Noninvasive and Safe Cell Viability Assay for Breast Cancer MCF-7 Cells Using Natural Food Pigment
Abstract
1. Introduction
- Dye exclusion test (DET) Cells stained with a synthetic dye such as trypan blue (TB) are judged as dead cells [3].
- Colony formation assay The number of live cells is evaluated by the number of colonies formed on an agar culture after an inoculation of diluted cell suspension and following a definite time of culture [4].
- Enzyme activity assay Enzymatic reaction in living cells or enzymes leaking out of dead cells are used for viability assay [5].
- Optical method The dead or living state of cells is diagnosed by deflection change of the probe light beam [9].
- Electro-orientation-based assay The positions of alternative electro-orientation of living or dead cylindrical yeast cells are dependent on the applied frequency [10].
2. Materials and Methods
2.1. Sample Preparation
2.2. Bright-Field Observation of Cells
- A fluorescence microscope, BZ-9000;
- A 4× objective lens, NA 0.20, CFI Plan Apochromat Lambda 4X, Nikon;
- A 20× objective lens, NA 0.75, CFI Plan Apochromat Lambda 20X, Nikon;
- Exposure time, 1/900 sec (4× objective lens, Figure 1);
- Exposure time, 1/130 sec (20× objective lens, Figure 2);
- Exposure time, 1/80 sec (20× objective lens, Figure 3);
- 4× objective lens, 1/800 sec or 1/900 sec.
2.3. Preparation of Dead Cells of MCF-7 Cells (Common for All Experiments)
- Microwave (MW) treatment Cells were treated with microwave at 2.45 GHz until it was boiled.
- Cisplatin treatment Cisplatin (Wako) was added to the culture and adjusted to a final concentration of 20 μM. According to the report, the survival rate of MCF-7 cells at 20 μM of cisplatin was estimated to be about 80% [35]. In this report, the relationship between survival rate and cisplatin concentration was 85.67% (12 μM) and 76.03% (25 μM) (Figure 1B in reference [35], not in this paper).
2.4. Pigment for Viability Assay
- Natural food pigment Food pigment Red (Monascus, Watashinodaidokoro Co., Ltd. http://watashinodaidokoro.net/);
- Synthetic dye Trypan blue (Merck KGaA).
2.5. Confirmation of Staining by Natural Pigment
2.6. Magnification of Observation and Staining of Dead Cells
2.7. Confirmation of Staining of Dead Cells
2.8. Immersion Time and Staining of Dead Cells
2.9. Staining of Cells in A Mixed Solution of MP and Cisplatin
2.10. Confirmation of Noninvasiveness of Pigment
2.11. Cytotoxicity Assay (WST-8)
3. Results
3.1. Confirmation of Staining by Natural Pigment
3.2. Magnification of Observation and Staining of Dead Cells
3.3. Confirmation of Staining of Dead Cells
3.4. Immersion Time and Staining of Dead Cells
3.5. Staining of Cells in a Mixed Solution of MP and Cisplatin
3.6. Confirmation of Noninvasiveness of Pigment
3.7. Immersion Time and Staining of Dead Cells
4. Discussion
5. Conclusions
6. Patents
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Moleiro, A.F.; Conceição, G.; Leite-Moreira, A.F.; Rocha-Sousa, A. A Critical Analysis of the Available In Vitro and Ex Vivo Methods to Study Retinal Angiogenesis. J. Ophthalmol. 2017, 2017. [Google Scholar] [CrossRef] [PubMed]
- Aslantürk, Ö.S. In Vitro Cytotoxicity and Cell Viability Assays: Principles, Advantages, and Disadvantages. In Genotoxicity—A Predictable Risk to Our Actual World; IntechOpen: London, UK, 2017. [Google Scholar] [CrossRef]
- Bonora, A.; Mares, D. A simple colorimetric method for detecting cell viability in cultures of eukaryotic microorganisms. Curr. Microbiol. 1982, 7, 217–221. [Google Scholar] [CrossRef]
- Collins, C.H.; Christopher, H.; Lyne, P.M. Microbiological Methods, 5th ed.; rev. reprint; Butterworths: London, UK, 1985. [Google Scholar]
- Kaja, S.; Payne, A.J.; Naumchuk, Y.; Koulen, P. Quantification of lactate dehydrogenase for cell viability testing using cell lines and primary cultured astrocytes. Curr. Protoc. Toxicol. 2017, 72, 2–26. [Google Scholar] [CrossRef] [PubMed]
- Hamalainen-Laanaya, H.K.; Orloff, M.S. Analysis of cell viability using time-dependent increase in fluorescence intensity. Anal. Biochem. 2012, 429, 32–38. [Google Scholar] [CrossRef]
- Yamada, K.; Suzuki, H.; Takeuchi, T.; Kazama, Y.; Mitra, S.; Abe, T.; Goda, K.; Suzuki, K.; Iwata, O. Efficient selective breeding of live oil-rich Euglena gracilis with fluorescence-activated cell sorting. Sci. Rep. 2016, 6, 26327. [Google Scholar] [CrossRef]
- Iwata, O.; Yamada, K.; Itou, T.; Ozeki, Y.; Suzuki, K.; Goda, K. Technology for Developing Super Microalgal Biofuels. Seibutsu Butsuri 2017, 57, 235–239. [Google Scholar] [CrossRef][Green Version]
- Wu, X.Z.; Terada, S. Noninvasive diagnosis of a single cell with a probe beam. Biotechnol. Prog. 2005, 21, 1772–1774. [Google Scholar] [CrossRef]
- Suga, M.; Kunimoto, A.; Shinohara, H. Non-invasive, electro-orientation-based viability assay using optically transparent electrodes for individual fission yeast cells. Biosens. Bioelectron. 2017, 97, 53–58. [Google Scholar] [CrossRef]
- Cells Detected, Alive or Dead. 2013. Available online: https://www.biocompare.com/Editorial-Articles/131962-Cells-Detected-Alive-or-Dead/ (accessed on 19 July 2018).
- Trypan Blue—An overview Science Direct Topics. Available online: https://www.sciencedirect.com/topics/biochemistry-genetics-and-molecular-biology/trypan-blue (accessed on 13 December 2018).
- Tran, S.L.; Puhar, A.; Ngo-Camus, M.; Ramarao, N. Trypan Blue Dye Enters Viable Cells Incubated with the Pore-Forming Toxin HlyII of Bacillus cereus. PLoS ONE 2011, 6, e22876. [Google Scholar] [CrossRef]
- Strober, W. Trypan Blue Exclusion Test of Cell Viability. Curr. Protoc. Immunol. 2015, 111, A3.B.1–A3.B.3. [Google Scholar] [CrossRef]
- Beck, F.; Lloyd, J.B. Dosage—Response Curves for the Teratogenic Activity of Trypan Blue. Nature 1964, 201, 1136–1137. [Google Scholar] [CrossRef] [PubMed]
- Tsaousis, K.T.; Kopsachilis, N.; Tsinopoulos, I.T.; Dimitrakos, S.A.; Kruse, F.E.; Welge-Luessen, U. Time-dependent morphological alterations and viability of cultured human trabecular cells after exposure to Trypan blue. Clin. Exp. Ophthalmol. 2013, 41, 484–490. [Google Scholar] [CrossRef] [PubMed]
- Tampion, J.; Tampion, M.D. Immobilized Cells: Principles and Applications; Cambridge University Press: Cambridge, UK, 1987. [Google Scholar]
- Feizi, A.; Zhang, Y.; Greenbaum, A.; Guziak, A.; Luong, M.; Lok Chan, R.Y.; Berg, B.; Ozkan, H.; Luo, W.; Wu, M.; et al. Rapid, portable and cost-effective yeast cell viability and concentration analysis using lensfree on-chip microscopy and machine learning. Lab Chip 2016, 16, 4350–4358. [Google Scholar] [CrossRef] [PubMed]
- Schrek, R. A Method for Counting the Viable Cells in Normal and in Malignant Cell Suspensions. Am. J. Cancer 1936, 28, 389–392. [Google Scholar] [CrossRef]
- Scharff, T.G.; Maupin, W.C. Correlation of the metabolic effects of benzalkonium chloride with its membrane effects in yeast. Biochem. Pharmacol. 1960, 5, 79–86. [Google Scholar] [CrossRef]
- Novelli, A. Amethyst violet as a stain for distinguishing cells with a damaged membrane from normal cells. Experientia 1962, 18, 295–296. [Google Scholar] [CrossRef] [PubMed]
- Shimizu, T.; Nakamura, M.; Fuji, M. Edible Natural Pigment (New edition); Korin: Tokyo, Japan, 2001; ISBN 978-4-7712-0010-4. [Google Scholar]
- Kiriya Chemi: Monascus Colour. Available online: http://www.kiriya-chem.co.jp/tennen/koji_red.html (accessed on 16 October 2019).
- Chen, W.; Chen, R.; Liu, Q.; He, Y.; He, K.; Ding, X.; Kang, L.; Guo, X.; Xie, N.; Zhou, Y.; et al. Orange, red, yellow: Biosynthesis of azaphilone pigments in Monascus fungi. Chem Sci 2017, 8, 4917–4925. [Google Scholar] [CrossRef]
- Wang, T.; Lin, T. Monascus Rice Products. In Advances in Food and Nutrition Research; Academic Press: Waltham, MA, USA, 2007; Volume 53, pp. 123–159. [Google Scholar]
- Feng, Y.; Shao, Y.; Chen, F. Monascus pigments. Appl. Microbiol. Biotechnol. 2012, 96, 1421–1440. [Google Scholar] [CrossRef]
- Japan’s Specifications and Standards for Food Additives (D. Monographs). Available online: http://dfa-j/shokuten_kikaku_j.html (accessed on 29 April 2020).
- Yamashita, K.; Tokunaga, E. Noninvasive and safe cell viability assay for Paramecium using natural pigment extracted from food. Sci. Rep. 2020, 10, 10996. [Google Scholar] [CrossRef]
- Comşa, Ş.; Cîmpean, A.M.; Raica, M. The Story of MCF-7 Breast Cancer Cell Line: 40 years of Experience in Research. Anticancer. Res. 2015, 35, 3147–3154. [Google Scholar]
- Martinho, N.; Santos, T.C.B.; Florindo, H.F.; Silva, L.C. Cisplatin-Membrane Interactions and Their Influence on Platinum Complexes Activity and Toxicity. Front. Physiol. 2019, 9, 1898. [Google Scholar] [CrossRef] [PubMed]
- Prabhakaran, P.; Hassiotou, F.; Blancafort, P.; Filgueira, L. Cisplatin Induces Differentiation of Breast Cancer Cells. Front. Oncol. 2013, 3, 134. [Google Scholar] [CrossRef] [PubMed]
- Sweeney, E.E.; McDaniel, R.E.; Maximov, P.Y.; Fan, P.; Jordan, V.C. Models and mechanisms of acquired antihormone resistance in breast cancer: Significant clinical progress despite limitations. Horm. Mol. Biol. Clin. Investig. 2012, 9, 143–163. [Google Scholar] [CrossRef] [PubMed]
- Gianfredi, V.; Nucci, D.; Vannini, S.; Villarini, M.; Moretti, M. In vitro Biological Effects of Sulforaphane (SFN), Epigallocatechin-3-gallate (EGCG), and Curcumin on Breast Cancer Cells: A Systematic Review of the Literature. Nutr. Cancer 2017, 69, 969–978. [Google Scholar] [CrossRef] [PubMed]
- Yamashita, K.; Yamada, K.; Suzuki, K.; Tokunaga, E. Noninvasive and safe cell viability assay for Euglena gracilis using natural food pigment. PeerJ 2019, 7, e6636. [Google Scholar] [CrossRef]
- Al-Taweel, N.; Varghese, E.; Florea, A.M.; Büsselberg, D. Cisplatin (CDDP) triggers cell death of MCF-7 cells following disruption of intracellular calcium ([Ca2+]i) homeostasis. J. Toxicol. Sci. 2014, 39, 765–774. [Google Scholar] [CrossRef]
- Cell Counting Kit-8 (Technical Manual). Available online: https://www.dojindo.co.jp/manual/CK04e.pdf (accessed on 9 October 2019).
- Steinhusen, U.; Badock, V.; Bauer, A.; Behrens, J.; Wittman-Liebold, B.; Dörken, B.; Bommert, K. Apoptosis-induced Cleavage of β-Catenin by Caspase-3 Results in Proteolytic Fragments with Reduced Transactivation Potential. J. Biol. Chem. 2000, 275, 16345–16353. [Google Scholar] [CrossRef]
- Okubo, T.; Kano, I. Studies on estrogenic activities of food additives with human breast cancer MCF-7 cells and mechanism of estrogenicity by BHA and OPP. Yakugaku Zasshi J. Pharm. Soc. Jpn. 2003, 123, 443–452. [Google Scholar] [CrossRef]
- Kim, S.I.; Kim, H.J.; Lee, H.J.; Lee, K.; Hong, D.; Lim, H.; Cho, K.; Jung, N.; Yi, Y.W. Application of a non-hazardous vital dye for cell counting with automated cell counters. Anal. Biochem. 2016, 492, 8–12. [Google Scholar] [CrossRef]
- Jennings, A.S.; Schwartz, S.L.; Balter, N.J.; Gardner, D.; Witorsch, R.J. Effects of oral erythrosine (2′,4′,5′,7′-tetraiodofluorescein) on the pituitary-thyroid axis in rats. Toxicol. Appl. Pharmacol. 1990, 103, 549–556. [Google Scholar] [CrossRef]
- Ap, F.D.A. Limits Red Dye No. 3. The New York Times, 30 January 1990. [Google Scholar]
- Food Dyes: Harmless or Harmful? Available online: https://www.healthline.com/nutrition/food-dyes (accessed on 4 August 2018).
- PubChem Erythrosin B (COMPOUND SUMMARY). Available online: https://pubchem.ncbi.nlm.nih.gov/compound/3259 (accessed on 20 October 2019).
- Mizoguchi, A.; Fujiwara, T.; Tanaka, K.; Wang, S.; Sai, K.; Kimura, K. Vital Stain. European Patent EP 14773865.2, 28 March 2014. [Google Scholar]


| Experimental Item | Table/Figure Number | Incubation Period (Before Pigment Addition) | Incubation Period (After Pigment Addition) |
|---|---|---|---|
| Confirmation of staining by natural pigment | Table S3, Supplementary Material | 2 day | 12 h |
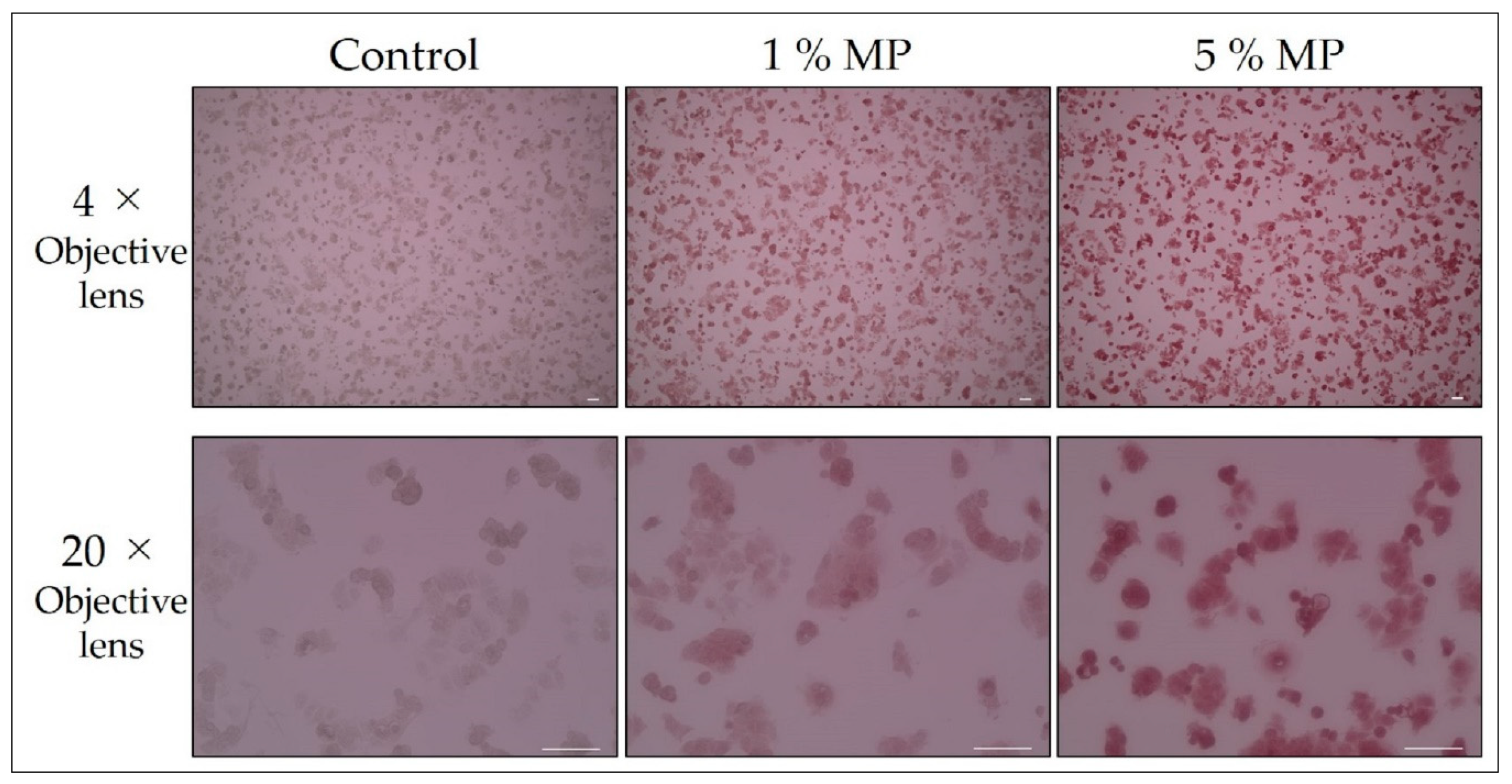
| Magnification of observation and staining of dead cells | Figure 1 | 2 day | 20 min |
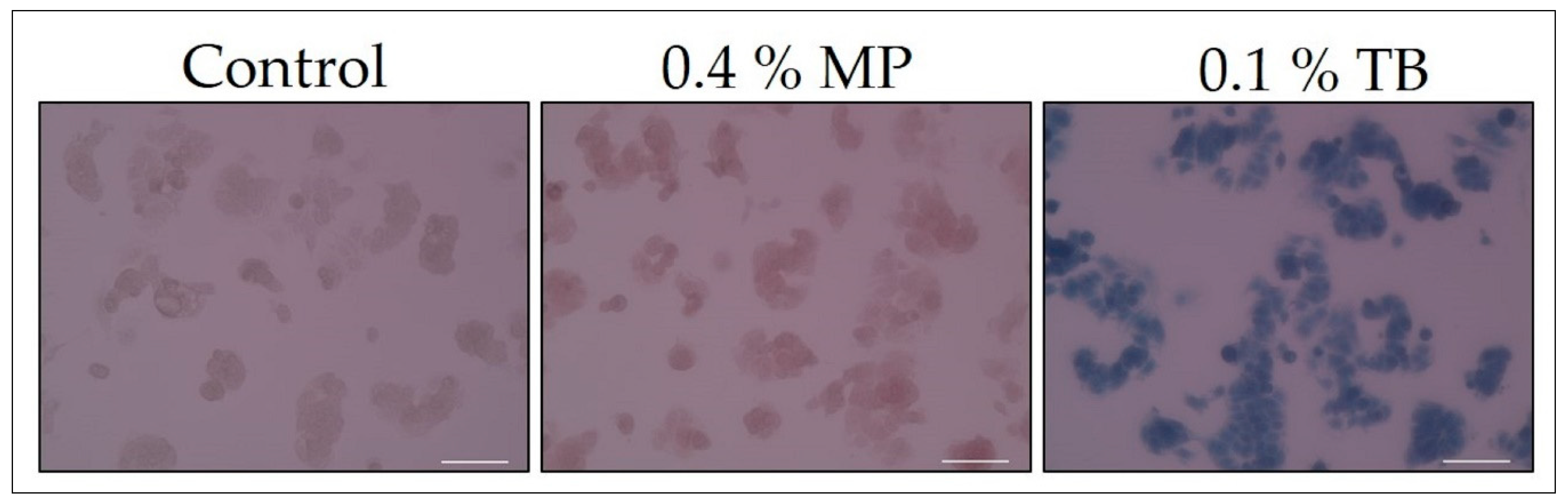
| Confirmation of staining of dead cells | Figure 2 | 2 day | 20 min |
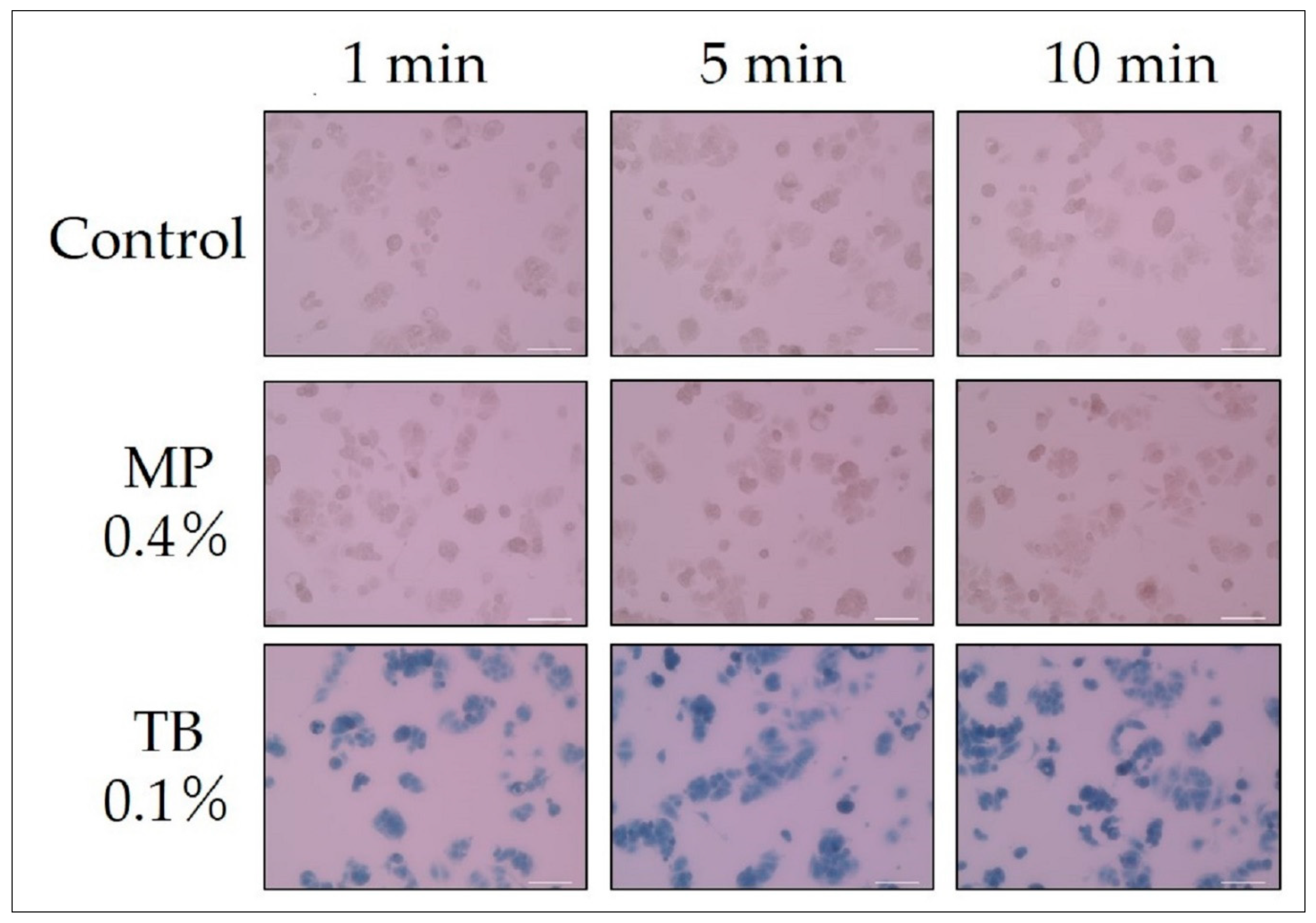
| Immersion time and staining of dead cells | Figure 3 | 2 day | 1 min, 5 min, 10 min |
| Staining of cells in a mixed solution of MP and cisplatin | Figure 4 | 2 day | 14 h |
| Confirmation of noninvasiveness of pigment | Figure 5 | 2 day | 47 h |
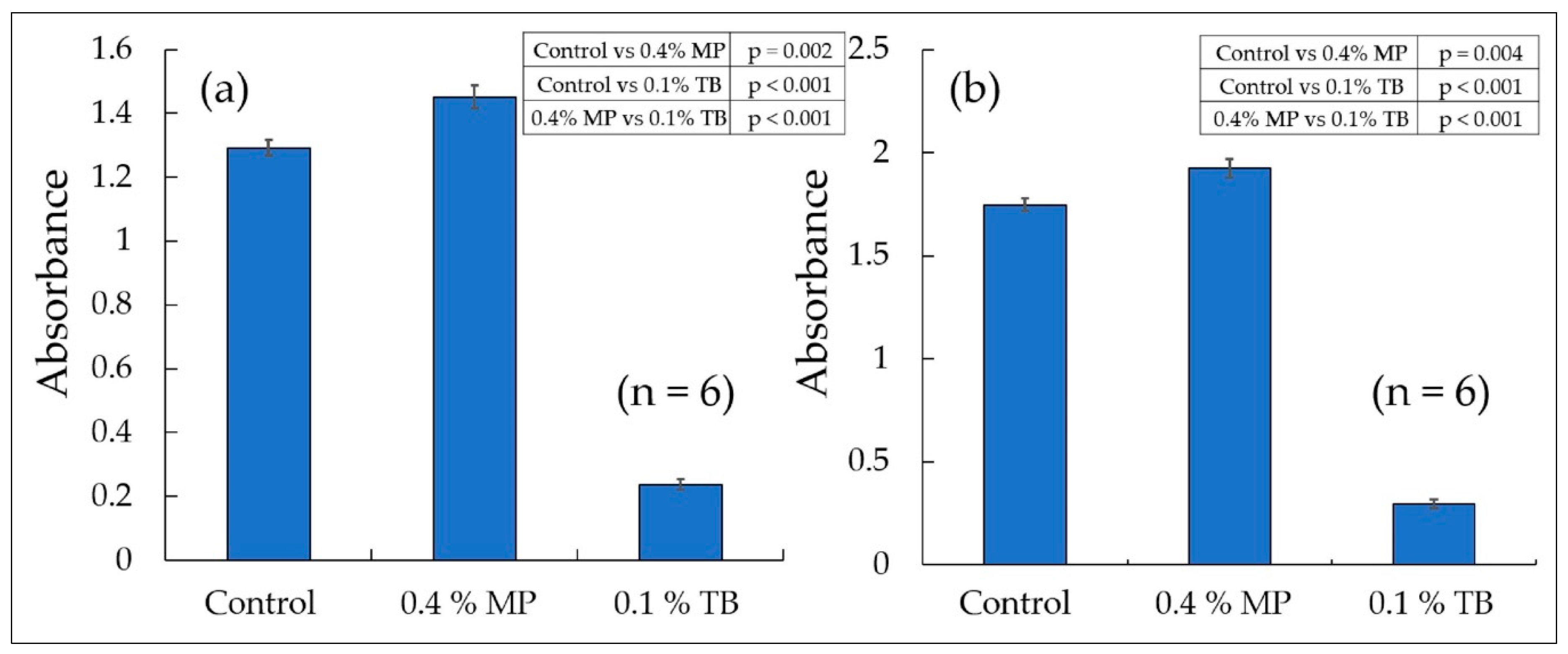
| Cytotoxicity assay (WST-8 *), Tukey test | Figure 6 | 2 day | 24 h, 48 h |
| Experimental Item | Table/Figure Number | Pigment/Dye Concentration (%) | Cisplatin (μM) | Objective Lens | ||
|---|---|---|---|---|---|---|
| Control | MP | TB | ||||
| Confirmation of staining by natural pigment | Table 3 | - | 1 ※ | - | 0 | 4×/20× |
| Magnification of observation and staining of dead cells | Figure 1 | 0 | 1, 5 | - | 0 | 4×/20× |
| Confirmation of staining of dead cells | Figure 2 | 0 | 0.4 | 0.1 | 0 | 20× |
| Immersion time and staining of dead cells | Figure 3 | 0 | 0.4 | 0.1 | 0 | 20× |
| Staining of cells in a mixed solution of MP and cisplatin | Figure 4 | 0 | 1, 5 | - | 20 | 20× |
| Confirmation of noninvasiveness of pigment | Figure 5 | 0 | 0.4 | 0.1 | 0 | 20× |
| Cytotoxicity assay (WST-8) | Figure 6 | 0 | 0.4 | 0.1 | 0 | - |
| Natural Food Pigment | Synthetic Dye | |||||
|---|---|---|---|---|---|---|
| Monascus | Purple sweet potato | Yellow gardenia | Green gardenia | Red beet | Spirulina | Trypan blue |
| ◯ | × | × | × | × | × | ◯ |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yamashita, K.; Tagawa, R.; Higami, Y.; Tokunaga, E. Noninvasive and Safe Cell Viability Assay for Breast Cancer MCF-7 Cells Using Natural Food Pigment. Biology 2020, 9, 227. https://doi.org/10.3390/biology9080227
Yamashita K, Tagawa R, Higami Y, Tokunaga E. Noninvasive and Safe Cell Viability Assay for Breast Cancer MCF-7 Cells Using Natural Food Pigment. Biology. 2020; 9(8):227. https://doi.org/10.3390/biology9080227
Chicago/Turabian StyleYamashita, Kyohei, Ryoma Tagawa, Yoshikazu Higami, and Eiji Tokunaga. 2020. "Noninvasive and Safe Cell Viability Assay for Breast Cancer MCF-7 Cells Using Natural Food Pigment" Biology 9, no. 8: 227. https://doi.org/10.3390/biology9080227
APA StyleYamashita, K., Tagawa, R., Higami, Y., & Tokunaga, E. (2020). Noninvasive and Safe Cell Viability Assay for Breast Cancer MCF-7 Cells Using Natural Food Pigment. Biology, 9(8), 227. https://doi.org/10.3390/biology9080227






