An Investigation into Incidences of Microbial Contamination in Cosmeceuticals in the UAE: Imbalances between Preservation and Microbial Contamination
Abstract
1. Introduction
2. Methodology/Materials
2.1. Collection of Samples (Sampling Methodology)
2.2. Standards and Guidelines Used for the Detection of Microorganisms
2.3. Enumeration of Aerobic Mesophilic Bacteria
2.4. Enumeration of Yeast and Mold
2.5. Detection of Escherichia coli
2.6. Detection of Staphylococcus aureus
2.7. Detection of Pseudomonas aeruginosa
2.8. Detection of Candida albicans
2.9. Reporting of Values
2.10. Quality Control and Assurance
2.11. Ethical Considerations
3. Statistical Analysis
4. Results
4.1. Sample Description
4.2. Evaluation of the Incidence of Bacterial and Fungal Contamination in the Cosmetics and Personal Care Products
4.3. Comparison of Microbial Contamination According to Sample Characteristics
5. Discussion
6. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- European Parliament, Council of the European Union. Regulation (EC) No 1223/2009 of the European Parliament and of the Council of 30 November 2009 on Cosmetic Products; European Parliament, Council of the European Union: Brussels, Belgium, 2009. [Google Scholar]
- Michalek, I.M.; John, S.M.; Caetano dos Santos, F.L. Microbiological contamination of cosmetic products–observations from Europe, 2005–2018. J. Eur. Acad. Dermatol. Venereol. 2019, 33, 2151–2157. [Google Scholar] [CrossRef] [PubMed]
- Kligman, A.M. Cosmeceuticals as a third category. Cosmet. Toilet 1998, 113, 33–41. [Google Scholar]
- Vermeer, B.J.; Gilchrest, B.A. Cosmeceuticals: A proposal for rational definition, evaluation and regulation. Arch. Dermatol. 1996, 132, 337–340. [Google Scholar] [CrossRef] [PubMed]
- Behravan, J.; Bazzaz, F.; Malaekeh, P. Survey of bacteriological contamination of cosmetic creams in Iran (2000). Int. J. Dermatol. 2005, 44, 482–485. [Google Scholar] [CrossRef] [PubMed]
- Becks, V.E.; Lorenzoni, N.M. Pseudomonas aeruginosa outbreak in a neonatal intensive care unit: A possible link to contaminated hand lotion. Am. J. Infect. Control 1995, 23, 396–398. [Google Scholar] [CrossRef] [PubMed]
- The Scientific Committee on Consumers Safety, Directorate-General for Health and Consumer Protection of the European Commission. The SSCS’s Notes of Guidance for the Testing of Cosmetic Ingredients and Their Safety Evaluation, 10th ed.; European Comission: Brussels, Belgium, 2018. [Google Scholar]
- Anelich, L.E.; Korsten, L. Survey of micro-organisms associated with spoilage of cosmetic creams manufactured in South Africa. Int. J. Cosmet. Sci. 1996, 18, 25–40. [Google Scholar] [CrossRef] [PubMed]
- Dadashi, L.; Dehghanzadeh, R. Investigating incidence of bacterial and fungal contamination in shared cosmetic kits available in the women beauty salons. Health Promot. Perspect. 2016, 6, 159–163. [Google Scholar] [CrossRef] [PubMed]
- Lundov, M.D.; Zacharian, C. Recall of microbiologically contaminated cosmetics is Eu from 2005 to May 2008. Int. J. Cosmet. Sci. 2008, 30, 471–474. [Google Scholar] [CrossRef] [PubMed]
- Scientific Committee on Consumer Products. The SCCP’s Notes of Guidance for the Testing of Cosmetic Ingredients and Their Safety Evaluation. 2006. Available online: http://ec.europa.eu/health/scientific_committees/consumer_safety/docs/sccs_s_006.pdf (accessed on 13 June 2020).
- Nasser, L.A. Fungal profiles isolated from open and used cosmetic products collected from different localities in Saudi Arabia. Saudi J. Biol. Sci. 2008, 15, 121–128. [Google Scholar]
- Flores, M.; Morillo, M.; Crespo, M.L. Deterioration of raw materials and cosmetic products by preservative resistant microorganisms. Int. Biodeterior. Biodegrad. 1997, 40, 157–160. [Google Scholar] [CrossRef]
- Abaas, I.S.; Mohammed, H.R.; Majeed, A.H. The microbial contamination study of some herbal cosmatics products used in traditional medicine in Iraq. Karbala J. Pharm. Sci. 2013, 6, 132–140. [Google Scholar]
- Kulkarni, S.B.; Bajpai, N.D.; Meghre, V.S. Evaluation of dome marketed facepacks and cakes for microbial load. Asian J. Microbiol. Biotechnol. Environ. Sci. 2011, 13, 213–216. [Google Scholar]
- Kabara, J.J.; Orth, D.S. (Eds.) Preservative-free and self-preserving cosmetics and drugs. In Principles for Product Preservation; Marcel Dekker: New York, NY, USA, 1997; Volume 31, pp. 1–14. [Google Scholar]
- Wedderburn, D.L. Preservation of emulsions against microbial attack. In Advances in Pharmaceutical Sciences; Bean, A.H., Beckett, J.E., Carless, H.S., Eds.; Academic Press: London, UK, 1964; Volume 1, pp. 195–268. [Google Scholar]
- Elmorsy, T.H.; Hafez, E.A. Microbial contamination of some cosmetic preparations in Egypt. J. Agric. Technol. 2016, 12, 471–481. [Google Scholar]
- Steinberg, D.C. Preservatives for Cosmetics, 2nd ed.; Allured Publishing: Carol Stream, IL, USA, 2006; pp. 2–7. [Google Scholar]
- Lundov, M.D.; Moesby, L.; Zachariae, C.; Johansen, J.D. Contamination versus preservation of cosmetics: A review on legislation, usage, infections, and contact allergy. Contact Dermat. 2009, 60, 70–78. [Google Scholar] [CrossRef] [PubMed]
- Okeke, I.N.; Lamikanra, A. Bacteriological quality of skin-moisturizing creams and lotions distributed in a tropical developing country. J. Appl. Microbiol. 2001, 23, 922–928. [Google Scholar] [CrossRef] [PubMed]
- Farrington, J.K.; Martz, E.L.; Wells, S.J.; Ennis, C.C.; Holder, J.; Levchuk, J.W.; Avis, K.E.; Hoffman, P.S.; Hitchins, A.D.; Madden, J.M. Ability of laboratory methods to predict in-use efficacy of antimicrobial preservatives in an experimental cosmetic. Appl. Environ. Microbiol. 1994, 1, 4553–4558. [Google Scholar] [CrossRef] [PubMed]
- Lintner, K.; Genet, V. A physical method for preservation of cosmetic products. Int. J. Cosmet. Sci. 1998, 20, 103–115. [Google Scholar] [CrossRef] [PubMed]
- Brannan, D.K.; Dille, J.C. Type of closure prevents microbial contamination of cosmetics during consumer use. Appl. Environ. Microbiol. 1990, 1, 1476–1479. [Google Scholar] [CrossRef] [PubMed]
- Campana, R.; Scesa, C.; Patrone, V.; Vittoria, E.; Baffone, W. Microbiological study of cosmetic products during their use by consumers: Health risk and efficacy of preservative systems. Lett. Appl. Microbiol. 2006, 43, 301–306. [Google Scholar] [CrossRef] [PubMed]
| Characteristics | Groups | Frequency | Percentage |
|---|---|---|---|
| Categories | Body care | 39 | 39% |
| Eye care | 8 | 8% | |
| Face and neck care | 21 | 21% | |
| Hair and scalp care | 21 | 21% | |
| Oral hygiene products | 11 | 11% | |
| Country of origin | China | 9 | 9% |
| EU | 15 | 15% | |
| India | 14 | 14% | |
| Korea | 8 | 8% | |
| The Middle East | 10 | 10% | |
| UAE | 32 | 32% | |
| USA | 12 | 12% |
| Microorganism Type | Number of Cosmetics with Microorganisms under the LOD | |
|---|---|---|
| Frequency | Percentage | |
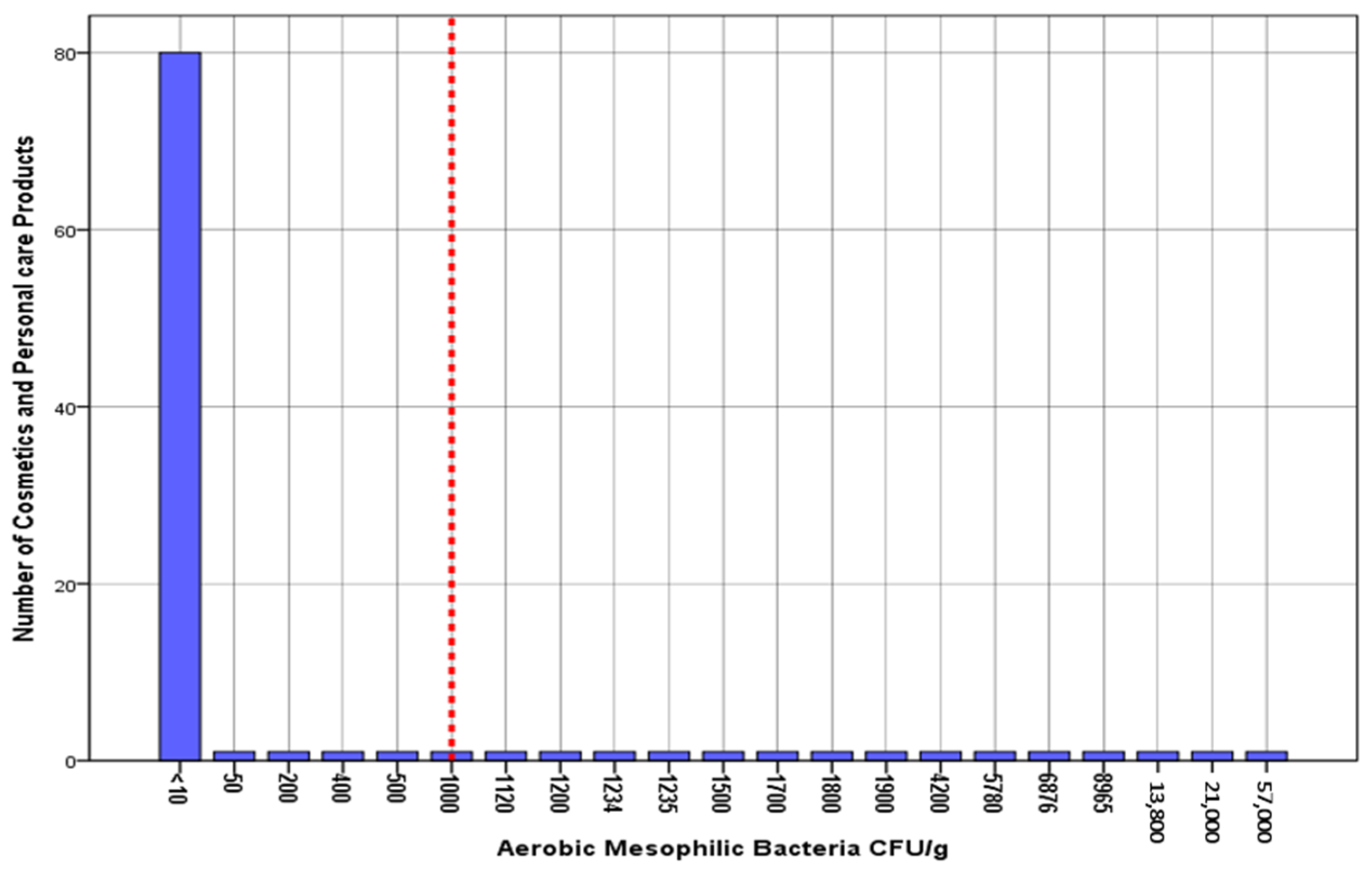
| Aerobic mesophilic bacteria | 80 | 80% |
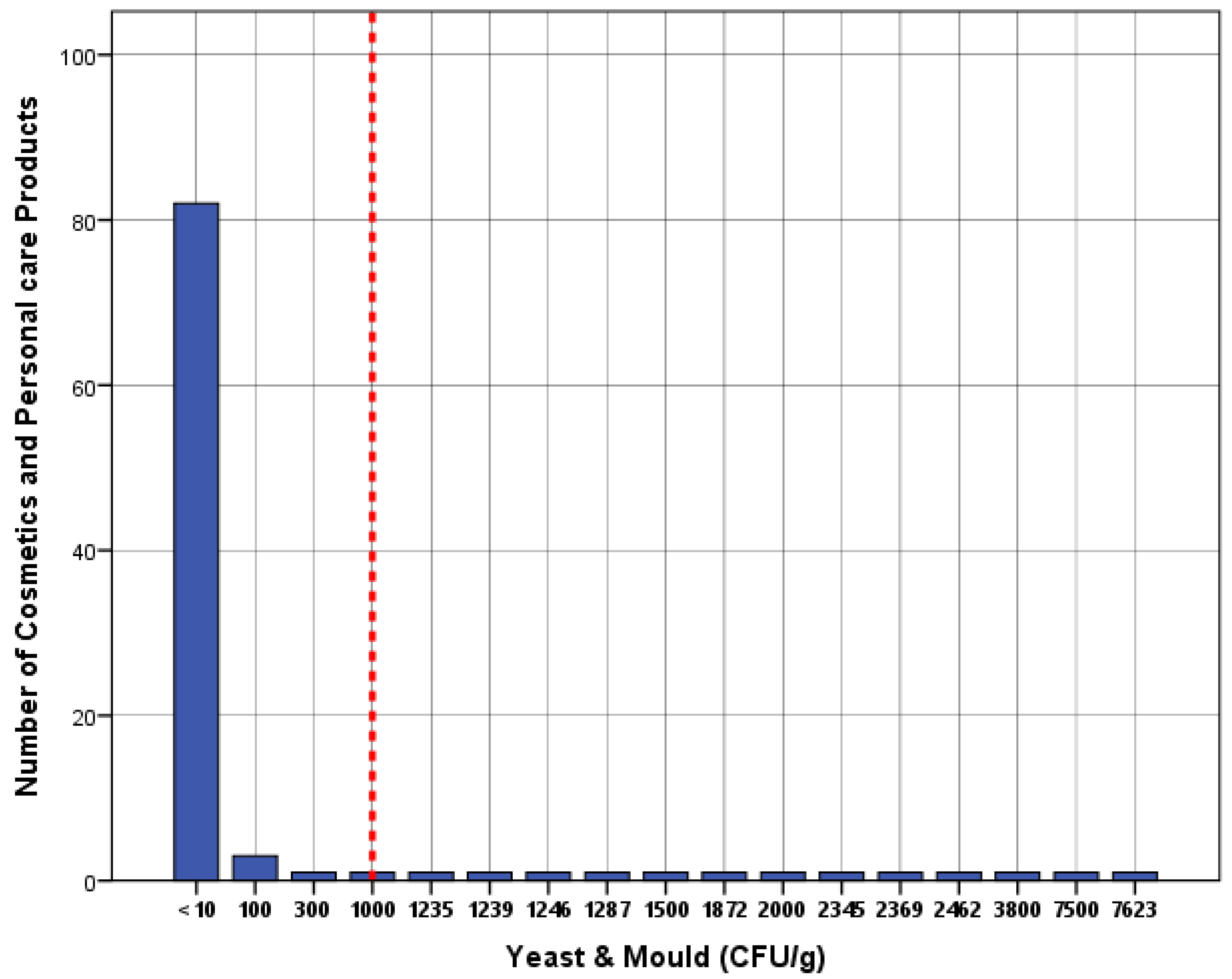
| Yeast and mold | 82 | 82% |
| Escherichia coli | 100 | 100% |
| Staphylococcus aureus | 100 | 100% |
| Candida albicans | 100 | 100% |
| Pseudomonas aeruginosa | 100 | 100% |
| Microorganism Type | Microbiological Maximum Limits | Cosmetics Exceeding the Maximum Limit | |||
|---|---|---|---|---|---|
| N | % | 95% CI | |||
| Lower | Upper | ||||
| Aerobic mesophilic bacteria | 1000 CFU/g | 15 | 15% | 0.79 | 22.1 |
| Yeast and mold | 1000 CFU/g | 13 | 13% | 0.63 | 19.7 |
| Escherichia coli | Absence | ND | ND | ND | |
| Staphylococcus aureus | Absence | ND | ND | ND | |
| Candida albicans | Absence | ND | ND | ND | |
| Pseudomonas aeruginosa | Absence | ND | ND | ND | |
| Name of the Product | Main Category | Country of Origin | Staphylococcus aureus (per g) | Yeast and Mold CFU/g | Candida albicans (per g) | Aerobic Mesophilic Bacteria CFU/g | Pseudomonas aeruginosa (per g) |
|---|---|---|---|---|---|---|---|
| Body lotion | Body care preparation | UAE | ND | <10 | ND | <10 | ND |
| Facial cleanser | Face and neck care | UAE | ND | <10 | ND | <10 | ND |
| Petroleum jelly | Body care preparation | UAE | ND | <10 | ND | <10 | ND |
| Petroleum jelly | Body care preparation | UAE | ND | <10 | ND | <10 | ND |
| Henna | Hair and scalp care | India | ND | 100 | ND | 500 | ND |
| Henna | Hair and scalp care | India | ND | 7500 | ND | 57,000 | ND |
| Herbal henna | Hair and scalp care | India | ND | 3800 | ND | 13,800 | ND |
| Shampoo | Hair and scalp care | UAE | ND | <10 | ND | <10 | ND |
| Hair crème | Hair and scalp care | UAE | ND | <10 | ND | <10 | ND |
| Hair styling gel | Hair and scalp care | EU | ND | <10 | ND | <10 | ND |
| Shower gel | Body care preparation | EU | ND | <10 | ND | <10 | ND |
| Face moisturizing cream | Face and neck care | UAE | ND | 2369 | ND | 6876 | ND |
| Facial scrub | Face and neck care | UAE | ND | <10 | ND | <10 | ND |
| Body crème milk | Body care preparation | USA | ND | 1500 | ND | 1235 | ND |
| Body wash | Body care preparation | EU | ND | <10 | ND | <10 | ND |
| Hair styling gel | Hair and scalp care | Korea | ND | <10 | ND | <10 | ND |
| Hair oil | Hair and scalp care | UAE | ND | <10 | ND | <10 | ND |
| Eyeliner | Eye care | UAE | ND | <10 | ND | 50 | ND |
| Hair oil | Hair and scalp care | EU | ND | <10 | ND | <10 | ND |
| Moisturizing body cream | Body care preparation | UAE | ND | <10 | ND | <10 | ND |
| Smooth hair cream | Hair and scalp care | India | ND | <10 | ND | <10 | ND |
| Vaseline body butter | Body care preparation | UAE | ND | <10 | ND | <10 | ND |
| Eyeliner | Eye care | UAE | ND | <10 | ND | <10 | ND |
| Moisturizing body emulsion | Body care preparation | China | ND | <10 | ND | <10 | ND |
| Facial nourishing cleanser | Face and neck care | China | ND | <10 | ND | <10 | ND |
| Mouthwash | Oral hygiene product | USA | ND | <10 | ND | <10 | ND |
| Vaseline body cream | Body care preparation | China | ND | <10 | ND | <10 | ND |
| Face toner | Face and neck care | China | ND | <10 | ND | <10 | ND |
| Petroleum jelly | Body care preparation | UAE | ND | <10 | ND | <10 | ND |
| Fair cream | Hair and scalp care | India | ND | <10 | ND | <10 | ND |
| Anti-stretch-mark moisturizing body cream | Body care preparation | Korea | ND | <10 | ND | <10 | ND |
| Anticellulite stretch mark firming body cream | Body care preparation | USA | ND | 2462 | ND | 1120 | ND |
| Cleansing facial soap | Face and neck care | Korea | ND | <10 | ND | <10 | ND |
| Face cleansing and brightening | Face and neck care | India | ND | <10 | ND | <10 | ND |
| Hair loss shampoo | Hair and scalp care | UAE | ND | <10 | ND | <10 | ND |
| Sunscreen | Body care preparation | USA | ND | <10 | ND | <10 | ND |
| Mouth rinse | Oral hygiene product | EU | ND | <10 | ND | <10 | ND |
| Face mask | Face and neck care | Middle East | ND | <10 | ND | <10 | ND |
| Hair styling gel | Hair and scalp care | China | ND | <10 | ND | <10 | ND |
| Toothpaste and mouth rinse | Oral hygiene product | USA | ND | <10 | ND | <10 | ND |
| Facial cleanser | Face and neck care | India | ND | <10 | ND | <10 | ND |
| Hair styling gel | Hair and scalp care | Middle East | ND | 2345 | ND | 1700 | ND |
| Soap | Body care preparation | Middle East | ND | <10 | ND | <10 | ND |
| Facial scrub | Face and neck care | EU | ND | <10 | ND | <10 | ND |
| Facial scrub | Face and neck care | Middle East | ND | <10 | ND | 4200 | ND |
| Mouthwash | Oral hygiene product | UAE | ND | <10 | ND | <10 | ND |
| Moisturizing body lotion | Body care preparation | EU | ND | 2000 | ND | <10 | ND |
| Hair styling gel | Hair and scalp care | UAE | ND | <10 | ND | <10 | ND |
| Toothpaste | Oral hygiene product | UAE | ND | <10 | ND | <10 | ND |
| Face foundation | Face and neck care | USA | ND | <10 | ND | <10 | ND |
| Mouthwash | Oral hygiene product | USA | ND | 1239 | ND | <10 | ND |
| Face cleaning water | Face and neck care | Korea | ND | <10 | ND | <10 | ND |
| Antistain body cream | Body care preparation | Middle East | ND | 1246 | ND | 5780 | ND |
| Firming and wrinkle removal body cream | Body care preparation | Middle East | ND | <10 | ND | <10 | ND |
| Body cream | Body care preparation | Middle East | ND | <10 | ND | <10 | ND |
| Body cream | Body care preparation | USA | ND | <10 | ND | <10 | ND |
| Face wipe off fleck | Face and neck care | Middle East | ND | <10 | ND | <10 | ND |
| Whitening skin cream | Body care preparation | Middle East | ND | <10 | ND | <10 | ND |
| Extensions volume mascara | Eye care | China | ND | <10 | ND | <10 | ND |
| Fluoride toothpaste | Oral hygiene product | EU | ND | <10 | ND | <10 | ND |
| Toothpaste | Oral hygiene product | China | ND | <10 | ND | <10 | ND |
| Toothpaste | Oral hygiene product | China | ND | <10 | ND | <10 | ND |
| Talc body powder | Body care preparation | UAE | ND | <10 | ND | 200 | ND |
| Talc body powder | Body care preparation | UAE | ND | 1235 | ND | 8965 | ND |
| Talc body powder | Body care preparation | UAE | ND | <10 | ND | <10 | ND |
| Talc body powder | Body care preparation | UAE | ND | <10 | ND | 400 | ND |
| Talc body powder | Body care preparation | UAE | ND | <10 | ND | 600 | ND |
| Talc body powder | Body care preparation | UAE | ND | 1872 | ND | <10 | ND |
| Talc body powder | Body care preparation | UAE | ND | <10 | ND | <10 | ND |
| Talc body powder | Body care preparation | UAE | ND | 100 | ND | 1200 | ND |
| Talc body powder | Body care preparation | UAE | ND | 100 | ND | 1500 | ND |
| Hair styling gel | Hair and scalp care | India | ND | 700 | ND | 21,000 | ND |
| Henna | Hair and scalp care | India | ND | <10 | ND | <10 | ND |
| Body oil | Body care preparation | EU | ND | <10 | ND | <10 | ND |
| Sun cream | Body care preparation | Korea | ND | <10 | ND | <10 | ND |
| Body oil | Body care preparation | Korea | ND | <10 | ND | <10 | ND |
| Face scrub | Face and neck care | India | ND | <10 | ND | <10 | ND |
| Deodorant powder | Body care preparation | EU | ND | <10 | ND | 1800 | ND |
| Face cream | Face and neck care | EU | ND | <10 | ND | <10 | ND |
| Eyeliner | Eye care | EU | ND | <10 | ND | <10 | ND |
| Milk body mask | Body care preparation | Korea | ND | <10 | ND | <10 | ND |
| Fragrance talc body powder | Body care preparation | USA | ND | <10 | ND | <10 | ND |
| Mouthwash | Oral hygiene product | Korea | ND | <10 | ND | <10 | ND |
| Dead Sea concentrated water | Body care preparation | Middle East | 0 | <10 | ND | <10 | ND |
| Eyeliner | Eye care | India | 0 | <10 | ND | <10 | ND |
| Eyebrow powder | Eye care | China | 0 | <10 | ND | <10 | ND |
| Eyes brow gel | Eye care | EU | 0 | 1287 | ND | <10 | ND |
| Facial balancing cleanser | Face and neck care | India | 0 | <10 | ND | <10 | ND |
| Hair conditioner | Hair and scalp care | USA | 0 | <10 | ND | <10 | ND |
| Face powder | Face and neck care | EU | 0 | <10 | ND | <10 | ND |
| Facial argan oil scrub | Face and neck care | UAE | 0 | <10 | ND | <10 | ND |
| Detoxifying scrub | Body care preparation | UAE | 0 | <10 | ND | <10 | ND |
| Hair oil | Hair and scalp care | India | 0 | <10 | ND | <10 | ND |
| Heat powder | Body care preparation | India | 0 | 300 | ND | 1900 | ND |
| Face cleansing gel | Face and neck care | UAE | 0 | <10 | ND | <10 | ND |
| Under eye cream | Eye care | UAE | 0 | <10 | ND | <10 | ND |
| Shampoo | Hair and scalp care | EU | 0 | <10 | ND | <10 | ND |
| Shampoo | Hair and scalp care | USA | 0 | <10 | ND | <10 | ND |
| Toothpaste | Oral hygiene product | USA | 0 | 7623 | ND | 1234 | ND |
| Facial scrub | Face and neck care | UAE | 0 | <10 | ND | <10 | ND |
| Incidence of Microbial Contamination | |||
|---|---|---|---|
| Aerobic Mesophilic Bacteria | Yeast and Mold | All | |
| Total | 15 (15%) | 13 (13%) | 19 (19%) |
| Categories | |||
| Body care preparations | 8 (20.5%) | 6 (15.4%) | 10 (25.6%) |
| Eye care | 0 | 1 (12.5%) | 1 (12.5%) |
| Face and neck care | 2 (9.5%) | 1 (4.8%) | 2 (9.5%) |
| Hair and scalp care | 4 (19.0%) | 3 (14.3%) | 4 (19%) |
| Oral hygiene products | 1 (9.1%) | 2 (18.2%) | 2 (18.2%) |
| p-Value | 0.492 | 0.782 | 0.633 |
| Country of origin | |||
| China | 0 | 0 | 0 |
| EU | 1 (6.7%) | 2 (13.3%) | 3 (20.0%) |
| India | 4 (28.6%) | 2 (14.3%) | 4 (28.6%) |
| Korea | 0 | 0 | 0 |
| Middle East | 3 (30.0%) | 2 (20.0%) | 3 (30.0%) |
| UAE | 4 (12.5%) | 3 (9.4%) | 5 (15.6%) |
| USA | 3 (25.0%) | 4 (33.3%) | 4 (33.3%) |
| p-Value | 0.191 | 0.257 | 0.281 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jairoun, A.A.; Al-Hemyari, S.S.; Shahwan, M.; Zyoud, S.H. An Investigation into Incidences of Microbial Contamination in Cosmeceuticals in the UAE: Imbalances between Preservation and Microbial Contamination. Cosmetics 2020, 7, 92. https://doi.org/10.3390/cosmetics7040092
Jairoun AA, Al-Hemyari SS, Shahwan M, Zyoud SH. An Investigation into Incidences of Microbial Contamination in Cosmeceuticals in the UAE: Imbalances between Preservation and Microbial Contamination. Cosmetics. 2020; 7(4):92. https://doi.org/10.3390/cosmetics7040092
Chicago/Turabian StyleJairoun, Ammar Abdulrahman, Sabaa Saleh Al-Hemyari, Moyad Shahwan, and Sa’ed H. Zyoud. 2020. "An Investigation into Incidences of Microbial Contamination in Cosmeceuticals in the UAE: Imbalances between Preservation and Microbial Contamination" Cosmetics 7, no. 4: 92. https://doi.org/10.3390/cosmetics7040092
APA StyleJairoun, A. A., Al-Hemyari, S. S., Shahwan, M., & Zyoud, S. H. (2020). An Investigation into Incidences of Microbial Contamination in Cosmeceuticals in the UAE: Imbalances between Preservation and Microbial Contamination. Cosmetics, 7(4), 92. https://doi.org/10.3390/cosmetics7040092





