Abstract
The potential of durian (Durio zibethinus Murr.) peel as feedstock for the production of fermentable sugars was evaluated. Durian peel biomass from two cultivars, monthong (Durio zibethinus Murr. cv. Monthong) and chanee (Durio zibethinus Murr. cv. Chanee), were pretreated with different concentrations (70%, 75%, 80%, and 85%) of phosphoric acid (H3PO4) at a moderate temperature of 60 °C for 60 min. The H3PO4-pretreated durian peel biomass was then subjected to enzymatic hydrolysis. Significantly higher glucan (44.74 ± 0.21%) content was observed in the monthong peel compared to the chanee peel (42.06 ± 0.28%). Phosphoric acid pretreatment caused the significant solubilization of the xylan and acid soluble lignin (ASL) contents. This enhanced the enzymatic hydrolysis process causing a significant increase in the hydrolysis efficiency and glucose concentration. The highest hydrolysis efficiency and glucose concentration were obtained after 72 h from the 75% H3PO4-pretreated peel biomass for both the monthong (90.33 ± 0.42% and 9.55 ± 0.11 g/L, respectively) and chanee (90.06 ± 0.40% and 8.56 ± 0.13 g/L, respectively) peels. Biomass to glucose recovery for monthong and chanee were improved by approximately 7- and 6-fold, respectively. The scanning electron microscopy (SEM) and X-ray diffraction (XRD) analysis showed destruction of the peel biomass structure and changes in the cellulose crystallinity index (CrIs).
1. Introduction
Lignocellulosic biomass is a cheap and abundant renewable resource for the sustainable production of bioethanol. This biomass includes agricultural and forestry waste products, grasses and other non-food plants [1]. Agro-residues are one of the most abundant and cheapest lignocellulosic resources worldwide [2]. Durian (Durio zibethinus Murr.) is a very popular fruit in Thailand and other countries in Southeast Asia. The flesh is delicious with a unique flavor and contains 27% carbohydrates, 5.33% fat, 3.1% fiber and 1.47% protein per 100 g of fresh fruit [3]. In Thailand, monthong (Durio zibethinus Murr. cv. Monthong) and chanee (Durio zibethinus Murr. cv. Chanee) are the most common cultivars of commercial importance [4]. Processing of this fruit generates different types of waste, including peel waste [5], that are disposed of by burning or dumping at landfills, creating pollution of the environment. This has necessitated the development of an alternative use of the durian peel by adding economic value to this renewable resource [6]. Durian peel waste, like other agro-residues, is an extremely attractive renewable resource for the production of value-added products such as bioethanol [7]. Production of fermentable sugars from this renewable resource will contribute to solving the problems associated with durian peel disposal, as well as the problem of food versus fuel competition in first-generation bioethanol production.
Despite its attractiveness as a substrate for bioethanol production, durian peel, like other lignocellulosic biomasses, has a very complex structure, consisting of hemicellulose and cellulose cross-linked and strongly bonded to lignin [8]. This structural complexity, coupled with the cellulose crystallinity, makes lignocellulosic biomass resistant to enzymatic hydrolysis [9]. Enzymatic hydrolysis, therefore, yields insufficient sugar concentrations for fermentation [10]. Pretreatment is required to disrupt the complex structure, and increase the surface area and porosity of the cellulose to make it more accessible during enzymatic hydrolysis [11]. Development of an effective pretreatment method is critical in the production of fermentable sugars from durian peel.
Different pretreatment techniques, including physical, chemical, biological, and their combinations, have been developed to enhance cellulose accessibility in enzymatic hydrolysis [12]. Chemical pretreatment with acid is considered one of the most effective pretreatment techniques [13]. Acid pretreatment exposes the cellulose to enzymatic attack by solubilizing hemicellulose and causing structural changes to the lignin and cellulose [14]. Acids, such as hydrochloric acid and sulfuric acid, are hazardous and very corrosive [15]. Phosphoric acid has been identified as one of the most safe and effective chemicals for the pretreatment of lignocellulosic biomass [16]. In addition to the pretreatment process, phosphoric acid serves as a source of phosphorus, an essential nutrient for numerous fermentation media. It has been used to pretreat other agricultural residues, including sugarcane bagasse [17], corn stover [18], corncobs [19] and wheat bran [20]. The present work is, therefore, aimed at developing a conversion process for the production of fermentable sugars from durian peel biomass from both monthong and chanee. The peel biomass was pretreated with different concentrations of phosphoric acid to determine the optimal condition(s) to enhance enzymatic hydrolysis.
2. Materials and Methods
2.1. Preparation of Durian Peel
Fresh durian peel from 2 cultivars (Monthong and Chanee) were collected from Phitsanulok Province, Thailand, cut into pieces and air-dried for 10 days. The dried biomass was ground with a grinding machine (SM 100, Rtsch, Rheinis-che StraBe 36-D-42781, Haan, North Rhine-Westphalia, Germany), after which it was sieved through a 150–300 µm screen. The 150–300 µm particle size biomass was stored at room temperature in tightly closed plastic bags for further study.
2.2. Analytical Methods
The chemical composition of the untreated and pretreated durian peel biomass, comprising the structural carbohydrates, acid soluble lignin (ASL), and acid insoluble lignin (AIL) were analyzed according to National Renewable Energy Laboratory [21]. The total ash component of the biomass before pretreatment was also determined by National Renewable Energy Laboratory [22]. Ethanol extractives were again analyzed as described by National Renewable Energy Laboratory [23]. Monomer sugars were determined by an Agilent 1100 high-performance liquid chromatography (HPLC; Agilent Technologies, Waldbronn, Germany) set-up equipped with a refractive index detector (RID, G1362A, Agilent Technologies, Waldbronn, Germany). Bio-Rad (300 × 7.8 mm, Hercules, CA, USA) Aminex HPX-87P column was used in the HPLC. The column was operated at 80 °C with the injection of 20 µL of samples. Ultra-pure water was used for the mobile phase at a flow rate of 0.6 mL/min.
2.3. Phosphoric Acid Pretreatment of the Durian Peel Biomass
Phosphoric acid was used for the pretreatment of the durian peel biomass. The biomass (3 g) was suspended in different concentrations (70%, 75%, 80% and 85% v/v) of H3PO4 in a 50 mL centrifuge tube at a ratio of 1.0 g/8.0 mL. The tubes were then incubated in a water bath (Eyela Pro Cool Bath NCB-3300, Tokyo Rikakikai Co. Ltd., Tokyo, Japan) at 60 °C for 60 min [15]. The durian peel biomass and the H3PO4 reaction slurry was stopped by adding approximately 30 mL of acetone. The tubes were then centrifuged at a relative centrifugal force of 1305× g for 15 min, after which the supernatant was discarded. The solids were again suspended in acetone, centrifuged, and the supernatant thrown away as before. This process was repeated 3 times after which the solids were washed with distilled water to attain a neutral pH.
2.4. Enzymatic Saccharification
The untreated and pretreated durian peel biomass were subjected to enzymatic hydrolysis using the method described by Siripong et al. [15] with some modifications. Durian peel biomass (0.1 g dry weight) was suspended in a 10 mL digestion solution containing 50 mM sodium citrate buffer (pH 4.8), 2% sodium azide (w/v) and an enzyme cocktail of 30 filter paper unit (FPU) cellulase (celluclast 1.5 L, Sigma-Aldrich, St. Louis, MO, USA) plus 60 U β-glucosidase (Oriental Yeast Co., Ltd., Tokyo, Japan) per g of dry biomass. The hydrolysis was conducted in a rotary shaker (Innova 4340, New Brunswick Scientific Company, Edison, NJ, USA) at a temperature of 50 °C and 150 rpm for 72 h. Liquid portions of the reaction solution (20 µL) were collected at 12, 24, 48 and 72 h to analyze the glucose concentration. The efficiency of hydrolysis was then calculated based on the glucose released, glucan in initial biomass, and the conversion factor of glucan to glucose (1.11). The biomass to glucose recovery was also calculated based on the solid recovery after pretreatment, glucan in biomass, the hydrolysis efficiency, and the conversion factor of glucan to glucose (1.11).
2.5. Scanning Electron Microscopy (SEM)
The morphology of the untreated and pretreated durian peel biomass was analyzed with a scanning electron microscope (SEM; LEO 1455VP, Zeiss, Gottingen, Germany). Samples were freeze dried, mounted on aluminum specimen stubs, and coated with gold before observing under the microscope.
2.6. X-ray Diffraction (XRD)
The degree of cellulose crystallinity in the untreated and pretreated durian peel biomass was determined using X-ray diffraction (PANalytical X’pert Pro, PW 3040/60 Diffractometer, Almelo, the Netherlands). Acetone was used to wash the biomass three times after which it was dried at room temperature. The biomass was then ground to a fine powder (150 µm mesh) and scanned over the range of 2θ = 10° to 40° at a rate of 0.1°/min. The crystallinity index (CrI) was calculated using Equation (1):
where I002 and Iam represent the intensities at 2θ = 22.2° and 2θ = 18.2°, respectively.
2.7. Statistical Analysis
All experiments were conducted in triplicate and the data expressed as the mean and standard deviation (SD). Analysis of Variance (ANOVA) and comparison of treatment means (Tukey’s test, 5% level) were performed using SPSS version 17.0 (SPSS Inc., Chicago, IL, USA).
3. Results
3.1. Composition of the Durian Peel Biomass
Average relative chemical contents (dry matter) of the durian peel biomass analyzed in this study are shown in Table 1. Glucan and xylan accounted for approximately 93% of the total carbohydrates in the durian peels from both cultivars. However, the monthong peel contained significantly (p < 0.05) higher glucan (44.74 ± 0.21%) and xylan (13.00 ± 0.15%) contents compared to the chanee peel (42.06 ± 0.28% and 12.28 ± 0.20%, respectively). The remaining 7% of the carbohydrates in the durian peels from both cultivars was made up of arabinose and galactose. The total lignin content of the chanee peel (14.93 ± 0.31%) was significantly higher than that of the monthong peel (13.44 ± 0.23%). The chanee peel again contained significantly more ash (4.87 ± 0.06%) and ethanol extractives (10.18 ± 0.08%) than the monthong peel (4.05 ± 0.04% and 9.10 ± 0.13%, respectively).

Table 1.
Chemical composition of the durian peel.
3.2. Changes in the Chemical Composition of the Durian Peel Biomass during Phosphoric Acid Pretreatment
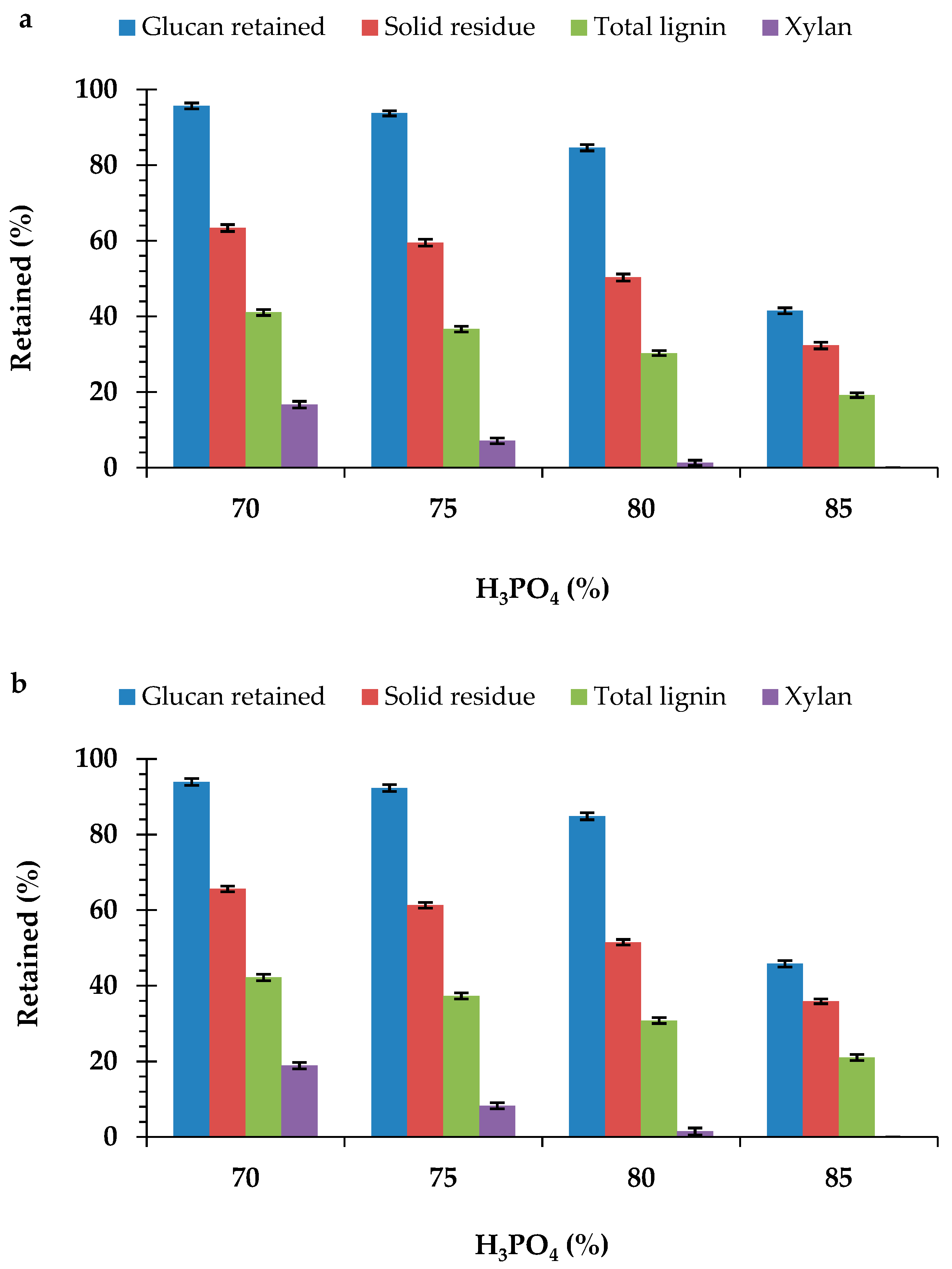
Pretreatment with different concentrations (70%, 75%, 80%, and 85%) of the H3PO4 resulted in significant changes in the chemical composition of the durian peel biomass from the two cultivars (Table 2 and Table 3). The effect of the H3PO4 pretreatment was more evident by the significant decrease and complete removal of the xylan content in the durian peel biomass. Xylan solubilization was more obvious with an increase in the H3PO4 concentration. Pretreatment with the 85% H3PO4 concentration completely removed the xylan content in the peel biomass from both monthong (Figure 1a) and chanee (Figure 1b). Results obtained also showed the significant decrease in the ASL content of the peel biomass from each of the cultivars after pretreatment (Table 2 and Table 3). However, increasing the concentration of the H3PO4 above 75% did not have any significant effect on the ASL content of the peel biomass from each of the cultivars. The decrease in the xylan and ASL content in the durian peel biomass was accompanied with a significant increase in the glucan content. This increase was more evident with an increase in the H3PO4 concentration. However, pretreatment of the peel biomass with the 85% H3PO4 concentration resulted in a significantly lower content of glucan compared to the other concentrations. It was also observed that AIL content in all of the pretreated peel biomasses under the various pretreatment conditions was higher, although not significant (p > 0.05), compared to the untreated peel biomass.

Table 2.
Relative chemical composition of the H3PO4-pretreated monthong peel.

Table 3.
Relative chemical composition of the H3PO4-pretreated chanee peel.
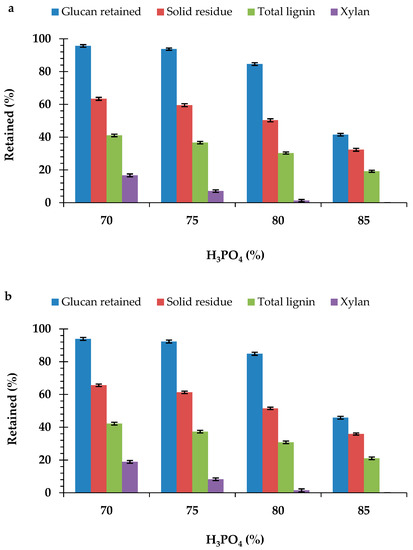
Figure 1.
Effect of the H3PO4 concentrations on the composition of the (a) monthong and (b) chanee peels.
By pretreatment with the 80% H3PO4 concentration and below, more than 80% and 50% of the xylan and total lignin, respectively, were removed in the peel biomass from all of the durian cultivars (Figure 1a,b). Under these pretreatment conditions, over 80% of the glucan content was retained in the peel biomass from all the cultivars. Nevertheless, the glucan content retained in the pretreated (85% H3PO4) peel biomass for all the durian cultivars was below 50%. Pretreatment with the 85% H3PO4 concentration resulted in the removal of more than 70% total lignin in both the monthong and chanee peels. The degradation of glucan, coupled with the solubilization of xylan and ASL, contributed to the significant loss in the total biomass weight (Figure 1a,b).
3.3. Enzymatic Hydrolysis of the Durian Peel Biomass
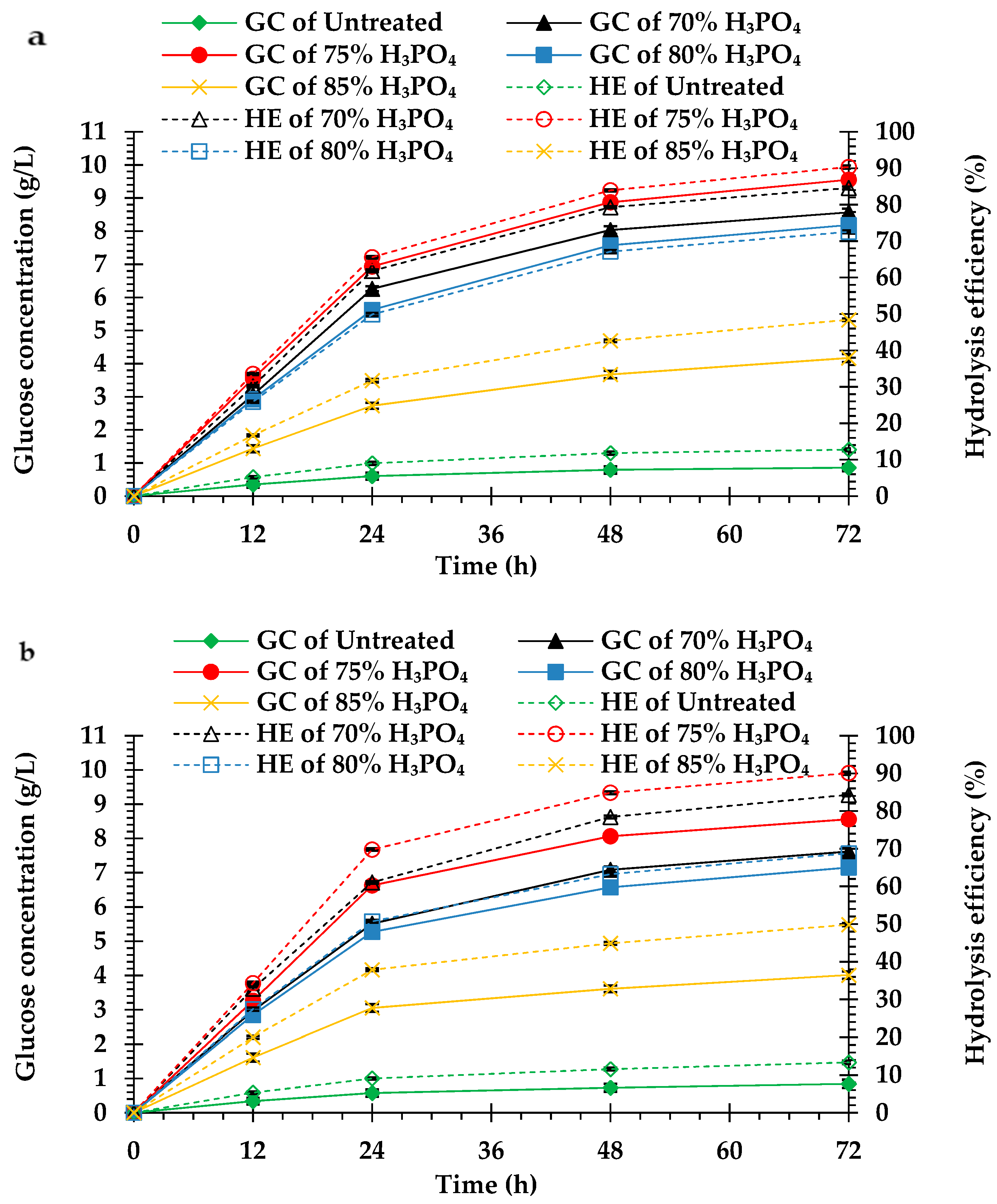
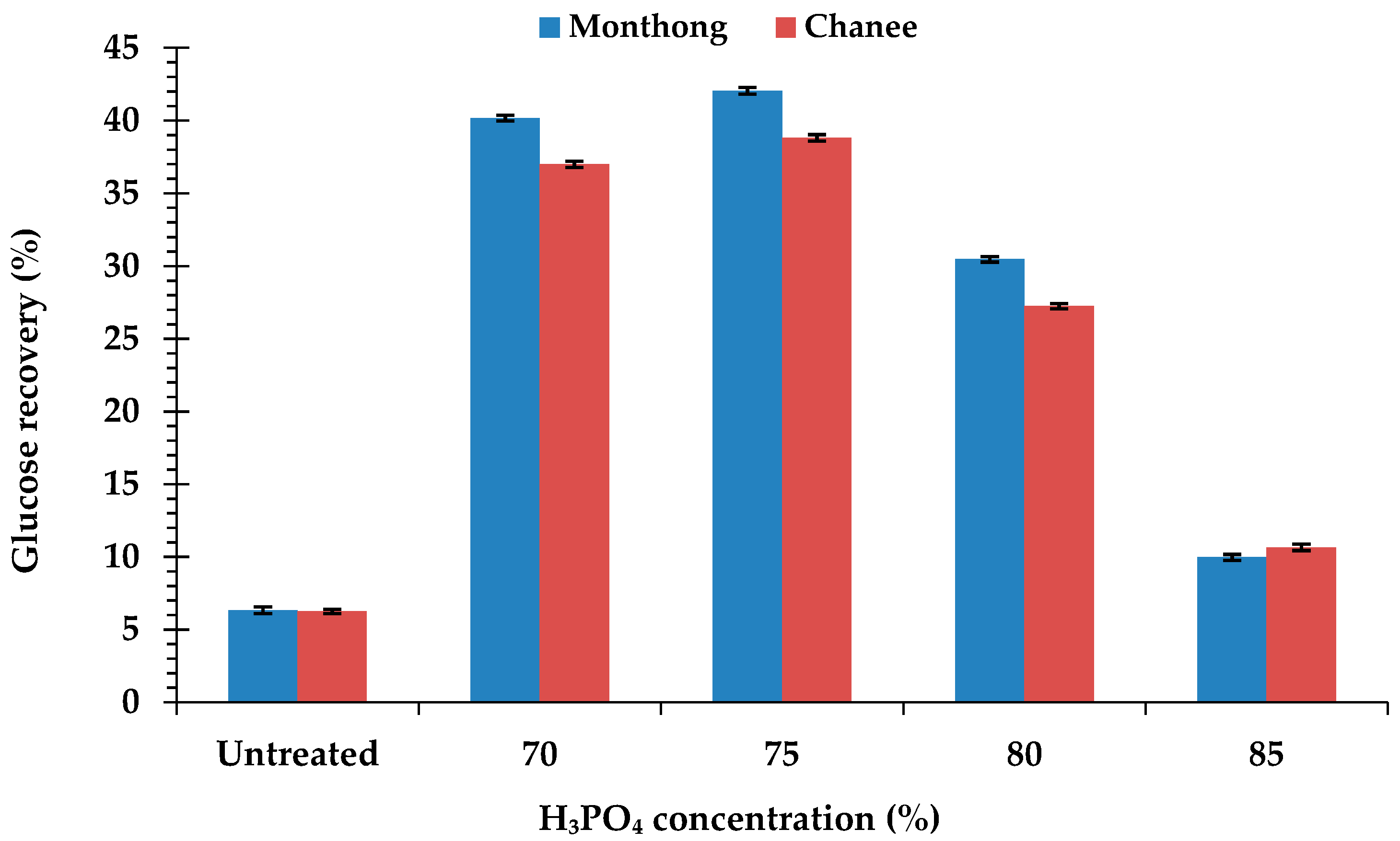
Compared to the untreated peel biomass, the H3PO4-pretreated peel biomass from both cultivars had higher hydrolysis efficiency and glucose concentration. The hydrolysis efficiency and glucose concentration of the H3PO4-pretreated peel biomass from monthong (Figure 2a) and chanee (Figure 2b) increased sharply in the initial 24 h and continued steadily until 72 h. Maximum hydrolysis efficiency and glucose concentration, in both the untreated and pretreated peel biomass from all the cultivars, were observed after 72 h of hydrolysis. The highest hydrolysis efficiency and glucose concentration were obtained from the 75% H3PO4-pretreated peel biomass for both monthong (90.33 ± 0.42% and 9.55 ± 0.11 g/L, respectively) and chanee (90.06 ± 0.40% and 8.56 ± 0.13 g/L, respectively). At this condition, the highest biomass to glucose recovery of approximately 42% (monthong peel) and 39% (chanee peel) were achieved (Figure 3). Further increases in the concentration of H3PO4 (80% and 85%), however, resulted in a decrease in the hydrolysis efficiency, glucose concentration and glucose recovery.
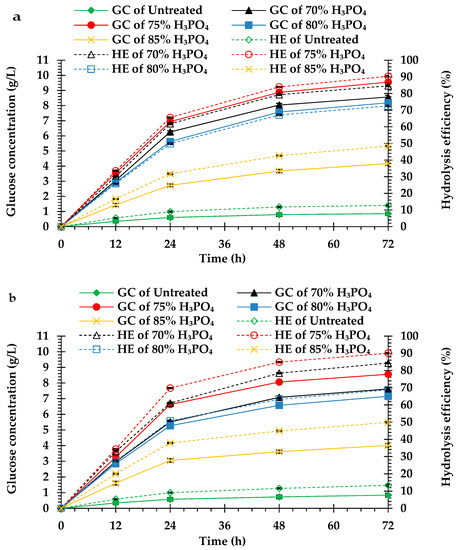
Figure 2.
Enzymatic hydrolysis of the untreated and pretreated (a) monthong and (b) chanee peels. GC and HE represent glucose concentration and hydrolysis efficiency, respectively.
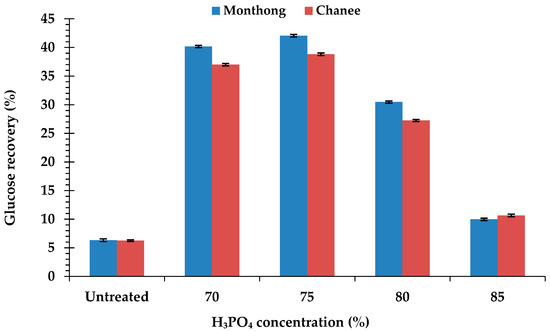
Figure 3.
Biomass to glucose recovery of the untreated and pretreated durian peel after 72 h of enzymatic hydrolysis.
3.4. Changes of the Durian Peel Biomass Surface Morphology
To further understand the role of biomass structure on the efficiency of cellulose digestibility by cellulase enzymes, the surface areas of the H3PO4-pretreated and untreated peel biomass were observed using the electron microscope. The electron microscope micrographs showed a severe disruption in the physical structure of the pretreated peel biomass from both monthong (Figure 4b–e) and chanee (Figure 5b–e) compared to the untreated samples (Figure 4a or Figure 5a, respectively). The cell surface of the pretreated peel biomass was characterized by more cracks and fiber destruction. More longitudinal grooves were observed after the pretreatment process. Structural destruction became more severe with an increase in the phosphoric acid concentration.

Figure 4.
Scanning electron micrographs of the (a) untreated and H3PO4-pretreated monthong peel at (b) 70%, (c) 75%, (d) 80%, and (e) 85% concentrations.

Figure 5.
Scanning electron micrographs of the (a) untreated and H3PO4-pretreated chanee peel at (b) 70%, (c) 75%, (d) 80%, and (e) 85% concentrations.
3.5. Changes of the Cellulose Crystalline Structure of the Durian Peel Biomass
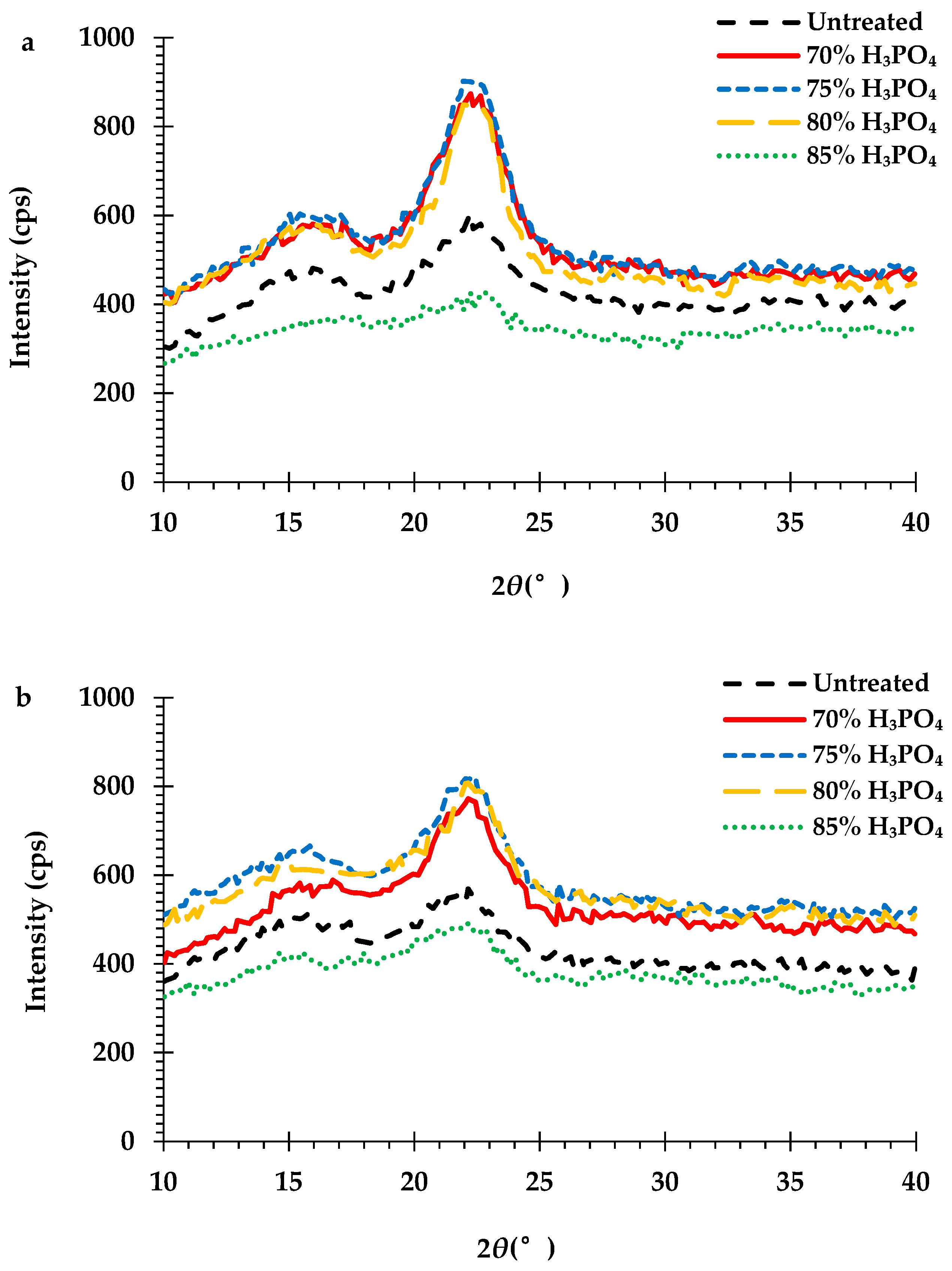
The effect of the H3PO4 pretreatment on the crystallinity of the cellulose in the peel biomass is a very important feature in the enzymatic hydrolysis process. The crystallinity index (CrI) of the cellulose in the untreated and pretreated monthong (Figure 6a) and chanee (Figure 6b) peel biomass is shown by the XRD pattern. The XRD spectra of the durian peel biomass showed two major diffraction peaks located at approximately 2θ = 15.6° and 22.2°, corresponding to cellulose II and I, respectively. The CrIs of the cellulose decreased from approximately 31% and 21% (untreated biomass) to around 19% and 18% (85% H3PO4-pretreated biomass) for both the monthong and chanee peel, respectively (Table 4). However, the CrIs of the cellulose in the peel biomass pretreated with the H3PO4 concentrations of 80% and below were relatively higher compared to that of the 85% H3PO4-pretreated and untreated peel biomass from the monthong and chanee peels (Table 4).
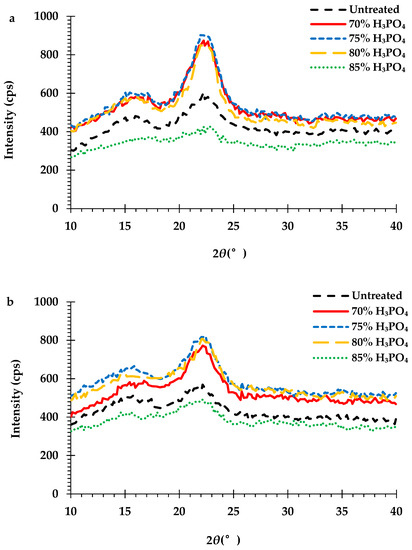
Figure 6.
X-ray diffraction (XRD) spectra of the cellulose-rich portions of the untreated and H3PO4-pretreated (a) monthong and (b) chanee peels.

Table 4.
The crystallinity index (CrIs) of the untreated and H3PO4-pretreated durian peels.
4. Discussion
Crop production and processing generate huge amounts of waste as renewable resources for the production of fermentable sugars [24]. The chemical composition of these agricultural residues, however, depends on many factors, including the plant species, location and conditions of storage [12]. Significant variations in the chemical contents were observed among the peels from the two durian cultivars in this study (Table 1). The glucan contents of the monthong and chanee peel biomass studied are higher than that observed in other agro-residues, including hazelnut shells (16.67 ± 0.76%) [25], almond shells (32.17 ± 1.32%) [26], corn stover (30.70 ± 0.90%) [27] and orange peel (16.37 ± 0.77%) [28]. The presence of the high glucan content in the durian peel biomass makes it a promising feedstock for the bioprocess industry. However, pretreatment of the durian peels is important to increase accessibility to the glucan contents by cellulase enzymes [9].
In spite of its effectiveness, acid pretreatment may be toxic and produces inhibitors, including furfural, hydroxymethylfurfural, or acetic acid, that might be inhibitory to the enzymatic hydrolysis and fermentation processes [17]. Phosphoric acid, on the other hand, is less corrosive and toxic compared to acids like hydrochloric acid and sulfuric acid, which have been widely used for the pretreatment of lignocellulosic biomass. It also services as a source of phosphorus, an essential nutrient for numerous fermentation media [15]. Phosphoric acid pretreatment in this study caused significant changes in the composition of the durian peel biomass (Table 2 and Table 3). Significant reduction in the xylan and ASL contents were observed after pretreatment with the H3PO4 concentrations of 80% and below. However, pretreatment with the 85% H3PO4 concentration lead to the complete removal of the xylan content (Figure 1a,b) in addition to a substantial decrease in the ASL (Table 2 and Table 3). Acid pretreatment mainly results in the solubilization of the hemicelluloses, as well as causing structural changes to the lignin and cellulose, leading to an increase in the cellulose digestibility by cellulase enzymes [14]. The changes in the xylan and ASL compositions of the durian peel biomass after pretreatment resulted in a substantial increase in the glucan content (Table 2 and Table 3). Nevertheless, the 85% H3PO4 concentration caused a substantial reduction in the glucan content (>50% removal). This reduction is attributed to the degradation of the glucan at this severe pretreatment condition. Glucan degradation (<20%) was also observed after pretreatment of the durian peel biomass with the 80% H3PO4 concentration and below (Figure 1a,b). In addition to the solubilization of hemicellulose, acid pretreatment of lignocellulosic materials results in the partial degradation of glucan [29]. It can be said that the majority of the glucan losses are associated with its amorphous structure, which is susceptible to the effects of chemical pretreatment [30]. Unlike the ASL content, the AIL content in the durian peel biomass increased after pretreatment (Table 2 and Table 3). This phenomenon has been reported by several researchers after acid pretreatment of different lignocellulosic biomasses [10,31,32]. Increase in the AIL content after pretreatment has been attributed to the presence of pseudo-lignin formed by the repolymerization of degradation products from polysaccharides [14]. In addition to the solubilization of hemicelluloses and structural changes in lignin and cellulose, acid pretreatment results in the formation of a substantial amount of pseudo-lignin [31]. Li et al. [33] extracted and analyzed the AIL content after acid pretreatment, and reported that only approximately half of the extract was real lignin. Yoon et al. [34] reported that degraded carbohydrates during phosphoric acid pretreatment may condense with tannins, suberin, or lignin, resulting in an increase in the lignin content.
The significant decrease and/or removal of the xylan and ASL contents during the H3PO4 pretreatment enhanced cellulose accessibility (Figure 2a,b). The H3PO4-pretreated durian peel biomass from both cultivars showed improved enzymatic hydrolysis at the enzyme loading of 30 FPU of cellulase and 60 U of β-glucosidase per g of biomass. This shows that the cellulose in the pretreated peel biomass was effectively hydrolyzed by the cellulase enzymes compared to the untreated peel biomass. The significant solubilization of the xylan and ASL, coupled with the reduced glucan removal (Figure 1a,b) in the peel biomass pretreated with the 75% H3PO4 concentration, contributed to the high hydrolysis efficiency and glucose (Figure 2a,b). The significantly high biomass to glucose recovery (Figure 3) at this pretreatment condition further confirms its effectiveness. Yoo et al. [35] reported that the optimal pretreatment condition is where the biomass to glucose recovery value is high, and close to the initial glucan content in the biomass. The decrease in hydrolysis efficiency and glucose concentration of the peel biomass pretreated with the H3PO4 concentrations of 80% and above is attributed to the sugar degradation during the pretreatment process (Figure 1a,b). Zhang et al. [36] attributed the high sugar yield after enzymatic digestibility to the absence of sugar degradation during pretreatment at 50 °C with 85% phosphoric acid. This is an indication that the amount of initial cellulose, among other factors, is key in attaining high hydrolysis efficiency and glucose concentration after hydrolysis. Sugar degradation must, therefore, be kept at the barest minimum during pretreatment. Under the optimum pretreatment condition (75% H3PO4 at 60 °C for 60 min) in the current study, the hydrolysis efficiency for the pretreated monthong (90.33 ± 0.42%) and chanee (90.06 ± 0.40%) peel biomass are greater than that of oak chips (50.2%), corn stalks (43.5%), wheat straw (68.7%), bamboo (39.3%), spruce chips (36.3%), Jerusalem artichoke stalks (59.6%) [37], Sida acuta (82.2%), and Achyranthes aspera (86.2%) [15] pretreated with an 80% H3PO4 concentration.
The structural modification of the pretreated peel biomass relates to the significant changes in the chemical composition after pretreatment. Lignin and hemicellulose removal during pretreatment destroy the strong bond between lignin, hemicellulose and cellulose, resulting in the destruction of the biomass structure [38]. By pretreatment, the structure of the durian peel biomass was more open, rougher, and porous, providing more access to the surface for cellulase enzymes to attack (Figure 4 and Figure 5). These structural changes may help to increase the adsorptivity potentials of the pretreated biomass compared to the untreated [39]. Changes in the structure of the durian peel biomass during the H3PO4 pretreatment confirm the results obtained from the enzymatic hydrolysis of the biomass.
The structural changes of the durian peel biomass during pretreatment were further evaluated by X-ray diffractogram of the cellulose rich portions. Cellulose crystallinity is a key factor that affects the efficiency of enzymatic hydrolysis [38]. The crystalline structure of the cellulose in the durian peel biomass pretreated with the 85% H3PO4 concentration in this study was transformed from cellulose I to the amorphous form. This result is consistent with reports from previous studies about the reduction of cellulose crystallinity after concentrated H3PO4 (approximately 85%) pretreatment [32,34,36,40,41]. However, pretreatment of the peel biomass with 80% H3PO4 concentration and below was not able to decrystallize the cellulose structure in the biomass (Figure 6a,b). Yoon et al. [34] reported that H3PO4 concentrations above 80% decrystallize cellulose, while concentrations below 80% swell the cellulose. Even though the CrIs of the durian peel biomass pretreated at these conditions (≤80% H3PO4) were higher than those of the untreated and 85% H3PO4-pretreated biomasses (Table 4), the enzymatic hydrolysis of these samples was greatly improved. Similar observations have been reported by several researchers [27,42,43]. It is evident from this study that the significant changes in the chemical composition of the durian peel biomass after the H3PO4 pretreatment enhanced enzymatic hydrolysis despite the increase in CrIs.
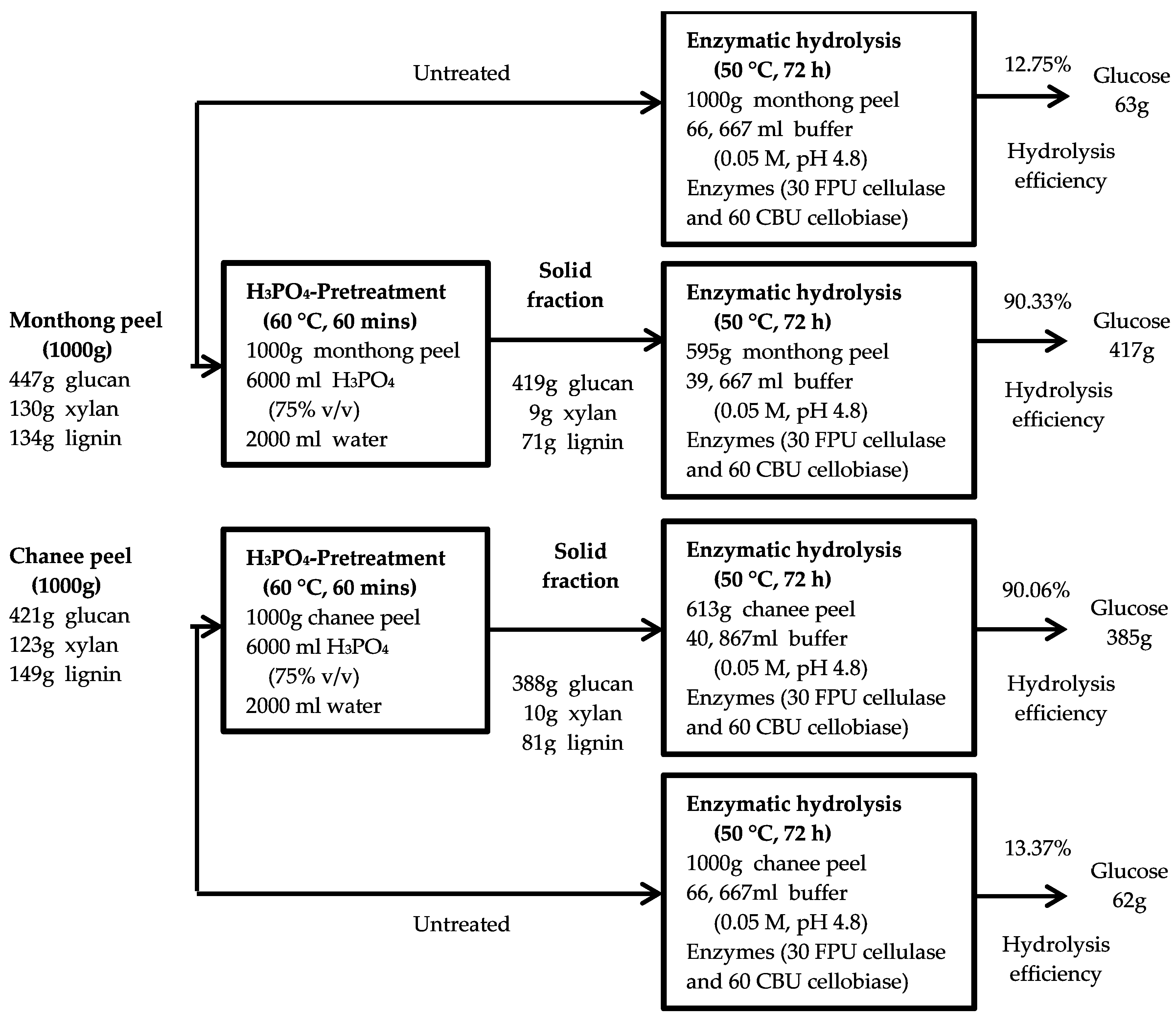
An overall mass balance based on the conversion of the peel biomass into glucose was established to further assess the optimal pretreatment condition on the production of fermentable sugars from both monthong and chanee (Figure 7). The untreated monthong and chanee peel biomass studied had maximum enzymatic digestibility of 12.75% and 13.37%, respectively, after 72 h of hydrolysis (Figure 2a,b). Based on this, the estimated glucose production from both cultivars were 63 and 62 g, respectively, per 1000 g of biomass processed. Enhancement of the conversion process through the optimum pretreatment condition of 75% H3PO4 at a moderate temperature of 60 °C for 60 min improved the enzymatic digestibility of both the monthong and chanee peel biomass up to 90.33% and 90.06%, respectively. Following this, estimated glucose yields of 417 and 385 g, respectively, were produced when 1000 g of the biomass is loaded in the process for each of the cultivars. The results from the mass balance show that glucose production from the monthong and chanee peel biomass was enhanced approximately 7- and 6-fold, respectively, after pretreatment with H3PO4 at the optimum condition.
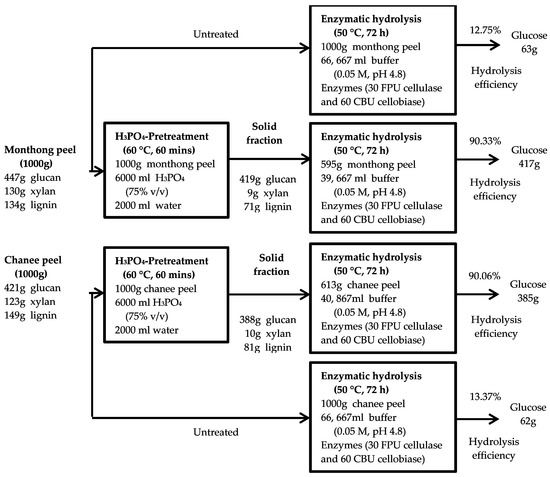
Figure 7.
Schematic diagram of fermentable sugar production from the durian peel biomass by H3PO4 pretreatment process.
5. Conclusions
The study has revealed that although the chemical composition of the monthong and chanee peel biomasses significantly differ, they both have similar structures and can be processed as a whole to produce fermentable sugars for bioethanol production. An optimum pretreatment condition, which was able to significantly increase the hydrolysis efficiency, resulting in higher glucose concentration, was established. Glucose production from the pretreated monthong and chanee peel biomass was greatly improved.
Author Contributions
Conceptualization, A.K.O., S.P. and D.P.; Methodology, data collection and analysis, S.P. and A.K.O.; Writing—Original Draft Preparation, A.K.O.; Writing—Review & Editing, S.P. and D.P.; Supervision, S.P. and D.P.; Funding Acquisition, S.P. and D.P.
Funding
This research was supported by the National Research Council of Thailand in the Fiscal year 2016–2017 (project number R2560B153).
Acknowledgments
The authors are thankful to the Naresuan University International Students Scholarship Scheme for providing the scholarship to study a PhD program in Biotechnology at the Department of Biology, Faculty of Science, Naresuan University, Thailand.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Yan, Q.; Wang, Y.; Rodiahwati, W.; Spiess, A.; Modigell, M. Alkaline-assisted screw press pretreatment affecting enzymatic hydrolysis of wheat straw. Bioprocess Biosyst. Eng. 2017, 40, 221–229. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, Q.A.; Yang, J.M.; Bae, H.J. Bioethanol production from individual and mixed agricultural biomass residues. Ind. Crops Prod. 2017, 95, 718–725. [Google Scholar] [CrossRef]
- Ho, L.-H.; Bhat, R. Exploring the potential nutraceutical values of durian (Durio zibethinus L.)—An exotic tropical fruit. Food Chem. 2015, 168, 80–89. [Google Scholar] [CrossRef] [PubMed]
- Wisutiamonkul, A.; Ampomah-Dwamena, C.; Allan, A.C.; Ketsa, S. Carotenoid accumulation and gene expression during durian (Durio zibethinus) fruit growth and ripening. Sci. Hortic. 2017, 220, 233–242. [Google Scholar] [CrossRef]
- Unhasirikul, M.; Narkrugsa, W.; Naranong, N. Sugar production from durian (Durio zibethinus Murray) peel by acid hydrolysis. Afr. J. Biotechnol. 2013, 12, 5244–5251. [Google Scholar]
- Foo, K.Y.; Hameed, B.H. Transformation of durian biomass into a highly valuable end commodity: Trends and opportunities. Biomass Bioenergy 2011, 35, 2470–2478. [Google Scholar] [CrossRef]
- Tan, Y.L.; Abdullah, A.Z.; Hameed, B.H. Fast pyrolysis of durian (Durio zibethinus L) shell in a drop-type fixed bed reactor: Pyrolysis behavior and product analyses. Bioresour. Technol. 2017, 243, 85–92. [Google Scholar] [CrossRef] [PubMed]
- Zaafouri, K.; Ziadi, M.; Ben Farah, R.; Farid, M.; Hamdi, M.; Regaya, I. Potential of Tunisian Alfa (Stipa tenassicima) fibers for energy recovery to 2G bioethanol: Study of pretreatment, enzymatic saccharification and fermentation. Biomass Bioenergy 2016, 94, 66–77. [Google Scholar] [CrossRef]
- Foston, M.; Ragauskas, A.J. Changes in lignocellulosic supramolecular and ultrastructure during dilute acid pretreatment of Populus and switchgrass. Biomass Bioenergy 2010, 34, 1885–1895. [Google Scholar] [CrossRef]
- Fougere, D.; Nanda, S.; Clarke, K.; Kozinski, J.A.; Li, K. Effect of acidic pretreatment on the chemistry and distribution of lignin in aspen wood and wheat straw substrates. Biomass Bioenergy 2016, 91, 56–68. [Google Scholar] [CrossRef]
- Lewandowska, M.; Szymańska, K.; Kordala, N.; Dąbrowska, A.; Bednarski, W.; Juszczuk, A. Evaluation of Mucor indicus and Saccharomyces cerevisiae capability to ferment hydrolysates of rape straw and Miscanthus giganteus as affected by the pretreatment method. Bioresour. Technol. 2016, 212, 262–270. [Google Scholar] [CrossRef] [PubMed]
- Obeng, A.K.; Premjet, D.; Premjet, S. A review article of biological pretreatment of agricultural biomass. Pertanika J. Trop. Agric. Sci. 2018, 41, 19–40. [Google Scholar]
- Parmar, I.; Rupasinghe, H.P.V. Optimization of dilute acid-based pretreatment and application of laccase on apple pomace. Bioresour. Technol. 2012, 124, 433–439. [Google Scholar] [CrossRef] [PubMed]
- Hu, F.; Jung, S.; Ragauskas, A. Pseudo-lignin formation and its impact on enzymatic hydrolysis. Bioresour. Technol. 2012, 117, 7–12. [Google Scholar] [CrossRef] [PubMed]
- Siripong, P.; Duangporn, P.; Takata, E.; Tsutsumi, Y. Phosphoric acid pretreatment of Achyranthes aspera and Sida acuta weed biomass to improve enzymatic hydrolysis. Bioresour. Technol. 2016, 203, 303–308. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.-H.P.; Cui, J.; Lynd, L.R.; Kuang, L.R. A Transition from Cellulose Swelling to Cellulose Dissolution by o-Phosphoric Acid: Evidence from Enzymatic Hydrolysis and Supramolecular Structure. Biomacromolecules 2006, 7, 644–648. [Google Scholar] [CrossRef] [PubMed]
- Nieves, I.U.; Geddes, C.C.; Miller, E.N.; Mullinnix, M.T.; Hoffman, R.W.; Fu, Z.; Tong, Z.; Ingram, L.O. Effect of reduced sulfur compounds on the fermentation of phosphoric acid pretreated sugarcane bagasse by ethanologenic Escherichia coli. Bioresour. Technol. 2011, 102, 5145–5152. [Google Scholar] [CrossRef] [PubMed]
- Sathitsuksanoh, N.; Zhu, Z.; Zhang, Y.-H.P. Cellulose solvent-based pretreatment for corn stover and avicel: Concentrated phosphoric acid versus ionic liquid [BMIM]Cl. Cellulose 2012, 19, 1161–1172. [Google Scholar] [CrossRef]
- Boonsombuti, A.; Luengnaruemitchai, A.; Wongkasemjit, S. Effect of Phosphoric Acid Pretreatment of Corncobs on the Fermentability of Clostridium beijerinckii TISTR 1461 for Biobutanol Production. Prep. Biochem. Biotechnol. 2015, 45, 173–191. [Google Scholar] [CrossRef] [PubMed]
- Nair, R.B.; Lundin, M.; Brandberg, T.; Lennartsson, P.R.; Taherzadeh, M.J. Dilute phosphoric acid pretreatment of wheat bran for enzymatic hydrolysis and subsequent ethanol production by edible fungi Neurospora intermedia. Ind. Crops Prod. 2015, 69, 314–323. [Google Scholar] [CrossRef]
- Sluiter, A.; Hames, B.; Ruiz, R.; Scarlata, C.; Sluiter, J.; Templeton, D.; Crocker, D. Determination of Structural Carbohydrates and Lignin in Biomass; National Renewable Energy Laboratory: Golden, CO, USA, 2012.
- Sluiter, A.; Hames, B.; Ruiz, R.; Scarlata, C.; Sluiter, J.; Templeton, D. Determination of Ash in Biomass; National Renewable Energy Laboratory: Golden, CO, USA, 2008.
- Sluiter, A.; Ruiz, R.; Scarlata, C.; Sluiter, J.; Templeton, D. Determination of Extractives in Biomass; National Renewable Energy Laboratory: Golden, CO, USA, 2008.
- Searle, S.Y.; Malins, C.J. Waste and residue availability for advanced biofuel production in EU Member States. Biomass Bioenergy 2016, 89, 2–10. [Google Scholar] [CrossRef]
- Hoşgün, E.Z.; Berikten, D.; Kıvanç, M.; Bozan, B. Ethanol production from hazelnut shells through enzymatic saccharification and fermentation by low-temperature alkali pretreatment. Fuel 2017, 196, 280–287. [Google Scholar] [CrossRef]
- Kacem, I.; Koubaa, M.; Maktouf, S.; Chaari, F.; Najar, T.; Chaabouni, M.; Ettis, N.; Chaabouni, E.S. Multistage process for the production of bioethanol from almond shell. Bioresour. Technol. 2016, 211, 154–163. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.W.; Kim, J.Y.; Jang, H.M.; Lee, M.W.; Park, J.M. Sequential dilute acid and alkali pretreatment of corn stover: Sugar recovery efficiency and structural characterization. Bioresour. Technol. 2015, 182, 296–301. [Google Scholar] [CrossRef] [PubMed]
- Vadivel, V.; Moncalvo, A.; Dordoni, R.; Spigno, G. Effects of an acid/alkaline treatment on the release of antioxidants and cellulose from different agro-food wastes. Waste Manag. 2017, 64, 305–314. [Google Scholar] [CrossRef] [PubMed]
- Jung, Y.H.; Kim, I.J.; Kim, H.K.; Kim, K.H. Dilute acid pretreatment of lignocellulose for whole slurry ethanol fermentation. Bioresour. Technol. 2013, 132, 109–114. [Google Scholar] [CrossRef] [PubMed]
- Rocha, G.J.M.; Martin, C.; Soares, I.B.; Maior, A.M.S.; Baudel, H.M.; de Abreu, C.A.M. Dilute mixed-acid pretreatment of sugarcane bagasse for ethanol production. Biomass Bioenergy 2011, 35, 663–670. [Google Scholar] [CrossRef]
- De Carvalho, D.M.; Sevastyanova, O.; Penna, L.S.; de Silva, B.P.; Lindström, M.E.; Colodette, J.L. Assessment of chemical transformations in eucalyptus, sugarcane bagasse and straw during hydrothermal, dilute acid, and alkaline pretreatments. Ind. Crops Prod. 2015, 73, 118–126. [Google Scholar] [CrossRef]
- Ishola, M.M.; Isroi; Taherzadeh, M.J. Effect of fungal and phosphoric acid pretreatment on ethanol production from oil palm empty fruit bunches (OPEFB). Bioresour. Technol. 2014, 165, 9–12. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Henriksson, G.; Gellerstedt, G. Lignin depolymerization/repolymerization and its critical role for delignification of aspen wood by steam explosion. Bioresour. Technol. 2007, 98, 3061–3068. [Google Scholar] [CrossRef] [PubMed]
- Yoon, S.-Y.; Kim, B.-R.; Han, S.-H.; Shin, S.-J. Different response between woody core and bark of goat willow (Salix caprea L.) to concentrated phosphoric acid pretreatment followed by enzymatic saccharification. Energy 2015, 81, 21–26. [Google Scholar] [CrossRef]
- Yoo, H.Y.; Lee, J.H.; Kim, D.S.; Lee, J.H.; Lee, S.K.; Lee, S.J.; Park, C.; Kim, S.W. Enhancement of glucose yield from canola agricultural residue by alkali pretreatment based on multi-regression models. J. Ind. Eng. Chem. 2017, 51, 303–311. [Google Scholar] [CrossRef]
- Zhang, Y.-H.P.; Ding, S.-Y.; Mielenz, J.R.; Cui, J.-B.; Elander, R.T.; Laser, M.; Himmel, M.E.; McMillan, J.R.; Lynd, L.R. Fractionating recalcitrant lignocellulose at modest reaction conditions. Biotechnol. Bioeng. 2007, 97, 214–223. [Google Scholar] [CrossRef] [PubMed]
- Wang, Q.; Wang, Z.; Shen, F.; Hu, J.; Sun, F.; Lin, L.; Yang, G.; Zhang, Y.; Deng, S. Pretreating lignocellulosic biomass by the concentrated phosphoric acid plus hydrogen peroxide (PHP) for enzymatic hydrolysis: Evaluating the pretreatment flexibility on feedstocks and particle sizes. Bioresour. Technol. 2014, 166, 420–428. [Google Scholar] [CrossRef] [PubMed]
- Li, M.-F.; Fan, Y.-M.; Xu, F.; Sun, R.-C.; Zhang, X.-L. Cold sodium hydroxide/urea based pretreatment of bamboo for bioethanol production: Characterization of the cellulose rich fraction. Ind. Crops Prod. 2010, 32, 551–559. [Google Scholar] [CrossRef]
- Cai, D.; Li, P.; Chen, C.; Wang, Y.; Hu, S.; Cui, C.; Qin, P.; Tan, T. Effect of chemical pretreatments on corn stalk bagasse as immobilizing carrier of Clostridium acetobutylicum in the performance of a fermentation-pervaporation coupled system. Bioresour. Technol. 2016, 220, 68–75. [Google Scholar] [CrossRef] [PubMed]
- Noppadon, S.; Zhiguang, Z.; Sungsool, W.; Zhang, Y.-H.P. Cellulose solvent-based biomass pretreatment breaks highly ordered hydrogen bonds in cellulose fibers of switchgrass. Biotechnol. Bioeng. 2011, 108, 521–529. [Google Scholar] [CrossRef]
- Takata, E.; Tsutsumi, K.; Tsutsumi, Y.; Tabata, K. Production of monosaccharides from napier grass by hydrothermal process with phosphoric acid. Bioresour. Technol. 2013, 143, 53–58. [Google Scholar] [CrossRef] [PubMed]
- Hideno, A.; Kawashima, A.; Endo, T.; Honda, K.; Morita, M. Ethanol-based organosolv treatment with trace hydrochloric acid improves the enzymatic digestibility of Japanese cypress (Chamaecyparis obtusa) by exposing nanofibers on the surface. Bioresour. Technol. 2013, 132, 64–70. [Google Scholar] [CrossRef] [PubMed]
- Behzad, S.; Keikhosro, K.; Akram, Z. Oil, chitosan, and ethanol production by dimorphic fungus Mucor indicus from different lignocelluloses. J. Chem. Technol. Biotechnol. 2016, 91, 1835–1843. [Google Scholar] [CrossRef]
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).