Genomic Influence in the Prevention of Cardiovascular Diseases with a Sterol-Based Treatment
Abstract
1. Introduction
2. Materials and Methods
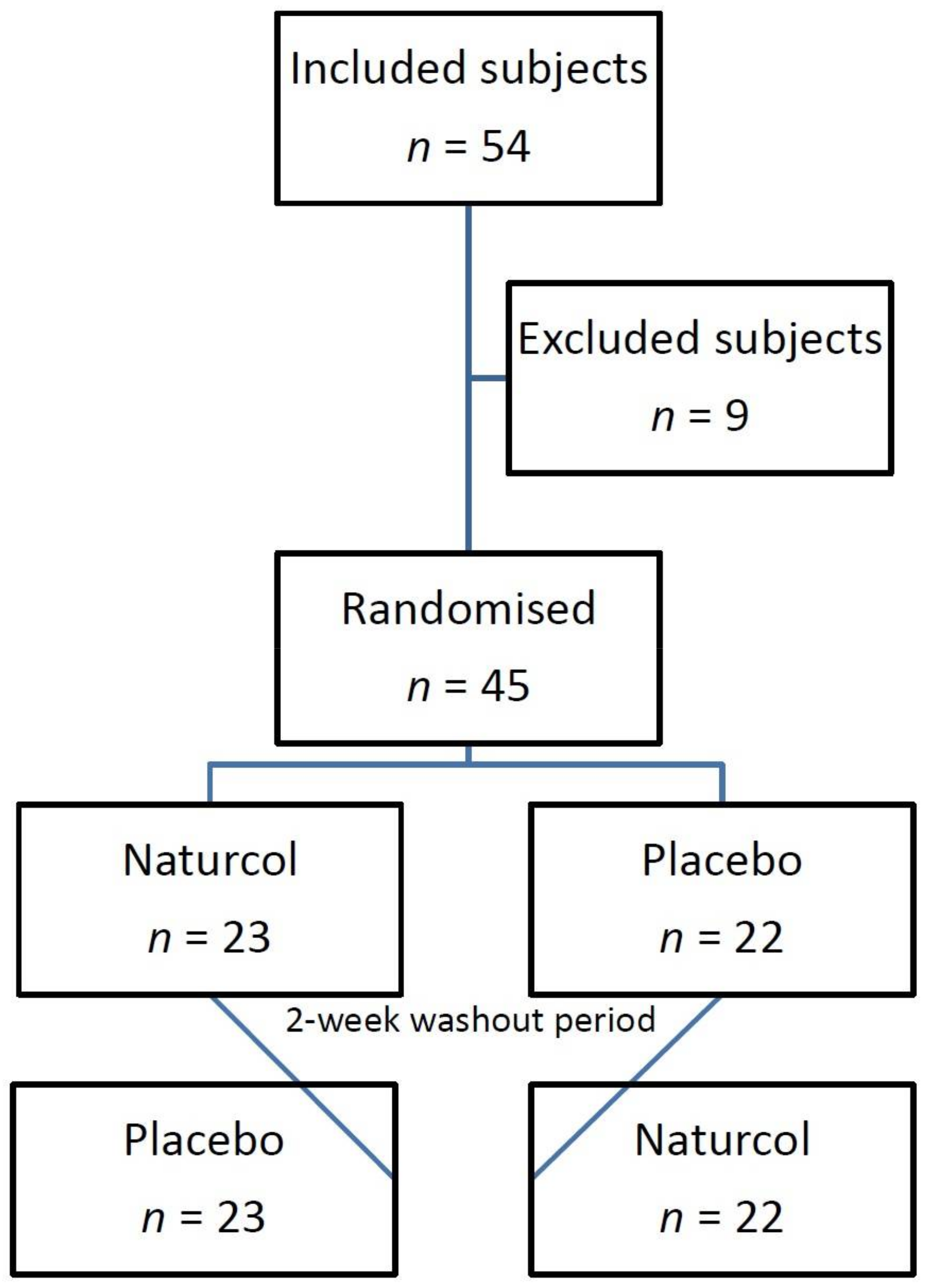
2.1. Sample Size
2.2. Inclusion and Exclusion Criteria
2.3. Clinical Analyses
2.4. Genetic
2.5. Variables and Study Factors
2.6. Statistical Analysis
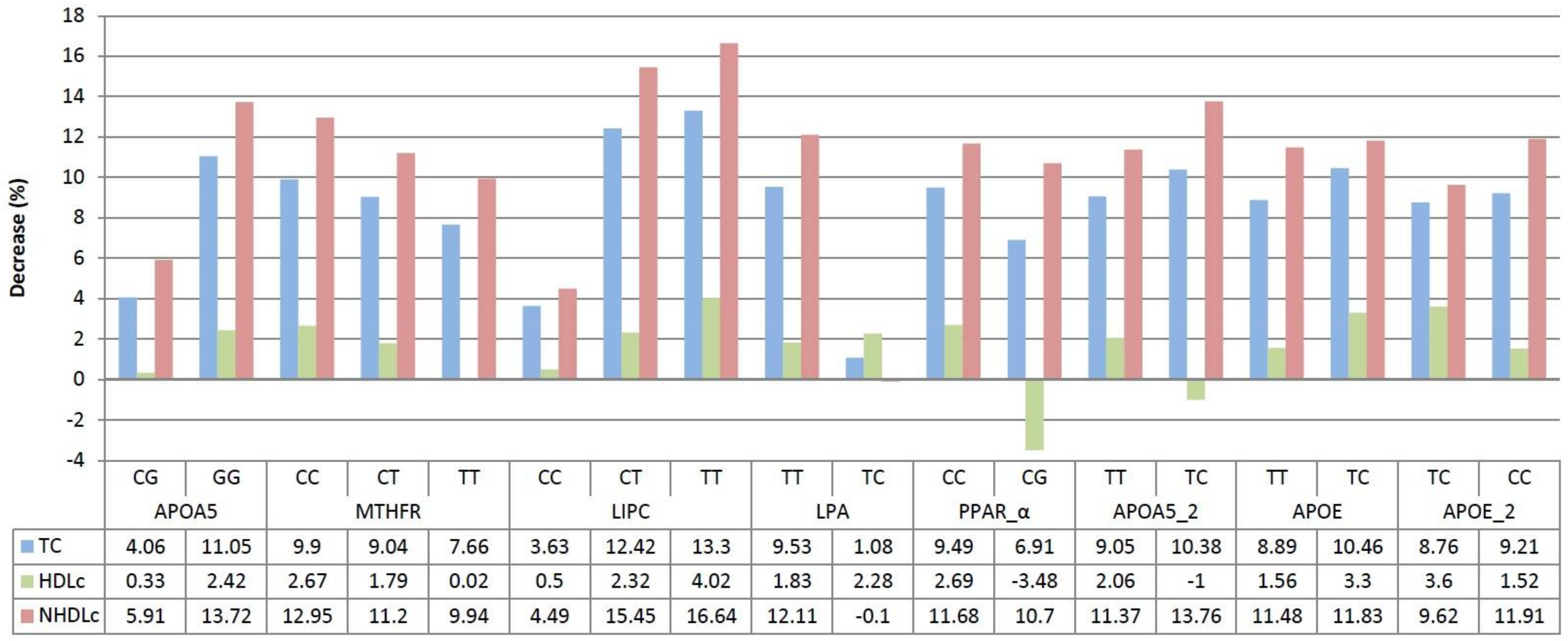
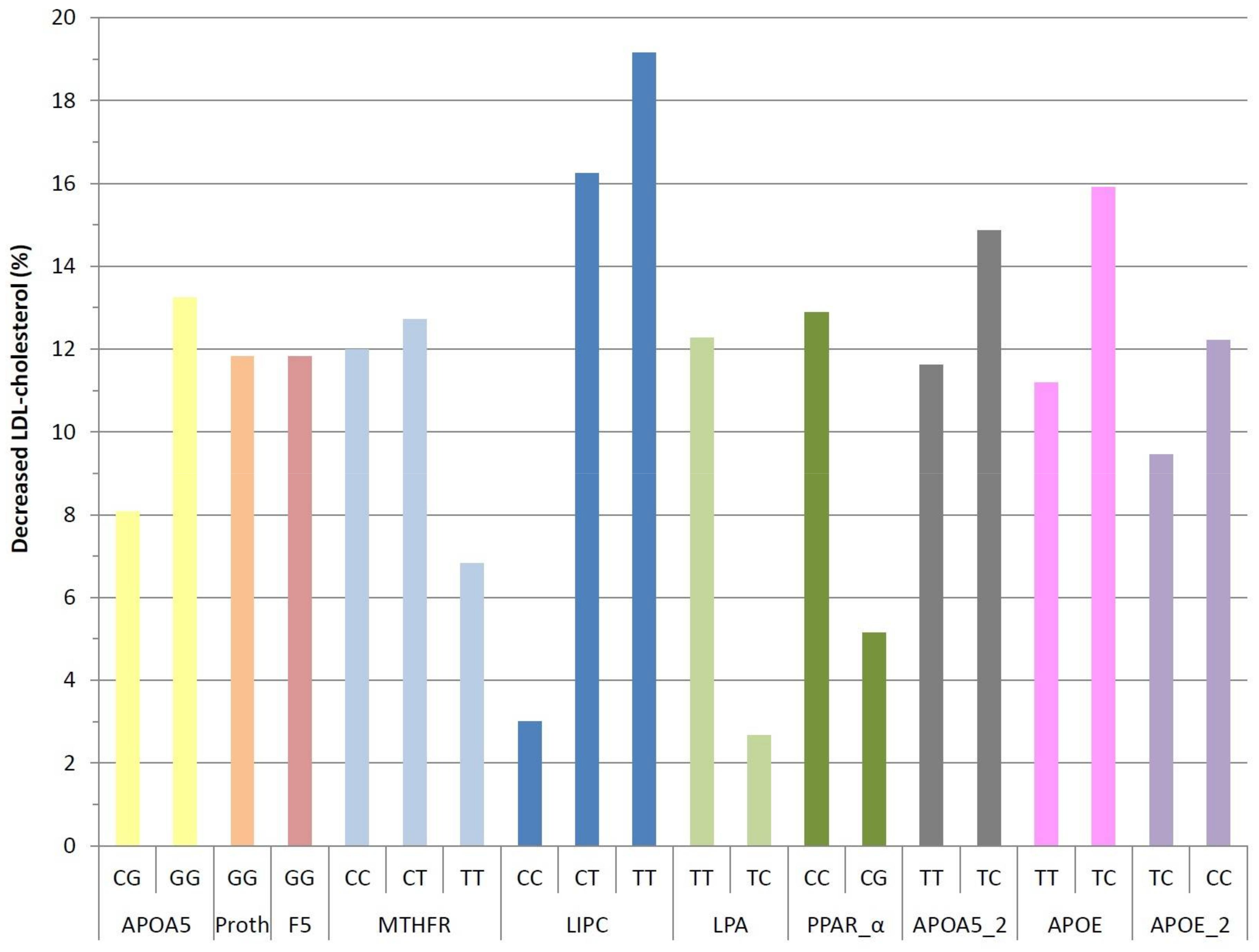
3. Results
4. Discussion
5. Conclusions
Limitations
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Frazier-Wood, A.C. Dietary patterns, genes, and health: Challenges and obstacles to be overcome. Curr. Nutr. Rep. 2015, 4, 82–87. [Google Scholar] [CrossRef] [PubMed]
- Doo, M.; Kim, Y. Obesity: Interactions of genome and nutrients intake. Prev. Nutr. Food Sci. 2015, 20, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Corella, D.; Peloso, G.; Arnett, D.K.; Demissie, S.; Cupples, L.A.; Tucker, K.; Lai, C.Q.; Parnell, L.D.; Coltell, O.; Lee, Y.C.; et al. APOA2, dietary fat, and body mass index: Replication of a gene-diet interaction in 3 independent populations. Arch. Intern. Med. 2009, 169, 1897–1906. [Google Scholar] [CrossRef] [PubMed]
- Seip, R.L.; Volek, J.S.; Windemuthl, A.; Kocherla, M.; Fernandez, M.L.; Kraemer, W.J.; Ruaño, G. Physiogenomic comparison of human fat loss in response to diets restrictive of carbohydrate or fat. Nutr. Metab. 2008, 5, 4. [Google Scholar] [CrossRef] [PubMed]
- Chen, K.H.; Chen, L.L.; Li, W.G.; Fang, Y.; Huang, G.Y. Maternal MTHFR C677T polymorphism and congenital heart defect risk in the Chinese Han population: A meta-analysis. Genet. Mol. Res. 2013, 12, 6212–6219. [Google Scholar] [CrossRef] [PubMed]
- Zhang, M.J.; Li, J.C.; Yin, Y.W.; Li, B.H.; Liu, Y.; Liao, S.Q.; Gao, C.Y.; Zhang, L.L. Association of MTHFR C677T polymorphism and risk of cerebrovascular disease in Chinese population: An updated meta-analysis. J. Neurol. 2014, 261, 925–935. [Google Scholar] [CrossRef] [PubMed]
- Kumar, A.; Kumar, P.; Prasad, M.; Sagar, R.; Yadav, A.K.; Pandit, A.K.; Jali, V.P.; Pathak, A. Association of C677T polymorphism in the methylenetetrahydrofolate reductase gene (MTHFR gene) with ischemic stroke: A meta-analysis. Neurol. Res. 2015, 37, 568–577. [Google Scholar] [CrossRef] [PubMed]
- Edmondson, A.C.; Braund, P.S.; Stylianou, I.M.; Khera, A.V.; Nelson, C.P.; Wolfe, M.L.; Derohannessian, S.L.; Keating, B.J.; Qu, L.; He, J.; et al. Dense genotyping of candidate gene loci identifies variants associated with high-density lipoprotein cholesterol. Circ. Cardiovasc. Genet. 2011, 4, 145–155. [Google Scholar] [CrossRef] [PubMed]
- Nordestgaard, B.G.; Chapman, M.J.; Ray, K.; Borén, J.; Andreotti, F.; Watts, G.F.; Ginsberg, H.; Amarenco, P.; Catapano, A.; Descamps, O.S.; et al. Lipoprotein(a) as a cardiovascular risk factor: Current status. Eur. Heart J. 2010, 31, 2844–2853. [Google Scholar] [CrossRef] [PubMed]
- Donnelly, L.A.; Van Zuydam, N.R.; Zhou, K.; Tavendale, R.; Carr, F.; Maitland-Van der Zee, A.H.; Leusink, M.; de Boer, A.; Doevendans, P.A.; Asselbergs, F.W.; et al. Robust association of the LPA locus with low-density lipoprotein cholesterol lowering response to statin treatment in a meta-analysis of 30467 individuals from both randomized control trials and observational studies and association with coronary artery disease outcome during statin treatment. Pharmacogenet. Genom. 2013, 23, 518–525. [Google Scholar]
- San Mauro-Martín, I.; de la Calle-de la Rosa, L.; Sanz-Rojo, S.; Garicano-Vilar, E.; Ciudad-Cabañas, M.J.; Collado-Yurrita, L. Genomic approach to cardiovascular disease. Nutr. Hosp. 2016, 33, 148–155. [Google Scholar]
- Abdullah, M.M.; Jones, P.J.; Eck, P.K. Nutrigenetics of cholesterol metabolism: Observational and dietary intervention studies in the postgenomic era. Nutr. Rev. 2015, 73, 523–543. [Google Scholar] [CrossRef] [PubMed]
- San Mauro Martin, I.; Collado Yurrita, L.; Ciudad Cabañas, M.J.; Cuadrado Cenzual, M.A.; Hernández Cabria, M.; Calle Purón, M.E. Manejo del riesgo de enfermedad cardiovascular con leche enriquecida en esteroles en población joven adulta; ensayo clínico controlado aleatorizado y cruzado. Nutr. Hosp. 2014, 30, 945–951. [Google Scholar] [PubMed]
- National Institutes of Health. Third Report of the National Cholesterol Educaction Program (NCEP) Expert Panel on Detection, Evaluation, and Treatment of High Blood Cholesterol in Adults (Adult Treatment Panel III). 2002. Available online: http://www.nhlbi.nih.gov/files/docs/resources/heart/atp3full.pdf (accessed on 26 April 2016).
- Alonso Karlezi, R.A.; Mata Pariente, N.; Mata López, P. Control de las hiperlipemias en la práctica clínica. Rev. Esp. Cardiol. Supl. 2006, 6, 24–35. [Google Scholar] [CrossRef]
- Matthan, N.R.; Zhu, L.; Pencina, M.; D’Agostino, R.B.; Schaefer, E.J.; Lichtenstein, A.H. Sex-specific differences in the predictive value of cholesterol homeostasis markers and 10-year cardiovascular disease event rate in Framingham offspring study participants. J. Am. Heart Assoc. 2013, 2, e005066. [Google Scholar] [CrossRef] [PubMed]
- EFSA Panel on Dietetic Products, Nutrition and Allergies (NDA). Scientific Opinion on the substantiation of health claims related to plant sterols and plant stanols and maintenance of normal blood cholesterol concentrations (ID 549, 550, 567, 713, 1234, 1235, 1466, 1634, 1984, 2909, 3140), and maintenance of normal prostate size and normal urination (ID 714, 1467, 1635) pursuant to Article 13 of Regulation (EC) No 1924/2006. EFSA J. 2010, 8, 1813. [Google Scholar]
- Ling, W.H.; Jones, P.J. Dietary phytosterols: A review of metabolism, benefits and side effects. Life Sci. 1995, 57, 195–206. [Google Scholar] [CrossRef]
- Lippi, G.; Avanzini, P.; Musa, R.; Sandei, F.; Aloe, R.; Cervellin, G. Evaluation of sample hemolysis in blood collected by S-Monovette® using vacuum or aspiration mode. Biochem. Med. (Zagreb) 2013, 23, 64–69. [Google Scholar] [CrossRef] [PubMed]
- Bowen, R.A.R.; Remaley, A.T. Interferences from blood collection tube components on clinical chemistry assays. Biochem. Med. (Zagreb) 2014, 24, 31–44. [Google Scholar] [CrossRef] [PubMed]
- Sarstedt. Blood Collection with the S-Monovette ®. Available online: https://www.sarstedt.com/fileadmin/user_upload/99_Gebrauchsanweisungen/Englisch_US_Code/644_c_PosterA3_AnleitungVenoeseBE_SafetyKanuele_GB_US_0314.pdf (accessed on 8 March 2018).
- Weale, M.E. Quality control for genome-wide association studies. Methods Mol. Biol. 2010, 628, 341–372. [Google Scholar] [PubMed]
- Ziegler, A. Genome-wide association studies: Quality control and population-based measures. Genet. Epidemiol. 2009, 33, S45–S50. [Google Scholar] [CrossRef] [PubMed]
- Vitagenes. Calidad Tecnica. Available online: https://www.vitagenes.com/Calidad/CalidadTecnica/ (accessed on 12 March 2018).
- Portao, J.; Bescós, R.; Irurtia, A.; Cacciatori, E.; Vallejo, L. Valoración de la grasa corporal en jóvenes físicamente activos: Antropometría vs. bioimpedancia. Nutr. Hosp. 2009, 24, 529–534. [Google Scholar] [PubMed]
- Rocha, M.; Banuls, C.; Bellod, L.; Jover, A.; Victor, V.M.; Hernandez-Mijares, A. A review on the role of phytosterols: New insights into cardiovascular risk. Curr. Pharm. Des. 2011, 17, 4061–4075. [Google Scholar] [CrossRef] [PubMed]
- Clifton, P.M.; Noakes, M.; Sullivan, D.; Erichsen, N.; Ross, D.; Annison, G.; Fassoulakis, A.; Cehun, M.; Nestel, P. Cholesterol-lowering effects of plant sterol esters differ in milk, yoghurt, bread and cereal. Eur. J. Clin. Nutr. 2004, 58, 503–509. [Google Scholar] [CrossRef] [PubMed]
- Katan, M.B.; Grundy, S.M.; Jones, P.; Law, M.; Miettinen, T.; Paoletti, R. Stresa Workshop Participants. Efficacy and safety of plant stanols and sterols in the management of blood cholesterol levels. Mayo Clin. Proc. 2003, 78, 965–978. [Google Scholar] [CrossRef]
- Abumweis, S.S.; Barake, R.; Jones, P.J. Plant sterols/stanols as cholesterol lowering agents: A meta-analysis of randomized controlled trials. Food Nutr. Res. 2008, 52. [Google Scholar] [CrossRef] [PubMed]
- Naumann, E.; Plat, J.; Kester, A.D.; Mensink, R.P. The baseline serum lipoprotein profile is related to plant stanol induced changes in serum lipoprotein cholesterol and triacylglycerol concentrations. J. Am. Coll. Nutr. 2008, 27, 117–126. [Google Scholar] [CrossRef] [PubMed]
- Maász, A.; Kisfali, P.; Szolnoki, Z.; Hadarits, F.; Melegh, B. Apolipoprotein A5 gene C56G variant confers risk for the development of large-vessel associated ischemic stroke. J. Neurol. 2008, 255, 649–654. [Google Scholar] [CrossRef] [PubMed]
- Zhou, J.; Xu, L.; Huang, R.S.; Huang, Y.; Le, Y.; Jiang, D.; Yang, X.; Xu, W.; Huang, X.; Dong, C.; et al. Apolipoprotein A5 gene variants and the risk of coronary heart disease: A case-control study and meta-analysis. Mol. Med. Rep. 2013, 8, 1175–1182. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Pi, Y.; Zhang, L.; Yang, Q.; Li, B.; Guo, L.; Fang, C.; Gao, C.; Wang, J.; Xiang, J.; Li, J. Apolipoprotein A5 gene promoter region -1131T/C polymorphism is associated with risk of ischemic stroke and elevated triglyceride levels: A meta-analysis. Cerebrovasc. Dis. 2012, 33, 558–565. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.Y.; Wu, X.Y.; Xu, J.; Qian, Y.; Zhou, C.W.; Wang, B. Apo A5 -1131T/C, FgB -455G/A, -148C/T, and CETP TaqIB gene polymorphisms and coronary artery disease in the Chinese population: A meta-analysis of 15,055 subjects. Mol. Biol. Rep. 2013, 40, 1997–2014. [Google Scholar] [CrossRef] [PubMed]
- Sánchez-Moreno, C.; Ordovás, J.M.; Smith, C.E.; Craza, J.C.; Lee, Y.; Garaulet, M. APOA5 gene variation interacts with dietary fat intake to modulate obesity and circulating triglycerides in a Mediterranean population. J. Nutr. 2011, 141, 380–385. [Google Scholar] [CrossRef] [PubMed]
- Zhang, M.D.; Gu, W.; Qiao, S.B.; Zhu, E.J.; Zhao, Q.M.; Ly, S.Z. Apolipoprotein E gene polymorphism and risk for coronary heart disease in the Chinese population: A meta-analysis of 61 studies including 6634 cases and 6393 controls. PLoS ONE 2014, 9, e95463. [Google Scholar] [CrossRef] [PubMed]
- Demirkan, A.; Van Duijn, C.M.; Ugocsai, P.; Isaacs, A.; Pramstaller, P.P.; Liebisch, G.; Wilson, J.F.; Johansson, Å.; Rudan, I.; Aulchenko, Y.S.; et al. Genome-wide association study identifies novel loci associated with circulating phospho- and sphingolipid concentrations. PLoS Genet. 2012, 8, e1002490. [Google Scholar] [CrossRef] [PubMed]
- Posadas-Sánchez, R.; Ocampo-Arcos, W.A.; López-Uribe, Á.R.; Posadas-Romero, C.; Villarreal-Molina, T.; León, E.Á.; Pérez-Hernández, N.; Rodríguez-Pérez, J.M.; Cardoso-Saldaña, G.; Medina-Urrutia, A.; et al. Hepatic lipase (LIPC) C-514T gene polymorphism is associated with cardiometabolic parameters and cardiovascular risk factors but not with fatty liver in Mexican population. Exp. Mol. Pathol. 2015, 98, 93–98. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.; Zhang, D.; Ling, J.; Lu, W.; Zhang, S.; Zhu, Y.; Lai, M. Gender specific effect of LIPC C-514T polymorphism on obesity and relationship with plasma lipid levels in Chinese children. J. Cell. Mol. Med. 2015, 19, 2296–2306. [Google Scholar] [CrossRef] [PubMed]
- Tai, E.S.; Corella, D.; Demissie, S.; Cupples, L.A.; Coltell, O.; Schaefer, E.J.; Tucker, K.L.; Ordovas, J.M.; Framingham Heart Study. Polyunsaturated fatty acids interact with the PPARA-L162V polymorphism to affect plasma triglyceride and apolipoprotein C-III concentrations in the Framingham Heart Study. J. Nutr. 2005, 135, 397–403. [Google Scholar] [CrossRef] [PubMed]
- Ordovas, J.M.; Corella, D.; Cupples, L.A.; Demissie, S.; Kelleher, A.; Coltell, O.; Wilson, P.W.; Schaefer, E.J.; Tucker, K. Polyunsaturated fatty acids modulate the effects of the APOA1 G-A polymorphism on HDL-cholesterol concentrations in a sex-specific manner: The Framingham Study. Am. J. Clin. Nutr. 2002, 75, 38–46. [Google Scholar] [CrossRef] [PubMed]
- Lai, C.Q.; Corella, D.; Demissie, S.; Cupples, L.A.; Adiconis, Y.; Zhu, Y.; Parnell, L.D.; Tucker, K.L.; Ordovas, J.M. Dietary intake of n-6 fatty acids modulates effect of apolipoprotein A5 gene on plasma fasting triglycerides, remnant lipoprotein concentrations, and lipoprotein particle size: The Framingham Heart Study. Circulation 2006, 113, 2062–2070. [Google Scholar] [CrossRef] [PubMed]
| Total (n = 45) | Males (n = 20) | Females (n = 25) | |
|---|---|---|---|
| Mean ± SD | Mean ± SD | Mean ± SD | |
| Age (years) | 37.9 ± 7.5 | 39.7 ± 6.9 | 36.5 ± 7.8 |
| Weight (kg) | 68.6 ± 12.4 | 77 ± 11.3 | 61.4 ± 8.03 |
| Height (m) | 1.7 ± 0.1 | 1.76 ± 0.1 | 1.64 ± 0.1 |
| BMI (Kg/m2) | 23.7 ± 3.1 | 24.8 ± 3.2 | 22.8 ± 2.7 |
| Body fat (%) | 25.5 ± 7.3 | 21.2 ± 6.5 | 29.4 ± 5.7 |
| Visceral fat (kg) | 6.8 ± 4.6 | 8 ± 4.7 | 5.7 ± 4.3 |
| Muscle (kg) | 35.5 ± 13.4 | 40.4 ± 14.1 | 31.2 ± 11.4 |
| Basal metabolic rate (kcal) | 1484.5 ± 278.6 | 1705 ± 230.1 | 1285 ± 125.3 |
| Decrease % (from baseline to final measures) | |||
| Total cholesterol (mg/dL) | 9.1 ± 9.3 | 8.2 ± 9.8 | 9.9 ± 9.3 |
| LDL-cholesterol (mg/dL) | 11.8 ± 13.04 | 9.9 ± 12.9 | 13.4 ± 13.8 |
| HDL-cholesterol (mg/dL) | 1.8 ± 8.1 | 2.1 ± 6.9 | 1.5 ± 9.3 |
| Non-HDL-cholesterol (mg/dL) | 11.5 ± 10.8 | 9.6 ± 11.7 | 13.2 ± 10.3 |
| Gene | Haplotype | Frequency (n) | Percentage (%) |
|---|---|---|---|
| APOA5 C56G Ser19Trp (rs3135506) | CG | 12 | 24.5 |
| GG | 37 | 75.5 | |
| Prothrombin G20210A (rs1799963) | GG | 49 | 100 |
| F5 Arg506Gln (rs6025) | GG | 49 | 100 |
| MTHFR C677T (rs1801133) | CC | 13 | 26.5 |
| CT | 31 | 63.3 | |
| TT | 5 | 10.2 | |
| LIPC C-514T (rs1800588) | CC | 19 | 38.8 |
| CT | 24 | 49 | |
| TT | 6 | 12.2 | |
| LPA I4300M (rs3798220) | TT | 47 | 95.9 |
| TC | 2 | 4.1 | |
| PPAR-alpha L162V (rs1800206) | CC | 43 | 87.8 |
| CG | 6 | 12.2 | |
| APOA5 1131T>C (rs662799) | TT | 46 | 93.9 |
| TC | 3 | 6.1 | |
| APOE Haplotipo APOE2/3/4 (rs429358) | TT | 42 | 85.7 |
| TC | 7 | 14.3 | |
| APOE Haplotipo APOE2,3,4 (rs7412) | TC | 7 | 14.3 |
| CC | 42 | 85.7 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
San Mauro Martín, I.; Blumenfeld Olivares, J.A.; Pérez Arruche, E.; Arce Delgado, E.; Ciudad Cabañas, M.J.; Garicano Vilar, E.; Collado Yurrita, L. Genomic Influence in the Prevention of Cardiovascular Diseases with a Sterol-Based Treatment. Diseases 2018, 6, 24. https://doi.org/10.3390/diseases6020024
San Mauro Martín I, Blumenfeld Olivares JA, Pérez Arruche E, Arce Delgado E, Ciudad Cabañas MJ, Garicano Vilar E, Collado Yurrita L. Genomic Influence in the Prevention of Cardiovascular Diseases with a Sterol-Based Treatment. Diseases. 2018; 6(2):24. https://doi.org/10.3390/diseases6020024
Chicago/Turabian StyleSan Mauro Martín, Ismael, Javier Andrés Blumenfeld Olivares, Eva Pérez Arruche, Esperanza Arce Delgado, María José Ciudad Cabañas, Elena Garicano Vilar, and Luis Collado Yurrita. 2018. "Genomic Influence in the Prevention of Cardiovascular Diseases with a Sterol-Based Treatment" Diseases 6, no. 2: 24. https://doi.org/10.3390/diseases6020024
APA StyleSan Mauro Martín, I., Blumenfeld Olivares, J. A., Pérez Arruche, E., Arce Delgado, E., Ciudad Cabañas, M. J., Garicano Vilar, E., & Collado Yurrita, L. (2018). Genomic Influence in the Prevention of Cardiovascular Diseases with a Sterol-Based Treatment. Diseases, 6(2), 24. https://doi.org/10.3390/diseases6020024





