Considerations for Augmenting Aripiprazole Long-Acting Injectables with Other Antipsychotics: A Mini-Review
Abstract
1. Introduction
2. Methods
3. Results
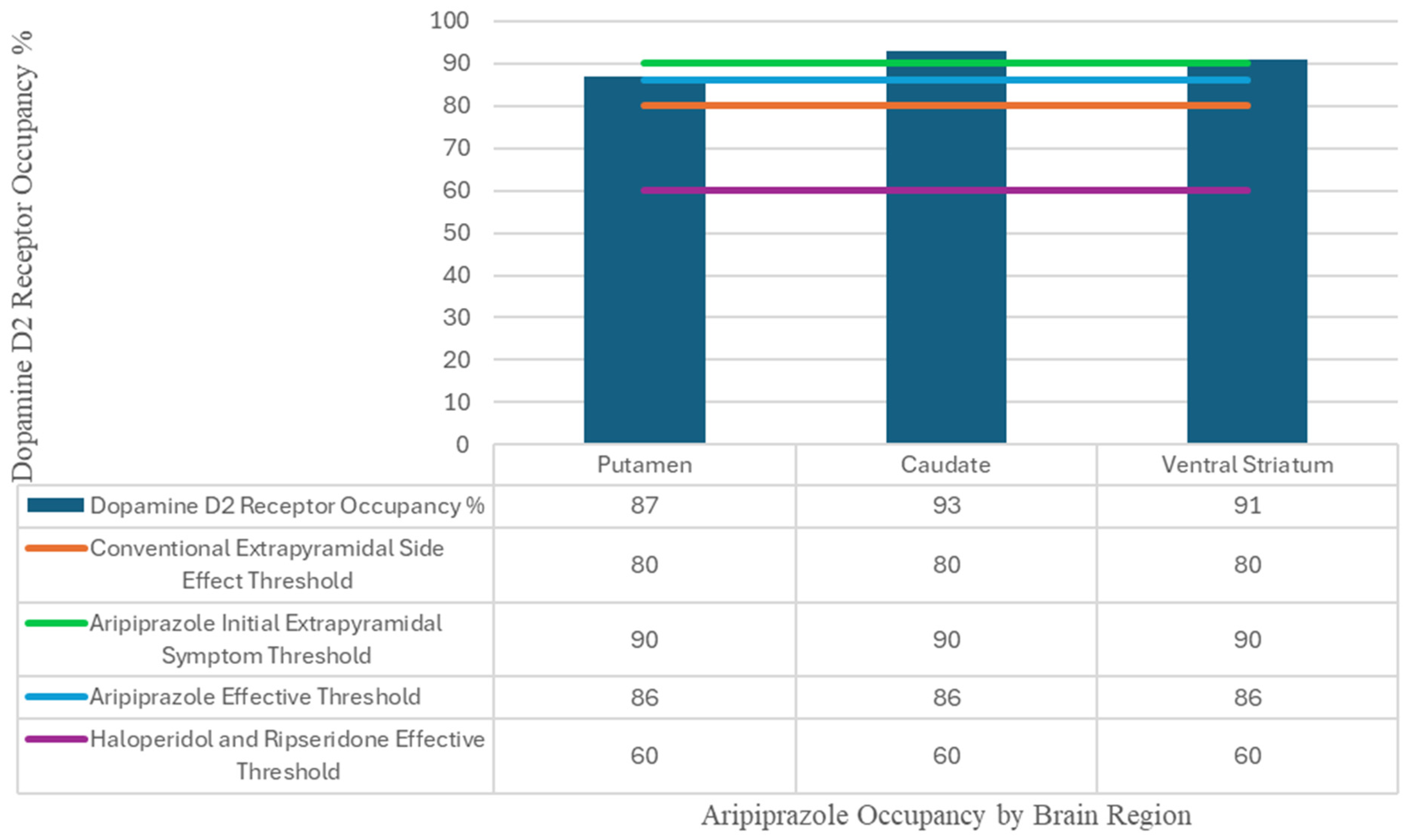
3.1. Mechanism Specifics
3.2. Side Effects Profiles of the Various Generations of Antipsychotics
3.3. Use with Other Antipsychotics and Psychotropic Medications
4. Discussion
4.1. Mechanistic Considerations
4.2. Patient Quality of Life and Logistical Considerations
4.3. Future Directions
5. Limitations
5.1. Methodological Limitations
5.2. Literature-Based Limitations
6. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| First-generation antipsychotic | FGA |
| Second-generation antipsychotic | SGA |
| Dopamine D2 receptor | DRD2 |
| Dopamine D2 receptor (long isoform) | D2LR |
| Long-acting injectable | LAI |
| Extrapyramidal symptoms | EPS |
| Richmond Agitation-Sedation Scale | RASS |
| Cytochrome P450 | CYP |
| Antipsychotic polypharmacy | APP |
| Antipsychotic monotherapy | APM |
| Significant adverse events | SAE |
| Muscarinic acetylcholine receptor | mAChR |
| N-desmethylclozapine | NDMC |
| Xanomeline-trospium | XT |
| Positive and Negative Syndrome Scale | PANSS |
References
- Casey, A.B.; Canal, C.E. Classics in Chemical Neuroscience: Aripiprazole. ACS Chem. Neurosci. 2017, 8, 1135–1146. [Google Scholar] [CrossRef]
- Siafis, S.; Wu, H.; Wang, D.; Burschinski, A.; Nomura, N.; Takeuchi, H.; Schneider-Thoma, J.; Davis, J.M.; Leucht, S. Antipsychotic dose, dopamine D2 receptor occupancy and extrapyramidal side-effects: A systematic review and dose-response meta-analysis. Mol. Psychiatry 2023, 28, 3267–3277. [Google Scholar] [CrossRef]
- Mamo, D.; Graff, A.; Mizrahi, R.; Shammi, C.M.; Romeyer, F.; Kapur, S. Differential Effects of Aripiprazole on D2, 5-HT2, and 5-HT1AReceptor Occupancy in Patients With Schizophrenia: A Triple Tracer PET Study. Am. J. Psychiatry 2007, 164, 1411–1417. [Google Scholar] [CrossRef] [PubMed]
- Burkat, P.M. Haloperidol dopamine receptor occupancy and antagonism correspond to delirium agitation scores and EPS risk: A PBPK-PD modeling analysis. J. Psychopharmacol. 2025, 39, 244–253. [Google Scholar] [CrossRef] [PubMed]
- Gründer, G.; Fellows, C.; Janouschek, H.; Veselinovic, T.; Boy, C.; Bröcheler, A.; Kirschbaum, K.M.; Hellmann, S.; Spreckelmeyer, K.M.; Hiemke, C.; et al. Brain and Plasma Pharmacokinetics of Aripiprazole in Patients With Schizophrenia: An [18F]Fallypride PET Study. Am. J. Psychiatry 2008, 165, 988–995. [Google Scholar] [CrossRef] [PubMed]
- Barnett, J.; Pappa, S. Long-Term Outcomes of Aripiprazole Long-Acting Injectable: A 10-Year Mirror Image Study of Patient Acceptability and Treatment Effectiveness. Schizophrenia 2025, 11, 92. [Google Scholar] [CrossRef]
- Sampogna, G.; Di Vincenzo, M.; Giuliani, L.; Menculini, G.; Mancuso, E.; Arsenio, E.; Cipolla, S.; Della Rocca, B.; Martiadis, V.; Signorelli, M.S.; et al. A Systematic Review on the Effectiveness of Antipsychotic Drugs on the Quality of Life of Patients with Schizophrenia. Brain Sci. 2023, 13, 1577. [Google Scholar] [CrossRef]
- rothlab. Rothlab. Unc.edu. Available online: https://pdsp.unc.edu/rothlab/publications.php (accessed on 14 May 2025).
- Shnayder, N.A.; Abdyrakhmanova, A.K.; Nasyrova, R.F. Phase I of Antipsychotics Metabolism and its Pharmacogenetic Testing. Pers. Psychiatry Neurol. 2022, 2, 4–21. [Google Scholar] [CrossRef]
- Crapanzano, C.; Laurenzi, P.F.; Amendola, C.; Casolaro, I. Clinical perspective on antipsychotic receptor binding affinities. Braz. J. Psychiatry 2021, 43, 680–681. [Google Scholar] [CrossRef]
- Kroeze, W.K.; Hufeisen, S.J.; Popadak, B.A.; Renock, S.M.; Steinberg, S.; Ernsberger, P.; Jayathilake, K.; Meltzer, H.Y.; Roth, B.L. H1-Histamine Receptor Affinity Predicts Short-Term Weight Gain for Typical and Atypical Antipsychotic Drugs. Neuropsychopharmacology 2003, 28, 519–526. [Google Scholar] [CrossRef]
- Shapiro, D.A.; Renock, S.; Arrington, E.; Chiodo, L.A.; Liu, L.-X.; Sibley, D.R.; Roth, B.L.; Mailman, R. Aripiprazole, A Novel Atypical Antipsychotic Drug with a Unique and Robust Pharmacology. Neuropsychopharmacology 2003, 28, 1400–1411. [Google Scholar] [CrossRef]
- Evernden, C.; Giang, I.; Anderson, M. The use of concurrent long-acting injectable antipsychotic therapy with paliperidone palmitate and aripiprazole monohydrate in a patient with schizophrenia. Ment. Health Clin. 2021, 11, 305–310. [Google Scholar] [CrossRef]
- Cipolla, S.; Catapano, P.; D’amico, D.; Monda, R.; Sallusto, N.P.; Perris, F.; De Santis, V.; Catapano, F.; Luciano, M.; Fiorillo, A. Combination of Two Long-Acting Antipsychotics in Schizophrenia Spectrum Disorders: A Systematic Review. Brain Sci. 2024, 14, 433. [Google Scholar] [CrossRef] [PubMed]
- Lähteenvuo, M.; Tiihonen, J. Antipsychotic Polypharmacy for the Management of Schizophrenia: Evidence and Recommendations. Drugs 2021, 81, 1273–1284. [Google Scholar] [CrossRef] [PubMed]
- Schneider-Thoma, J.; Efthimiou, O.; Bighelli, I.; Dörries, C.; Huhn, M.; Krause, M.; Reichelt, L.; Röder, H.; Furukawa, T.A.; Davis, J.M.; et al. Second-generation antipsychotic drugs and short-term somatic serious adverse events: A systematic review and meta-analysis. Lancet Psychiatry 2019, 6, 753–765. [Google Scholar] [CrossRef] [PubMed]
- Kishi, T.; Sakuma, K.; Saito, T.; Nakagawa, A.; Kato, M.; Iwata, N. Comparison of Brexpiprazole, Aripiprazole, and Placebo for Japanese Major Depressive Disorder: A Systematic Review and Network Meta-Analysis. Neuropsychopharmacol. Rep. 2024, 44, 165–175. [Google Scholar] [CrossRef]
- Ishigooka, J.; Inada, K.; Niidome, K.; Aoki, K.; Kojima, Y.; Iwashita, S.; Yamada, S. Safety of Switching to Brexpiprazole in Japanese Patients with Schizophrenia: A Post-Hoc Analysis of a Long-Term Open-Label Study. Hum. Psychopharmacol. Clin. Exp. 2021, 36, e2777. [Google Scholar] [CrossRef]
- Paul, S.M.; Yohn, S.E.; Popiolek, M.; Miller, A.C.; Felder, C.C. Muscarinic Acetylcholine Receptor Agonists as Novel Treatments for Schizophrenia. Am. J. Psychiatry 2022, 179, 611–627. [Google Scholar] [CrossRef]
- Freibüchler, A.; Seifert, R. Analysis of Clinical Studies on Clozapine from 2012–2022. Naunyn-Schmiedeberg’s Arch. Pharmacol. 2024, 397, 9745–9765. [Google Scholar] [CrossRef]
- Correll, C.U.; Agid, O.; Crespo-Facorro, B.; de Bartolomeis, A.; Fagiolini, A.; Seppälä, N.; Howes, O.D. A Guideline and Checklist for Initiating and Managing Clozapine Treatment in Patients with Treatment-Resistant Schizophrenia. CNS Drugs 2022, 36, 659–679. [Google Scholar] [CrossRef]
- Dean, B. IUPHAR Review on Muscarinic M1 and M4 Receptors as Drug Treatment Targets Relevant to the Molecular Pathology of Schizophrenia. Pharmacol. Res. 2024, 210, 107510. [Google Scholar] [CrossRef] [PubMed]
- Bartolomeis, A.; Vellucci, L.; Barone, A.; Manchia, M.; De Luca, V.; Iasevoli, F.; Correll, C.U. Clozapine’s multiple cellular mechanisms: What do we know after more than fifty years? A systematic review and critical assessment of translational mechanisms relevant for innovative strategies in treatment-resistant schizophrenia. Pharmacol. Ther. 2022, 236, 108236. [Google Scholar] [CrossRef] [PubMed]
- Fabiano, N.; Wong, S.; Zhou, C.; Correll, C.U.; Højlund, M.; Solmi, M. Efficacy, Tolerability, and Safety of Xanomeline-Trospium Chloride for Schizophrenia: A Systematic Review and Meta-Analysis. Eur. Neuropsychopharmacol. 2024, 92, 62–73. [Google Scholar] [CrossRef] [PubMed]
- Moran, S.P.; Maksymetz, J.; Conn, P.J. Targeting Muscarinic Acetylcholine Receptors for the Treatment of Psychiatric and Neurological Disorders. Trends Pharmacol. Sci. 2019, 40, 1006–1020. [Google Scholar] [CrossRef]
- Biso, L.; Carli, M.; Scarselli, M.; Longoni, B. Overview of Novel Antipsychotic Drugs: State of the Art, New Mechanisms, and Clinical Aspects of Promising Compounds. Biomedicines 2025, 13, 85. [Google Scholar] [CrossRef]
- Smith, C.M.; Augustine, M.S.; Dorrough, J.; Szabo, S.T.; Shadaram, S.; Hoffman, E.O.; Muzyk, A. Xanomeline-trospium (CobenfyTM) for Schizophrenia: A Review of the Literature. Clin. Psychopharmacol. Neurosci. 2024, 23, 2–14. [Google Scholar] [CrossRef]
- Kaul, I.; Claxton, A.; Sawchak, S.; Sauder, C.; Brannan, S.K.; Raj, E.; Ruan, S.; Konis, G.; Brown, D.; Cutler, A.J.; et al. Safety and Tolerability of Xanomeline and Trospium Chloride in Schizophrenia. J. Clin. Psychiatry 2025, 86, 24m15497. [Google Scholar] [CrossRef]
- Meyer, J.M.; Kramer, K.; Vuocolo, S.; Kaul, I.; Miller, A.C. From Theory to Therapy: Unlocking the Potential of Muscarinic Receptor Activation in Schizophrenia with the Dual M1/M4 Muscarinic Receptor Agonist Xanomeline and Trospium Chloride and Insights from Clinical Trials. Int. J. Neuropsychopharmacol. 2025, 28, pyaf015. [Google Scholar] [CrossRef]
- Kaul, I.; Sawchak, S.; Walling, D.P.; Tamminga, C.A.; Breier, A.; Zhu, H.; Miller, A.C.; Paul, S.M.; Brannan, S.K. Efficacy and Safety of Xanomeline-Trospium Chloride in Schizophrenia. JAMA Psychiatry 2024, 81, 749–756. [Google Scholar] [CrossRef]
- Bristol Myers Squibb Announces Topline Results from Phase 3 ARISE Trial Evaluating Cobenfy (Xanomeline and Trospium Chloride) as an Adjunctive Treatment to Atypical Antipsychotics in Adults with Schizophrenia 2020. Available online: https://news.bms.com/news/details/2025/Bristol-Myers-Squibb-Announces-Topline-Results-from-Phase-3-ARISE-Trial-Evaluating-Cobenfy-xanomeline-and-trospium-chloride-as-an-Adjunctive-Treatment-to-Atypical-Antipsychotics-in-Adults-with-Schizophrenia/default.aspx (accessed on 14 May 2025).
- Clinicaltrials.gov. 2025. Available online: https://clinicaltrials.gov/study/NCT05145413#study-plan (accessed on 14 May 2025).
- Citrome, L.; Neugebauer, N.; Meli, A.; Kando, J. Xanomeline and Trospium Chloride versus Placebo for the Treatment of Schizophrenia: A Post Hoc Analysis of Number Needed to Treat, Number Needed to Harm, and Likelihood to Be Helped or Harmed. Neuropsychiatr. Dis. Treat. 2025, 21, 761–773. [Google Scholar] [CrossRef]
| Antipsychotic | Average Ki Value (nM) | Ki Value Range | Metabolism | Notes/Considerations |
|---|---|---|---|---|
| Fluphenazine | 0.54 | 0.540000 a,b | CYP2D6 | More susceptible to CYP2D6 inhibitors than other antipsychotics. d |
| Pimozide | 0.65 | 0.650000 b | CYP2D6 | Moderate sensitivity to CYP2D6. d |
| Thiothixene | 1.015 | 0.630000–1.400000 a,b | - | - |
| Trifluoperazine | 1.3 | 1.300000 b | - | - |
| Perphenazine | 1.4 | 1.400000 b | - | Multiple pathways of clearance, limited impact of CYP induction/inhibition on serum levels. d |
| Aripiprazole | 2.455 | 0.660000–3.300000 a,b,c | CYP2D6, CYP3A4 | Manufacturer recommends twofold dose increase when administered with inducers like carbamazepine and a two-fold dose reduction with inhibitors such as fluoxetine, quinidine, or ketoconazole. d |
| Chlorpromazine | 3 | 2.000000–4.000000 a,b | CYP1A2 | Recommended to avoid smoking tobacco as it induces CYP1A2. d |
| Haloperidol | 3 | 2.000000–4.000000 a,b | Primarily CYP2D6, CYP3A4, and CYP3A5 | Has multiple pathways, limiting impact of CYP induction/inhibition on serum levels. d |
| Risperidone | 5.7 | 4.900000–6.500000 a,b | CYP2D6 | Serum levels may rise with CYP2D6 inhibitors like fluoxetine or paroxetine, will increase to a lesser degree with bupropion. d |
| Ziprasidone | 6.85 | 4.000000–9.700000 a,b | Primarily through glutathione and aldehyde oxidase with minor contribution from CYP3A4 | Inhibitors and inducers of CYP only cause modest effects in serum levels. d |
| Sertindole | 9.1 | 9.100000 b | - | - |
| Thioridazine | 10.5 | 10.000000–11.000000 a,b | - | Multiple pathways of clearance, limited impact of CYP induction/inhibition on serum levels. d |
| Loxapine | 11 | 10.000000–12.000000 a,b | - | Multiple pathways of clearance, limited impact of CYP induction/inhibition on serum levels. d |
| Olanzapine | 53 | 34.000000–72.000000 a,b | CYP1A2 | Avoid strong inhibitors or inducers of CYP1A2 as well as smoking tobacco. d |
| Molindone | 63 | 63.000000 b | - | - |
| Clozapine | 343.5 | 256.000000–431.000000 a,b | Primarily CYP1A2 and CYP3A4, some CYP2D6 | May require reduction of one-third dose with fluvoxamine or ciprofloxacin, smoking may require twofold increase in dose with 30–40% reduction of dose in settings where smoker is unable to smoke. d |
| Quetiapine | 410.5 | 254.000000–567.000000 a,b | CYP3A4 | Manufacturer recommends up to a fivefold increase in dose with CYP3A4 inducers like carbamazepine and a reduction to one-sixth with inhibitors like voriconazole or ritonavir. d |
| Receptor | G Protein Subtype | Function | Regions of the Brain Mainly Found in | Dopamine Pathway(s) Affected |
|---|---|---|---|---|
| M1 | Gq | Excitatory | Cerebral Cortex, basal ganglia | Nigrostriatal pathway more than Mesolimbic pathway |
| M2 | Gi/o | Inhibition | Nucleus Basalis, hippocampus, basal ganglia | Mesolimbic pathway more than Nigrostriatal pathway |
| M3 | Gq | Excitatory | Cerebral cortex, basal ganglia | Mesocortical pathway |
| M4 | Gi/o | Inhibition | Basal ganglia and cortex | Nigrostriatal pathway more than Mesolimbic pathway |
| M5 | Gq | Excitatory | Hippocampus, substantia nigra, and VTA | Mesolimbic pathway more than Mesocortical pathway |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Shaw, J.; Kim, E.; Ton, E.; Lai, C.; Bota, P.; Allee, T. Considerations for Augmenting Aripiprazole Long-Acting Injectables with Other Antipsychotics: A Mini-Review. Diseases 2025, 13, 274. https://doi.org/10.3390/diseases13080274
Shaw J, Kim E, Ton E, Lai C, Bota P, Allee T. Considerations for Augmenting Aripiprazole Long-Acting Injectables with Other Antipsychotics: A Mini-Review. Diseases. 2025; 13(8):274. https://doi.org/10.3390/diseases13080274
Chicago/Turabian StyleShaw, Jonathan, Ethan Kim, Emily Ton, Charles Lai, Peter Bota, and Tina Allee. 2025. "Considerations for Augmenting Aripiprazole Long-Acting Injectables with Other Antipsychotics: A Mini-Review" Diseases 13, no. 8: 274. https://doi.org/10.3390/diseases13080274
APA StyleShaw, J., Kim, E., Ton, E., Lai, C., Bota, P., & Allee, T. (2025). Considerations for Augmenting Aripiprazole Long-Acting Injectables with Other Antipsychotics: A Mini-Review. Diseases, 13(8), 274. https://doi.org/10.3390/diseases13080274






