The Potential Phosphorus Crisis: Resource Conservation and Possible Escape Technologies: A Review
Abstract
1. Introduction
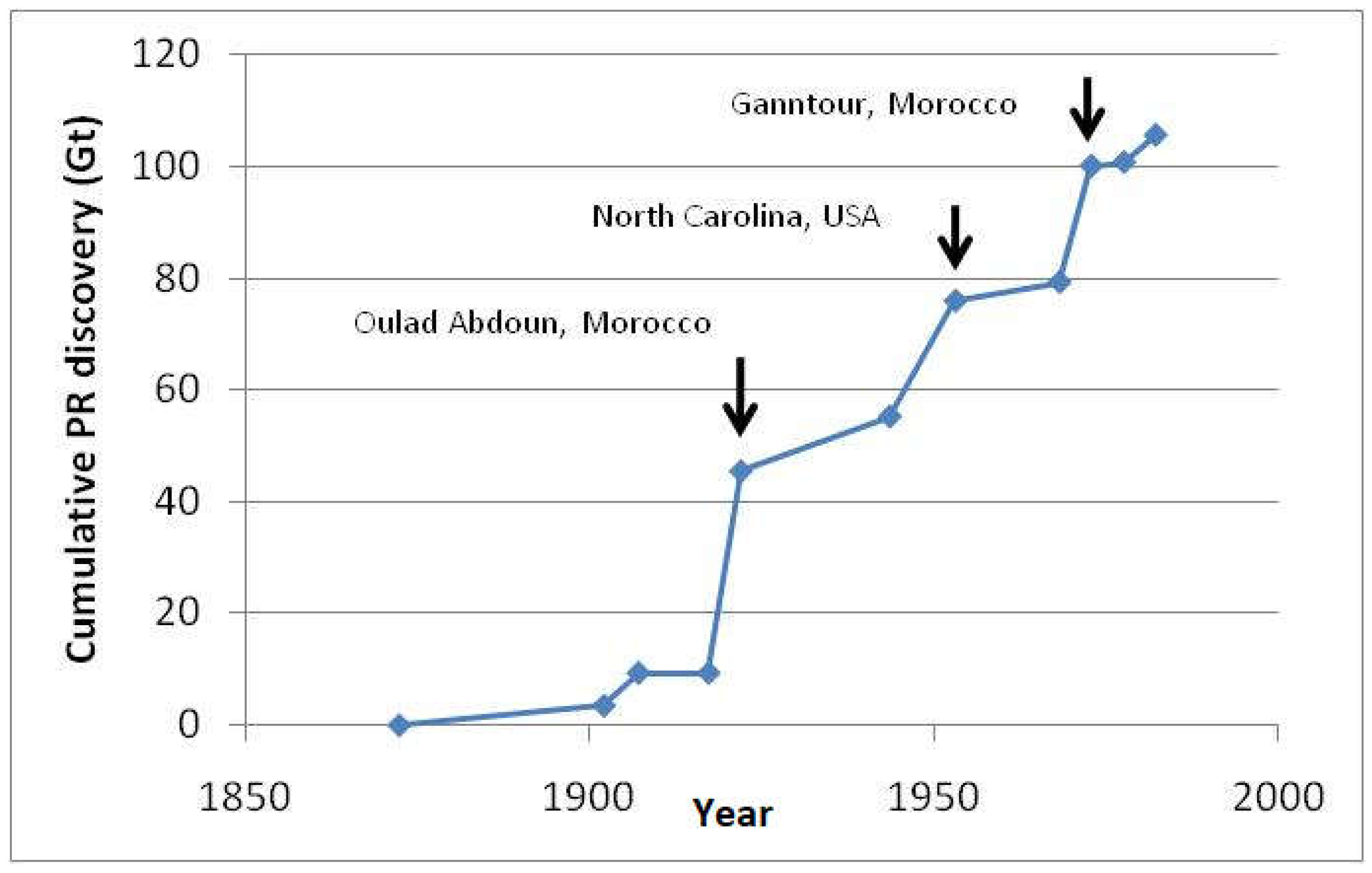
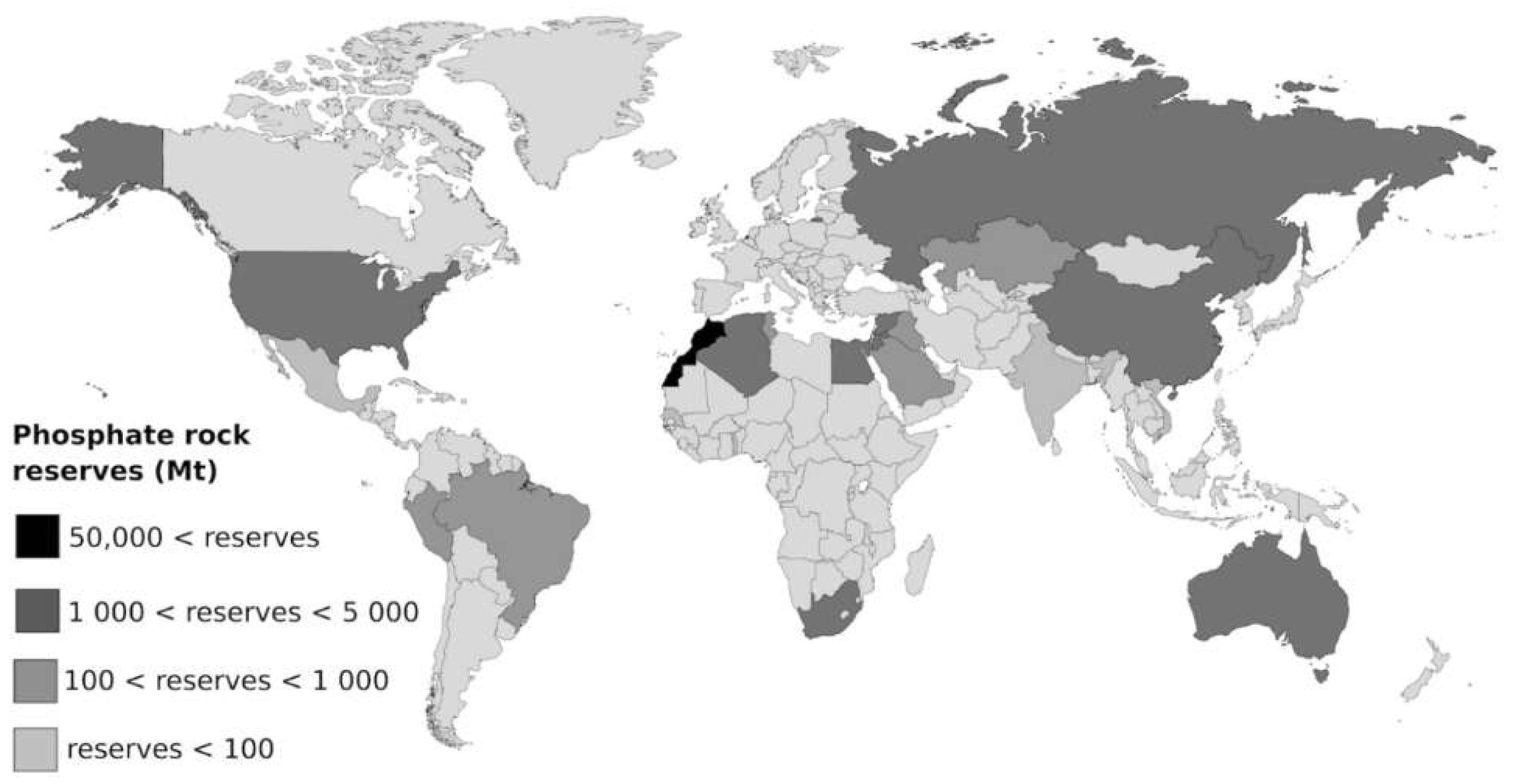
2. Phosphorus Rock Reserves
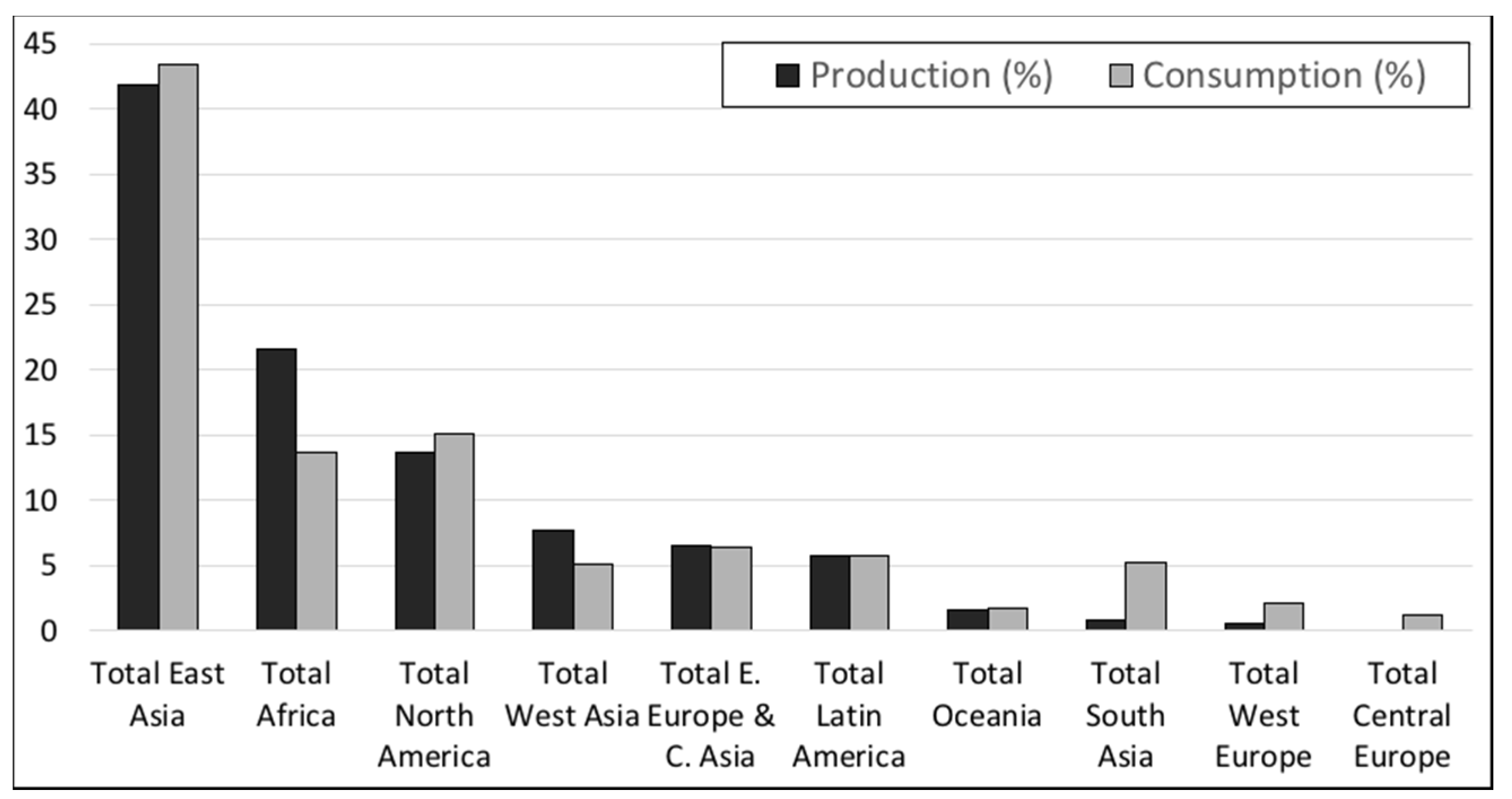
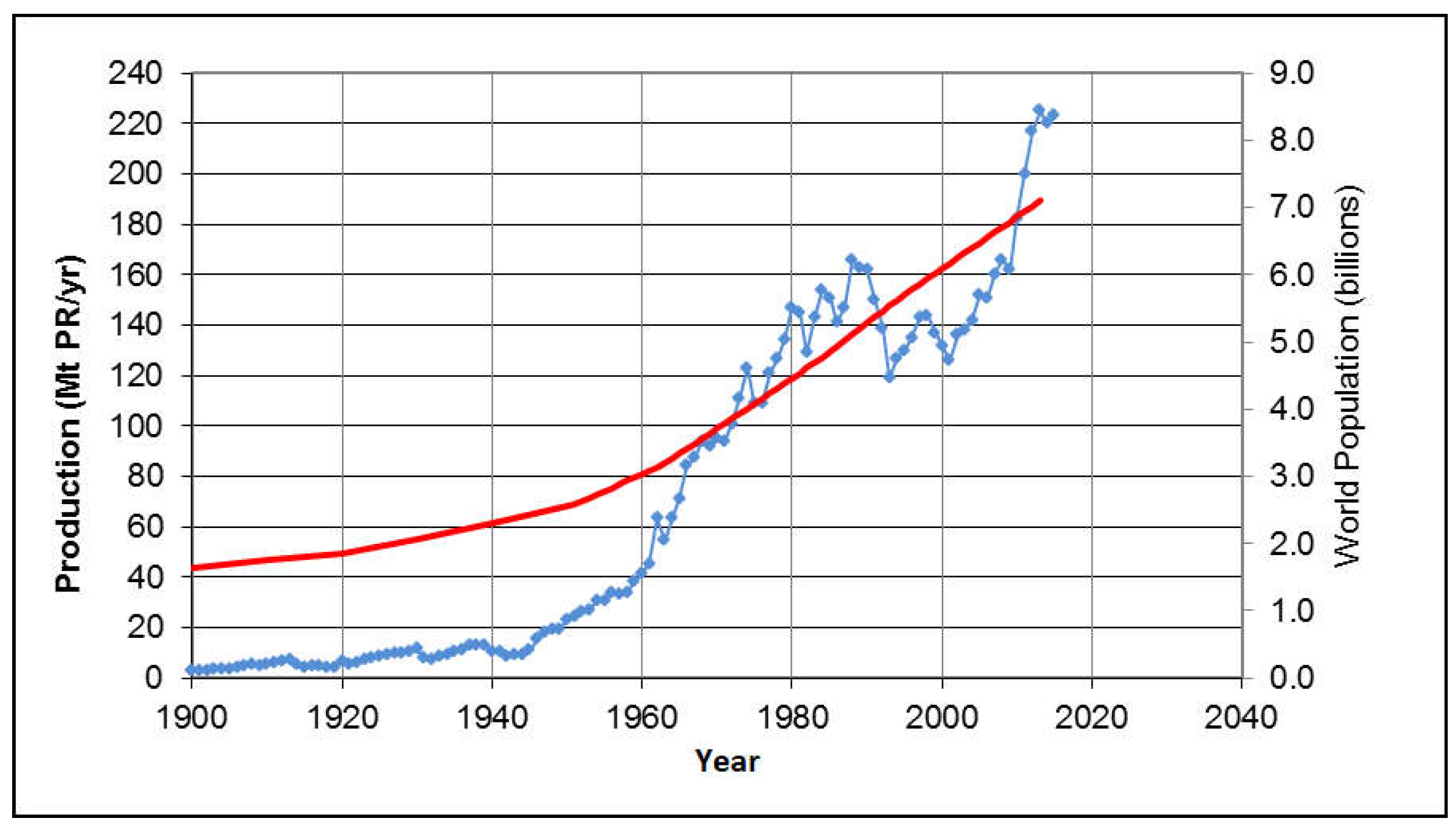
2.1. Current Phosphorus Production, Consumption and Resources
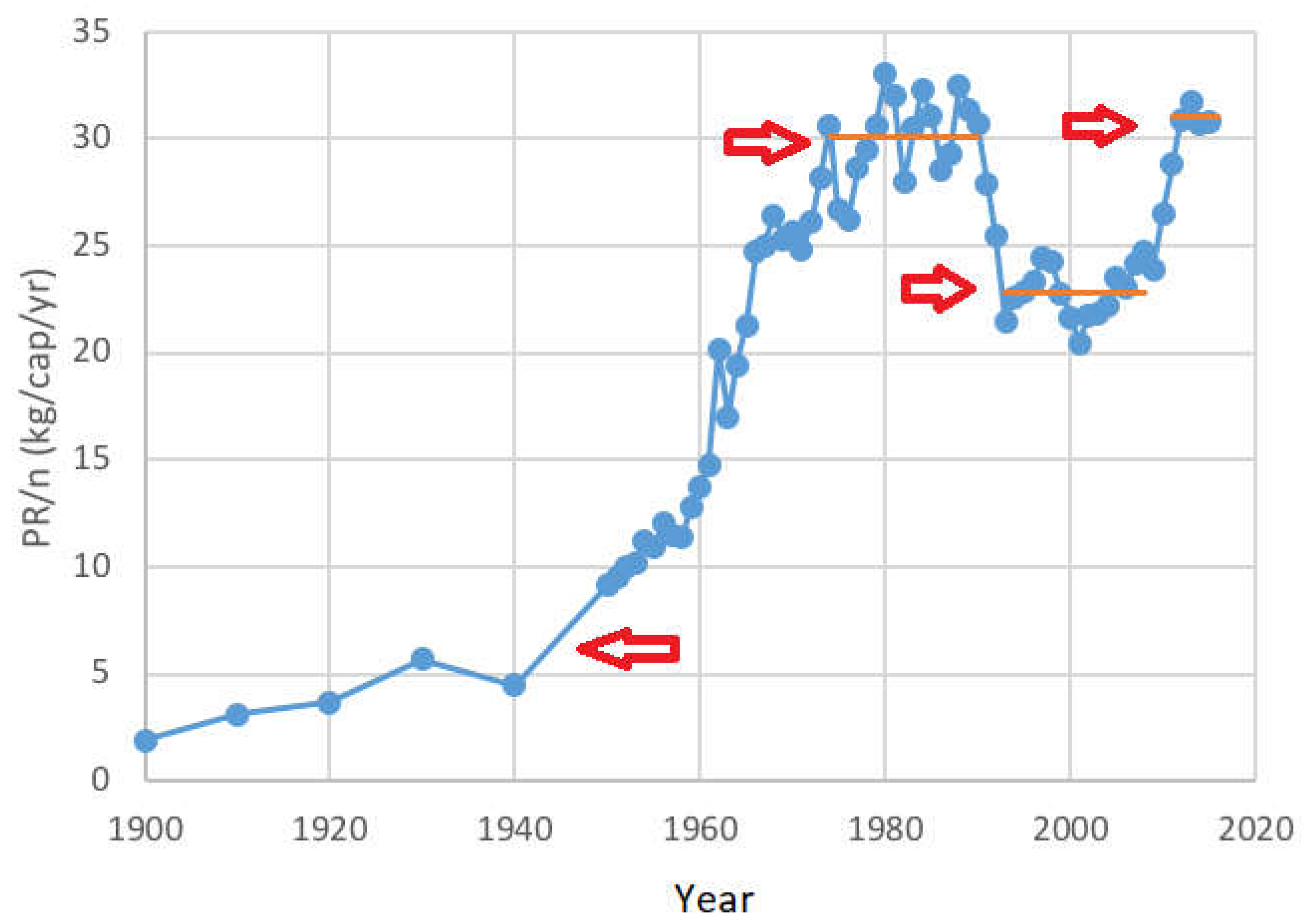
2.2. Future Trends
- (1)
- There was a large per-capita PR increase after World War II through the 1970s, which could be attributed to the “green revolution” of the time;
- (2)
- These high levels stabilized from 1975 to 1991 at an average of 30.1 kg PR per capita, per year;
- (3)
- This was followed by a 24% reduction for reasons described in Section 2.1, to about 22.8 kg PR/cap/yr from 1993 to 2006;
- (4)
- In the last decade, the per capita consumption recovered to about 31.0 kg PR/cap/yr. This may be due to improved diet in historically undernourished regions, and due to an increased amount of meat in the diet of nations such as China that have experienced rapid economic development.
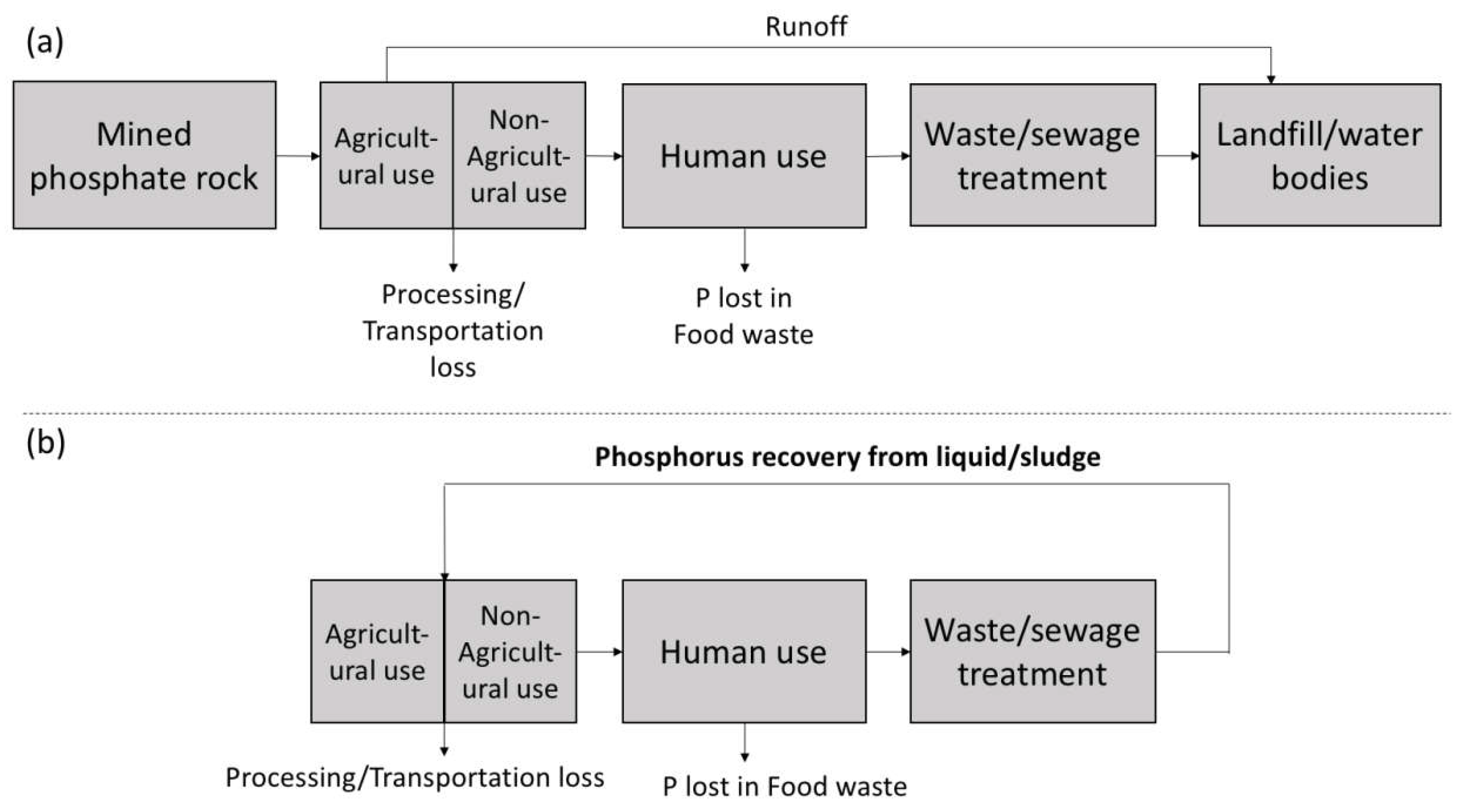
3. Phosphorus Cycle
Streamlining the Human P Cycle
4. Environmental Effects of Phosphorus Pollution
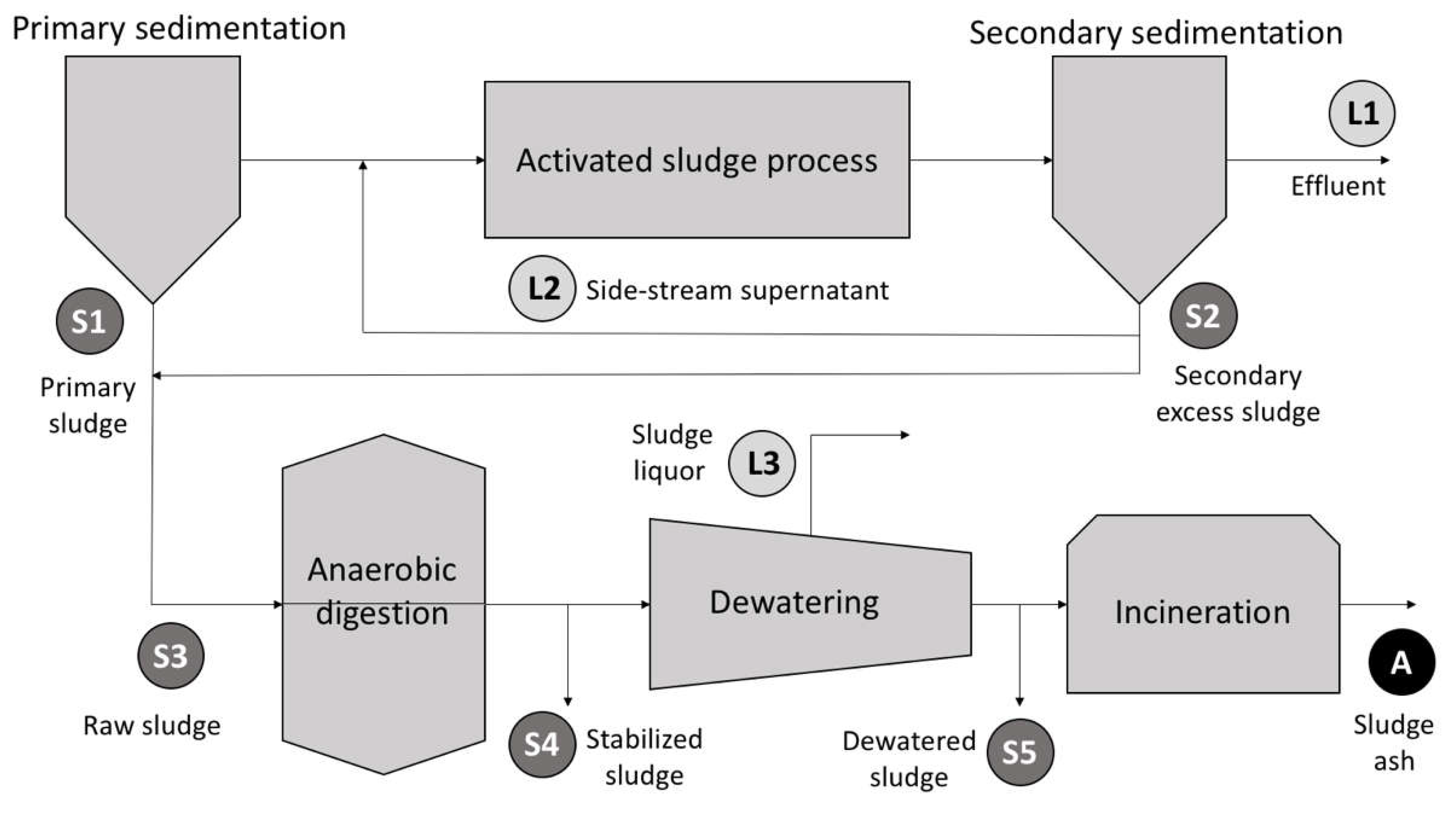
Phosphorus Removal
5. Phosphorus Recovery
5.1. Struvite Crystallization
5.2. P Recovery by Assimilation
5.3. Membrane Technologies
6. Considerations on the Energy-Nutrient Nexus in WWTPs
7. Conclusions
Author Contributions
Acknowledgments
Conflicts of Interest
References
- Pfitzner, A.; Bräu, M.F.; Zweck, J.; Brunklaus, G.; Eckert, H. Phosphorus Nanorods—Two allotropic modifications of a long-known element. Angew. Chem. Int. Ed. 2004, 43, 4228–4231. [Google Scholar] [CrossRef] [PubMed]
- Boskey, A. Bone composition: Relationship to bone fragility and antiosteoporotic drug effects. BoneKey Rep. 2013, 2, 447. [Google Scholar] [CrossRef] [PubMed]
- Childers, D.L.; Corman, J.; Edwards, M.; Elser, J.J. Sustainability Challenges of Phosphorus and Food: Solutions from Closing the Human Phosphorus Cycle. Bioscience 2011, 61, 117–124. [Google Scholar] [CrossRef]
- Pasek, M.A.; Sampson, J.M.; Atlas, Z. Redox chemistry in the phosphorus biogeochemical cycle. PNAS 2014, 111, 15468–15473. [Google Scholar] [CrossRef] [PubMed]
- Glindemann, D.; Edwards, M.; Liu, J.; Kuschk, P. Phosphine in soils, sludges, biogases and atmospheric implications—A review. Ecol. Eng. 2005, 24, 457–463. [Google Scholar] [CrossRef]
- Filippelli, G.M. Phosphate rock formation and marine phosphorus geochemistry: The deep time perspective. Chemosphere 2011, 84, 759–766. [Google Scholar] [CrossRef] [PubMed]
- Sheldon, R.P. Industrial Minerals—With Emphasis on Phosphate Rock. In Resources and World Development; McLaren, D.J., Skinner, B.J., Eds.; John Wiley & Sons Ltd.: New York, NY, USA, 1987; pp. 347–361. [Google Scholar]
- IFA. International Fertilizer Association Production and International Trade Report. 2016. Available online: https://fertilizer.org/Statistics (accessed on 20 March 2017).
- Smil, V. Phosphorus: Global Transfers, Causes and consequences of global environmental change. In Encyclopedia of Global Environmental Change; Douglas, I., Munn, T., Eds.; John Wiley & Sons, Ltd.: Chichester, UK, 2002; Volume 3, pp. 536–542. ISBN 0-471-97796-9. [Google Scholar]
- Vaccari, D.A. How do you spot a trend? An examination of recent phosphate rock production. In the special issue on Sustainable Phosphorus. Glob. Environ. Res. 2015, 19, 3–8. [Google Scholar]
- Jasinski, S.M. Phosphate Rock, USGS Mineral Commodities Summary. Available online: http://minerals.usgs.gov/minerals/pubs/commodity/phosphate_rock/ (accessed on 15 March 2017).
- Notholt, A.J.G.; Sheldon, R.P.; Davidson, D.F. Phosphate Deposits of the World. Volume 2: Phosphate Rock Resources; Cambridge University Press: Cambridge, UK, 1989. [Google Scholar]
- Cordell, D.; Drangert, J.O.; White, S. The story of phosphorus: Global food security and food for thought. Glob. Environ. Chang. 2009, 19, 292–305. [Google Scholar] [CrossRef]
- De Ridder, M.; de Jong, S.; Polchar, J.; Lingermann, S. Risks and Opportunities in the Global Phosphate Rock Market; The Hague Centre for Strategic Studies (HCSS): The Hague, The Netherlands, 2012. [Google Scholar]
- Vaccari, D.A. Phosphorus: A Looming Crisis. Sci. Am. 2009, 300, 54–59. [Google Scholar] [CrossRef] [PubMed]
- Van Kauwenbergh, S.J. World Phosphate Rock Reserves and Resources; International Fertilizer Development Center: Muscle Shoals, AL, USA, 2010; ISBN 978-9-88999-167-3. [Google Scholar]
- Edixhoven, J.D.; Gupta, J.; Savenije, H.H.G. Recent revisions of phosphate rock reserves and resources: Reassuring or misleading? An in-depth literature review of global estimates of phosphate rock reserves and resources. Earth Syst. Dyn. 2013, 4, 1005–1034. [Google Scholar] [CrossRef]
- Gharbi, A. Les Phosphates Marocains, Chronique de la Recherche Minière; Bureau de Récherches Géologiques et Minières: Orléans, France, 1998; pp. 531–532.
- Terrab, M. IMPHOS Annual Report; World Phosphate Institute: Casablanca, Morocco, 2006. [Google Scholar]
- Cordell, D.; White, S.; Lindström, T. Peak phosphorus: The crunch time for humanity? The Sustainability Review. 2011. Available online: http://www.thesustainabilityreview.org/articles/peak-phosphorus-the-crunch-time-for-humanity (accessed on 20 April 2017).
- Mew, M. Future Phosphate Rock Production—Peak or Plateau? Fertecon Research Centre Limited—A Matter of Opinion. 2011. Available online: http://www.fertecon-frc.info/page15.htm (accessed on 1 October 2012).
- Vaccari, D.A.; Strigul, N. Extrapolating phosphorus production to estimate resource reserves. Chemosphere 2011, 84, 792–797. [Google Scholar] [CrossRef] [PubMed]
- Scholz, R.W.; Wellmer, F.W. Approaching a dynamic view on the availability of mineral resources: What we may learn from the case of phosphorus? Glob. Environ. Chang. 2013, 23, 11–27. [Google Scholar] [CrossRef]
- Walan, P.; Davidsson, S.; Johansson, S.; Höök, M. Phosphate rock production and depletion: Regional disaggregated modeling and global implications. Resour. Conserv. Recycl. 2014, 93, 178–187. [Google Scholar] [CrossRef]
- Cooper, J.; Lombardi, R.; Boardman, D.; Carliell-Marquet, C. The future distribution and production of global phosphate rock reserves. Resour. Conserv. Recycl. 2011, 57, 78–86. [Google Scholar] [CrossRef]
- Leopold, A. A Sand County Almanac and Sketches Here and There; Oxford University Press: New York, NY, USA, 1943; ISBN 978-0-19-500777-0. [Google Scholar]
- Filippelli, G.M. The Global Phosphorus Cycle: Past, Present, and Future. Elements 2013, 4, 89–95. [Google Scholar] [CrossRef]
- Pierrou, U. The Global Phosphorus Cycle. Ecol. Bull. Nitrogen Phosphorus Sulphur Glob. Cycles Scope Rep. 1976, 22, 75–88. [Google Scholar]
- Broecker, W.S. Chemical Oceanography; Harcourt Brace Jovanowich Inc.: San Diego, CA, USA, 1976; ISBN 0-15-506437-1. [Google Scholar]
- Desmidt, E.; Ghyselbrecht, K.; Zhang, Y.; Pinoy, P.; Van der Bruggen, B.; Verstraete, W.; Rabaey, K.; Meesschaert, B. Global Phosphorus Scarcity and Full-Scale P-Recovery Techniques: A Review. Crit. Rev. Environ. Sci. Technol. 2015, 45, 336–384. [Google Scholar] [CrossRef]
- Søndergaard, M.; Jensen, J.P.; Jeppesen, E. Role of sediment and internal loading of phosphorus in shallow lakes. Hydrobiologia 2003, 506–509, 135–145. [Google Scholar] [CrossRef]
- Scharf, W. Restoration of the highly eutrophic lingese reservoir. Hydrobiologia 1999, 416, 85–96. [Google Scholar] [CrossRef]
- Granéli, W. Internal phosphorus loading in Lake Ringsjön. In Nutrient Reduction and Biomanipulation as Tools to Improve Water Quality: The Lake Ringsjön Story; Hansson, L.A., Bergman, E., Eds.; Developments in Hydrobiology; Springer: Dordrecht, The Netherlands, 1999; Volume 140, pp. 19–26. ISBN 978-94-017-2462-3. [Google Scholar]
- Bendoricchio, G.; Di Luzio, M.; Baschieri, P.; Capodaglio, A.G. Diffuse pollution in the Lagoon of Venice. Water Sci. Technol. 1993, 28, 69–78. [Google Scholar] [CrossRef]
- Capodaglio, A.G.; Muraca, A.; Becchi, G. Accounting for water quality effects of future urbanization: Diffuse pollution loads estimates and control in Mantua’s Lakes (Italy). Water Sci. Technol. 2003, 47, 291–298. [Google Scholar] [CrossRef] [PubMed]
- Capodaglio, A.G.; Boguniewicz, J.; Llorens, E.; Salerno, F.; Copetti, D.; Legnani, E.; Buraschi, E.; Tartari, G. Integrated lake/catchment approach as a basis for the implementation of the WFD in the Lake Pusiano watershed. In Proceedings of the Conference on River Basin Management—Progress TOWARDS Implementation of the European Water Framework Directive; Scimago Lab: Budapest, Hungary, 2005; pp. 77–86. [Google Scholar]
- Capodaglio, A.G.; Hlavínek, P.; Raboni, M. Physico-chemical technologies for nitrogen removal from wastewaters: A review. Rev. Ambient. Agua 2015, 10, 481–498. [Google Scholar] [CrossRef]
- Capodaglio, A.G.; Hlavínek, P.; Raboni, M. Advances in wastewater nitrogen removal by biological processes: State of the art review. Rev. Ambient. Agua 2016, 11, 250–267. [Google Scholar] [CrossRef]
- Capodaglio, A.G.; Ghilardi, P.; Boguniewicz-Zablocka, J. New paradigms in urban water management for conservation and sustainability. Water Pract. Technol. 2016, 11, 176–186. [Google Scholar] [CrossRef]
- Capodaglio, A.G. Integrated, Decentralized Wastewater Management for Resource Recovery in Rural and Peri-Urban Areas. Resources 2017, 6, 22. [Google Scholar] [CrossRef]
- Capodaglio, A.G.; Callegari, A.; Cecconet, D.; Molognoni, D. Sustainability of decentralized wastewater treatment technologies. Water Pract. Technol. 2017, 12, 463–477. [Google Scholar] [CrossRef]
- Council Directive 91/271/EEC, Official Journal of the European Communities. 1991. 135/40. Available online: https://eurlex.europa.eu/legalcontent/EN/TXT/PDF/?uri=CELEX:31991L0271&from=EN (accessed on 20 April 2017).
- Yang, L.; Zhou, H.; Moccia, R. Membrane Filtration Coupled with Chemical Precipitation to Treat Recirculating Aquaculture System Effluent. J. Environ. Qual. 2006, 35, 2419–2424. [Google Scholar] [CrossRef] [PubMed]
- Barnard, J.L. A Review of Biological Phosphorus Removal in the Activated Sludge Process. Water SA 1976, 2, 136–144. [Google Scholar]
- Schoumans, O.F.; Rulkens, W.H.; Oenema, O.; Ehlert, P.A.I. Phosphorus Recovery from Animal Manure: Technical Opportunities and Agro-Economical Perspectives; Alterra Report; Alterra: Wageningen, The Netherlands, 2010. [Google Scholar]
- Cornel, P.; Schaum, C. Phosphorus recovery from wastewater: Needs, techniques and costs. Water Sci. Technol. 2009, 59, 1069–1076. [Google Scholar] [CrossRef] [PubMed]
- Donatello, S.; Cheeseman, C.R. Recycling and recovery routes for incinerated sewage sludge ash (ISSA): A review. Waste Manag. 2013, 33, 2328–2340. [Google Scholar] [CrossRef] [PubMed]
- Mayer, B.K.; Baker, L.A.; Boyer, T.H.; Drechsel, P.; Gifford, M.; Hanjra, M.A.; Parameswaran, P.; Stoltzfus, J.; Westerhodd, P.; Rittmann, B.E. Total value of phosphorus recovery. Environ. Sci. Technol. 2016, 50, 6606–6620. [Google Scholar] [CrossRef] [PubMed]
- Schipper, W.J.; Klapwijk, A.; Potjer, B.; Rulkens, W.H.; Temmink, B.G.; Kiestra, F.D.G.; Lijmbach, A.C.M. Phosphate recycling in the phosphorus industry. Environ. Technol. 2001, 22, 1337–1345. [Google Scholar] [CrossRef] [PubMed]
- Sartorius, C.; von Horn, J.; Tettenborn, F. Phosphorus recovery from wastewater expert survey on present use and future potential. Water Environ. Res. 2012, 84, 313–322. [Google Scholar] [CrossRef] [PubMed]
- Cost-Effective and Eco-Friendly Struvite Wastewater Treatment Products. Available online: http://www.struvite.info (accessed on 8 April 2018).
- Borgerding, J. Phosphate deposits in digestion systems. J. Water Pollut. Control Fed. 1972, 44, 813–819. [Google Scholar]
- Parsons, S.A. Recent scientific and technical developments: Struvite precipitation. CEEP Scope Newsl. 2001, 41, 15–22. [Google Scholar]
- Le Corre, K.S.; ValsamieJones, E.; Hobbs, P.; Parsons, S.A. Phosphorus recovery from wastewater by struvite crystallization: A review. Crit. Rev. Environ. Sci. Technol. 2009, 39, 433–477. [Google Scholar] [CrossRef]
- Le Corre, K.S.; Valsami-Jones, E.; Hobbs, P.; Parsons, S.A. Impact of calcium on struvite crystal size, shape and purity. J. Cryst. Growth 2005, 283, 514–522. [Google Scholar] [CrossRef]
- Crutchik, D.; Garrido, J.M. Struvite crystallization versus amorphous magnesium and calcium phosphate precipitation during the treatment of a saline industrial wastewater. Water Sci. Technol. 2011, 64, 2460–2467. [Google Scholar] [CrossRef] [PubMed]
- Pastor, L.; Mangin, D.; Barat, R.; Seco, A. A pilot-scale study of struvite precipitation in a stirred tank reactor: Conditions influencing the process. Bioresour. Technol. 2008, 99, 6285–6291. [Google Scholar] [CrossRef] [PubMed]
- Hao, X.D.; Wang, C.C.; Lan, L.; Van Loosdrecht, M.C.M. Struvite formation, analytical methods and effects of pH and Ca2+. Water Sci. Technol. 2008, 58, 1687–1692. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.; Yoo, B.; Lim, S.J.; Kim, T.; Kim, S.; Kim, J.Y. Development and validation of an equilibrium model for struvite formation with calcium co-precipitation. J. Cryst. Growth 2013, 372, 129–137. [Google Scholar] [CrossRef]
- Peng, L.; Dai, H.; Wu, Y.; Peng, Y.; Lu, X. A comprehensive review of the available media and approaches for phosphorus recovery from wastewater. Water Air Soil Pollut. 2018, 229, 115. [Google Scholar] [CrossRef]
- Liu, Y.H.; Rahman, M.M.; Kwag, J.H.; Kim, J.H.; Ra, C.S. Eco-friendly production of maize using struvite re- covered from swine wastewater as a sustainable fertilizer source. Asian Austral. J. Anim. Sci. 2011, 24, 1699–1705. [Google Scholar] [CrossRef]
- Regy, S.; Mangin, D.; Klein, J.P.; Lieto, J. Phosphate Recovery by Struvite Precipitation in a Stirred Reactor, Phosphate Recovery in Waste Water by Crystallization; CEEP: Southampton, UK, 2002; pp. 54–58. [Google Scholar]
- Yu, R.; Geng, J.; Ren, H.; Wang, Y.; Xu, K. Struvite pyrolysate recycling combined with dry pyrolysis for ammonium removal from wastewater. Bioresour. Technol. 2013, 132, 154–159. [Google Scholar] [CrossRef] [PubMed]
- Shih, Y.J.; Abarca, R.R.M.; Luna, M.D.G.D.; Huang, Y.H.; Lu, M.C. Recovery of phosphorus from synthetic wastewaters by struvite crystallization in a fluidized-bed reactor: Effects of pH, phosphate concentration and coexisting ions. Chemosphere 2017, 173, 466–473. [Google Scholar] [CrossRef] [PubMed]
- Bojcevska, H.; Raburu, P.O.; Tonderski, K.S. Free water surface wetlands for polishing sugar factory effluent in western kenya-macrophyte nutrient recovery and treatment results. In Proceedings of the 10th International Conference on Wetland Systems for Water Pollution Control, Lisbon, Portugal, 23–29 September 2006; pp. 709–718. [Google Scholar]
- Yuan, Z.; Pratt, S.; Batstone, D.J. Phosphorus recovery from wastewater through microbial processes. Curr. Opin. Biotechnol. 2012, 23, 878–883. [Google Scholar] [CrossRef] [PubMed]
- Shilton, A.N.; Powell, N.; Guieysse, B. Plant-based phosphorus recovery from wastewater via algae and macrophytes. Curr. Opin. Biotechnol. 2012, 23, 884–889. [Google Scholar] [CrossRef] [PubMed]
- Abis, K.L.; Mara, D.D. Primary facultative ponds in the UK: The effect of operational parameters on performance and algal populations. Water Sci. Technol. 2005, 51, 61–67. [Google Scholar] [CrossRef] [PubMed]
- Yaakob, Z.; Kamarudin, K.F.; Rajkumar, R.; Takriff, M.S.; Badar, S.N. The current methods for the biomass production of the microalgae from wastewaters: An overview. World Appl. Sci. J. 2014, 31, 1744–1758. [Google Scholar] [CrossRef]
- Garcia, J.; Mujeriego, R.; Bourrouet, A.; Penuelas, G.; Freixes, A. Wastewater treatment by pond systems: Experiences in Catalonia, Spain. Water Sci. Technol. 2000, 42, 35–42. [Google Scholar] [CrossRef]
- Richmond, A. Handbook of Microalgal Culture: Biotechnology and Applied Phycology; Wiley-Blackwell: Hoboken, NJ, USA, 2004. [Google Scholar]
- El Hamouri, B. Rethinking natural, extensive systems for tertiary treatment purposes: The high-rate algae pond as an example. Desalination Water Treat. 2009, 4, 128–134. [Google Scholar] [CrossRef]
- Tarayre, C.; De, C.L.; Charlier, R.; Michels, E.; Meers, E.; Camargo-Valero, M.; Delvigne, F. New perspectives for the design of sustainable bioprocesses for phosphorus recovery from waste. Bioresour. Technol. 2016, 206, 264–274. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Chang, W.C.; Tang, C.Y. Osmotic membrane bioreactor (OMBR) technology for wastewater treatment and reclamation: Advances, challenges and prospects for the future. J. Membr. Sci. 2016, 504, 113–132. [Google Scholar] [CrossRef]
- Qiu, G.; Ting, Y.P. Direct phosphorus recovery from municipal wastewater via osmotic membrane bioreactor (OMBR) for wastewater treatment. Bioresour. Technol. 2014, 170, 221–229. [Google Scholar] [CrossRef] [PubMed]
- Achilli, A.; Cath, T.Y.; Marchand, E.A.; Childress, A.E. The forward osmosis membrane bioreactor: A low fouling alternative to MBR processes. Desalination 2009, 239, 10–21. [Google Scholar] [CrossRef]
- Holloway, R.W.; Achilli, A.; Cath, T.Y. The osmotic membrane bioreactor: A critical review. Environ. Sci. Water Res. Technol. 2015, 1, 581–605. [Google Scholar] [CrossRef]
- Qui, G.; Zhang, S.; Raghavan, D.S.S.; Das, S.; Ting, Y. The potential of hybrid forward osmosis membrane bioreactor (FOMBR) processes in achieving high throughput treatment of municipal wastewater with enhanced phosphorus recovery. Water Res. 2016, 105, 370–382. [Google Scholar] [CrossRef]
- Qiu, G.; Law, Y.M.; Das, S.; Ting, Y.P. Direct and complete phosphorus recovery from municipal wastewater using a hybrid microfiltration-forward osmosis membrane bioreactor process with seawater brine as draw solution. Environ. Sci. Technol. 2015, 49, 6156–6163. [Google Scholar] [CrossRef] [PubMed]
- Luo, W.; Hai, F.I.; Price, W.E.; Guo, W.; Ngo, H.H.; Yamamoto, K.; Nghiem, L.D. Phosphorus and water recovery by a novel osmotic membrane bioreactor-reverse osmosis system. Bioresour. Technol. 2016, 200, 297–304. [Google Scholar] [CrossRef] [PubMed]
- Thong, Z.; Cue, Y.; Ong, Y.K.; Chung, T.S. Molecular design of nanofiltration membranes for the recovery of phosphorus from sewage sludge. ACS Sustain. Chem. Eng. 2016, 4, 5570–5577. [Google Scholar] [CrossRef]
- Hau, N.T.; Chen, S.S.; Nguyen, N.C.; Huang, K.Z.; Ngo, H.H.; Guo, W. Exploration of EDTA sodium salt as novel draw solution in forward osmosis process for dewatering of high nutrient sludge. J. Membr. Sci. 2014, 455, 305–311. [Google Scholar] [CrossRef]
- Electric Power Research Institute (EPRI). Water & Sustainability: U.S. Electricity Consumption for the Water Supply & Treatment; The Next Half Century Topical Report; EPRI: Palo Alto, CA, USA, 2002. [Google Scholar]
- De Haas, D.; Dancey, M. Wastewater treatment energy efficiency, a review with current Australian prospectives. AWA J. 2015. [Google Scholar] [CrossRef]
- MURL—Ministry for Environment. Nature Protection, Agriculture & Consumer Protection in the German State of North Rhine Westphalia: Energy in WWTPs; MURL: Düsseldorf, Germany, 1999. (In German)
- Gellings, C.W.; Parmenter, K.E. Energy Efficiency in Fertilizer Production and Use. Efficient Use and Conservation of Energy; Encyclopedia of life support systems (EOLSS): Oxford, UK, 2004; Volume 2. [Google Scholar]
- Daw, J.; Hallett, K.; DeWolfe, J.; Venner, I. Energy Efficiency Strategies for Municipal Wastewater Treatment Facilities; NREL/TP-7A30-53341; National Renewable Energy Laboratory: Golden, CO, USA, 2012.
- Vanrolleghem, P.A.; Vaneeckhaute, C. Resource Recovery from Wastewater and Sludge: Modelling and Control Challenges. In Proceedings of the IWA Specialist Conference on Global Challenges: Sustainable Wastewater Treatment and Resource Recovery, Kathmandu, Nepal, 26–30 October 2014. [Google Scholar]
- Ráduly, B.; Gernaey, K.V.; Capodaglio, A.G.; Mikkelsen, P.S.; Henze, M. Artificial neural networks for rapid WWTP performance evaluation: Methodology and case study. Environ. Model. Softw. 2007, 22, 1208–1216. [Google Scholar] [CrossRef]
- Vaneeckhaute, C.; Claeys, F.H.A.; Tack, F.M.G.; Meers, E.; Belia, E.; Vanrolleghem, P.A. Development, implementation, and validation of a generic nutrient recovery model (NRM) library. Environ. Model. Softw. 2018, 99, 170–209. [Google Scholar] [CrossRef]
- Batstone, D.J.; Amerlinck, Y.; Ekama, G.; Goel, R.; Grau, P.; Johnson, B.; Kaya, I.; Steyer, J.-P.; Tait, S.; Takács, I.; et al. Towards a generalized physicochemical framework. Water Sci. Technol. 2012, 66, 1147–1161. [Google Scholar] [CrossRef] [PubMed]
- Wett, B.; Buchauer, K.; Fimml, C. Energy self-sufficiency as a feasible concept for wastewater treatment systems. In Proceedings of the IWA Leading Edge Technology Conference, Asian Water, Singapore, 3–6 June 2007. [Google Scholar]
- Chen, S.; Chen, B. Net energy production and emissions mitigation of domestic wastewater treatment system: A comparison of different biogas-sludge use alternatives. Bioresour. Technol. 2013, 144, 296–303. [Google Scholar] [CrossRef] [PubMed]
- McCarty, P.L.; Kim, J.; Bae, J. Domestic wastewater treatment as a net energy producer e can this be achieved? Environ. Sci. Technol. 2011, 45, 7100–7106. [Google Scholar] [CrossRef] [PubMed]
- Kelly, P.T.; He, Z. Nutrients removal and recovery in bioelectrochemical systems: A review. Bioresour. Technol. 2014, 153, 351–360. [Google Scholar] [CrossRef] [PubMed]
- Capodaglio, A.G.; Molognoni, D.; Dallago, E. Microbial Fuel Cells for Direct Electrical Energy Recovery from Urban Wastewaters. Sci. World J. 2013, 634738. [Google Scholar] [CrossRef] [PubMed]
- Capodaglio, A.G.; Molognoni, D.; Puig, S.; Balaguer, M.D.; Colprim, J. Role of Operating Conditions on Energetic Pathways in a Microbial Fuel Cell. Energy Procedia 2015, 74, 728–735. [Google Scholar] [CrossRef]
- Liu, H.; Ramnarayanan, R.; Logan, B.E. Production of electricity during wastewater treatment using a single chamber microbial fuel cell. Environ. Sci. Technol. 2004, 38, 2281–2285. [Google Scholar] [CrossRef] [PubMed]
- Cecconet, D.; Molognoni, D.; Callegari, A.; Capodaglio, A.G. Agro-food industry wastewater treatment with microbial fuel cells: Energetic recovery issues. Int. J. Hydrog. Energy 2018, 43, 500–511. [Google Scholar] [CrossRef]
- Kim, B.H. Microbial fuel cell. KISToday 2009, 2, 4–8. [Google Scholar]
- Mo, W.; Zhang, Q. Energy-nutrients-water nexus: Integrated resource recovery in municipal wastewater treatment plants. J. Environ. Manag. 2013, 127, 255–267. [Google Scholar] [CrossRef] [PubMed]
- Fischer, F.; Bastian, C.; Happe, M.; Mabillard, E.; Schmidt, N. Microbial fuel cell enables phosphate recovery from digested sewage sludge as struvite. Bioresour. Technol. 2011, 102, 5824–5830. [Google Scholar] [CrossRef] [PubMed]
- Ichihashi, O.; Hirooka, K. Removal and recovery of phosphorus as struvite from swine wastewater using microbial fuel cell. Bioresour. Technol. 2012, 114, 303–307. [Google Scholar] [CrossRef] [PubMed]
- Cusick, R.D.; Logan, B.E. Phosphate recovery as struvite within a single chamber microbial electrolysis cell. Bioresour. Technol. 2012, 107, 110–115. [Google Scholar] [CrossRef] [PubMed]
- You, J.; Greenman, J.; Melhuish, C.; Ieropoulos, I. Electricity generation and struvite recovery from human urine using microbial fuel cells. J. Chem. Technol. Biotechnol. 2014, 91, 647–654. [Google Scholar] [CrossRef]
- Jaffer, Y.; Clark, T.A.; Pearce, P.; Parsons, S.A. Potential phosphorus recovery by struvite formation. Water Res. 2002, 36, 1834–1842. [Google Scholar] [CrossRef]

| Source | Production | Reserves | R/C | ||
|---|---|---|---|---|---|
| (Mt/yr) | (%) | (Mt) | (%) | (yrs) | |
| Morocco & Western Sahara | 30 | 13 | 50,000 | 73 | 1667 |
| China | 100 | 45 | 3700 | 5.4 | 37 |
| United States | 25.3 | 12 | 1100 | 1.6 | 40 |
| MENA-M/WS * | 25.7 | 12 | 8166 | 12 | 318 |
| Rest of the world | 37.2 | 17 | 5810 | 8 | 156 |
| World total | 218 | 68,776 | 315 | ||
| As P ** | 28.6 | 9005 | |||
| Sludge Type | Process Name | Method | Products | Operational Scale |
|---|---|---|---|---|
| Sludge liquor | P-ROC | Adsorption | CaP, CaP on CSH | Semi-industrial |
| RECYPHOS | Adsorption | FeP | Semi-industrial | |
| PHOSIEDI | Adsorption | CaP | Lab scale | |
| PHOSTRIP | Precipitation | CaP | Full scale | |
| PRISA | Precipitation | Struvite | Semi-industrial | |
| CRYSTALACTOR | Pellets | CaP, struvite | Full scale | |
| PEARL | Pellets | Struvite | Full scale | |
| Digested sludge | BERLINER VERFAHREN | Without leaching | Struvite | Full scale |
| FIX-PHOS | Without leaching | CaP on CSH | Lab scale | |
| SEABORNE | With leaching | Struvite | Full scale | |
| STUTTGARTER VERFAHREN | With leaching | Struvite | Full scale | |
| LOPROX/PHOXAN | With leaching | Phosphoric acid | Full scale | |
| CAMBI | With leaching | FeP, AlP, CaP | Lab scale/Full scale | |
| AQUA RECI | With leaching | FeP, AlP, CaP | Lab scale/Full scale | |
| K REPO | With leaching | FeP | - | |
| SEPHOS | With leaching | AlP, CaP | Lab scale | |
| SESAL-PHOS | With leaching | CaP | Lab scale | |
| P ASCH | With leaching | Struvite | Semi-industrial | |
| BIOLEACHING | With leaching | Struvite | Lab scale | |
| BIO CON | With leaching | Phosphoric acid | Semi-industrial | |
| Sludge ash | MEPHREC | Thermal treatment | CaP | Semi-industrial |
| ASH DEC | Thermal treatment | Fertilizer | Semi-industrial | |
| THERMPHOS | Thermal treatment | Elemental phosphorus | Industrial process | |
| PHOSPHORUS INDUSTRY | Thermal treatment | Fertilizer | Industrial process |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Daneshgar, S.; Callegari, A.; Capodaglio, A.G.; Vaccari, D. The Potential Phosphorus Crisis: Resource Conservation and Possible Escape Technologies: A Review. Resources 2018, 7, 37. https://doi.org/10.3390/resources7020037
Daneshgar S, Callegari A, Capodaglio AG, Vaccari D. The Potential Phosphorus Crisis: Resource Conservation and Possible Escape Technologies: A Review. Resources. 2018; 7(2):37. https://doi.org/10.3390/resources7020037
Chicago/Turabian StyleDaneshgar, Saba, Arianna Callegari, Andrea G. Capodaglio, and David Vaccari. 2018. "The Potential Phosphorus Crisis: Resource Conservation and Possible Escape Technologies: A Review" Resources 7, no. 2: 37. https://doi.org/10.3390/resources7020037
APA StyleDaneshgar, S., Callegari, A., Capodaglio, A. G., & Vaccari, D. (2018). The Potential Phosphorus Crisis: Resource Conservation and Possible Escape Technologies: A Review. Resources, 7(2), 37. https://doi.org/10.3390/resources7020037