Abstract
Metal mobilization in general, as well as the number of metals used in products to increase performance and provide sometimes unique functionalities, has increased steadily in the past decades. Materials, such as indium, gallium, platinum group metals (PGM), and rare earths (RE), are used ever more frequently in high-tech applications and their criticality as a function of economic importance and supply risks has been highlighted in various studies. Nevertheless, recycling rates are often below one percent. Against this background, secondary flows of critical metals from three different end-of-life products up to 2020 are modeled and losses along the products’ end-of-life (EOL) chain are identified. Two established applications of PGM and RE–industrial catalysts and thermal barrier coatings–and CIGS photovoltaic cells as a relatively new product have been analyzed. In addition to a quantification of future EOL flows, the analysis showed that a relatively well working recycling system exists for PGM-bearing catalysts, while a complete loss of critical metals occurs for the other applications. The reasons include a lack of economic incentives, technologically caused material dissipation and other technological challenges.
1. Introduction
The conservation and considerate use of non-renewable resources is an important element of sustainability goals and strategies in Germany [1] and the EU [2]. These strategies aim at improving material efficiency, at substituting scarce resources with more abundant or renewable resources, and at significantly improving material recycling, which is an essential aspect of a sustainable management of materials (e.g., [3]).
The issue of material recycling is of particular importance, especially given the fact that global metals mobilization is significantly increasing [4] and an increasing number of elements from the periodic table are being used in products [5]. In various products, especially the so-called critical metals, provide a variety of new functionalities, increase efficiency of established applications, or enable miniaturization of products.
The issue of material criticality as the combination of a materials economic importance and its supply risks has already been discussed frequently (see e.g., [4,6]) and various criticality assessments have been carried out (e.g., [7,8,9]) for varying technological scopes and geographic areas, including the EU [10] and Germany [11]. An outline of the criticality assessment used in this study is given in the supplementary information. Metals, such as indium, gallium, germanium, platinum group metals (PGM), and rare earths (RE), are commonly among the materials labeled as critical. With the PGM as an exception, these critical metals share the same insufficiencies in their products’ life cycles: a high scale of dissipative losses along the life cycle [12] and barely any recycling at end-of-life [13].
Against this background, this article discusses several aspects of the situation described. First, three exemplary applications of critical metals are analyzed regarding the amounts of critical metals embodied in end-of-life products in 2020, with a regional focus on Germany. Knowledge about these stocks and flows is crucial for a development towards sustainable materials management. It is required for building up a recycling infrastructure to avoid the materials’ dissipation, as well as for urban mining activities. Then, for the three applications, the end-of-life chains in Germany are explored, identifying magnitude and location of material losses.
In addition to two established applications of PGM and RE–industrial catalysts and thermal barrier coatings, that have so far received only little attention in material flow analysis (MFA) and related studies, copper-indium-gallium-(di)selenide (CIGS) photovoltaic cells have been analyzed as a relatively new product, i.e., with currently no significant recycling activities.
2. Description of Methods
2.1. Modeling of End-of-Life Flows
To quantify the amounts of critical metals from the assessed end-of-life products, a product-centric approach has been chosen. The study focuses on Germany and the time horizon is 2020.
The applied approach as shown in Figure 1 focuses on the use phase and is determined by mainly three parameters: the amount of products entering the use phase per year (P), the concentration of critical metals per product (c), and the probability distribution for time spent in the use phase (V(t)), also called lifespan distribution. Additionally, exports from the use phase (E) have been considered but did not show any relevance for the three assessed products. Based on these parameters the amount of secondary materials embodied in EOL products (A) has been determined. Details on the parameters for each product are given in the respective sections.
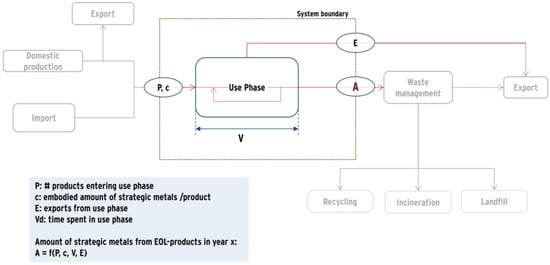
Figure 1.
Modeling approach and system boundaries.
As the analysis as well as the analyzed system is dynamic, the time spent in the use phase is a particularly important parameter. As highlighted by Murakami [14], various differing definitions of lifespan are used in science and literature. In this study, lifespan is understood as the time span from entering the use phase to leaving the use phase. This complies with the “domestic service lifespan” described by Murakami [14]. Instead of using average values, a life span probability distribution is used in the model. From the many plausible distributions functions, the Weibull distribution has been chosen as most suitable. The suitability of the Weibull distribution function for modeling product lifespans has been described in multiple studies (e.g., [15,16,17,18]). Examples for using the Weibull distribution to model end-of-life material flows are still very limited but can for example be found in [19] for wind energy converters or in [20,21], and [22], for photovoltaic cells.
The Weibull probability density function (WPDF) can be defined as:
where t is the time in years and k and λ are the shape and scale parameters, respectively. V(t, λ, k) is then the probability that a product has a lifespan of t years. Further details on the application of the Weibull function and related functions are given elsewhere (e.g., [15,23,24,25,26,27]) and will not be explained in detail here.
V(t, λ, k) = λk(λt)k−1e−(λt)k
For a given point in time T (e.g., the year 2020), the amount A(T) of end-of-life products is dependent on the amounts of products being sold in the past and their respective lifespan distribution. Here, we assume that the lifespan distribution does not change with time. Since the sales data is only available per full year, the amount of sold products and the amount of metals in end-of-life products can only be calculated on a per-year basis. With exports showing no particular relevance for the analyzed products (i.e., E = 0) the amount of strategic metals in end-of-life products in year T can be calculated from P, V and c as follows:
with
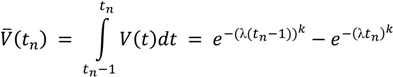
A(T) = cΣtn = 1,2,3…P(T − tn)V(tn)
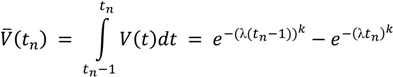
V(tn) is, thus, the cumulated probability that a product has a lifespan between (tn − 1) and tn years. Equation (2) represents a delay model (cf. [14]), and time series of past P flows are required for the calculation of A(t). Regarding the metal concentration c and the average lifespan, which is needed to specify the Weibull Function, it has to be noted that they are in fact dynamic parameters which vary over time. This fact could not be accounted for in the modeling approach due to data availability and reliability. Even though there was some data available referring to different years, this did not allow any conclusions on the development of metal concentration and average lifespan over time. To account for this, ranges have been used in the model where appropriate. Regarding the shape parameter k of the WPDF different references as described in Section 3.1.3 and Section3.2.3 have been used.
2.2. Analysis of Material Losses along Recycling/End-of-Life Chain
For the analysis of the material losses along the end-of-life chain, the idealized end-of-life chain comprising collection, sorting, pre-processing, and the final step of metal recovery is analyzed (see Figure 2). Similar approaches for the analysis of the efficiency of end-of-life chains can for example be found in [28,29,30,31,32]. Depending on the characteristics of the respective products, steps of the EOL chain are sometimes aggregated to streamline the analysis.
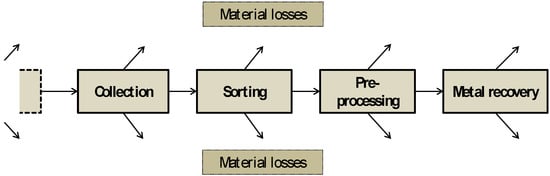
Figure 2.
Idealized end-of-life chain.
While for some of the analyzed metals (i.e., indium, gallium, and rare earth metals) it is well known that EOL recycling rates are below one percent, the chosen approach provides additional information about where the losses occur and what the aim of potential measures should be.
3. Exemplary Product Analyses
3.1. Thermal Barrier Coatings
Thermal barrier coatings (TBC) are used in stationary gas turbines and aircraft engines to isolate turbine components from the hot gas stream whose temperatures can go up to 1400 °C [33,34,35]. Commonly, yttria stabilized zirconia (YSZ) is used in these applications. While the purpose of TBC in stationary gas turbines and aircraft engines is practically identical, the two applications differ in parameters, such as thickness and service life of the coating, as well as yttrium concentration.
3.1.1. Metal Concentration
The amount of yttrium in the TBC per final product (i.e., per megawatt for stationary gas turbines and per engine for aircrafts) has been analyzed by literature research and consultation of experts. Based on this and additional information from experts, the amount of strategic metal in the final product is determined.
For stationary gas turbines the majority of data indicates an Y2O3 content of 7–8 wt% that has also been confirmed by manufacturers [34,35,36,37,38,39,40,41,42,43,44,45]. The density of YSZ is about 6 g/cm3. Layer thickness varies between 0.4 mm and 1.5 mm with an average of about 0.85 mm, the coated surface area ranges from 0.16 to 0.53 m2/MW (average 0.29 m2/MW) [42,46].
Based on this, a specific yttrium concentration of 71 to 165 g per megawatt has been determined. An average value of 118 gram per megawatt is used in the baseline scenario.
For aircraft engines, the Y2O3 content varies between 7 and 8 mol-% (about 13.7 wt%) [47,48,49,50,51]. The thickness of the coating is in the range of 50 and 250 µm (average 150 µm) [46,51,52,53,54]. Due to limited data availability, the density of YSZ has been assumed to be about 6 g/cm3 analogously to the YSZ used in stationary gas turbines. The size of the coated surface per engine can range from about 3 to 8 m2. Based on this and additional literature and expert information [41,47,51,55], the yttrium amount per engine can be estimated to be 411 to 959 g. Further details are given in the supplementary information (see Tables S1 and S2). For the baseline scenario a specific concentration of 685 g per engine is used.
3.1.2. Products Placed on the Market and Lifespan
In both analyzed applications TBC are only part of a component of the actual product (central and decentral power plants and aircrafts or their turbines, respectively). The amount of TBC placed on the market is assessed based on the stock (-development) of the main products. It is composed of products newly put on the market and replacements within the stock. The former parameter can be determined based on the amount of (main) products newly put on the market per year using the specific metal concentration described above. For the latter, we distinguished between the stock in 2012 and TBC added to the stock through newly installed gas turbines (or aircrafts put into operation, respectively) from 2013 on. For the stock of TBC in 2012, due to lack of data, the replacements in the following years have been modeled assuming a simultaneous exit (SE) function and a homogeneous age distribution within the stock of TBC in 2012. For new installations from 2012 on, replacements have been modeled assuming a Weibull distribution, i.e., the flows into use are calculated using the following equation:


Regarding stationary gas turbines, one has to distinguish between smaller decentralized industrial applications and bigger power plants. For the latter, the lifespan of the TBC is estimated to be four years, while for decentralized turbines five years are assumed (based on data and information from [35,37,38,40,41,46,56,57,58,59,60,61]).
Data regarding the historic and future stock development of gas turbines has been taken from statistics and technology forecasts [62,63,64,65] and is compiled in Table 1 including the resulting amount of yttrium for the years 2012 to 2020.

Table 1.
Yttrium flows into use-stationary gas turbines.
| Year | Installed capacity–decentralized gas turbines [MW] | Resulting Y flows into use [kg] (baseline scenario) | Installed capacity–centralized power plants [MW] | Resulting Y flows into use [kg] (baseline scenario) |
|---|---|---|---|---|
| 2006 | 7,480 | – | 17,760 | – |
| 2007 | – | – | 18,317 | – |
| 2008 | – | – | 19,761 | – |
| 2009 | – | – | 20,469 | – |
| 2010 | 8,200 | – | 20,916 | – |
| 2011 | 8,500 | – | 23,302 | – |
| 2012 | 8,800 | 238 | 24,796 | 812 |
| 2013 | 9,100 | 242 | 25,077 | 713 |
| 2014 | 9,400 | 248 | 25,997 | 820 |
| 2015 | 9,700 | 254 | 26,530 | 802 |
| 2016 | 10,000 | 261 | 26,530 | 760 |
| 2017 | 10,300 | 268 | 29,030 | 1,067 |
| 2018 | 10,600 | 275 | 29,630 | 874 |
| 2019 | 10,900 | 283 | 30,830 | 666 |
| 2020 | 11,200 | 290 | 30,830 | 837 |
When it comes to TBC in aircraft engines, the geographic focus on Germany is not strictly applicable. Therefore, to analyze the amount of products placed on the market we have focused on German airlines (Lufthansa German Airlines, Air Berlin, German wings, TUI fly and Condor Flugdienst). Stock and planned expansion of their aircraft fleets have been taken into account [66,67,68,69,70,71,72,73]. The lifespan of the coating differs between short- and mid-/long-distance planes. While both have an equal average number of flight hours per year of about 4000 h, due to the higher numbers of take-offs and landings, short-distance planes have a higher frequency of service intervals. Hence for short-distance planes a lifetime of the coating of about 2.25 years and for mid-/long-distance planes of about 5.5 years was assumed (based on information and data from [41,46,47,74,75,76,77,78,79,80,81,82,83]; see also Tables S3 and S4 in the supplementary information).
The stock development over time is presented in Table 2.

Table 2.
Development of aircraft stock of German airlines up to 2020 and resulting yttrium flows into use (in baseline scenario) [66,67,68,69,70,71,72,73].
| Year | Short-Distance | Mid-/Long distance | ||||
|---|---|---|---|---|---|---|
| Aircrafts | Engines | Y flows into use (kg) | Aircrafts | Engines | Y flows into use (kg) | |
| 2012 | 264 | 528 | 161 | 326 | 844 | 105 |
| 2013 | 295 | 590 | 203 | 362 | 936 | 168 |
| 2014 | 325 | 650 | 208 | 398 | 1028 | 170 |
| 2015 | 350 | 700 | 215 | 423 | 1096 | 158 |
| 2016 | 350 | 700 | 196 | 453 | 1156 | 118 |
| 2017 | 350 | 700 | 204 | 482 | 1214 | 125 |
| 2018 | 350 | 700 | 204 | 496 | 1242 | 130 |
| 2019 | 350 | 700 | 204 | 498 | 1246 | 133 |
| 2020 | 350 | 700 | 204 | 498 | 1246 | 134 |
3.1.3. Calculation and Results
Following the approach and data described above, the calculation of the yttrium amounts in end-of-life thermal barrier coatings has been conducted. For the additionally required parameter for the application of the Weibull distribution, the shape parameter k, the Japanese lifespan database for vehicles, equipment, and structures (LiVES) has been consulted [84]. Here, a k-value of 2.03 can be found for engines and turbines. To the best of the authors’ knowledge, other data has not yet been published. Based on this, a k-value of 2 is used in the calculations of the baseline scenario for both turbine applications. Additionally, due to the uncertainty of the k-value, values of k = 1.5 and k = 2.5 are analyzed in a sensitivity analysis to investigate the potential influence on the results. In the baseline scenario, the scale parameter λ has been determined to 0.221 for centralized gas turbines, to 0.177 for decentralized gas turbines, to 0.394 for short-distance aircrafts, and to 0.161 for mid- and long-distance aircrafts (e.g., [27]).
The results are shown in Figure 3 and Figure 4 for the baseline scenarios as well as selected alternative scenarios in which the parameters have been varied as described above. In the baseline scenario, a steady increase from 805 kg in 2013 to 1062 kg in 2020 can be observed. Naturally, the assumptions about the metal concentration in the product have a significant influence on the results. The variation of the shape parameter k, however, has only little influence on the results. Results of additional parameter variations for selected years as well as a summary of parameters used in the calculation are given in the supplementary information (Tables S5, S6 and S7).
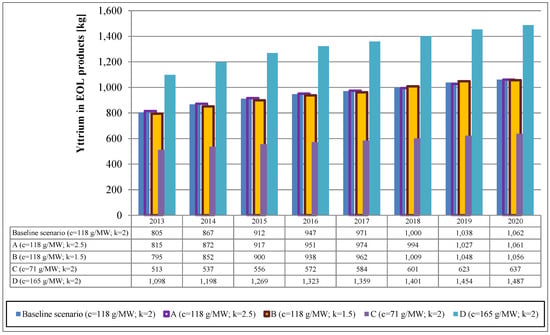
Figure 3.
Yttrium from TBC in stationary gas turbines.
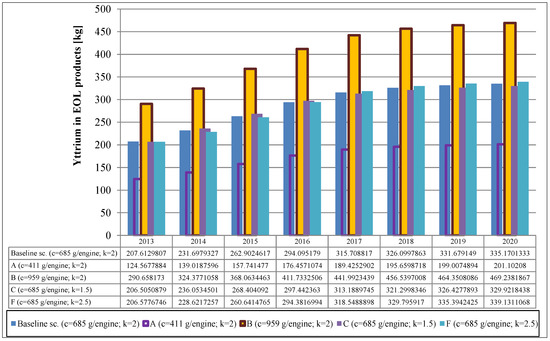
Figure 4.
Yttrium from TBC in aircraft engines.
3.1.4. Material Losses along EOL Chain
There are two possible end-of-life options for thermal barrier coatings from aircraft engines. Either the coating is removed during regular maintenance intervals or it is discarded with the coated components at its end-of-life. In regular maintenance–coated components of aircraft engines are used for 1 to 4 cycles—the coating is commonly removed using processes such as high pressure water blasting (for the combustor), aluminum oxide blasting (for blades and vanes) [51]. 2 cycles can be assumed as an average [51], meaning a 50–50 split of the relevant wastes between regular maintenance and final disposal.
In regular maintenance, the blasting wastes are collected manually as well as as filter cake with minor differences between the different blasting processes. In the following steps, however, further processing aiming at a recovery of rare earth elements is not practiced. The respective wastes are commonly landfilled [85].
In the final disposal of the coated components, rare earth recovery is not a priority either. Components usually go to metal recovery, but without any ambitions to recover the contained rare earth metals [85]. The losses at the different end-of-life stages are summarized in Table 3.

Table 3.
Losses along the EOL chain of TBC.
| Life cycle stage | Collection | Sorting, pre-processing | Recovery | EOL chain |
|---|---|---|---|---|
| Maintenance | <10% | irrelevant | 100% | 100% |
| Final disposal | <10% | irrelevant | 100% | 100% |
3.2. CIGS Photovoltaic Cells
Photovoltaic cells play an important role in most strategies aimed at supporting a low carbon society. In Germany, promoted by instruments, such as feed in tariffs, the installed capacity of photovoltaic cells has grown significantly in the past years and is today comparable in size with wind energy. On the technological side, two types of photovoltaic cells can be distinguished: silicon based cells and thin film cells. Other types of PV cells like compound semiconductors and nanotechnology cells are not yet relevant for commercial energy production [86]. Especially their costs, temperature robustness, and versatility of thin film cells led to a significant gain in the market share [86]. The following analysis will focus on copper-indium-gallium-di-selenide (CIGS) cells.
3.2.1. Metal Concentration
CIGSis a mixture of CIS (copper-indium-di-selenide) and CGS (copper-gallium-di-selenide). Its chemical formula is CuInxGa(1-x)Se2. The x value can vary from 0 and 1 meaning different ratios of CIS to CGS (e.g., [87]). The thickness of the active layer of CIGS cells ranges from 1 to 3 µm. Most modules show a thickness of around 2 µm. The efficiency of commercial modules ranges between 10 and 12 percent (e.g., [86,88]). Although CIGS cells differ in their gallium content, it can be said that modules without gallium do not play a significant role while cells with gallium can be considered as common [89]. Additional differences in the material intensity result from different thicknesses of the active layer and differences in the module’s efficiency. These variations in the material intensity are reflected in the literature and primary data.
The data for the material intensity has been taken from literature but also includes primary data from manufacturers and expert judgments. Literature data is partly referring to different reference units (e.g., kg/kW) and needs to be normalized to kg/MWp.
In addition to literature data [21,22,88,89,90,91,92,93,94,95,96] primary data from two manufacturers (a German and a Japanese manufacturer) could be obtained and expert judgments (from “Helmholtz-Zentrum Berlin für Materialien und Energie GmbH” and “German Solar Industry Association–BSW Solar”) for complementing and validating the collected data have been included. Original data, excluded data and harmonized data as well as details on how the data have been harmonized and compiled are given in the supplementary information (see Tables S8 and S9). The resulting compiled data is presented in Table 4.

Table 4.
Material intensity of copper-indium-gallium-di-selenide (CIGS) photovoltaic cells. Data from literature, experts, and manufacturers.
| Metal | Material intensity (kg/MW) | ||
|---|---|---|---|
| Lower bound | Mean value–baseline scenario | Upper bound | |
| Indium | 9.8 | 16.5 | 23.1 |
| Gallium | 2.3 | 11.0 | 19.7 |
The relatively great spread in the data could not be explained by its age, technological development, trends, or other factors, but is supposed to be caused by differences in the design between different producers, such as a variation in the gallium-indium ratio as described above.
3.2.2. Products Placed on the Market
CIGS photovoltaic cells are a relatively young technology and thus empiric data on the service life span of the modules is rare. Commonly, a lifespan of between 20 and 30 years is assumed for all kinds of photovoltaic cells (e.g., [22,96,97,98,99,100,101,102,103]). In addition, in some studies it is indicated that the lifespan of thin film modules is more than 25 years [96,104].
In some of the few studies dealing with life span observations of PV-modules lifespans of 29.6 years [105], 27.7, and 28 years [106] are reported.
Against this background, a lifespan of 25 years is assumed in the baseline scenario and a lifespan of 30 years is analyzed in a sensitivity analysis.
The amount of products placed on the market is available from statistics [107] and forecasts [108] providing information about photovoltaic installations in general. For the shares of the different photovoltaic technologies data from [109] and [92] has been used.
Based on this, CIGS installations and resulting metal flows could be determined as shown in Table 5. Before 1999, no relevant installations of CIGS photovoltaic cells have been reported.

Table 5.
CIGS installations and metal flows into use.
| Year | CIGS installations (MW) | Metal flows into use (kg)–baseline scenario | Year | CIGS installations (MW) | Metal flows into use (kg)–baseline scenario | ||
|---|---|---|---|---|---|---|---|
| In | Ga | In | Ga | ||||
| 1999 | 0.009 | 0.15 | 0.10 | 2010 | 111.8 | 1844.83 | 1229.89 |
| 2000 | 0.066 | 1.09 | 0.73 | 2011 | 179.6 | 2964.06 | 1976.04 |
| 2001 | 0.22 | 3.63 | 2.42 | 2012 | 101.6 | 1677.06 | 1118.04 |
| 2002 | 0.22 | 3.63 | 2.42 | 2013 | 101.2 | 1670.04 | 1113.36 |
| 2003 | 0.83 | 13.76 | 9.17 | 2014 | 104.5 | 1724.26 | 1149.51 |
| 2004 | 2.7 | 44.22 | 29.48 | 2015 | 110.2 | 1817.49 | 1211.66 |
| 2005 | 1.9 | 31.38 | 20.92 | 2016 | 115.7 | 1908.71 | 1272.47 |
| 2006 | 1.7 | 27.82 | 18.55 | 2017 | 123.9 | 2043.95 | 1362.64 |
| 2007 | 6.4 | 104.86 | 69.91 | 2018 | 132.4 | 2184.62 | 1456.41 |
| 2008 | 19.5 | 321.75 | 214.50 | 2019 | 141.1 | 2327.34 | 1551.56 |
| 2009 | 75.6 | 1247.10 | 831.40 | 2020 | 149.6 | 2467.58 | 1645.05 |
3.2.3. Calculation and Results
Based on the approach and parameters described above, the flows of indium and gallium in end-of-life CIGS modules have been calculated.
For the shape parameter a value of k = 5.4 has been chosen based on a study by Kuitche (2010) [105]. The flows of indium and gallium from EOL CIGS cells in the baseline and alternative scenarios are shown in Figure 5 and Figure 6, respectively.
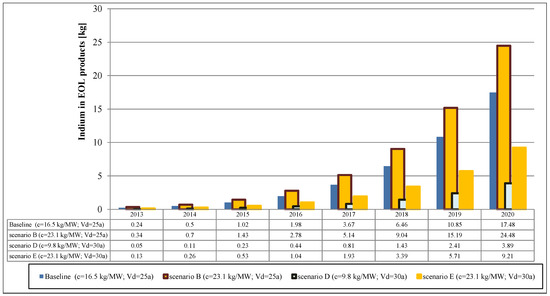
Figure 5.
Secondary indium flows from EOL CIGS modules.
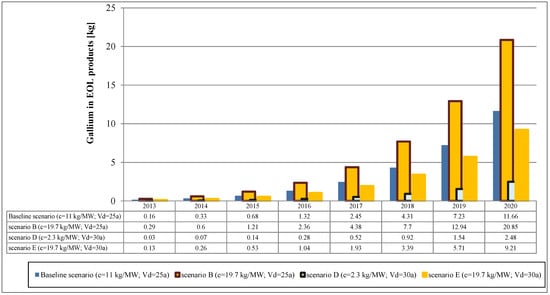
Figure 6.
Secondary gallium flows from EOL CIGS modules.
3.2.4. Material Losses along EOL Chain
On 14 August, 2013, the new WEEE (Waste Electrical and Electronic Equipment) directive became effective. Until February 2014 it needs to be adapted to national laws, e.g., in Germany through an adaption of the ElektroG. How exactly this adaptation will look and what implications it will bring for photovoltaic cells remains to be seen. A quota for the collection of 85% of the total amount of waste modules is being discussed [110]. Apart from that, it has to be noted that there are already different systems for the taking-back and recycling of photovoltaic modules, e.g., PV cycle, the E-Bell recycling system or the First Solar recycling system (see [111,112,113,114]) providing an infrastructure that makes a high collection rate of EOL PV modules likely. Against this background, a collection rate of at least 85% can be assumed which is also in accordance with similar studies [115].
In the following steps–sorting, processing, and recycling–there is so far only very little experience regarding the treatment of thin-film modules and the little amounts of thin-film modules do not allow for an economical feasible recovery of semiconductor materials. Nevertheless, higher amounts can be expected soon and different recycling processes are currently being developed by different German companies, such as Loser Chemie, Solarcycle, Lobbe, and Saperatec [111,113,116,117,118]. Recovery rates for indium and gallium in the different processes can be quantified to 70% to 90% [119].
The material losses along the end-of-life chain of CIGS photovoltaic cells can be summed up as shown in Table 6. The table shows the present situation (no recycling of semiconductor materials) as well as an outlook based on processes currently under development on how the future EOL situation might look like.

Table 6.
Material losses along the end-of-life (EOL) chain of CIGS photovoltaic cells.
| Scenario | Collection | Sorting, pre-processing, recovery | EOL chain |
|---|---|---|---|
| Today | <15% | 100% In, Ga | 100% In |
| Outlook | <15% | 10%–30% In, Ga | 24%–41% In, Ga |
3.3. Industrial Catalysts
Catalysts are of great importance in various industries where they are a key factor for production efficiency. In the chemical industry over 90% of all products are produced using catalysts [120,121]. The industrial fields of application of catalysts can be divided into polymerization, pollution control/environment protection, and chemical industry [121]. From a material point of view, another distinction can be made between PGM, rare earth and germanium bearing catalysts. The following analysis will follow this distinction and PGM-, rare earth- and germanium bearing catalysts will be analyzed in separate sections.
Contrary to the other analyzed applications, catalysts have a relatively short lifespan of often below one year. Because of that, the application of the Weibull distribution is not feasible for catalysts. Instead, the amount of products placed on the market can function as a proxy for the amount of end-of-life products and metals, respectively. In addition, it has to be noted that in most fields of applications the amount of catalysts placed on the market is constant over time disregarding minor annual fluctuation.
3.4. PGM Bearing Catalysts
For PGM bearing catalysts (and other PGM applications in Germany) a detailed study has been published by Hagelüken and colleagues [122]. Additional information was available from [32,120,122,123,124,125]. From these studies, information about material flows including secondary material flows was already available as well as forecasts about the future development up to 2020. Therefore parameters such as the metal concentration and the amount of products placed on the market did not need to be investigated further.
3.4.1. Calculation and Results
The secondary flows of platinum, palladium, and rhodium over time are shown in Table 7. In addition to catalysts in nitric acid production and catalysts in homogeneous catalysis, constant annual consumptions have been assumed.

Table 7.
Secondary flows of PGM from industrial catalysts [kg].
| Catalyst type | Platinum | Palladium | Rhodium | |||
|---|---|---|---|---|---|---|
| 2013 | 2020 | 2013 | 2020 | 2013 | 2020 | |
| Refinery catalysts | 830 | 90 | – | |||
| Catalysts in nitric acid production | 760 | 200 | 40 | |||
| Catalysts in cyanhydric acid production | 180 | 187 | – | 20 | 27 | |
| Powdered catalyst | 533–588 | 746–842 | 4801–5294 | 6876–7581 | – | |
| Packed and fluidized-bed catalysts | 320 | 2900 | – | |||
| Homogeneous catalysis | 2286–2520 | 3274–3610 | 114–126 | 164–180 | 594–656 | 851–939 |
| Environmental catalysts | 150 | – | – | |||
| Total | 5059–5349 | 6285–6699 | 8106–8610 | 10,230–10,951 | 654–716 | 918–1006 |
3.4.2. Material Losses along EOL Chain
Generally, a rather well-functioning recycling system for industrial catalysts exists; high recovery rates of platinum group metals from PGM bearing industrial catalysts are achieved (e.g., [123,126,127]). This well-working system has existed for some time now and already in 2001 generally low losses of precious metals in the end-of-life treatment of industrial catalysts have been identified [120]. In a subsequent study in 2003 the European recycling infrastructure for the recycling of precious metals from industrial catalysts was found to be functioning and sufficient [125].
A summary of the losses in the end-of-life treatment of different catalyst types along the end-of-life chain is shown in Table 8. Losses are generally below five percent, environmental catalysts being one exception of minor relevance in terms of mass. Reasons for this well-working recycling system are described in detail in [123]. The high concentration of PGM in the catalysts compared to its concentration in ore, its economic value, the availability of suitable processes and well-functioning co-operations between users and manufacturers/recyclers can be named as the main reasons.

Table 8.
Losses of critical metals along EOL chain of PGM bearing catalysts (calculated based on data from [123]).
| Catalyst type | Collection and sorting | Pre-processing and recovery | EOL chain |
|---|---|---|---|
| Refinery catalysts | irrelevant | 1.5% (Pt und Pd) | 1.5% |
| Cat. in nitric acid production | irrelevant | irrelevant | irrelevant |
| Cat. in cyanhydric acid production | irrelevant | irrelevant | irrelevant |
| Chemical industry: powdered catalyst | irrelevant | 2% | 2% |
| Chemical industry: packed and fluidized-bed catalysts | irrelevant | irrelevant | <4.5% |
| Chemical industry: homogeneous catalysis | Irrelevant for Pt, Rh48% Pd | 3.3% Pt; 4.4% Rh; 3.8% Pd | 3.3% Pt; 4.4% Rh; 50% Pd |
| Environmental catalysts | 80%–90% | <10% | 82%–91% |
3.5. Rare Earth Bearing Catalysts
In petroleum refining, catalysts bearing cerium and lanthanum are used for fluid-catalytic-cracking (FCC) [90,120,128,129]. Cerium and lanthanum increase the gasoline yield and reduce air emissions from the oil refining process [91].
3.5.1. Metal Concentration
The rare earth element mainly used in FCC catalysts is lanthanum; cerium is of minor importance [91]. Data regarding the metal content in the catalyst is to a large extent in the same region and ranges from 2 to 6 mass percent [91,128,130,131,132,133,134] with the vast majority of values between 2 and 3 mass percent.
Based on this, a rare earth content of 2.5 wt% is assumed for the baseline scenario; values of 2 and 3 wt% are assessed in a sensitivity analysis.
Additionally, it has to be noted that in fluid catalytic cracking there are no special requirements regarding the purity of the rare earths and up to 20% other rare earth elements like cerium, neodymium, praseodymium, and samarium can be added [135].
3.5.2. Products Placed on the Market and Lifespan
Statistical or other data on the consumption of FCC catalysts in Germany has not been available. Therefore, the catalyst consumption has been estimated based on the production output of FCC-processes.
Germany has a FCC capacity of about 350,000 barrels-per-day. Catalysts are replaced continuously and about 0.2 lb (about 91 g) catalysts are consumed per barrel [90,131]. In the past years, the capacity utilization of refineries in Germany varied between 78 and 95% [136], but general trends towards an increase or decrease of the total capacity and capacity utilization cannot be identified. Therefore, the average capacity utilization rate of the years 1990 to 2012 of 81.3% is used as baseline scenario for the years 2013 to 2020 [137]; in addition 78% and 95% values are assessed in a sensitivity analysis.
3.5.3. Calculations and Results
Based on the parameters described above the annual amounts of rare earths in FCC-catalysts reaching their end-of-life have been calculated. The results are shown in Table 9. For the baseline scenario, an amount of 235 tons has been calculated. Considering possible deviations of the metal concentration and the capacity utilization the results can vary from 181 to 330 tons.

Table 9.
Rare earths in EOL FCC catalysts.
| Rare earths in EOL FCC catalysts, 2013 to 2020 (t) | Capacity utilization rate | |||
|---|---|---|---|---|
| 81.3% | 78% | 95% | ||
| Metal concentration | 2.5% | 235 | 226 | 275 |
| 2% | 188 | 181 | 220 | |
| 3% | 282 | 271 | 330 | |
3.5.3. Material Losses along End-of-Life Chain
Contrary to PGM in industrial catalysts, rare earths from FCC catalysts are commonly not recovered [132]. After their collection they are usually landfilled, or, due to the material properties of the substrate, used as filling material in the construction industry or cement production [120,128,132]. Based on this, a complete loss of critical metals along the end-of-life chain can be assumed as shown in Table 10.

Table 10.
Losses of critical metals along EOL chain of FCC catalysts.
| Application | Collection, sorting | Pre-processing and recovery | EOL chain |
|---|---|---|---|
| FCC-catalysts | <10% (RE*) | 100% (RE*) | 100% (RE*) |
| *La, Ce, Nd, Pr, Sm | |||
3.6. Ge Bearing Catalysts
In PET production, GeO2 is used as polymerization catalyst. PET manufactured using GeO2 catalysts is mainly used for drink bottles and shows particularly good properties in terms of brightness and shine [128]. Antimony and titanium are alternative catalysts in PET manufacturing and account for the majority of the market, i.e., about 90% of all PET is produced using antimony catalysts [138,139]; while titanium and germanium account for the balance.
In PET manufacturing, GeO2 from polymerization catalysts dissipates completely into the product and is not recovered [120,125,128,140]. Therefore, the potential secondary flows are not quantified based on the number of polymerization catalysts but on the number of PET bottles placed on the market.
3.6.1. Metal Concentration
The germanium content in GeO2 catalysts is about 69%. As mentioned above, the catalyst dissipates entirely into the product. Data regarding the GeO2 concentration ranges from 1:10,000 [125] to 1:100,000 to 7:100,000 [128] or 10 to 70 mg/kg, respectively. As it is more up-to-date, the OakdeneHollins data (1:100,000 to 7:100,000 with an average of 1:25,000) is used in the following.
In combination with data on the weight of PET bottles of different sizes [141,142] a specific GeO2 concentration per 1l-bottles can be calculated. Assuming an average GeO2 concentration in the PET of 1:25,000, the specific concentration is 0.88 to 1.22 mg/1l-bottle for one-way, and 2.48 to 2.83 mg/1l-bottle for returnable bottles.
3.6.2. Products Placed on the Market
The amount of products placed on the market, i.e., the number of PET bottles on the market, is analyzed based on the beverage consumption [143] and statistics regarding the shares of different types of packaging [141,143,144] (see supplementary information for further details; Tables S10, S11 and S12). The statistics show that the beverage consumption remained relatively constant for the majority of beverages and no significant changes in the total beverage consumption are expected for the future. Following this, the average beverage consumption of the years 2004 to 2009 has been used for calculating the baseline scenario. Additionally, fluctuations of ±10% have been analyzed in a sensitivity analysis. Regarding the share of different packaging types, different scenarios regarding the future development have been analyzed (see Table 11).

Table 11.
Share of PET bottles for different beverage types.
| Beverage type | Year | Returnable PET bottles | One-way PET Bottles |
|---|---|---|---|
| Beer and shandy | 2010–2020 | – | 6%/7%/8% |
| Water | 2010–2020 | 15%/20% | 50%/56.1%/70% |
| Soft drinks | 2010–2020 | 15%/25% | 50%/60.5%/70% |
3.6.3. Calculation and Results
As stated above, constant beverage consumption and probable fluctuations of ±10% have been assumed. Therefore, no strict distinction is being made between the years 2013 to 2020 but a probable range valid for the entire period is calculated. Besides the fluctuations of the beverage consumption, the described ranges of the specific GeO2 concentration have been considered.
Following this, a germanium amount in EOL PET bottles of 837 to 1835 kg (1336 kg in the baseline scenario) has been calculated.
3.6.4. Material Losses along End-of-Life Chain
Since the germanium dissipates completely into the PET, the PET bottles have to be considered the relevant product for analyzing the end-of-life chain. Within the current end-of-life treatment of PET bottles, the recovery of germanium is not an issue in any form and a complete loss has to be noted. To the authors’ knowledge, no findings exist regarding potential options to recover germanium from PET. According to OakdeneHollins [128] the feasibility of a recovery has to be questioned owing to the low concentration in the product. Additional problems result from the mixing with bottles manufactured with alternative catalysts and further dissipation in the subsequent applications of the recycled material, e.g., fleece clothing.
3.7. Assessing the Results’ Scale
To place the results (secondary material flows up to 2020) into context, i.e., to provide a sense of scale, they have been put into relation to the world mine production of 2011 (based on data from British Geological Survey and U.S. Geological Survey). Here, only the results of the baseline scenarios are considered. As no reliable data on the production of the different rare earth elements has been available, the rare earth elements considered in the study (yttrium and rare earths from FCC catalysts) are put into relation to the accumulated production of all rare earth elements.
As shown in Table 12, the largest secondary material flows compared to the world production are those of PGM from industrial catalysts. The secondary flows of indium and gallium from CIGS photovoltaic cells, on the other hand, are relatively small. However, since CIGS photovoltaic cells are a young technology and due to their long lifespan, a significant growth of secondary flows can be expected for the following years.

Table 12.
Ratios of secondary metal flows to world production.
| Metal | World production | 2013 | 2014 | 2015 | 2016 | 2017 | 2018 | 2019 | 2020 |
|---|---|---|---|---|---|---|---|---|---|
| Y | 125,384 t (RE) | 0.001% | 0.001% | 0.001% | 0.001% | 0.001% | 0.001% | 0.001% | 0.001% |
| In | 641 t | ~0% | 0.0001% | 0.0002% | 0.0003% | 0.0006% | 0.0010% | 0.0017% | 0.0027% |
| Ga | 216 t | ~0% | 0.0002% | 0.0003% | 0.0006% | 0.0011% | 0.0020% | 0.0033% | 0.0054% |
| Pt | 179 t | 2.91% | 3.01% | 3.11% | 3.22% | 3.32% | 3.42% | 3.52% | 3.63% |
| Pd | 200 t | 4.18% | 4.34% | 4.50% | 4.66% | 4.82% | 4.98% | 5.14% | 5.30% |
| Rh | 28 t | 2.45% | 2.59% | 2.73% | 2.87% | 3.01% | 3.15% | 3.29% | 3.44% |
| RE in FCC Catalysts | 125,384 t (RE) | 0.20% | |||||||
| Ge | 120 t | 1.11% | |||||||
The total secondary flows of rare earth metals from the analyzed applications (thermal barrier coatings and FCC catalysts) are located somewhere between the two, if looked at together. Comparing them to the world production of the respective rare earth element would probably result in somewhat higher values. However, this step could not be performed due to data availability.
4. Discussion and Conclusions
In this study, the flows of selected critical metals at the end-of-life of three different applications have been quantified and the respective end-of-life chains have been analyzed. Besides for the products with relatively short lifespans (i.e., around one year or less) or constant stocks, for which a simplified approach has been followed, the Weibull function has been used to model the secondary material flows of the analyzed products. Especially for products with high growth rates such as photovoltaic cells or otherwise volatile markets, the Weibull distribution has to be considered preferable compared to a simultaneous exit functionas different studies indicate (see e.g., [15,16,17,18,19]).
Even though the chosen methodological approach is connected with some uncertainties regarding parameters such as metal concentration in the product, average lifespan, and the future market development of the products, it provides valuable knowledge that is required for any measures contributing to more sustainable materials management. Especially for building up a recycling infrastructure aiming at an improved recovery of critical metals and for urban mining activities, knowledge about stocks and flows is essential.
While for PGM bearing catalysts–just like for most other PGM applications–a relatively well-functioning recycling system exists, there is quite a lot of room for improvements regarding the other analyzed products. In the analyzed rare earth applications–thermal barrier coatings and catalysts for fluid catalytic cracking–a well-functioning collection system already exists. Still, due to their historically particularly low prices, the recovery of the rare earths does not play a role in the subsequent steps of the end-of-life chain even though a significant potential for improvements of the recycling situation can be identified. Considering their criticality [6,7,8,9,10,11], i.e., their economic importance and high supply risks, and past events like the Chinese export restrictions, such improvements should get a much higher attention in the future.
Contrary to the relatively well established applications FCC catalysts and TBC, recycling of critical metals from CIGS photovoltaic cells has already gained considerable attention; even though end-of-life flows will probably not reach a significant scale for at least the next 8 to 15 years. In part, this is triggered by the higher economic value of indium and gallium compared to lanthanum or cerium, but existing and planned legislation as well as research funding contributed to this development as well.
The last analyzed product–germanium bearing polymerization catalysts–does not show any recycling potential since the metals dissipate completely into the product. Recycling of germanium from PET is–as described above–unfeasible, but in-process recovery in the PET production is apparently also economically unfeasible. Disregarding the apparent lack of economic incentives and considering the fact that polymerization accounts for 37% of the total germanium consumption, a highly significant potential for optimizations in terms of a sustainable metals management can be identified here.
Comparing the three different products, we can also identify specific reasons for the failing recycling of metals. In the cases analyzed here, it is not the interfaces between elements of the recycling chain that are ineffective, as is typically the case for automotive catalysts or some WEEE wastes. The reasons here are either economic (low prices for target metal), or technical (low concentration of metal contained in low-value/high-volume waste streams), or both. Early consideration of the (potential) criticality of metals, as in the case of PV modules, helps in avoiding such threats to a closed loop metal economy and maintains options for a future metals management that is resilient against supply disruptions.
Acknowledgments
The presented results originate from an ongoing research project carried out on behalf of the German Federal Environmental Agency (UBA) under the Environmental Research Plan-funding reference number 371193 339–and is funded by the German Federal Ministry for the Environment, Nature Conservation and Nuclear Safety (BMU). The responsibility for the content of the publication lies with the authors.
We thank Anna Schaffner, Uwe Schulz, Jörg Sopka, O. Görke, Jürgen Lemke, Thilo Seitz, Ralf Burmeister, Philipp Döbber, Renate Gülzow, Max Seier, Viktor Knobloch, Jan Kosmol, the journal editor and two anonymous reviewers for providing feedback, support, expertise and/or helpful comments.
Author Contribution
Till Zimmermann and Stefan Gößling-Reisemann designed the research and the methodological framework. Till Zimmermann performed the research and analyzed the data. Both co-authors wrote the paper.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Deutsches Ressourceneffizienzprogramms (ProgRess)—Programm zur Nachhaltigen Nutzung und zum Schutz natürlicher Ressourcen; Bundesministerium für Umwelt, Naturschutz und Reaktorsicherheit (BMU; German Federal Ministry for the Environment, Nature Convervation, and Nuclear Safety): Berlin, Germany, 2012. (in German)
- European Commission. Mitteilung der Kommission an das europäische Parlament, den Rat, den europäischen Wirtschafts- und Sozialausschuss und den Ausschuss der Regionen: Fahrplan für ein ressourcenschonendes Europa, Kom (2011) 571; Communication from the Commission to the European Parliament. Europeam Commission: Brussels, Belgium, 2011. (in German) [Google Scholar]
- Von Gleich, A. Outlines of a Sustainable Metals Industry. In Sustainable Metals Management: Securing Our Future-Steps towards a Closed Loop Economy; von Gleich, A., Ayres, R.U., Gößling-Reisemann, S., Eds.; Springer: Berlin, Germany, 2006; pp. 3–39. [Google Scholar]
- Graedel, T.E.; Barr, R.; Chandler, C.; Chase, T.; Choi, J.; Christoffersen, L.; Friedlander, E.; Henly, C.; Jun, C.; Nassar, N.T.; et al. Methodology of metal criticality determination. Environ. Sci. Technol. 2012, 46, 1063–1070. [Google Scholar] [CrossRef]
- Reuter, M.; Hudson, C.; Hagelüken, C.; Heiskanen, K.; Meskers, C.; van Schaik, A. Metal Recycling: Opportunities, Limits, Infrastructure; UNEP Report; United Nations Environment Programme: Paris, France, 2013. [Google Scholar]
- Erdmann, L.; Graedel, T.E. Criticality of non-fuel minerals: A review of major approaches and analyses. Environ. Sci. Technol. 2011, 45, 7620–7630. [Google Scholar] [CrossRef]
- American Physical Society; Materials Research Society. Energy Critical Elements: Securing Materials for Emerging Technologies; A Report by the APS Panel on Public Affairs & the Materials Research Society: Washington, DC, USA, 2011. [Google Scholar]
- Buchert, M.; Schüler, D.; Bleher, D. Critical Metals for Future Sustainable Technologies and their Recycling Potential; UNEP Report: Darmstadt, Germany, July 2009. [Google Scholar]
- Reller, A. Rohstoffsituation Bayern: Keine Zukunft ohne Rohstoffe: Strategien und Handlungsoptionen; vbw Bayern, IW Consult GmbH: Munich, Germany, 2009. [Google Scholar]
- European Commission. Critical Raw Materials for the EU; Report of the Ad-hoc Working Group on Defining Critical Raw Materials; European Commission: Brussels, Belgium, 2010. [Google Scholar]
- Erdmann, L.; Behrendt, S. Kritische Rohstoffe für Deutschland: Identifikation aus Sicht Deutscher Unternehmen Wirtschaftlich Bedeutsamer Mineralischer Rohstoffe, Deren Versorgungslage sich Mittel- bis Langfristig als Kritisch Erweisen Könnte; Final Report; kfw Bankengruppe: Berlin, Germany, 2011. (in German) [Google Scholar]
- Zimmermann, T.; Gößling-Reisemann, S. Critical materials and dissipative losses: A screening study. Sci. Total Environ. 2013, 461–462, 774–780. [Google Scholar] [CrossRef]
- Graedel, T.E.; Allwood, J.; Birat, J.-P.; Buchert, M.; Hagelüken, C.; Reck, B.K.; Sibley, S.F.; Sonnemann, G. What do we know about metal recycling rates? J. Ind. Ecol. 2011, 15, 355–366. [Google Scholar] [CrossRef]
- Murakami, S.; Oguchi, M.; Tasaki, T.; Daigo, I.; Hashimoto, S. Lifespan of commodities, part I. J. Ind. Ecol. 2010, 14, 598–612. [Google Scholar] [CrossRef]
- Cullen, A.C.; Frey, H.C. Probabilistic Techniques in Exposure Assessment: A Handbook for Dealing with Variability and Uncertainty in Models and Inputs; Plenum Press: New York, NY, USA, 1999. [Google Scholar]
- Tasaki, T.; Takasuga, T.; Osako, M.; Sakai, S.-I. Substance flow analysis of brominated flame retardants and related compounds in waste TV sets in Japan. Waste Manag. 2004, 24, 571–580. [Google Scholar] [CrossRef]
- Kagawa, S.; Tasaki, T.; Moriguchi, Y. The environmental and economic consequences of product lifetime extension: Empirical analysis for automobile use. Ecol. Econ. 2006, 58, 108–118. [Google Scholar] [CrossRef]
- Oguchi, M.; Kameya, T.; Yagi, S.; Urano, K. Product flow analysis of various consumer durables in Japan. Resour. Conserv. Recycl. 2008, 52, 463–480. [Google Scholar] [CrossRef]
- Zimmermann, T.; Rehberger, M.; Gößling-Reisemann, S. Material flows resulting from large scale deployment of wind energy in Germany. Resources 2013, 2, 303–334. [Google Scholar] [CrossRef]
- Marwede, M.; Reller, A. Future recycling flows of tellurium from cadmium telluride photovoltaic waste. Resour. Conserv. Recycl. 2012, 69, 35–49. [Google Scholar] [CrossRef]
- Zimmermann, T. Historic and Future Flows of Critical Materials Resulting from Deployment of Photovoltaics. In Proceedings of the 6th International Conference on Life Cycle Management (LCM 2013), Gothenburg, Sweden, 25–28 August 2013.
- Zimmermann, T. Dynamic Material Flow Analysis of Critical Metals Embodied in Thin-Film Photovoltaic Cells. Available online: http://www.uni-bremen.de/fileadmin/user_upload/single_sites/artec/artec_Dokumente/artec-paper/194_paper.pdf (accessed on 4 January 2013).
- Wilker, H. Leitfaden zur Zuverlässigkeitsermittlung technischer Komponenten: Mit 86 Tabellen, 86 Beispielen, 2nd ed.; BoD (Books on demand): Norderstedt, Germany, 2010. (in German) [Google Scholar]
- Qiu, S.-L.; Vuorinen, M. Special Functions in Geometric Function Theory. In Handbook of Complex Analysis: Geometric Function Theory, 1st ed.; Kühnau, R., Ed.; Elsevier North Holland: Amsterdam, The Netherlands, 2005. [Google Scholar]
- Abramowitz, M.; Stegun, I.A. Handbook of Mathematical Functions with Formulas, Graphs, and Mathematical Tables, 7th ed.; Dover Publications: New York, NY, USA, 1972. [Google Scholar]
- Lehman, E.H. Shapes, moments and estimators of the weibull distribution. IEEE Trans. Reliab. 1963, 12, 32–38. [Google Scholar] [CrossRef]
- Nomura, K. Duration of Assets: Examination of Directly Observed Discard Data in Japan; KEO Discussion Paper No. 99; Keio Economic Observatory, Keio University: Tokyo, Japan, 2005. [Google Scholar]
- Chancerel, P. Substance Flow Analysis of the Recycling of Small Waste Electrical and Electronic Equipment: An Assessment of the Recovery of Gold and Palladium. Ph.D. Thesis, Technische Universität Berlin, Berlin, Germany, 2010. [Google Scholar]
- Chancerel, P.; Meskers, C.E.; Hagelüken, C.; Rotter, V.S. Assessment of precious metal flows during preprocessing of waste electrical and electronic equipment. J. Ind. Ecol. 2009, 13, 791–810. [Google Scholar] [CrossRef]
- Chancerel, P.; Rotter, S. Edelmetallrückgewinnung aus Elektro- und Elektronikaltgeräten durch Aufbereitung. Müll Abfall 2009, 2, 78–82. (in German). [Google Scholar]
- Buchert, M.; Manhart, A.; Bleher, D.; Pingel, D. Recycling Critical Raw Material from Waste Electronic Equipment; Oeko-Institut e.V.: Freiburg, Darmstadt, Germany, 2012. [Google Scholar]
- Hagelüken, C. Edelmetallrecycling-Status und Entwicklungen. In Sondermetalle und Edelmetalle: Vorträge beim 44. Metallurgischen Seminar des Fachausschusses für Metallurgische Aus- und Weiterbildung der GDMB vom 03.02. bis 05.02.2010 in Hanau; Harre, J., Ed.; GDMB-Info GmbH: Clausthal-Zellerfeld, 2010; Volume 121. (in German) [Google Scholar]
- Batista, C. Laser-Glazing of Plasma-Sprayed Thermal Barrier Coatings-Experimental and Computational Studies. Master Thesis, Universidade do Minho, Braga, Portugal, 2007. [Google Scholar]
- Schweda, M.E. Optimierung von APS-ZrO2-Wärmedämmschichten durch Variation der Kriechfestigkeit und der Grenzflächenrauhigkeit. Ph.D. Thesis, RWTH Aachen, Aachen, Germany, 2010. [Google Scholar]
- Clarke, D.R.; Oechsner, M.; Padture, N.P. Thermal-barrier coatings for more efficient gas-turbine engines. MRS Bull. 2012, 37, 891–898. [Google Scholar] [CrossRef]
- Batista, C. Thermal barrier coatings (TBCs)-State of the art. In Laser-Glazing of Plasma-Sprayed Thermal Barrier Coatings-Experimental and Computational Studies; Universidade do Minho: Braga, Portugal, 2007. [Google Scholar]
- Nelson, W.A.; Orenstein, R.M. TBC experience in land-based gas turbines. J. Therm. Spray Technol. 1997, 6, 176–180. [Google Scholar] [CrossRef]
- Subanovic, M. Einfluss der Bondcoatzusammensetzung und Herstellungsparameter auf die Lebensdauer von Wärmedämmschichten bei zyklischer Temperaturbelastung. In Schriften des Forschungszentrum Jülich; Forschungszentrum Jülich: Jülich, Germany, 2009. (in German) [Google Scholar]
- Vaßen, R.; Jarligo, M.O.; Steinke, T.; Mack, D.E.; Stöver, D. Overview on advanced thermal barriercoatings. Surf. Coat. Technol. 2010, 205, 938–942. [Google Scholar] [CrossRef]
- Czech, N. Korrosion und Beschichtung. In Stationäre Gasturbinen; Lechner, C., Seume, J., Eds.; Springer-Verlag Berlin Heidelberg: Berlin, Germany, 2014. (in German) [Google Scholar]
- Schulz, U.; Head of Department, Hochtemperatur-und Funktionsschichten, DLR (Deutsches Zentrum für Luft- und Raumfahrt), Köln, Germany. Personal Communication, January 2013.
- Sopka, J.; Technology/Development, ALSTOM Power GmbH, Mannheim, Germany. Personal Communication, October 2012.
- Bacos, M.-P.; Dorvaux, J.-M.; Landais, S.; Lavigne, O.; Mévrel, R.; Poulain, M.; Rio, C.; Vidal-Sétif, M.-H. 10 years-activities at onera on advanced thermal barrier coatings. J. Aerosp. Lab. 2011, 3, 1–14. [Google Scholar]
- Trunova, O.; Beck, T.; Herzog, R.; Steinbrech, R.; Singheiser, L. Damage mechanisms and lifetime behavior of plasma sprayed thermal barrier coating systems for gas turbines—Part I: Experiments. Surf. Coat. Technol. 2008, 202, 5027–5032. [Google Scholar] [CrossRef]
- Görke, O.; Institut für Werkstoffwissenschaften, FG Keramische Werkstoffe, TU Berlin, Berlin, Germany. Personal Communication, November 2012.
- Lemke, J.; ALD Vacuum Technologies GmbH, Mannheim, Germany. Personal Communication, January 2013.
- Seitz, T.; Director Propulsion Systems Engineering, Lufthansa Technik AG, Hamburg, Germany. Personal Communication, 2013.
- SulzerMetco. 8% Yttria Stabilized Zirconia Agglomerated and HOSP Thermal Spray Powders: Material Product Data Sheet. 2012. Available online: http://www.sulzer.com/en/-/media/Documents/ProductsAndServices/Coating_Materials/Thermal_Spray/ProductInformation/Ceramics_Zirconium_Oxide/DSMTS_0001_2_8YOZrOHOSP.pdf (Accessed on 2 January 2013).
- SulzerMetco. 8% Yttria Stabilized Zirconia Agglomerated and Sintered Thermal Spray Powders, Material Product Data Sheet. 2012. Available online: http://www.sulzer.com/en/-/media/Documents/ProductsAndServices/Coating_Materials/Thermal_Spray/ProductInformation/Ceramics_Zirconium_Oxide/DSMTS_0047_1_8YOZrOaggsint.pdf (accessed on 2 January 2013).
- SulzerMetco. Ceria-Yttria Stabilized Zirconium Oxide HOSP Powder: Material Product Data Sheet. 2012. Available online: http://www.sulzer.com/en/-/media/Documents/ProductsAndServices/Coating_Materials/Thermal_Spray/ProductInformation/Ceramics_Zirconium_Oxide/DSMTS_0038_0_CeZrO.pdf? (Accessed on 02 January 2013).
- Burmeister, R.; Döbber, P.; MTU Maintenance Hannover GmbH, Hannover, Germany. Personal Communication, 2013.
- Strangman, T.E. Thermal barrier coatings for turbine airfoils. Thin Solid Films 1985, 127, 93–106. [Google Scholar] [CrossRef]
- Nissley, D.M. Thermal barrier coating life modeling in aircraft gas turbine engines. JTST 1997, 6, 91–98. [Google Scholar] [CrossRef]
- Rolls-Royce. Journey through a Jet Engine. 2011. Available online: http://www.rolls-royce.com/interactive_games/journey03/index.html (accessed on 15 January 2013).
- Parsons, D.; Chatterton, J. Ceramic Coatings for Jet Engine Turbine Blades; Carbon Brainprint Case Study, Center for Environmental Risks and Futures, Cranfield University: Cranfield, UK, 2011. [Google Scholar]
- Padture, N.P. Thermal barrier coatings for gas-turbine engine applications. Science 2002, 296, 280–284. [Google Scholar] [CrossRef]
- Heinloth, K. Die Energiefrage: Bedarf und Potentiale, Nutzen, Risiken und Kosten, 2nd ed.; Vieweg: Braunschweig, Germany, 2003. (in German) [Google Scholar]
- Wenk, E. Neue Turbine, Neue Computer. 2011. Available online: http://www.pnn.de/potsdam/554325/ (accessed on 2 January 2013).
- Stadtwerke Leipzig. Turbinentausch im Leipziger Kraftwerk. 2012. Available online: http://www.swl.de/web/swl/DE/Unternehmen/presse/Pressemeldungen/2012/turbinentausch.htm (accessed on 2 January 2013).
- Lechner, C.; Seume, J. Stationäre Gasturbinen; Springer-Verlag: Berlin, Germany, 2010. (in German) [Google Scholar]
- Babar, F.-U.-R. Gas Turbines Maintenance Inspections and Calculations for Equivalent Operating Hours. 2013. Available online: http://de.scribd.com/doc/37753088/Gas-Turbine-Equivalent-Op-Hours-for-Maintenance (accessed on 4 February 2013).
- ASUE. Gasturbinen-Kenndaten-Referenzen; Arbeitsgemeinschaft für Sparsamen und Umweltfreundlichen Energieverbrauch e.V.: Kaiserslautern, Germany, 2006. (in German) [Google Scholar]
- ASUE. Hocheffiziente Kraft-Wärme-Kopplung mit Gasturbinen: Gesetzliche Rahmenbedingungen, Aktuelle Entwicklungen, Betriebserfahrungen. In Proceedings of ASUE-Fachtagung, Berlin, Germany, 29–30 September 2009; Arbeitsgemeinschaft für Sparsamen und umweltfreundlichen Energieverbrauch e.V.: Berlin, Germany, 2009. (in German). [Google Scholar]
- Bundesverband der Energie- und Wasserwirtschaft e.V. (BDEW). 69 Kraftwerke bis 2020 geplant, Anlage zur Presseinformation “Trotz Milliardeninvestition keine Entwarnung beim Kraftwerksbau”. 2012. (in German). Available online: http://bdew.de/internet.nsf/id/91C0FC9A8D7AD3EEC12579E9002F8CBF/$file/120424%20Anlage%20zur%20PM%20Hannover_Kraftwerksliste%20aktuell.pdf (accessed on 3 January 2013).
- Umweltbundesamt. Kraftwerke in Deutschland. 2012. (in German). Available online: http://www.umweltbundesamt.de/energie/archiv/kraftwerke_in_deutschland_datenbank.xls (accessed on 4 February 2013).
- AirBerlin. Airberlin IR–Die Flotte. 2013. (in German). Available online: http://ir.airberlin.com/de/ir/fakten-zur-gruppe/die-flotte (accessed on 8 February 2013).
- AirBerlin. UnsereFlotte. 2013. (in German). Available online: http://www.airberlin.com/de-DE/site/flotte.php (accessed on 7 February 2013).
- Condor. Condor-Unternehmen|Unsere Geschichte. 2012. (in German). Available online: http://www.condor.com/de/unternehmen/unsere-geschichte.jsp (accessed on 8 February 2013).
- Condor. Unsere Flotte. 2013. (in German). Available online: http://www.condor.com/de/unternehmen/unsere-flotte.jsp (accessed on 7 February 2013).
- TAI. World of TUI als verborgener Airline-Riese. 2012. (in German). Available online: http://www.tai.at/index.php?option=com_content&view=article&id=2719:world-of-tui-als-verborgener-airline-riese&catid=9&Itemid=4&lang=de (accessed on 8 February 2013).
- TUIfly. Über TUIfly–Das Unternehmen|TUIfly. (in German). Available online: http://www.tuifly.com/de/unternehmen_tuifly/index.html (accessed on 7 February 2013).
- Lufthansa, A.G. Geschäftsbericht. 2011. (in German). Available online: http://investor-relations.lufthansa.com/fileadmin/downloads/de/finanzberichte/geschaeftsberichte/LH-GB-2011-d.pdf (accessed on 8 February 2013).
- Lufthansa, A.G. Lufthansa-Flotte. (in German). Available online: http://konzern.lufthansa.com/flotte.html (accessed on 7 Februar 2013).
- Steinke, S. Luftfahrt-Nachrichten und –Community. (in German). Available online: http://www.aero.de/news-14328/Lufthansa-mustert-ihre-erste-Boeing-747-400-aus.html (accessed on 15 January 2013).
- Pandit, P.N. Tenets of MRO Strategy for Airlines. Available online: http://www.infosys.com/industries/airlines/white-papers/Documents/tenets-MRO-strategy.pdf (accessed on 7 February 2013).
- Aircraft-commerce. Aircraft Owner’s and Operator’s Guide: 747-200/-300. 2005. Available online: http://www.aircraft-commerce.com/sample_articles/sample_articles/owners_guide.pdf (accessed on 7 February 2013).
- Heermann, J. Warum sie oben bleiben-FAQ vom Autor. 2011. (in German). Available online: http://www.flugingenieur.de/faq/faq_vom_autor.htm (accessed on 15 January 2013).
- Aviation Broker GmbH. Fragen rund ums Fliegen. 2012. (in German). Available online: http://www.aviation-broker.com/flugzeuge/technik.html (accessed on 7 February 2013).
- China Economic Net. In China entwickelter Passagierjet Comac C919 Soll im Jahr 2014 starten. 2012. (in German). Available online: http://de.ce.cn/ga/un/unternehmen/201209/18/t20120918_581146.shtml (accessed on 7 February 2013).
- Krummheuer, E. Verlängertes Leben: Airbus macht Jets fit. 2008. (in German). Available online: http://www.handelsblatt.com/unternehmen/industrie/verlaengertes-leben-airbus-macht-jets-fit/2910418.html;%20https://www.bit-ag.com/downloads/drpeters_vpinfo_08_2008_lebenszyklusa320.pdf (accessed on 7 February 2013).
- Stadt Cuxhaven. Stadt Cuxhaven-Flugzeuge, n.d. (in German). Available online: http://www.cuxhaven.de/staticsite/staticsite.php?menuid=66&topmenu=13 (accessed on 7 February 2013).
- Flugzeugforum. durchschnittliche Flugleistung? n.d. (in German). Available online: http://www.flugzeugforum.de/durchschnittliche-flugleistung-47174.html (accessed on 7 February 2013).
- Flugzeugforum. Lebensdauer von Flugzeugen, n.d. (in German). Available online: http://www.flugzeugforum.de/lebensdauer-flugzeugen-37810.html (accessed on 7 February 2013).
- NIES. Lifespan database for Vehicles, Equipment, and Structures: LiVES. 2010. Available online: http://www.nies.go.jp/lifespan/index-e.html (accessed on 30 January 2013).
- Gülzow, R.; Umweltmanagement, Abfallentsorgung, Lufthansa Technik GmbH, Hamburg, Germany. Personal Communication, 2013.
- El Chaar, L.; lamont, L.; El Zein, N. Review of photovoltaic technologies. Renew. Sustain. Energy Rev. 2011, 15, 2165–2175. [Google Scholar] [CrossRef]
- Speirs, J.; Gross, R.; Candelise, C.; Gross, B. Materials Availability: Potential Constraints to the Future Low-Carbon Economy; Working Paper, Imperial College: London, UK, 2011. [Google Scholar]
- Fthenakis, V. Sustainability of photovoltaics: The case for thin-film solar cells. Renew. Sustain. Energy Rev. 2009, 13, 2746–2750. [Google Scholar] [CrossRef]
- Sander, K.; Schilling, S.; Wambach, K.; Schlenker, S.; Müller, A.; Springer, J.; Fouquet, D.; Jelitte, A.; Stryi-Hipp, G.; Chrometzka, T. Studie zur Entwicklung eines Rücknahme- und Verwertungssystems für photovoltaische Produkte; Ökopol: Hamburg, Germany, 2007. (in German) [Google Scholar]
- Critical Materials Strategy; US DOE Report, U.S. Department of Energy: Washington, DC, USA, 2010.
- Critical Materials Strategy; US DOE Report, U.S. Department of Energy: Washington, DC, USA, 2011.
- Moss, R.L.; Tzimas, E.; Kara, H.; Kooroshy, J. Critical Metals in Strategic Energy Technologies: Assessing Rare Metals as Supply-Chain Bottlenecks in Low-Carbon Energy Technologies; JRC Scientific and Technical Reports, Joint Research Center, European Commission: Petten, Netherlands, 2011. [Google Scholar]
- Andersson, B.A. Materials availability for large-scale thin-film photovoltaics. Progr. Photov. Res. Appl. 2000, 8, 61–76. [Google Scholar] [CrossRef]
- Jungbluth, N.; Tuchschmid, M. Photovoltaics. In Sachbilanzen von Energiesystemen: Grundlagen für den ökologischen Vergleich von Energiesystemen und den Einbezug von Energiesystemen in Ökobilanzen für die Schweiz; Dones, R., Ed.; Paul Scherrer Institute Villigen, Swiss Centre for Life Cycle Inventories: Dübendorf, Switzerland, 2007. [Google Scholar]
- Bleiwas, D.I. Byproduct Mineral Commodities Used for the Production of Photovoltaic Cells. Circular 1365; 2010. Available online: http://pubs.usgs.gov/circ/1365/Circ1365.pdf (accessed on 19 July 2012). [Google Scholar]
- Berger, W.; Simon, F.-G.; Weimann, K.; Alsema, E.A. A novel approach for the recycling of thin film photovoltaic modules. Resourc. Conserv. Recycl. 2010, 54, 711–718. [Google Scholar] [CrossRef]
- Sherwani, A.F.; Usmani, J.A.; Varun. Life cycle assessment of solar PV based electricity generation systems: A review. Renew. Sustain. Energy Rev. 2010, 14, 540–544. [Google Scholar] [CrossRef]
- García-Valverde, R.; Miguel, C.; Martínez-Béjar, R.; Urbina, A. Life cycle assessment study of a 4.2 kWp stand-alone photovoltaic system. Solar Energy 2009, 83, 1434–1445. [Google Scholar] [CrossRef]
- Stoppato, A. Life cycle assessment of photovoltaic electricity generation. Energy 2008, 33, 224–232. [Google Scholar] [CrossRef]
- Briem, S.; Blesl, M.; Fahl, U.; Ohl, M.; Moerschner, J.; Eltrop, L.; Voß, A.; Viebahn, P.; Krewitt, W.; Gürzenich, D.; et al. Lebenszyklusanalysen Ausgewählter zuküNftiger Stromerzeugungstechnologien. Forschungsvorhaben mit Finanzieller Unterstützung des Bundesministeriums für Wirtschaft und Arbeit; University of Stuttgart: Stuttgart, Germany, 2004. (in German) [Google Scholar]
- Azzopardi, B.; Mutale, J. Life cycle analysis for future photovoltaic systems using hybrid solar cells. Renew. Sustain. Energy Rev. 2010, 14, 1130–1134. [Google Scholar] [CrossRef]
- Held, M.; Ilg, R. Update of environmental indicators and energy payback time of CdTe PV systems in Europe. Progr. Photovolt Res. Appl. 2011, 19, 614–626. [Google Scholar] [CrossRef]
- Raugei, M.; Fthenakis, V. Cadmium flows and emissions from CdTe PV: Future expectations. Energy Policy 2010, 38, 5223–5228. [Google Scholar] [CrossRef]
- European Photovoltaic Industry Association (EPIA). Solar Generation 6: Solar Photovoltaic Electricity Empowering the World; EPIA: Brussels, Belgium, 2011. [Google Scholar]
- Kuitche, J.M. Statistical Lifetime Predictions for PV Modules. 2010. Available online: http://www1.eere.energy.gov/solar/pdfs/pvrw2010_kuitche.pdf (accessed on 12 March 2013).
- Kumar, S.; Sarkan, B. Design for reliability with weibull analysis for photovoltaic modules. Int. J. Curr. Eng. Technol. 2013, 3, 129–134. [Google Scholar]
- Bundesministerium für Umwelt, Naturschutz und Reaktorsicherheit (BMU). Erneuerbare Energien-Zeitreihen zur Entwickung der erneuerbaren Energien in Deutschland. 2012. Available online: http://www.erneuerbare-energien.de/erneuerbare_energien/datenservice/zeitreihen/doc/45919.php (accessed on 15 October 2012).
- Bundesministerium für Umwelt, Naturschutz und Reaktorsicherheit (BMU). Langfristszenarien und Strategien für den Ausbau der erneuerbaren Energien in Deutschland bei Berücksichtigung der Entwicklung in Europa und Global. Schlussbericht-BMU-FKZ 03MAP146. 2012. (in German). Available online: http://www.fvee.de/fileadmin/publikationen/Politische_Papiere_anderer/12.03.29.BMU_Leitstudie2011/BMU_Leitstudie2011.pdf.
- PHOTON. Das Solarstrommagazin: April 2012-Anteile der verschiedenen Zelltechnologien, weltweite Solarzellenproduktion 1999 bis 2011. Photon Das Solarstrom Mag. 2012, 4, 43–53. [Google Scholar]
- Wambach, K. Erfassung von Solarmodulen-Zukünftige Entsorgungsmengen. In Proceedings of Urban Mining Kongress 2013, Iserlohn, Germany, 12–13 June 2013; Available online: http://urban-mining-kongress.de/fileadmin/pdfs/vortraege_2013/Wambach_Urbanmining12062013.pdf (accessed on 10 October 2013).
- PV Cycle. PV CYCLE, Rücknahme und Recycling von PV-Altmodulen. 2013. (in German). Available online: http://www.pvcycle.org/de/ (accessed on 30 May 2013).
- Wambach, K.; Wade, A. Rückführungskonzepte und Aufbereitungsverfahren für Photovoltaikmodule-Stand der Technik und rechtliche Rahmenbedingungen. First Solar, Wambach Consulting; Urban Mining Kongress: Iserlohn, Germany, 2013. (in German) [Google Scholar]
- Beckmann, J. Wieso Abfall? Entsorgung von photovoltaik-anlagen. Installateur 2012, 2, 28–31. [Google Scholar]
- Landbell AG. Neues Rücknahmesystem für ausgediente Solarmodule. 2013. (in German). Available online: http://www.landbell.de/news/102-neues-ruecknahmesystem-fuer-ausgediente-solarmodule.html (accessed on 17 July 2013).
- Sinha, P.; Cossette, M.; Ménard, J.-F. End-of-life CdTe PV recycling with semiconductor refining. In 27th European Photovoltaic Solar Energy Conference and Exhibition, Proceedings of the International Conference, Frankfurt, Germany, 24–28 September 2012; Nowak, S., Jäger-Waldau, A., Helm, P., Eds.; European Photovoltaic Solar Energy Conference and Exhibition (EU PVSEC): München, Germany, 2012; pp. 4653–4656. [Google Scholar]
- Palitzsch, W. PV Recyclingstrategien der Loser Chemie GmbH; Loser Chemie GmbH: Langenweißbach, Germany. (in German)
- Lobbe. Recycling von PV-Modulen. 2012. (in German). Available online: http://www.lobbe.de/fileadmin/Lobbe_Downloads/TatSachen/Tat39_web.pdf (accessed on 17 July 2013).
- Betrieb einer Recyclinganlage für Photovoltaik-Module. 2013. (in German). Available online: http://www.saperatec.de/index.php/technologie/referenzen (accessed on 17 July 2013).
- Marwede, M.; TU Berlin, Fraunhofer IZM, Berlin, Germany. Personal Communication, 2013.
- Hassan, A. Rohstoffeinsparung durch Kreislaufführung von verbrauchten Katalysatoren aus der chemischen Industrie; Forschungsbericht 299 93 301,UBA-FB 000139; Umweltbundesamt: Berlin, Germany, 2001. (in German) [Google Scholar]
- Weitkamp, J.; Gläser, R. Katalyse. In Chemische Technik: Prozesse und Produkte, 5th ed.; Dittmeyer, R., Keim, W., Kreysa, G., Oberholz, A., Eds.; Wiley-VCH: Weinheim, Germany, 2004–2006. (in German) [Google Scholar]
- Hagelüken, C.; Buchert, M.; Stahl, H. Stoffströme der Platingruppenmetalle: Systemanalyse und Massnahmen für eine nachhaltige Optimierung der Stoffströme der Platingruppenmetalle; Endbericht; GDMB-Medienverl: Clausthal-Zellerfeld, Germany, 2005. (in German) [Google Scholar]
- Hagelüken, C. Material Flows of Platinum Group Metals in Germany. Presentation at GFMS Precious & Base Metals Seminar. 2005. Available online: http://www.gfms.co.uk/Market%20Commentary/Materials%20Flow%20of%20PGMs%20in%20Germany.pdf (accessed on 17 March 2011).
- Hagelüken, C.; Meskers, C. Technology challenges to recover precious and special metals from complex products. 2009. Available online: http://ewasteguide.info/files/Hageluecken_2009_R09.pdf (accessed on 28 January 2014).
- Hassan, A. Stand der Verwertung von verbrauchten Katalysatoren aus der chemischen Industrie sowie Einflussfaktoren zur Verbesserung der Kreislaufführung. Forschungsbericht 363 01 046, UBA-FB 000443; Umweltbundesamt: Berlin, Germany, 2003. (in German) [Google Scholar]
- Hagelüken, C.; Director EU Governmental Affairs, Umicore Precious Metals Refining. Hanau, Germany. Personal Communication, 2013.
- Zimmer, H.-O.; GmbH & Co. KG. Hürth, Germany. Managing director Zimmer Katalysatoren-Recycling. Personal Communication, 2012. [Google Scholar]
- Oakdene Hollins. Study into the Feasibility of Protecting and Recovering Critical Raw Materials through Infrastructure Development in the South East of England; Final Report; Oakdene Hollins, Ed.; European Pathway to Zero Waste Environment Agency: Reading, Berkshire, UK, 2011. [Google Scholar]
- Yang, W.-C. Handbook of Fluidization and Fluid-Particle Systems; Marcel Dekker: New York, NY, USA, 2003. [Google Scholar]
- Schüler, D.; Buchert, M.; Liu, R.; Dittrich, S.; Merz, C. Study on Rare Earths and Their Recycling; Final Report for The Greens/EFA Group in the European Parliament: Darmstadt, Germany, 2011. [Google Scholar]
- Campagna, R. FCC Network Editor, Refining Process Servicesn.C.. Personal Communication, 10 December 2012.
- Goonan, T.G. Rare Earth Elements-End use and Reyclability; Scientific Investigations Report 2011–5094; U.S. Geological Survey; U.S. Geological Survey; U.S. Department of the Interior: Reston, VA, USA, 2011. [Google Scholar]
- Ismall, S. Fluid Catalytic Cracking (FCC) Catalyst Optimization to Cope with High Rare Earth Oxide Price Environment; Technical Note; BASF: Middlesex/Essex, UK, 2011. [Google Scholar]
- Young, G.H.; Cowfer, J.A.; Johnston, V.J. Katalysator und Verfahren zu Oxychlorierung von Ethylen in Dichlorethan. EP0582165 B1, 21 May 1997. (in German). [Google Scholar]
- Auer, A.; Treibacher Industrie AG, Althofen, Austria. Personal Communication, 13 December 2012.
- EID. EID Daten und Fakten Energiepreise und Statistiken zum Download|Energie Informationsdienst. 2012. (in German). Available online: http://www.eid-aktuell.de/inhalt/statistiken/raffineriedurchsatz/ (accessed on 18 December 2012).
- Jahresbericht Mineralöl-Zahlen 2011; Mineralölwirtschaftsverband e.V.: Berlin, German, 2012. (in German)
- Shotyk, W.; Krachler, M. Contamination of bottled waters with antimony leaching from polyethylene terephthalate (PET) Increases upon storage. Environ. Sci. Technol. 2007, 41, 1560–1563. [Google Scholar] [CrossRef]
- Thiele, U. Quo vadis polyester catalyst? Chem. Fibers Int. 2004, 54, 162–163. [Google Scholar]
- Landesanstalt für Umwelt, M.u.N.B.-W.(Abfallsteckbriefe-1608 Gebrauchte Katalysatoren, Informationsportal zur Abfallbewertung–IPA. 2012. (in German). Available online: http://www.abfallbewertung.org/repgen.php?report=ipa&char_id=1608_Kat&lang_id=de&avv=&synon=&kapitel=2 (accessed on 10 April 2013).
- Kauertz, B.; Wellenreuther, F.; Busch, S.; Krüger, M.; Detzel, A. Ökobilanz der Glas- und PET-Mehrwegflaschen der GDB im Vergleich zu PET-Einwegflaschen. Studie im Auftrag der Genossenschaft Deutscher Brunnen eG; PGenossenschaft Deutscher Brunnen eG: Heidelberg, Germany, 2008. (in German) [Google Scholar]
- Kauertz, B.; Döhner, A.; Detzel, A. Ökobilanz von Getränkeverpackungen in Österreich Sachstand 2010; IFEU-Institut: Heidelberg, Germany, 2011. (in German) [Google Scholar]
- Heinisch, J. Verbrauch von Getränken in Einweg- und Mehrweg-Verpackungen: Berichtsjahr 2009; UBA-Texte 37/2011; Umweltbundesamt: Dessau-Roßlau, Germany, 2011. (in German) [Google Scholar]
- Umweltbundesamt. Trend zu Einwegflaschen aus Kunststoff Ungebrochen. Presse-Information 031/2011; Umweltbundesamt: Dessau-Roßlau, Germany, 2011. (in German) [Google Scholar]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).