Abstract
Biotechnological production of lactic acid (LA) is based on the so-called first generation feedstocks, meaning sugars derived from food and feed crops such as corn, sugarcane and cassava. The aim of this study was to exploit the potential of a second generation resource: Common reed (Phragmites australis) is a powerfully reproducing sweet grass which grows in wetlands and creates vast monocultural populations. This lignocellulose biomass bears the possibility to be refined to value-added products, without competing with agro industrial land. Besides utilizing reed as a renewable and inexpensive substrate, low-cost nutritional supplementation was analyzed for the fermentation of thermophilic Bacillus coagulans. Various nutritional sources such as baker’s and brewer’s yeast, lucerne green juice and tryptone were investigated for the replacement of yeast extract. The structure of the lignocellulosic material was tackled by chemical treatment (1% NaOH) and enzymatic hydrolysis (Cellic® CTec2). B. coagulans DSM ID 14-300 was employed for the homofermentative conversion of the released hexose and pentose sugars to polymerizable L-(+)-LA of over 99.5% optical purity. The addition of autolyzed baker’s yeast led to the best results of fermentation, enabling an LA titer of 28.3 g L−1 and a yield of 91.6%.
1. Introduction
Lactic acid (LA), or 2-hydroxy propionic acid, is considered to be one of the main chemical building blocks for enabling the transition to a sustainable bio-based economy [1,2]. Traditionally used in the food industry as preserver and mild acidifier, the applications for LA have broadened since its capacity to polymerize under certain conditions to polylactic acid (PLA) has been put to use [3]. Naturally occurring in the two stereochemical forms L-(+) and D-(−), the L-LA is preferably used for the production of PLA [4]. Initially, PLA had been introduced for medical applications like bone fixation devices, stents or drug delivery systems, benefiting from L-LA’s compatibility with the human body [5]. Nowadays, with the decline of PLA’s competitive cost, its production for various industrial applications is on the rise. Various consumer goods packaging like bags, cups or bottles are already available on the market [4,6].
The most commercial value is gained by PLA of high molecular weight (MW), and this property depends on the stereochemical purity of the material. Consequently, the optical purity of the starting material, the initial L-LA, has to meet this requirement [4]. Biotechnology proved to be advantageous over potential chemical routes, not only providing higher optical purities of the product, but also offering the utilization of inexpensive renewable substrates [3]. NatureWorks LLC, for instance, which is jointly owned by PTT Global Chemical and Cargill and is the major producer of PLA today, utilizes LA originating from the fermentation of corn for their process [4]. Though feasible, research nowadays is focused on feedstocks of the so-called second generation to avoid competition with the provision of food [7,8].
Common reed (Phragmites australis) is one example for a renewable substrate that bears fermentable sugars and grows abundantly in wetlands, therefore not competing with land intended for the production of food. It is a highly productive grass that reaches heights of several meters and has been harvested for uses like thatching, in construction and pulp & paper or landscape managing [9]. Macrophytes like reed exhibit many properties that support the water treatment of natural or constructed wetlands, being advantageous for erosion control, filtration, surface provision for microbial growth and nutrient uptake [10]. Notably, sustainable harvesting of reed is beneficial to the wetland ecosystem, while at the same time expected to provide viable income to local communities [11]. In their recent study, Baibagyssov et al. [12] estimated a quantity of at least 30 million tonnes of annually available biomass worldwide.
Regarding reed, few studies focus on the production of LA: Perttunen et al. [13] utilized hemicellulose liquor from reed, which occurred as a by-product in the pulping process for fermentation by L. pentosus. Zhang et al. [14] pretreated reed with sulfuric acid and compared separate and integrated fermentation strategies by employing B. coagulans IPE22. The best result was attained by a simultaneous saccharification and co-fermentation approach (SSCF), leading to an LA-titer of 35.05 g L−1 after a course of 55 h [14]. Reports on other grassy crops like corn stover are more frequent in the literature. Fermentation of acid pretreated corn stover by B. coagulans MXL-9 resulted in 40.2 g L−1 LA with a productivity of 0.5 g L−1 h−1 [15]. Final LA-titers could be increased by prior concentration of corn stover hydrolysate (e.g., by vacuum evaporation) or through the implementation of fed-batch approaches [16,17,18,19]. Fed-batch SSF of alkaline pretreated corn stover by B. coagulans LA204 resulted in 97.6 g L−1 L-LA at a productivity of 1.63 g L−1 h−1 [18].
Besides the utilization of inexpensive carbon sources like lignocellulose residues, the replacement of costly nutritional supplements is another option to improve the overall process costs [20]. Commonly, yeast extract (YE) is added to the fermentation as a source of nitrogen for microbial growth. Several studies have tested low-cost alternatives, starting out with trials on lactic acid bacteria (LAB) of the genus Lactobacillus and more recently, focusing on Bacillus strains. Since LAB are fastidious concerning their nutrient requirement, replacement of YE can be challenging in comparison to Bacillus strains, which are able to grow in simple mediums [21]. Studies employing corn steep liquor (CSL) or powder (CSP) in fermentations of L. delbrueckii and L. pentosus noted its cost-effectiveness in comparison to YE, though admitting lower performances [22,23]. Concerning several Bacillus strains as well as Sporolactobacillus sp. CASD, trials with enzymatic hydrolyzed peanut meal showed promising results [24,25,26]. Fermentations with B. coagulans in particular have been supplemented with CSL, CSP, peptone, dry yeast cells (DYC) and excess sludge hydrolysate [15,18,27,28]. Best results were achieved in a study by Ooi & Wu [27], concerning the SSF of cassava starch. Compared to supplementation with YE, adding autolyzed DYC to the fermentation led to an increase in productivity without compromising values of titer nor yield [27].
In this study, we investigated reed biomass as an abundant and inexpensive renewable resource for the production of polymerizable L-LA. The recalcitrant chemical structure of the reed lignocellulose was tackled by chemical and enzymatic hydrolysis. B. coagulans 14-300 was employed for the biotechnological conversion of the released sugars to L-LA. To meet its nitrogen demand, various nutritional sources such as baker’s yeast, lucerne green juice or corn steep liquor were investigated as low-cost alternatives to YE. So far, studies on the biotechnological production of L-LA from reed are rarely found in literature. Furthermore, to our knowledge, only one study tested the feasibility of alternative nutrient sources for the fermentation of lignocellulose substrates in respect to B. coagulans [15].
2. Materials and Methods
2.1. Raw Material & Processing
Reed stems (P. australis) were kindly provided by the HISS REET Schilfrohrhandel GmbH (Bad Oldesloe, Germany) and mechanically processed by chopping and grinding until an average size of 2 mm × 2–10 mm was reached. The reed stems contained 92.8% of total solids (TS) and an ash content of 5.36% (w/w TS basis). Various substances were obtained to investigate their potential as nutrient source for the fermentation of B. coagulans 14-300: Yeast extract and tryptone (Carl Roth, Karlsruhe, Germany), peptone from soymeal and malt extract (Merck Millipore, Darmstadt, Germany), baker’s yeast (RUF, Quakenbrück, Germany), brewer’s yeast (T.T. Baits, Baiersdorf; Germany) and insect meal (ground Hermetia illucens; Hermetia, Baruth, Germany). Additionally, lucerne green juice (GJ) was investigated, since it showed promising results concerning the nutrition of L. paracasei [29]. GJ was provided as stated in our former publication [29], already in liquid form and not further processed. For all other nutrients, stock solutions were prepared by suspension in deionized water and autoclaved for 15 min at 121 °C. Furthermore, as proposed by Ooi and Wu [27], an autolysis of the two yeast cell sources—namely, baker’s and brewer’s yeast—was carried out. For this, stock solutions were kept overnight at 55 °C and 100 rpm in a shaking incubator.
2.2. Chemical and Enzymatic Pretreatment
The ground reed stems were chemically and enzymatically treated to gain a lignocellulosic hydrolysate for the fermentation studies. As chemical agents, sodium hydroxide (NaOH) and sulfuric acid (H2SO4) were tested. Subsequent enzymatic hydrolysis was conducted with Cellic® CTec2 (Novozymes, Denmark). Preliminarily, experiments were carried out in 0.3 L shaking flasks working with a volume of 50 mL and a load of 10% (w/v). NaOH or H2SO4 (0.5%–4%) were added to the reed and the flasks were kept for 30 min at 121 °C in the autoclave. After adjustment to pH 5, an enzymatic hydrolysis was conducted by adding Cellic® CTec2 to each shaking flask (0.053 mL g−1 cellulose or 14 FPU g−1 cellulose), keeping them at 50 °C and 150 rpm for 48 h in a shaking incubator. For scale-up, 20% (w/v) reed was pretreated with 1% NaOH in a 5 L Schott bottle with a working volume of 2 L. The material was kept for 30 min at 121 °C in the autoclave and, after cooling, transferred to a 5 L lab scale fermenter unit equipped with 2 × 6 blade Rushton turbines. After adjustment to pH 5 with 50% H2SO4, enzymatic hydrolysis was conducted for 48 h at 50 °C and 600 rpm. The obtained hydrolysate was passed through a mesh with 150 µm pore size to receive a medium suitable for the fermentation studies.
2.3. Bacterial Strain and Preculture Conditions
Bacillus coagulans DSM ID 14-300 (short 14-300) was chosen, since it performed well in hemicellulose hydrolysate of an earlier study and exhibited tolerance against typical inhibitor compounds found among such substrates [30]. B. coagulans 14-300 preculture was grown in 60 mL MRS (Merck Millipore, Darmstadt, Germany), supplemented with 0.67 g of EVERZIT® Dol (0.5–2.5 mm sized CaCO3∙MgCO3; Evers, Hopsten, Germany) for pH regulation and incubated for 14 h at 40 °C and 100 rpm.
2.4. Fermentation Studies with Varying Nutrient Sources
Preliminary shaking flask experiments were carried out to determine nutrient sources that were most favorable for the fermentation of reed hydrolysate by B. coagulans 14-300. Experiments were conducted in 0.25 L flasks, working with a total volume of 100 mL. Nutrient stock solution was added, reaching a nutrient concentration of 5 g L−1 within each flask. Concerning GJ, the same volume was added to the flask, resulting in a concentration of 10% (v/v). After the addition of 6% (v/v) preculture, the flasks were kept at 52 °C and 100 rpm in a shaking incubator and samples were taken after 2, 4 and 24 h. The first shaking flask experiment included all the obtained nutrient sources and was conducted with synthetic media supplemented with 1.2 g EVERZIT® Dol (Evers, Germany) as a buffer. Synthetic media composed of 60% glucose, 35% xylose and 5% arabinose, to reproduce the monosaccharide fractions found in NaOH pretreated reed hydrolysate. A second shaking flask experiment was performed to test a selected number of nutrient sources, this time using reed hydrolysate as a substrate. For lab scale fermentations, the EloFerm-dual System (Biotronix, Hennigsdorf, Germany) equipped with 0.5 L glass reactors and stirring rods was employed. Reed hydrolysate was used as the substrate, running the fermentations with a working volume of 0.25 L at 52 °C and 400 rpm. Nutrients were added, reaching a concentration of 5 g L−1, except for autolyzed brewer’s yeast, which was additionally tested for a concentration of 10 g L−1. Neutralizing agent (20% NaOH) was automatically added throughout the experiment to keep the medium stable at pH 6. Preculture concentration of B. coagulans 14-300 was 6% (v/v). All experiments were performed in duplicates.
2.5. Analytics
The lignocellulose composition of the reed stems was analyzed according to laboratory analytical procedures (LAP) suggested by the National Renewable Energy Laboratory (NREL) [31,32,33]. The cellulase activity of Cellic® CTec2 was measured following the guidelines of the NREL, using an adapted version of the reagents as found in Arnaut de Toledo et al. to avoid the application of hazardous phenol [34,35]. The total nitrogen content was analyzed by the Kjeldahl method using the Kjeldahl Sampler System K-370/371 (BÜCHI, Essen, Germany) [36]. Lactic acid, acetic acid and monosaccharide concentrations of glucose, xylose and arabinose were measured via high performance liquid chromatography (HPLC; DIONEX, Sunnyvale, CA, USA). As HPLC column, Eurokat H (300 mm × 8 mm × 10 µm; Knauer, Berlin, Germany) was used, running 10 µL sample at a flow rate of 0.8 mL min−1 within a mobile phase of 5 mM H2SO4. The HPLC was equipped with a refractive index detector RI-71 (SHODEX, Tokyo, Japan). Determination of the L-/D-isomer ratio in the sample was also conducted via HPLC, using a Chiralpak®MA(+) column (50 mm × 4.6 mm × 3 µm; DAICEL, Tokyo, Japan) with a 2 mM CuSO4 mobile phase and an ultraviolet detector.
2.6. Calculations
The yield of enzymatic hydrolysis was calculated by dividing the total released monosaccharides by the theoretical amount of monosaccharides in g g−1. Converting factors for glucan and xylan were 0.9 and 0.88, respectively, as reported in literature [37]. Regarding fermentation experiments, the addition of neutralizing agent led to the dilution of the media and sample withdrawal to a loss of monosaccharides, as well as produced LA. These factors were taken into account for the calculation of the fermentation yield. Therefore, fermentation yield was calculated by dividing the total produced LA by the available monosaccharides in the fermentation broth in g g−1. Average and maximum productivity were determined by dividing the measured LA concentration by the fermentation duration or the specific time interval in g L−1 h−1.
3. Results
3.1. Reed Analysis & Pretreatment
Lignocellulose of the reed stems was analyzed according to the guidelines of the NREL [31,32,33]. However, since no standard has yet been provided for P. australis by the National Institute of Standards and Technology (NIST), method errors cannot be excluded [38]. Nevertheless, as can be seen in Table 1, our data lies well within the range of earlier reported values concerning the lignocellulose composition of P. australis analyzed according to NREL guidelines [14,39].

Table 1.
Lignocellulose composition of reed (Phragmites australis) 1.
Sodium hydroxide (NaOH) and sulfuric acid (H2SO4) were chosen as chemical agents for affecting the lignocellulose structure of reed, employing concentrations ranging from 0.5%–4%. Biomass with added chemical agent was kept for 30 min at 121 °C in the autoclave, followed by an enzymatic hydrolysis of 48 h with Cellic® CTec2. In Figure 1, results of shaking flask experiments with diluted NaOH and H2SO4 are compared. The blank displays an enzymatic hydrolysis of the reed stems without prior treatment with chemical agent. This alone leads to a hydrolysis yield of around 10%. The exposure of reed stems to each chemical agent led to an improvement of the hydrolysis yield: The experiments with H2SO4 present the highest yields, reaching 49.7% for an addition of 2% acid. Shaking flask trials with NaOH resulted in a lower saccharification yield, producing a maximum of 35.5% by adding 1% of base (Figure 1a). Though pretreatment with acid resulted in the highest yield, the glucose ratio was also taken into account for choosing an optimum fermentation medium. While B. coagulans is able to metabolize hexose as well as pentose sugars, glucose is consumed preferably causing higher productivity in comparison to xylose [16,30]. The glucose ratios for all the pretreatment experiments are shown in Figure 1b. The blank experiment, though exhibiting the lowest hydrolysis yield, led to the highest glucose ratio of around 70%, which itself is due to the main function of Cellic® CTec2 being the enzymatic degradation of cellulose. Though hemicellulose is tackled as well, it is affected to a lesser extent. Concerning the pretreatment with chemical agents, the share of glucose in the monosaccharide solution descents with an increasing amount of added H2SO4. At the highest yield of hydrolysis (2% H2SO4), only 29.1% of the monosaccharides are of glucose. On the other hand, applying 1% of NaOH led to 60.9% of glucose in the solution.
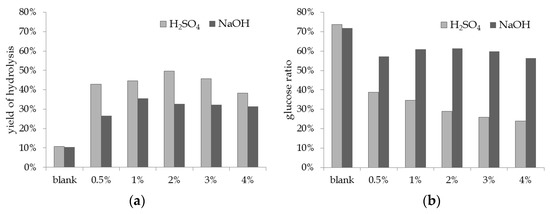
Figure 1.
Pretreatment of reed stems: (a) yield of hydrolysis and (b) glucose ratio after chemical pretreatment and 48 h of enzymatic hydrolysis with Cellic® CTec2 (shaking flask experiments).
These findings go well in accordance with the literature. An acid pretreatment of lignocellulose leads to a hydrolysis of the hemicellulose, resulting in higher proportions of pentose sugars in the hydrolysate [40]. The application of base leads to the swelling of the biomass and therefore increases the surface of the material. Extent of polymerization and crystallinity of the cellulose are reduced. Bases particularly affect the lignin structure by loosening chemical bonds between the lignin components and the carbohydrates, without promoting the solubilization of hemicellulose [40,41].
Despite the fact that the actual monosaccharide yield was higher when applying sulfuric acid as the chemical agent, lower chemical input and higher glucose ratio was favored for our process. Based on these results, a chemical treatment with 1% NaOH was chosen as a basis for the following fermentation experiments.
3.2. Shaking Flask Fermentation Experiments
To study the effect of various nutrient sources on B. coagulans 14-300, a shaking flask experiment with simplified synthetic medium was carried out. Medium composition was based on the monosaccharide present in reed hydrolysate, containing 60% of glucose and 40% of pentose sugars. Shaking flasks contained a concentration of 5 g L−1 nutrient, except for lucerne green juice, which was added at a concentration of 10% (v/v). Abbreviations and N-Kjeldahl content for all the tested nutrients are listed in Table 2. Tryptone contains the highest content of 133.2 mg gFM−1 nitrogen, followed by yeast extract, peptone from soymeal and brewer’s yeast. All other sources tested for nutrient supplementation hold N-Kjeldahl contents of lower than 70 mg gFM−1.

Table 2.
List of all the tested nutrient sources: Abbreviations, dry matter (DM) and total nitrogen content (NKjel = Nitrogen measured by the Kjeldahl method, FM = fresh matter, SF = shaking flask).
In Figure 2a the results of the shaking flask experiment are displayed against the total nitrogen content of the nutrient sources. The best lactic acid concentration of 30.2 g L−1 was obtained by supplementing the fermentation with yeast extract, followed by the addition of tryptone and lucerne green juice, reaching 22.6 and 20.7 g L−1, respectively. It can be noted that an increase in total nitrogen content of the nutrient applied did not necessarily result in higher LA concentration. Besides the total nitrogen content, specific nutrient components can play a role for the growth of B. coagulans. Marshall & Beers [42] found five strains of B. coagulans requiring thiamine as well as biotin, while a difference in temperature regimes (37, 45 & 55 °C) necessitated additional components such as aspartic acid and folic acid.
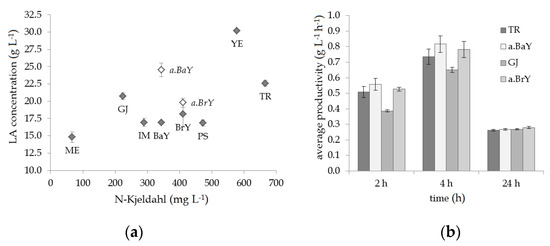
Figure 2.
Results of the shaking flask experiments with various nutrient sources (B. coagulans 14-300, 100 mL working volume, 52 °C): (a) fermentation of simplified medium with synthetic sugars (hexose:pentose ratio = 3:2); (b) fermentation of reed hydrolysate (pretreated with 1% NaOH and Cellic® CTec2). Concentrations of nutrients were 5 g L−1 or 10% (v/v) for lucerne green juice (GJ).
For two of the tested nutrients, baker’s yeast (BaY) and brewer’s yeast (BrY), a modification of an autolysis method described by Ooi & Wu [27] was applied to enhance the release of nitrogen and other nutritional compounds. Experiments with untreated baker’s and brewer’s yeast resulted in 16.9 and 18.1 g L−1 LA. Employing autolysis beforehand led to increased LA concentrations of 24.5 and 19.8 g L−1, respectively (see Figure 2a).
Nutrient sources which led to the highest LA titers were tested in a second shaking flask experiment, this time using reed stem hydrolysate as the fermentation medium. The average productivity for each point of sampling allowed a comparison regarding the performance of the individual fermentations (see Figure 2b). At 2 h and 4 h, the fermentations with autolyzed baker’s yeast (a.BaY) displayed the highest average productivity of 0.56 and 0.82 g L−1 h−1, followed by the ones supplied with autolyzed brewer’s yeast (a.BrY) and tryptone. Supplementation with lucerne green juice resulted in a slightly lower average productivity compared to the other nutrient sources. To limit the expense of substrate, only the three nutrients, which led to the best fermentation performances were further tested in lab-scale fermentations.
3.3. Lab-Scale Fermentation Experiments
Two batches of reed hydrolysate were utilized as medium for the lab-scale fermentations. Though their total sugar content was similar, the composition of monosaccharides deviated slightly. The first batch (B1) contained 34.9 ± 1.4 g L−1 total monosaccharides comprised of 66.2% glucose, 30.5% xylose and 3.3% arabinose. The second batch (B2) held 35.0 ± 0.8 g L−1 of total monosaccharides with a slight increased ratio of the hexose sugars, namely 68.4% glucose, 28.8% xylose and 2.8% arabinose. Both batches contained acetic acid with a concentration of 6.6 ± 0.2 g L−1 and 7.2 ± 0.2 g L−1, respectively. Reed hydrolysate of B1 was employed for the fermentations supplemented with YE, TR and a.BrY. Batch B2 was used for application of a.BaY and 10 g L−1 a.BrY, as well as for the fermentations without added nutrients.
The results for the fermentations of reed hydrolysate by B. coagulans 14-300 are shown in Figure 3. Running the experiment without application of any nutrients resulted in no growth of B. coagulans 14-300 (data not shown). Figure 3a displays the course of the fermentations over time. Observing the fermentations with supplementation of 5 g L−1 nutrient sources, YE and a.BaY led to the best performances. After 48 h of fermentation, LA titers resulted in 25.2 and 28.3 g L−1, respectively. Notably, the produced L-LA had a high optical purity of over 99.5%. The increase of the a.BrY concentration from 5 to 10 g L−1, led to an improvement in sugar consumption regarding the first 10 h of fermentation. However, final titer, overall productivity and yield were not enhanced by this approach. After 48 h, yields of fermentation ranged between 75%–80% for all experiments except the ones supplemented with a.BaY, which resulted in an average yield of 91.6% (see Figure 3b). However, according to ANOVA analysis, none of the results deviates significantly from the group of 48 h which is due to the deviations between the duplicate runs. It should also be noted that the fermentations supplemented with a.BaY were conducted with reed hydrolysate of B2, which held a slightly increased amount of glucose. Though no positive effect of a.BaY can be statistically validated for our study, still, it can be noted that a.BaY supports B. coagulans 14-300 as good as YE concerning the LA titer and the yield of fermentation. Furthermore, the average productivity of the fermentations supplemented with a.BaY was comparable to that conducted with YE (see Figure 3c). After 48 h of fermentation, the average productivity for B. coagulans 14-300 was 0.53 and 0.59 g L-1 h−1 regarding the addition of YE and a.BaY, respectively. Maximum productivity of 4.5 g L−1 h−1 was observed for YE application, followed by 3.5 g L−1 h−1 regarding a.BaY and 3.3 g L−1 h−1 for the addition of 10 g L−1 a.BrY. These findings are compliant with the study conducted by Ooi & Wu [27]. Concerning the utilization of cassava starch, autolyzed DYC were found to be comparable to YE in respect to resulting LA titer and yield. Moreover, the average productivity of B. coagulans was slightly enhanced [27].
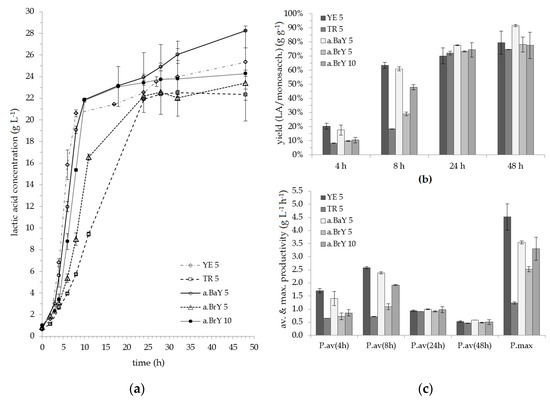
Figure 3.
Results of the lab scale fermentations with the nutrient sources YE, TR, a.BaY & a.BrY (B. coagulans 14-300, 0.25 L working volume, 52 °C, pH 6; 5 or 10 = nutrient concentration of 5 or 10 g L−1): (a) course of fermentation; (b) yield of fermentation; (c) average (P.av) and maximum productivity (P.max).
The acetic acid concentration was measured throughout the fermentations, and although the final concentrations did not deviate significantly from the beginning, minor changes could be observed over the course of fermentation (data not shown). A decrease in acetic acid concentration was noted during the phase of glucose consumption, since addition of neutralizing agent resulted in slight dilution of the medium. After the depletion of glucose, a slight increase of acetic acid was detected, indicating low by-product formation by B. coagulans 14-300. Nevertheless, for all lab-scale experiments the increase in acetic acid concentration was calculated to be less than 0.8 g L−1 after 48 h of fermentation.
In Figure 4a,b the course of monosaccharide consumption and LA production, regarding the fermentations supplemented with YE and a.BaY, are shown for a detailed comparison. Utilizing YE, B. coagulans 14-300 consumed the glucose rapidly within the first 8 h of the fermentation, exerting a volumetric rate of glucose consumption of around 2.7 g L−1 h−1. Notably, the pentoses are already utilized during this stage, indicating that carbon catabolite repression (CCR) is reduced. However, the average volumetric rates of xylose and arabinose consumption were 0.15 and 0.05 g L−1 h−1, leading to a second fermentation stage with lowered productivity after glucose was depleted, resulting in a productivity of 2.58 g L−1 h−1 for the first 8 h and 0.12 g L−1 h−1 for the following 38 h of fermentation. In the fermentations supplemented with a.BaY, B. coagulans 14-300 dealt with slightly decreased ratios of pentose sugars, seemingly consuming them more efficiently and leading to an increased L-LA titer and fermentation yield. Again, though statistical analysis does not support the conclusion of a positive effect for this study, our results still indicate a.BaY as a promising alternative for the use of YE.
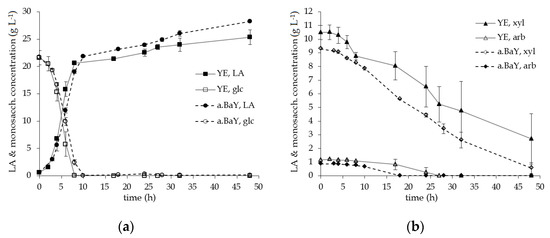
Figure 4.
Sugar consumption and LA production for the lab scale fermentations with addition of YE and a.BaY as nutrient sources (B. coagulans 14-300, 0.25 L working volume, 52 °C, pH 6, 5 g L−1 YE or a.BaY concentration): (a) consumption of glucose (glc) and production of LA over time; (b) consumption of xylose (xyl) and arabinose (arb) over time.
4. Discussion
To date, research on biotechnological production of LA is focused on feedstocks of the second generation, such as lignocellulose biomass, as well as municipal, industrial and agricultural wastes or residues [7,8,43]. In our study, we analyzed common reed as an inexpensive lignocellulose substrate for LA production. Reed grows abundantly and its harvest, if conducted in a sustainable way, is reported to be beneficial for its ecosystem and local communities [11]. Besides working with a renewable and inexpensive substrate, this study particularly compared various low-cost resources in respect to their applicability as nutrients for the fermentation of B. coagulans. Ground reed stems were pretreated with NaOH in the autoclave, followed by enzymatic hydrolysis. After filtration of the hydrolysate, thermophilic B. coagulans DSM ID 14-300 was employed for the fermentation of the liquid supernatant to optical pure L-LA. Supplementation with 5 g L−1 autolyzed baker’s yeast resulted in the highest values, concerning LA titer, yield and overall productivity (28.3 g L−1, 91.6% and 0.59 g L−1 h−1). However, maximum productivity of B. coagulans 14-300 was increased from 3.5 to 4.5 g L−1 h−1 when 5 g L−1 YE was applied, instead.
The LA titer achieved in this study is comparable to results from other studies describing the fermentation of pretreated reed. Besides SSF trials, Zhang et al. [14] conducted a batch experiment on the filtrated supernatant of acid pretreated reed. Modified MRS medium was added as a rather costly source of nutrients, containing various compounds such as peptone, beef extract and yeast extract. After 19 h of fermentation, a concentration of 20.8 g L−1 LA was achieved from 24.6 g L−1 mixed sugars by employing B. coagulans IPE22. Though the LA titer was increased to 35.05 g L−1 by performing SSCF of liquid and solid fraction together, the overall process duration was prolonged to 55 h [14]. Utilizing L. pentosus, Perttunen et al. [13] gained 33 g L−1 LA from hemicellulose liquor which contained 50 g L−1 of total sugars via 48 h of batch fermentation. However, since L. pentosus produces LA heterofermentatively, an additional 17 g L−1 of acetic acid were generated as by-product [13].
While results on LA production from reed are rarely found in literature [13,14], cereal grasses like corn are discussed more frequently [15,16,17,18,19]. Hu et al. [18] employed B. coagulans LA204 for SSF with alkali pretreated corn stover, gaining 29.9 g L−1 L-LA at a productivity of 0.50 g L−1 h−1, which is quite similar to the results found in our study. Titer and productivity were significantly improved to 97.6 g L−1 and 1.63 g L−1 h−1 when fed-batch SSF was performed instead [18]. Hence, further investigations on fermentation processes like SSF and fed-batch should be considered for future experiments on reed to enhance the overall performance.
Regarding the investigations on low-cost nutrients, Altaf et al. [44] were one of the few to test baker’s yeast lysate for the production of L-LA, studying the fermentation of L. amylophilus. Baker’s yeast was autoclaved and utilized in combination with red lentil gram to replace YE and peptone [44]. Though feasible, autoclaving is energy intensive and, according to Ooi & Wu [27], leads to the disintegration of valuable nutritional compounds. The latter autolyzed DYC at a mild temperature of 50 °C and utilized them for the fermentation of cassava starch by B. coagulans WCP 10-4. While the provision of 20 g L−1 YE was superior to non-treated DYC, its substitution with pre-autolyzed DYC let to an increase in productivity from 6.4 to 7.7 g L−1 h−1 [27].
To our knowledge, only one study investigated the effect of low-cost nutrition on the fermentation of lignocellulose residues by B. coagulans. Bischoff et al. [15] replaced YE with CSL for the fermentation of corn fiber hydrolysate to LA, resulting in reduced maximum and average productivity. While a titer of 45.6 g L−1 LA was achieved in SSF combined with 18 h of prehydrolysis, the productivity of B. coagulans was reduced to 0.21 g L−1 h−1 [15]. In our study, we demonstrated that autolyzed baker’s yeast could effectively be used for the fermentation of B. coagulans 14-300, using mixed sugars from lignocellulose hydrolysate as a substrate. Titer, yield and overall productivity were not compromised in comparison to experiments conducted with the addition of YE.
It has been argued that due to the energy consumption of autolyzing yeast cells overnight, this might not be economically feasible for practical applications [27]. On the other hand, it can be considered that an implemented production plant creates exhaust heat, which could be redirected and utilized for such an autolyzing process unit. Since expenses for YE were about ninefold higher at the time of our study, baker’s yeast appeared to be a promising nutrition alternative in regards to the feedstock costs. However, the actual viability of the autolysis method should be assessed in an economic analysis, provided, that data on scale-up and whole process concepts is available.
In conclusion, this study demonstrated that yeast extract could effectively be replaced by autolyzed baker’s yeast, regarding the fermentation of the lignocellulose substrate reed by B. coagulans 14-300. Reed is an abundant and inexpensive feedstock, whose cultivation does not compete with agricultural land uses. Alkali and enzymatically pretreated reed was filtrated and fermented to 28.3 g L−1 L-LA at an overall productivity of 0.59 g L−1 h−1. The generated L-LA had an optical purity of over 99.5%, making it an ideal candidate for the production of high molecular weight PLA.
Author Contributions
Conceptualization, L.S., R.S. and J.V.; methodology, L.S. and R.S.; validation, L.S.; formal analysis, L.S. and R.S.; investigation, L.R., R.S. and L.S.; resources, J.V.; data curation, R.S.; writing—original draft preparation, L.S.; writing—review and editing, J.V.; visualization, L.S.; supervision, J.V.; project administration, J.V.; funding acquisition, J.V. All authors have read and agreed to the published version of the manuscript.
Funding
This work was financially supported by the German Ministry for Education and Research (BMBF) through the grant “ReedBiom: Nachhaltige Biomassenutzung aus Schilf” (FKZ01DK18009). The publication of this article was funded by the Open Access Fund of the Leibniz Association.
Conflicts of Interest
The authors declare no conflict of interest. The funders had no role in the design of the study; in the collection, analyses, or interpretation of data; in the writing of the manuscript, or in the decision to publish the results.
References
- de Jong, E.; Stichnothe, H.; Bell, G.; Jørgensen, H. Bio-Based Chemicals: A 2020 Update; IEA Bioenergy Task 42; IEA Bioenergy, 2020; ISBN 978-1-910154-69-4. Available online: https://www.ieabioenergy.com/wp-content/uploads/2020/02/Bio-based-chemicals-a-2020-update-final-200213.pdf (accessed on 20 July 2020).
- Bozell, J.J.; Petersen, G.R. Technology development for the production of biobased products from biorefinery carbohydrates—The US Department of Energy’s “top 10” revisited. Green Chem. 2010, 12, 539–555. [Google Scholar] [CrossRef]
- Komesu, A.; Allan Rocha de Oliveira, J.; Helena da Silva Martins, L.; Lee, H.D.; Lee, M.Y.; Hwang, Y.S.; Cho, Y.H.; Kim, H.W. Lactic Acid Production to Purification: A Review. BioResources 2017, 12, 4364–4383. [Google Scholar] [CrossRef]
- Castro-Aguirre, E.; Iñiguez-Franco, F.; Samsudin, H.; Fang, X.; Auras, R. Poly(lactic acid)—Mass production, processing, industrial applications, and end of life. Adv. Drug Deliv. Rev. 2016, 107, 333–366. [Google Scholar] [CrossRef] [PubMed]
- Lasprilla, A.J.R.; Martinez, G.A.R.; Lunelli, B.H.; Jardini, A.L.; Filho, R.M. Poly-lactic acid synthesis for application in biomedical devices—A review. Biotechnol. Adv. 2012, 30, 321–328. [Google Scholar] [CrossRef]
- Narancic, T.; Cerrone, F.; Beagan, N.; O’Connor, K.E. Recent advances in bioplastics: Application and biodegradation. Polymers 2020, 12, 920. [Google Scholar] [CrossRef]
- Koutinas, A.A.; Vlysidis, A.; Pleissner, D.; Kopsahelis, N.; Lopez Garcia, I.; Kookos, I.K.; Papanikolaou, S.; Kwan, T.H.; Lin, C.S.K. Valorization of industrial waste and by-product streams via fermentation for the production of chemicals and biopolymers. Chem. Soc. Rev. 2014, 43, 2587–2627. [Google Scholar] [CrossRef]
- Alonso, S.; Rendueles, M.; Díaz, M. Microbial production of specialty organic acids from renewable and waste materials. Crit. Rev. Biotechnol. 2015, 35, 497–513. [Google Scholar] [CrossRef]
- Köbbing, J.F.; Thevs, N.; Zerbe, S. The utilisation of reed (Phragmites australis): A review. Mires Peat 2013, 13, 1–14. [Google Scholar]
- Brix, H. Functions of macrophytes in constructed wetlands. Water Sci. Technol. 1994, 29, 71–78. [Google Scholar] [CrossRef]
- van der Sluis, T.; Poppens, R.; Kraisvitnii, P.; Rii, O.; Lesschen, J.P.; Galytska, M.; Elbersen, W. Reed Harvesting from Wetlands for Bioenergy; Alterra Report 2460; Alterra Wageningen UR: Wageningen, Netherlands, 2013. [Google Scholar]
- Baibagyssov, A.; Thevs, N.; Nurtazin, S.; Waldhardt, R.; Beckmann, V.; Salmurzauly, R. Biomass Resources of Phragmites australis in Kazakhstan: Historical Developments, Utilization, and Prospects. Resources 2020, 9, 74. [Google Scholar] [CrossRef]
- Perttunen, J.; Myllykoski, L.; Keiski, R.L. Lactic Acid Fermentation of Hemicellulose Liquors and Their Activated Carbon Pretreatments. In Engineering and Manufacturing for Biotechnology; Hofman, M., Thonart, P., Eds.; Kluwer Academic Publishers: Berlin, Germany, 2001; ISBN 0-7923-6927-0. [Google Scholar]
- Zhang, Y.; Li, M.; Nie, T.; Ni, Z. A Process Study of Lactic Acid Production from Phragmites australis Straw by a Thermophilic Bacillus coagulans Strain under Non-Sterilized Conditions. Processes 2018, 6, 175. [Google Scholar] [CrossRef]
- Bischoff, K.M.; Liu, S.; Hughes, S.R.; Rich, J.O. Fermentation of corn fiber hydrolysate to lactic acid by the moderate thermophile Bacillus coagulans. Biotechnol. Lett. 2010, 32, 823–828. [Google Scholar] [CrossRef] [PubMed]
- Ma, K.; Hu, G.; Pan, L.; Wang, Z.; Zhou, Y.; Wang, Y.; Ruan, Z.; He, M. Highly efficient production of optically pure L-lactic acid from corn stover hydrolysate by thermophilic Bacillus coagulans. Bioresour. Technol. 2016, 219, 114–122. [Google Scholar] [CrossRef] [PubMed]
- Xue, Z.; Wang, L.; Ju, J.; Yu, B.; Xu, P.; Ma, Y. Efficient production of polymer-grade L-lactic acid from corn stover hydrolyzate by thermophilic Bacillus sp. strain XZL4. Springerplus 2012, 1, 43. [Google Scholar] [CrossRef]
- Hu, J.; Zhang, Z.; Lin, Y.; Zhao, S.; Mei, Y.; Liang, Y.; Peng, N. High-titer lactic acid production from NaOH-pretreated corn stover by Bacillus coagulans LA204 using fed-batch simultaneous saccharification and fermentation under non-sterile condition. Bioresour. Technol. 2015, 182, 251–257. [Google Scholar] [CrossRef]
- Peng, L.; Wang, L.; Che, C.; Yang, G.; Yu, B.; Ma, Y. Bacillus sp. strain P38: An efficient producer of l-lactate from cellulosic hydrolysate, with high tolerance for 2-furfural. Bioresour. Technol. 2013, 149, 169–176. [Google Scholar] [CrossRef]
- Pleissner, D.; Venus, J. Utilization of protein-rich residues in biotechnological processes. Appl. Microbiol. Biotechnol. 2016, 100, 2133–2140. [Google Scholar] [CrossRef]
- Michelson, T.; Kask, K.; Jõgi, E.; Talpsep, E.; Suitso, I.; Nurk, A. L(+)-Lactic acid producer Bacillus coagulans SIM-7 DSM 14043 and its comparison with Lactobacillus delbrueckii ssp. lactis DSM 20073. Enzym. Microb. Technol. 2006, 39, 861–867. [Google Scholar] [CrossRef]
- de la Torre, I.; Ladero, M.; Santos, V.E. Production of D-lactic acid by L. delbrueckii growing on orange peel waste hydrolysates and model monosaccharide solutions: Effects of pH and temperature on process kinetics. Biomass Convers. Biorefinery 2019, 9, 565–575. [Google Scholar] [CrossRef]
- Hu, J.; Lin, Y.; Zhang, Z.; Xiang, T.; Mei, Y.; Zhao, S.; Liang, Y.; Peng, N. High-titer lactic acid production by Lactobacillus pentosus FL0421 from corn stover using fed-batch simultaneous saccharification and fermentation. Bioresour. Technol. 2016, 214, 74–80. [Google Scholar] [CrossRef]
- Meng, Y.; Xue, Y.; Yu, B.; Gao, C.; Ma, Y. Efficient production of l-lactic acid with high optical purity by alkaliphilic Bacillus sp. WL-S20. Bioresour. Technol. 2012, 116, 334–339. [Google Scholar] [CrossRef]
- Assavasirijinda, N.; Ge, D.; Yu, B.; Xue, Y.; Ma, Y. Efficient fermentative production of polymer-grade d-lactate by an engineered alkaliphilic Bacillus sp. strain under non-sterile conditions. Microb. Cell Fact. 2016, 15, 3. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Zhao, B.; Li, F.; Xu, K.; Ma, C.; Tao, F.; Li, Q.; Xu, P. Highly efficient production of d-lactate by Sporolactobacillus sp. CASD with simultaneous enzymatic hydrolysis of peanut meal. Appl. Microbiol. Biotechnol. 2011, 89, 1009–1017. [Google Scholar] [CrossRef] [PubMed]
- Ooi, K.Y.; Wu, J.C. Use of dry yeast cells as a cheap nitrogen source for lactic acid production by thermophilic Bacillus coagulans WCP10-4. Front. Chem. Sci. Eng. 2015, 9, 381–385. [Google Scholar] [CrossRef]
- Ma, K.; Maeda, T.; You, H.; Shirai, Y. Open fermentative production of l-lactic acid with high optical purity by thermophilic Bacillus coagulans using excess sludge as nutrient. Bioresour. Technol. 2014. [Google Scholar] [CrossRef] [PubMed]
- Vodnar, D.C.; Venus, J.; Schneider, R.; Socaciu, C. Lactic acid production by Lactobacillus paracasei 168 in discontinuous fermentation using lucerne green juice as nutrient substitute. Chem. Eng. Technol. 2010, 33, 468–474. [Google Scholar] [CrossRef]
- Alves de Oliveira, R.; Schneider, R.; Vaz Rossell, C.E.; Maciel Filho, R.; Venus, J. Polymer grade l-lactic acid production from sugarcane bagasse hemicellulosic hydrolysate using Bacillus coagulans. Bioresour. Technol. Rep. 2019, 6, 26–31. [Google Scholar] [CrossRef]
- Hames, B.; Ruiz, R.; Scarlata, C.; Sluiter, A.; Sluiter, J.; Templeton, D. Preparation of Samples for Compositional Analysis; Technical Report NREL/TP-510-42620; National Renewable Energy Laboratory: Golden, CO, USA, 2008. [Google Scholar]
- Sluiter, A.; Ruiz, R.; Scarlata, C.; Sluiter, J.; Templeton, D. Determination of Extractives in Biomass; Technical Report NREL/TP-510-42619; National Renewable Energy Laboratory: Golden, CO, USA, 2005. [Google Scholar]
- Sluiter, A.; Hames, B.; Ruiz, R.; Scarlata, C.; Sluiter, J.; Templeton, D.; Crocker, D.L.A.P. Determination of Structural Carbohydrates and Lignin in Biomass; Technical Report NREL/TP-510-42618; National Renewable Energy Laboratory: Golden, CO, USA, 2012. [Google Scholar]
- Adney, B.; Baker, J. Measurement of Cellulase Activities; Technical Report NREL/TP-510-42628; National Renewable Energy Laboratory: Golden, CO, USA, 1996. [Google Scholar]
- de Toledo, V.D.A.A.; Ruvolo-Takasusuki, M.C.C.; de Oliveira, A.J.B.; Dechechi Chambó, E.; Sanches Lopes, S.M. Spectrophotometry as a Tool for Dosage Sugars in Nectar of Crops Pollinated by Honeybees. In Macro to Nano Spectroscopy; Uddin, J., Ed.; Intech Open: London, UK, 2012. [Google Scholar]
- VDLUFA (Association of German Agricultural Analytic and Research Institutes). VDLUFA Methodenbuch Band III Die Chemische Untersuchung Von Futtermitteln, 3rd ed.; VDLUFA-Verlag: Darmstadt, Germany, 1976; ISBN 978-3-941273-14-6. [Google Scholar]
- Wyman, C.; Decker, S.; Himmel, M.; Brady, J.; Skopec, C.; Viikari, L. Hydrolysis of Cellulose and Hemicellulose. In Polysaccharides; Dumitriu, S., Ed.; CRC Press: Boca Raton, FL, USA, 2004; ISBN 978-0429131660. [Google Scholar]
- Templeton, D.W.; Scarlata, C.J.; Sluiter, J.B.; Wolfrum, E.J. Compositional analysis of lignocellulosic feedstocks. 2. Method uncertainties. J. Agric. Food Chem. 2010, 58, 9054–9062. [Google Scholar] [CrossRef]
- Cotana, F.; Cavalaglio, G.; Pisello, A.L.; Gelosia, M.; Ingles, D.; Pompili, E. Sustainable ethanol production from common reed (Phragmites australis) through simultaneuos saccharification and fermentation. Sustainability 2015, 7, 12149–12163. [Google Scholar] [CrossRef]
- Seidl, P.R.; Goulart, A.K. Pretreatment processes for lignocellulosic biomass conversion to biofuels and bioproducts. Curr. Opin. Green Sustain. Chem. 2016, 2, 48–53. [Google Scholar] [CrossRef]
- Agbor, V.B.; Cicek, N.; Sparling, R.; Berlin, A.; Levin, D.B. Biomass pretreatment: Fundamentals toward application. Biotechnol. Adv. 2011, 29, 675–685. [Google Scholar] [CrossRef] [PubMed]
- Marshall, R.; Beers, R.J. Growth of Bacillus coagulans in chemically defined media. J. Bacteriol. 1967, 94, 517–521. [Google Scholar] [CrossRef] [PubMed]
- López-Gómez, J.P.; Unger, P.; Schneider, R.; Venus, J. From Upstream to Purification: Production of Lactic Acid from the Organic Fraction of Municipal Solid Waste. Waste Biomass Valoriz. 2020. [Google Scholar] [CrossRef]
- Altaf, M.; Naveena, B.J.; Ready, G. Screening of inexpensive nitrogen sources for production of L(+) lactic acid from starch by amylolytic lactobacillus amylophilus GV6 in single step fermentation. Food Technol. Biotechnol. 2005, 43, 235–239. [Google Scholar]
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).