Distribution of Phosphorus Forms Depends on Compost Source Material
Abstract
:1. Introduction
2. Materials and Method
2.1. Preparations of Composts
2.2. Chemical Analysis
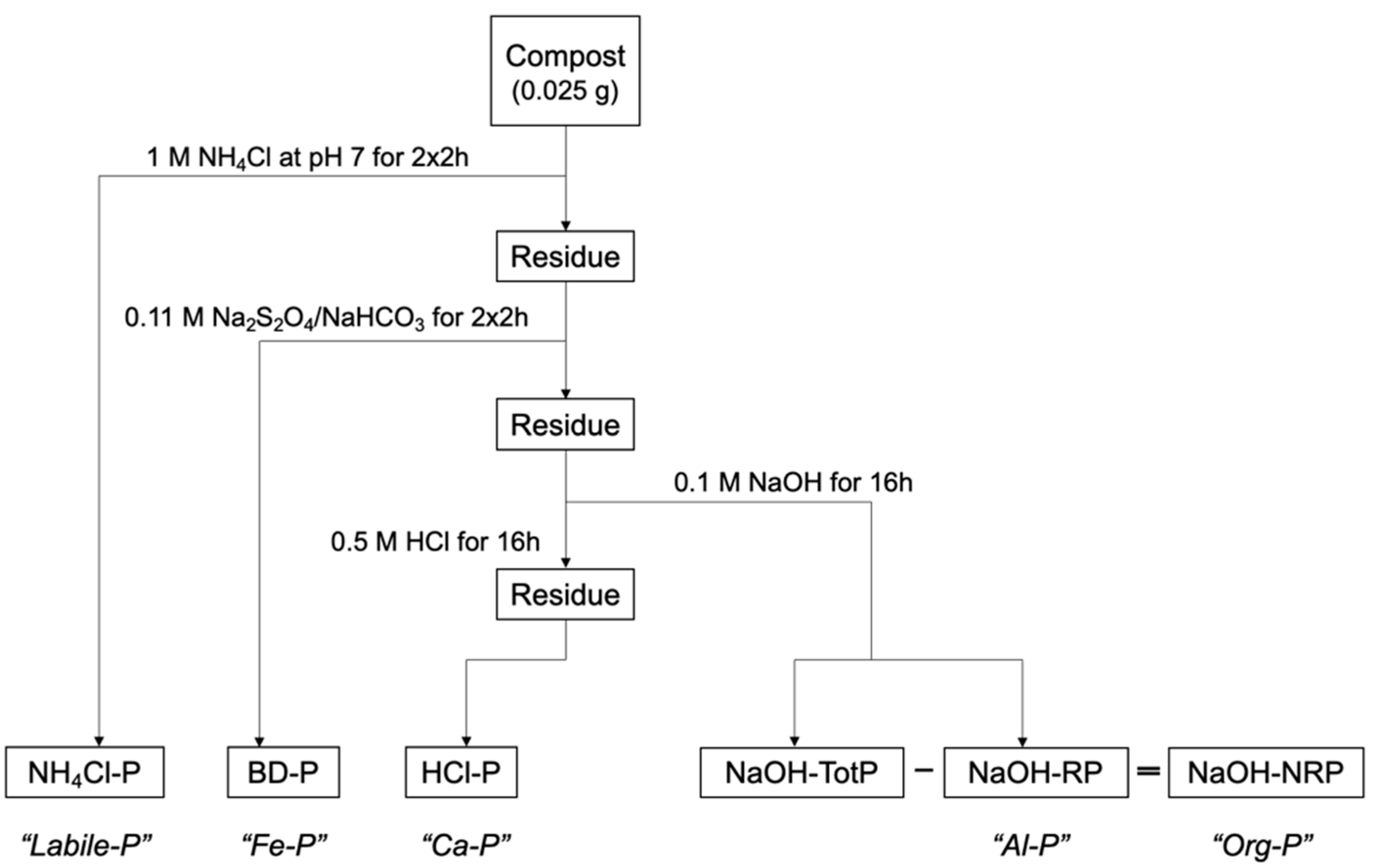
2.3. Determination of P Forms
- NH4Cl-RP—“Labile P”, immediately available P, which is loosely bound;
- BD-RP P—“Fe-P”, bound to reducible metals (Fe, Mn);
- NaOH-RP—“Al-P”, phosphates absorbed to metals (Al, MxOy);
- NaOH-NRP—“Org-P”, bound to organic material and colloids, determined as difference between soluble reactive P and total P in NaOH extract; and
- HCl-RP—“Ca-P”, bound to calcium (Ca).
2.4. Statistical Analysis
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Bajwa, A.A.; Khan, M.J.; Bhowmik, P.C.; Walsh, M.; Chauhan, B.S. Sustainable Weed Management. In Innovations in Sustainable Agriculture; Farooq, M., Pisante, M., Eds.; Springer: Cham, Switzerland, 2019; ISBN 978-3-030-23168-2. [Google Scholar] [CrossRef]
- Holford, I.C.R. Soil phosphorus: Its measurement, and its uptake by plants. Soil Res. 1997, 35, 227. [Google Scholar] [CrossRef]
- Driver, J.; Lijmbach, D.; Steen, I. Why Recover Phosphorus for Recycling, and How? Environ. Technol. 1999, 20, 651–662. [Google Scholar] [CrossRef]
- Savci, S. Investigation of Effect of Chemical Fertilizers on Environment. APCBEE Procedia 2012, 1, 287–292. [Google Scholar] [CrossRef] [Green Version]
- Massah, J.; Azadegan, B. Effect of Chemical Fertilizers on Soil Compaction and Degradation. Agric. Mech. Asia, Afr. Lat. Am. 2016, 47, 44–50. [Google Scholar]
- Rittmann, B.E.; Mayer, B.; Westerhoff, P.; Edwards, M. Capturing the lost phosphorus. Chemosphere 2011, 84, 846–853. [Google Scholar] [CrossRef] [PubMed]
- European Comission. Communication from the Commission to the European Parliament, the Council, the European Economic and Social Committee and the Committee of the Regions. A New Circular Economy Action Plan. For a Cleaner and More Competitive Europe. COM/2020/98 final. European Comission: Brussels, Belgium, 2020; p. 20. Available online: https://eur-lex.europa.eu/legal-content/EN/TXT/?qid=1583933814386&uri=COM:2020:98:FIN (accessed on 7 October 2021).
- Withers, P.J.A.; Van Dijk, K.C.; Neset, T.-S.S.; Nesme, T.; Oenema, O.; Rubæk, G.H.; Schoumans, O.F.; Smit, B.; Pellerin, S. Stewardship to tackle global phosphorus inefficiency: The case of Europe. AMBIO 2015, 44, 193–206. [Google Scholar] [CrossRef] [Green Version]
- Powers, S.M.; Chowdhury, R.; Macdonald, G.K.; Metson, G.S.; Beusen, A.H.W.; Bouwman, A.F.; Hampton, S.E.; Mayer, B.K.; McCrackin, M.L.; Vaccari, D.A. Global Opportunities to Increase Agricultural Independence Through Phosphorus Recycling. Earth’s Futur. 2019, 7, 370–383. [Google Scholar] [CrossRef] [Green Version]
- Mayer, B.K.; Baker, L.A.; Boyer, T.H.; Drechsel, P.; Gifford, J.; Hanjra, M.A.; Parameswaran, P.; Stoltzfus, J.; Westerhoff, P.; Rittmann, B.E. Total Value of Phosphorus Recovery. Environ. Sci. Technol. 2016, 50, 6606–6620. [Google Scholar] [CrossRef]
- European Environment Agency. Bio-Waste—Mainly Food and Garden Waste—Is a Key Waste Stream with a High Potential for Contributing to a More Circular Economy. This Report Provides an Overview of Bio-Waste Generation, Prevention, Collection, and Treatment in Europe; European Environment Agency: Copenhagen, Denmark, 2020. [Google Scholar] [CrossRef]
- European Parliament. Regulation (EU) 2019/1009 of the European Parliament and of the Council of 5 June 2019 Laying down Rules on the Making Available on the Market of EU Fertilising Products and Amending Regulations (EC) No 1069/2009 and (EC) No 1107/2009 and Repealing Regulation (EC) No 2003/2003; EUR-Lex: Luxembourg, Luxembourg, 2019; Available online: http://data.europa.eu/eli/reg/2019/1009/oj (accessed on 7 October 2021).
- Sánchez, J.; Ospina, D.A.; Montoya, S. Compost supplementation with nutrients and microorganisms in composting process. Waste Manag. 2017, 69, 136–153. [Google Scholar] [CrossRef]
- Busato, J.G.; Lima, L.S.; Aguiar, N.O.; Canellas, L.P.; Olivares, F.L. Changes in labile phosphorus forms during maturation of vermicompost enriched with phosphorus-solubilizing and diazotrophic bacteria. Bioresour. Technol. 2012, 110, 390–395. [Google Scholar] [CrossRef] [Green Version]
- Ngo, P.-T.; Rumpel, C.; Ngo, Q.-A.; Alexis, M.; Vargas, G.V.; Gil, M.D.L.L.M.; Dang, D.-K.; Jouquet, P. Biological and chemical reactivity and phosphorus forms of buffalo manure compost, vermicompost and their mixture with biochar. Bioresour. Technol. 2013, 148, 401–407. [Google Scholar] [CrossRef]
- Wei, Y.; Zhao, Y.; Xi, B.; Wei, Z.; Li, X.; Cao, Z. Changes in phosphorus fractions during organic wastes composting from different sources. Bioresour. Technol. 2015, 189, 349–356. [Google Scholar] [CrossRef]
- Gagnon, B.; Demers, I.; Ziadi, N.; Chantigny, M.H.; Parent, L.; Forge, T.A.; Larney, F.J.; Buckley, K.E. Forms of phosphorus in composts and in compost-amended soils following incubation. Can. J. Soil Sci. 2012, 92, 711–721. [Google Scholar] [CrossRef] [Green Version]
- Shen, J.; Yuan, L.; Zhang, J.; Li, H.; Bai, Z.; Chen, X.; Zhang, W.; Zhang, F. Phosphorus Dynamics: From Soil to Plant. Plant Physiol. 2011, 156, 997–1005. [Google Scholar] [CrossRef] [Green Version]
- Naidu, R.; Bolan, N.S.; Megharaj, M.; Juhasz, A.L.; Gupta, S.K.; Clothier, B.E.; Schulin, R. Chemical Bioavailability in Terrestrial Environments. In Developments in Soil Science; Elsevier: Amsterdam, The Netherlands, 2008; ISBN 978-0-444-52169-9. [Google Scholar]
- Jakubus, M. Estimation of phosphorus bioavailability from composted organic wastes. Chem. Speciat. Bioavailab. 2016, 28, 189–198. [Google Scholar] [CrossRef] [Green Version]
- Siebert, S.; Auweele, W.V. ECN-Qas European Quality Assurance Scheme for Compost and Digestate; European Compost Network ECN e.V.: Bochum, Germany, 2014; ISBN 978-3-00-047599-3. [Google Scholar]
- Regulation of the Republic of Estonia. Biolagunevatest Jäätmetest Komposti Tootmise Nõuded. RT I, 18.12.2020, 23; Keskkonnaminister: Tallinn, Estonia, 2021. [Google Scholar]
- Sharpley, A.; Moyer, B. Phosphorus Forms in Manure and Compost and Their Release during Simulated Rainfall. J. Environ. Qual. 2000, 29, 1462–1469. [Google Scholar] [CrossRef]
- European Parliament. Commission Regulation (EU) No 142/2011 of 25 February 2011 Implementing Regulation (EC) No 1069/2009 of the European Parliament and of the Council Laying down Health Rules as Regards Animal By-Products and Derived Products Not Intended for Human Consumption and Implementing Council Directive 97/78/EC as Regards Certain Samples and Items Exempt from Veterinary Checks at the Border under That Directive (Text with EEA Relevance); EUR-Lex: Luxembourg, Luxembourg, 2011; Available online: http://data.europa.eu/eli/reg/2011/142/oj (accessed on 7 October 2021).
- Regulation of the Republic of Estonia. Haljastuses, Rekultiveerimisel Ja Põllumajanduses Kasutatava Reoveesette Kvaliteedi Piirväärtused Ning Kasutamise Nõuded. RT I, 06.08.2019, 7; Keskkonnaminister: Tallinn, Estonia, 2019. [Google Scholar]
- Egner, H.; Riehm, H.; Domingo, W.R. Investigations on the Chemical Soil Analysis as a Basis for Assessing the Soil Nutrient Status. II: Chemical Extraction Methods for Phosphorus and Potassium Determination. K. Lantbr. Ann. 1960, 26, 199–215. [Google Scholar]
- Rydin, E. Potentially mobile phosphorus in Lake Erken sediment. Water Res. 2000, 34, 2037–2042. [Google Scholar] [CrossRef]
- Murphy, J.; Riley, J. A modified single solution method for the determination of phosphate in natural waters. Anal. Chim. Acta 1962, 27, 31–36. [Google Scholar] [CrossRef]
- R Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2020. [Google Scholar]
- Mendiburu, F.D. Agricolae: Statistical Procedures for Agricultural Research. R Package Version 1.2-3. 2015. [Google Scholar]
- Rydin, E.; Otabbong, E. Potential Release of Phosphorus from Soil Mixed with Sewage Sludge. J. Environ. Qual. 1997, 26, 529–534. [Google Scholar] [CrossRef]
- Blake, L.; Johnston, A.; Poulton, P.; Goulding, K. Changes in soil phosphorus fractions following positive and negative phosphorus balances for long periods. Plant Soil 2003, 254, 245–261. [Google Scholar] [CrossRef]
- Carter, M.R.; Gregorich, E.G. Soil Sampling and Methods of Analysis; CRC Press: Boca Raton, FL, USA, 2007. [Google Scholar]
- Radziemska, M.; Vaverková, M.D.; Adamcová, D.; Brtnický, M.; Mazur, Z. Valorization of Fish Waste Compost as a Fertilizer for Agricultural Use. Waste Biomass-Valorization 2019, 10, 2537–2545. [Google Scholar] [CrossRef] [Green Version]
- Illera-Vives, M.; Labandeira, S.S.; Loureiro, L.I.; López-Mosquera, M.E. Agronomic assessment of a compost consisting of seaweed and fish waste as an organic fertilizer for organic potato crops. Environ. Boil. Fishes 2017, 29, 1663–1671. [Google Scholar] [CrossRef]
- Ahuja, I.; Dauksas, E.; Remme, J.F.; Richardsen, R.; Løes, A.-K. Fish and fish waste-based fertilizers in organic farming—With status in Norway: A review. Waste Manag. 2020, 115, 95–112. [Google Scholar] [CrossRef] [PubMed]
- Dantas, M.C.; Attayde, J.L. Nitrogen and phosphorus content of some temperate and tropical freshwater fishes. J. Fish Biol. 2007, 70, 100–108. [Google Scholar] [CrossRef]
- Ramadhani, S.; Iswanto, B.; Purwaningrum, P. Waste utilization of red snapper (Lutjanus sp.) fish bone to improve phosphorus contents in compost. IOP Conf. Series: Earth Environ. Sci. 2018, 106, 12091. [Google Scholar] [CrossRef]
- Yan, X.; Yang, W.; Chen, X.; Wang, M.; Wang, W.; Ye, D.; Wu, L. Soil Phosphorus Pools, Bioavailability and Environmental Risk in Response to the Phosphorus Supply in the Red Soil of Southern China. Int. J. Environ. Res. Public Health 2020, 17, 7384. [Google Scholar] [CrossRef]
- Ahuja, I.; Løes, A.K. Effect of Fish Bones and Algae Fibre as Fertilisers for Ryegrass; NORSØK: Tingvoll, Norway, 2019; pp. 1–64. [Google Scholar]
- Zhang, L.; Sun, X. Addition of fish pond sediment and rock phosphate enhances the composting of green waste. Bioresour. Technol. 2017, 233, 116–126. [Google Scholar] [CrossRef]
- Saleque, M.A.; Naher, U.A.; Islam, A.; Pathan, A.B.M.B.U.; Hossain, A.T.M.S.; Meisner, C.A. Inorganic and Organic Phosphorus Fertilizer Effects on the Phosphorus Fractionation in Wetland Rice Soils. Soil Sci. Soc. Am. J. 2004, 68, 1635–1644. [Google Scholar] [CrossRef]
- Syers, J.K.; Johnston, A.E.; Curtin, D. Efficiency of Soil and Fertilizer Phosphorus Use: Reconciling Changing Concepts of Soil Phosphorus Behaviour with Agronomic Information; FAO Fertilizer and Plant Nutrition Bulletin; Food and Agriculture Organization of the United Nations: Rome, Italy, 2008; ISBN 978-92-5-105929-6. [Google Scholar]
- Council Directive of 12 June 1986 on the Protection of the Environment, and in Particular of the Soil, When Sewage Sludge Is Used in Agriculture (86/278/EEC). EUR-Lex: Luxembourg, 2018. Available online: http://data.europa.eu/eli/dir/1986/278/2018-07-04 (accessed on 7 October 2021).
- Tomócsik, A.; Makádi, M.; Orosz, V.; Aranyos, T.; Demeter, I.; Mészáros, J.; Füleky, G. Effect of sewage sludge compost treatment on crop yield. AGROFOR 2016, 1. [Google Scholar] [CrossRef]
- Elsalam, H.; Sharnouby, M.E.; Mohamed, A.; Raafat, B.; El-Gamal, E. Effect of Sewage Sludge Compost Usage on Corn and Faba Bean Growth, Carbon and Nitrogen Forms in Plants and Soil. Agronomy 2021, 11, 628. [Google Scholar] [CrossRef]
- Eid, E.M.; Alamri, S.A.; Shaltout, K.H.; Galal, T.M.; Ahmed, M.T.; Brima, E.I.; Sewelam, N. A sustainable food security approach: Controlled land application of sewage sludge recirculates nutrients to agricultural soils and enhances crop productivity. Food Energy Secur. 2020, 9. [Google Scholar] [CrossRef]
- Meena, M.; Narjary, B.; Sheoran, P.; Jat, H.; Joshi, P.; Chinchmalatpure, A.; Yadav, G.; Yadav, R. Changes of phosphorus fractions in saline soil amended with municipal solid waste compost and mineral fertilizers in a mustard-pearl millet cropping system. CATENA 2018, 160, 32–40. [Google Scholar] [CrossRef]

| Treatment Name | Source of Waste | Amendments | Active Composting Time (d) |
|---|---|---|---|
| FW1 | Fish waste (80%) | Un-shredded straw (20%) | 185 |
| FW2 | Fish waste (57%) | Shredded straw (43%), | 70 |
| FW3 | Fish waste (22%) | Shredded straw (22%), peat (22%), inoculum from mature HM (21%) and FW1 compost (13%) | 50 |
| HM | Horse manure (60%) | Straw (40%) | 90 |
| GW | Green waste (100%) | None | >180 |
| SS | Sewage sludge (90%) | Un-shredded straw (10%) | 120 |
| Treatment | N | C | C/N | K | Ca | Mg | Dry Matter | Loss of Ignition | Stability |
|---|---|---|---|---|---|---|---|---|---|
| (%) | (mg/100g) | (%) | (mg O2/g) | ||||||
| FW1 | 2.43 | 30.22 | 12.44 | 790.51 | 4471.04 | 703.31 | 53.80 | 75.40 | 3.00 |
| FW2 | 2.75 | 33.04 | 12.01 | 1329.22 | 4625.55 | 664.00 | 47.94 | 85.78 | 11.20 |
| FW3 | 2.73 | 35.66 | 13.06 | 1675.63 | 3166.32 | 680.48 | 34.79 | 87.05 | 10.40 |
| HM | 2.47 | 30.99 | 12.55 | 2550.90 | 2986.40 | 649.17 | 37.83 | 79.67 | 3.20 |
| GW | 0.60 | 13.35 | 22.25 | 552.08 | 6681.84 | 223.96 | 41.78 | 65.46 | 0.90 |
| SS | 3.74 | 27.02 | 7.22 | 551.81 | 1169.69 | 517.71 | 44.16 | 61.34 | 6.70 |
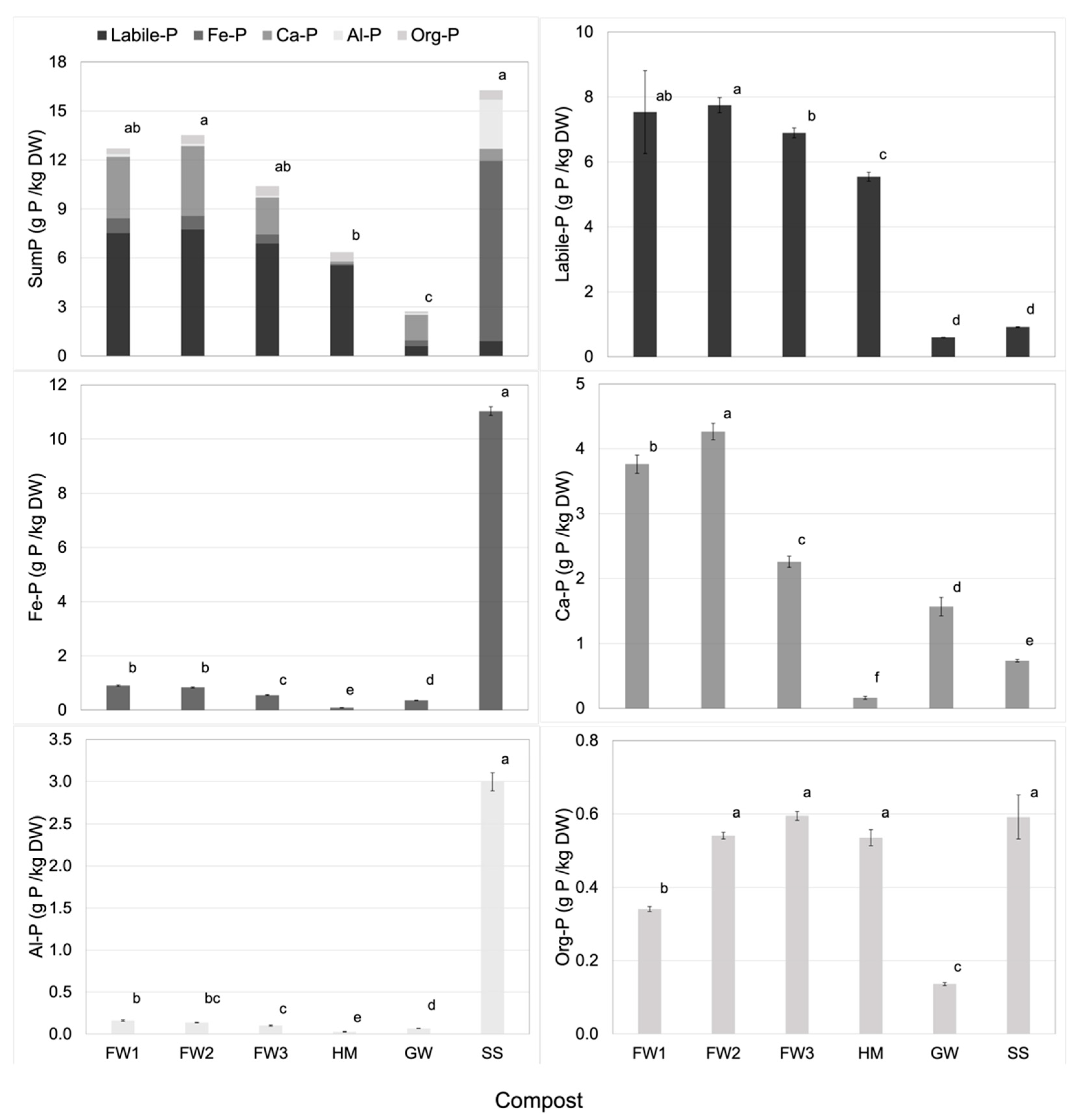
| Compost | Labile-P | Fe-P | Ca-P | Al-P | Org-P | SumP |
|---|---|---|---|---|---|---|
| FW1 | 7.53 | 0.90 | 3.76 | 0.16 | 0.34 | 12.70 |
| FW2 | 7.75 | 0.83 | 4.27 | 0.14 | 0.54 | 13.53 |
| FW3 | 6.90 | 0.55 | 2.26 | 0.10 | 0.59 | 10.40 |
| HM | 5.55 | 0.09 | 0.16 | 0.03 | 0.54 | 6.36 |
| GW | 0.60 | 0.35 | 1.57 | 0.07 | 0.14 | 2.73 |
| SS | 0.92 | 11.03 | 0.74 | 3.00 | 0.59 | 16.27 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lanno, M.; Kriipsalu, M.; Shanskiy, M.; Silm, M.; Kisand, A. Distribution of Phosphorus Forms Depends on Compost Source Material. Resources 2021, 10, 102. https://doi.org/10.3390/resources10100102
Lanno M, Kriipsalu M, Shanskiy M, Silm M, Kisand A. Distribution of Phosphorus Forms Depends on Compost Source Material. Resources. 2021; 10(10):102. https://doi.org/10.3390/resources10100102
Chicago/Turabian StyleLanno, Marge, Mait Kriipsalu, Merrit Shanskiy, Maidu Silm, and Anu Kisand. 2021. "Distribution of Phosphorus Forms Depends on Compost Source Material" Resources 10, no. 10: 102. https://doi.org/10.3390/resources10100102
APA StyleLanno, M., Kriipsalu, M., Shanskiy, M., Silm, M., & Kisand, A. (2021). Distribution of Phosphorus Forms Depends on Compost Source Material. Resources, 10(10), 102. https://doi.org/10.3390/resources10100102







