com/10.1111/j.1469-8986.1992.tb02021.x (accessed on 17 May 2018). [CrossRef] [PubMed]
- Haider, M.; Spong, P.; Lindsley, D.B.; John, E.R. Attention, Vigilance, and Cortical Evoked-Potentials in Humans. Science 1964, 145, 180–182. Available online: http://www.ncbi.nlm.nih.gov/pubmed/14171563 (accessed on 17 May 2018). [CrossRef] [PubMed]
- Goodin, D.; Desmedt, J.; Maurer, K.; Nuwer, M.R. IFCN Recommended Standards for Long-Latency Auditory Event-Related Potentials. Report of an IFCN Committee. Electroencephalogr. Clin. Neurophysiol. 1994, 91, 18–20. Available online: http://linkinghub.elsevier.com/retrieve/pii/0013469494900140 (accessed on 17 May 2018). [CrossRef]
- Ferrazzoli, D.; Ortelli, P.; Madeo, G.; Giladi, N.; Petzinger, G.M.; Frazzitta, G. Basal Ganglia and Beyond: The Interplay between Motor and Cognitive Aspects in Parkinson’s Disease Rehabilitation. Neurosci. Biobehav. Rev. 2018, 90, 294–308. Available online: https://www.sciencedirect.com/science/article/pii/S0149763418301611 (accessed on 14 June 2018). [CrossRef] [PubMed]
- Buccino, G.; Gatti, R.; Giusti, M.C.; Negrotti, A.; Rossi, A.; Calzetti, S.; Cappa, S.F. Action Observation Treatment Improves Autonomy in Daily Activities in Parkinson’s Disease Patients: Results from a Pilot Study. Mov. Disord. 2011, 26, 1963–1964. Available online: http://doi.wiley.com/10.1002/mds.23745 (accessed on 28 March 2018). [CrossRef] [PubMed]
- Pelosin, E.; Avanzino, L.; Bove, M.; Stramesi, P.; Nieuwboer, A.; Abbruzzese, G. Action Observation Improves Freezing of Gait in Patients With Parkinson’s Disease. Neurorehabil. Neural Repair 2010, 24, 746–752. Available online: http://journals.sagepub.com/doi/10.1177/1545968310368685 (accessed on 28 March 2018). [CrossRef] [PubMed]
- Gil, R.; Neau, J.P.; Toullat, G.; Rivasseau-Jonveaux, T.; Lefèvre, J.P. Parkinson Disease and Cognitive Evoked Potentials. Rev. Neurol. 1989, 145, 201–207. Available online: http://www.ncbi.nlm.nih.gov/pubmed/2749097 (accessed on 28 March 2018). [PubMed]
- Jiang, C.; Kaseda, Y.; Kumagai, R.; Nakano, Y.; Nakamura, S. Habituation of Event-Related Potentials in Patients with Parkinson’s Disease. Physiol. Behav. 2000, 68, 741–747. Available online: http://www.ncbi.nlm.nih.gov/pubmed/10764905 (accessed on 28 March 2018). [CrossRef]
- Lukhanina, E.P.; Kapustina, M.T.; Berezetskaya, N.M.; Karaban, I.N. Reduction of the Postexcitatory Cortical Inhibition Upon Paired-Click Auditory Stimulation in Patients with Parkinson’s Disease. Clin. Neurophysiol. 2009, 120, 1852–1858. Available online: http://www.ncbi.nlm.nih.gov/pubmed/19767236 (accessed on 28 March 2018). [CrossRef] [PubMed]
- Matsui, H.; Nishinaka, K.; Oda, M.; Kubori, T.; Udaka, F. Auditory Event-Related Potentials in Parkinson’s Disease: Prominent Correlation with Attention. Parkinsonism. Relat. Disord. 2007, 13, 394–398. Available online: http://www.ncbi.nlm.nih.gov/pubmed/17329143 (accessed on 28 March 2018). [CrossRef] [PubMed]
- Da Silva Lopes, M.; de Souza Melo, A.; Nóbrega, A.C. Delayed Latencies of Auditory Evoked Potential P300 Are Associated with the Severity of Parkinson’s Disease in Older Patients. Arquivos de Neuro-Psiquiatria 2014, 72, 296–300. Available online: http://www.ncbi.nlm.nih.gov/pubmed/24760094 (accessed on 28 March 2018). [CrossRef]
- Sohn, Y.H.; Kim, G.W.; Huh, K.; Kim, G.-S. Dopaminergic influences on the P300 abnormality in Parkinson’s disease. J. Neurol. Sci. 1998, 158, 83–87. [Google Scholar] [CrossRef]
- Solís-Vivanco, R.; Rodríguez-Violante, M.; Rodríguez-Agudelo, Y.; Schilmann, A.; Rodríguez-Ortiz, U.; Ricardo-Garcell, J. The P3a Wave: A Reliable Neurophysiological Measure of Parkinson’s Disease Duration and Severity. Clin. Neurophysiol. 2015, 126, 2142–2149. Available online: http://www.ncbi.nlm.nih.gov/pubmed/25655938 (accessed on 14 June 2018). [CrossRef] [PubMed]
- Tang, H.; Huang, J.; Nie, K.; Gan, R.; Wang, L.; Zhao, J.; Huang, Z.; Zhang, Y.; Wang, L. Cognitive Profile of Parkinson’s Disease Patients: A Comparative Study between Early-Onset and Late-Onset Parkinson’s Disease. Int. J. Neurosci. 2016, 126, 227–234. Available online: http://www.ncbi.nlm.nih.gov/pubmed/26001202 (accessed on 14 June 2018). [CrossRef] [PubMed]
- Emek-Savaş, D.D.; Özmüş, G.; Güntekin, B.; Dönmez Çolakoğlu, B.; Çakmur, R.; Başar, E.; Yener, G.G. Decrease of Delta Oscillatory Responses in Cognitively Normal Parkinson’s Disease. Clin. EEG Neurosci. 2017, 48, 355–364. Available online: http://www.ncbi.nlm.nih.gov/pubmed/27582502 (accessed on 14 June 2018). [CrossRef] [PubMed]
- Te Woerd, E.S.; Oostenveld, R.; de Lange, F.P.; Praamstra, P. Impaired Auditory-To-Motor Entrainment in Parkinson’s Disease. J. Neurophysiol. 2017, 117, 1853–1864. Available online: http://www.ncbi.nlm.nih.gov/pubmed/28179479 (accessed on 14 June 2018). [CrossRef] [PubMed]
- Tokic, K.; Titlic, M.; BeganovicPetrovic, A.; Suljic, E.; Romac, R.; Silic, S. P300 Wave Changes in Patients with Parkinson’s Disease. Med. Arch. 2016, 70, 453. Available online: http://www.ncbi.nlm.nih.gov/pubmed/28210020 (accessed on 14 June 2018). [CrossRef] [PubMed]
- Agosta, F.; Gatti, R.; Sarasso, E.; Volonté, M.A.; Canu, E.; Meani, A.; Sarro, L.; Copetti, M.; Cattrysse, E.; Kerckhofs, E.; Comi, G. Brain Plasticity in Parkinson’s Disease with Freezing of Gait Induced by Action Observation Training. J. Neurol. 2017, 264, 88–101. Available online: http://www.ncbi.nlm.nih.gov/pubmed/27778161 (accessed on 14 June 2018). [CrossRef] [PubMed]
| Mean | Std. Deviation | Std. Error of Mean | T0 vs. T1 | r | 25% Percentile | Median | 75% Percentile | ||
|---|---|---|---|---|---|---|---|---|---|
| Ages of diseases | 8 | 4.528 | 2.025 | 4.5 | 6 | 12.5 | |||
| Gender | 5 Male | 0 female | |||||||
| Age | 71.6 | 6.731 | 3.01 | 66 | 74 | 76 | |||
| UPDRS III | T0 | 34.6 | 12.82 | 5.732 | 23.5 | 33 | 46.5 | ||
| UPDRS III | T1 | 24.2 | 13.33 | 5.962 | p = 0.0082 | −0.102 | 13 | 22 | 36.5 |
| Hoeh&Yahr | T0 | 2.5 | 2.5 | 2.5 | |||||
| Hoeh&Yahr | T1 | n.s. | −0.288 | 2 | 2.5 | 2.5 | |||
| BBS | T0 | 45.4 | 10.45 | 4.675 | 37 | 50 | 51.5 | ||
| BBS | T1 | 52.8 | 2.95 | 1.319 | n.s. | 0.790 | 50.5 | 53 | 55 |
| 10 m walk Sec | T0 | 13.36 | 6.628 | 2.964 | 7.4 | 11.9 | 20.05 | ||
| 10 m walk Sec | T1 | 10.41 | 2.258 | 1.01 | n.s. | 0.200 | 8.35 | 9.95 | 12.7 |
| 10 m walk Step | T0 | 20.8 | 4.266 | 1.908 | 17 | 20 | 25 | ||
| 10 m walk Step | T1 | 20.6 | 2.881 | 1.288 | n.s. | −0.011 | 18.5 | 20 | 23 |
| Time U&G | T0 | 17.96 | 11.41 | 5.101 | 10.35 | 15.3 | 26.9 | ||
| Time U&G | T1 | 12.62 | 3.259 | 1.458 | n.s. | 0.300 | 9.7 | 12.4 | 15.65 |
| SF12 PCS | T0 | 33.12 | 2.758 | 1.233 | 30.65 | 32.6 | 35.85 | ||
| SF12 PCS | T1 | 41.28 | 9.055 | 4.05 | n.s. | −0.300 | 33.4 | 45.1 | 47.25 |
| SF12 MCS | T0 | 37.86 | 6.433 | 2.877 | 31.85 | 40.8 | 42.4 | ||
| SF12 MCS | T1 | 49.9 | 6.6 | 2.952 | p = 0.0007 | −0.900 | 44 | 51.2 | 55.15 |
| FOG-Q | T0 | 15 | 1.871 | 0.8367 | 13.5 | 15 | 16.5 | ||
| FOG-Q | T1 | 8.4 | 2.302 | 1.03 | p = 0.003 | 0.300 | 6.5 | 8 | 10.5 |
| PDQ-39 | T0 | 60.4 | 34.54 | 15.45 | 32 | 49 | 94.5 | ||
| PDQ-39 | T1 | 36 | 23.87 | 10.68 | p = 0.01 | −0.900 | 16 | 27 | 60.5 |
| FIM | T0 | 92.6 | 8.792 | 3.932 | 84.5 | 92 | 101 | ||
| FIM | T1 | 103.2 | 10.52 | 4.705 | n.s. | 0.700 | 93.5 | 108 | 110.5 |
| MMSE | T0 | 25.71 | 1.63 | 0.7292 | 24.37 | 24.97 | 27.43 | ||
| MMSE | T1 | 25 | 0.9925 | 0.4438 | n.s. | −0.300 | 24 | 25.3 | 25.85 |
| P300 LD | T0 | 379.5 | 42.08 | 21.04 | |||||
| P300 LD | T1 | 349.8 | 33.01 | 16.50 | p = 0.0077 | ||||
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Di Iorio, W.; Ciarimboli, A.; Ferriero, G.; Feleppa, M.; Baratto, L.; Matarazzo, G.; Gentile, G.; Masiero, S.; Sale, P. Action Observation in People with Parkinson’s Disease. A Motor–Cognitive Combined Approach for Motor Rehabilitation. A Preliminary Report. Diseases 2018, 6, 58. https://doi.org/10.3390/diseases6030058
Di Iorio W, Ciarimboli A, Ferriero G, Feleppa M, Baratto L, Matarazzo G, Gentile G, Masiero S, Sale P. Action Observation in People with Parkinson’s Disease. A Motor–Cognitive Combined Approach for Motor Rehabilitation. A Preliminary Report. Diseases. 2018; 6(3):58. https://doi.org/10.3390/diseases6030058
Chicago/Turabian StyleDi Iorio, Walter, Alessandro Ciarimboli, Giorgio Ferriero, Michele Feleppa, Luigi Baratto, Giuseppe Matarazzo, Giovanni Gentile, Stefano Masiero, and Patrizio Sale. 2018. "Action Observation in People with Parkinson’s Disease. A Motor–Cognitive Combined Approach for Motor Rehabilitation. A Preliminary Report" Diseases 6, no. 3: 58. https://doi.org/10.3390/diseases6030058
APA StyleDi Iorio, W., Ciarimboli, A., Ferriero, G., Feleppa, M., Baratto, L., Matarazzo, G., Gentile, G., Masiero, S., & Sale, P. (2018). Action Observation in People with Parkinson’s Disease. A Motor–Cognitive Combined Approach for Motor Rehabilitation. A Preliminary Report. Diseases, 6(3), 58. https://doi.org/10.3390/diseases6030058






