The Cardiovascular Effects of Cocoa Polyphenols—An Overview
Abstract
:1. Introduction
2. Polyphenols Composition of Cocoa
- (i)
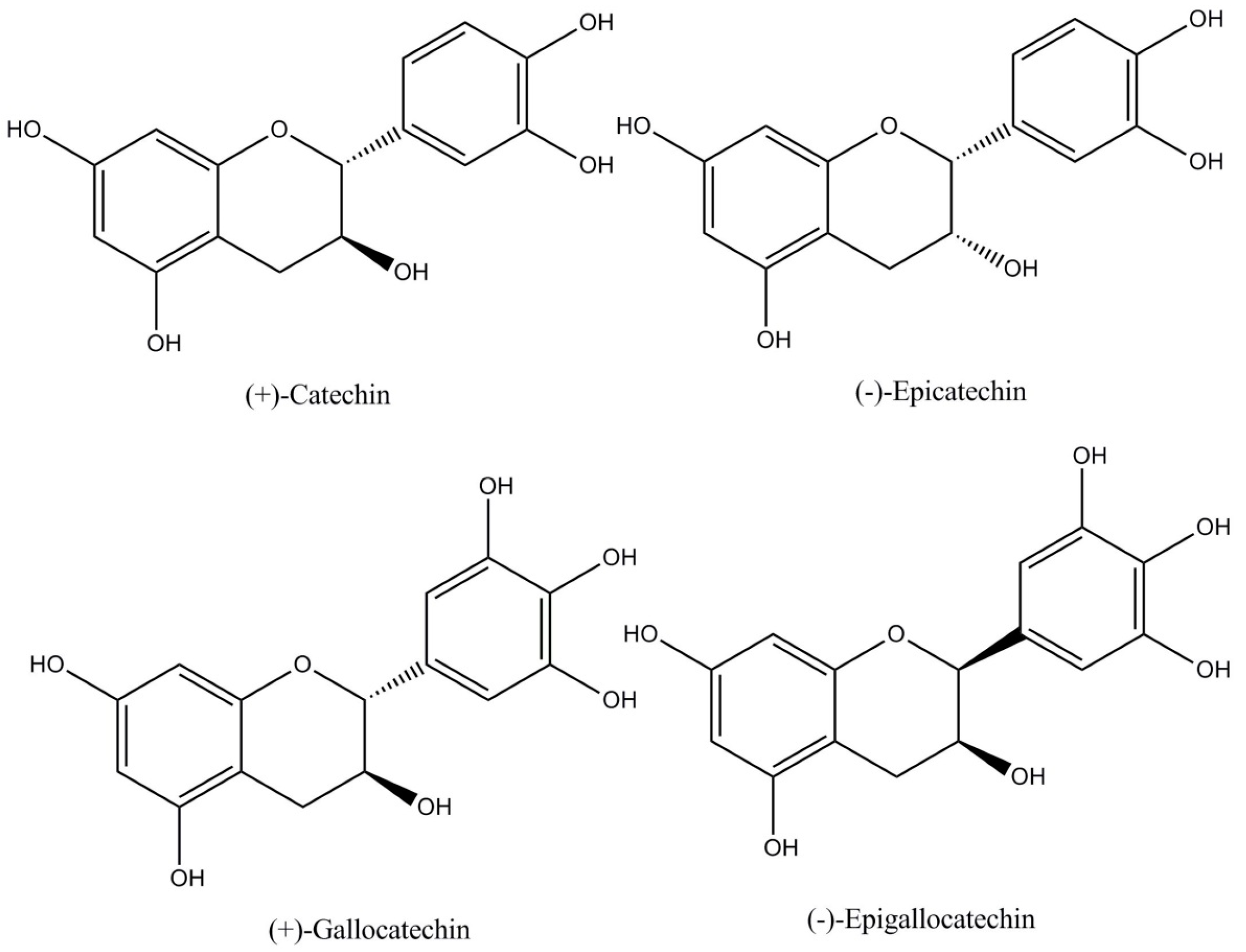
- Flavan-3-ols or catechins (29%–38% of total polyphenols) as monomers and polymers. The monomers include (−)-epicatechin, (+)-catechin, (+)-gallocatechin, and (−)-epigallocatechin (Figure 1). (−)-Epicatechin constitutes up to 35% of total polyphenols being the most abundant polyphenol both in cocoa and cocoa derived products (chocolate) [4,6]. Flavanols are commonly found in cocoa as R-forms [7].
- (ii)
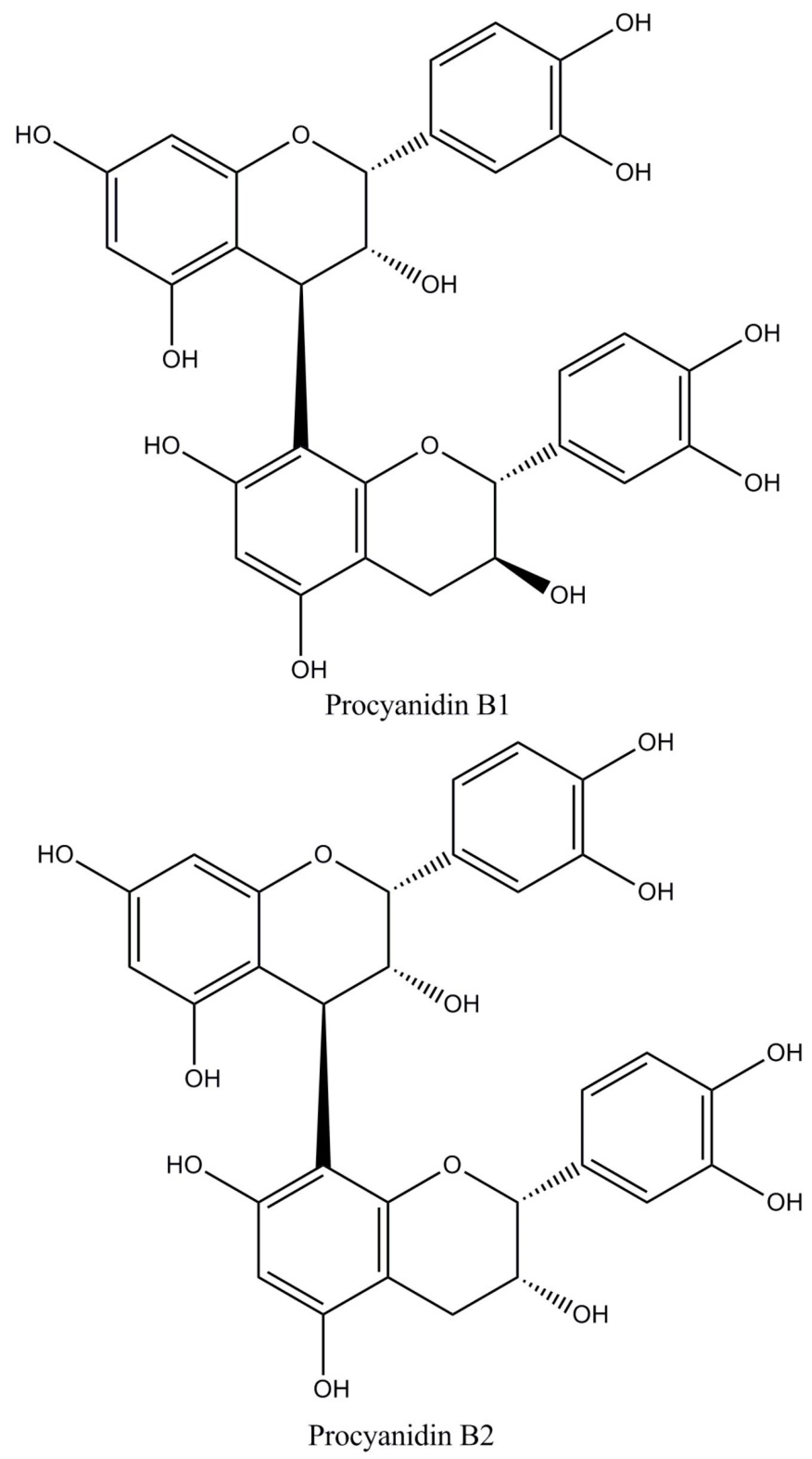
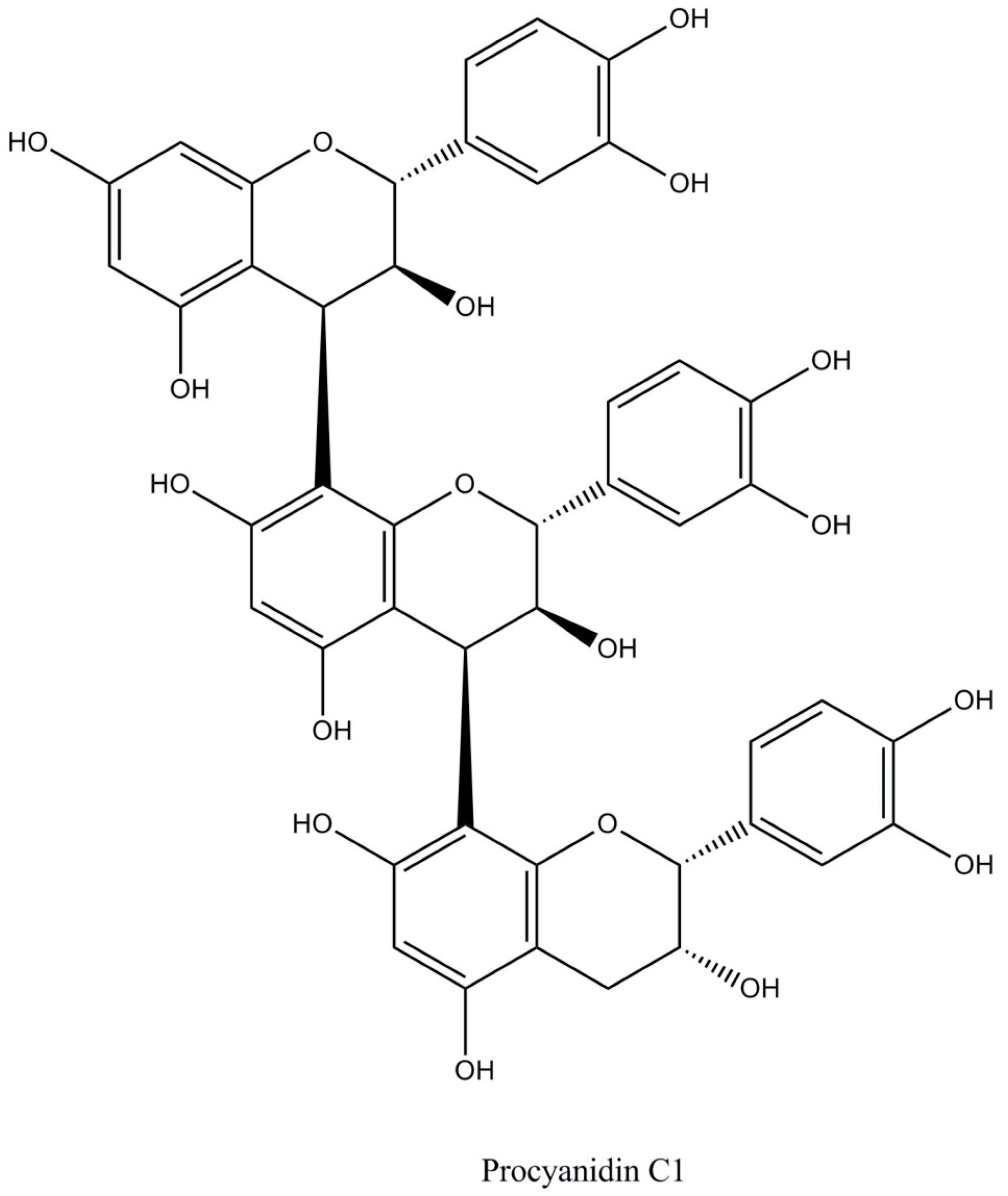
- Proanthocyanidins (58%–65% of total polyphenols) are polymers of epicatechin and catechin molecules linked by 4→8 and 4→6 bonds. In cocoa, mainly oligomeric proanthocyanidins (degree of polymerization = 10) are found, and dimers to hexamers predominate. The most important cocoa proathocyanidins are dimers: B1 [epicatechin-(4β→8)-catechin], B2 [epicatechin-(4β→8)-epicatechin] (Figure 2), B3 [catechin-(4α→8)-catechin], B4 [catechin-(4α→8)-epicatechin], B5 [epicatechin-(4β→6)-epicatechin], and trimers: C1 [epicatechin-(4β→8)2-epicatehin] (Figure 3) [4].
- (iii)
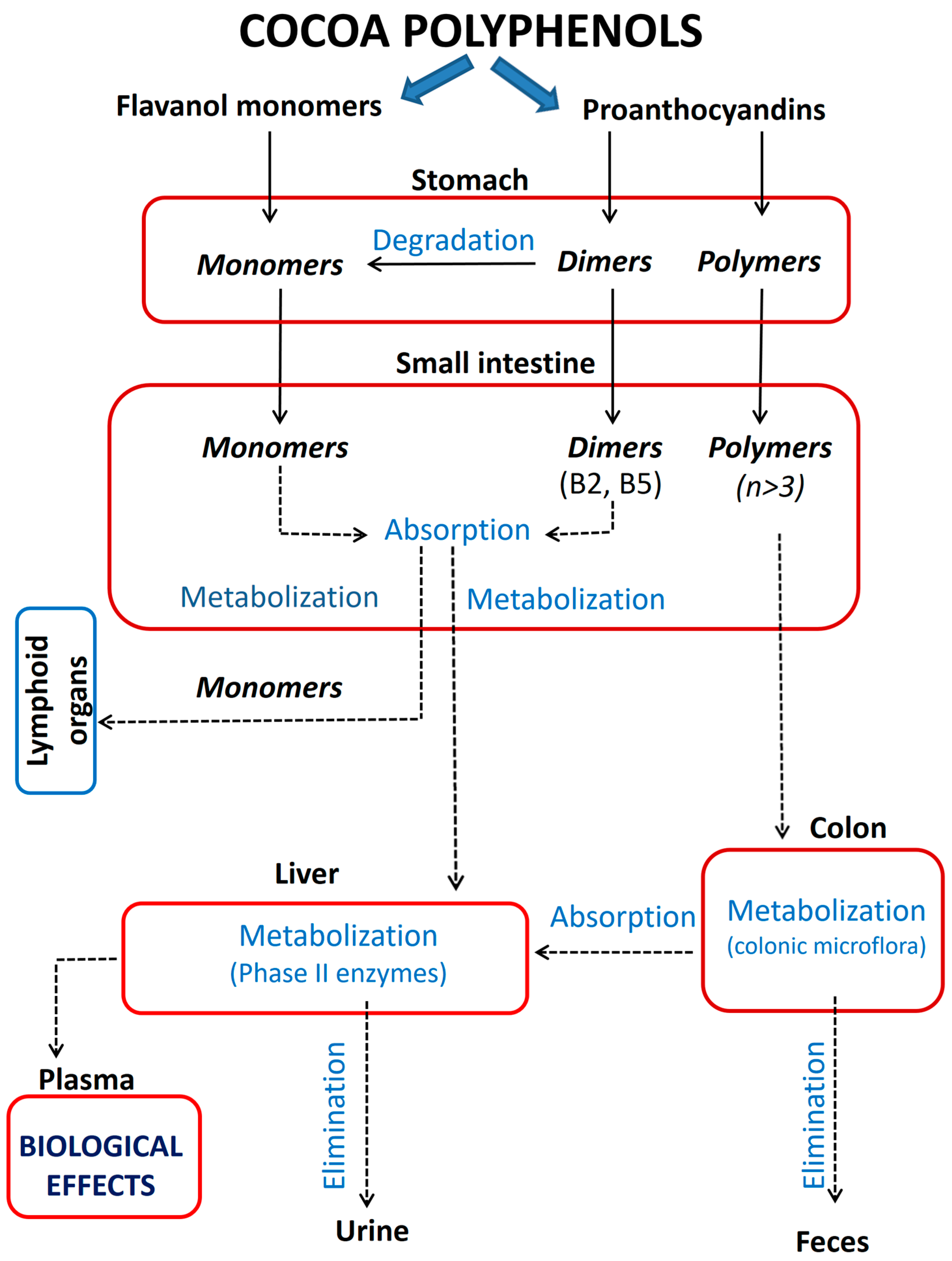
3. Bioavailability of Cocoa Polyphenols
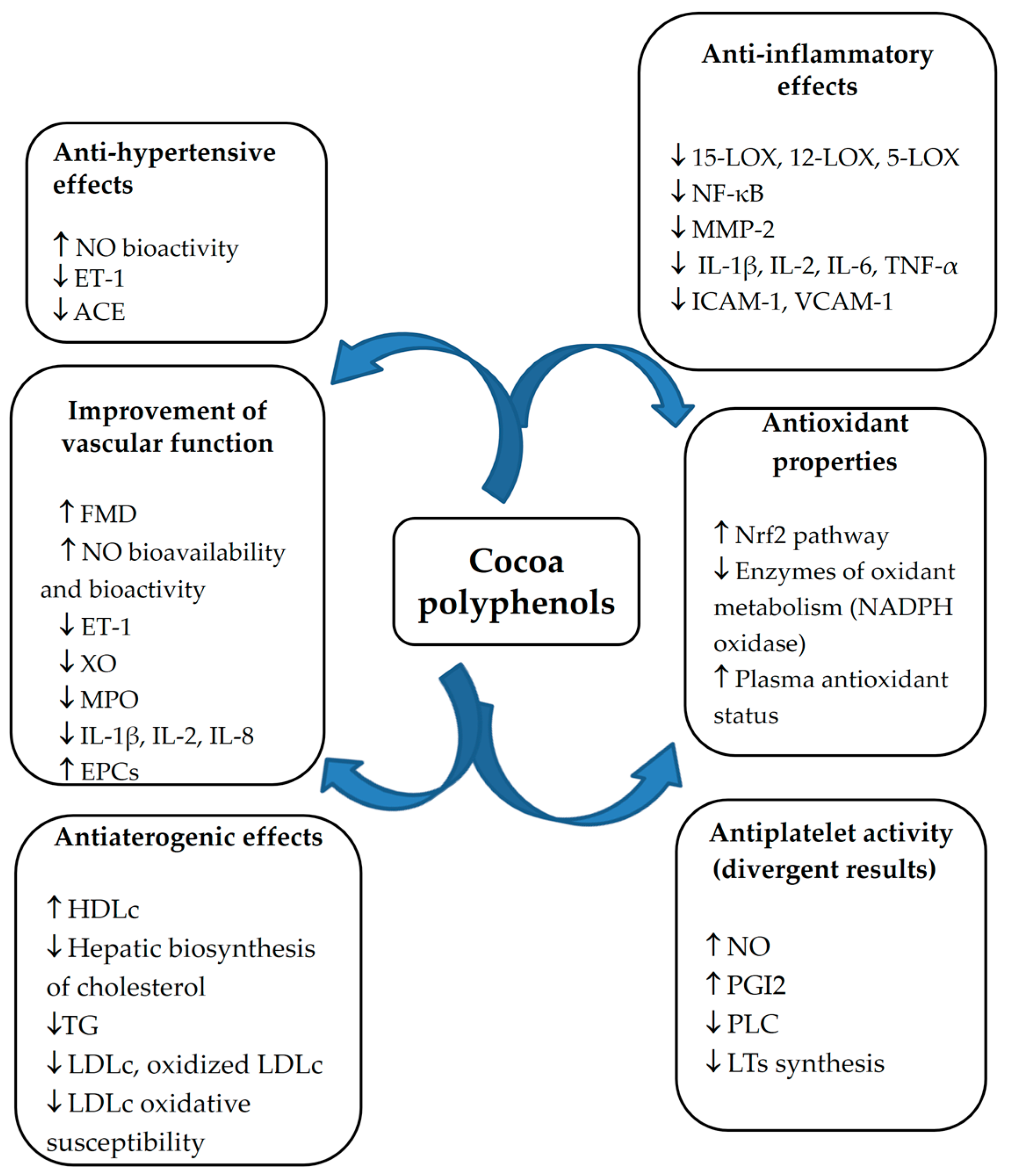
4. Cardioprotective Effects of Cocoa Polyphenols
4.1. Antioxidant Activity
4.2. Modulation of Endothelium-Dependent Vasomotor Function
4.3. Effects on Blood Pressure
4.4. Antiplatelet Effects
- (i)
- Health status/gender of participants. The different basal level of platelet function may result in different responses after the exposure to compounds with antiplatelet potential. In a randomized controlled human intervention trial, Ostertag et al. [70] showed gender-dependent antiplatelet effects of dark chocolate. Acute intake of flavan-3-ol-enriched dark chocolate (907.4 mg flavanols/60 g chocolate) significantly reduces ADP-induced platelet aggregation and P-selectin expression in men and decreases thrombin receptor-activating peptide (TRAP)-induced platelet aggregation in women. The platelets from men are more sensitive to activation via adrenergic and serotoninergic pathways and show more intense thromboxane A2 receptor-related aggregation responses. The women’s platelets have a lower amount of thromboxane A2 receptors, which would explain the inhibitory effect of flavanol-enriched dark chocolate on TRAP-induced aggregation. Also, the use of oral contraceptives and menstrual phase may influence the effects on platelet function. In a critical review on antiplatelet effects of dietary polyphenols, Ostertag et al. [69] noted that the most significant changes on platelet functions have been reported for subjects with single or multiple cardiovascular risk factors. Also, acute intake of dark chocolate (40 g, cocoa > 85%) reduces platelet activation via antioxidant mechanisms only in smokers who have a higher baseline generation of oxidative stress compared to healthy subjects [71];
- (ii)
- Acute or chronic intake. The antiplatelet effects of cocoa flavanols appear to be more intense and meaningful in the case of acute intake. Even a modest amount of flavanols may modulate platelet reactivity in acute studies. The metabolism of cocoa flavanols, bioactivity of metabolites and their persistence could contribute to these findings. The existence of possible different mechanisms for acute and chronic administration does not allow a direct comparison of results from such studies. A better assessment of cocoa flavanols antiplatelet activity should measure both acute and chronic effects in the same study [69];
- (iii)
- Methodology. The assessment of antiplatelet effects of cocoa polyphenols was performed by various experimental approaches that differ in terms of principle, sensitivity, evaluated markers or functions. Besides, the correlations between methods are low. Bleeding time (BT) assesses primary hemostasis by in vivo measurement of bleeding block. Although BT is a simple and quick method, it has the disadvantage that is poorly standardized and is influenced by many variables (skin thickness, temperature among patients). Light transmission platelet aggregometry on platelet-rich plasma (PRP)-LTA is a standard test that evaluates various platelet functions such as platelet activation under action of different agonists (ADP, AA, collagen, and epinephrine, TRAP, thromboxane A2 mimetic U46619) and platelet-to-platelet clump formation in a glycoprotein (GP) IIb/IIIa-dependent manner. The preanalytical conditions (type of anticoagulant, lipid plasma, hemolysis, or low platelet count) as well as procedural conditions (manual sample processing, PRP preparation, use of different concentrations of agonists) may alter the final outcomes [72]. Besides, LTA is a relative non-physiological method, and platelets are not subjected to intense shear conditions [69]. The Platelet Function Analyzer—PFA-100 assesses platelet function in whole blood at the point-of-care under shear stress using collagen (C) plus ADP or collagen plus epinephrine as stimulators of hemostasis. The method presents some limitations such as platelet count-hematocrit-dependence and insensitivity to platelet secretion defects [72]. Platelet analysis based on flow cytometry provides information on platelet functional status in vivo, and includes different methods such as the assessment of platelet activation biomarkers, leukocyte–platelet aggregates or platelet-derived microparticles. However, the preanalytical phase may induce errors, and the measurement of circulating monocyte–platelet is performed under low shear conditions that do not accurately reproduce in vivo processes [69,72];
- (iv)
- Small size of subject lots and different populations [73].
4.5. Modulation of Lipid Profile
4.6. Anti-Inflammatory Activity
4.7. Is (−)-Epicatechin the Main Compound Responsible for the Cardioprotective Properties of Cocoa Products?
- (i)
- Small number of subjects and statistical underpowered trials [87];
- (ii)
- Large heterogeneity of the study population (40–80 years) and biological variations among subjects [84];
- (iii)
- Dose of epicatechin. As we already noted, EFSA recommends 200 mg of cocoa polyphenols daily for a beneficial effect on endothelial function. Although, Dower et al. [83,84] chosed the dosage of epicatechin in line with the amount of epicatechin present in previous cocoa/chocolate intervention studies (46–107 mg/day); in those studies, the level of total polyphenols was significantly higher (more than 200 mg and even higher than 800 mg). In cocoa products, the effects of epicatechin can be boosted by the pharmacokinetic and pharmacological interactions with other cocoa flavonoids and compounds. The activity of a compound within the natural phytocomplexes may be different as intensity or even sense from that of pure compound. The interactions between compounds in the phytocomplex affect their solubility, bioavailability or bioactivity and lead to a nuanced expression of biological response. In fact, even the authors mentioned that the dose of 100 mg epicatechin is likely to be too low to exert an effect on NO metabolism, the main target of vasodilatory mechanisms. Moreover, using a nonlinear meta-regression model with a Bayesian approach, Ellinger et al. (2012) [88] showed that the dose of ingested epicatechin influences the mean treatment effect. In this respect, authors showed that the daily intake of 25 mg epicatechin via cocoa consumption (but not as pure compound) can reduce BP through an increased availability of NO. In the study of Schroeter et al. [87], the dosage of epicatechin can be even lower or higher than 100 mg, depending on the body weight of subjects. In this context, the occurrence of the effects at low doses is difficult to explain. Maybe the fact that the subjects are more age-homogenous (25–32 years) exerts a positive influence;
- (iv)
- Health status of subjects. Baseline values of cardiovascular status and metabolism are different for young adults (25–32 years) and older adults (over 50 years), the last category being included in the studies of Dower et al. [83,84]. The type of treatment with epicatechin (acute vs. acute-on-chronic effect) may influence the final outcome. Dower et al. [84] showed that it is possible to reach a plateau regarding the effect on the endothelial function after 4 weeks of epicatechin administration, and one additional acute dose could not elicit more effect on FMD. The present data do not allow to define the cardiovascular profile of epicatechin in humans. The topic remains in discussion, and further long-term well-designed studies, with a larger number of subjects and appropriate methodology, are needed to gain more insight into the cardioprotective potential of epicatechin.
5. Safety of Cocoa Polyphenols
6. Limitations of Cocoa Studies
7. Future Perspectives
8. Conclusions
Conflicts of Interest
References
- World Health Organization. Cardiovascular Diseases. Available online: http://www.who.int/mediacentre/factsheets/fs317/en/ (accessed on 10 July 2016).
- Field, D.; Newton, G. The antihypertensive actions of cocoa polyphenols—A review. Curr. Top. Nutraceuticals Res. 2013, 1, 113–128. [Google Scholar]
- World Health Organization. A Global Brief on Hypertension-Silent Killer, Global Public Health Crisis; WHO Press: Geneva, Switzerland, 2013; pp. 7–12. [Google Scholar]
- Aprotosoaie, A.C.; Luca, S.V.; Miron, A. Flavor chemistry of cocoa and cocoa products—An overview. Compr. Rev. Food Sci. Food Saf. 2016, 15, 73–91. [Google Scholar] [CrossRef]
- Jalil, A.M.M.; Ismail, A. Polyphenols in cocoa and cocoa products: Is there a link between antioxidant properties and health. Molecules 2008, 13, 2190–2219. [Google Scholar] [CrossRef] [PubMed]
- Fraga, C.G.; Litterio, M.C.; Prince, P.D.; Calabro, V.; Piotrkowski, B.; Galleano, M. Cocoa flavanols: Effects on vascular nitric oxide and blood pressure. J. Clin. Biochem. Nutr. 2010, 48, 63–67. [Google Scholar] [CrossRef] [PubMed]
- Keen, C.L. Chocolate: Food as medicine/medicine as food. J. Am. Coll. Nutr. 2001, 20, 436S–439S. [Google Scholar] [CrossRef] [PubMed]
- Rimbach, G.; Melchin, M.; Moehring, J.; Wagner, A.E. Polyphenols from cocoa and vascular health—A critical review. Int. J. Mol. Sci. 2009, 10, 4290–4309. [Google Scholar] [CrossRef] [PubMed]
- Sudano, I.; Flammer, A.J.; Noll, G.; Corti, R. Vascular and platelet effects of cocoa. In Chocolate and Health; Paoletti, R., Poli, A., Conti, A., Visioli, F., Eds.; Springer: Milan, Italy, 2012; pp. 103–111. [Google Scholar]
- Cooper, K.A.; Donovan, J.L.; Waterhouse, A.L.; Williamson, G. Cocoa and health: A decade of research. Br. J. Nutr. 2008, 99, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Schinella, G.; Mosca, S.; Cienfuegos-Jovellanos, E.; Pasamar, M.A.; Miguerza, B.; Ramón, D.; Rios, J.L. Antioxidant properties of polyphenol-rich cocoa products industrially processed. Food Res. Int. 2010, 43, 1614–1623. [Google Scholar] [CrossRef]
- Goya, L.; Martin, M.A.; Sarriá, B.; Ramos, S.; Mateos, R.; Bravo, L. Effect of cocoa and its flavonoids on biomarkers of inflammation: Studies of cell culture, animals and humans. Nutrients 2016, 8, 212. [Google Scholar] [CrossRef] [PubMed]
- Steffen, Y.; Gruber, C.; Schewe, T.; Sies, H. Mono-O-methylated flavanols and other flavonoids as inhibitors of endothelial NADPH oxidase. Arch. Biochem. Biophys. 2008, 469, 209–219. [Google Scholar] [CrossRef] [PubMed]
- Keen, C.L.; Holt, R.R.; Polagruto, J.A.; Wang, J.F.; Schmitz, H.H. Cocoa flavanols and cardiovascular health. Phytochem. Rev. 2002, 1, 231–240. [Google Scholar] [CrossRef]
- Khan, N.; Monagas, M.; Andres-Lacueva, C.; Casas, R.; Urpi-Sardà, M.; Lamuela-Raventos, R.M.; Estruch, R. Regular consumption of cocoa powder with milk increases HDL cholesterol and reduces oxidized LDL levels in subjects at high-risk of cardiovascular disease. Nutr. Metab. Cardiovasc. 2012, 22, 1046–1053. [Google Scholar] [CrossRef] [PubMed]
- Schroeter, H.; Heiss, C.; Spencer, J.P.E.; Keen, C.L.; Lupton, J.R.; Schmitz, H.H. Recommending flavanols and procyanidins for cardiovascular health: Current knowledge and future needs. Mol. Aspects Med. 2010, 31, 546–557. [Google Scholar] [CrossRef] [PubMed]
- Schewe, T.; Steffen, Y.; Sies, H. How do dietary flavanols improve vascular function? A position paper. Arch. Biochem. Biophys. 2008, 476, 102–106. [Google Scholar] [CrossRef] [PubMed]
- Heiss, C.; Jahn, S.; Taylor, M.; Real, W.M.; Angeli, F.S.; Wong, M.L.; Amabile, N.; Prasad, M.; Rassaf, T.; Ottaviani, J.I.; et al. Improvement of endothelial function with dietary flavanols is associated with mobilization of circulating angiogenic cells in patients with coronary artery diseases. J. Am. Coll. Cardiol. 2010, 56, 218–224. [Google Scholar] [CrossRef] [PubMed]
- De Palma, R.; Sotto, I.; Wood, E.G.; khan, N.Q.; Butler, J.; Johnston, A.; Rothman, M.T.; Corder, R. Cocoa flavanols reduce N-terminal pro-B-type natriuretic peptide in patients with chronic heart failure. ESC Heart Fail. 2016, 3, 97–106. [Google Scholar] [CrossRef] [PubMed]
- Dower, J.I.; Geleijnse, J.M.; Kroon, P.A.; Philo, M.; Mensink, M.; Kromhout, D.; Hollman, P.C.H. Does epicatechin contribute to the acute vascular function effects of dark chocolate? A randomized, crossover study. Mol. Nutr. Food Res. 2016, 60, 2379–2386. [Google Scholar] [CrossRef] [PubMed]
- Rassaf, T.; Rammos, C.; Hendgen-Cotta, U.B.; Heiss, C.; Kleophas, W.; Dellanna, F.; Floege, J.; Hetzel, G.R.; Kelm, M. Vasculoprotective effects of dietary cocoa flavanols in patients on hemodialysis: A double-blind, randomized, placebo-controlled trial. Clin. J. Am. Soc. Nephrol. 2016, 11, 108–118. [Google Scholar] [CrossRef] [PubMed]
- Balboa-Castillo, T.; López-García, E.; León-Muñoz, L.M.; Pérez-Tasigchana, R.F.; Banegas, J.R.; Rodríguez-Artaleji, F.; Guallar-Castillón, P. Chocolate and health-related quality of life: A prospective study. PLoS ONE 2015, 10, e0123161. [Google Scholar] [CrossRef] [PubMed]
- Basu, A.; Betts, N.M.; Leyva, M.J.; Fu, D.; Aston, C.E.; Lyons, T.J. Acute cocoa supplementation increases postprandial HDL cholesterol and insulin in obese adults with type 2 diabetes after consumption of a high-fat breakfast. J. Nutr. 2015, 145, 2325–2332. [Google Scholar] [CrossRef] [PubMed]
- Grassi, D.; Desideri, G.; Necozione, S.; di Giosia, P.; Barnabei, R.; Allegaert, L.; Bernaert, H.; Bernaert, H.; Ferri, C. Cocoa consumption dose-dependently improves flow-mediated dilation and arterial stiffness decreasing blood pressure in healthy individuals. J. Hypertens. 2015, 33, 294–303. [Google Scholar] [CrossRef] [PubMed]
- Hammer, A.; Koppensteiner, R.; Steiner, S.; Niessner, A.; Goliasch, G.; Gschwandtner, M.; Hoke, M. Dark chocolate and vascular function in patients with peripheral artery disease: A randomized, controlled cross-over trial. Clin. Hemorrheol. Microcirc. 2015, 59, 145–153. [Google Scholar]
- Heiss, C.; Sansone, R.; Karimi, H.; Krabbe, M.; Schuler, D.; Rodriguez-Mateos, A.; Kraemer, T.; Cortese-Krott, M.M.; Kuhnle, G.G.; Spencer, J.P.; et al. Impact of cocoa flavanol intake on age-dependent vascular stiffness in healthy men: A randomized, controlled, double-masked trial. Age 2015, 37, 9794. [Google Scholar] [CrossRef] [PubMed]
- De Jesús Romero-Prado, M.M.; Curiel-Beltrán, J.A.; Miramontes-Espino, M.V.; Cardona-Muñoz, E.G.; Rios-Arellano, A.; Balam-Salazar, L.B. Dietary flavonoids added to pharmacological antihypertensive therapy are effective in improving blood pressure. Basic Clin. Pharmacol. Toxicol. 2015, 117, 57–64. [Google Scholar] [CrossRef] [PubMed]
- Koli, R.; Köhler, K.; Tonteri, E.; Peltonen, J.; Tikkanen, H.; Fogelholm, M. Dark chocolate and reduced snack consumption in midly hypertensive adults: An intervention study. Nutr. J. 2015, 14, 84. [Google Scholar] [CrossRef] [PubMed]
- Massee, L.A.; Ried, K.; Pase, M.; Travica, N.; Yoganathan, J.; Scholey, A.; Macpherson, H.; Kennedy, G.; Sali, A.; Pipingas, A. The acute and sub-chronic effects of cocoa flavanols on mood, cognitive and cardiovascular health in young healthy adults: A randomized, controlled trial. Front. Pharmacol. 2015, 6, 93. [Google Scholar] [CrossRef] [PubMed]
- McFarlin, B.K.; Venable, A.S.; Henning, A.L.; Prado, E.A.; Sampson, J.N.B.; Vingren, J.L.; Hill, D.W. Natural cocoa consumption: Potential to reduce atherogenic factors? J. Nutr. Biochem. 2015, 26, 626–632. [Google Scholar] [CrossRef] [PubMed]
- Ottaviani, J.I.; Balz, M.; Kimball, J.; Ensunsa, J.L.; Fong, R.; Momma, T.Y.; Kwik-Uribe, C.; Schroeter, H.; Keen, C.L. Safety and efficacy of cocoa flavanol intake in healthy adults: A randomized, controlled, double-masked trial. Am. J. Clin. Nutr. 2015, 102, 1425–1435. [Google Scholar] [CrossRef] [PubMed]
- Rodriguez-Mateos, A.; Hezel, M.; Aydin, H.; Kelm, M.; Lundberg, J.O.; Weitzberg, E.; Spencer, J.P.; Heiss, C. Interactions between cocoa flavanols and inorganic nitrate: Additive effects on endothelial function at achievable dietary amounts. Free Radic. Biol. Med. 2015, 80, 121–128. [Google Scholar] [CrossRef] [PubMed]
- Rostami, A.; Khalili, M.; Haghighat, N.; Eghtesadi, S.; Shidfar, F.; Heidari, I.; Ebrahimpour-Koujan, S.; Eghtesadi, M. High-cocoa polyphenol-rich chocolate improves blood pressure in patients with diabetes and hypertension. ARYA Atheroscler. 2015, 11, 21–29. [Google Scholar] [PubMed]
- Rull, G.; Mohd-Zain, Z.N.; Shiel, J.; Lundberg, M.H.; Collier, D.J.; Johnston, A.; Warner, T.D.; Corder, R. Effects of high flavanol dark chocolate on cardiovascular function and platelet aggregation. Vascul. Pharmacol. 2015, 71, 70–78. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sansone, R.; Rodriguez-Mateos, A.; Heuel, J.; Falk, D.; Schuler, D.; Wagstaff, R.; Kuhnle, G.G.C.; Spencer, J.P.; Schroeter, H.; Merx, M.W.; et al. Cocoa flavanol intake improves endothelial function and Framingham Risk Score in healthy men and women: A randomised, controlled, double-masked trial: The Flaviola Health Study. Br. J. Nutr. 2015, 114, 1246–1255. [Google Scholar] [CrossRef] [PubMed]
- Sarriá, B.; Martínez-López, S.; Sierra-Cinos, J.L.; Garcia-Diz, L.; Mateos, R.; Bravo, L. Regular consumption of a cocoa product improves the cardiometabolic profile in healthy and moderately hypercholesterolaemic adults. Br. J. Nutr. 2014, 111, 122–134. [Google Scholar] [CrossRef] [PubMed]
- Wiese, S.; Esatbeyoglu, T.; Winterhalter, P.; Kruse, H.P.; Winkler, S.; Bub, A.; Kulling, S.E. Comparative biokinetics and metabolism of pure monomeric, dimeric, and polymeric flavan-3-ols: A randomized cross-over study in humans. Mol. Nutr. Food Res. 2015, 59, 610–621. [Google Scholar] [CrossRef] [PubMed]
- Horn, P.; Amabile, N.; Angeli, F.S.; Sansone, R.; Stegemann, B.; Kelm, M.; Springer, M.L.; Yeghiazarianis, Y.; Schroeter, H.; Heiss, C. Dietary flavanol intervention lowers the levels of endothelial microparticles in coronary artery disease patients. Br. J. Nutr. 2014, 111, 1245–1252. [Google Scholar] [CrossRef] [PubMed]
- Ibero-Baraibar, I.; Abete, I.; Navas-Carretero, S.; Massias-Zaid, A.; Martinez, J.A.; Zulet, M.A. Oxidised LDL levels decreases after the consumption of ready-to-eat meals supplemented with cocoa extract within a hypocaloric diet. Nutr. Metab. Cardiovasc. Dis. 2014, 24, 416–422. [Google Scholar] [CrossRef] [PubMed]
- Martínez-López, S.; Sarriá, B.; Sierra-Cinos, J.L.; Goya, L.; Mateos, R.; Bravo, L. Realistic intake of a flavanol-rich soluble cocoa produt increases HDL-cholesterol without inducing anthropometric changes in healthy and moderately hypercholesterolemic subjects. Food Funct. 2014, 5, 364–374. [Google Scholar] [CrossRef] [PubMed]
- Mastroiacovo, D.; Kwik-Uribe, C.; Grassi, D.; Necozione, S.; Raffaele, A.; Pistacchio, L.; Righetti, R.; Bocale, R.; Lechiara, M.C.; Marini, C.; et al. Cocoa flavanol consumption improve cognitive function, blood pressure control, and metabolic profile in elderly subjects: The Cocoa, Cognition, and Aging (CoCoA) Study—A randomized controlled trial. Am. J. Clin. Nutr. 2015, 101, 538–548. [Google Scholar] [CrossRef] [PubMed]
- Sarriá, B.; Martínez-López, S.; Sierra-Cinos, J.L.; Garcia-Diz, L.; Goya, L.; Mateos, R.; Bravo, L. Effects of bioactive constituents in functional cocoa products on cardiovascular health in humans. Food Chem. 2015, 174, 214–218. [Google Scholar] [CrossRef] [PubMed]
- Terai, N.; Gedenk, A.; Spoerl, E.; Pillunat, L.E.; Stodtmeister, R. The short-term effect of flavonoid-rich dark chocolate on retinal vessel diameter in glaucoma patients and age-matched controls. Acta Ophthalmol. 2014, 92, e341–e345. [Google Scholar] [CrossRef] [PubMed]
- West, S.G.; McIntyre, M.D.; Piotrowski, M.J.; Poupin, N.; Miller, D.I.; Preston, A.G.; Wagner, P.; Groves, L.F.; Skulas-Ray, A.C. Effects of dark chocolate and cocoa consumption on endothelial function and arterial stiffness in overweight adults. Br. J. Nutr. 2014, 111, 653–661. [Google Scholar] [CrossRef] [PubMed]
- Franzini, L.; Ardigò, D.; Valtueňa, S.; Pellegrini, N.; del Rio, D.; Bianchi, M.A.; Scazzina, F.; Piatti, P.M.; Brighenti, F.; Zavaroni, I. Food selection based on high total antioxidant capacity improves endothelial function in a low cardiovascular risk population. Nutr. Metab. Cardiovasc. 2012, 22, 50–57. [Google Scholar] [CrossRef] [PubMed]
- Scapagnini, G.; Davinelli, S.; di Renzo, L.; de Lorenzo, A.; Olarte, H.H.; Micali, G.; Cicero, A.F.; Gonzalez, S. Cocoa Bioactive Compounds: Significance and Potential for the Maintenance of Skin Health. Nutrients 2014, 6, 3202–3213. [Google Scholar] [CrossRef] [PubMed]
- Habauzit, V.; Morand, C. Evidence for a protective effects of polyphenols-containing foods on cardiovascular health: An update for clinicians. Ther. Adv. Chronic Dis. 2012, 3, 87–106. [Google Scholar] [CrossRef] [PubMed]
- Galleano, M.; Oteiza, P.I.; Fraga, C. Cocoa, chocolate, and cardiovascular disease. J. Cardiovasc. Pharmacol. 2009, 54, 483–490. [Google Scholar] [CrossRef] [PubMed]
- Monahan, K.D. Effect of cocoa/chocolate ingestion on brachial artery flow-mediated dilation and its relevance to cardiovascular health and disease in humans. Arch. Biochem. Biophys. 2012, 527, 90–94. [Google Scholar] [CrossRef] [PubMed]
- Campia, U.; Panza, J.A. Flavanol-rich cocoa—A promising new dietary intervention to reduce cardiovascular risk in type 2 diabetes? J. Am. Coll. Cardiol. 2008, 51, 2150–2152. [Google Scholar] [CrossRef] [PubMed]
- Heiss, C.; Kleinbongard, P.; Dejam, A.; Perré, S.; Schroeter, H.; Sies, H.; Kelm, M. Acute consumption of flavanol-rich cocoa and the reversal on endothelial dysfunction in smokers. J. Am. Coll. Cardiol. 2005, 46, 1276–1283. [Google Scholar] [CrossRef] [PubMed]
- Kerimi, A.; Williamson, G. The cardiovascular benefits of dark chocolate. Vascul. Pharmacol. 2015, 71, 11–15. [Google Scholar] [CrossRef] [PubMed]
- Liu, H.-B.; Gong, Y.-F.; Yu, C.-J.; Sun, Y.-Y.; Li, X.-Y.; Zhao, D.; Zhang, Z.-R. Endothelial progenitor cells in cardiovascular diseases: From biomarker to therapeutic agent. Regen. Med. Res. 2013, 1, 9. [Google Scholar] [CrossRef] [PubMed]
- Alexandru, N.; Badila, E.; Georgescu, A. The Role of Endothelial Progenitor Cells in the Cardiovascular Disease Pathogenesis. J. Stem Cells Res. Rev. Rep. 2014, 1, 1006. [Google Scholar]
- Balaji, S.; King, A.; Crombleholme, T.M.; Keswani, S.G. The role of endothelial progenitor cells in postnatal vasculogenesis: Implications for therapeutic neovascularization and wound healing. Adv. Wound Care 2013, 2, 283–295. [Google Scholar] [CrossRef] [PubMed]
- Grisar, J.C.; Haddad, F.; Gomari, F.A.; Wu, J.C. Endothelial progenitor cells in cardiovascular disease and chronic inflammation: From biomarker to therapeutic agent. Biomark. Med. 2011, 5, 731–744. [Google Scholar] [CrossRef] [PubMed]
- European Food Safety Authority (EFSA). Scientific opinion on the substantiation of a health claim related to cocoa flavanols and maintenance of normal endothelium-dependent vasodilatation pursuant to Article 13 (5) of Regulation (EC) No 1924/20061. EFSA J. 2012, 10, 2809–2830. [Google Scholar]
- Grassi, D.; Desideri, G.; Ferri, C. Blood pressure and cardiovascular risk: What about cocoa and chocolate? Arch. Biochem. Biophys. 2010, 501, 112–115. [Google Scholar] [CrossRef] [PubMed]
- Cienfuegos-Jovellanos, E.; del Mar Quiñones, M.; Muguerza, B.; Moulay, L.; Miguel, M.; Aleixandre, A. Antihypertensive effect of a polyphenol-rich cocoa powder industrially processed to preserve the original flavonoids of the cocoa beans. J. Agric. Food Chem. 2009, 57, 6156–6162. [Google Scholar] [CrossRef] [PubMed]
- Yamazaki, K.G.; Taub, P.R.; Barraza-Hidalgo, M.; Rivas, M.M.; Zambon, A.C.; Ceballos, G.; Villarreal, F.J. Effects of (−)-epicatechin on myocardial infarct size and left ventricular remodeling after permanent coronary occlusion. J. Am. Coll. Cardiol. 2010, 55, 2869–2876. [Google Scholar] [CrossRef] [PubMed]
- Ried, K.; Sullivan, T.R.; Fakler, P.; Frank, O.R.; Stocks, N.P. Effect of cocoa on blood pressure. Cochrane Database Syst. Rev. 2012, 15, CD008893. [Google Scholar]
- Desch, S.; Schmidt, J.; Kobler, D.; Sonnabend, M.; Eitel, I.; Sareban, M.; Rahimi, K.; Schuler, G.; Thiele, H. Effect of cocoa products on blood pressure: Systematic review and meta-analysis. Am. J. Hypertens. 2010, 23, 97–103. [Google Scholar] [CrossRef] [PubMed]
- Taubert, D.; Roesen, R.; Schomig, E. Effect of cocoa and tea intake on blood pressure: A meta-analysis. Arch. Intern. Med. 2007, 167, 626–634. [Google Scholar] [CrossRef] [PubMed]
- Grassi, D.; Necozione, S.; Lippi, C.; Croce, G.; Valeri, L.; Pasqualetti, P.; Desideri, G.; Blumberg, J.B.; Ferri, C. Cocoa reduces blood pressure and insulin resistance and improves endothelium-dependent vasodilation in hypertensives. Hypertension 2005, 46, 398–405. [Google Scholar] [CrossRef] [PubMed]
- Desideri, G.; Kwik-Uribe, C.; Grassi, D.; Necozione, S.; Ghiadoni, L.; Mastroiacovo, D.; Raffaele, A.; Ferri, L.; Bocale, R.; Lechiara, M.C.; et al. Benefits in cognitive function, blood pressure, and insulin resistance through cocoa flavanol consumption in elderly subjects with mild cognitive impairment: The Cocoa, Cognition, and Aging (CoCoA) study. Hypertension 2012, 60, 794–801. [Google Scholar] [CrossRef] [PubMed]
- Hooper, L.; Kroon, P.A.; Rimm, E.B.; Cohn, J.S.; Harvey, I.; le Cornu, K.A.; Ryder, J.J.; Hall, W.L.; Cassidy, A. Flavonoids, flavonoid-rich foods and cardiovascular risk: A meta-analysis of randomized controlled trials. Am. J. Clin. Nutr. 2008, 88, 38–50. [Google Scholar] [PubMed]
- Vlachopoulos, C.V.; Alexopoulos, N.A.; Aznaouridis, K.A.; Ioakeimidis, N.C.; Dima, I.A.; Dagre, A.; Vasiliadou, C.; Stefanadi, E.C.; Stefanadis, C.I. Relation of habitual cocoa consumption to aortic stiffness and wave reflections, and to central hemodynamics in healthy individuals. Am. J. Cardiol. 2007, 99, 1473–1475. [Google Scholar] [CrossRef] [PubMed]
- Arranz, S.; Valdera-Martinez, P.; Chiva-Blanch, G.; Casas, R.; Urpi-Sarda, M.; Lamuela-Raventos, R.M.; Estruch, R. Cardioprotective effects of cocoa: Clinical evidence from randomized clinical intervention trials in humans. Mol. Nutr. Food Res. 2013, 57, 936–947. [Google Scholar] [CrossRef] [PubMed]
- Ostertag, L.M.; O’Kennedy, N.; Kroon, P.A.; Duthie, G.G.; de Roos, B. Impact of dietary polyphenols on human platelet function—A critical review of controlled dietary intervention studies. Mol. Nutr. Food Res. 2010, 54, 60–81. [Google Scholar] [CrossRef] [PubMed]
- Ostertag, L.M.; Kroon, P.A.; Wood, S.; Horgan, G.H.; Cienfuegos-Jovellanos, E.; Saha, S.; Duthie, G.G.; de Roos, B. Flavan-3-ol-enriched dark chocolate and white chocolate improve acute measures of platelet function in a gender-specific way—A randomized-controlled human intervention trial. Mol. Nutr. Food Res. 2013, 57, 191–202. [Google Scholar] [CrossRef] [PubMed]
- Carnevale, R.; Loffredo, L.; Pignatelli, P.; Nocella, C.; Bartimoccia, S.; di Santo, S.; Martino, F.; Catasca, E.; Perri, L.; Violi, F. Dark chocolate inhibits platelet isoprostanes via NOX2 down-regulation in smokers. J. Thromb. Haemost. 2012, 10, 125–132. [Google Scholar] [CrossRef] [PubMed]
- Paniccia, R.; Priora, R.; Liotta, A.A.; Abbate, R. Platelet function tests: A comparative review. Vasc. Health Risk Manag. 2015, 11, 133–148. [Google Scholar] [CrossRef] [PubMed]
- Loffredo, L.; Perri, L.; Nocella, C.; Violi, F. Antioxidant and antiplatelet activity by polyphenol-rich nutrients: Focus on extra virgin olive and cocoa. Br. J. Clin. Pharmacol. 2016. [Google Scholar] [CrossRef] [PubMed]
- Quiñones, M.; Miguel, M.; Aleixandre, A. Beneficial effects of polyphenols on cardiovasculare disease. Pharmacol. Res. 2013, 68, 125–131. [Google Scholar] [CrossRef] [PubMed]
- Di Castelnuovo, A.; di Giuseppe, R.; Iacoviello, L.; de Gaetano, G. Consumption of cocoa, tea and coffee and risk of cardiovascular disease. Eur. J. Intern. Med. 2012, 23, 15–25. [Google Scholar] [CrossRef] [PubMed]
- Holt, R.R.; Actis-Goretta, L.; Momma, T.Y.; Keen, C.L. Dietary flavanols and platelet reactivity. J. Cardiovasc. Pharmacol. 2006, 47, S187–S193. [Google Scholar] [CrossRef] [PubMed]
- Tokede, O.A.; Gaziano, J.M.; Djousse, L. Effects of cocoa products/dark chocolate on serum lipids: A meta-analysis. Eur. J. Clin. Nutr. 2011, 65, 879–886. [Google Scholar] [CrossRef] [PubMed]
- Lee, K.W.; Kang, N.J.; Oak, M.-H.; Hwang, M.K.; Kim, J.H.; Schini-Kerth, V.B.; Lee, H.J. Cocoa procyanidins inhibit expression and activation of MMP-2 in vascular smooth muscle cells by direct inhibition of MEK and MT1-MMP activities. Cardiovasc. Res. 2008, 79, 34–41. [Google Scholar] [CrossRef] [PubMed]
- Paramo, J.A. A new favourable effect of cocoa on atherosclerosis? Cardiovasc. Res. 2008, 79, 3–4. [Google Scholar] [CrossRef] [PubMed]
- Jiménez, R.; Duarte, J.; Perez-Vizcaino, F. Epicatechin: Endothelial function and blood pressure. J. Agric. Food Chem. 2012, 60, 8823–8830. [Google Scholar] [CrossRef] [PubMed]
- Gutiérrez-Salméan, G.; Meaney, E.; Lanaspa, M.A.; Cicerchi, C.; Johnson, R.J.; Dugar, S.; Taub, P.; Ramírez-Sánchez, I.; Villarreal, F. A randomized, placebo-controlled, double-blind study on the effects of (−)-epicatechin on the triglyceride/HDLc ratio and cardiometabolic profile of subjects with hypertriglyceridemia: Unique in vitro effects. Int. J. Cardiol. 2016, 223, 500–506. [Google Scholar] [CrossRef] [PubMed]
- Barnett, C.F.; Moreno-Ulloa, A.; Shiva, S.; Ramirez-Sanchez, I.; Taub, P.R.; Su, Y.; Ceballos, G.; Dugar, S.; Schreiner, G.; Villarreal, F. Pharmacokinetic, partial pharmacodynamics and initial safety analysis of (−)-epicatechin in healthy volunteers. Food Funct. 2015, 6, 824–833. [Google Scholar] [CrossRef] [PubMed]
- Dower, J.I.; Geleijnse, J.M.; Gijsbers, L.; Schalkwijk, C.; Kromhout, D.; Hollman, P.C. Supplementation of the pure flavonoids epicatechin and quercetin effects some biomarkers of endothelial dysfunction and inflammation in (pre)hypertensive adults: A randomized double-blind, placebo-controlled, crossover trial. J. Nutr. 2015, 145, 1459–1463. [Google Scholar] [CrossRef] [PubMed]
- Dower, J.I.; Geleijnse, J.M.; Gijsbers, L.; Zock, P.L.; Kromhout, D.; Hollman, P.C.H. Effects of the pure flavonoids epicatechin and quercetin on vascular function and cardiometabolic health: A randomized, double-blind, placebo-controlled, crossover trial. Am. J. Clin. Nutr. 2015, 101, 914–921. [Google Scholar] [CrossRef] [PubMed]
- Gutiérrez-Salméan, G.; Ortiz-Vilchis, P.; Vacaseydel, C.M.; Rubio-Gayosso, I.; Meaney, E.; Villareal, F.; Ramírez-Sánchez, I.; Ceballos, G. Acute effects of an oral supplement of (−)-epicatechin on postprandial fat and carbohydrate metabolism in normal and overweight subjects. Food Funct. 2014, 5, 521–527. [Google Scholar]
- Loke, W.M.; Hodgson, J.M.; Proudfoot, J.M.; McKinley, A.J.; Puddey, I.B.; Croft, K.D. Pure dietary flavonoids quercetin and (−)-epicatechin augment nitric oxide products and reduce endothelin-1 acutely in healthy men. Am. J. Clin. Nutr. 2008, 88, 1018–1025. [Google Scholar] [PubMed]
- Schroeter, H.; Heiss, C.; Balzer, J.; Kleinbongard, P.; Keen, C.L.; Hollenberg, N.K.; Sies, H.; Kwik-Uribe, C.; Schmitz, H.H.; Kelm, M. (−)-Epicatechin mediates beneficial effects of flavanol-rich cocoa on vascular function in humans. Proc. Natl. Acad. Sci. USA 2006, 103, 1024–1029. [Google Scholar] [CrossRef] [PubMed]
- Ellinger, S.; Reusch, A.; Stehle, P.; Helfrich, H.-P. Epicatechin ingested via cocoa products reduces blood pressure in humans: A nonlinear regression model with a Bayesian approach. Am. J. Clin. Nutr. 2012, 95, 1365–1377. [Google Scholar] [CrossRef] [PubMed]
- Braun, L.; Cohen, M. Herbs and Natural Supplements, an Evidence-Based Guide, 4th ed.; Elsevier Churchill Livingstone: Sydney, Australia, 2015; Volume 2, pp. 202–214. [Google Scholar]
| Year | Study Design | No. of Participants | Aprox. Mean Age (years) | Sex | Comorbidities | Cocoa Product Intervention | TF (mg/day) | EC (mg/day) | Duration (weeks) | Outcomes | Ref. |
|---|---|---|---|---|---|---|---|---|---|---|---|
| 2016 | Randomized, double-blind, placebo-controlled, crossover | 32 | 70 | M + F | Chronic heart failure (stable on GDMT) | Dark chocolate | 1064 (HFDC); 88 (LFDC) | ns | 4 |  Plasma NT-proBNP (HFDC) vs. LFDC and baseline; Plasma NT-proBNP (HFDC) vs. LFDC and baseline; DBP vs. LFDC and baseline; DBP vs. LFDC and baseline;♦ No change in platelet function. | De Palma et al. [19] |
| 2016 | Randomized, placebo controlled crossover | 20 | 62 | M | None | Dark chocolate | 770 | 150 | 1 dose |  FMD vs. control; FMD vs. control; AIx vs. control. AIx vs. control. | Dower et al. [20] |
| 2016 | Randomized, placebo-controlled, double-blind | 57 | 65 | M + F | End-stage renal disease with chronic hemodialysis (pharmacological medication) | Cocoa powder | 900 | ns | 4 |  FMD vs. control; FMD vs. control; DBP vs. control. DBP vs. control. | Rassaf et al. [21] |
| 2015 | ENRICA cohort study | 1272 | 18–65 and over | M + F | Coronary heart disease, Hypertension, Diabetes, Hypercholesterolemia, Stroke | Chocolate | ns | ns | 3 years | No evidence of beneficial effects of chocolate regular consumption on physical or mental components of Health-related Quality of Life | Balboa-Castillo et al. [22] |
| 2015 | Randomized, double-blind, crossover, postprandial study | 18 | ns | M + F | Type 2 diabetes, Obese | Cocoa beverage | 480 | 40 | 1 dose |  HDLc vs. placebo; HDLc vs. placebo; Serum insulin concentration vs. placebo; Serum insulin concentration vs. placebo; Large artery elasticity vs. placebo; Large artery elasticity vs. placebo;♦ No effect on total cholesterol, LDLc, TG, glucose, hsCRP vs. placebo; ♦ No changes in SBP, DBP vs. placebo. | Basu et al. [23] |
| 2015 | Randomized, controlled, double-blind, crossover | 20 | 18–70 | ns | None | Cocoa powder | 80–800 | 17–168 | 1 |  FMD vs. control; FMD vs. control; SBP, DBP, PWV, ET1 vs. control SBP, DBP, PWV, ET1 vs. control | Grassi et al. [24] |
| 2015 | Blinded, randomized, controlled, crossover | 21 | ns | ns | Symptomatic peripheral artery disease | Dark chocolate | ns | ns | 1 dose | ♦ No effect on microvascular function vs. cocoa-free control chocolate. | Hammer et al. [25] |
| 2015 | Randomized, double-blind, controlled, parallel-group | 22 young 20 elderly | 22/60 | M | None | Cocoa powder | 900 | 128 | 2 |  FMD, arteriolar and microvascular vasodilator capacity, red blood cell deformability vs. baseline; FMD, arteriolar and microvascular vasodilator capacity, red blood cell deformability vs. baseline; PWV, total peripheral resistance vs. baseline. PWV, total peripheral resistance vs. baseline. | Heiss et al. [26] |
| 2015 | Randomized, controlled, blind open-label | 79 | 42 | M + F | Hypertension (grades I and II) treated with captopril or telmisartan | Dark chocolate + dehydrated red apples + green tea | 425.8 | ns | 24 |  SBP, DBP, TG, hsCRP vs. control SBP, DBP, TG, hsCRP vs. control | De Jesús Romero-Prado et al. [27] |
| 2015 | Randomized, controlled, crossover | 22 | 33–64 | M + F | Mild hypertension | Dark chocolate | 602.70 | ns | 8 |  DBP, SBP vs. baseline; DBP, SBP vs. baseline;♦ Improvement of subjects status from hypertensive to normotensive; ♦ No significant changes in CAVI vs. baseline; ♦ No significant changes in lipid profile, blood glucose, blood insulin vs. baseline. | Koli et al. [28] |
| 2015 | Randomized, placebo-controlled, double-blind, parallel | 40 | 18–43 | ns | None | Cocoa extract | 250 | ns | 1 dose; 4 | ♦ No significant changes in peripheral and central blood pressure vs. baseline; ♦ No significant changes in central blood flow vs. baseline. | Massee et al. [29] |
| 2015 | Randomized, double-blind | 24 | 22 | F | None/Overweight Obesity | Cocoa powder | 640 | 48 | 4 |  Haptoglobin, total and proinflammatory monocyte CD62L expression in obese women vs. baseline; Haptoglobin, total and proinflammatory monocyte CD62L expression in obese women vs. baseline; Endothelial microparticles in obese and overweight women vs. baseline. Endothelial microparticles in obese and overweight women vs. baseline. | McFarlin et al. [30] |
| 2015 | Randomized, parallel-arm, double-masked, controlled dietary intervention | 46 | 35–55 | M + F | None | Food-grade cocoa extract | 2000 | 220 | 12 | ♦ No significant in BP, platelet function, liver panel (albumin, bilirubin, ALT, AST, AlkPhos), metabolic markers (glucose, hemoglobin, hematocrit, urea, Na+, K+, Ca2+, Cl-, leucocytes, erythrocytes) vs. cocoa flavanols-free control | Ottaviani et al. [31] |
| 2015 | Double-blind, randomized, crossover | 15 | 18–35 | M | None | Cocoa powder | 1.4–10.9 /kg | 0.37–1.5 /kg | 1 dose |  FMD vs. baseline FMD vs. baseline | Rodriguez-Mateos et al. [32] |
| 2015 | Randomized, placebo-controlled, double-blind | 60 | 57 | M + F | Type 2 diabetes and Hypertension | Dark chocolate | 450 | ns | 8 |  FBG, SBP, DBP, hsCRP, apolipoprotein B vs. baseline; FBG, SBP, DBP, hsCRP, apolipoprotein B vs. baseline; FBG, SBP, DBP in dark chocolate group vs. control. FBG, SBP, DBP in dark chocolate group vs. control. | Rostami et al. [33] |
| 2015 | Randomized, double-blind, placebo-controlled | 32 | 45–70 | M | Pre-hypertension, Mild hypertension | Dark chocolate | 1064 (HFDC); 88 (LFDC) | ns | 6 |  Platelet aggregation induced by thrombin-receptor activation, ADP vs. baseline; Platelet aggregation induced by thrombin-receptor activation, ADP vs. baseline; Heart rate (LFDC) vs. baseline; Heart rate (LFDC) vs. baseline;♦No changes in total cholesterol, LDLc, HDLc, TG vs. Baseline ♦No significant changes in SBP, DBP (HFDC ) vs. baseline or LFDC. | Rull et al. [34] |
| 2015 | Randomized, double-blind, controlled | 100 | 44 | M + F | None | Cocoa powder | 450 | 64 | 4 |  FMD, HDLc vs. control; FMD, HDLc vs. control; SBP, DPB, PWV, total and LDLc vs. control. SBP, DPB, PWV, total and LDLc vs. control. | Sansone et al. [35] |
| 2015 | Two consecutive controlled, crossover | 44 | 29 | M + F | None/Mild hypercholesterolemia | Cocoa powder rich in dietary fibers/polyphenols | 43.8/45.3 | 9.3/18.9 | 4 |  HDLc after the 2nd intervention vs. baseline; HDLc after the 2nd intervention vs. baseline; Plasma glucose and IL-1β in cocoa product rich in dietary fiber group vs. baseline. Plasma glucose and IL-1β in cocoa product rich in dietary fiber group vs. baseline. | Sarriá et al. [36] |
| 2015 | Randomized, double-blind, crossover | 7 | 24–31 | M | None | Pure flavanols (monomers and polymers) | 1 mg/kg bw (EC, PCB1); 2 mg/kg bw PPC | - | Single dose | Glucuronidated, sulfated, methylated (-)EC and DHPV are predominant metabolites in blood and urine. | Wiese et al. [37] |
| 2014 | Randomised, double-blind, crossover | 40 | 64/27/60 | M + F | None (young and old)/Coronary artery disease | Cocoa beverage | 375 | 59 | 4 |  CD144+ EMP, CD31+/412− vs. baseline CD144+ EMP, CD31+/412− vs. baseline | Horn et al. [38] |
| 2014 | Randomised, parallel and double-blind | 50 | 57 | M + F | None | Cocoa powder | 414.26 | 153.44 | 4 |  Oxidized LDLc vs. control and vs. baseline; Oxidized LDLc vs. control and vs. baseline; MPO, ICAM1 vs. baseline. MPO, ICAM1 vs. baseline. | Ibero-Baibar et al. [39] |
| 2014 | Non-randomized, controlled, crossover | 44 | 29 | M + F | None/ Hypercholesterolemia | Cocoa powder | 45.3 | 18.9 | 4 |  HDLc vs. baseline; HDLc vs. baseline; IL 10 vs. baseline. IL 10 vs. baseline. | Martinez-López et al. [40] |
| 2014 | Double-blind, controlled, parallel-arm | 90 | 61–85 | M + F | Diabetes, Hypercholesterolemia Hypertension (pharmacological treatments), Former smokers | Cocoa drinks | 993 (HF); 520 (IF); 48 (LF) | 185 (HF); 95 (IF); 5 (LF) | 8 |  SBP, DBP (HF, IF) vs. LF; SBP, DBP (HF, IF) vs. LF; Plasma glucose concentrations (HF, IF) vs. LF; Plasma glucose concentrations (HF, IF) vs. LF; Insulin resistance (HF, IF) vs. LF; Insulin resistance (HF, IF) vs. LF; LDLc, TG, total cholesterol vs. LF; LDLc, TG, total cholesterol vs. LF; Plasma total 8-iso-prostaglandin F2α (HF, LF) vs. LF; Plasma total 8-iso-prostaglandin F2α (HF, LF) vs. LF; Circulating insulin (HF, IF) vs. LF; Circulating insulin (HF, IF) vs. LF; HDLc (HF, IF) vs. LF. HDLc (HF, IF) vs. LF. | Mastroiacovo et al. [41] |
| 2014 | Randomized, controlled, crossover | 44 | 29 | M + F | None/Hypercholeste-romia | Cocoa powder | 44.1 | 9.3 | 4 |  HDLc vs. baseline; HDLc vs. baseline; Glucose, IL-1β and IL-10 vs. baseline. Glucose, IL-1β and IL-10 vs. baseline. | Sarría et al. [42] |
| 2014 | Randomized, single-blinded, prospective placebo-controlled | 60 | 65 | ns | None/Glaucoma | Dark chocolate | ns | ns | 1 dose |  Mean dilatation of the retinal venules in age-matched controls, but not in glaucoma patients; Mean dilatation of the retinal venules in age-matched controls, but not in glaucoma patients; Venous vasodilatation in control group, but not in the glaucoma group. Venous vasodilatation in control group, but not in the glaucoma group. | Terai et al. [43] |
| 2014 | Randomized, double-blind, 2 period, crossover | 30 | 52 | M + F | Overweight, Moderate obesity | Dark chocolate and sugar-free cocoa powder | 814 | 73.6 | 4 |  Basal blood flow, basal and peak diameter of the brachial artery, vs. baseline Basal blood flow, basal and peak diameter of the brachial artery, vs. baseline | West et al. [44] |
| Year | Study Design | No. of Participants | Aprox. Mean Age (years) | Sex | Comorbidities | Form of EC Intervention | EC (mg/day) | Duration (weeks) | Outcomes | Ref. |
|---|---|---|---|---|---|---|---|---|---|---|
| 2016 | Randomized, multicenter, placebo-controlled, double-blind | 30 | 18–55 | M + F | Hypertriglyceridemia | Capsules | 100 | 4 |  TG, TG/HDLc ratio, hsCRP vs. baseline TG, TG/HDLc ratio, hsCRP vs. baseline | Gutiérrez-Salméan et al. [81] |
| 2015 | Non-randomized open label | 9 (single-dose); 8 (multiple-dose) | 23–68 (single-dose); 22–45 (multiple-dose) | ns | None | Dissolved in water | 50; 100 | Single-dose; multiple-dose, 5 days | ↑ Plasma nitrite vs. baseline; ↑ Platelet mitochondria complexes I, IV, citrate synthase activities (multi-dose) vs. baseline; ↑ Plasma follistatin levels (multi-dose) vs. baseline. | Barnett et. al. [82] |
| 2015 | Randomized, double blind, placebo-controlled crossover | 35 | 40–80 | M + F | Pre-hypertension | Capsules | 100 | 4 |  Plasma soluble endothelial selectin vs. control Plasma soluble endothelial selectin vs. control | Dower et al. [83] |
| 2015 | Randomized, double-blind, placebo-controlled, crossover | 33 | 40–80 | M + F | None | Capsules | 100 | 4 |  Fasting plasma insulin and insulin resistance vs. control Fasting plasma insulin and insulin resistance vs. control | Dower et al. [84] |
| 2014 | Pilot, open-labeled, crossover | 20 | 28 | M + F | None/Overweight | Capsules | 1 mg/kg | 1 dose |  Blood glucose and TG after 2 h vs. control Blood glucose and TG after 2 h vs. control | Gutiérrez-Salméan et al. [85] |
| 2008 | Randomized, placebo-controlled, crossover | 12 | 43 | M | None | Dissolved in water | 200 | 1 dose |  Plasma S-nitrosothiols and nitrite after 2 h and urinary nitrate after 5 h vs. Baseline Plasma S-nitrosothiols and nitrite after 2 h and urinary nitrate after 5 h vs. Baseline Plasma entothelin-1 after 2 h vs. baseline Plasma entothelin-1 after 2 h vs. baseline | Loke et al. [86] |
| 2006 | Randomized, double-blind, crossover | 6 (3 + 3) | 25–32 | M | None | Dissolved in water | 1 or 2 mg/kg | 1 dose |  FMD and PAT responses after 2 h vs. baseline and vs. control FMD and PAT responses after 2 h vs. baseline and vs. control | Schroeter et al. [87] |
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Aprotosoaie, A.C.; Miron, A.; Trifan, A.; Luca, V.S.; Costache, I.-I. The Cardiovascular Effects of Cocoa Polyphenols—An Overview. Diseases 2016, 4, 39. https://doi.org/10.3390/diseases4040039
Aprotosoaie AC, Miron A, Trifan A, Luca VS, Costache I-I. The Cardiovascular Effects of Cocoa Polyphenols—An Overview. Diseases. 2016; 4(4):39. https://doi.org/10.3390/diseases4040039
Chicago/Turabian StyleAprotosoaie, Ana Clara, Anca Miron, Adriana Trifan, Vlad Simon Luca, and Irina-Iuliana Costache. 2016. "The Cardiovascular Effects of Cocoa Polyphenols—An Overview" Diseases 4, no. 4: 39. https://doi.org/10.3390/diseases4040039
APA StyleAprotosoaie, A. C., Miron, A., Trifan, A., Luca, V. S., & Costache, I.-I. (2016). The Cardiovascular Effects of Cocoa Polyphenols—An Overview. Diseases, 4(4), 39. https://doi.org/10.3390/diseases4040039







