Cold Anaphylaxis in Children: Italian Case Series and Review of the Literature
Abstract
:1. Introduction
1.1. Definition
1.2. Prevalence
1.3. Pathogenesis
1.4. Genetics
1.5. Diagnosis
1.5.1. Physical Tests
1.5.2. Differential Diagnosis
1.5.3. Laboratory Tests
1.6. Desensitization
1.7. Aims of the Study
2. Material and Methods
3. Results
3.1. Demographic Characteristics and Comorbidities
3.2. Reaction Triggers
3.3. Clinical Manifestations of ColdA
3.4. Provocation Test and Laboratory Studies
3.5. Management and Follow-Up
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
References
- Lee, S.J.; Ha, E.K.; Jee, H.M.; Lee, K.S.; Lee, S.W.; Kim, M.A.; Kim, D.H.; Jung, Y.-H.; Sheen, Y.H.; Sung, M.S.; et al. Prevalence and Risk Factors of Urticaria with a Focus on Chronic Urticaria in Children. Allergy Asthma Immunol. Res. 2017, 9, 212. [Google Scholar] [CrossRef]
- Frank, J.P. De curandis Hominum Morbis Epitome Praelectonibus Academicis Dicata. Liber III: De Exanthematibus; Apud CF Schwan et CG Goetz Bibliop: Mannhemii, Germany, 1792. [Google Scholar]
- Zuberbier, T.; Abdul Latiff, A.H.; Abuzakouk, M.; Aquilina, S.; Asero, R.; Baker, D.; Ballmer-Weber, B.; Bangert, C.; Ben-Shoshan, M.; Bernstein, J.A.; et al. The international EAACI/GA2LEN/EuroGuiDerm/APAAACI guideline for the definition, classification, diagnosis, and management of urticaria. Allergy 2022, 77, 734–766. [Google Scholar] [CrossRef]
- Ben-Shoshan, M.; Grattan, C.E. Management of Pediatric Urticaria with Review of the Literature on Chronic Spontaneous Urticaria in Children. J. Allergy Clin. Immunol. Pract. 2018, 6, 1152–1161. [Google Scholar] [CrossRef]
- Vadas, P.; Sinilaite, A.; Chaim, M. Cholinergic Urticaria with Anaphylaxis: An Underrecognized Clinical Entity. J. Allergy Clin. Immunol. Pract. 2016, 4, 284–291. [Google Scholar] [CrossRef] [PubMed]
- Wanderer, A.A.; Grandel, K.E.; Wasserman, S.I.; Farr, R.S. Clinical characteristics of cold-induced systemic reactions in acquired cold urticaria syndromes: Recommendations for prevention of this complication and a proposal for a diagnostic classification of cold urticaria. J. Allergy Clin. Immunol. 1986, 78, 417–423. [Google Scholar] [CrossRef]
- Wanderer, A.A. Cold urticaria syndromes: Historical background, diagnostic classification, clinical and laboratory characteristics, pathogenesis, and management. J. Allergy Clin. Immunol. 1990, 85, 965–981. [Google Scholar] [CrossRef]
- Maltseva, N.; Borzova, E.; Fomina, D.; Bizjak, M.; Terhorst-Molawi, D.; Košnik, M.; Kulthanan, K.; Meshkova, R.; Thomsen, S.F.; Maurer, M. Cold urticaria—What we know and what we do not know. Allergy 2021, 76, 1077–1094. [Google Scholar] [CrossRef] [PubMed]
- Bizjak, M.; Košnik, M.; Dinevski, D.; Thomsen, S.F.; Fomina, D.; Borzova, E.; Kulthanan, K.; Meshkova, R.; Ahsan, D.M.; Al-Ahmad, M.; et al. Risk factors for systemic reactions in typical cold urticaria: Results from the COLD-CE study. Allergy 2022, 77, 2185–2199. [Google Scholar] [CrossRef] [PubMed]
- Kring Tannert, L.; Stahl Skov, P.; Bjerremann Jensen, L.; Maurer, M.; Bindslev-Jensen, C. Cold Urticaria Patients Exhibit Normal Skin Levels of Functional Mast Cells and Histamine after Tolerance Induction. Dermatology 2012, 224, 101–105. [Google Scholar] [CrossRef]
- Kobza Black, A.; Sibbald, R.; Greaves, M. Cold urticaria treated by induction of tolerance. Lancet 1979, 314, 964. [Google Scholar] [CrossRef]
- Bizjak, M.; Košnik, M.; Dinevski, D.; Thomsen, S.F.; Fomina, D.; Borzova, E.; Kulthanan, K.; Meshkova, R.; Aarestrup, F.M.; Ahsan, D.M.; et al. Adrenaline autoinjector is underprescribed in typical cold urticaria patients. Allergy 2022, 77, 2224–2229. [Google Scholar] [CrossRef] [PubMed]
- Stepaniuk, P.; Vostretsova, K.; Kanani, A. Review of cold-induced urticaria characteristics, diagnosis and management in a Western Canadian allergy practice. Allergy Asthma Clin. Immunol. 2018, 14, 85. [Google Scholar] [CrossRef]
- Prosty, C.; Gabrielli, S.; Le, M.; Ensina, L.F.; Zhang, X.; Netchiporouk, E.; Ben-Shoshan, M. Prevalence, Management, and Anaphylaxis Risk of Cold Urticaria: A Systematic Review and Meta-Analysis. J. Allergy Clin. Immunol. Pract. 2022, 10, 586–596.e4. [Google Scholar] [CrossRef]
- Manca, E.; Calò, A.; Sica, F.; Baiardi, C.; Di Toma, M.; Campanozzi, A. Anaphylaxis and cold urticaria in an 11-year-old girl. Minerva Pediatr. 2023, 75, 615–617. [Google Scholar] [CrossRef]
- Inuzuka, Y.; Yamamoto-Hanada, K.; Saito-Abe, M.; Ohya, Y. Pediatric cold-induced anaphylaxis and evaluation using TempTest®. Allergol. Int. 2022, 71, 412–413. [Google Scholar] [CrossRef]
- Benelli, E.; Longo, G.; Barbi, E.; Berti, I. Anaphylaxis in atypical cold urticaria: Case report and review of literature. Ital. J. Pediatr. 2018, 44, 135. [Google Scholar] [CrossRef]
- Maciag, M.C.; Nargozian, C.; Broyles, A.D. Intraoperative anaphylaxis secondary to systemic cooling in a pediatric patient with cold-induced urticaria. J. Allergy Clin. Immunol. Pract. 2018, 6, 1394–1395. [Google Scholar] [CrossRef]
- Romita, P.; Mascia, P.; Calogiuri, G.; Foti, C. Cold-induced Anaphylaxis: The Case of a 9-year-old Child and Review of the Literature. Endocr. Metab. Immune Disord. Drug Targets 2017, 17, 96–99. [Google Scholar] [CrossRef]
- Gaspar, Â.; Santos, N.; Piedade, S.; Santa-Marta, C.; Pires, G.; Sampaio, G.; Arêde, C.; Borrego, L.M.; Morais-Almeida, M. One-year survey of paediatric anaphylaxis in an allergy department. Eur. Ann. Allergy Clin. Immunol. 2015, 47, 197–205. [Google Scholar] [PubMed]
- Işk, S.; Arkan-Ayyldz, Z.; Sozmen, S.C.; Karaman, Ö.; Uzuner, N. Idiopathic Cold Urticaria and Anaphylaxis. Pediatr. Emerg. Care 2014, 30, 38–39. [Google Scholar] [CrossRef] [PubMed]
- Hochstadter, E.F.; Ben-Shoshan, M. Cold-induced urticaria: Challenges in diagnosis and management. Case Rep. 2013, 2013, bcr2013010441. [Google Scholar] [CrossRef]
- Silva, R.; Gomes, E.; Cunha, L.; Falcão, H. Anaphylaxis in children: A nine years retrospective study (2001–2009). Allergol. Immunopathol. 2012, 40, 31–36. [Google Scholar] [CrossRef]
- Fernando, S.L. Cold-Induced Anaphylaxis. J. Pediatr. 2009, 154, 148–148.e1. [Google Scholar] [CrossRef] [PubMed]
- Kulthanan, K.; Church, M.K.; Grekowitz, E.M.; Hawro, T.; Kiefer, L.A.; Munprom, K.; Nanchaipruek, Y.; Rujitharanawong, C.; Terhorst-Molawi, D.; Maurer, M. Evidence for histamine release in chronic inducible urticaria—A systematic review. Front. Immunol. 2022, 13, 901851. [Google Scholar] [CrossRef] [PubMed]
- Meyer, J.; Gorbach, A.M.; Liu, W.-M.; Medic, N.; Young, M.; Nelson, C.; Arceo, S.; Desai, A.; Metcalfe, D.D.; Komarow, H.D. Mast Cell Dependent Vascular Changes Associated with an Acute Response to Cold Immersion in Primary Contact Urticaria. PLoS ONE 2013, 8, e56773. [Google Scholar] [CrossRef] [PubMed]
- Bizjak, M.; Košnik, M.; Terhorst-Molawi, D.; Dinevski, D.; Maurer, M. Cold Agglutinins and Cryoglobulins Associate With Clinical and Laboratory Parameters of Cold Urticaria. Front. Immunol. 2021, 12, 665491. [Google Scholar] [CrossRef]
- Ginter, K.; Ahsan, D.M.; Bizjak, M.; Krause, K.; Maurer, M.; Altrichter, S.; Terhorst-Molawi, D. Cryoglobulins, Cryofibrinogens, and Cold Agglutinins in Cold Urticaria: Literature Review, Retrospective Patient Analysis, and Observational Study in 49 Patients. Front. Immunol. 2021, 12, 675451. [Google Scholar] [CrossRef]
- Murphy, G.; Austen, K.; Fonferko, E.; Sheffer, A. Morphologically distinctive forms of cutaneous mast cell degranulation induced by cold and mechanical stimuli: An ultrastructural study. J. Allergy Clin. Immunol. 1987, 80, 603–611. [Google Scholar] [CrossRef]
- Kaplan, A.P.; Gray, L.; Shaff, R.E.; Horakova, Z.; Beaven, M.A. In vivo studies of mediator release in cold urticaria and cholinergic urticaria. J. Allergy Clin. Immunol. 1975, 55, 394–402. [Google Scholar] [CrossRef]
- Tillie-Leblond, I.; Gosset, P.; Janin, A.; Dalenne, R.; Joseph, M.; Wallaert, B.; Tonnel, A.-B. Tumor necrosis factor-α release during systemic reaction in cold urticaria. J. Allergy Clin. Immunol. 1994, 93, 501–509. [Google Scholar] [CrossRef]
- Ormerod, A.D.; Black, A.K.; Dawes, J.; Murdoch, R.D.; Koro, O.; Barr, R.M.; Greaves, M.W. Prostaglandin D2 and histamine release in cold urticaria unaccompanied by evidence of platelet activation. J. Allergy Clin. Immunol. 1988, 82, 586–589. [Google Scholar] [CrossRef] [PubMed]
- Grandel, K.E.; Farr, R.S.; Wanderer, A.A.; Eisenstadt, T.C.; Wasserman, S.I. Association of Platelet-Activating Factor with Primary Acquired Cold Urticaria. N. Engl. J. Med. 1985, 313, 405–409. [Google Scholar] [CrossRef] [PubMed]
- Kolkhir, P.; Giménez-Arnau, A.M.; Kulthanan, K.; Peter, J.; Metz, M.; Maurer, M. Urticaria. Nat. Rev. Dis. Prim. 2022, 8, 61. [Google Scholar] [CrossRef] [PubMed]
- Kaplan, A.P.; Garofalo, J.; Sigler, R.; Hauber, T. Idiopathic cold urticaria: In vitro demonstration of histamine release upon challenge of skin biopsies. N. Engl. J. Med. 1981, 305, 1074–1077. [Google Scholar] [CrossRef]
- Houser, D.D.; Arbesman, C.E.; Ito, K.; Wicher, K. Cold urticaria. Am. J. Med. 1970, 49, 23–33. [Google Scholar] [CrossRef]
- Metz, M.; Schütz, A.; Weller, K.; Gorczyza, M.; Zimmer, S.; Staubach, P.; Merk, H.F.; Maurer, M. Omalizumab is effective in cold urticaria-results of a randomized placebo-controlled trial. J. Allergy Clin. Immunol. 2017, 140, 864–867.e5. [Google Scholar] [CrossRef]
- Ombrello, M.J.; Remmers, E.F.; Sun, G.; Freeman, A.F.; Datta, S.; Torabi-Parizi, P.; Subramanian, N.; Bunney, T.D.; Baxendale, R.W.; Martins, M.S.; et al. Cold Urticaria, Immunodeficiency, and Autoimmunity Related to PLCG2 Deletions. N. Engl. J. Med. 2012, 366, 330–338. [Google Scholar] [CrossRef]
- Magerl, M.; Altrichter, S.; Borzova, E.; Giménez-Arnau, A.; Grattan, C.E.H.; Lawlor, F.; Mathelier-Fusade, P.; Meshkova, R.Y.; Zuberbier, T.; Metz, M.; et al. The definition, diagnostic testing, and management of chronic inducible urticarias—The EAACI/GA 2 LEN/EDF/UNEV consensus recommendations 2016 update and revision. Allergy 2016, 71, 780–802. [Google Scholar] [CrossRef]
- Siebenhaar, F.; Staubach, P.; Metz, M.; Magerl, M.; Jung, J.; Maurer, M. Peltier effect–based temperature challenge: An improved method for diagnosing cold urticaria. J. Allergy Clin. Immunol. 2004, 114, 1224–1225. [Google Scholar] [CrossRef]
- Holm, J.G.; Agner, T.; Thomsen, S.F. Diagnostic properties of provocation tests for cold, heat, and delayed-pressure urticaria. Eur. J. Dermatol. 2017, 27, 406–408. [Google Scholar] [CrossRef]
- Magerl, M.; Abajian, M.; Krause, K.; Altrichter, S.; Siebenhaar, F.; Church, M.K. An improved Peltier effect-based instrument for critical temperature threshold measurement in cold- and heat-induced urticaria. J. Eur. Acad. Dermatol. Venereol. 2015, 29, 2043–2045. [Google Scholar] [CrossRef] [PubMed]
- Booshehri, L.M.; Hoffman, H.M. CAPS and NLRP3. J. Clin. Immunol. 2019, 39, 277–286. [Google Scholar] [CrossRef] [PubMed]
- Mari, D.C.; Banks, T.A. Pearls and pitfalls: Cold-induced urticaria. Allergy Asthma Proc. 2020, 41, 301–304. [Google Scholar] [CrossRef] [PubMed]
- Bizjak, M.; Maurer, M.; Košnik, M.; Terhorst-Molawi, D.; Zver, S.; Burmeister, T.; Siebenhaar, F. Severe cold urticaria can point to an underlying clonal mast cell disorder. Allergy 2021, 76, 2609–2613. [Google Scholar] [CrossRef]
- von Mackensen, Y.A.; Sticherling, M. Cold urticaria: Tolerance induction with cold baths. Br. J. Dermatol. 2007, 157, 835–836. [Google Scholar] [CrossRef]
- Prosty, C.; Gabrielli, S.; Mule, P.; Le Gallee, M.; Miles, L.M.; Le, M.; Zhang, X.; Ensina, L.F.; Netchiporouk, E.; Ben-Shoshan, M. Cold urticaria in a pediatric cohort: Clinical characteristics, management, and natural history. Pediatr. Allergy Immunol. 2022, 33, e13751. [Google Scholar] [CrossRef]
- Yee, C.S.K.; El Khoury, K.; Albuhairi, S.; Broyles, A.; Schneider, L.; Rachid, R. Acquired Cold-Induced Urticaria in Pediatric Patients: A 22-Year Experience in a Tertiary Care Center (1996–2017). J. Allergy Clin. Immunol. Pract. 2019, 7, 1024–1031.e3. [Google Scholar] [CrossRef]
- Alangari, A.A.; Twarog, F.J.; Shih, M.-C.; Schneider, L.C. Clinical Features and Anaphylaxis in Children With Cold Urticaria. Pediatrics 2004, 113, e313–e317. [Google Scholar] [CrossRef]
- Azkur, D.; Civelek, E.; Toyran, M.; Misirlioǧlu, E.D.; Erkoçoǧlu, M.; Kaya, A.; Vezir, E.; Giniş, T.; Akan, A.; Kocabaş, C.N. Clinical and etiologic evaluation of the children with chronic urticaria. Allergy Asthma Proc. 2016, 37, 450–457. [Google Scholar] [CrossRef]
- Neittaanmäki, H. Cold urticaria: Clinical findings in 220 patients. J. Am. Acad. Dermatol. 1985, 13, 636–644. [Google Scholar] [CrossRef]
- Diluvio, L.; Piccolo, A.; Marasco, F.; Vollono, L.; Lanna, C.; Chiaramonte, B.; Niolu, C.; Campione, E.; Bianchi, L. Improving of psychological status and inflammatory biomarkers during omalizumab for chronic spontaneous urticaria. Futur. Sci. OA 2020, 6, FSO618. [Google Scholar] [CrossRef] [PubMed]
- Maurer, M.; Metz, M.; Brehler, R.; Hillen, U.; Jakob, T.; Mahler, V.; Pföhler, C.; Staubach, P.; Treudler, R.; Wedi, B.; et al. Omalizumab treatment in patients with chronic inducible urticaria: A systematic review of published evidence. J. Allergy Clin. Immunol. 2018, 141, 638–649. [Google Scholar] [CrossRef] [PubMed]
- Briand, C.; Tetart, F.; Soria, A.; Staumont-Sallé, D.; Sterling, B.; Boralevi, F.; Castelain Lakkis, F.; Du-Thanh, A.; Raison-Peyron, N.; Chiaverini, C. Omalizumab in cold urticaria in children: Retrospective case series of 13 patients, review of the literature. Ann. Dermatol. Venereol. 2021, 148, 269–271. [Google Scholar] [CrossRef] [PubMed]
- Deza, G.; Brasileiro, A.; Bertolín-Colilla, M.; Curto-Barredo, L.; Pujol, R.M.; Giménez-Arnau, A.M. Acquired cold urticaria: Clinical features, particular phenotypes, and disease course in a tertiary care center cohort. J. Am. Acad. Dermatol. 2016, 75, 918–924.e2. [Google Scholar] [CrossRef]
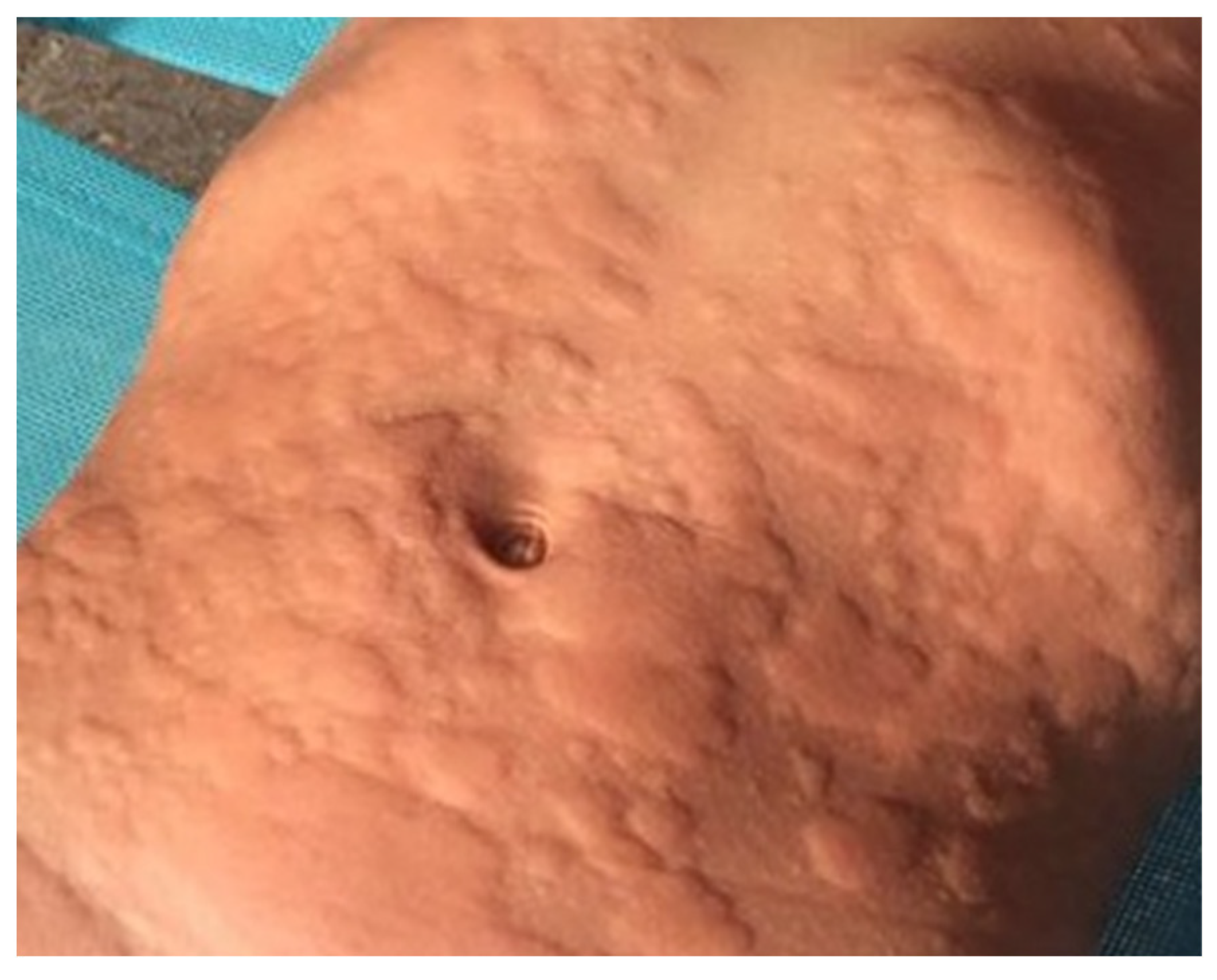
| Case Description | Management of the Anaphylaxis | AAI Prescription | Ref. | ||
|---|---|---|---|---|---|
| Age, Sex | Trigger | Clinical Manifestation | |||
| 11-year-old girl | Swimming in cold water | Urticaria, pallor, loss of consciousness | Oral steroids and antihistamines | Yes | [15] |
| 6-year-old boy | Swimming in cold water | Vomit, pallor, loss of consciousness | Improved spontaneously | Yes | [16] |
| 9-year-old boy | Swimming in cold water | Urticaria, dyspnea, conjunctival hyperemia, blurred vision and loss of strength | IM steroids and IV antihistamine | Yes | [17] |
| 15-year-old girl | Systemic cooling during surgery | Urticaria, angioedema, hypotension, desaturation | IV epinephrine, IV steroids, and antihistamine | No | [18] |
| 9-year-old girl | Walking in seawater | Urticaria, dry throat, dyspnea, loss of consciousness | Improved spontaneously | Yes | [19] |
| 14-year-old girl | Cold drink | Anaphylaxis (unspecified) | Not reported | Not reported | [20] |
| 14-year-old boy | Swimming in cold water | ||||
| 2-year-old boy | Swimming in cold water | ||||
| 13-year-old girl | Swimming in cold water | Loss of consciousness, dizziness | Improved spontaneously | Yes | [21] |
| 12-year-old boy | Swimming in cold water | Loss of consciousness, generalized seizure | Improved spontaneously | Yes | [22] |
| One child with ColdA | Not reported | Not reported | Not reported | Yes | [23] |
| 9-year-old girl | Swimming in cold water | Urticaria, loss of consciousness | Improved spontaneously | Yes | [24] |
| N of patients (%) | |
|---|---|
| Urticaria | 21 (100%) |
| Angioedema | 6 (28.57%) |
| Syncope/hypotension | 18 (85.71%) |
| Gastrointestinal symptoms | 8 (38.10%) |
| 7 vomit | |
| 1 vomit, abdominal pain, diarrhea | |
| Respiratory symptoms | 2 (9.52%) |
| 2 dyspnoea | |
| 2 cough | |
| 1 bronchospasm |
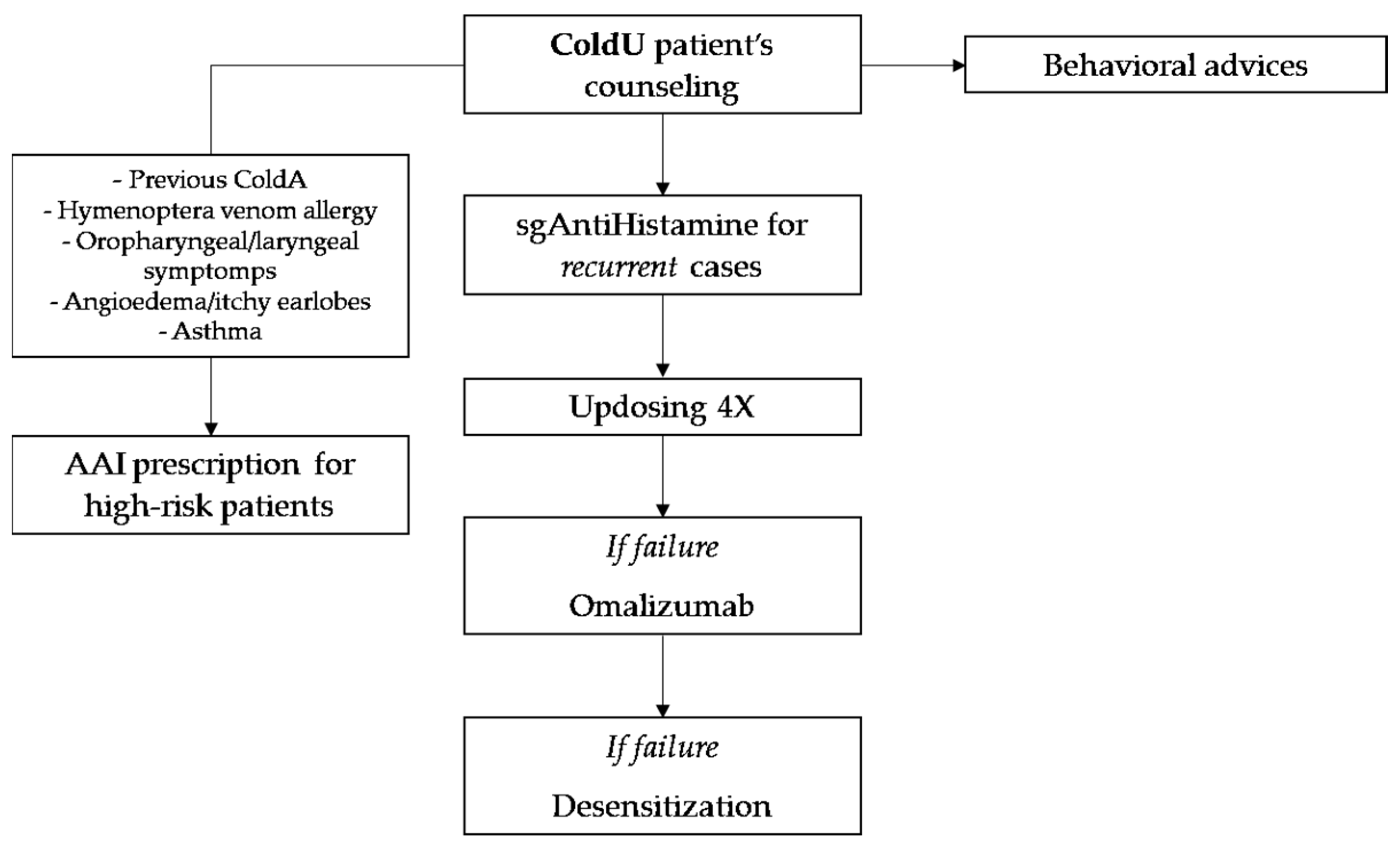
| Management Recommendations in ColdU and ColdA |
| Detailed anamnesis and family counseling |
| Avoidance of immersion in cold water |
| Swimming under surveillance |
| Avoidance of systemic cold exposure (rapid indoor/outdoor passage, refrigerated section at the supermarket, ice skating rink) |
| Warming of intravenous or irrigation fluids and premedication before surgery |
| Appropriate selections of clothes in cold weather |
| Pharmacological therapy (antihistamine and omalizumab) |
| Desensitization |
| Epinephrine auto-injector in high-risk cases |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tomei, L.; Saretta, F.; Arasi, S.; Sarti, L.; Licari, A.; Giovannini, M.; Barni, S.; Liccioli, G.; Tallarico, V.; Piccorossi, A.; et al. Cold Anaphylaxis in Children: Italian Case Series and Review of the Literature. Diseases 2023, 11, 143. https://doi.org/10.3390/diseases11040143
Tomei L, Saretta F, Arasi S, Sarti L, Licari A, Giovannini M, Barni S, Liccioli G, Tallarico V, Piccorossi A, et al. Cold Anaphylaxis in Children: Italian Case Series and Review of the Literature. Diseases. 2023; 11(4):143. https://doi.org/10.3390/diseases11040143
Chicago/Turabian StyleTomei, Leonardo, Francesca Saretta, Stefania Arasi, Lucrezia Sarti, Amelia Licari, Mattia Giovannini, Simona Barni, Giulia Liccioli, Valeria Tallarico, Alessandra Piccorossi, and et al. 2023. "Cold Anaphylaxis in Children: Italian Case Series and Review of the Literature" Diseases 11, no. 4: 143. https://doi.org/10.3390/diseases11040143
APA StyleTomei, L., Saretta, F., Arasi, S., Sarti, L., Licari, A., Giovannini, M., Barni, S., Liccioli, G., Tallarico, V., Piccorossi, A., Caffarelli, C., Novembre, E., & Mori, F. (2023). Cold Anaphylaxis in Children: Italian Case Series and Review of the Literature. Diseases, 11(4), 143. https://doi.org/10.3390/diseases11040143








