Abstract
Residual sludge disposal costs may constitute up to, and sometimes above, 50% of the total cost of operation of a Wastewater Treatment Plant (WWTP) and contribute approximately 40% of the total greenhouse gas (GHG) emissions associated with its operation. Traditionally, wastewater sludges are processed for: (a) reduction of total weight and volume to facilitate their transfer and subsequent treatments; (b) stabilization of contained organic material and destruction of pathogenic microorganisms, elimination of noxious odors, and reduction of putrefaction potential and, at an increasing degree; (c) value addition by developing economically viable recovery of energy and residual constituents. Among several other processes, pyrolysis of sludge biomass is being experimented with by some researchers. From the process, oil with composition not dissimilar to that of biodiesels, syngas, and a solid residue can be obtained. While the advantage of obtaining sludge-derived liquid and gaseous fuels is obvious to most, the solid residue from the process, or char (also indicated as biochar by many), may also have several useful, initially unexpected applications. Recently, the char fraction is getting attention from the scientific community due to its potential to improve agricultural soils’ productivity, remediate contaminated soils, and supposed, possible mitigation effects on climate change. This paper first discusses sludge-pyrolysis-derived char production fundamentals (including relationships between char, bio-oil, and syngas fractions in different process operating conditions, general char properties, and possible beneficial uses). Then, based on current authors’ experiments with microwave-assisted sludge pyrolysis aimed at maximization of liquid fuel extraction, evaluate specific produced char characteristics and production to define its properties and most appropriate beneficial use applications in this type of setting.
1. Introduction
Residual sludge disposal costs may constitute up to, and sometimes above, 50% of the total cost of operation of a Wastewater Treatment Plant (WWTP) and contribute approximately 40% of the total greenhouse gas (GHG) emissions associated with its operation [1]. The safe disposal of such sludge is literally a “big” issue in urban wastewater treatment: at the European Union level, the 2012 sludge production was estimated at 11 million tons dry weight, and was expected by the year 2020 to have a further increase of more than 30%. Traditionally, wastewater sludges are processed for: (a) reduction of total weight and volume to facilitate their transport and subsequent treatments; (b) stabilization of contained organic material and destruction of pathogenic microorganisms, elimination of noxious odours, and reduction of putrefaction potential; (c) value addition by developing economically viable recovery of energy and residual constituents. After appropriate treatment, sludges are mostly disposed of in landfill, agriculture (including composting), or incinerated.
Landfill Directive 99/31/EC set restrictions (quantitative targets) for biodegradable municipal wastes (such as sewage sludge) disposed of in landfills; in addition, national legislations of some Member States have set very strict limits for organic matter or total organic carbon (TOC) contained in disposed sludge, prohibiting de facto its landfilling. According to recent Eurostat data, in fact, significant abandonment of sludge landfilling practice is occurring in most of Europe, except for Italy, Denmark, and Estonia.
Sewage Sludge Directive 86/278/EEC, concerning beneficial use of sludge on soils, sought to encourage safe use of sewage sludge in agriculture (composting is often included as a form of agricultural disposal in official reporting), regulating this form of disposal to prevent harmful effects. After concerns were raised about possible harmful compound accumulation in soils, 16 (out of 27) EU countries have set more stringent requirements for heavy metals in sludge, compared to the Directive’s provisions, and 10 countries set stricter limit values for heavy metals in soil. Such restrictions are being strengthened periodically, while most EU countries have outright prohibited the disposal of untreated sludge in soil. Composting is applied more often in the new EU-12 countries, compared to the “old” EU-15 members. According to Eurostat, an increasing trend of overall agricultural uses (including composting) is observed in 7 (out of 15) old countries and 6 (out of 12) new countries. The most significant increases are observed in Portugal, Cyprus, and Bulgaria, while significant decreasing trends are observed in Slovakia and Czech Republic, where direct agricultural disposal seems to be replaced by composting.
Finally, incineration is enforced in most EU-15 countries. Greece, Slovenia, Germany, and the Netherlands present the greatest increasing trends, even though the first two countries export sludge for incineration; Denmark, Austria, Belgium, and Italy show instead decreasing incineration trends [2].
Thermal processing of sludge remains, however, a common and efficient approach for the disposal of waste urban sludge without causing excess secondary pollution, used as much as possible in many countries. Thermal utilization of sludge comes into play when the sludge does not comply with, or is in excess of, requirements for disposal in agriculture, and allows forms of energetic recovery. Thermal processing of sludge can take several forms. Co-firing in power plants and heating plants with coal (approximately 5% sludge) do not significantly decrease the temperature of the combustion process and usually do not require extra investment costs for off-gas cleaning, as filters and separators can usually handle this component. Co-firing in cement kilns (in adequate proportions) was considered the most convenient technology in terms of both sludge disposal and utilization until recently: one ton of dried sludge can substitute up to 0.33 t of raw clinker material. Since ash from the sludge is bound to the cement clinker, this can also be considered a waste-less technology, or a ‘‘waste-to-energy’’ system, and is acceptable if no other environmentally friendly technology can be applied [3]. Incineration (with urban solid waste or in special sludge incinerators) is another common option, where the energy contained in the sludge contributes to the energy balance of the process.
An alternative option to classical sludge incineration is pyrolysis, which can achieve 50% or more reduction in waste volume, stabilization of organic matter, as well as recovery of valuable end products. Pyrolysis is a thermal degradation of organic material in an oxygen-deficient atmosphere, a second-generation, alternative bioenergy production technology that is relatively simple, inexpensive, and robust, and can be used for transforming biomass into products such as bio-oil, solid residue, and syngas. Among several other processes, microwave-assisted pyrolysis (MAP) of sludge biomass was experimented with by some researchers [4]. In MAP, the feedstock (usually at an optimal humidity of around 10%) is targeted in the reaction vessel by microwaves with uniform and efficient heating, therefore the process takes generally a much shorter time than conventional pyrolysis, where heating is induced by convection. Obtained oil composition is not dissimilar to that of biodiesels obtained from common feedstock (food) crops, although with a slightly lower calorific value [5].
While the advantage of obtaining sludge-derived liquid fuels is easily obvious to most, the solid fraction residue from the process has also been found to have several useful, unexpected applications. In most of the literature on this topic, this solid residual is indifferently termed char or biochar, with a slight preference for the latter form in recent publications. Biochar is a new technical term indicating “the porous carbonaceous solid produced by the thermochemical conversion of organic materials in an oxygen depleted atmosphere that has physicochemical properties suitable for safe and long-term storage of carbon in the environment” [6]. Originally, the term “biochar” referred to the solid product of biomass pyrolysis and, strictly speaking, sewage-sludge solid residue satisfies this definition. At the moment, however, there is intensive research on pyrolysis application to a wide range of other feedstock, like sewage sludge, poultry litter waste, etc., in which ash content may be quite high, with the resulting solid pyrolysis products containing more than 50% (by weight) of ash. In 2012, the European Biochar Foundation (EBC) published the “Guidelines for a Sustainable Production of Biochar”, where it is stated that “a biochar’s carbon content must be higher than 50% of the dry mass, while pyrolysed organic matter with a carbon content lower than 50% is classified as Pyrogenic Carbonaceous Material (PCM)”. In such cases, the pyrolysis end product should not be defined as biochar [7]. In this paper, therefore, the term PCM will be used to indicate the solid residual from sewage sludge of other feedstocks with organic content most likely lower than 50%, while biochar will be used only for residuals that fulfil EBC’s definition. As a general term, the wording “(bio)char” or simply “char” will be used when referring indistinctively to solid residuals from pyrolysis.
Biochar and PCMs could be burned to generate energy, however, recently, both are getting attention from the scientific community due to their potential to improve agricultural soils’ productivity, remediate contaminated soils, and their supposed, possible mitigation effects on climate change. Process operational temperature has a substantial effect on the quality of (bio)char produced: that produced at low temperatures is most suitable for agricultural uses, due to higher carbon content and nutrient availability, while higher temperatures can improve its porosity and thus enhance its effectiveness in adsorbing contaminants present in soils. Researchers have shown that the pyrolysis process can suppress heavy metal release by non-impregnated biochars, resulting in an extremely low environmental risk using sludge-derived biochar as soil ammendant (unlike the use of sludge as is) [8]. (Bio)chars obtained under different processes significantly differ from one to another in their properties, depending on the type of biomass used to produce them, its growth conditions, and also on pyrolysis operating conditions.
This paper will first discuss sludge-pyrolysis-derived PCM production fundamentals (including relationships between PCM, bio-oil, and syngas fractions in different process operating conditions, general (bio)char properties, and possible beneficial uses). Based on current authors’ experiments with sludge pyrolysis aimed at maximization of liquid fuel extraction, specific PCM characteristics and production processes are evaluated to define PCM properties and the most appropriate beneficial use applications in this type of setting.
Recent trends and future possibilities of future (bio)char applications are discussed.
2. Fundamentals of Sludge Pyrolysis: Technologies and Final Products
Pyrolysis is a thermochemical process that can be used to transform biomass and other waste materials (e.g., rubber tyres) into bio-oil (energy content ~17 MJ/kg), (bio)char (energy content up to 18 MJ/kg), and syngas (energy content ~6 MJ/kg). The actual energy content values of these components vary significantly depending on feedstock and process conditions. The process has been used to produce “charcoal” for thousands of years; initially, the volatile fraction was usually dispersed in air, giving these systems a bad environmental reputation (causing, in fact, deforestation and severe air pollution). In modern facilities, technology has been modified to avoid gross pollution and transform all of the biomass into renewable energy products. Four classes of traditional pyrolysis processes can be identified (Table 1): slow pyrolysis, flash pyrolysis, gasification, and fast pyrolysis. In actuality, gasification cannot be properly classified as pyrolysis, since the latter consists of a devolatalization/decomposition of solid carbonaceous material, where gasification is a reaction of O2/CO2/H2O with the solid product of pyrolysis. In current literature, however, these two processes are often compared side-by-side, given the similarities of process conditions. As such they are both included in the following table.

Table 1.
Classes of pyrolysis processes (elaborated from: [9,10]).
2.1. Pyrolysis Products
Syngas is primarily composed of H2 and CO, with smaller quantities of CH4, CO2, H2O, and other low-molecular-weight volatile organics. While its heating value is usually low (~6 MJ/kg) compared to natural gas (~54 MJ/kg), it can provide fuel for hot water, drying, or electricity. Some time ago, before widespread availability of natural gas, syngas was generated to provide energy for home heating, cooking, street lighting, etc. (a.k.a. “town gas”).
Sanchez et al. [11] studied pyrolysis oil composition with gas chromatography–mass spectroscopy (GC–MS) and found that pyrolysis oil was a complex mixture of organic compounds. Volatile oil produced in the pyrolysis process showed high heat value, slightly lower than some fossil fuels; bio-oil consists of a wide range of oxygenated organic compounds including organic acids, aldehydes, alcohols, phenols, carbohydrates, and lignin-derived oligomers. Its chemical composition and energy value are quite variable and may vary substantially, depending on original feedstock and process conditions. Most bio-oils are acidic (pH ~ 2) and tend to solidify after being stored for prolonged periods of time, therefore their use as fuel oil in many desirable applications (i.e., home heating, diesel transportation fuel, exception made for industrial boilers) requires post-processing to correct acidity, improve stability, or upgrade it to synthetic transportation-grade fuels (biogasoline, biodiesel). Bio-oils can also be refined into ethanol and/or other chemical compounds. The European Union (EU) is currently pursuing an extended use of alternative liquid fuels in transportation uses, including bio-oils derived from biomasses [12].
(Bio)char is the solid residue of the pyrolysis process, with some energy content (generally speaking, the higher the carbon residue in the char, the higher its energy content) that can therefore be burned in systems fed with pulverized coal. NOx emissions from (bio)char combustion are comparable to those of coal, requiring similar abatement technologies. Some types of feedstock (i.e., urban sludge) may contain relatively high levels of metals that concentrate in the PCM residue after pyrolysis, and low levels of carbon, making it a poor fuel. Chars, however, may also have many other attractive, high-value uses in fields such as chemistry, metallurgy, agriculture, waste treatment, etc., that will be analysed in a later section.
2.2. Pyrolysis Technologies
In conventional biomass pyrolysis processes, energy production is the main target; in sewage sludge pyrolysis, the main purpose is its safe and economic disposal of a waste material, with energy (oil, syngas) and material (char) recovery as added benefit. Fundamentally, pyrolysis involves the heating of organic materials to high temperatures (usually greater than 400 °C) in the absence of oxygen. In these conditions, organic materials thermally decompose releasing a vapor phase and a residual solid phase. Both oil and gas leave the pyrolysis furnace in volatile form. After cooling, polar and high-molecular-weight compounds condense as liquid (bio-oil) from this phase, while low-molecular-weight volatile compounds remain in gas form (syngas). The main operating parameters in pyrolysis are heating rate, process temperature, and residence time, on which depend the physics and chemistry of the rather complex pyrolytic reactions.
Microwave technology has recently emerged as one of the most promising methods of enhancing and accelerating chemical reactions, due to effective heat transfer profiles. It is therefore being adopted as one of the best technologies available in pyrolytic processes, since it reduces residence time and brings significant energy savings [9]. So-called microwave-assisted pyrolysis (MAP) technology is an alternative heating method presenting several advantages over conventional pyrolysis, including: uniform internal heating for material particles, since electromagnetic energy is directly converted into heat at the molecular level; ease of control due to process instantaneous response; simple set-up, facilitating its adaptation to large-scale industrial processes; reduced need for feedstock grinding; low cost, as microwave is a mature and energy-efficient technology. The different heating mechanisms allowed by microwave properties (heat diffuses from within the microwaved mass and not from its exterior) make MAP products retain different characteristics than those obtained with conventional heating. MAP develops according to two stages: drying and pyrolysis (Figure 1). Since water has excellent microwave absorbability, biomass shows high temperature rising rates during initial drying. As the temperature continues to increase, the biomass becomes less absorptive towards microwave radiation, and temperature rising rates slow down [13]. Masek et al. [14] also confirmed that in the case of MAP, pyrolysis of some feedstocks can occur at even lower temperatures than those reported for conventional heating.
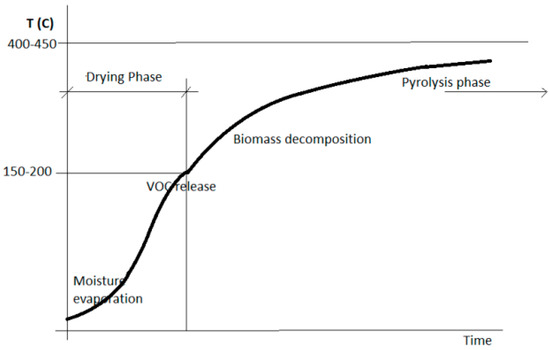
Figure 1.
Pyrolysis phases temperature profile (indicative): max. temperature and rates of increase depend on the actual process used and specific settings.
Due to the properties of microwaves, MAP could thus provide faster heating, better efficiency, and a faster, more controllable process compared to conventional pyrolysis. Unfortunately, while some feedstocks behave well with this process, sewage sludge biosolids are generally poor absorbers of microwaves [15]; dielectric materials, able to better absorb microwave energy, are often added to the sludge to reach the temperatures required for pyrolysis. These work as “hot spots”, absorbing microwave energy and conductively transferring it as heat to the surrounding material. Carbon-based materials, including (bio)char itself, are often selected as microwave absorbers because of their effectiveness and relatively low cost. Using monomodal microwave synthetizers (MMS) instead of multimodal ones (such as those commonly used in the kitchen), the need for preliminary mixing of dry sludge with microwave receptors additives, reported by earlier researchers, could be eliminated, and the process temperature needed for process completion could also be significantly lowered (as low as 270 °C), allowing the production of a larger liquid product (biodiesel) fraction. This process is discussed in greater detail in Capodaglio et al. [5].
Despite these and other advantages of microwave over conventional heating methods, only a few studies have been conducted on sewage sludge pyrolysis with microwave technology, and the effects of catalysts on the process.
MAP applied to bio-oil production from sewage sludge was reported by Tian et al. [16] as a feasible method to obtain increased yields from the process. It was shown that by adjusting applied power, it was possible to select the desired gas, bio-oil, and solid fraction yields. Maximum bio-oil yield of 49.8% was obtained at 400 W microwave exit power for 6 min; while increasing power from 200 W to 1200 W, the solid fraction yield decreased from 77.3 wt % to 32.6 wt %, and bio-gas yield increased from 4.4 wt % to 60.21 wt %, respectively. Bio-oil production from sewage sludge by MAP was also tested by Lin et al. [17], who studied the effects of reaction parameters and chemical additives on produced oil yield and quality. Five types of additives (KOH, H2SO4, H3BO3, ZnCl2, and FeSO4) were tested. All catalysts decreased the quantity of bio-oil produced, while KOH, H2SO4, H3BO3, and FeSO4 improved its quality (calorific value, density, viscosity, carbon content). ZnCl2 had a negative effect on the product’s quality. Dominguez et al. [18] compared bio-oil characteristics obtained from MAP and conventional pyrolysis. Three types of sewage sludge (two from urban WWTPs and one from a dairy factory) enriched with graphite (0.5–3 g/kg) were used as feedstock. Results showed that oil produced using a conventional furnace at high temperatures was of considerable environmental concern, containing “dirty” compounds such as PAHs in high quantity, while the one obtained from MAP had a higher calorific value and lower proportion of PAHs. The same group of researchers [19] later investigated MAP of sewage sludge for bio-fuel preparation using single-mode and multimode microwave cavities at constant input power, with tests duplication in an electric oven, applying graphite and char as microwave absorbers. The conclusion was that MAP improved the quality of gases produced (up to 38% increase in H2), in comparison with the conventional method, also improving char production and decreasing reaction time by over 51% to 10 min. Single-mode MAP was recognized as a better microwave source for gas production, while multimode yielded higher char production in the tests.
Monomodal Microwave assisted Pyrolysis (MMAP) is a MAP-alternative process that occurs when microwaves produced by a magnetron travel through special wave guides to a circulator that diverts reflected microwave power into an electromagnetically matched load. A microwave directional coupler, which allows measurement of forward and reflected power, and a three-stub tuner, matching the impedance of the wave-guide segments to the load, direct the microwaves into the single-mode chamber [20].
3. Sewage Sludge Char Characteristics
Excess (waste) sewage sludge contains valuable nutrients such as nitrogen, phosphorus, organic matter, and essential trace elements that can improve soil’s physical properties and, as fertilizers, increase crop yields. The concomitant contents of heavy metals and other toxic/dangerous compounds (micropollutants, pharmaceutical compounds), however, may affect soil–plant systems and threaten human health. It has been shown that pyrolytic conversion of sewage sludge is advantageous over conventional incineration concerning fuel economy, nutrient recovery, and control of heavy metal emissions. Most studies on sewage sludge pyrolysis refer to fuel recovery, however, the effects of sewage sludge char on soil, plant nutrients, and bioavailability of heavy metals in plants have seldom been studied [21].
Perhaps some of the most comprehensive studies on systematic evaluation of the properties of chars produced from different sewage sludges at different temperatures are those carried out by Zielinska et al. [22] and Capodaglio and Callegari [23]. Sewage sludge samples were subject to (traditional) pyrolysis in a laboratory furnace at temperatures of 500, 600, and 700 °C, with a heating rate of 25 °C/min, in an oxygen-free atmosphere maintained by constant flow of nitrogen gas. The results from the studies are compared with results obtained from other authors on char obtained from traditional and microwave-assisted sludge pyrolysis (Table 2).

Table 2.
Characteristics of biochar derived from sewage sludges with traditional and microwave-assisted pyrolysis.
3.1. Yield
From the results reported in Table 2, some considerations can be drawn considering sewage sludge char characteristics. First, an increase in pyrolysis temperature generally results in a decrease in char yield (as % dry weight), due to the volatilization of some organic fractions.
3.2. pH
Based on available literature, the pH of chars produced at low temperatures (≤500 °C) mostly depends on sludge pH, resulting in PCMs with neutral pH (≈7.3). Higher pyrolysis temperatures (≥550 °C), on the other hand, promote the formation of PCMs with alkaline pH, regardless of the sludge pH. A pH increase with increasing pyrolysis temperature is typically observed for chars derived from sewage sludge and other feedstocks. The phenomenon can be significant: chars produced from sludge at neutral pH, at high temperatures (up to 700 °C), were characterized by pHs ranging from 12.0 to 13.0 [22]. Such increases could be due to polymerization/condensation reactions of aliphatic compounds, dehydration associated with decrease of acidic surface groups during thermal treatment, and the concentration of inorganic constituents in the char resulting from the separation of metal salts from the organic matrix at increasing temperatures [24].
3.3. Carbon and Inorganic Constitutents
The carbon percentage in sewage sludges investigated by literature is summarized in Table 2, ranging from 21.6% to 33.2%. Studies show a reduction of C content relative to the original feedstock, due to increased volatility during the process.
Both sewage sludges and PCMs analysed in Table 2 show high ash content, ranging from 55.8 to 61.3% in sewage sludges and from 64.1 to 79.1% in chars, showing an increase in ash content, compared to the original feedstock, with increasing pyrolysis temperature. Such increase is a typical tendency for sewage sludge (and other feedstock) chars, due to the concentration of the non-volatile mineral constituents that form the ash, and the removal of volatile organic decomposition products. Mineral fractions are, in fact, dominant in both untreated sewage sludges and chars, usually much higher than in (bio)chars derived from other materials. This seems to be the result of the complexity of sewage sludge, and the diversity of its components. Sewage sludges, in fact, usually contain high concentrations of silica (19–58% in sewage sludge before pyrolysis). It is difficult to exactly predict ash content of chars based on the mineral components content of original sewage sludge, although an increase in process temperature will cause an increase in ash percentage from pyrolyzed sewage sludge [25].
3.4. Phosphorus and Nitrogen
The initial P content of sewage sludge depends on the wastewater treatment processes adopted, and is further concentrated (by about 40–100%) when pyrolysed at temperatures above 600 °C, indicating that phosphorus is associated with the inorganic fraction of the sludge. On the other hand, the N content of the sludge is decreased in the PCM when process temperature is increased, due to volatilisation and loss of the NH4-N and NO3-N fractions, as well as that of volatile matter containing N groups.
3.5. Porosity
Sewage sludges are practically nonporous. As a result of pyrolysis, a material with a more developed surface texture is obtained. Increase in surface area can vary widely, from 6 to almost 40 times [22], although it is difficult to define a straightforward relationship between specific surface area of the initial sewage sludges and surface area of the PCM produced. Increase of surface area with increasing temperature up to 600 °C and decrease at higher temperatures were observed by Zelinska et al. [22] and Lu et al. [26].
Kinetic sorption mechanisms onto PCMs from aqueous solutions were investigated by several researchers on samples obtained from traditional sludge pyrolysis. Char exhibited a significant ability in adsorbing Cr(III), with removal at equilibrium of approximately 70%, while it was not as effective in removing As(V), for which maximum removal capacity (at equilibrium) was approximately 30%. The high differences observed in metal removal could be attributed to favorable electrostatic interactions between char negative surface charge and metal cations, contrary to anions [8]. In terms of phosphorus adsorption, (bio)char exhibits a similar capacity to biosolids, about 15 mg/g, which is almost seven times higher than activated carbon.
3.6. Metals in Char
The composition of sewage sludge is naturally varied and heterogeneous, however, analysis of total heavy metals concentrations in samples from different WWTP feedstocks has been found to be surprisingly consistent (Table 3). Organic wastes, like sewage sludge, are known to generally contain high concentrations of metals which increasingly accumulate in the solid residue with increasing pyrolytic temperature. Data demonstrates that metal concentrations are usually less than regulatory limits stipulated in the European Union Council Directive 86/278/EEC (The Sewage Sludge Directive). Some metals, however (e.g., Zn and Cu), exceed Chinese GB4284-84 standard, which is more stringent than those of the Sewage Sludge Directive 86/278/EEC. In addition to concentration, stability of heavy metals also increases with pyrolysis process temperature, therefore greatly reducing their leaching potential as indicated in Table 4.

Table 3.
Main ash components for some reported biomass feedstocks.

Table 4.
Total and extractable (DTPA) heavy metals in sludges.
Diethyl–enetriamine–pentaacetic acid (DTPA) extractable fraction has been widely used to estimate bioavailability of metals in soils and sludge due to its capacity to chelate a wide range of metallic elements [27]. Many researchers have concluded that heavy metals are practically immobile in char and that the pyrolysis process itself is effective to reduce their potential release.
3.7. Advantages of PCM vs. Dewatered Sludge Spreading
In biologic wastewater treatment processes, sorption to biosolids is one of the primary removal pathways for many hydrophobic, and especially persistent, bioaccumulative toxic organic chemicals, including pharmaceutical and endocrine-disrupting compounds [28] and metals [29]. Even volatile organics, such as benzene, are commonly found in sewage sludges as a result of sorption to the sludge matrix. Land application of sewage sludge was widely used in the past (and still is in some countries) in view of its soil conditioning properties. Macronutrients in sewage sludge could serve as a source of plant nutrients, and organic constituents could provide beneficial soil additives. Its heterogeneous nature and variability, however, require knowledge of its composition prior to such applications. Industrial and non-domestic effluents may also cause the sludge to contain many toxic compounds in addition to organic material. Uninformed sludge soil amendment may disturb soil properties, especially when containing high concentrations of metals and toxic constituents, which may accumulate in the long term and could become highly persistent in soil and be forebearers of potential problems, including transfer into the food chain, with possible serious risks to human health. Much of the existing literature on organic contaminants focused on once predominant chemicals such as PCBs, chlorinated pesticides, and chlorophenols, whose importance as inputs to sewage collection systems, however, has declined in the last decade. The biggest scientific interest relating to pollutant concentrations in sewage sludge currently includes endocrine-disrupting substances and other common-use potential toxicants, such as linear alkyl benzenesulfonates.
Very few countries have rules limiting the concentration of organic chemicals in sewage sludges. While accumulation of heavy metals in plants and their effects has been assessed in sludge-amended soil, no extensive evaluation has been conducted for xenobiotic organics (XOCs). Giger et al. [30] investigated the occurrence and fate of antibiotics in wastewaters and sewage sludge in Switzerland. Mass balance studies on antibiotics for human use show that, while wastewater treatment as a whole resulted in a reduction of compounds mass flow of about 90%, approximately 84% was due to sorption on sewage sludge without further significant removal under methanogenic conditions in sludge digesters [31]. Jelic et al. [32] observed the presence of 32 target pharmaceutical compounds in three WWTPs influents, and 29 in their effluents, in concentrations ranging from low ng/L to a few mg/L. An analysis of sludge samples showed that 21 pharmaceuticals accumulated in sewage sludge from all three WWTPs in concentrations up to 100 ng/g. This indicates that even good removal rates obtained in the aqueous phase (i.e., determined by comparison of influent and effluent wastewater concentrations) do not actually imply degradation of the compounds to the same extent [33]. The process of PCM production, by destroying most of the organic contaminants contained in the sludge, is by itself capable of eliminating such risks.
4. Beneficial Uses of PCMs: Current and Potential Applications
In the introductory section, a brief review of the existing methods of excess sewage sludge disposal was presented. This section analyses in greater detail the beneficial uses by which char can be disposed of, since the reuse pathways for bio-oils and syngas are obvious enough. The most appealing feature of char is the fact that it is an inexpensive, sustainable, and easily-produced material with potentially extensive applications, with a much lower cost than materials from petrochemical or other chemical processes. In fact, PCM from sewage sludge (a waste material) should be available at a cost very close to the cost of simple wastewater sludge disposal under current technologies. Even though most applications are still in infancy, PCM has already a number of identified applications with potentially extraordinary effects, including soil amendment, process catalysis, water purification, and many others, existing or still to be invented. As mentioned in the introductory section, (bio)char is the final solid residue of a pyrolysis process. Due to the elimination of the embedded humidity, it has a higher energy content than the original feedstock, therefore, similarly to sludge, it could be burned in systems fed with pulverized coal, with similar emissions issues, requiring similar pollution abatement technologies. In reality, chars have many other much more attractive, high value uses that will be herein analysed.
4.1. Agricultural Uses
The beneficial effects of (bio)char produced from various feedstocks on agricultural crop yield and properties of soil have been studied, with findings that show it significantly improves the yield of some crops [34]. (Bio)char addition is also known to improve nitrogen fertilisers’ efficiency by improving the chemical properties of soil. Wastewater sludge PCM application was specifically found to increase soil cation exchange capacity (CEC) by up to 40% and soil pH by up to one pH unit [35], improving nutrients’ plant availability and carbon sequestration. It should be noted that PCM itself will not contribute meaningful amounts of nutrients in a short period given its high stability (it could do so, however, in the long term), but will make those already present more available due to its chemical characteristics.
Heavy metals accumulation, particularly As, Cd, Cr, Cu, Pb, Ni, Se, and Zn, is of specific concern as far as agricultural activities are concerned, and is also one of the principal reasons for the existing limitations on continuing sludge agricultural disposal practices. Hossain et al. [35] identified 16 metals and trace elements in the char applied to a cherry tomato farm at a dose of 10 t/ha, which was showing poor soil properties due to low nutrient availability. Produce analysis showed that all elements present in char were uptaken in insignificant amounts: Se, Pb, and Sn in the produce were all below detection limits, as well as Cr and As. Cu and Zn showed the lowest bioavailability and also that of other trace elements was very low, while the improvement of crop production increased by 64%. The study confirmed that char improves also the availability of P, total N, and other major cations, and has a positive liming effect when applied to low pH soils, increasing it while enhancing nutrient use efficiency. In addition, char had positive effects on soil quality, enhancing soil aeration, increasing water holding capacity, and improving environmental conditions for the growth and development of plant root systems, significantly increasing soil C. Although char may increase total N uptake by crops, this does not imply that lesser amounts of N fertilizers may be needed, as char-bound N is not directly available to plants but is fused in its carbon matrix. Evidence nonetheless showed that soil application of (bio)char often positively affects crop productivity and is beneficial in many situations. Therefore, it may be also considered a potential substitute for agricultural lime, especially in regions with acidic soils [36].
4.2. Remediation and Restoration of Contaminated Soils
Regulators are now concerned with anthropogenic contaminants bioavailability and mobility effects on environmental risk, rather than just their total concentration. (Bio)char has shown great potential for managing waste streams originated by animals or plants while decreasing their associated pollution loading to the environment. Soils amendment is a long-standing remediation procedure, aimed at reducing the risk of pollutants transfer to ground or surface waters, or receptor organisms. (Bio)chars have demonstrated potential for reducing the diffusion of a variety of organic and inorganic contaminants present in soils in mobile forms; from the study of environmental pollutants’ fate, in fact, it is known that by increasing the organic fraction content of soil, its pollutants adsorption properties are increased, as is its capacity to reduce their bioavailability. Carbon-rich amendments, such as activated carbon, have in fact been employed for soil and sediment remediation purposes for some time due to their ability to reduce contaminant bioavailability and risk [37].
In recent literature, the benefits associated with applying (bio)chars to soils, such as their conditioning properties, have been described [38]. These materials are capable to complex metal ions surfaces into the soil, reducing their bioavailability (Figure 2). Furthermore, it was shown earlier that increasing the pyrolysis temperature of chars increases their degree of carbonisation and surface area, and reduces the abundance of amorphous organic matter, increasing their capability to adsorb organic contaminants. Uptake of pesticides by crops is also reduced [39]. Chen et al. [40] compared (bio)char surface areas obtained at different pyrolysis temperatures and found that those pyrolysed at 700 °C had half the surface area compared to activated carbon; this indicates that (bio)chars obtained at high temperatures (or “activated”) will have a high organic contaminant remediation potential. In a study comparing activated carbon to low-temperature-processed (bio)chars, the latter was shown to linearly absorb atrazine [41], suggesting possible advantages in the remediation of soils with organic and inorganic contaminants competing for sorption sites. Sewage sludge biochar seems to be more or less effective for the absorption of most of the contaminants for which Activated Carbon has traditionally been used, namely heavy metals, organic pollutants, pesticides, and others. A careful consideration of the role of nanoporosity, hydrophobicity and surface characteristics, the importance of black carbon in sorption, and pollutants sequestration should be taken into account to investigate adequate adsorption kinetic models to biochar from different sources and production processes [42,43,44].
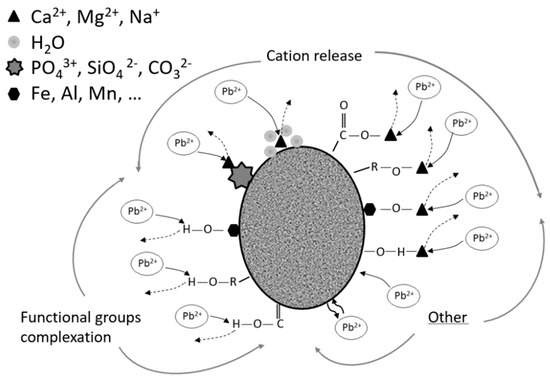
Figure 2.
Conceptualization of Pb adsorption mechanisms by biochar granules.
Care must be taken as char can have different effects on the mobility of metals in soils, compared to that in water. In a study where char was applied to multi-element (As, Cu, Cd, and Zn) contaminated soil, Cd and Zn were immobilized, as expected, in soils amended with char, however, Cu and As were mobilized. This phenomenon was associated with the soil’s increased pH resulting from the application [45]. Mobility of As can also be increased as biochar soil amendments can cause reduction of As(V) to As(III) (more environmentally mobile) [46].
Currently, little information is available concerning char applications in the treatment of contaminated sediment, however, considering biochar’s excellent adsorption capabilities for many pollutants in solution, all the elements suggest that char could be used as a new, in situ potential amendment sorbent for remediation and management of contaminated sediment sites, with advantages in both carbon sequestration and sediment remediation opportunities.
4.3. Water and Wastewater Treatment
The specific properties of char, including large specific surface area, porous structure, enriched surface functional groups, and mineral components, make it possible to use this material as a proper adsorbent to remove pollutants from aqueous solutions. Compared to activated carbon, char can be considered a potential low-cost and effective new adsorbent. Activated carbon is treated (activated) with oxygen to increase its microporosity and surface area, and is the most commonly used carbonaceous sorbent. Its production needs high temperatures and the additional activation processes, therefore, comparatively, the production of (bio)char is cheaper since it has lower initial energy requirements; in addition, feedstocks are abundant and low cost, especially when char is obtained from urban sludge. Studies report that PCM showed excellent ability to remove contaminants such as heavy metals and organic and other pollutants from aqueous solutions, and some even exhibit comparable or better adsorption properties than commercial activated carbon [47].
In available literature about water treatment applications, 46% of the studies concern the (bio)char removal ability of heavy metals, 39% of organic pollutants, 13% of fertilizers N&P, and 2% of other pollutants. Both Langmuir’s and Freundlich’s isotherm models fit well with data to describe heavy metals equilibrium adsorption by (bio)chars. Since these also show high affinity for organic pollutants (dyes, pesticides, herbicides, antibiotics, and others), a perfect fit of the experimental data with Langmuir’s or Freundlich’s models was also demonstrated. Ahmad et al. [48] covered an extended overview of (bio)char use as sorbent for contaminant management in soil and water. Adsorption efficiency is influenced by its properties, dosage, competitive anions, solution temperature, and pH; as (bio)char surfaces are negatively charged, they also facilitate electrostatic attraction of cationic organic compounds. Dosage has significant influence on adsorption efficiency [49,50], hence applying an optimum (bio)char-to-contaminant ratio is essential for cost-effective application. Chen et al. [51] reported that increased (bio)char concentration decreases adsorption effectiveness, although the increase of adsorbent concentration may result in an increased total removal efficiency, due also to the increase of active site availability. Adsorption of contaminants onto (bio)chars appears to be an endothermic process, with capacity increasing at increasing temperatures: increase of ΔG0 absolute value with temperature suggests that adsorption is more favorable at higher temperatures [52].
Co-existence and interaction of pollutants’ ions have significant influence on equilibrium adsorption capacity, especially for applications of (bio)char in real water systems. Studies on its adsorption capacity for co-existing contaminants (e.g., atrazine and simazine, phenanthrene and Hg(II)) showed that adsorption of the two contaminants decreased when they co-existed in solution [53]. Presence of heavy metals had different effects on oxytetracycline adsorption onto biochar, ranging from insignificant (Cd2+), slight facilitation (Zn2+), slight inhibition (Pb2+), and facilitation (Cu2+), regardless of solution pH value [54].
A solution’s pH is one of the most relevant parameters for the adsorption process, and depends on (bio)char type and target contaminants, affecting not only adsorbent surface charge, but also the degree of speciation of adsorbate. Surface functional groups (e.g., carboxylate, -COOH; and hydroxyl, -OH), whose behavior changes with solution pH, are present on (bio)char: at low pH most of these groups are positively charged, favoring anion adsorption. (Bio)char surface, negatively charged in the higher pH range, when pH > pH0 (point of zero charge), on the other hand, allows cations to be easily captured.
All published adsorption studies so far were conducted on simulated (artificial) wastewater, in order to simplify the underlying hypotheses. This is a normal procedure which, however, presents wide gaps with actual situations. Applications of biochar for real wastewater treatment are still unpublished, probably due to the complex pollutants-and-ions combinations that co-exist in such systems, which could have significant influence on the equilibrium adsorption capacity.
Since application of (bio)char for removal of pollutants from aqueous solutions is mainly dealing with toxic pollutants (organic or inorganic), the final disposal of the spent material becomes an important issue. (Bio)char loaded with ammonium, nitrate, and phosphate, and without toxic pollutants, can be used as slow-release fertilizer to enhance soil fertility; however, if used to adsorb toxic pollutants, it needs appropriate handling, following standards of hazardous wastes treatment. (Bio)char desorption/regeneration properties have been investigated to determine the feasibility of economical reuse: results indicate that food-waste biochar could be used repeatedly without significant loss of total adsorption capacity, however, a wide availability of waste biomass for its production, and increasingly limited use cycles, may render its recovery process economically un-effective. At the present time, little information is available about disposal of spent (bio)char: its stability, risks of secondary pollution, effect on carbon sequestration, and economical feasibility are still unclear and require further investigation [55]. It is also assumed that it could be used for combustion, upon verification that adsorbed compounds are destroyed or immobilized in the ashes.
4.4. Soil Properties Amelioration
PCM from urban sludges has been proven to be a strong adsorbent; when spread into soils it not only increases their capacity to better adsorb plant nutrients and agricultural chemicals, reducing leaching potential of those chemicals into surface and ground water, but it itself also contains significant quantities of carbon and plant nutrients that are slowly released to growing plants. Last, it has a relatively low density that helps improve soils’ properties, lowers bulk density, improves drainage, aeration, and root penetration properties of clay soils, and increases the water and nutrient retention ability of sandy ones by increasing their carbon content. The following list summarizes some of the positive effects of PCM soil amelioration:
Enhances soil properties and plant growth. PCM could raise and sustain crop yields and improve problematic nutrient-poor soils, including acidic tropical, humid, and dry environment soils. Having nutrient affinity, it can retain plant nutrients, notably N, in permeable soils under rainy conditions. Reducing soil acidity raises its pH and improves productivity of many crops. Supporting nitrogen fixation, it can also reduce the need for fertilizers, reducing dependency of farmers on suppliers. It increases soil cation exchange capacity and enhances moisture retention, reducing irrigation demand and making crops more resilient to droughts. It can increase soil microbial biomass, supporting beneficial organisms like earthworms and arbuscular mycorrhizal fungi in soil.
Helps reduce agrochemical pollution. PCM may bind agrochemicals and help reduce phosphate and nitrate pollution of streams and groundwater, resolving major problems (e.g., eutrophication, nitrates contamination) that hinder intensive agriculture. As a consequence, it could help reduce pressures for new forest clearances (biodiversity conservation benefits). It can also reduce plant uptake of pesticides from contaminated soils, which is a form of bioremediation. In periurban/urban agriculture, PCM may counter harmful compounds like heavy metals, dioxins, and PAHs (polycyclic aromatic hydrocarbons) present in raw sewage or refuse inputs. It can also reduce aluminum toxicity.
Helps compensate for GHG emissions associated with agricultural development. PCM stores carbon in the soil for a long time, while compost and manures are subject to rapid microbial breakdown. Sequestration in char is instead likely to last for centuries, possibly even thousands of years. It could suppress methane and N2O (nitrous oxide gas) emissions from cultivated soil. A laboratory study in Japan found that soils amended with 10 wt % PCM suppressed 89% of previous N2O emissions [56].
Combats climate change. PCM applied to soils offers effective, long-term carbon storage. It can increase adaptability to environmental change by improving soil moisture retention, increasing agricultural resilience against climatic change effects like increased droughts and floods. Supporting biofuel production (a by-product of the process), its carbon footprint may even be completely carbon neutral.
4.5. Carbon Sequestration by Biochar
(Bio)char is emerging as not only as an ameliorant to reduce bioavailability of contaminants with additional benefits of soil fertilization, but also as a possible mitigation agent of climate change, in the sense that it can act as an efficient and economic carbon sequestration means. (Bio)char contains a considerable fraction (roughly 1/4 to 1/3) of the carbon initially contained in sewage sludge (or in any other feedstock for its production). It has been shown that the half-life of C in soils is in excess of 1000 yr [57], indicating that soil-applied (bio)char will make not only a lasting contribution to soil properties and quality, but also that carbon it contains will be removed from the atmosphere and sequestered in the soil for millennia, at a lower cost than some of the Carbon Capture and Storage (CCS) technologies commonly used today (i.e., storage in geologic traps). It has been estimated that a US-wide system of pyrolyzers for processing biomass into bio-oil and charcoal, replacing fossil fuels with bio-oil and returning (bio)char to soils, could reduce that nation’s demand for fossil oil by 25% and its carbon emissions by 10%, all the while strengthening its rural economy. If extrapolated to the global scale, such a strategy could make a major contribution to world energy supply and be a solution to global warming.
It should be noted that, while substitution of fossil fuels with crop-derived bio-oils could raise some ethical and sustainability issues (i.e., food crops use competition), waste sludge is by all means an available and obligatorily disposed of by-product of wastewater treatment, and that may constitute itself, if not properly handled, an environmental problem. As indicated in prior sections of this paper, processing of urban sludge with pyrolysis not only allows recovery of valuable products, but also improves the characteristics of environmental compatibility of the final resulting solid fraction.
4.6. (Bio)char as a Secondary Prime Material for Activated Carbon Production
Activated carbon is produced according to two main steps: (a) carbonization of organic material, such as agricultural residues, to generate char in inert or oxygen-limited atmosphere, and (b) char activation by chemical or physical means, at temperatures usually ranging between 600 °C and 1200 °C. Physical activation occurs in the presence of an oxidizing atmosphere (a mix of CO2, steam and air) and does not involve use of chemicals. Chemical activation, on the other hand, uses chemicals as activating agents in one-step or two-step activation. The most common agents are ZnCl2, KOH, H3PO4, and K2CO3. The former offers an advantage over the latter due to the lack of chemicals involved, however, two-step chemical activation could produce a highly microporous, high specific surface activated carbon with better properties. Tests on activating biochar from wood biomass for use as activated carbon substitute have met with positive results [58]. Although no such experiments have been performed so far on sewage-sludge-originated PCM, the possibilities for utilization of (bio)char from different combinations of biomasses are yet to be exhaustively investigated. In this context, recently ascertained applications of (bio)char from other sources are summarized in the following paragraphs, just to illustrate some of the multitude of innovative applications that these materials could have in the near future.
4.7. (Bio)char for Catalyst Production
Syngas from gasification or pyrolysis of biomasses contain non-negligible amounts of tars that are detrimental to downstream uses with high value, requiring subsequent treatment, such as water/oil scrubbing and thermal (typically at T > 1000 °C) and catalytic cracking. The latter is considered the most promising technology for syngas cleaning, requiring low temperatures (<700 °C) and energy to achieve high tar removal (>90%) by using appropriate catalysts. Marin [59] showed that (bio)char can be used as a gas catalyst for this purpose, with and without active metal loading.
(Bio)char can also work as a catalyst for conversion of syngas into liquefied hydrocarbons by Fischer–Tropsch synthesis as shown by Yan et al. [60], and be a precursor for producing heterogeneous acid catalysts for esterification or transesterification for biodiesel production from vegetable oils and/or animal fats [61]. (Bio)char-based catalysts from various lignin biomasses were tested to this end by Kastner et al. [62], achieving 90–100% conversion within 30–60 min, with a high degree of reusability.
4.8. (Bio)char as Gas Adsorbent
CO2 capture and storage is a promising strategy to reduce GHG-CO2 emissions, whose main challenges are high flow rates and low partial pressure of CO2 in flue gases. For effective capture and removal of the CO2, high selectivity and adsorption capacity of absorbents are required, in addition to their durability in time, low cost, and ease of regeneration. (Bio)char-based activated carbons have shown adsorption capacity similar to the highest reported for other carbon materials [63].
Deployment of hydrogen-based fuel technologies as an industry standard is seriously hindered by the difficulty in safely storing this gas, which is considered a promising and clean energy carrier, with ample potential affects on future transportation industry development worldwide. Zhang et al. [64] tested activated new carbon material based on biochar from corncobs, using KOH for its chemical activation, obtaining a high surface area (3500 m2/g) together with large pores volume (1.3–1.94 cm3/g), that could adsorb hydrogen economically. The small pore activated carbon exhibited the highest hydrogen uptake capacity so far with >2.85 wt % at 1.0 bar and 196 °C.
4.9. (Bio)char in Fuel Cell Systems
The direct conversion of molten, carbonaceous solid fuel into electricity, without the need of pre- conversion into gaseous form by direct carbon fuel cell (DCFC) was recently achieved [65]. Results showed the feasibility of using (bio)char as a low-cost, renewable fuel for DCFCs notwithstanding its relatively low carbon and high ash contents, achieving fuel cell power density of about 60–70% compared to coal-based fuel cells. In a comparison of carbon fuels for DCFCs (commercial graphite, carbon black, two types of commercial coal, five biochar types) on cell performance, commercially available (bio)char achieved the second highest generation (64.2 mA/cm2) and power densities (32.8 mW/cm2) [66].
(Bio)char has also been tested as a low-cost substitute anode material in microbial fuel cells (MFCs). This technology is capable of simultaneously removing organic matter from wastewater and soil, with direct generation of electricity [67,68], that can be used for other environmental purposes, such as groundwater nitrates decontamination [69,70]. MFC electrode materials are normally granules of graphite or activated carbon, which cost, on the average, from $500 to $2500 per ton, making their construction cost prohibitive at the large scale. (Bio)char was shown to be an alternative, promising material for MFC construction. By using new wood-based biochar for electrodes, cost and power output of tested systems were comparable to those made with activated carbon and graphite electrodes. Power output of biochar systems (532–457 mW/m2) was slightly lower than that of activated carbon (674 mW/m2) and graphite (566 mW/m2) systems, at a specific cost f material that was about 90% lower than the others (biochar $17–$35/W, activated carbon $402/W, graphite $392/W) [71].
Finally, PCM was also used as catalyst in MFCs with a carbon cloth air cathode and char catalytic layer coating on both sides of the wet-proofed membrane. The catalytic layer made of sewage-sludge-derived PCM was compared with a hugely more expensive Pt/C layer. Power density of the char-coated cathode reached 500 mW/m2, comparable to that of Pt/C-coated cathode. This showed that sewage sludge char was active in catalyzing redox reactions in MFCs, and could become an alternative to more expensive Pt catalysts, with even better stability than the latter [72].
4.10. (Bio)char-Based Supercapacitors
Supercapacitors are energy storage devices indispensable to store energy from renewable sources, thanks to high-power densities, long lifecycle, and quick charge/discharge capabilities. They can store 10 to 100 times more energy per unit volume than other capacitor types, and can accept and deliver charge much faster than batteries, tolerating many more charge/discharge cycles than rechargeable batteries. They can be used as uninterruptible power sources in electric vehicles like cars, buses, trains, and elevators, and can be used for regenerative braking, short-term energy storage, or burst-mode power delivery. The electrodes’ microstructure of these devices has a great influence on their performance.
The preferred raw materials for making supercapacitors is carbon material with high specific surface area and porous structure, due to its wide availability, relatively low cost, and environmental impacts. Recently, biochar from different biomass feedstocks (paper cardboard and woody biomass) was used for supercapacitor fabrication, indicating that its use of biochar is promising thanks to low cost and satisfactory performance. Supercapacitor electrodes from biochar materials showed potential of about 1.3 V and fast charging–discharging behavior with gravimetric capacitance of about 14 F/g, that could be increased by activating the biochar with nitric acid to115 F/g [72].
5. Discussion
Sewage sludge is a diverse material in terms of physico-chemical properties, depending on its origin. It is therefore a huge simplification to try to generalize conclusions on this product based on results from only one or a few types of sludge, since this may lead to wrong assumptions. Although there is some literature concerning the evaluation of chars produced from different sewage sludge of varying properties, with different processes and at different temperatures, systematic methodologies on practical investigation of physical and chemical properties of chars still do not exist, mainly because of a poor focus towards a myriad of possible final uses. Relating how the initial properties of a generic sludge (and how the other variables, such as process and process conditions, influence them) determine the properties of the char produced is of key importance in a moment when interest in sewage-sludge-originated char is becoming relevant.
(Bio)chars could virtually be a panacea for innumerable environmental and industrial uses; for example, activated char could replace activated carbon in most of its current applications, as it has equivalent or even greater sorption efficiency for various contaminants due to its cost-effective production from waste resources, that makes it less expensive compared to activated carbon. In fact, the estimated break-even price for (bio)char is US $246/ton, which is approximately 1/6 of commercially available activated carbon cost (US $1500/t) [73].
As discussed above, (bio)char has been applied to the most varied applications, with others that undoubtedly will be proposed in the near future. A thorough understanding of (bio)char properties will be critical in mitigating its possible undesired impacts while harnessing its benefits. Research on in-depth characterization of its properties and relationships to reaction conditions in its production are critical to optimizing and tailoring properties for maximum effectiveness in any application [74].
In catalysis uses, (bio)char has potential roles in different applications, such as syngas reforming and conditioning, and bio-oils upgrading; its use will increase net sustainability of bioenergy refinery systems by reducing the need for external materials. Use in fuel cells and supercapacitors also suggests economic and environmental benefits, however, properties of (bio)char-based, novel functional materials depend highly on biomass precursors, and have still space for substantial improvement. For example, capacitance of Co3O4 nanotube-based supercapacitors can reach 500 F/g while the maximum of (bio)char-based ones is only 250 F/g, so far.
Recently, the concept of engineered (bio)chars has been postulated [75], its key aspect being similar but more advanced to that of designer (bio)char, where this would be produced by a controlled process with specific properties for particular purposes, ranging from carbon sequestration to soil fertility improvement, or waste management and pollution control, as previously illustrated. Engineered (bio)char is a term to indicate application-oriented char modification involving various possible methods such as acid/base treatment, amination, surfactant modification, impregnation of mineral sorbents, steam activation, and magnetic modification [76]. (Bio)char-based nanocomposites with improved physicochemical and sorptive properties have been manufactured and used for environmental applications including contaminant removal and reclamation of polluted sites [77]; engineered (bio)chars, coated with graphene, have been found to have excellent reversible discharge capacity, and are thus suitable for use as anode material for making batteries [78]. A paradigm shift towards the development of “next generation” (bio)chars is expected, with applications in fields including but not limited to industrial manufacturing of electronics, sensors, smart building materials, healthcare products, and renewable energy, where feedstock combination could help achieve innovative products.
6. Conclusions
Sewage sludge pyrolysis-generated char may be an additional, important resource for reuse in agricultural and many other environmental or industrial applications, and its additional contribution to the selection of a final strategy for wastewater sludge disposal must be carefully evaluated.
Feedstock used for (bio)char production, as well as the production process itself, influences these materials characteristics. Optimizing (designing) (bio)char for a specific end-use may require feedstock selection as well as specific pyrolysis production techniques and conditions. Several studies are starting to address these relationships. Literature has shown that pyrolysis process parameters (temperature, residence time, heating rate, and feedstock particle size) affect quality and quantity of the produced (bio)char, and thus its environmental behavior. Among these, pyrolysis temperature has the largest effect on final product quality, as its increase decreases its yield and N content, increasing at the same time its pH, specific surface area, carbon content, available nutrients, and heavy metal stability. (Bio)char as a product of pyrolysis of biomass can be produced inexpensively, more so in the case of sludge pyrolysis, which, being a waste that is expensive to dispose of, could be had at close to no cost.
Increased sewage sludge char exploitation can provide new incentives for agriculture improvement and investment development, offer farmers additional sources of income, help in carbon sequestration, increase crop and land yield and productivity, improve sustainable land use in agriculture, and provide new incentives for municipal solid waste (MSW) and sludge treatment research and development as well.
This paper discussed initially how sewage sludge char characteristics are developed during its production process, then existing and prospective environmental and technological uses of this by-product. The use of waste biomass for (bio)char production is not only economical but also highly beneficial. Initially, expected benefits mainly included soil improvements, energy production, and climate change mitigation, however, as research on potential uses picks up momentum, their range seems to expand exponentially. Despite this justified optimism, however, a number of research gaps and uncertainties still exist, as partly identified in the previous discussion, that need more specific investigations.
Author Contributions
The Authors declare that they have equally contributed to the manuscript
Conflicts of Interest
The authors declare no conflict of interest.
References
- Liu, B.; Wei, Q.; Zhang, B.; Bi, J. Life cycle GHG emissions of sewage sludge treatment and disposal options in Tai Lake watershed, China. Sci. Total Environ. 2013, 447, 361–369. [Google Scholar] [CrossRef] [PubMed]
- Kelessidis, A.; Stasinakis, A.S. Comparative study of the methods used for treatment and final disposal of sewage sludge in European countries. Waste Manag. 2012, 32, 1186–1195. [Google Scholar] [CrossRef] [PubMed]
- Capodaglio, A.G.; Callegari, A.; Lopez, M.V. European Framework for the Diffusion of Biogas Uses: Emerging Technologies, Acceptance, Incentive Strategies, and Institutional-Regulatory Support. Sustainability 2016, 8, 298. [Google Scholar] [CrossRef]
- Menendez, J.A.; Inguanzo, M.; Pis, J.J. Microwave-induced pyrolysis of sewage sludge. Water Res. 2002, 36, 3261–3264. [Google Scholar] [CrossRef]
- Capodaglio, A.G.; Callegari, A.; Dondi, D. Microwave-induced pyrolysis for production of sustainable biodiesel from waste sludges. Waste Biomass Valoriz. 2016, 7, 703–709. [Google Scholar] [CrossRef]
- Shackley, S.; Carter, S.; Knowles, T.; Middelink, E.; Haefele, S.; Sohi, S.; Cross, A.; Haszeldine, S. Sustainable gasification-biochar systems? A case-study of rice-husk gasification in Cambodia, Part 1: Context, chemical properties, environmental and health and safety issues. Energy Policy 2012, 42, 49–58. [Google Scholar] [CrossRef]
- European Biochar Foundation (EBC). European Biochar Certificate—Guidelines for a Sustainable Production of Biochar; European Biochar Foundation (EBC): Arbaz, Switzerland, 2012; Available online: http://www.europeanbiochar.org/en/download (accessed on 14 August 2017).
- Agrafioti, E.; Bouras, G.; Kalderis, D.; Diamadopoulos, E. Biochar production by sewage sludge pyrolysis. J. Anal. Appl. Pyrolysis 2013, 101, 72–78. [Google Scholar] [CrossRef]
- Motasemi, F.; Afzal, M.T. A review on the microwave-assisted pyrolysis technique. Renew. Sustain. Energy Rev. 2013, 28, 317–330. [Google Scholar] [CrossRef]
- Zhang, Y.; Chen, P.; Liu, S.; Peng, P.; Min, M.; Cheng, Y.; Anderson, E.; Zhou, N.; Fan, L.; Liu, C.; et al. Effects of feedstock characteristics on microwave-assisted pyrolysis—A review. Bioresour. Technol. 2017, 230, 143–151. [Google Scholar] [CrossRef] [PubMed]
- Sanchez, M.E.; Menéndez, J.A.; Dominguez, A. Effect of pyrolysis temperature on the composition of the oils obtained from sewage sludge. Biomass Bioenergy 2009, 33, 933–940. [Google Scholar] [CrossRef]
- Raboni, M.; Viotti, P.; Capodaglio, A.G. A comprehensive analysis of the current and future role of biofuels for transport in the European Union (EU). Ambiente & Água—An Interdisciplinary. J. Appl. Sci. 2015, 10, 9–21. [Google Scholar] [CrossRef]
- Budarin, V.L.; Shuttleworth, P.L.; De bruyn, M.; Farmer, T.J.; Gronnow, M.J.; Pfaltzgraff, L.; Macquarrie, D.J.; Clark, J.H. The potential of microwave technology for the recovery, synthesis and manufacturing of chemicals from bio-wastes. Catal. Today 2015, 239, 80–89. [Google Scholar] [CrossRef]
- Masek, O.; Budarin, V.; Gronnow, M.; Crombie, W.; Brownsort, P.; Fitzpatrick, E.; Hurst, P. Microwave and slow pyrolysis biochar—Comparison of physical and functional properties. J. Anal. Appl. Pyrolysis 2013, 100, 41–48. [Google Scholar] [CrossRef]
- Brodie, G.; Destefani, R.; Schneider, P.A.; Airey, L.; Jacob, M.V. Dielectric Properties of Sewage Biosolids: Measurement and Modeling. J. Microw. Power Electromagn. Energy 2014, 48, 147–157. [Google Scholar] [CrossRef]
- Tian, Y.; Zuo, W.; Ren, Z.; Chen, D. Estimation of a novel method to produce bio-oil from sewage sludge by microwave pyrolysis with the consideration of efficiency and safety. Bioresour. Technol. 2011, 102, 2053–2061. [Google Scholar] [CrossRef] [PubMed]
- Lin, Q.; Chen, G.; Liu, Y. Scale-up of microwave heating process for the production of bio-oil from sewage sludge. J. Anal. Appl. Pyrolysis 2012, 94, 114–119. [Google Scholar] [CrossRef]
- Dominguez, A.; Menéndez, J.A.; Inguanzo, M.; Bernad, P.L.; Pis, J.J. Gas chromatographic–mass spectrometric study of the oil fractions produced by microwave-assisted pyrolysis of different sewage sludges. J. Chromatogr. A 2003, 1012, 193–206. [Google Scholar] [CrossRef]
- Dominguez, A.; Menendez, J.A.; Inguanzo, M.; Pís, J.J. Production of bio-fuels by high temperature pyrolysis of sewage sludge using conventional and micro-wave heating. Bioresour. Technol. 2006, 97, 1185–1193. [Google Scholar] [CrossRef] [PubMed]
- Antunes, E.; Schumann, J.; Brodie, G.; Jacob, M.V.; Schneider, P.A. Biochar produced from biosolids using a single-mode microwave: Characterisation and its potential for phosphorus removal. J. Environ. Manag. 2017, 196, 119–126. [Google Scholar] [CrossRef] [PubMed]
- Liu, T.; Liu, B.; Zhang, W. Nutrients and Heavy Metals in Biochar Produced by Sewage Sludge Pyrolysis: Its Application in Soil Amendment. Pol. J. Environ. Stud. 2014, 23, 271–275. [Google Scholar]
- Zielinska, A.; Oleszczuk, P.; Charmas, B.; Skubiszewska-Zieba, J.; Pasieczna-Patkowska, Z. Effect of sewage sludge properties on the biochar characteristic. J. Anal. Appl. Pyrolysis. 2015, 112, 201–213. [Google Scholar] [CrossRef]
- Capodaglio, A.G.; Callegari, A. Feedstock and process influence on biodiesel produced from waste sewage sludge. J. Environ. Manag. 2017, in press. [Google Scholar] [CrossRef] [PubMed]
- Gascó, G.; Blanco, C.G.; Guerrero, F.; Méndez Lázaro, A.M. The influence of organic matter on sewage sludge pyrolysis. J. Anal. Appl. Pyrolysis 2005, 74, 413–420. [Google Scholar] [CrossRef]
- Novak, J.; Lima, I.; Xing, B.; Gaskin, J.; Steiner, C.; Das, K. Characterization of designer biochar produced at different temperatures and their effects on a loamy sand. Ann. Environ. Sci. 2009, 3, 195–206. [Google Scholar]
- Lu, H.; Zhang, W.; Wang, S.; Zhuang, L.; Yang, Y.; Qiu, R. Characterization of sewage sludge-derived biochars from different feedstocks and pyrolysis temperatures. J. Anal. Appl. Pyrolysis 2013, 102, 137–143. [Google Scholar] [CrossRef]
- Halim, M.; Conte, P.; Piccolo, A. Potential availability of heavy metals to phytoextraction from contaminated soils induced by exogenous humic substances. Chemosphere 2003, 52, 265–275. [Google Scholar] [CrossRef]
- Cecconet, D.; Molognoni, D.; Callegari, A.; Capodaglio, A.G. Biological combination processes for efficient removal of pharmaceutically active compounds from wastewater. J. Environ. Chem. Eng. 2017, 5, 3590–3603. [Google Scholar] [CrossRef]
- Callegari, A.; Capodaglio, A.G. Effects of selected industrial pollutants on urban WWTPs activated sludge population, and possible mitigation strategies. Water Pract. Technol. 2017, 12, 619–637. [Google Scholar] [CrossRef]
- Giger, W.; Alder, A.C.; Golet, E.M.; Kohler, H.P.E.; McArdell, C.S.; Molnar, E.; Siegrist, H.; Suter, M.J.F. Occurrence and Fate of Antibiotics as Trace Contaminants in Wastewaters, Sewage Sludges, and Surface Waters. CHIMIA Int. J. Chem. 2003, 57, 485–491. [Google Scholar] [CrossRef]
- Capodaglio, A.G. High-energy oxidation process: An efficient alternative for wastewater organic contaminants removal. Clean Technol. Environ. Policy 2017, 19, 1995–2006. [Google Scholar] [CrossRef]
- Jelic, A.; Gros, M.; Ginebreda, A.; Cespedes-Sanchez, R.; Ventura, F.; Petrovic, M.; Barcelo, D. Occurrence, partition and removal of pharmaceuticals in sewage water and sludge during wastewater treatment. Water Res. 2011, 45, 1165–1176. [Google Scholar] [CrossRef] [PubMed]
- Trojanowicz, M.; Bojanowska-Czajka, A.; Capodaglio, A.G. Can radiation chemistry supply a highly efficient AO (R) P process for organics removal from drinking and waste water? A review. Environ. Sci. Pollut. Res. 2017, 24, 20187–20208. [Google Scholar] [CrossRef] [PubMed]
- Chan, K.Y.; Van Zwieten, L.; Meszaros, I.; Downie, A.; Joseph, S. Using poultry litter biochar as soil amendments. Soil Res. 2008, 46, 437–444. [Google Scholar] [CrossRef]
- Hossain, H.K.; Strezov, V.; Chan, K.Y.; Nelson, P.F. Agronomic properties of wastewater sludge biochar and bioavailability of metals in production of cherry tomato (Lycopersicon esculentum). Chemosphere 2010, 78, 1167–1171. [Google Scholar] [CrossRef] [PubMed]
- Collins, H. Use of biochar from the pyrolysis of waste organic material as a soil amendment: Laboratory and greenhouse analyses. In Quarterly Progress Report Prepared for the Biochar Project; USDA-ARS: Prosser, WA, USA, 2008. [Google Scholar]
- Brändli, R.C.; Hartnik, T.; Henriksen, T.; Cornelissen, G. Sorption of native polyaromatic hydrocarbons (PAH) to black carbon and amended activated carbon in soil. Chemosphere 2008, 73, 1805–1810. [Google Scholar] [CrossRef] [PubMed]
- Beesley, L.; Moreno-Jiménez, E.; Gomez-Eyles, J.L.; Harris, E.; Robinson, B.; Sizmur, T. A review of biochars’ potential role in the remediation, revegetation and restoration of contaminated soils. Environ. Pollut. 2011, 159, 3269–3282. [Google Scholar] [CrossRef] [PubMed]
- Yu, X.Y.; Ying, G.G.; Kookana, R.S. Reduced plant uptake of pesticides with biochar additions to soil. Chemosphere 2009, 76, 665–671. [Google Scholar] [CrossRef] [PubMed]
- Chen, B.L.; Zhou, D.D.; Zhu, L.Z. Transitional adsorption and partition of nonpolar and polar aromatic contaminants by biochars of pine needles with different pyrolytic temperatures. Environ. Sci. Technol. 2008, 42, 5137–5143. [Google Scholar] [CrossRef] [PubMed]
- Cao, X.D.; Ma, L.N.; Gao, B.; Harris, W. Dairy-manure derived biochar effectively sorbs lead and atrazine. Environ. Sci. Technol. 2009, 43, 3285–3291. [Google Scholar] [CrossRef] [PubMed]
- Lohmann, R.; MacFarlane, J.K.; Gschwend, P.M. Importance of black carbon to sorption of native PAHs, PCBs, and PCDDs in Boston and New York harbor sediments. Environ. Sci. Technol. 2005, 39, 141–148. [Google Scholar] [CrossRef] [PubMed]
- Nam, K.; Alexander, M. Role of nanoporosity and hydrophobicity in sequestration and bioavailability: Tests with model solids. Environ. Sci. Technol. 1998, 32, 71–74. [Google Scholar] [CrossRef]
- Demiral, H.; Demiral, I. Surface properties of activated carbon prepared from wastes. Surf. Interface Anal. 2008, 40, 612–615. [Google Scholar] [CrossRef]
- Beesley, L.; Jiménez, E.M.; Eyles, J.L.G. Effects of biochar and greenwaste compost amendments on mobility, bioavailability and toxicity of inorganic and organic contaminants in a multi-element polluted soil. Environ. Pollut. 2010, 158, 2282–2287. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Wang, H.; He, L.; Lu, K.; Sarmah, A.; Li, J.; Bolan, N.S.; Pei, J.; Huang, H. Using biochar for remediation of soils contaminated with heavy metals and organic pollutants. Environ. Sci. Pollut. Res. 2013, 20, 8472–8483. [Google Scholar] [CrossRef] [PubMed]
- Zhang, C.; Geng, Z.; Cai, M.; Zhang, J.; Liu, X.; Xin, H.; Ma, J. Microstructure regulation of superactivated carbon from biomass source corncob with enhanced hydrogenuptake. Int. J. Hydrog. Energy 2013, 38, 9243–9250. [Google Scholar] [CrossRef]
- Ahmad, M.; Rajapaksha, A.U.; Lim, J.E.; Zhang, M.; Bolan, N.; Mohan, D.; Vithanage, M.; Lee, S.S.; Ok, Y.S. Biochar as a sorbent for contaminant management in soil and water: A review. Chemosphere 2014, 99, 19–33. [Google Scholar] [CrossRef] [PubMed]
- Aliverti, N.; Callegari, A.; Capodaglio, A.G.; Sauvignet, P. NOM removal from freshwater supplies by advanced separation technology. In Advanced Water Supply and Wastewater Treatment; Springer: Dordrecht, The Netherlands, 2011; pp. 49–61. [Google Scholar]
- Callegari, A.; Boguniewicz-Zablocka, J.; Capodaglio, A.G. Experimental Application of an Advanced Separation Process for NOM Removal from Surface Drinking Water Supply. Separations 2017, 4, 32. [Google Scholar] [CrossRef]
- Chen, X.; Chen, G.; Chen, L.; Chen, Y.; Lehmann, J.; McBride, M.B.; Hay, A.G. Adsorption of copper and zinc by biochars produced from pyrolysis of hardwood and corn straw in aqueous solution. Bioresour. Technol. 2011, 102, 8877–8884. [Google Scholar] [CrossRef] [PubMed]
- Meng, J.; Feng, X.; Dai, Z.; Liu, X.; Wu, J.; Xu, J. Adsorption characteristics of Cu(II) from aqueous solution onto biochar derived from swine manure. Environ. Sci. Pollut. Res. 2014, 21, 7035–7046. [Google Scholar] [CrossRef] [PubMed]
- Zheng, W.; Guo, M.; Chow, T.; Bennett, D.N.; Rajagopalan, N. Sorption properties of greenwaste biochar for two triazine pesticides. J. Hazard. Mater. 2010, 181, 121–126. [Google Scholar] [CrossRef] [PubMed]
- Jia, M.; Wang, F.; Bian, Y.; Jin, X.; Song, Y.; Kengara, F.O.; Xu, R.; Jiang, X. Effects of pH and metal ions on oxytetracycline sorption to maize-straw-derived biochar. Bioresour. Technol. 2013, 136, 87–93. [Google Scholar] [CrossRef] [PubMed]
- Tan, X.; Liu, Y.; Zeng, G.; Wang, X.; Hua, X.; Gu, Y.; Yang, Z. Application of biochar for the removal of pollutants from aqueous solutions. Chemosphere 2015, 125, 70–85. [Google Scholar] [CrossRef] [PubMed]
- Yanai, Y.; Toyota, K.; Okazaki, M. Effects of charcoal addition on N2O emissions from soil resulting from rewetting air-dried soil in short-term laboratory experiments. Soil Sci. Plant Nutr. 2007, 53, 181–188. [Google Scholar] [CrossRef]
- Glaser, B.; Lehmann, J.; Zech, W. Ameliorating physical and chemical properties of highly weathered soils in the tropics with charcoal—A review. Biol. Fertil. Soils 2002, 35, 219–230. [Google Scholar] [CrossRef]
- Azargohar, R.; Dalai, A.K. Steam and KOH activation of biochar: Experimental and modelling studies. Microporous Mesoporous Mater. 2008, 110, 413–421. [Google Scholar] [CrossRef]
- Marin, L.S. Treatment of Biomass-Derived Synthesis Gas Using Commercial Steam Reforming Catalysts and Biochar. Ph.D. Thesis, Oklahoma State University, Stillwater, OK, USA, 2011. [Google Scholar]
- Yan, Q.; Wan, C.; Liu, J.; Gao, J.; Yu, F.; Zhang, J.; Cai, Z. Iron nanoparticles in situ encapsulated in biochar-based carbon as an effective catalyst for the conversion of biomass-derived syngas to liquid hydrocarbons. Green Chem. 2013, 15, 1631–1640. [Google Scholar] [CrossRef]
- Dehkhoda, A.M.; Ellis, N. Biochar-based catalyst for simultaneous reactions of esterification and transesterification. Catal. Today 2013, 207, 86–92. [Google Scholar] [CrossRef]
- Kastner, J.R.; Miller, J.; Geller, D.P.; Locklin, J.; Keith, L.H.; Johnson, T. Esterification of fatty acids using solid acid catalysts generated from biochar and activatedcarbon. Catal. Today 2012, 190, 122–132. [Google Scholar] [CrossRef]
- González, A.S.; Plaza, M.G.; Rubiera, F.; Pevida, C. Sustainable biomass-based carbon adsorbents for post-combustion CO2 capture. Chem. Eng. J. 2013, 230, 456–465. [Google Scholar] [CrossRef]
- Zhang, M.; Gao, B.; Li, Y.; Zhang, X.W.; Hardin, I.R. Graphene-coated pyrogenic carbon as an anode material for lithium battery. Chem. Eng. J. 2013, 229, 399–403. [Google Scholar] [CrossRef]
- Ahn, S.Y.; Eom, S.Y.; Rhie, Y.H.; Sung, Y.M.; Moon, C.E.; Choi, G.M.; Kim, D.J. Utilization of wood biomass char in a direct carbon fuel cell (DCFC) system. Appl. Energy 2013, 105, 207–216. [Google Scholar] [CrossRef]
- Kacprzak, A.; Kobyłecki, R.; Włodarczyk, R.; Bis, Z. The effect of fuel type on the performance of a direct carbon fuel cell with molten alkaline electrolyte. J. Power Sources 2014, 255, 179–186. [Google Scholar] [CrossRef]
- Capodaglio, A.G.; Molognoni, D.; Dallago, E.; Liberale, A.; Cella, R.; Longoni, P.; Pantaleoni, L. Microbial Fuel Cells for direct electrical energy recovery from urban wastewaters. Sci. World J. 2013, 3, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Capodaglio, A.G.; Cecconet, D.; Molognoni, D. An integrated mathematical model of microbial fuel cells processes: Bioelectrochemical and microbiologic aspects. Processes 2017, 5, 73. [Google Scholar] [CrossRef]
- Cecconet, D.; Devecseri, M.; Callegari, A.; Capodaglio, A.G. Effects of process operating conditions on the autotrophic denitrification of nitrate-contaminated groundwater using bioelectrochemical systems. Sci Total Environ. 2017, 613–614, 663–671. [Google Scholar] [CrossRef] [PubMed]
- Molognoni, D.; Devecseri, M.; Cecconet, D.; Capodaglio, A.G. Cathodic groundwater denitrification with a bioelectrochemical system. J. Water Process Eng. 2017, 19, 67–73. [Google Scholar] [CrossRef]
- Huggins, T.; Wang, H.; Kearns, J.; Jenkins, P.; Ren, Z.J. Biochar as a sustainable electrode material for electricity production in microbial fuel cells. Bioresour. Technol. 2014, 157, 114–119. [Google Scholar] [CrossRef] [PubMed]
- Yuan, Y.; Yuan, T.; Wang, D.; Tang, J.; Zhou, S. Sewage sludge biochar as an efficient catalyst for oxygen reduction reaction in a microbial fuel cell. Bioresour. Technol. 2013, 144, 115–120. [Google Scholar] [CrossRef] [PubMed]
- Liu, M.-C.; Kong, L.-B.; Zhang, P.; Luo, Y.-C.; Kang, L. Porous wood carbon monolith for high-performance supercapacitors. Electrochimica Acta 2012, 60, 443–448. [Google Scholar] [CrossRef]
- McCarl, B.A.; Peacocke, C.; Chrisman, R.; Kung, C.C.; Sands, R.D. Economics of biochar production, utilization and greenhouse gas offsets. In Biochar for Environmental Management: Science and Technology; Lehmann, J., Joseph, A.S., Eds.; Earthscan: London, UK, 2009; pp. 341–358. [Google Scholar]
- Qian, K.; Kumar, A.; Zhang, H.; Bellmer, D.; Huhnke, R. Recent advances in utilization of biochar. Renew. Sustain. Energy Rev. 2015, 42, 1055–1064. [Google Scholar] [CrossRef]
- Ok, Y.S.; Chang, S.X.; Gao, B.; Chung, H.J. SMART biochar technology—A shifting paradigm towards advanced materials and healthcare research. Environ. Technol. Innov. 2015, 4, 206–209. [Google Scholar] [CrossRef]
- Rajapaksha, A.U.; Chen, S.S.; Tsang, D.C.W.; Zhang, M.; Vithanage, M.; Mandal, S.; Gao, B.; Bolan, N.S.; Ok, Y.S. Engineered/designer biochar for contaminant removal/immobilization from soil and water: Potential and implication of biochar modification. Chemosphere 2016, 148, 276–291. [Google Scholar] [CrossRef] [PubMed]
- Zhang, M.; Gao, B.; Yao, Y.; Xue, Y.; Inyang, M. Synthesis, characterization, and environmental implications of graphene-coated biochar. Sci. Total Environ. 2012, 435–436, 567–572. [Google Scholar] [CrossRef] [PubMed]
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).