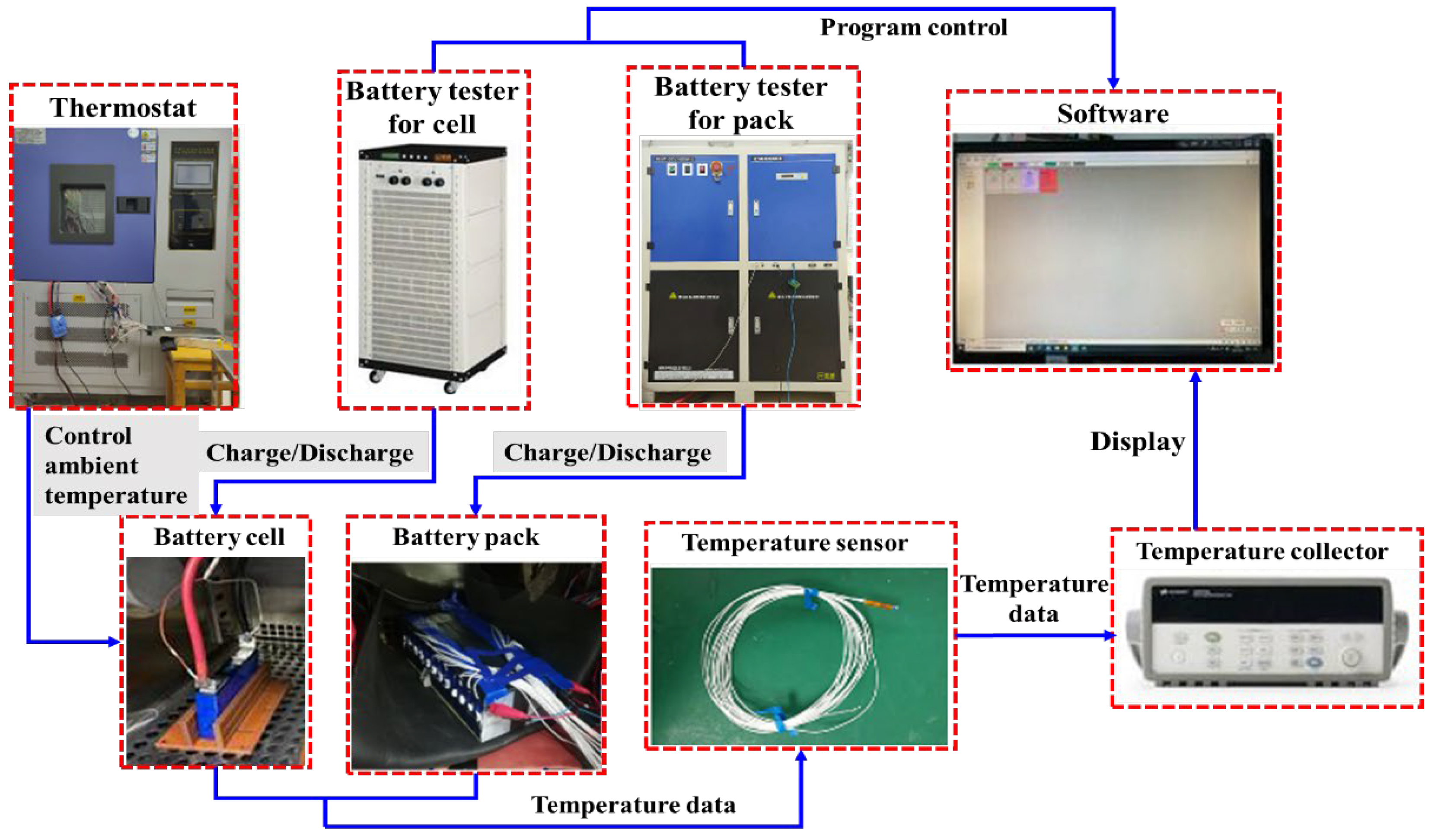
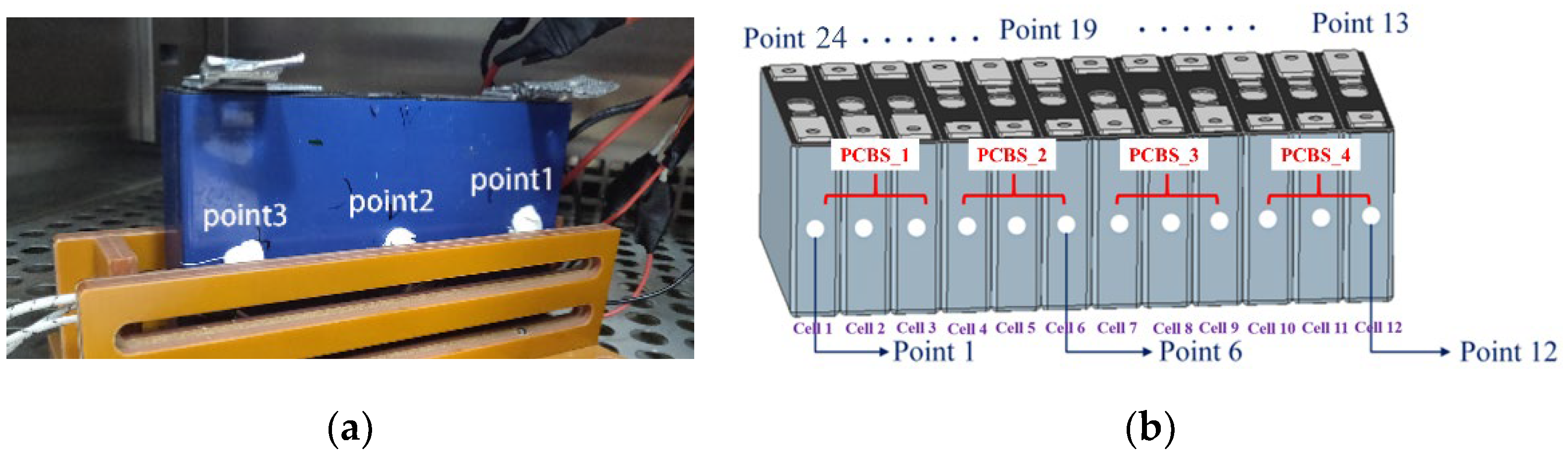
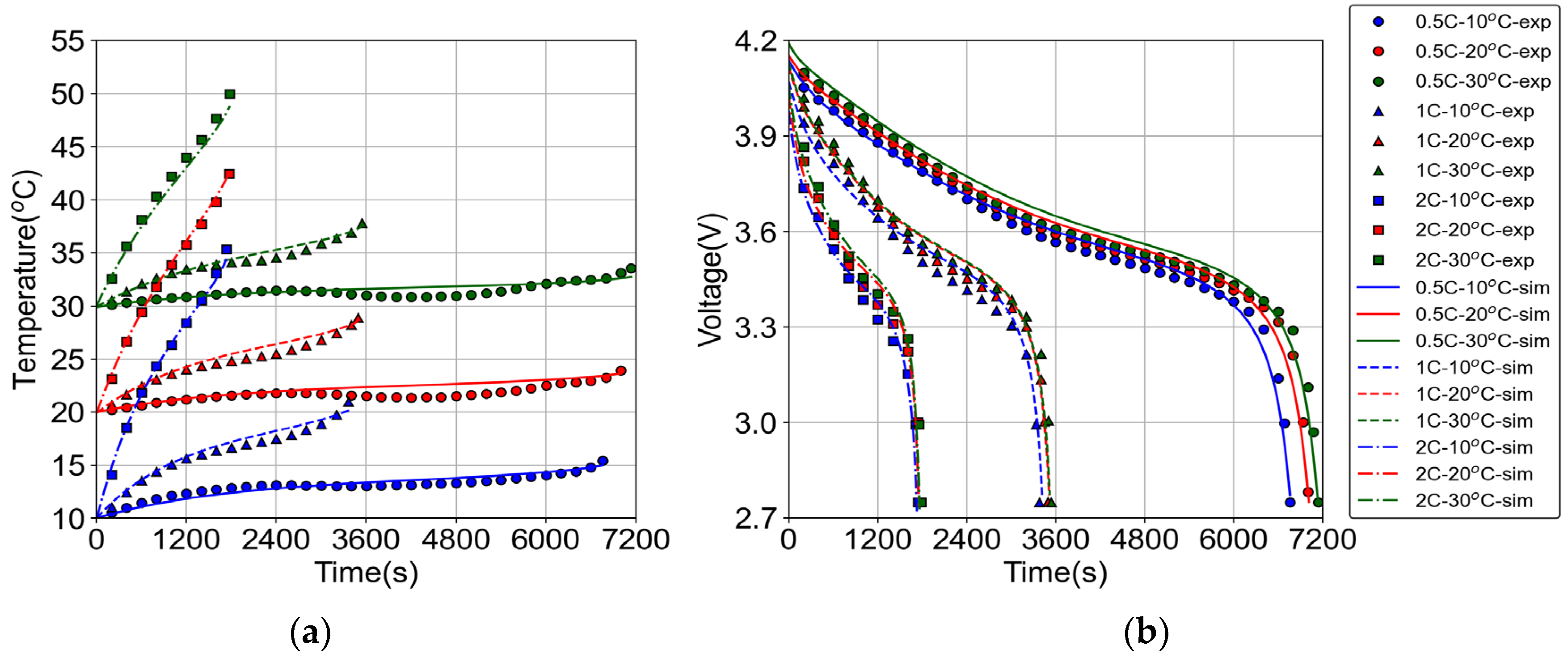
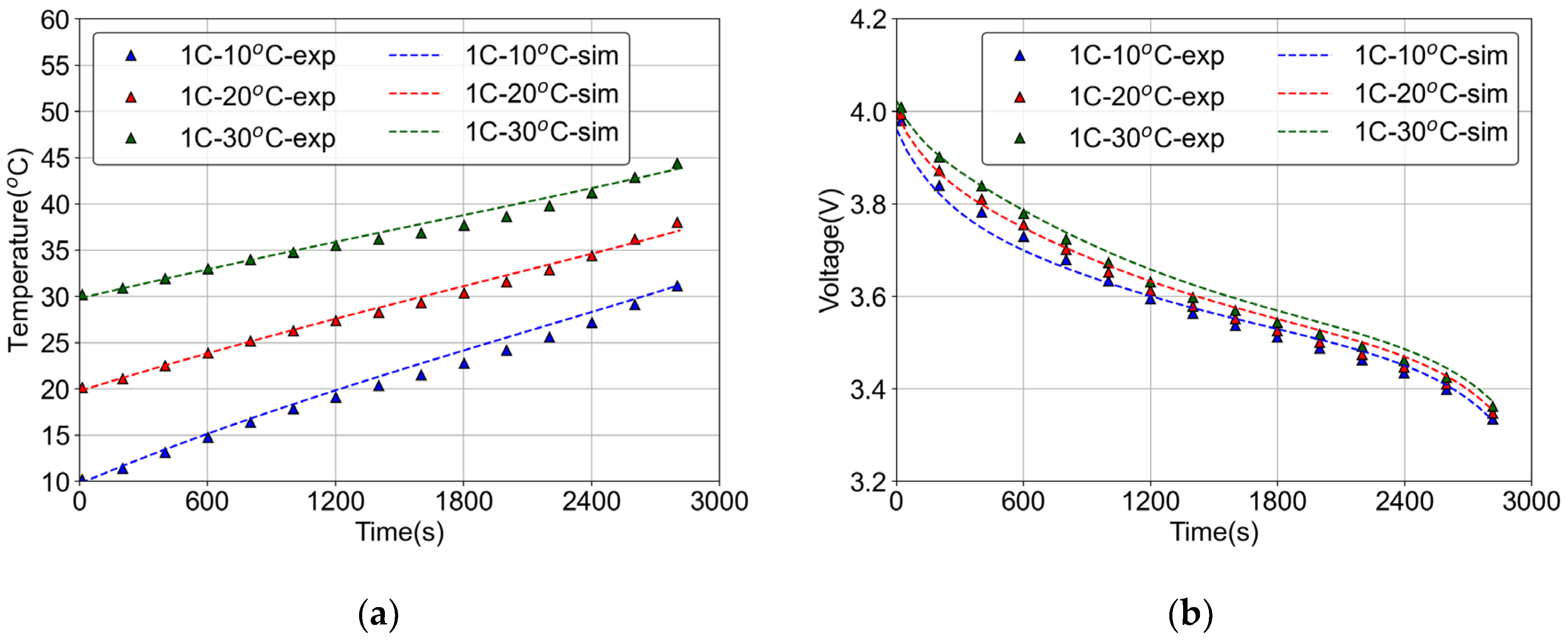
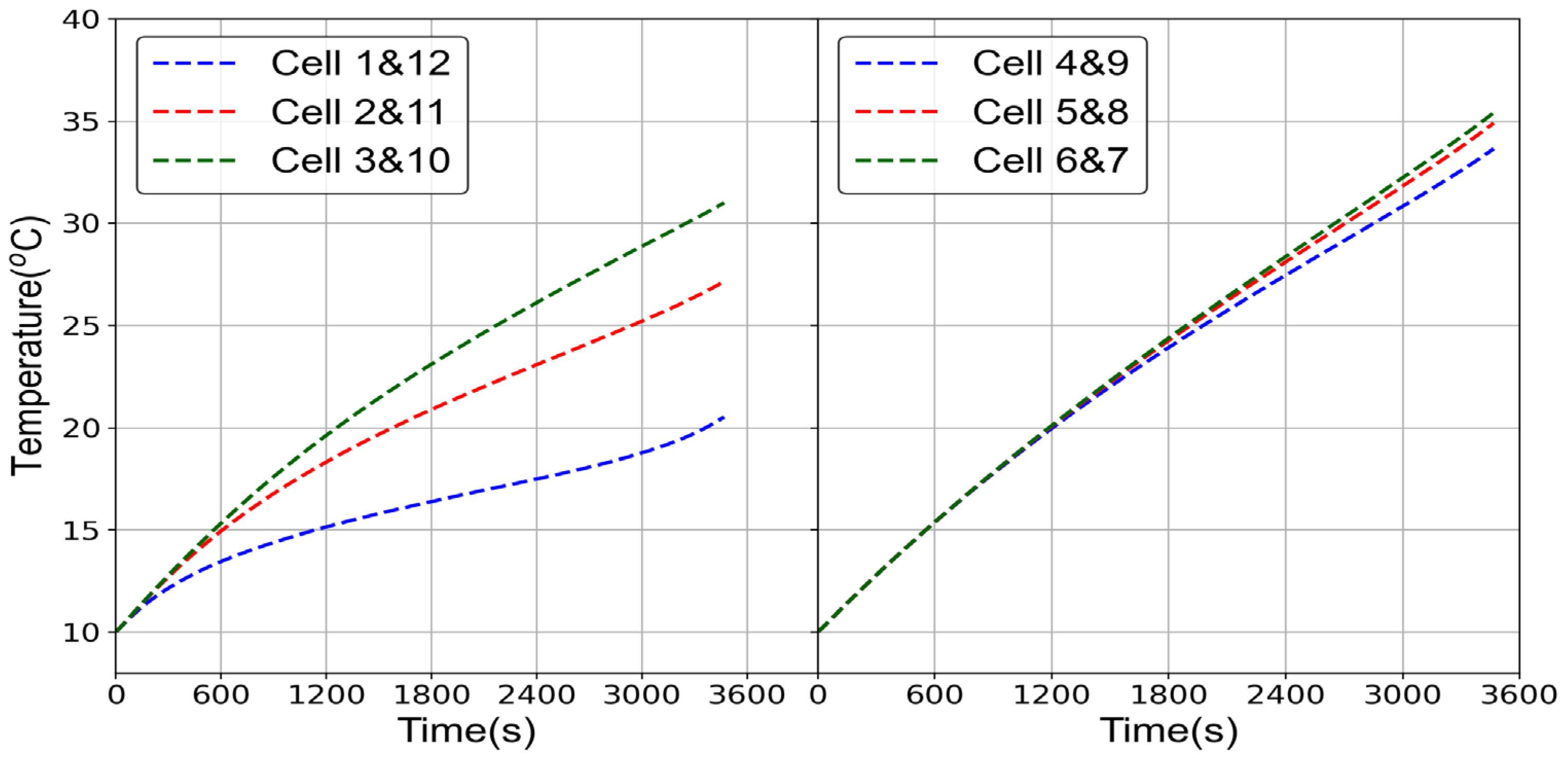
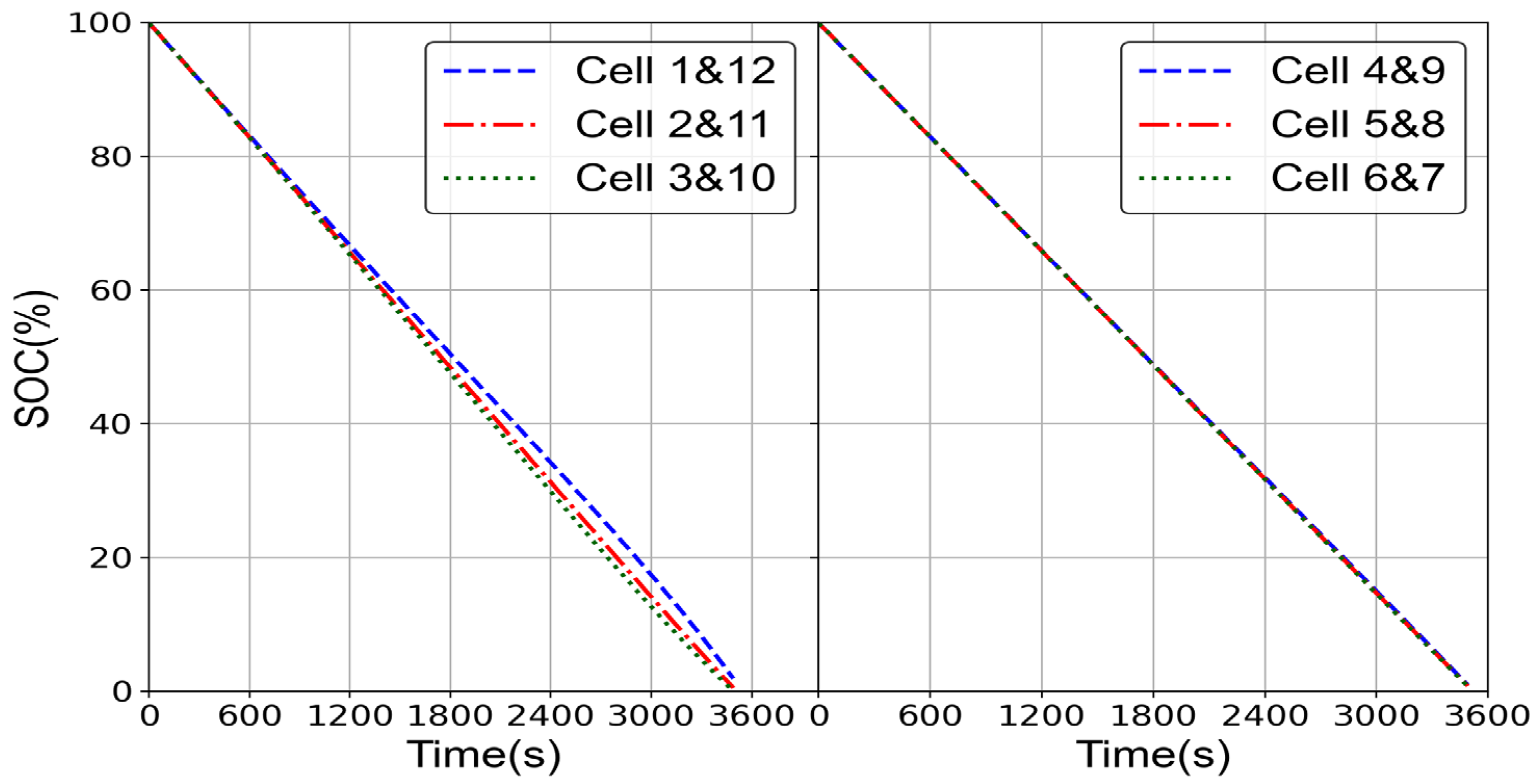
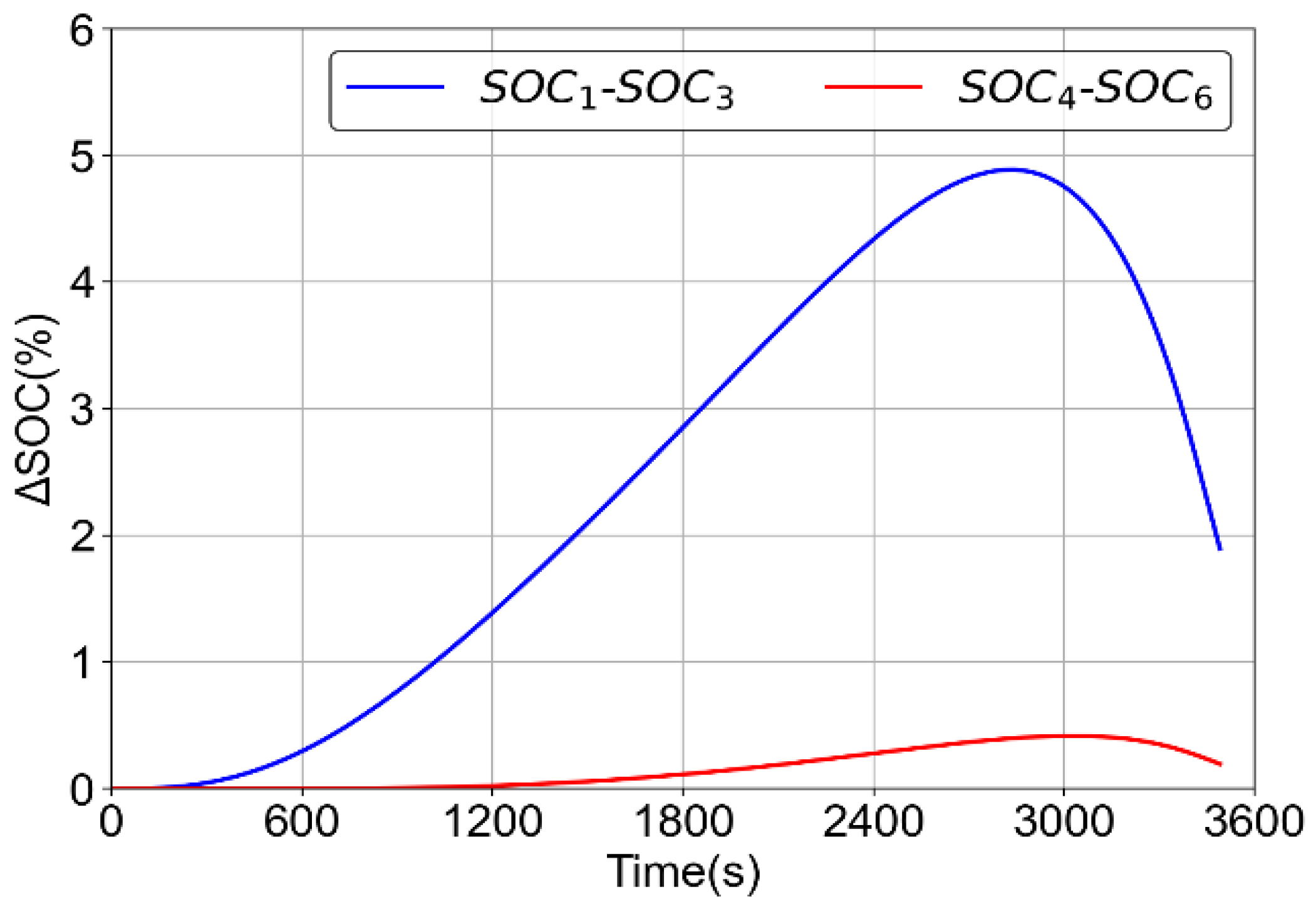
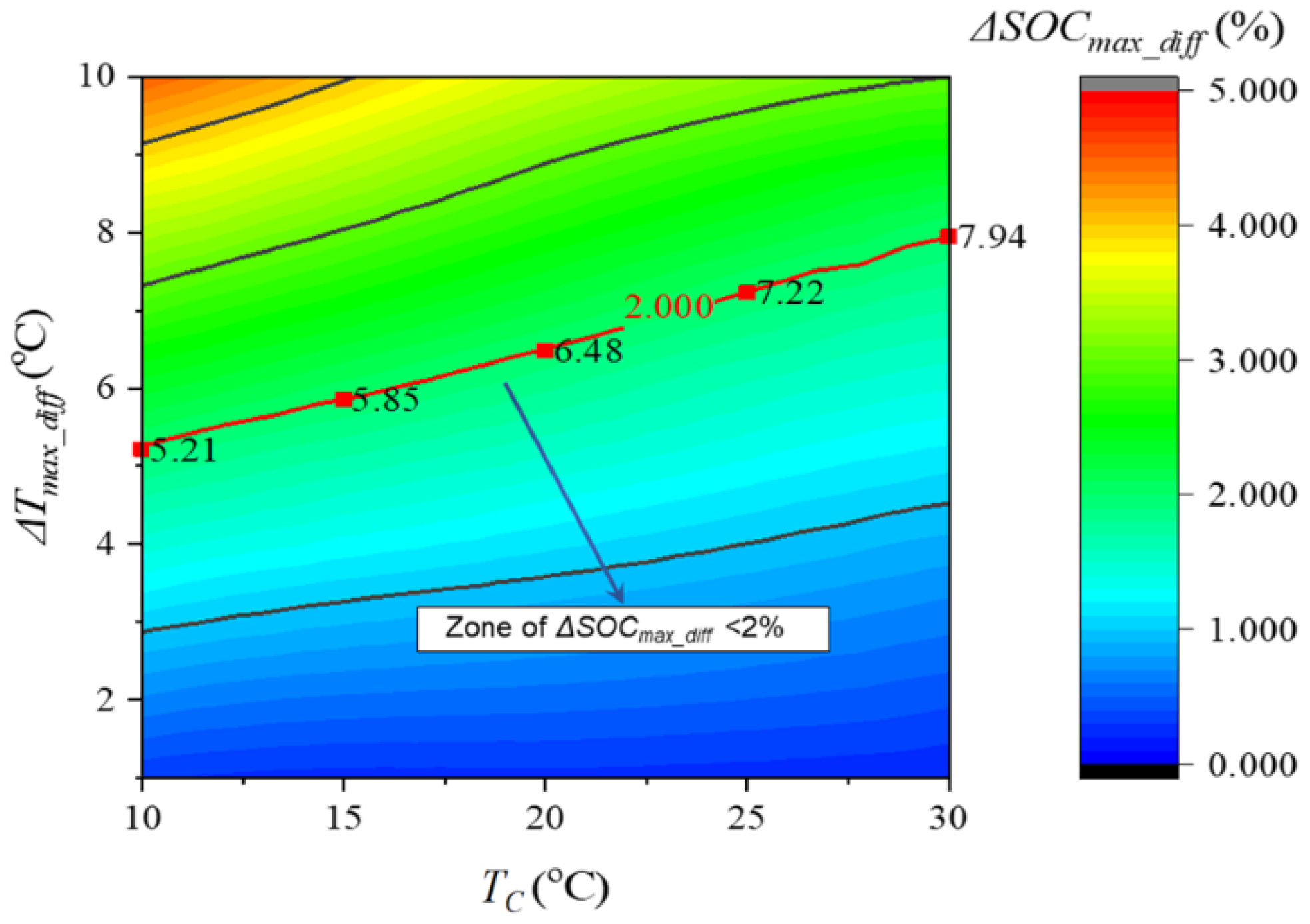
2.1.1. Electrochemical Model
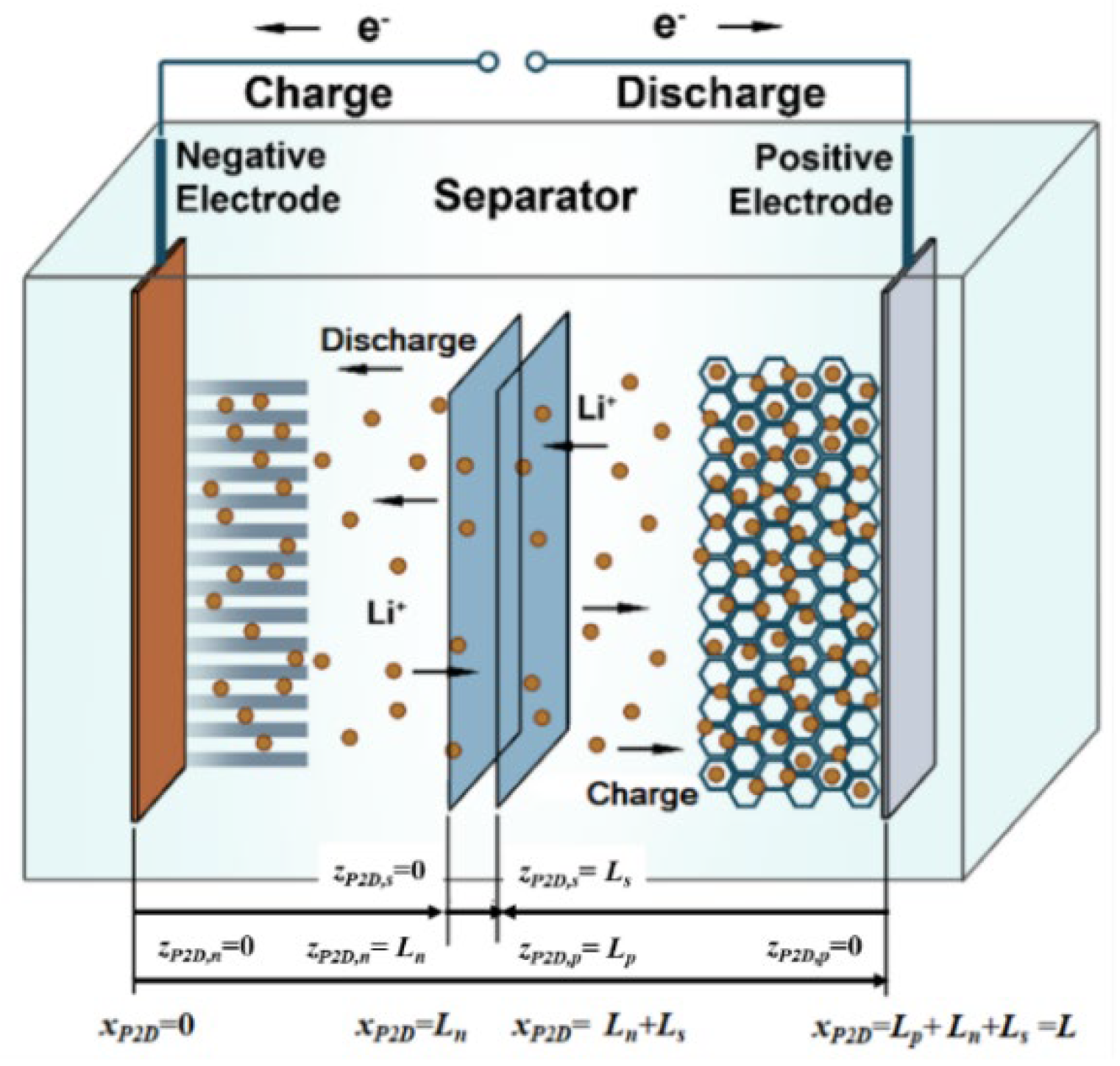
The pseudo two-dimensional model (P2D) is a strictly physics-based model and describes the electrochemical process in the batteries.
Table 1 shows the structure of the P2D model, and the meanings of the variables are given in the “Nomenclature”. The
xP2D coordinate is used for the P2D model at the microscale, and its direction shown in
Figure 2 is from the negative electrode to the positive electrode. The additional pseudo dimension
r for the P2D model is along the radial distance of the electrode particle
r.
The high-order partial differential equations (PDEs) involved in the P2D model require complex calculation, which makes the application difficult. Therefore, it is meaningful and necessary for the electrochemical model to be simplified and reduce the loss of model accuracy. The polynomial has a simple structure and can maintain high accuracy. Moreover, solving it requires little calculation. Therefore, we use the polynomial to approximate the solutions of the PDEs above, and the P2D model becomes the polynomial P2D model or PP2D model. Another major difference between the P2D model and the PP2D model lies in the selection of the coordinate axis. In order to easily calculate the PP2D model and apply it to each area of the cell, the
zP2D coordinate is used to replace the
xP2D coordinate in
Table 1. Because
zP2D is defined in the electrode and the diaphragm areas, respectively, axis
zP2D is divided into
zP2D,n for the negative electrode,
zP2D,s for the separator, and
zP2D,p for the positive electrode.
Figure 2 shows the regions of
zP2D,n,
zP2D,s and
zP2D,p, and
Table 2 shows the P2D model in the
zP2D coordinate.
- (a)
Electrolyte concentration approximation
Because the Li-ion diffusion equation given in Equation (5) is the second-order PDE, the distribution of the electrolyte concentration along the
z axis is assumed to be parabolic. The second-order polynomials are used for the concentration distributions in the electrolyte and separator, and they are
In Equation (9),
ce is the electrolyte concentration and the subscript n represents the negative electrode. The subscript
s represents the separator, and the subscript
p represents the positive electrode. In order to satisfy the boundary condition of the Li-ion diffusion equation given the third equation of Equation (6), the coefficients
a1,1 and
a3,1 are 0, and Equation (9) is simplified as
The coefficients [
a0,
a1,
a2,
a3,
a4,
a5,
a6] in Equation (10) are time-varying parameters, which need to be calculated in each time step. Substituting the first four equations in Equation (8) with Equation (10) gives
where
De is the liquid diffusion coefficient,
Deeff is the effective liquid diffusion coefficient, and their relationship is
De =
Deeffεe−1.5.
εe is the volume fraction of Li-ion in the electrolyte, and
εece is the concentration of Li-ion. The mole number of Li-ion at the unit area in the regions of the negative electrode
Qe,n(
t), the separator
Qs(
t), and the positive electrode
Qe,p(
t) are obtained by integrating its concentration along the
z-axis, and they are
The surface density of the current at the electrode (
I/
A) can be achieved by integrating the volumetric current density
jf along the
z axis, and the expressions are
where
A means the electrode plate area and
I is the current through the cell.
Substituting the second equation in Equation (5) and the first equation in Equation (7) with Equation (10), integrating these equations along the
z axis, and combing them with Equations (15)–(19) gives the expressions of
Qe,n(
t),
Qe,s(
t), and
Qe,p(
t), which are
In Equations (20)–(22), t is the time and t+ is the transfer number of Li-ion.
Because the initial electrolyte concentration is evenly distributed in regions of the electrodes and separator, the initial total mole numbers of Li-ion
Qe,n(0),
Qe,s(0), and
Qe,p(0) can be achieved by solving
where
ce,0 is the initial electrolyte concentration. In order to satisfy Equation (23), the initial polynomial coefficients [
a0,
a1,
a2,
a3,
a4,
a5,
a6] in Equations (15)–(17) should be meet the requirement below.
The forward Euler method is used to solve the Equations (20)–(22) to obtain the Qe,n(t), Qe,s(t), and Qe,p(t) in each time step. Then, these total mole numbers of Li-ion are used for Equations (11)–(17) to calculate [a0, a1, a2, a3, a4, a5, a6]. After the coefficient matrix is achieved, we can employ Equation (10) to approximate the distribution of the electrolyte concentration in the electrodes and separator.
- (b)
Reaction flux approximation
The reaction flux
j is non-uniform, especially when the large current rate is loaded on the cell. The local volumetric current density
jf is used to describe its distribution between the negative and positive electrodes. Because
jf is distributed like a parabola, a quadratic polynomial is applied to approximating the
jf. It is
In the electrode, the activation overpotential
η is calculated by the potential balance equation, which is
In Equation (26),
φs is the potential of the solid phase.
U is the electrode equilibrium potential, and
RSEI represents the resistance of SEI film.
φe represents the potential of the electrolyte.
as means the specific interfacial surface area, and the subscript
k is
n for the negative electrode or
p for the positive electrode.
η can also be calculated by solving the Bulter–Volmer given in Equation (5), and it is
where
T is cell temperature,
R is the gas constant,
F is Faraday constant, and
i0 represents the exchange current density. Substituting the
η in Equation (26) with Equation (27) gives
where
P(
ZP2D,k) is
Integrating the third and fourth equations in Equation (5) along the
ZP2D,n and
ZP2D,p, respectively, gives the expressions of the
φs and
φe. They are
where
σeff means solid-phase effective conductivity,
t+ is the transfer number of lithium-ion, and
κeff is the effective conductivity of the electrolyte.
With the Equations (25)–(31) and their corresponding boundary conditions, the coefficients in Equation (25) can be solved. We take the negative electrode as an example to show how
c2,n,
c1,n and
c0,n in Equation (25) are reached. The subscript
k in Equations (26)–(31) becomes
n. Substituting [∂
φs(
ZP2D,n)/∂
ZP2D,n] and [∂
φe(
ZP2D,n)/∂
ZP2D,n] in Equation (28) with Equations (30) and (31) gives
where
P(
ZP2D,n) is
When
ZP2D,n is 0, Equation (32) is solved by replacing
jf (
ZP2D,n) with Equation (25), and the result is
When
ZP2D,n is
Ln, the difference between the first second term on right side of Equation (32) is 0 according to Equation (28) and the fifth equation in Equation (6), and the integral value in the four term is (
I/
A) according to Equation (18). With the calculation above, Equation (32) under the condition of
ZP2D,n =
Ln becomes
In order to obtain
c2,n and
c1,n in Equations (34) and (35), it is necessary to calculate the electrode equilibrium potential
U(
ZP2D,n) at
ZP2D,n = 0 and
ZP2D,n =
Ln. According to Ref. [
24],
U(
ZP2D,n) in the negative electrode is decided by the stoichiometry of the electrode concentration
θn, and it is
where
θn(
ZP2D,n) is
The third equation for
c2,n,
c1,n, and
c0,n in Equation (25) is achieved by integrating Equation (18), and it is
By solving Equations (34), (35), and (38), we have the coefficients of
c2,n,
c1,n, and
c0,n in Equation (25) and the local volumetric current density in the negative electrode. Because the cell is in a stable state in the beginning of discharging or charging, the
jf is evenly distributed and the initial
jf for Equation (25) is
The calculation process of jf in the positive electrode is almost same as that in the negative electrode. The only difference is that the current I in the positive electrode should be multiplied by −1 because the direction of the ZP2D,p-axis is opposite to that of the ZP2D,n-axis.
- (c)
Solid-phase surface concentration approximation
According to Ref. [
25], the solid-phase surface concentration of lithium-ion
cs,e(
t) is not only related to its volume-averaged concentration
but also to its volume-averaged concentration flux
, and it can be calculated through [
25]
where
and
are
In Equations (40)–(42), t is time, Ds is the solid-phase diffusivity of lithium-ion, Rs is the particle radius, and the subscript i is n for the negative electrode or p for the positive electrode.
When
t = 0, the cell is about to charge or discharge, and the initial value of
is 0. Moreover, the initial value of
can be achieved by solving
where
s,n(0) and
s,p(0) are the volume-averaged concentration of lithium-ion in the positive and negative electrodes;
SOCini is the initial SOC,
cns,max and
cps,max are the maximum solid-phase concentration of lithium-ion in the positive and negative electrodes;
θ is the stoichiometry of electrode concentration; superscripts
n and
p represent the positive and negative electrode, and subscript 0 and 100 are 0% and 100% of the cell SOC, respectively.
- (d)
Other parameters for PP2D model
This section shows how the cell terminal voltage is calculated. Moreover, the calculation methods of the variables and the constants for the PP2D model are also given in this section. Integrating Equation (31) in the electrodes and separator and using the third equation in Equation (6) as the boundary condition for Equation (31) gives the formula of the electrolyte potential, which is
where
The potential difference can be achieved through Equation (45). It is
where
φe is the electrolyte potential; (0) is the current collector; and the subscript
p and
n represent positive and negative electrode separately. Terminal voltage
V of cell is reached by solving
where
φe is the solid-phase potential. Combining Equations (26), (37), (48), and (49) gives the expression of the cell terminal voltage. It is
where “
p(0)” and “
n(0)” refer to the variable values at
ZP2D,p = 0 and
ZP2D,n = 0, respectively. A 50Ah NCM/graphite prismatic cell is used in this paper, and its electrochemical parameters are given in
Table 3 and
Table 4.