A Review on Solution-Processed Organic Phototransistors and Their Recent Developments
Abstract
:1. Introduction
2. Organic Semiconductors and Fundamental Principles of OFETs
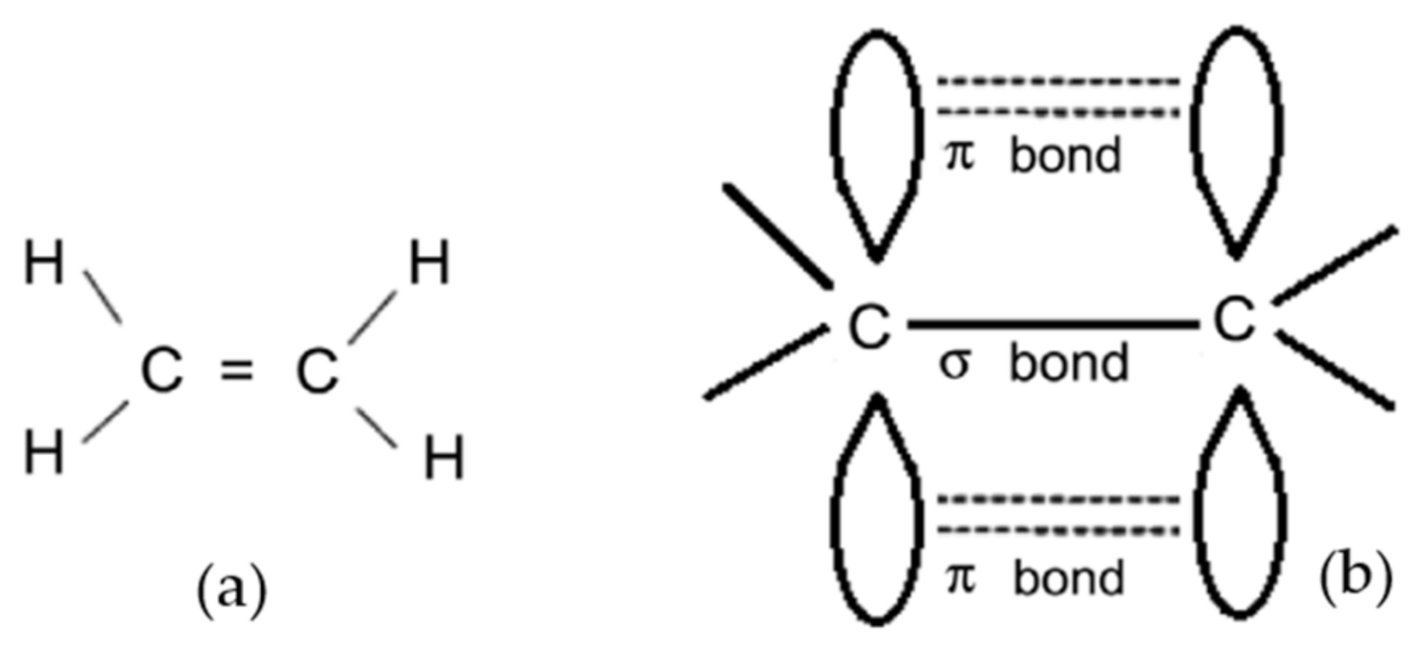
2.1. Organic Semiconductors
2.2. Fundamental Principles of OFETs
2.3. Solution Processing Techniques for Device Fabrication
3. Organic Phototransistors
3.1. Working Modes of OPTs
3.1.1. Photovoltaic Mode
3.1.2. Photoconductive Mode
3.2. Performance Parameters of OPTs
3.3. Charge Photogeneration and Separation in OPTs
- Interface/surface engineering
- Doping in channel/dielectric
- Direct use at channel layer
3.4. OPTs Based on Solution-Processed Small Molecules, Polymers and Blends
3.5. Novel Organic Semiconductors for Photosensing
4. The Applications of Organic Phototransistors
4.1. Photoactivated Memory Devices and Their Applications
4.2. Synaptic Phototransistors and Their Applications
5. Outlook and Future Perspectives
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Wakayama, Y.; Hayakawa, R.; Seo, H.S. Recent progress in photoactive organic field-effect transistors. Sci. Technol. Adv. Mater. 2014, 15, 024202. [Google Scholar] [CrossRef] [Green Version]
- Wang, C.; Zhang, X.; Hu, W. Organic photodiodes and phototransistors toward infrared detection: Materials, devices, and applications. Chem. Soc. Rev. 2020, 49, 653–670. [Google Scholar] [CrossRef] [PubMed]
- Baeg, K.J.; Binda, M.; Natali, D.; Caironi, M.; Noh, Y.Y. Organic light detectors: Photodiodes and phototransistors. Adv. Mater. 2013, 25, 4267–4295. [Google Scholar] [CrossRef]
- Xie, C.; Liu, C.K.; Loi, H.L.; Yan, F. Perovskite-Based Phototransistors and Hybrid Photodetectors. Adv. Funct. Mater. 2020, 30, 1–28. [Google Scholar] [CrossRef]
- Wu, Z.; Zhai, Y.; Kim, H.; Azoulay, J.D.; Ng, T.N. Emerging Design and Characterization Guidelines for Polymer-Based Infrared Photodetectors. Acc. Chem. Res. 2018, 51, 3144–3153. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Öberg, P.Å. Optical Sensors in Medical Care. Sensors Appl. 2008, 3, 15–43. [Google Scholar] [CrossRef]
- Chow, P.C.Y.; Someya, T. Organic Photodetectors for Next-Generation Wearable Electronics. Adv. Mater. 2020, 32, 1–26. [Google Scholar] [CrossRef] [PubMed]
- Zaumseil, J.; Sirringhaus, H. Electron and ambipolar transport in organic field-effect transistors. Chem. Rev. 2007, 107, 1296–1323. [Google Scholar] [CrossRef] [PubMed]
- Xu, H.; Li, J.; Leung, B.H.K.; Poon, C.C.Y.; Ong, B.S.; Zhang, Y.; Zhao, N. A high-sensitivity near-infrared phototransistor based on an organic bulk heterojunction. Nanoscale 2013, 5, 11850–11855. [Google Scholar] [CrossRef]
- Rim, Y.S.; Yang, Y.; Bae, S.H.; Chen, H.; Li, C.; Goorsky, M.S.; Yang, Y. Ultrahigh and Broad Spectral Photodetectivity of an Organic-Inorganic Hybrid Phototransistor for Flexible Electronics. Adv. Mater. 2015, 27, 6885–6891. [Google Scholar] [CrossRef]
- García De Arquer, F.P.; Armin, A.; Meredith, P.; Sargent, E.H. Solution-processed semiconductors for next-generation photodetectors. Nat. Rev. Mater. 2017, 2, 1–17. [Google Scholar] [CrossRef] [Green Version]
- Liu, J.; Wang, Y.; Wen, H.; Bao, Q.; Shen, L.; Ding, L. Organic Photodetectors: Materials, Structures, and Challenges. Sol. RRL 2020, 4, 1–8. [Google Scholar] [CrossRef]
- Nasiri, N.; Bo, R.; Wang, F.; Fu, L.; Tricoli, A. Ultraporous Electron-Depleted ZnO Nanoparticle Networks for Highly Sensitive Portable Visible-Blind UV Photodetectors. Adv. Mater. 2015, 27, 4336–4343. [Google Scholar] [CrossRef] [PubMed]
- Kurz, W.; Yetisen, A.K.; Kaito, M.V.; Fuchter, M.J.; Jakobi, M.; Elsner, M.; Koch, A.W. UV-Sensitive Wearable Devices for Colorimetric Monitoring of UV Exposure. Adv. Opt. Mater. 2020, 8, 1901969. [Google Scholar] [CrossRef] [Green Version]
- Xu, H.; Liu, J.; Zhang, J.; Zhou, G.; Luo, N.; Zhao, N. Flexible Organic/Inorganic Hybrid Near-Infrared Photoplethysmogram Sensor for Cardiovascular Monitoring. Adv. Mater. 2017, 29, 1–6. [Google Scholar] [CrossRef]
- Li, N.; Lan, Z.; Lau, Y.S.; Xie, J.; Zhao, D.; Zhu, F. SWIR Photodetection and Visualization Realized by Incorporating an Organic SWIR Sensitive Bulk Heterojunction. Adv. Sci. 2020, 7, 2000444. [Google Scholar] [CrossRef]
- Lee, Y.H.; Jang, M.; Lee, M.Y.; Kweon, O.Y.; Oh, J.H. Flexible Field-Effect Transistor-Type Sensors Based on Conjugated Molecules. Chem 2017, 3, 724–763. [Google Scholar] [CrossRef] [Green Version]
- Gu, P.; Yao, Y.; Feng, L.; Niu, S.; Dong, H. Recent advances in polymer phototransistors. Polym. Chem. 2015, 6, 7933–7944. [Google Scholar] [CrossRef]
- Dou, L.; Liu, Y.; Hong, Z.; Li, G.; Yang, Y. Low-Bandgap Near-IR Conjugated Polymers/Molecules for Organic Electronics. Chem. Rev. 2015, 115, 12633–12665. [Google Scholar] [CrossRef]
- Mei, J.; Diao, Y.; Appleton, A.L.; Fang, L.; Bao, Z. Integrated materials design of organic semiconductors for field-effect transistors. J. Am. Chem. Soc. 2013, 135, 6724–6746. [Google Scholar] [CrossRef]
- Guo, Y.; Yu, G.; Liu, Y. Functional organic field-effect transistors. Adv. Mater. 2010, 22, 4427–4447. [Google Scholar] [CrossRef] [PubMed]
- Chortos, A.; Liu, J.; Bao, Z. Pursuing prosthetic electronic skin. Nat. Mater. 2016, 15, 937–950. [Google Scholar] [CrossRef] [PubMed]
- Serdar Sariciftci, N. Plastic photovoltaic devices. Mater. Today 2004, 7, 36–40. [Google Scholar] [CrossRef]
- Liu, J.; Jiang, L.; Shi, J.; Li, C.; Shi, Y.; Tan, J.; Li, H.; Jiang, H.; Hu, Y.; Liu, X.; et al. Relieving the Photosensitivity of Organic Field-Effect Transistors. Adv. Mater. 2020, 32, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Huang, J.; Du, J.; Cevher, Z.; Ren, Y.; Wu, X.; Chu, Y. Printable and Flexible Phototransistors Based on Blend of Organic Semiconductor and Biopolymer. Adv. Funct. Mater. 2017, 27, 1–8. [Google Scholar] [CrossRef]
- Park, H.L.; Kim, M.H.; Kim, H. Improvement of photoresponse in organic phototransistors through bulk effect of photoresponsive gate insulators. Materials 2020, 13, 1565. [Google Scholar] [CrossRef] [Green Version]
- Huang, X.; Ji, D.; Fuchs, H.; Hu, W.; Li, T. Recent Progress in Organic Phototransistors: Semiconductor Materials, Device Structures and Optoelectronic Applications. ChemPhotoChem 2020, 4, 9–38. [Google Scholar] [CrossRef]
- Horowitz, B.G. Organic Field-Effect Transistors. Adv. Mater. 1998, 10, 365–377. [Google Scholar] [CrossRef]
- Braga, D.; Horowitz, G. High-Performance organic field-effect transistors. Adv. Mater. 2009, 21, 1473–1486. [Google Scholar] [CrossRef]
- Sirringhaus, H. Device physics of solution-processed organic field-effect transistors. Adv. Mater. 2005, 17, 2411–2425. [Google Scholar] [CrossRef]
- Sirringhaus, H. 25th anniversary article: Organic field-effect transistors: The path beyond amorphous silicon. Adv. Mater. 2014, 26, 1319–1335. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yang, S.Y.; Shin, K.; Park, C.E. The effect of gate-dielectric surface energy on pentacene morphology and organic field-effect transistor characteristics. Adv. Funct. Mater. 2005, 15, 1806–1814. [Google Scholar] [CrossRef]
- Ren, X.; Yang, F.; Gao, X.; Cheng, S.; Zhang, X.; Dong, H.; Hu, W. Organic Field-Effect Transistor for Energy-Related Applications: Low-Power-Consumption Devices, Near-Infrared Phototransistors, and Organic Thermoelectric Devices. Adv. Energy Mater. 2018, 8, 1–27. [Google Scholar] [CrossRef]
- Han, H.; Lee, C.; Kim, H.; Kim, Y. Flexible Near-Infrared Plastic Phototransistors with Conjugated Polymer Gate-Sensing Layers. Adv. Funct. Mater. 2018, 28, 1–8. [Google Scholar] [CrossRef]
- Park, J.; Kim, H.; Kim, T.; Lee, C.; Song, D.I.; Kim, Y. Effect of top channel thickness in near infrared organic phototransistors with conjugated polymer gate-sensing layers. Electronics 2019, 8, 1493. [Google Scholar] [CrossRef] [Green Version]
- Lei, Y.; Li, N.; Chan, W.K.E.; Ong, B.S.; Zhu, F. Highly sensitive near infrared organic phototransistors based on conjugated polymer nanowire networks. Org. Electron. 2017, 48, 12–18. [Google Scholar] [CrossRef]
- Lee, G.H.; Moon, H.; Kim, H.; Lee, G.H.; Kwon, W.; Yoo, S.; Myung, D.; Yun, S.H.; Bao, Z.; Hahn, S.K. Multifunctional materials for implantable and wearable photonic healthcare devices. Nat. Rev. Mater. 2020, 5, 149–165. [Google Scholar] [CrossRef]
- Li, N.; Lei, Y.; Chan, W.K.E.; Zhu, F. Broadband phototransistors realised by incorporating a bi-layer perovskite/NIR light absorbing polymer channel. J. Mater. Chem. C 2019, 7, 4808–4816. [Google Scholar] [CrossRef]
- Xia, Y.; Wang, L.; Deng, X.; Li, D.; Zhu, X.; Cao, Y. Photocurrent response wavelength up to 1.1 μm from photovoltaic cells based on narrow-band-gap conjugated polymer and fullerene derivative. Appl. Phys. Lett. 2006, 89, 89–92. [Google Scholar] [CrossRef]
- Li, F.; Chen, Y.; Ma, C.; Buttner, U.; Leo, K.; Wu, T. High-Performance Near-Infrared Phototransistor Based on n-Type Small-Molecular Organic Semiconductor. Adv. Electron. Mater. 2017, 3, 1600430. [Google Scholar] [CrossRef]
- Li, D.; Du, J.; Tang, Y.; Liang, K.; Wang, Y.; Ren, H.; Wang, R.; Meng, L.; Zhu, B.; Li, Y. Flexible and Air-Stable Near-Infrared Sensors Based on Solution-Processed Inorganic–Organic Hybrid Phototransistors. Adv. Funct. Mater. 2021, 31, 1–10. [Google Scholar] [CrossRef]
- Zhu, Z.; Guo, Y.; Liu, Y. Application of organic field-effect transistors in memory. Mater. Chem. Front. 2020, 4, 2845–2862. [Google Scholar] [CrossRef]
- Ling, Y.; An, T.; Yap, L.W.; Zhu, B.; Gong, S.; Cheng, W. Disruptive, Soft, Wearable Sensors. Adv. Mater. 2020, 32, 1904664. [Google Scholar] [CrossRef] [PubMed]
- Ebisawa, F.; Kurokawa, T.; Nara, S. Electrical properties of polyacetylene/polysiloxane interface. J. Appl. Phys. 1983, 54, 3255–3259. [Google Scholar] [CrossRef]
- Wang, C.; Dong, H.; Hu, W.; Liu, Y.; Zhu, D. Semiconducting π-Conjugated Systems in Field-Effect Transistors. Chem. Rev. 2012, 112, 2208–2267. [Google Scholar] [CrossRef]
- Fratini, S.; Nikolka, M.; Salleo, A.; Schweicher, G.; Sirringhaus, H. Charge transport in high-mobility conjugated polymers and molecular semiconductors. Nat. Mater. 2020, 19, 491–502. [Google Scholar] [CrossRef] [Green Version]
- Shi, L.; Liang, Q.; Wang, W.; Zhang, Y.; Li, G.; Ji, T.; Hao, Y.; Cui, Y. Research progress in organic photomultiplication photodetectors. Nanomaterials 2018, 8, 713. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Xie, W.; Menke, S.M.; Frisbie, C.D.; Holmes, R.J. Experimental Characterization of Charge and Exciton Transport in Organic Semiconductors. WSPC Ref. Org. Electron. Org. Semicond. 2016, 1, 231–291. [Google Scholar] [CrossRef]
- Forrest, S.R.; Thompson, M.E. Introduction: Organic electronics and optoelectronics. Chem. Rev. 2007, 107, 923–925. [Google Scholar] [CrossRef]
- Rivnay, J.; Inal, S.; Salleo, A.; Owens, R.M.; Berggren, M.; Malliaras, G.G. Organic electrochemical transistors. Nat. Rev. Mater. 2018, 3, 17086. [Google Scholar] [CrossRef]
- Russ, B.; Glaudell, A.; Urban, J.J.; Chabinyc, M.L.; Segalman, R.A. Organic thermoelectric materials for energy harvesting and temperature control. Nat. Rev. Mater. 2016, 1, 16050. [Google Scholar] [CrossRef]
- van de Burgt, Y.; Melianas, A.; Keene, S.T.; Malliaras, G.; Salleo, A. Organic electronics for neuromorphic computing. Nat. Electron. 2018, 1, 386–397. [Google Scholar] [CrossRef]
- Schlesinger, R. Chapter 2—Fundamentals. In Energy-Level Control at Hybrid Inorganic/Organic Semiconductor Interfaces, 1st ed.; Springer Theses; Springer International Publishing: Berlin/Heidelberg, Germany, 2017; pp. 7–45. ISBN 9783319466248. [Google Scholar]
- Kalyani, N.T.; Swart, H.; Dhoble, S.J. Luminescence in Organic Semiconductors. In Principles and Applications of Organic Light Emitting Diodes (OLEDs); Elsevier: Amsterdam, The Netherlands, 2017; pp. 39–64. ISBN 9780081012130. [Google Scholar]
- Nunzi, J.-M. Organic Materials and Devices for Photovoltaic Applicaitons. Photovolt. Photoactive Mater. Prop. Technol. Appl. Kluwer Acad. Publ. 2002, 3, 197–224. [Google Scholar]
- Sturge, M.D. Electrons and Holes in Semiconductors. Stat. Therm. Phys. 2020, 269–290. [Google Scholar] [CrossRef]
- Brütting, W. Physics of Organic Semiconductors, 1st ed.; Wiley-VCH Verlag GmbH & Co. KGaA: Weinheim, Germany, 2006; ISBN 352740550X. [Google Scholar]
- Zhu, M.; Lv, S.; Wang, Q.; Zhang, G.; Lu, H.; Qiu, L. Enhanced near-infrared photoresponse of organic phototransistors based on single-component donor-acceptor conjugated polymer nanowires. Nanoscale 2016, 8, 7738–7748. [Google Scholar] [CrossRef]
- Nam, S.; Han, H.; Seo, J.; Song, M.; Kim, H.; Anthopoulos, T.D.; McCulloch, I.; Bradley, D.D.C.; Kim, Y. Ambipolar Organic Phototransistors with p-Type/n-Type Conjugated Polymer Bulk Heterojunction Light-Sensing Layers. Adv. Electron. Mater. 2016, 2, 1–8. [Google Scholar] [CrossRef]
- Zhang, F.; Qu, G.; Mohammadi, E.; Mei, J.; Diao, Y. Solution-Processed Nanoporous Organic Semiconductor Thin Films: Toward Health and Environmental Monitoring of Volatile Markers. Adv. Funct. Mater. 2017, 27, 17–20. [Google Scholar] [CrossRef]
- RAO, M.C.; Shekhawat, M.S. A Brief Survey on Basic Properties of Thin Films for Device Application. Int. J. Mod. Phys. Conf. Ser. 2013, 22, 576–582. [Google Scholar] [CrossRef]
- Huang, W.; Feng, L.; Wang, G.; Reichmanis, E. Wearable Organic Nano-sensors. In Flexible and Wearable Electronics for Smart Clothing, 1st ed.; Wiley-VCH Verlag GmbH & Co. KGaA.: Hoboken, NJ, USA, 2020; pp. 1–27. [Google Scholar] [CrossRef]
- Street, R.A. Thin-Film Transistors. Adv. Mater. 2009, 21, 2007–2022. [Google Scholar] [CrossRef]
- Ong, B.S.; Wu, Y.; Liu, P.; Gardner, S. High-Performance Semiconducting Polythiophenes for Organic Thin-Film Transistors. J. Am. Chem. Soc. 2004, 126, 3378–3379. [Google Scholar] [CrossRef]
- Torsi, L.; Magliulo, M.; Manoli, K.; Palazzo, G. Organic field-effect transistor sensors: A tutorial review. Chem. Soc. Rev. 2013, 42, 8612–8628. [Google Scholar] [CrossRef] [PubMed]
- Li, H.; Shi, W.; Dailey, J.; Jang, H.-J.; Song, J.; Yu, J.; Katz, H.E. Vapor Sensing Using Organic, Polymer, and Nanomaterial Field-Effect Transistors, 2nd ed.; Elsevier: Amsterdam, The Netherlands, 2019; ISBN 9780081022849. [Google Scholar]
- Park, S.J.; Lee, S.H.; Yang, H.; Park, C.S.; Lee, C.S.; Kwon, O.S.; Park, T.H.; Jang, J. Human Dopamine Receptor-Conjugated Multidimensional Conducting Polymer Nanofiber Membrane for Dopamine Detection. ACS Appl. Mater. Interfaces 2016, 8, 28897–28903. [Google Scholar] [CrossRef] [PubMed]
- Someya, T.; Dodabalapur, A.; Huang, J.; See, K.C.; Katz, H.E. Chemical and Physical Sensing by Organic Field-Effect Transistors and Related Devices. Adv. Mater. 2010, 22, 3799–3811. [Google Scholar] [CrossRef] [PubMed]
- Hu, J.; Wei, W.; Ke, S.; Zeng, X.; Lin, P. A novel and sensitive sarcosine biosensor based on organic electrochemical transistor. Electrochim. Acta 2019, 307, 100–106. [Google Scholar] [CrossRef]
- Zhu, H.; Luo, W.; Ciesielski, P.N.; Fang, Z.; Zhu, J.Y.; Henriksson, G.; Himmel, M.E.; Hu, L. Wood-Derived Materials for Green Electronics, Biological Devices, and Energy Applications. Chem. Rev. 2016, 116, 9305–9374. [Google Scholar] [CrossRef]
- Huang, J.; Zhu, H.; Chen, Y.; Preston, C.; Rohrbach, K.; Cumings, J.; Hu, L. Highly transparent and flexible nanopaper transistors. ACS Nano 2013, 7, 2106–2113. [Google Scholar] [CrossRef] [PubMed]
- Tobjörk, D.; Österbacka, R. Paper electronics. Adv. Mater. 2011, 23, 1935–1961. [Google Scholar] [CrossRef]
- Qian, C.; Sun, J.; Yang, J.; Gao, Y. Flexible organic field-effect transistors on biodegradable cellulose paper with efficient reusable ion gel dielectrics. RSC Adv. 2015, 5, 14567–14574. [Google Scholar] [CrossRef]
- Kleemann, H.; Krechan, K.; Fischer, A.; Leo, K. A Review of Vertical Organic Transistors. Adv. Funct. Mater. 2020, 30, 1907113. [Google Scholar] [CrossRef]
- Mohammadian, N.; Faraji, S.; Sagar, S.; Das, B.C.; Turner, M.L.; Majewski, L.A. One-volt, solution-processed organic transistors with self-assembled monolayer-Ta2O5 gate dielectrics. Materials 2019, 12, 2563. [Google Scholar] [CrossRef] [Green Version]
- Seck, M.; Mohammadian, N.; Diallo, A.K.; Faraji, S.; Saadi, M.; Erouel, M.; Ly, E.H.B.; Khirouni, K.; Majewski, L.A. Low voltage organic transistors with water-processed gum arabic dielectric. Synth. Met. 2020, 267, 116447. [Google Scholar] [CrossRef]
- Lucas, B.; Trigaud, T.; Videlot-Ackermann, C. Organic transistors and phototransistors based on small molecules. Polym. Int. 2012, 61, 374–389. [Google Scholar] [CrossRef]
- Ortiz, R.P.; Facchetti, A.; Marks, T.J. High-k organic, inorganic, and hybrid dielectrics for low-voltage organic field-effect transistors. Chem. Rev. 2010, 110, 205–239. [Google Scholar] [CrossRef] [PubMed]
- Chen, X.; Zhang, H.; Zhang, Y.; Guan, X.; Zhang, Z.; Chen, D. Low-power flexible organic field-effect transistors with solution-processable polymer-ceramic nanoparticle composite dielectrics. Nanomaterials 2020, 10, 518. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Faraji, S.; Danesh, E.; Tate, D.J.; Turner, M.L.; Majewski, L.A. Cyanoethyl cellulose-based nanocomposite dielectric for low-voltage, solution-processed organic field-effect transistors (OFETs). J. Phys. D Appl. Phys. 2016, 49, 185102. [Google Scholar] [CrossRef] [Green Version]
- Faraji, S.; Hashimoto, T.; Turner, M.L.; Majewski, L.A. Solution-processed nanocomposite dielectrics for low voltage operated OFETs. Org. Electron. 2015, 17, 178–183. [Google Scholar] [CrossRef]
- Gao, Y.; Yi, Y.; Wang, X.; Meng, H.; Lei, D.; Yu, X.F.; Chu, P.K.; Li, J. A Novel Hybrid-Layered Organic Phototransistor Enables Efficient Intermolecular Charge Transfer and Carrier Transport for Ultrasensitive Photodetection. Adv. Mater. 2019, 31, 1–9. [Google Scholar] [CrossRef]
- Carr, J.A.; Chaudhary, S. The identification, characterization and mitigation of defect states in organic photovoltaic devices: A review and outlook. Energy Environ. Sci. 2013, 6, 3414–3438. [Google Scholar] [CrossRef]
- Lee, H.; Ahn, J.; Im, S.; Kim, J.; Choi, W. High-Responsivity Multilayer MoSe2 Phototransistors with Fast Response Time. Sci. Rep. 2018, 8, 1–8. [Google Scholar] [CrossRef] [Green Version]
- Pesavento, P.V.; Chesterfield, R.J.; Newman, C.R.; Frisble, C.D. Gated four-probe measurements on pentacene thin-film transistors: Contact resistance as a function of gate voltage and temperature. J. Appl. Phys. 2004, 96, 7312–7324. [Google Scholar] [CrossRef]
- Kymissis, I.; Dimitrakopoulos, C.D.; Purushothaman, S. High-Performance Bottom Electrode Organic. IEEE Trans. Electron Devices 2001, 48, 1060–1064. [Google Scholar] [CrossRef]
- Eggers, H.; Schackmar, F.; Abzieher, T.; Sun, Q.; Lemmer, U.; Vaynzof, Y.; Richards, B.S.; Hernandez-Sosa, G.; Paetzold, U.W. Inkjet-Printed Micrometer-Thick Perovskite Solar Cells with Large Columnar Grains. Adv. Energy Mater. 2020, 10, 1903184. [Google Scholar] [CrossRef] [Green Version]
- Corzo, D.; Almasabi, K.; Bihar, E.; Macphee, S.; Rosas-Villalva, D.; Gasparini, N.; Inal, S.; Baran, D. Digital Inkjet Printing of High-Efficiency Large-Area Nonfullerene Organic Solar Cells. Adv. Mater. Technol. 2019, 4, 1900040. [Google Scholar] [CrossRef]
- Tang, W.; Huang, Y.; Han, L.; Liu, R.; Su, Y.; Guo, X.; Yan, F. Recent progress in printable organic field effect transistors. J. Mater. Chem. C 2019, 7, 790–808. [Google Scholar] [CrossRef]
- Feng, L.; Jiang, C.; Ma, H.; Guo, X.; Nathan, A. All ink-jet printed low-voltage organic field-effect transistors on flexible substrate. Org. Electron. 2016, 38, 186–192. [Google Scholar] [CrossRef] [Green Version]
- Perinot, A.; Passarella, B.; Giorgio, M.; Caironi, M. Walking the Route to GHz Solution-Processed Organic Electronics: A HEROIC Exploration. Adv. Funct. Mater. 2020, 30, 1907641. [Google Scholar] [CrossRef]
- Yokota, T.; Sekitani, T.; Kato, Y.; Kuribara, K.; Zschieschang, U.; Klauk, H.; Yamamoto, T.; Takimiya, K.; Kuwabara, H.; Ikeda, M.; et al. Low-voltage organic transistor with subfemtoliter inkjet source–drain contacts. MRS Commun. 2011, 1, 3–6. [Google Scholar] [CrossRef] [Green Version]
- Sele, C.W.; von Werne, T.; Friend, R.H.; Sirringhaus, H. Lithography-Free, Self-Aligned Inkjet Printing with Sub-Hundred-Nanometer Resolution. Adv. Mater. 2005, 17, 997–1001. [Google Scholar] [CrossRef]
- Huang, Y.; Tang, W.; Chen, S.; Han, L.; Hou, X.; Guo, X. Scalable Processing of Low Voltage Organic Field Effect Transistors With a Facile Soft-Contact Coating Approach. IEEE Electron Device Lett. 2019, 40, 1945–1948. [Google Scholar] [CrossRef]
- Duan, S.; Wang, T.; Geng, B.; Gao, X.; Li, C.; Zhang, J.; Xi, Y.; Zhang, X.; Ren, X.; Hu, W. Solution-Processed Centimeter-Scale Highly Aligned Organic Crystalline Arrays for High-Performance Organic Field-Effect Transistors. Adv. Mater. 2020, 32, 1–8. [Google Scholar] [CrossRef]
- Yao, Y.; Zhang, L.; Leydecker, T.; Samorì, P. Direct Photolithography on Molecular Crystals for High Performance Organic Optoelectronic Devices. J. Am. Chem. Soc. 2018, 140, 6984–6990. [Google Scholar] [CrossRef] [Green Version]
- Zhang, Z.; Peng, B.; Ji, X.; Pei, K.; Chan, P.K.L. Marangoni-Effect-Assisted Bar-Coating Method for High-Quality Organic Crystals with Compressive and Tensile Strains. Adv. Funct. Mater. 2017, 27, 1703443. [Google Scholar] [CrossRef]
- Sze, S.; Ng, K.K. Photodetectors and Solar Cells. In Physics of Semiconductor Devices, 3rd ed.; John Wiley & Sons: Hoboken, NJ, USA, 2006; pp. 663–742. [Google Scholar]
- Bansal, A.K.; Hou, S.; Kulyk, O.; Bowman, E.M.; Samuel, I.D.W. Wearable Organic Optoelectronic Sensors for Medicine. Adv. Mater. 2015, 27, 7638–7644. [Google Scholar] [CrossRef] [Green Version]
- Wu, Z.; Zhai, Y.; Yao, W.; Eedugurala, N.; Zhang, S.; Huang, L.; Gu, X.; Azoulay, J.D.; Ng, T.N. The Role of Dielectric Screening in Organic Shortwave Infrared Photodiodes for Spectroscopic Image Sensing. Adv. Funct. Mater. 2018, 28, 1805738. [Google Scholar] [CrossRef] [Green Version]
- Choi, C.S.; Kang, H.S.; Choi, W.Y.; Kim, H.J.; Choi, W.J.; Kim, D.H.; Jang, K.C.; Seo, K.S. High optical responsivity of InAlAs-InGaAs metamorphic high-electron mobility transistor on GaAs substrate with composite channels. IEEE Photonics Technol. Lett. 2003, 15, 846–848. [Google Scholar] [CrossRef]
- Mok, S.M.; Yan, F.; Chan, H.L.W. Organic phototransistor based on poly(3-hexylthiophene)/ TiO2 nanoparticle composite. Appl. Phys. Lett. 2008, 93, 2–4. [Google Scholar] [CrossRef] [Green Version]
- Liu, X.; Dong, G.; Duan, L.; Wang, L.; Qiu, Y. High performance low-voltage organic phototransistors: Interface modification and the tuning of electrical, photosensitive and memory properties. J. Mater. Chem. 2012, 22, 11836–11842. [Google Scholar] [CrossRef]
- Park, H.L.; Kim, M.H.; Kim, H. Organic phototransistor by adopting photo-responsive polymer in hybrid gate-insulator configuration. Polymers 2020, 12, 527. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Trujillo Herrera, C.; Hong, M.J.; Labram, J.G. Role of the Blend Ratio in Polymer:Fullerene Phototransistors. ACS Appl. Electron. Mater. 2020, 2, 2257–2264. [Google Scholar] [CrossRef]
- Yang, Y.; Rim, Y.S.; Ok, K.C.; Yang, Y.M.; Chen, H.; Bae, S.H.; Wang, C.; Huang, Y.; Park, J.S. Boosting Responsivity of Organic-Metal Oxynitride Hybrid Heterointerface Phototransistor. ACS Appl. Mater. Interfaces 2016, 8, 14665–14670. [Google Scholar] [CrossRef]
- Zhang, X.; Hou, L.; Samorì, P. Coupling carbon nanomaterials with photochromic molecules for the generation of optically responsive materials. Nat. Commun. 2016, 7. [Google Scholar] [CrossRef] [PubMed]
- Wakayama, Y.; Hayakawa, R.; Higashiguchi, K.; Matsuda, K. Photochromism for optically functionalized organic field-effect transistors: A comprehensive review. J. Mater. Chem. C 2020, 8, 10956–10974. [Google Scholar] [CrossRef]
- Fu, L.N.; Leng, B.; Li, Y.S.; Gao, X.K. Photoresponsive organic field-effect transistors involving photochromic molecules. Chin. Chem. Lett. 2016, 27, 1319–1329. [Google Scholar] [CrossRef]
- Shen, Q.; Wang, L.; Liu, S.; Cao, Y.; Gan, L.; Guo, X.; Steigerwald, M.L.; Shuai, Z.; Liu, Z.; Nuckolls, C. Photoactive gate dielectrics. Adv. Mater. 2010, 22, 3282–3287. [Google Scholar] [CrossRef] [PubMed]
- Kang, M.; Cha, A.N.; Lee, S.A.; Lee, S.K.; Bae, S.; Jeon, D.Y.; Hong, J.M.; Fabiano, S.; Berggren, M.; Kim, T.W. Light-sensitive charge storage medium with spironaphthooxazine molecule-polymer blends for dual-functional organic phototransistor memory. Org. Electron. 2019, 78, 105554. [Google Scholar] [CrossRef]
- Ishiguro, Y.; Hayakawa, R.; Yasuda, T.; Chikyow, T.; Wakayama, Y. Unique device operations by combining optical-memory effect and electrical-gate modulation in a photochromism-based dual-gate transistor. ACS Appl. Mater. Interfaces 2013, 5, 9726–9731. [Google Scholar] [CrossRef] [PubMed]
- Hayakawa, R.; Higashiguchi, K.; Matsuda, K.; Chikyow, T.; Wakayama, Y. Optically and electrically driven organic thin film transistors with diarylethene photochromic channel layers. ACS Appl. Mater. Interfaces 2013, 5, 3625–3630. [Google Scholar] [CrossRef] [PubMed]
- Osaka, I.; Takimiya, K. Backbone orientation in semiconducting polymers. Polymer 2015, 59, A1–A15. [Google Scholar] [CrossRef]
- Xu, T.; Yu, L. How to design low bandgap polymers for highly efficient organic solar cells. Mater. Today 2014, 17, 11–15. [Google Scholar] [CrossRef]
- Isci, R.; Gunturkun, D.; Yalin, A.S.; Ozturk, T. Copolymers of 4-thieno[3,2-b]thiophen-3-ylbenzonitrile with anthracene and biphenyl; synthesis, characterization, electronic, optical, and thermal properties. J. Polym. Sci. 2021, 59, 117–123. [Google Scholar] [CrossRef]
- Bronstein, H.; Chen, Z.; Ashraf, R.S.; Zhang, W.; Du, J.; Durrant, J.R.; Shakya Tuladhar, P.; Song, K.; Watkins, S.E.; Geerts, Y.; et al. Thieno[3,2-b]thiophene-diketopyrrolopyrrole-containing polymers for high-performance organic field-effect transistors and organic photovoltaic devices. J. Am. Chem. Soc. 2011, 133, 3272–3275. [Google Scholar] [CrossRef]
- Cho, M.Y.; Kim, S.J.; Han, Y.D.; Park, D.H.; Kim, K.H.; Choi, D.H.; Joo, J. Highly sensitive, photocontrolled, organic thin-film transistors using soluble star-shaped conjugated molecules. Adv. Funct. Mater. 2008, 18, 2905–2912. [Google Scholar] [CrossRef]
- Han, S.T.; Zhou, Y.; Yang, Q.D.; Lee, C.S.; Roy, V.A.L. Poly(3-hexylthiophene)/gold nanoparticle hybrid system with an enhanced photoresponse for light-controlled electronic devices. Part. Part. Syst. Charact. 2013, 30, 599–605. [Google Scholar] [CrossRef]
- Han, S.T.; Zhou, Y.; Xu, Z.X.; Roy, V.A.L. Controllable threshold voltage shifts of polymer transistors and inverters by utilizing gold nanoparticles. Appl. Phys. Lett. 2012, 101, 033306. [Google Scholar] [CrossRef]
- Nam, S.; Seo, J.; Park, S.; Lee, S.; Jeong, J.; Lee, H.; Kim, H.; Kim, Y. Hybrid phototransistors based on bulk heterojunction films of poly(3-hexylthiophene) and zinc oxide nanoparticle. ACS Appl. Mater. Interfaces 2013, 5, 1385–1392. [Google Scholar] [CrossRef]
- Lim, D.H.; Lim, D.H.; Kang, M.; Jang, S.Y.; Hwang, K.; Kim, I.B.; Jung, E.; Jo, Y.R.; Kim, Y.J.; Kim, J.; et al. Unsymmetrical Small Molecules for Broad-Band Photoresponse and Efficient Charge Transport in Organic Phototransistors. ACS Appl. Mater. Interfaces 2020, 12, 25066–25074. [Google Scholar] [CrossRef] [PubMed]
- Wang, Q.; Zhu, M.; Wu, D.; Zhang, G.; Wang, X.; Lu, H.; Wang, X.; Qiu, L. Phototransistors based on a donor-acceptor conjugated polymer with a high response speed. J. Mater. Chem. C 2015, 3, 10734–10741. [Google Scholar] [CrossRef]
- Zhong, J.; Wu, X.; Lan, S.; Fang, Y.; Chen, H.; Guo, T. High Performance Flexible Organic Phototransistors with Ultrashort Channel Length. ACS Photonics 2018, 5, 3712–3722. [Google Scholar] [CrossRef]
- Vella, J.H.; Huang, L.; Eedugurala, N.; Mayer, K.S.; Ng, T.N.; Azoulay, J.D. Broadband infrared photodetection using a narrow bandgap conjugated polymer. Sci. Adv. 2021, 7, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Li, M.; Zeng, M.; Tang, X.; Zheng, T.; Weng, C.; Shen, P. Synergetic Effect of Side-Chain Engineering of Polymer Donors and Conformation Tuning of Small-Molecule Acceptors on Molecular Properties, Morphology, and Photovoltaic Performance. ACS Appl. Energy Mater. 2021, 4, 8117–8129. [Google Scholar] [CrossRef]
- Nguyen, T.; Nguyen, V.; Song, J.; An, J.; Truong, N. Molecular Weight-Dependent Physical and Photovoltaic Properties of Poly(3-alkylthiophene)s with Butyl, Hexyl, and Octyl Side-Chains. Polymers 2021, 13, 3440. [Google Scholar] [CrossRef]
- Lin, Y.-H.; Huang, W.; Pattanasattayavong, P.; Lim, J.; Li, R.; Sakai, N.; Panidi, J.; Hong, M.J.; Ma, C.; Wei, N.; et al. Deciphering photocarrier dynamics for tuneable high-performance perovskite-organic semiconductor heterojunction phototransistors. Nat. Commun. 2019, 10, 4475. [Google Scholar] [CrossRef] [PubMed]
- Isci, R.; Tekin, E.; Kaya, K.; Piravadili Mucur, S.; Gorkem, S.F.; Ozturk, T. Tetraphenylethylene substituted thienothiophene and dithienothiophene derivatives: Synthesis, optical properties and OLED applications. J. Mater. Chem. C 2020, 8, 7908–7915. [Google Scholar] [CrossRef]
- Kim, S.; Lee, D.; Lee, J.; Cho, Y.; Kang, S.-H.; Choi, W.; Oh, J.H.; Yang, C. Diazapentalene-Containing Ultralow-Band-Gap Copolymers for High-Performance Near-Infrared Organic Phototransistors. Chem. Mater. 2021, 33, 7499–7508. [Google Scholar] [CrossRef]
- Singh, A.K.; Chourasia, N.K.; Pal, B.N.; Pandey, A.; Chakrabarti, P. Low Operating Voltage Solution Processed (Li2ZnO2) Dielectric and (SnO2) Channel-Based Medium Wave UV-B Phototransistor for Application in Phototherapy. IEEE Trans. Electron Devices 2020, 67, 2028–2034. [Google Scholar] [CrossRef]
- Santiago, I. Trends and Innovations in Biosensors for COVID-19 Mass Testing. ChemBioChem 2020, 21, 2880–2889. [Google Scholar] [CrossRef]
- Choi, J.R. Development of Point-of-Care Biosensors for COVID-19. Front. Chem. 2020, 8, 517. [Google Scholar] [CrossRef] [PubMed]
- Asif, M.; Ajmal, M.; Ashraf, G.; Muhammad, N.; Aziz, A.; Iftikhar, T.; Wang, J.; Liu, H. The role of biosensors in coronavirus disease-2019 outbreak. Curr. Opin. Electrochem. 2020, 23, 174–184. [Google Scholar] [CrossRef]
- Şahin, S.; Ünlü, C.; Trabzon, L. Affinity biosensors developed with quantum dots in microfluidic systems. Emerg. Mater. 2021, 4, 1–23. [Google Scholar] [CrossRef]
- Lee, I.; Park, N.; Lee, H.; Hwang, C.; Kim, J.H.; Park, S. Systematic review on human skin-compatible wearable photoplethysmography sensors. Appl. Sci. 2021, 11, 2313. [Google Scholar] [CrossRef]
- Parak, J.; Korhonen, I. Evaluation of wearable consumer heart rate monitors based on photopletysmography. In Proceedings of the 36th Annual International Conference of the IEEE Engineering in Medicine and Biology Society, Chicago, IL, USA, 26–30 August 2014; pp. 3670–3673. [Google Scholar] [CrossRef]
- Rozi, R.M.; Usman, S.; Mohd Ali, M.A.; Reaz, M.B.I. Second Derivatives of Photoplethysmography (PPG) for Estimating Vascular Aging of Atherosclerotic Patients. In Proceedings of the 2012 IEEE-EMBS Conference on Biomedical Engineering and Sciences, Langkawi, Malaysia, 17–19 December 2012; pp. 256–259. [Google Scholar] [CrossRef]
- Beachey, W. Respiratory Care Anatomy and Physiology: Foundations for Clinical Practice, 2nd ed.; Mosby Elsevier: St. Louis, MO, USA, 2007. [Google Scholar]
- Lee, H.; Ko, H.; Lee, J. Reflectance pulse oximetry: Practical issues and limitations. ICT Express 2016, 2, 195–198. [Google Scholar] [CrossRef] [Green Version]
- Lauterbach, C.J.; Romano, P.A.; Greisler, L.A.; Brindle, R.A.; Ford, K.R.; Kuennen, M.R. Accuracy and Reliability of Commercial Wrist-Worn Pulse Oximeter During Normobaric Hypoxia Exposure Under Resting Conditions. Res. Q. Exerc. Sport 2020, 92, 549–558. [Google Scholar] [CrossRef]
- Nitzan, M.; Romem, A.; Koppel, R. Pulse oximetry: Fundamentals and technology update. Med. Devices Evid. Res. 2014, 7, 231–239. [Google Scholar] [CrossRef]
- Tao, J.; Liu, D.; Qin, Z.; Shao, B.; Jing, J.; Li, H.; Dong, H.; Xu, B.; Tian, W. Organic UV-Sensitive Phototransistors Based on Distriphenylamineethynylpyrene Derivatives with Ultra-High Detectivity Approaching 1018. Adv. Mater. 2020, 32, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Jiang, C.Y.; Tsai, C.H.; Tzou, C.Y.; Hsu, F.C.; Chen, Y.F. Magnetically controllable and flexible phototransistor for artificial intelligent skin with additional perception. Org. Electron. 2020, 85, 105849. [Google Scholar] [CrossRef]
- Liu, X.; Lee, E.K.; Kim, D.Y.; Yu, H.; Oh, J.H. Flexible Organic Phototransistor Array with Enhanced Responsivity via Metal-Ligand Charge Transfer. ACS Appl. Mater. Interfaces 2016, 8, 7291–7299. [Google Scholar] [CrossRef] [PubMed]
- Lee, C.; Kim, H.; Kim, Y. Short-wave infrared organic phototransistors with strong infrared-absorbing polytriarylamine by electron-transfer doping. NPJ Flex. Electron. 2021, 5, 1–9. [Google Scholar] [CrossRef]
- Li, Q.; Ran, Y.; Shi, W.; Qin, M.; Sun, Y.; Kuang, J.; Wang, H.; Chen, H.; Guo, Y.; Liu, Y. High-performance near-infrared polymeric phototransistors realized by combining cross-linked polymeric semiconductors and bulk heterojunction bilayer structures. Appl. Mater. Today 2021, 22, 100899. [Google Scholar] [CrossRef]
- Kim, M.-K.; Lee, J.-S. Ferroelectric Analog Synaptic Transistors. Nano Lett. 2019, 19, 2044–2050. [Google Scholar] [CrossRef]
- Yu, Y.; Ma, Q.; Ling, H.; Li, W.; Ju, R.; Bian, L.; Shi, N.; Qian, Y.; Yi, M.; Xie, L.; et al. Small-Molecule-Based Organic Field-Effect Transistor for Nonvolatile Memory and Artificial Synapse. Adv. Funct. Mater. 2019, 29, 1–31. [Google Scholar] [CrossRef]
- Gao, S.; Yi, X.; Shang, J.; Liu, G.; Li, R.-W. Organic and hybrid resistive switching materials and devices. Chem. Soc. Rev. 2019, 48, 1531–1565. [Google Scholar] [CrossRef]
- Wang, H.; Zhao, Q.; Ni, Z.; Li, Q.; Liu, H.; Yang, Y.; Wang, L.; Ran, Y.; Guo, Y.; Hu, W.; et al. A Ferroelectric/Electrochemical Modulated Organic Synapse for Ultraflexible, Artificial Visual-Perception System. Adv. Mater. 2018, 30, e1803961. [Google Scholar] [CrossRef]
- Wang, Z.; Zhang, S.-R.; Zhou, L.; Mao, J.-Y.; Han, S.-T.; Ren, Y.; Yang, J.-Q.; Wang, Y.; Zhai, Y.; Zhou, Y. Functional Non-Volatile Memory Devices: From Fundamentals to Photo-Tunable Properties. Phys. Status Solidi—Rapid Res. Lett. 2019, 13, 1800644. [Google Scholar] [CrossRef]
- Zhao, Q.; Wang, H.; Jiang, L.; Zhen, Y.; Dong, H.; Hu, W. Solution-Processed Flexible Organic Ferroelectric Phototransistor. ACS Appl. Mater. Interfaces 2017, 9, 43880–43885. [Google Scholar] [CrossRef] [PubMed]
- Chiang, Y.C.; Hung, C.C.; Lin, Y.C.; Chiu, Y.C.; Isono, T.; Satoh, T.; Chen, W.C. High-Performance Nonvolatile Organic Photonic Transistor Memory Devices using Conjugated Rod–Coil Materials as a Floating Gate. Adv. Mater. 2020, 32, 1–8. [Google Scholar] [CrossRef]
- Xu, X.; Deng, W.; Zhang, X.; Huang, L.; Wang, W.; Jia, R.; Wu, D.; Zhang, X.; Jie, J.; Lee, S.T. Dual-Band, High-Performance Phototransistors from Hybrid Perovskite and Organic Crystal Array for Secure Communication Applications. ACS Nano 2019, 13, 5910–5919. [Google Scholar] [CrossRef]
- Tzou, C.Y.; Cai, S.Y.; Tseng, C.Y.; Chang, C.Y.; Chiang, S.Y.; Jiang, C.Y.; Li, Y.H.; Ma, J.M.; Liao, Y.M.; Hsu, F.C.; et al. An ultra-fast two-terminal organic phototransistor with vertical topology for information technologies. Appl. Phys. Lett. 2019, 114, 193301. [Google Scholar] [CrossRef]
- Huang, T.Y.; Chen, C.H.; Lin, C.C.; Lee, Y.J.; Liu, C.L.; Liou, G.S. UV-sensing organic phototransistor memory devices with a doped organic polymer electret composed of triphenylamine-based aggregation-induced emission luminogens. J. Mater. Chem. C 2019, 7, 11014–11021. [Google Scholar] [CrossRef]
- Medintz, I.L.; Uyeda, H.T.; Goldman, E.R.; Mattoussi, H. Quantum dot bioconjugates for imaging, labelling and sensing. Nat. Mater. 2005, 4, 435–446. [Google Scholar] [CrossRef] [PubMed]
- Hu, J.; Odom, T.W.; Lieber, C.M. Chemistry and Physics in One Dimension: Synthesis and Properties of Nanowires and Nanotubes. Acc. Chem. Res. 1999, 32, 435–445. [Google Scholar] [CrossRef]
- Novoselov, K.S.; Jiang, D.; Schedin, F.; Booth, T.J.; Khotkevich, V.V.; Morozov, S.V.; Geim, A.K. Two-dimensional atomic crystals. Proc. Natl. Acad. Sci. USA 2005, 102, 10451–10453. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Fang, H.; Hu, W. Photogating in Low Dimensional Photodetectors. Adv. Sci. 2017, 4, 1700323. [Google Scholar] [CrossRef]
- He, Z.; Han, J.; Du, X.; Cao, L.; Wang, J.; Zheng, C.; Lin, H.; Tao, S. Photomemory and Pulse Monitoring Featured Solution-Processed Near-Infrared Graphene/Organic Phototransistor with Detectivity of 2.4 × 1013 Jones. Adv. Funct. Mater. 2021, 31, 1–8. [Google Scholar] [CrossRef]
- Ljubic, D.; Liu, W.; González-Espinoza, C.E.; Hu, N.-X.; Wu, Y.; Zhu, S. Binary Blends of Polyimide and Benzothienobenzothiophene for High-Performance Solution-Processed Organic Phototransistors. Adv. Electron. Mater. 2017, 3, 1700284. [Google Scholar] [CrossRef]
- Li, J.; Fu, W.H.; Li, L.K.; Jiang, D.L.; He, L.C.; Zhu, W.Q.; Zhang, J.H. Recent advances in solid electrolytes for synaptic transistors. Org. Electron. 2021, 95, 106196. [Google Scholar] [CrossRef]
- Yoon, C.; Lee, J.H.; Lee, S.; Jeon, J.H.; Jang, J.T.; Kim, D.H.; Kim, Y.H.; Park, B.H. Synaptic Plasticity Selectively Activated by Polarization-Dependent Energy-Efficient Ion Migration in an Ultrathin Ferroelectric Tunnel Junction. Nano Lett. 2017, 17, 1949–1955. [Google Scholar] [CrossRef]
- Lu, K.; Li, X.; Sun, Q.; Pang, X.; Chen, J.; Minari, T.; Liu, X.; Song, Y. Solution-processed electronics for artificial synapses. Mater. Horiz. 2021, 8, 447–470. [Google Scholar] [CrossRef] [PubMed]
- Wan, C.J.; Liu, Y.H.; Zhu, L.Q.; Feng, P.; Shi, Y.; Wan, Q. Short-Term Synaptic Plasticity Regulation in Solution-Gated Indium–Gallium–Zinc-Oxide Electric-Double-Layer Transistors. ACS Appl. Mater. Interfaces 2016, 8, 9762–9768. [Google Scholar] [CrossRef]
- Yang, Y.; Wen, J.; Guo, L.; Wan, X.; Du, P.; Feng, P.; Shi, Y.; Wan, Q. Long-Term Synaptic Plasticity Emulated in Modified Graphene Oxide Electrolyte Gated IZO-Based Thin-Film Transistors. ACS Appl. Mater. Interfaces 2016, 8, 30281–30286. [Google Scholar] [CrossRef]
- Yang, J.-T.; Ge, C.; Du, J.-Y.; Huang, H.-Y.; He, M.; Wang, C.; Lu, H.-B.; Yang, G.-Z.; Jin, K.-J. Artificial Synapses Emulated by an Electrolyte-Gated Tungsten-Oxide Transistor. Adv. Mater. 2018, 30, 1801548. [Google Scholar] [CrossRef] [PubMed]
- Qian, C.; Sun, J.; Kong, L.; Gou, G.; Yang, J.; He, J.; Gao, Y.; Wan, Q. Artificial Synapses Based on in-Plane Gate Organic Electrochemical Transistors. ACS Appl. Mater. Interfaces 2016, 8, 26169–26175. [Google Scholar] [CrossRef]
- Raeis-Hosseini, N.; Park, Y.; Lee, J.-S. Flexible Artificial Synaptic Devices Based on Collagen from Fish Protein with Spike-Timing-Dependent Plasticity. Adv. Funct. Mater. 2018, 28, 1800553. [Google Scholar] [CrossRef]
- Nawrocki, R.A.; Voyles, R.M.; Shaheen, S.E. A Mini Review of Neuromorphic Architectures and Implementations. IEEE Trans. Electron Devices 2016, 63, 3819–3829. [Google Scholar] [CrossRef]
- Liu, J.; Yang, Z.; Gong, Z.; Shen, Z.; Ye, Y.; Yang, B.; Qiu, Y.; Ye, B.; Xu, L.; Guo, T.; et al. Weak Light-Stimulated Synaptic Hybrid Phototransistors Based on Islandlike Perovskite Films Prepared by Spin Coating. ACS Appl. Mater. Interfaces 2021, 13, 13362–13371. [Google Scholar] [CrossRef]
- Huang, X.; Li, Q.; Shi, W.; Liu, K.; Zhang, Y.; Liu, Y.; Wei, X.; Zhao, Z.; Guo, Y.; Liu, Y. Dual-Mode Learning of Ambipolar Synaptic Phototransistor Based on 2D Perovskite/Organic Heterojunction for Flexible Color Recognizable Visual System. Small 2021, 17, 1–8. [Google Scholar] [CrossRef] [PubMed]





















| Parameter | Organic SC | Inorganic SC |
|---|---|---|
| Binding energy | Weak (Van der Waals) | Strong (Covalent, metallic, ionic bonding) |
| Mechanical resistance | Weak (prone to gases and oxidation) | Strong |
| Charge carriers | Polarons, excitons (localized) | e−, h+, ions (delocalized) |
| Dielectric constant | 3–4 | 11 |
| Polarisation | Strong | Weak |
| Charge transport | Hopping | Band |
| Exciton | Frenkel | Wannier-Mott |
| Mobility | ~10−6–1 cm2 V−1 s−1 | ~1000 cm2 V−1 s−1 |
| Conductivity | Extrinsic | Intrinsic |
| Figures of Merit | Definition | Unit |
|---|---|---|
| Photoresponsivity (R) | (Ilight − Idark)/Popt 1 | A W−1 |
| Photosensitivity (P) | (Ilight − Idark)/Idark | Unitless |
| Detectivity (D*) | The minimum input optical signal to generate photocurrent. D* = (AΔf)1/2R/Sn 2 | 1 Jones = 1 cm Hz1/2 W−1 |
| Linear Dynamic Range (LDR) | Range of incident optical powers for which the detector responds linearly 3 | dB |
| External Quantum Efficiency (EQE) | The ratio between the numbers of collected carriers and incident photon | EQE = hc/λq % |
| Noise Equivalent Power (NEP) | Sn/R | W Hz−1/2 |
| Noise Density (Sn) | It has multiple components, including thermal noise, which is significant in narrow band gap, shot noise and 1/f noise | A/Hz1/2 |
| Photoconductive Gain (G) | Number of collected carriers divided by the number of photons absorbed | G = τlifetimeτdrift−1 Unitless |
| Device Structure | OSC Type | Dielectric | Mobility (µ) (cm2 V−1 s−1) | Photoresponsivity (R) (A W−1) | Photosensitivity (P) Iph/Idark | Type of Radiation | Wavelength Range | Refs |
|---|---|---|---|---|---|---|---|---|
| BGBC | P3HT:PCBM | SiO2 | - | - | - | UV-Vis | 300–1000 nm | [105] |
| TGBC | DPPT-TT/TFP:PS | Cyanoresin (high-k) | 0.84 | 0.091 | - | UV-Vis-NIR | 250–1000 nm | [122] |
| BGBC | DPP-DTT:PCBM | SiO2 | 0.3 | 5 × 105 | ~1.6 × 104 | NIR | 350–1000 nm, max at 808 nm | [9] |
| BGBC | DPP-DTT:PCBM Encapsulation layer: PTFE/Parylene | Al2O3/SiO2 | - | 3.5 × 105 | - | NIR | max 810 nm | [15] |
| BGTC | PODTPPD-BT (active layer), P3HT (channel layer) | PMMA | - | 388.43 mA W−1 (max R value at 780 nm) | - | NIR | 200–1000 nm | [35] |
| BGTC | BODIPY-BF2 | SiO2 | 1.13 × 10−1 | 1.14 × 104 | 1.04 × 104 | NIR | 600–1000 nm, max at 840 nm | [40] |
| BGTC | C8-BTBT:PC61BM (BHJ layer), MoO3 (Multifunctional layer), C8-BTBT (Conduction channel) | SiO2 | - | 8.6 × 103 | 2.9 × 106 | UV-Vis | 352–700 nm | [82] |
| BGTC | DPP-DTT nanowire | SiO2 | - | ̴ 246 | 103 | NIR | 600–1000 nm, max at 850 nm | [36] |
| TGBC | P3HT:PDPPTTT | PMMA/PVA Low k/high-k | 0.14 | 2.43 (p-channel) 4.29 (n-channel) | 170 (p-channel) 61 (n-channel) | Vis-NIR | 350–900 nm | [59] |
| BGTC | PBIBDF-BT | OTS/SiO2 | - | 108.43 mA W−1 (p-channel) 38.72 mA W−1 (n-channel) | 4552 (p-channel) 1044 (n-channel) | Vis-NIR | 600–1000 nm, max at 650 nm | [123] |
| BGTC | PBIBDF-TT nanowire | SiO2 | - | 440 mA W−1 (p-channel) 70 mA W−1 (n-channel) | 1.3 × 104 (p-channel) 3.3 × 104 (n-channel) | Vis-NIR | 400–1200 nm, max at 808 nm | [58] |
| BGTC | PDVT-8/PC61BM | SiO2 | - | 750 | 1 × 106 | Vis | 720 nm | [124] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tavasli, A.; Gurunlu, B.; Gunturkun, D.; Isci, R.; Faraji, S. A Review on Solution-Processed Organic Phototransistors and Their Recent Developments. Electronics 2022, 11, 316. https://doi.org/10.3390/electronics11030316
Tavasli A, Gurunlu B, Gunturkun D, Isci R, Faraji S. A Review on Solution-Processed Organic Phototransistors and Their Recent Developments. Electronics. 2022; 11(3):316. https://doi.org/10.3390/electronics11030316
Chicago/Turabian StyleTavasli, Aybuke, Betul Gurunlu, Dilara Gunturkun, Recep Isci, and Sheida Faraji. 2022. "A Review on Solution-Processed Organic Phototransistors and Their Recent Developments" Electronics 11, no. 3: 316. https://doi.org/10.3390/electronics11030316
APA StyleTavasli, A., Gurunlu, B., Gunturkun, D., Isci, R., & Faraji, S. (2022). A Review on Solution-Processed Organic Phototransistors and Their Recent Developments. Electronics, 11(3), 316. https://doi.org/10.3390/electronics11030316







