Nanocarrier Drug Conjugates Exhibit Potent Anti-Naegleria fowleri and Anti-Balamuthia mandrillaris Properties
Abstract
1. Introduction
2. Materials and Methods
2.1. Nanoparticle Preparation
2.2. Morphology, Hydrodynamic Diameter, and Polydispersity Index (PDI)
2.3. Efficient Drug Entrapment Determination
2.4. HeLa Cells
2.5. Naegleria fowleri Culture
2.6. Balamuthia mandrillaris Culture
2.7. Amoebicidal Assay
2.8. Cytotoxicity Assays
2.9. Cytopathogenicity Experiments
2.10. Statistical Analyses
3. Results
3.1. Effective Preparation of AMPB−Lecithin Nanoformulations
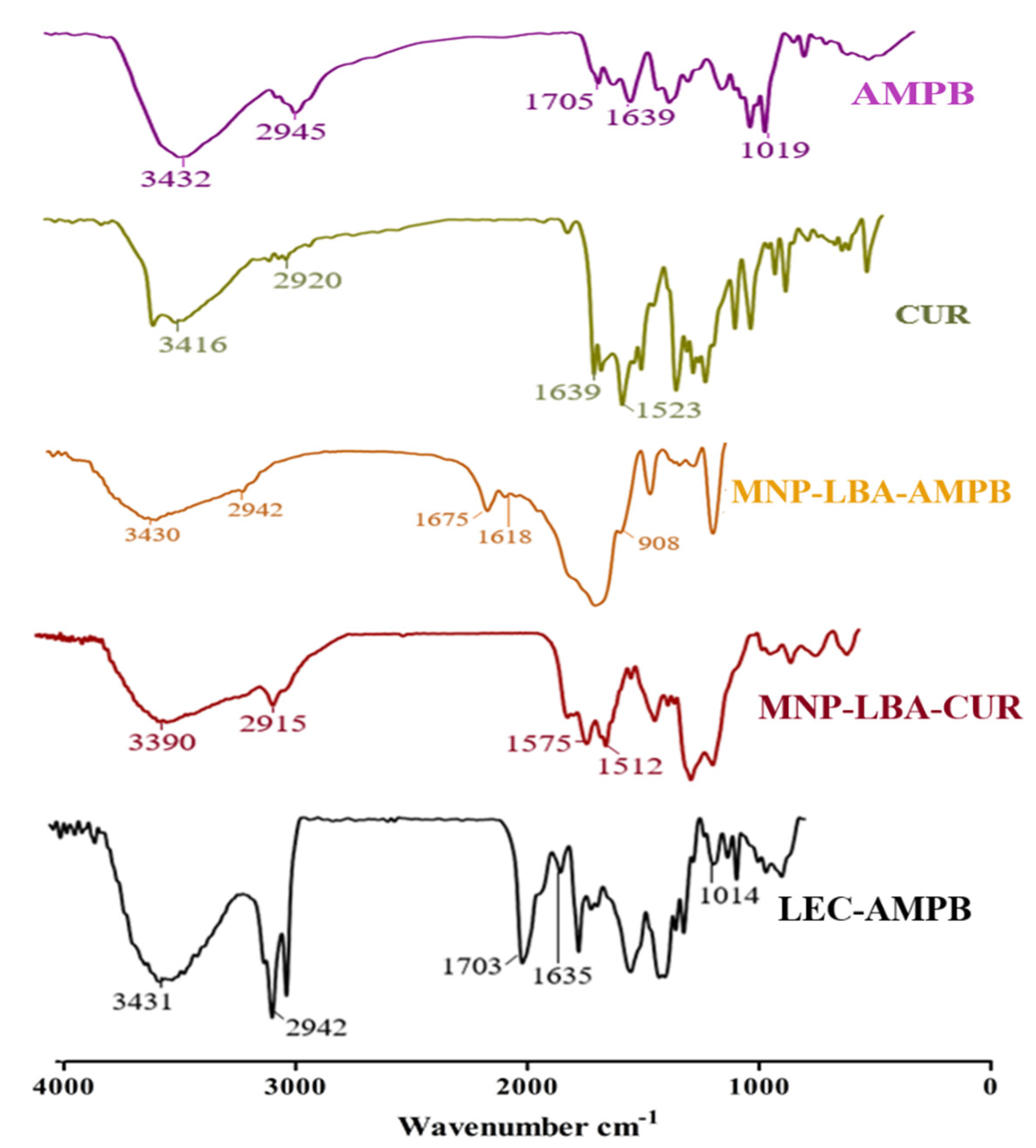
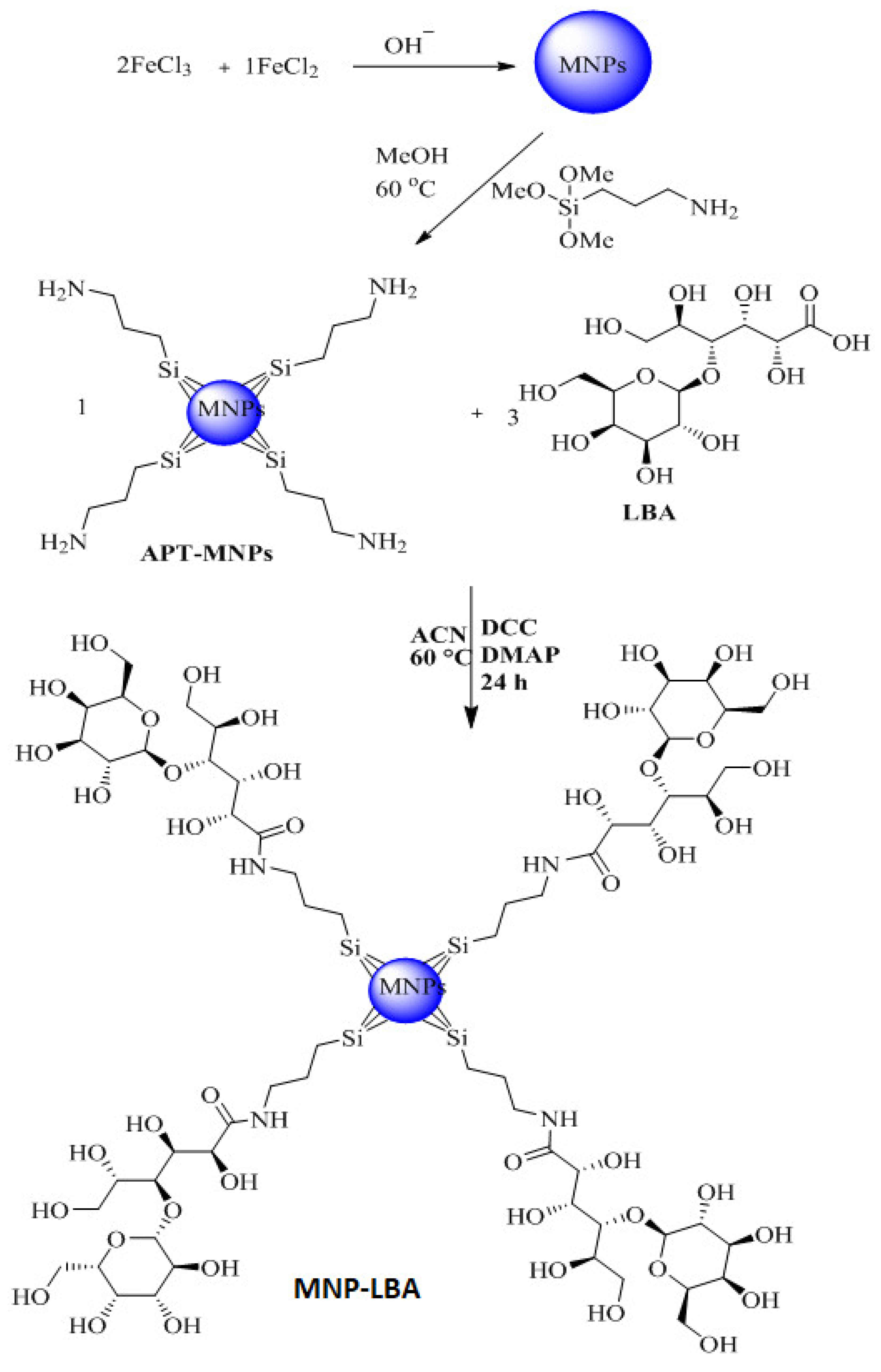
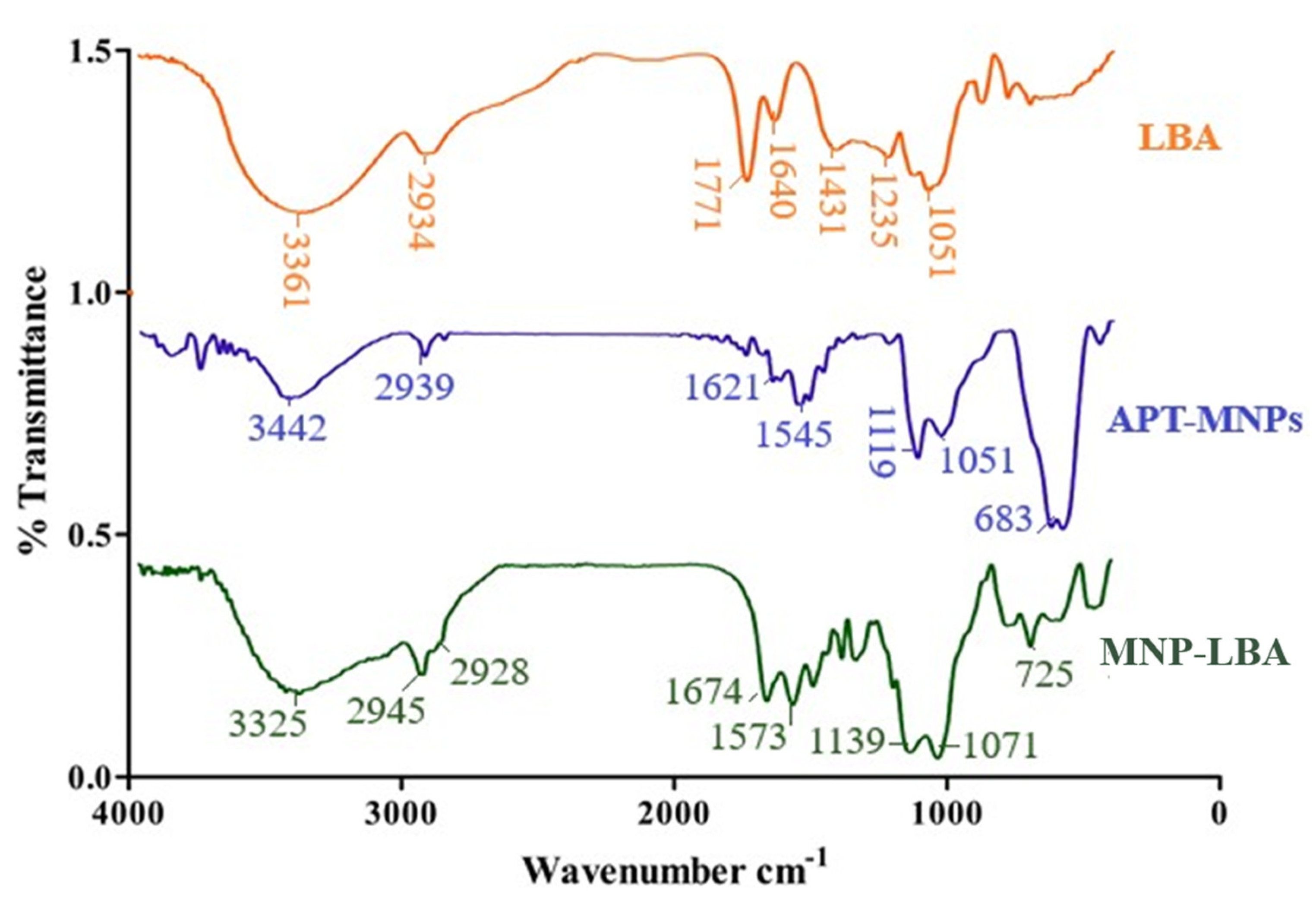
3.2. Effective Formulation of MNP−LBA, MNP−LBA−CUR and MNP−LBA−AMPB
3.3. Nanoformulations Properties
3.4. Drug Entrapment Efficiency
3.5. Nanocarrier-Conjugated Curcumin and Amphotericin B Showed Significant Amoebicidal Effects against Balamuthia mandrillaris and Naegleria fowleri
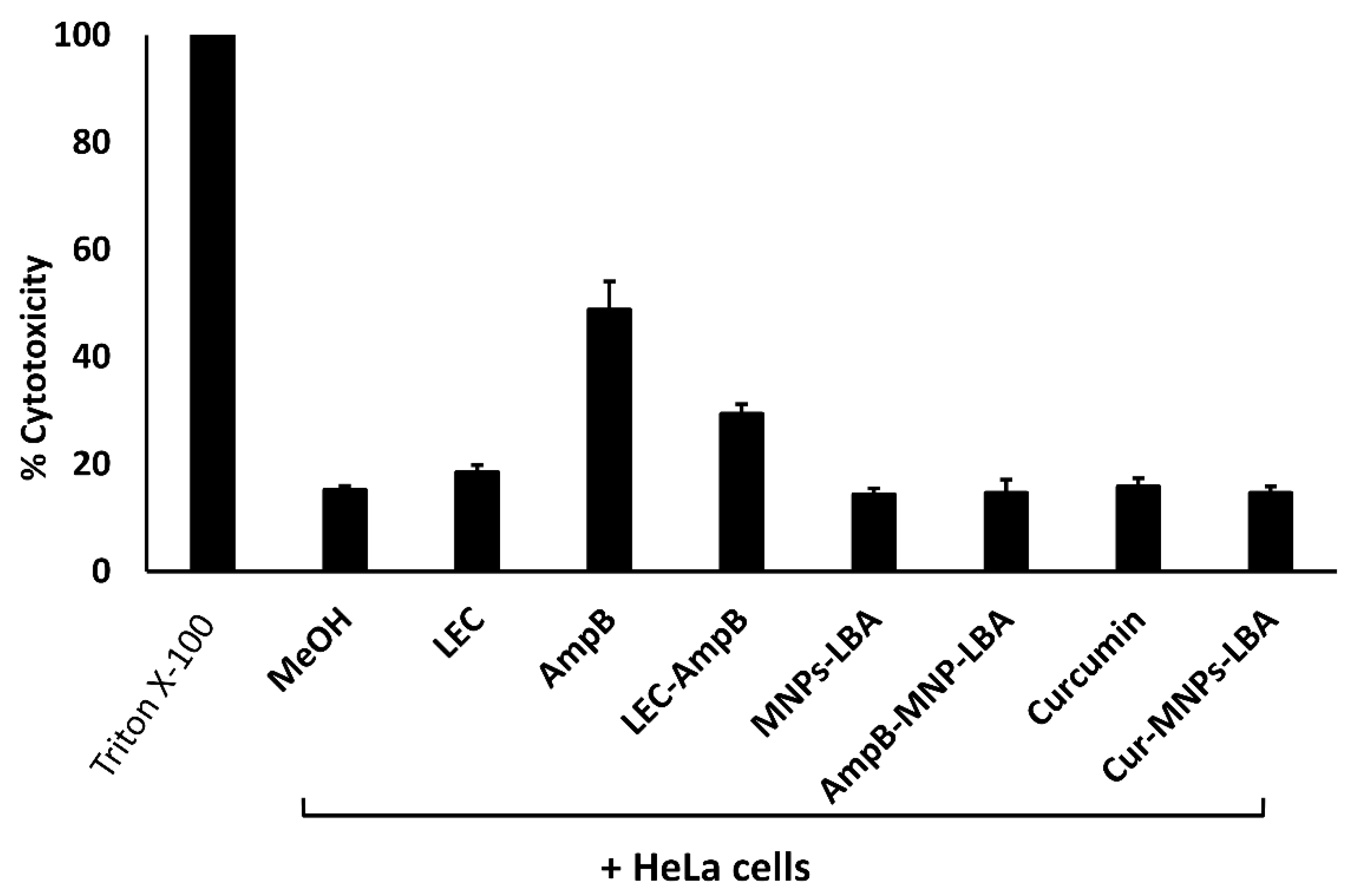
3.6. Nanocarrier-Conjugation Reduced Curcumin and Amphotericin B cytotoxicity to Human Cells
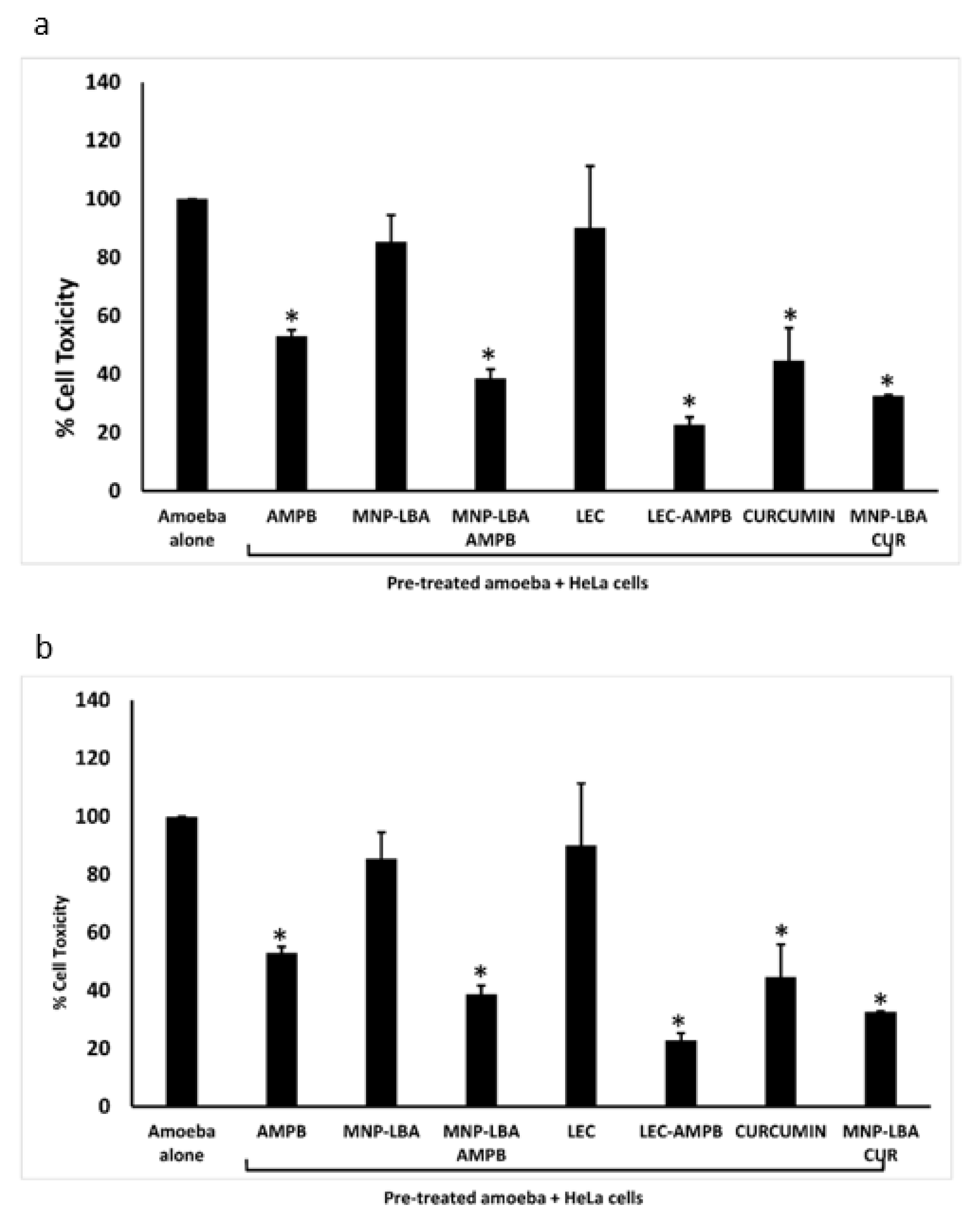
3.7. Nanocarrier-Conjugated Curcumin and Amphotericin B Reduced Human Cell Cytopathogenicity Caused by Balamuthia mandrillaris and Naegleria fowleri
4. Discussion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Jain, A.K.; Thareja, S. In vitro and in vivo characterization of pharmaceutical nanocarriers used for drug delivery. Artif. Cells Nanomed. Biotechnol. 2019, 47, 524–539. [Google Scholar] [CrossRef] [PubMed]
- Masri, A.; Anwar, A.; Khan, N.A.; Siddiqui, R. The use of nanomedicine for targeted therapy against bacterial infections. Antibiotics 2019, 8, 260. [Google Scholar] [CrossRef] [PubMed]
- Taravaud, A.; Fechtali-Moute, Z.; Loiseau, P.M.; Pomel, S. Drugs used for the treatment of cerebral and disseminated infections caused by free-living amoebae. Clin. Transl. Sci. 2021, 14, 791–805. [Google Scholar] [CrossRef]
- Schuster, F.L. Cultivation of pathogenic and opportunistic free-living amebas. Clin. Microbiol. Rev. 2002, 15, 342–354. [Google Scholar] [CrossRef] [PubMed]
- Król-Turmińska, K.; Olender, A. Human infections caused by free-living amoebae. Ann. Agric. Environ. Med. 2017, 24, 254–260. [Google Scholar] [CrossRef]
- Mungroo, M.R.; Anwar, A.; Khan, N.A.; Siddiqui, R. Gold-Conjugated Curcumin as a Novel Therapeutic Agent against Brain-Eating Amoebae. ACS Omega 2020, 5, 12467–12475. [Google Scholar] [CrossRef]
- Matin, A.; Siddiqui, R.; Jayasekera, S.; Khan, N.A. Increasing importance of Balamuthia mandrillaris. Clin. Microbiol. Rev. 2008, 21, 435–448. [Google Scholar] [CrossRef] [PubMed]
- Rajendran, K.; Anwar, A.; Khan, N.A.; Siddiqui, R. Brain-Eating Amoebae: Silver Nanoparticle Conjugation Enhanced Efficacy of Anti-Amoebic Drugs against Naegleria fowleri. ACS Chem Neurosci. 2017, 8, 2626–2630. [Google Scholar] [CrossRef]
- Anwar, A.; Mungroo, M.R.; Khan, S.; Fatima, I.; Rafique, R.; Kanwal; Khan, K.M.; Siddiqui, R.; Khan, N.A. Novel Azoles as Antiparasitic Remedies against Brain-Eating Amoebae. Antibiotics 2020, 9, 188. [Google Scholar] [CrossRef]
- Rodriguez-Anaya, L.Z.; Félix-Sastré, Á.J.; Lares-Villa, F.; Lares-Jiménez, L.F.; Gonzalez-Galaviz, J.R. Application of the omics sciences to the study of Naegleria fowleri, Acanthamoeba spp., and Balamuthia mandrillaris: Current status and future projections. Parasite 2021, 28, 36. [Google Scholar] [CrossRef]
- Siddiqui, R.; Ali, I.K.M.; Cope, J.R.; Khan, N.A. Biology and pathogenesis of Naegleria fowleri. Acta Trop. 2016, 164, 375–394. [Google Scholar] [CrossRef] [PubMed]
- Siddiqui, R.; Khamis, M.; Ibrahim, T.; Khan, N.A. Brain-Eating Amoebae in the United Arab Emirates? ACS Pharmacol. Transl. Sci. 2021, 4, 1014–1015. [Google Scholar] [CrossRef] [PubMed]
- Maciver, S.K.; Piñero, J.E.; Lorenzo-Morales, J. Is Naegleria fowleri an emerging parasite? Trends Parasitol. 2020, 36, 19–28. [Google Scholar] [CrossRef] [PubMed]
- Gondim, B.L.; da Silva Catarino, J.; de Sousa, M.A.D.; de Oliveira Silva, M.; Lemes, M.R.; de Carvalho-Costa, T.M.; de Lima Nascimento, T.R.; Machado, J.R.; Rodrigues, V.; Oliveira, C.J.F.; et al. Nanoparticle-mediated drug delivery: Blood-brain barrier as the main obstacle to treating infectious diseases in CNS. Curr. Pharm. Des. 2019, 25, 3983–3996. [Google Scholar] [CrossRef]
- Siddiqui, R.; Khan, N.A. Contemporary approaches to treat Naegleria fowleri: A patent overview. Pharm. Pat. Anal. 2020, 10, 99–101. [Google Scholar] [CrossRef]
- Siddiqui, R.; Khan, N.A. Current treatment options of Balamuthia mandrillaris: A patent overview. Pharm. Pat. Anal. 2020, 9, 121–123. [Google Scholar] [CrossRef]
- Grace, E.; Asbill, S.; Virga, K. Naegleria fowleri: Pathogenesis, diagnosis, and treatment options. Antimicrob. Agents Chemother. 2015, 59, 6677–6681. [Google Scholar] [CrossRef]
- Rice, C.A.; Colon, B.L.; Chen, E.; Hull, M.V.; Kyle, D.E. Discovery of repurposing drug candidates for the treatment of diseases caused by pathogenic free-living amoebae. PLoS Negl. Trop. Dis. 2020, 14, e0008353. [Google Scholar] [CrossRef]
- Trabelsi, H.; Dendana, F.; Sellami, A.; Sellami, H.; Cheikhrouhou, F.; Neji, S.; Makni, F.; Ayadi, A. Pathogenic free-living amoebae: Epidemiology and clinical review. Pathol. Biol. 2012, 60, 399–405. [Google Scholar] [CrossRef]
- Rajendran, K.; Anwar, A.; Khan, N.A.; Shah, M.R.; Siddiqui, R. trans-Cinnamic acid conjugated gold nanoparticles as potent therapeutics against brain-eating amoeba Naegleria fowleri. ACS Chem. Neurosci. 2019, 10, 2692–2696. [Google Scholar] [CrossRef]
- Anwar, A.; Masri, A.; Rao, K.; Rajendran, K.; Khan, N.A.; Shah, M.R.; Siddiqui, R. Antimicrobial activities of green synthesized gums-stabilized nanoparticles loaded with flavonoids. Sci. Rep. 2019, 9, 3122. [Google Scholar] [CrossRef] [PubMed]
- Amiri, H.; Bordonali, L.; Lascialfari, A.; Wan, S.; Monopoli, M.P.; Lynch, I.; Laurent, S.; Mahmoudi, M. Protein corona affects the relaxivity and MRI contrast efficiency of magnetic nanoparticles. Nanoscale 2013, 5, 8656–8665. [Google Scholar] [CrossRef] [PubMed]
- Lemke, A.; Kiderlen, A.F.; Petri, B.; Kayser, O. Delivery of amphotericin B nanosuspensions to the brain and determination of activity against Balamuthia mandrillaris amebas. Nanomed. Nanotechnol. Biol. Med. 2010, 6, 597–603. [Google Scholar] [CrossRef] [PubMed]
- Akbar, N.; Kawish, M.; Khan, N.A.; Shah, M.R.; Alharbi, A.M.; Alfahemi, H.; Siddiqui, R. Hesperidin-, Curcumin-, and Amphotericin B-Based Nano-Formulations as Potential Antibacterials. Antibiotics 2022, 11, 696. [Google Scholar] [CrossRef]
- Javed, I.; Hussain, S.Z.; Shahzad, A.; Khan, J.M.; Rehman, M.; Usman, F.; Razi, M.T.; Shah, M.R.; Hussain, I. Lecithin-gold hybrid nanocarriers as efficient and pH selective vehicles for oral delivery of diacerein—In-vitro and in-vivo study. Colloids Surf. B Biointerfaces 2016, 141, 1–9. [Google Scholar] [CrossRef]
- Kawish, M.; Jabri, T.; Elhissi, A.; Zahid, H.; Muhammad Iqbal, K.; Rao, K.; Gul, J.; Abdullah, M.; Shah, M.R. Galactosylated iron oxide nanoparticles for enhancing oral bioavailability of ceftriaxone. Pharm. Dev. Technol. 2021, 26, 291–301. [Google Scholar] [CrossRef]
- Akbar, N.; Siddiqui, R.; Iqbal, M.; Khan, N.A. Antibacterial activities of selected pure compounds isolated from gut bacteria of animals living in polluted environments. Antibiotics 2020, 9, 190. [Google Scholar] [CrossRef]
- Aqeel, Y.; Iqbal, J.; Siddiqui, R.; Gilani, A.H.; Khan, N.A. Anti-Acanthamoebic properties of resveratrol and demethoxycurcumin. Exp. Parasitol. 2012, 132, 519–523. [Google Scholar] [CrossRef]
- Jeyamogan, S.; Khan, N.A.; Anwar, A.; Shah, M.R.; Siddiqui, R. Cytotoxic effects of Benzodioxane, Naphthalene diimide, Porphyrin and Acetamol derivatives on HeLa cells. SAGE Open Med. 2018, 6, 2050312118781962. [Google Scholar] [CrossRef]
- Jabri, T.; Imran, M.; Rao, K.; Ali, I.; Arfan, M.; Shah, M.R. Fabrication of lecithin-gum tragacanth muco-adhesive hybrid nano-carrier system for in-vivo performance of Amphotericin B. Carbohydr. Polym. 2018, 194, 89–96. [Google Scholar] [CrossRef]
- Costa, V.M.; De Souza, M.C.M.; Fechine, P.B.A.; Macedo, A.C.; Gonçalves, L.R.B. Nanobiocatalytic systems based on lipase-Fe3O4 and conventional systems for isoniazid synthesis: A comparative study. Braz. J. Chem. Eng. 2016, 33, 661–673. [Google Scholar] [CrossRef]
- Pan, Q.; Lv, Y.; Williams, G.R.; Tao, L.; Yang, H.; Li, H.; Zhu, L. Lactobionic acid and carboxymethyl chitosan functionalized graphene oxide nanocomposites as targeted anticancer drug delivery systems. Carbohydr. Polym. 2016, 151, 812–820. [Google Scholar] [CrossRef] [PubMed]
- Sun, B.; Tian, Y.; Chen, L.; Jin, Z. Linear dextrin as curcumin delivery system: Effect of degree of polymerization on the functional stability of curcumin. Food Hydrocoll. 2018, 77, 911–920. [Google Scholar] [CrossRef]
- Stewart, M.; Bartholomew, B.; Currie, F.; Abbiw, D.K.; Latif, Z.; Sarker, S.D.; Nash, R.J. Pyranoisoflavones from Rinoreawelwitschii. Fitoterapia 2000, 71, 595–597. [Google Scholar] [CrossRef] [PubMed]
- Kong, Z.-L.; Kuo, H.-P.; Johnson, A.; Wu, L.-C.; Chang, K.L.B. Curcumin-Loaded Mesoporous Silica Nanoparticles Markedly Enhanced Cytotoxicity in Hepatocellular Carcinoma Cells. Int. J. Mol. Sci. 2019, 20, 2918. [Google Scholar] [CrossRef]
- Kawish, M.; Elhissi, A.; Jabri, T.; Muhammad Iqbal, K.; Zahid, H.; Shah, M.R. Enhancement in oral absorption of ceftriaxone by highly functionalized magnetic iron oxide nanoparticles. Pharmaceutics 2020, 12, 492. [Google Scholar] [CrossRef]
- Kaasalainen, M.; Aseyev, V.; von Haartman, E.; Karaman, D.Ş.; Mäkilä, E.; Tenhu, H.; Rosenholm, J.; Salonen, J. Size, stability, and porosity of mesoporous nanoparticles characterized with light scattering. Nanoscale Res. Lett. 2017, 12, 74. [Google Scholar] [CrossRef]
- ISO 10993-5 2009; Biological Evaluation of Medical Devices-Part 5: Tests for In Vitro Cytotoxicity. International Organization for Standardization: Geneva, Switzerland, 2009.
- Visvesvara, G.S.; Moura, H.; Schuster, F.L. Pathogenic and opportunistic free-living amoebae: Acanthamoeba spp., Balamuthia mandrillaris, Naegleria fowleri, and Sappiniadiploidea. FEMS Immunol. Med. Microbiol. 2007, 50, 1–26. [Google Scholar] [CrossRef]
- Ong, T.Y.Y.; Khan, N.A.; Siddiqui, R. Brain-eating amoebae: Predilection sites in the brain and disease outcome. J. Clin. Microbiol. 2017, 55, 1989–1997. [Google Scholar] [CrossRef]
- Bhosale, N.K.; Parija, S.C. Balamuthia mandrillaris: An opportunistic, free-living ameba–An updated review. Trop. Parasitol. 2021, 11, 78–88. [Google Scholar]
- Debnath, A. Drug discovery for primary amebic meningoencephalitis: From screen to identification of leads. Expert Rev. Anti-Infect. Ther. 2021, 19, 1099–1106. [Google Scholar] [CrossRef] [PubMed]
- Basniwal, R.K.; Buttar, H.S.; Jain, V.K.; Jain, N. Curcumin nanoparticles: Preparation, characterization, and antimicrobial study. J. Agric. Food Chem. 2011, 59, 2056–2061. [Google Scholar]
- Saeed, B.Q.; Hussain, K.; Akbar, N.; Khan, H.; Siddiqui, R.; Shah, R.M.; Khan, N.A. Nanovesicles containing curcumin hold promise in the development of new formulations of anti-Acanthamoebic agents. Mol. Biochem. Parasitol. 2022, 247, 111430. [Google Scholar] [CrossRef] [PubMed]
- Cuoco, J.A.; Klein, B.J.; LeBel, D.P.; Faulhaber, J.; Apfel, L.S.; Witcher, M.R. Successful Treatment of a Balamuthia mandrillaris Cerebral Abscess in a Pediatric Patient with Complete Surgical Resection and Antimicrobial Therapy. Pediatr. Infect. Dis. J. 2022, 41, e54–e57. [Google Scholar] [CrossRef]
- Farnon, E.C.; Kokko, K.E.; Budge, P.J.; Mbaeyi, C.; Lutterloh, E.C.; Qvarnstrom, Y.; da Silva, A.J.; Shieh, W.J.; Roy, S.L.; Paddock, C.D.; et al. Transmission of Balamuthia mandrillaris by organ transplantation. Clin. Infect. Dis. 2016, 63, 878–888. [Google Scholar] [CrossRef]
- Wang, L.; Li, B.; Zhao, T.; Wang, L.; Jian, Z.; Cheng, W.; Chen, J.; Li, C.; Wang, G.; Gao, T. Treatment of cutaneous Balamuthia mandrillaris infection with diminazene aceturate: A report of 4 cases. Clin. Infect. Dis. 2022, 75, 1637–1640. [Google Scholar] [CrossRef]

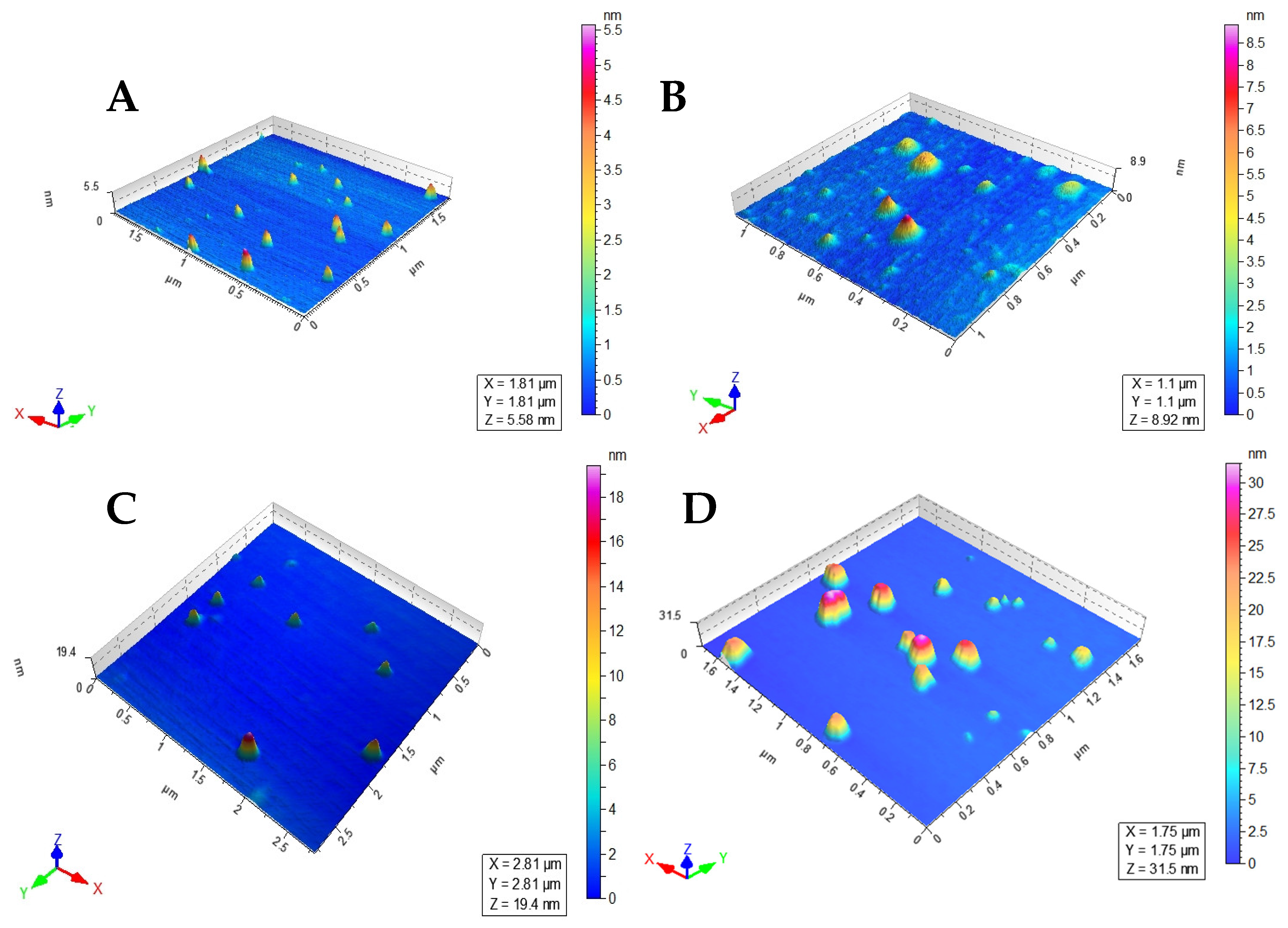

| Nanoparticles | Average Size (nm) | PDI |
|---|---|---|
| LEC−AmpB | 144 ± 16.2 | 0.26 ± 0.050 |
| MNP−LBA | 171.9 ± 2.23 | 0.24 ± 0.015 |
| MN−-LBA−CUR | 230.1 ± 3.13 | 0.32 ± 0.040 |
| MNP−LBA−AMPB | 189.7 ± 7.89 | 0.35 ± 0.020 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Siddiqui, R.; Boghossian, A.; Kawish, M.; Jabri, T.; Shah, M.R.; Anuar, T.S.; Al-Shareef, Z.; Khan, N.A. Nanocarrier Drug Conjugates Exhibit Potent Anti-Naegleria fowleri and Anti-Balamuthia mandrillaris Properties. Diseases 2023, 11, 58. https://doi.org/10.3390/diseases11020058
Siddiqui R, Boghossian A, Kawish M, Jabri T, Shah MR, Anuar TS, Al-Shareef Z, Khan NA. Nanocarrier Drug Conjugates Exhibit Potent Anti-Naegleria fowleri and Anti-Balamuthia mandrillaris Properties. Diseases. 2023; 11(2):58. https://doi.org/10.3390/diseases11020058
Chicago/Turabian StyleSiddiqui, Ruqaiyyah, Anania Boghossian, Muhammad Kawish, Tooba Jabri, Muhammad Raza Shah, Tengku Shahrul Anuar, Zainab Al-Shareef, and Naveed Ahmed Khan. 2023. "Nanocarrier Drug Conjugates Exhibit Potent Anti-Naegleria fowleri and Anti-Balamuthia mandrillaris Properties" Diseases 11, no. 2: 58. https://doi.org/10.3390/diseases11020058
APA StyleSiddiqui, R., Boghossian, A., Kawish, M., Jabri, T., Shah, M. R., Anuar, T. S., Al-Shareef, Z., & Khan, N. A. (2023). Nanocarrier Drug Conjugates Exhibit Potent Anti-Naegleria fowleri and Anti-Balamuthia mandrillaris Properties. Diseases, 11(2), 58. https://doi.org/10.3390/diseases11020058








