Human Rabies by Secondary Transmission in Argentina, 2021
Abstract
:1. Introduction
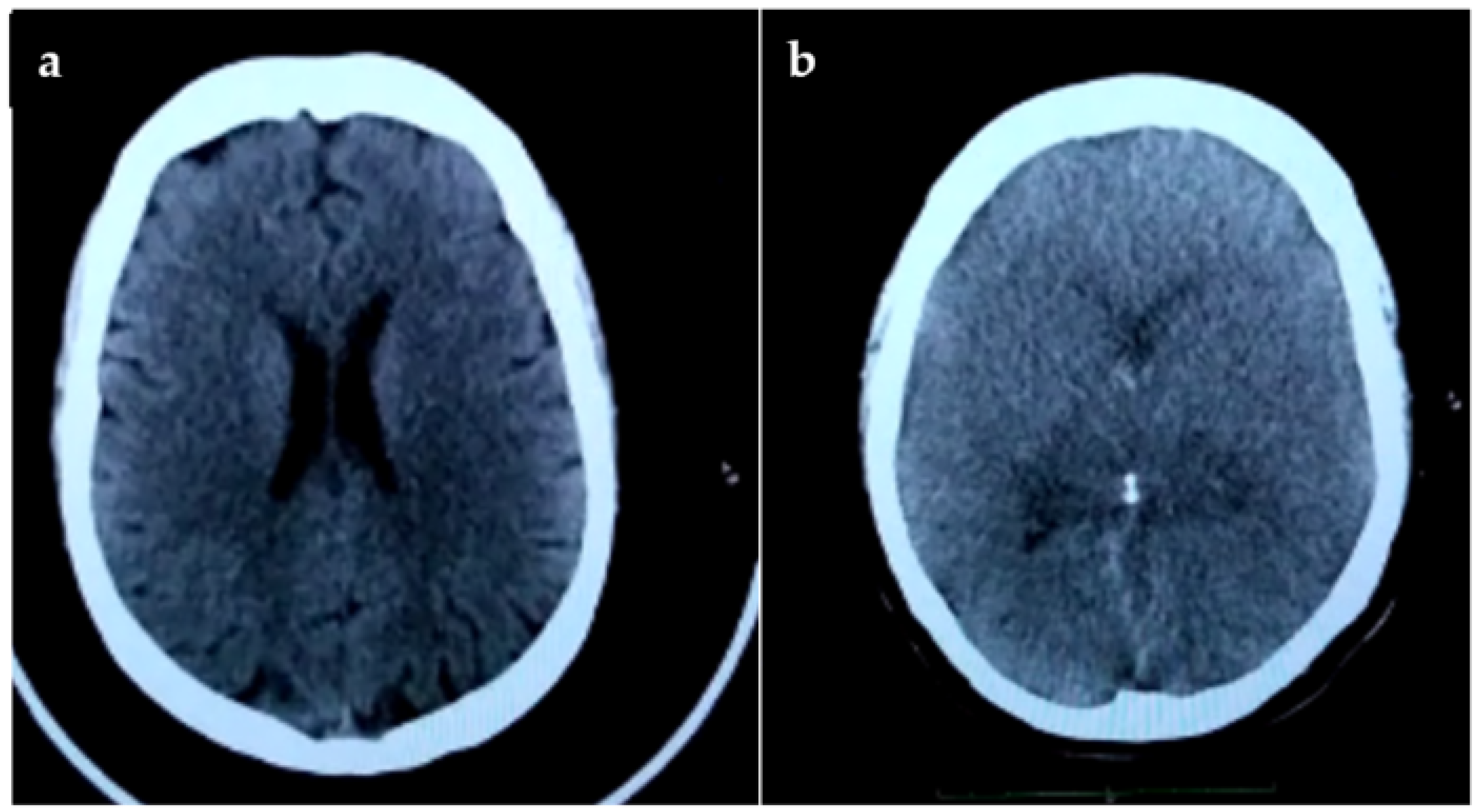
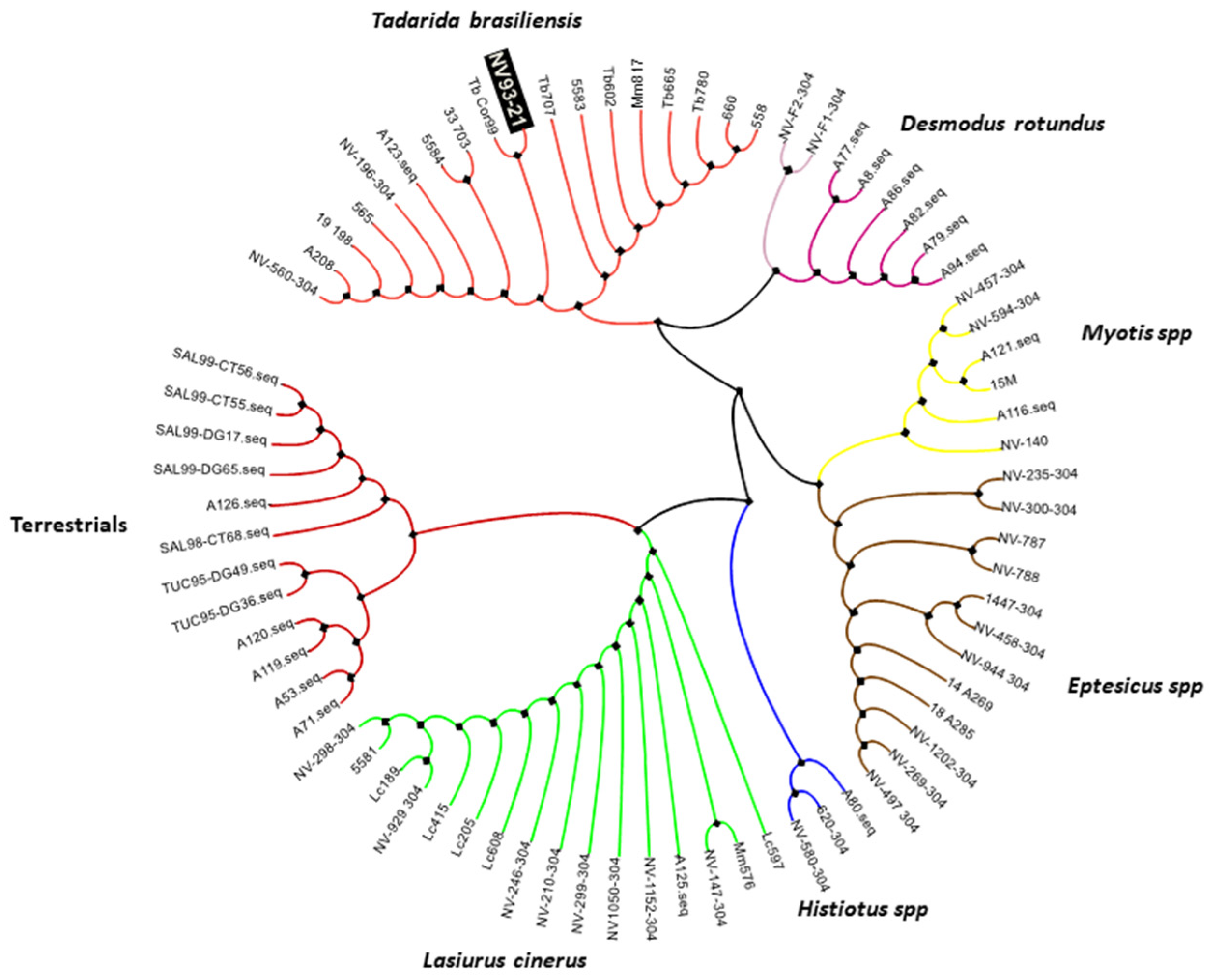
2. Case Report
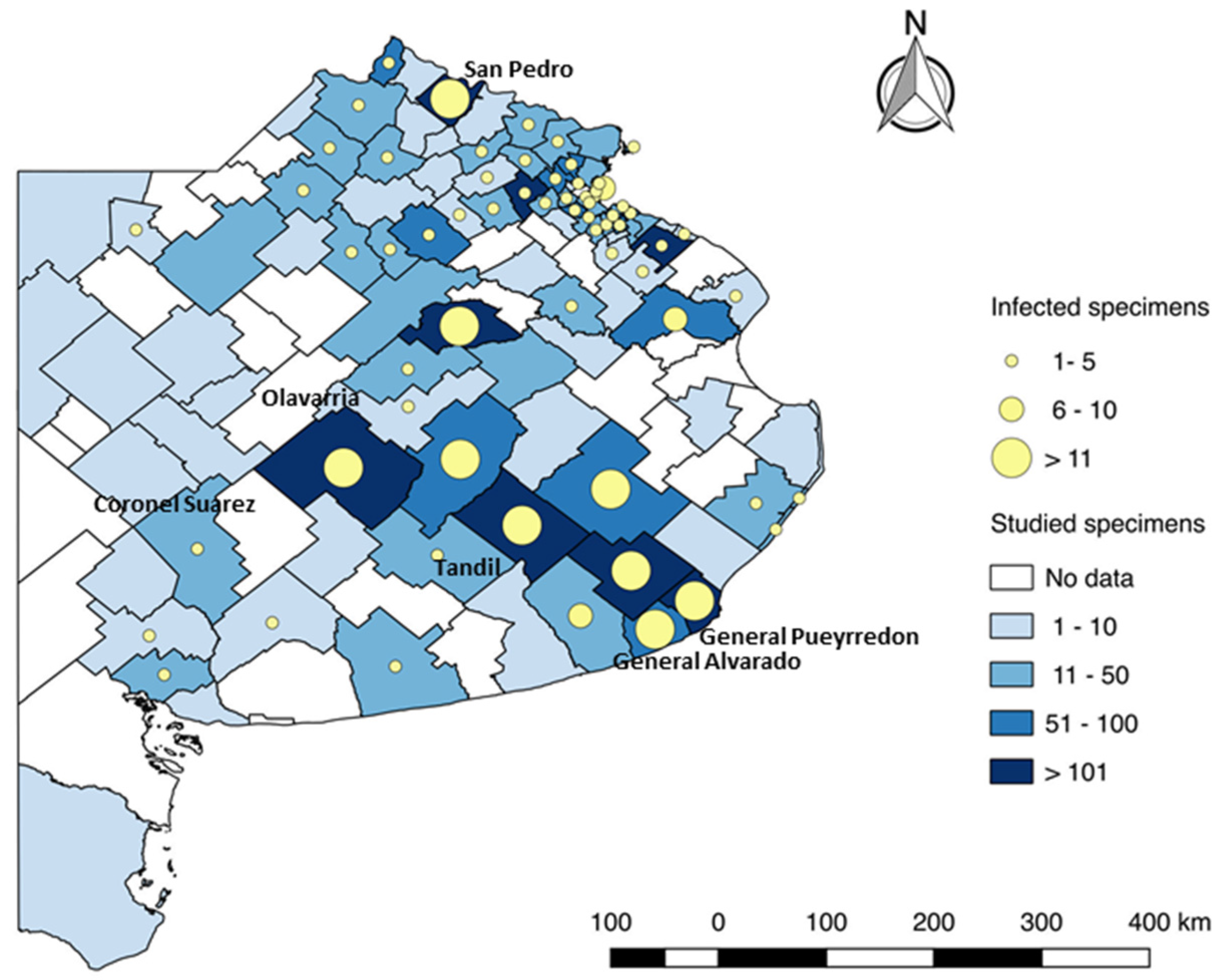
3. Discussion
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Markotter, W.; Coertse, J. Bat lyssaviruses. Rev. Sci. Tech. 2018, 37, 385–400. [Google Scholar] [CrossRef] [PubMed]
- Oliveira, R.N.; Freire, C.C.; Iamarino, A.; Zanotto, P.M.; Pessoa, R.; Sanabani, S.S.; De Souza, S.P.; Castilho, J.G.; Batista, H.B.C.R.; Carnieli, P., Jr.; et al. Rabies virus diversification in aerial and terrestrial mammals. Genet. Mol. Biol. 2020, 43, e20190370. [Google Scholar] [CrossRef] [PubMed]
- Fisher, C.R.; Streicker, D.G.; Schnell, M.J. The spread and evolution of rabies virus: Conquering new frontiers. Nat. Rev. Microbiol. 2018, 16, 241–255. [Google Scholar] [CrossRef] [PubMed]
- Dato, V.M.; Campagnolo, E.; Long, J.; Rupprecht, C.E. A Systematic Review of Human Bat Rabies Virus Variant Cases: Evaluating Unprotected Physical Contact with Claws and Teeth in Support of Accurate Risk Assessments. PLoS ONE 2016, 11, e0159443. [Google Scholar] [CrossRef] [PubMed]
- Ministerio de Salud. Caso de rabia humana variante murciélago: Provincia de Buenos Aires. In Alerta Epidemiologica; Ministerio de Salud: Buenos Aires, Argentina, 2021; p. 10. [Google Scholar]
- Wadhwa, A.; Wilkins, K.; Gao, J.; Condori, R.E.C.; Gigante, C.M.; Zhao, H.; Ma, X.; Ellison, J.A.; Greenberg, L.; Velasco-Villa, A.; et al. A Pan-Lyssavirus Taqman Real-Time RT-PCR Assay for the Detection of Highly Variable Rabies virus and Other Lyssaviruses. PLoS Negl. Trop. Dis. 2017, 11, e0005258. [Google Scholar] [CrossRef] [PubMed]
- WHO. Diagnosis. In WHO Expert Consultation on Rabies; WHO Press, World Health Organization: Geneva, Switzerland, 2018; pp. 21–36. [Google Scholar]
- Pinero, C.; Dohmen, F.G.; Beltran, F.; Martinez, L.; Novaro, L.; Russo, S.; Palacios, G.; Cisterna, D.M. High diversity of rabies viruses associated with insectivorous bats in Argentina: Presence of several independent enzootics. PLoS Negl. Trop. Dis. 2012, 6, e1635. [Google Scholar] [CrossRef] [PubMed]
- Ministerio de Salud de la Provincia de Buenos Aires. Informe sobre situación del programa provincial del control de la rabia en pandemia COVID-19. In Boletin Epidemiologico; Ministerio de Salud Provincia de Buenos Aires: Buenos Aires, Argentina, 2020; p. 7. [Google Scholar]
- Kotait, I.; Carrieri, M.L.; Rupprecht, C. Secondary transmission of rabies in Latin America. Rev. Educ. Cont. Med. Vet. Zootec. 2012, 10, 48–49. [Google Scholar]
- Sanchez, M.P.; Sanmiguel, R.A.; Díaz Sanchez, O.A.; Ramirez, A.A.; Escobar, L. Rabies in the Americas, various challenges and «One Health»: Review article. Rev. Inv. Vet. Perú. 2019, 30, 1361–1381. [Google Scholar]
- Brunt, S.; Solomon, H.; Brown, K.; Davis, A. Feline and Canine Rabies in New York State, USA. Viruses 2021, 13, 450. [Google Scholar] [CrossRef] [PubMed]
- Castilho, J.G.; Achkar, S.M.; Oliveira, R.D.N.; Mori, E.; Carnieli, P.; Macedo, C.I. Analysis of rabies diagnosis in dogs and cats in the state of Sao Paulo, Brazil. Arch. Virol. 2018, 163, 2369–2376. [Google Scholar] [CrossRef] [PubMed]
- Khayat, R.O.S.; Grant, R.A.; Ryan, H.; Melling, L.M.; Dougill, G.; Killick, D.R.; Shaw, K.J. Investigating cat predation as the cause of bat wing tears using forensic DNA analysis. Ecol. Evol. 2020, 10, 8368–8378. [Google Scholar] [CrossRef] [PubMed]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Firpo, S.; Piccirilli, M.G.; Urizar, R.; Vitta, N.; Hirmas Riade, S.M.; Leguizamón, C.; Vico, M.L.; Martínez, G.; Beltrán, F.J.; Cisterna, D.M. Human Rabies by Secondary Transmission in Argentina, 2021. Diseases 2022, 10, 17. https://doi.org/10.3390/diseases10010017
Firpo S, Piccirilli MG, Urizar R, Vitta N, Hirmas Riade SM, Leguizamón C, Vico ML, Martínez G, Beltrán FJ, Cisterna DM. Human Rabies by Secondary Transmission in Argentina, 2021. Diseases. 2022; 10(1):17. https://doi.org/10.3390/diseases10010017
Chicago/Turabian StyleFirpo, Soledad, María Guadalupe Piccirilli, Rogelio Urizar, Nicolas Vitta, Stella Maris Hirmas Riade, Constanza Leguizamón, María Lorena Vico, Gustavo Martínez, Fernando J. Beltrán, and Daniel M. Cisterna. 2022. "Human Rabies by Secondary Transmission in Argentina, 2021" Diseases 10, no. 1: 17. https://doi.org/10.3390/diseases10010017
APA StyleFirpo, S., Piccirilli, M. G., Urizar, R., Vitta, N., Hirmas Riade, S. M., Leguizamón, C., Vico, M. L., Martínez, G., Beltrán, F. J., & Cisterna, D. M. (2022). Human Rabies by Secondary Transmission in Argentina, 2021. Diseases, 10(1), 17. https://doi.org/10.3390/diseases10010017






