The Relationship between Sperm Oxidative Stress Alterations and IVF/ICSI Outcomes: A Systematic Review from Nonhuman Mammals
Abstract
1. Introduction
2. Methods
2.1. Systematic Review Execution and Registration
2.2. Data Sources and Systematic Search Strategy
2.3. Study Eligibility
2.4. Study Selection Procedure
2.5. Data Extraction for Systematic Review and Statistics
3. Results
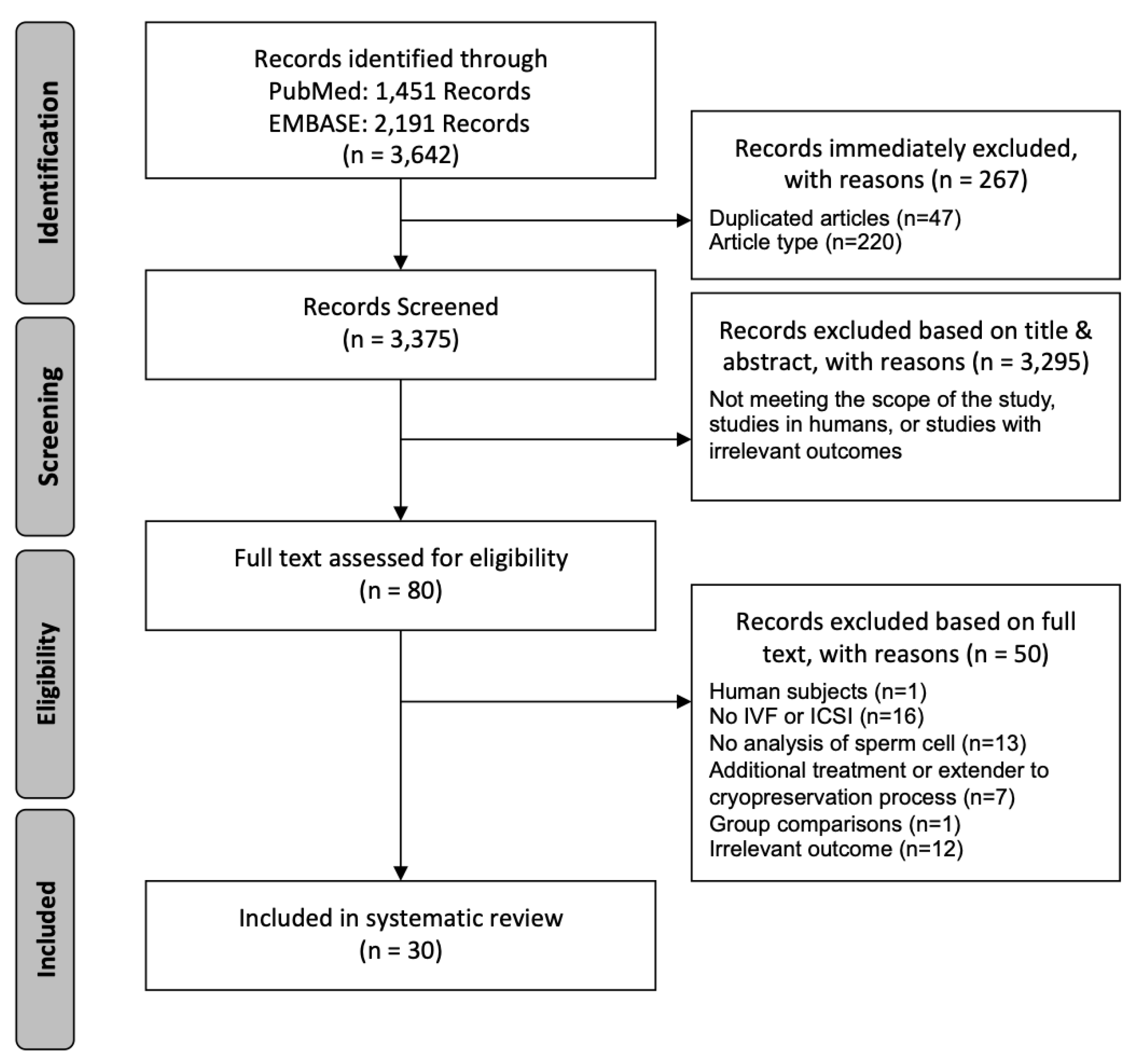
3.1. Identification and Selection of Articles for Qualitative Analysis
3.2. Systematic Review: Qualitative Analysis
3.2.1. Effect of Different Treatments on Spermatozoa
3.2.2. Effect of the Different Treatments on IVF/ICSI Outcomes
4. Discussion
4.1. Strengths and Limitations
4.2. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
Appendix A
References
- Yeste, M.; Jones, C.; Amdani, S.N.; Patel, S.; Coward, K. Oocyte activation deficiency: A role for an oocyte contribution? Hum. Reprod. Update 2016, 22, 23–47. [Google Scholar] [CrossRef]
- De Geyter, C.; Calhaz-Jorge, C.; Kupka, M.S.; Wyns, C.; Mocanu, E.; Motrenko, T.; Scaravelli, G.; Smeenk, J.; Vidakovic, S.; Goossens, V.; et al. ART in Europe, 2015: Results generated from European registries by ESHRE. Hum. Reprod. Open 2020, 2020, hoz038. [Google Scholar] [CrossRef] [PubMed]
- Gilchrist, R.B.; Lane, M.; Thompson, J.G. Oocyte-secreted Factors: Regulators of Cumulus Cell Function and Oocyte Quality. Hum. Reprod. Update 2008, 14. [Google Scholar] [CrossRef]
- Colaco, S.; Sakkas, D. Paternal Factors Contributing to Embryo Quality. J. Assist. Reprod. Genet. 2018, 35. [Google Scholar] [CrossRef] [PubMed]
- Munné, S.; Howles, C.M.; Wells, D. The Role of Preimplantation Genetic Diagnosis in Diagnosing Embryo Aneuploidy. Curr. Opin. Obstet. Gynecol. 2009, 21. [Google Scholar] [CrossRef] [PubMed]
- Munné, S. Status of Preimplantation Genetic Testing and Embryo Selection. Reprod. Biomed. Online 2018, 37. [Google Scholar] [CrossRef] [PubMed]
- Castelló, D.; Motato, Y.; Basile, N.; Remohí, J.; Espejo-Catena, M.; Meseguer, M. How much have we learned from time-lapse in clinical IVF? Mol. Hum. Reprod. 2016, 22, 719–727. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Matzuk, M.M.; Lamb, D.J. The biology of infertility: Research advances and clinical challenges. Nat. Med. 2008, 14, 1197. [Google Scholar] [CrossRef] [PubMed]
- Bracke, A.; Peeters, K.; Punjabi, U.; Hoogewijs, D.; Dewilde, S. A search for molecular mechanisms underlying male idiopathic infertility. Reprod. Biomed. Online 2018, 36, 327–339. [Google Scholar] [CrossRef] [PubMed]
- Rubino, P.; Viganò, P.; Luddi, A.; Piomboni, P. The ICSI procedure from past to future: A systematic review of the more controversial aspects. Hum. Reprod. Update 2015, 22, dmv050. [Google Scholar] [CrossRef] [PubMed]
- Lamirande, E.; Harakat, A.; Gagnon, C. Human Sperm Capacitation Induced by Biological Fluids and Progesterone, but Not by NADH or NADPH, Is Associated With the Production of Superoxide Anion. J. Androl. 1998, 19, 215–225. [Google Scholar] [PubMed]
- Herrero, M.B.; de Lamirande, E.; Gagnon, C. Nitric Oxide Regulates Human Sperm Capacitation and Protein-Tyrosine Phosphorylation In Vitro1. Biol. Reprod. 1999, 61, 575–581. [Google Scholar] [CrossRef] [PubMed]
- Aitken, R.J.; Gibb, Z.; Baker, M.A.; Drevet, J.; Gharagozloo, P. Causes and consequences of oxidative stress in spermatozoa. Reprod. Fertil. Dev. 2016, 28, 1–10. [Google Scholar] [CrossRef] [PubMed]
- O’Flaherty, C.; Matsushita-Fournier, D. Reactive oxygen species and protein modifications in spermatozoa. Biol. Reprod. 2017, 97, 577–585. [Google Scholar] [CrossRef]
- Drevet, J.R.; Aitken, R.J. Oxidative Damage to Sperm DNA: Attack and Defense. In Advances in Experimental Medicine and Biology; Baldi, E., Muratori, M., Eds.; Springer: Berlin, Germany, 2019; Volume 1166, pp. 107–117. [Google Scholar]
- Ribas-Maynou, J.; Benet, J. Single and Double Strand Sperm DNA Damage: Different Reproductive Effects on Male Fertility. Genes (Basel) 2019, 10, 105. [Google Scholar] [CrossRef] [PubMed]
- Lewis, S.E.M.; Aitken, R.J. DNA damage to spermatozoa has impacts on fertilization and pregnancy. Cell Tissue Res. 2005, 322, 33–41. [Google Scholar] [CrossRef]
- Simon, L.; Lewis, S.E.M. Sperm DNA damage or progressive motility: Which one is the better predictor of fertilization in vitro? Syst. Biol. Reprod. Med. 2011, 57, 133–138. [Google Scholar] [CrossRef]
- Gawecka, J.E.; Marh, J.; Ortega, M.; Yamauchi, Y.; Ward, M.A.; Ward, W.S. Mouse zygotes respond to severe sperm DNA damage by delaying paternal DNA replication and embryonic development. PLoS ONE 2013, 8, e56385. [Google Scholar] [CrossRef] [PubMed]
- Toyoshima, M.; Shimura, T.; Adiga, S.-K.; Taga, M.; Shiraishi, K.; Inoue, M.; Yuan, Z.-M.; Niwa, O. Transcription-independent suppression of DNA synthesis by p53 in sperm-irradiated mouse zygotes. Oncogene 2005, 24, 3229–3235. [Google Scholar] [CrossRef]
- Dorostghoal, M.; Kazeminejad, S.R.; Shahbazian, N.; Pourmehdi, M.; Jabbari, A. Oxidative stress status and sperm DNA fragmentation in fertile and infertile men. Andrologia 2017, 49. [Google Scholar] [CrossRef]
- Barbato, V.; Talevi, R.; Braun, S.; Merolla, A.; Sudhakaran, S.; Longobardi, S.; Gualtieri, R. Supplementation of sperm media with zinc, D-aspartate and co-enzyme Q10 protects bull sperm against exogenous oxidative stress and improves their ability to support embryo development. Zygote 2017, 25, 168–175. [Google Scholar] [CrossRef] [PubMed]
- Zini, A. Are sperm chromatin and DNA defects relevant in the clinic? Syst. Biol. Reprod. Med. 2011, 57, 78–85. [Google Scholar] [CrossRef] [PubMed]
- Zhao, J.; Zhang, Q.; Wang, Y.; Li, Y. Whether sperm deoxyribonucleic acid fragmentation has an effect on pregnancy and miscarriage after in vitro fertilization/intracytoplasmic sperm injection: A systematic review and meta-analysis. Fertil. Steril. 2014, 102, 998–1005.e8. [Google Scholar] [CrossRef] [PubMed]
- Cissen, M.; van Wely, M.; Scholten, I.; Mansell, S.; de Bruin, J.P.; Mol, B.W.; Braat, D.; Repping, S.; Hamer, G. Measuring Sperm DNA Fragmentation and Clinical Outcomes of Medically Assisted Reproduction: A Systematic Review and Meta-Analysis. PLoS ONE 2016, 11, e0165125. [Google Scholar] [CrossRef]
- Simon, L.; Aston, K.I.; Emery, B.R.; Hotaling, J.; Carrell, D.T. Sperm DNA damage output parameters measured by the alkaline Comet assay and their importance. Andrologia 2017, 49, e12608. [Google Scholar] [CrossRef] [PubMed]
- Deng, C.; Li, T.; Xie, Y.; Guo, Y.; Yang, Q.; Liang, X.; Deng, C.; Liu, G. Sperm DNA fragmentation index influences assisted reproductive technology outcome: A systematic review and meta-analysis combined with a retrospective cohort study. Andrologia 2019, 51, e13263. [Google Scholar] [CrossRef]
- Simon, L.; Zini, A.; Dyachenko, A.; Ciampi, A.; Carrell, D. A systematic review and meta-analysis to determine the effect of sperm DNA damage on in vitro fertilization and intracytoplasmic sperm injection outcome. Asian J. Androl. 2017, 19, 80–90. [Google Scholar] [CrossRef]
- Thirumavalavan, N.; Gabrielsen, J.S.; Lamb, D.J. Where are we going with gene screening for male infertility? Fertil. Steril. 2019, 111, 842–850. [Google Scholar] [CrossRef]
- Krausz, C.; Escamilla, A.R.; Chianese, C. Genetics of male infertility: From research to clinic. Reproduction 2015, 150, R159–R174. [Google Scholar] [CrossRef] [PubMed]
- Alvarez, M.; Anel-Lopez, L.; Boixo, J.C.; Chamorro, C.; Neila-Montero, M.; Montes-Garrido, R.; de Paz, P.; Anel, L. Current challenges in sheep artificial insemination: A particular insight. Reprod. Domest. Anim. 2019, 54, 32–40. [Google Scholar] [CrossRef]
- Waberski, D.; Riesenbeck, A.; Schulze, M.; Weitze, K.F.; Johnson, L. Application of preserved boar semen for artificial insemination: Past, present and future challenges. Theriogenology 2019, 137, 2–7. [Google Scholar] [CrossRef] [PubMed]
- Yeste, M.; Rodríguez-Gil, J.E.; Bonet, S. Artificial insemination with frozen-thawed boar sperm. Mol. Reprod. Dev. 2017, 84, 802–813. [Google Scholar] [CrossRef] [PubMed]
- Fowler, K.E.; Mandawala, A.A.; Griffin, D.K.; Walling, G.A.; Harvey, S.C. The production of pig preimplantation embryos in vitro: Current progress and future prospects. Reprod. Biol. 2018, 18, 203–211. [Google Scholar] [CrossRef]
- Aurich, C.; Schneider, J. Sex determination in horses—Current status and future perspectives. Anim. Reprod. Sci. 2014, 146, 34–41. [Google Scholar] [CrossRef] [PubMed]
- Espinosa-Cervantes, R.; Córdova-Izquierdo, A. Sexing sperm of domestic animals. Trop. Anim. Health Prod. 2012, 45, 1–8. [Google Scholar] [CrossRef]
- Yamauchi, Y.; Shaman, J.A.; Boaz, S.M.; Ward, W.S. Paternal pronuclear DNA degradation is functionally linked to DNA replication in mouse oocytes. Biol. Reprod. 2007, 77, 407–415. [Google Scholar] [CrossRef] [PubMed]
- Burruel, V.; Klooster, K.L.; Chitwood, J.; Ross, P.J.; Meyers, S.A. Oxidative Damage to Rhesus Macaque Spermatozoa Results in Mitotic Arrest and Transcript Abundance Changes in Early Embryos1. Biol. Reprod. 2013, 89, 1–11. [Google Scholar] [CrossRef]
- Liberati, A.; Altman, D.G.; Tetzlaff, J.; Mulrow, C.; Gøtzsche, P.C.; Ioannidis, J.P.A.; Clarke, M.; Devereaux, P.J.; Kleijnen, J.; Moher, D. The PRISMA Statement for Reporting Systematic Reviews and Meta-Analyses of Studies That Evaluate Health Care Interventions: Explanation and Elaboration. PLoS Med. 2009, 6, e1000100. [Google Scholar] [CrossRef] [PubMed]
- Ahmadi, A.; Ng, S.C. Developmental capacity of damaged spermatozoa. Hum. Reprod. 1999, 14, 2279–2285. [Google Scholar] [CrossRef]
- Arias, M.E.; Sánchez, R.; Risopatrón, J.; Pérez, L.; Felmer, R. Effect of sperm pretreatment with sodium hydroxide and dithiothreitol on the efficiency of bovine intracytoplasmic sperm injection. Reprod. Fertil. Dev. 2014, 26, 847–854. [Google Scholar] [CrossRef] [PubMed]
- Bittner, L.; Wyck, S.; Herrera, C.; Siuda, M.; Wrenzycki, C.; Van Loon, B.; Bollwein, H. Negative effects of oxidative stress in bovine spermatozoa on in vitro development and DNA integrity of embryos. Reprod. Fertil. Dev. 2018, 30, 1359–1368. [Google Scholar] [CrossRef]
- De Castro, L.S.; De Assis, P.M.; Siqueira, A.F.P.; Hamilton, T.R.S.; Mendes, C.M.; Losano, J.D.A.; Nichi, M.; Visintin, J.A.; Assumpção, M.E.O.A. Sperm oxidative stress is detrimental to embryo development: A dose-dependent study model and a new and more sensitive oxidative status evaluation. Oxid. Med. Cell. Longev. 2016, 2016, 8213071. [Google Scholar] [CrossRef] [PubMed]
- Castro, L.S.; Siqueira, A.F.P.; Hamilton, T.R.S.; Mendes, C.M.; Visintin, J.A.; Assumpção, M.E.O.A. Effect of bovine sperm chromatin integrity evaluated using three different methods on in vitro fertility. Theriogenology 2018, 107, 142–148. [Google Scholar] [CrossRef] [PubMed]
- Cho, C.; Jung-Ha, H.; Willis, W.D.; Goulding, E.H.; Stein, P.; Xu, Z.; Schultz, R.M.; Hecht, N.B.; Eddy, E.M. Protamine 2 Deficiency Leads to Sperm DNA Damage and Embryo Death in Mice1. Biol. Reprod. 2003, 69, 211–217. [Google Scholar] [CrossRef]
- Manas, G.E.; Hasanzadeh, S.; Najafi, G.; Parivar, K.; Yaghmaei, P. The effects of pyridaben pesticide on the DNA integrity of sperms and early in vitro embryonic development in mice. Int. J. Reprod. Biomed. 2013, 11, 605–610. [Google Scholar]
- Fatehi, A.N.; Bevers, M.M.; Schoevers, E.; Roelen, B.A.J.; Colenbrander, B.; Gadella, B.M. DNA damage in bovine sperm does not block fertilization and early embryonic development but induces apoptosis after the first cleavages. J. Androl. 2006, 27, 176–188. [Google Scholar] [CrossRef]
- Gonçalves, F.S.; Barretto, L.S.S.; Arruda, R.P.; Perri, S.H.V.; Mingoti, G.Z. Effect of antioxidants during bovine in vitro fertilization procedures on spermatozoa and embryo development. Reprod. Domest. Anim. 2010, 45, 129–135. [Google Scholar] [CrossRef] [PubMed]
- Gonzalez-Castro, R.A.; Carnevale, E.M. Association of equine sperm population parameters with outcome of intracytoplasmic sperm injections. Theriogenology 2018, 119, 114–120. [Google Scholar] [CrossRef] [PubMed]
- Hourcade, J.D.; Pérez-Crespo, M.; Fernández-González, R.; Pintado, B.; Gutiérrez-Adán, A. Selection against spermatozoa with fragmented DNA after postovulatory mating depends on the type of damage. Reprod. Biol. Endocrinol. 2010, 8, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Izquierdo-Vega, J.A.; Sánchez-Gutiérrez, M.; Del Razo, L.M. Decreased in vitro fertility in male rats exposed to fluoride-induced oxidative stress damage and mitochondrial transmembrane potential loss. Toxicol. Appl. Pharmacol. 2008, 230, 352–357. [Google Scholar] [CrossRef] [PubMed]
- Jang, H.Y.; Kim, Y.H.; Kim, B.W.; Park, I.C.; Cheong, H.T.; Kim, J.T.; Park, C.K.; Kong, H.S.; Lee, H.K.; Yang, B.K. Ameliorative effects of melatonin against hydrogen peroxide-induced oxidative stress on boar sperm characteristics and subsequent in vitro embryo development. Reprod. Domest. Anim. 2010, 45, 943–950. [Google Scholar] [CrossRef] [PubMed]
- Li, C.; Mizutani, E.; Ono, T.; Wakayama, T. Production of normal mice from spermatozoa denatured with high alkali treatment before ICSI. Reproduction 2009, 137, 779–792. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Li, M.W.; Lloyd, K.C.K. DNA fragmentation index (DFI) as a measure of sperm quality and fertility in mice. Sci. Rep. 2020, 10, 1–11. [Google Scholar] [CrossRef]
- Llamas Luceño, N.; de Souza Ramos Angrimani, D.; de Cássia Bicudo, L.; Szymańska, K.J.; Van Poucke, M.; Demeyere, K.; Meyer, E.; Peelman, L.; Mullaart, E.; Broekhuijse, M.L.W.J.; et al. Exposing dairy bulls to high temperature-humidity index during spermatogenesis compromises subsequent embryo development in vitro. Theriogenology 2020, 141, 16–25. [Google Scholar] [CrossRef]
- Makvandi, A.; Kowsar, R.; Hajian, M.; Mahdavi, A.H.; Tanhaei Vash, N.; Nasr-Esfahani, M.H. Alpha lipoic acid reverses the negative effect of LPS on mouse spermatozoa and developmental competence of resultant embryos in vitro. Andrology 2019, 7, 350–356. [Google Scholar] [CrossRef]
- Matini Behzad, A.; Ebrahimi, B.; Alizadeh, A.R.; Esmaeili, V.; Dalman, A.; Rashki, L.; Shahverdi, A.H. Improvement in In Vitro fertilization rate, decrease in reactive oxygen species and spermatozoa death incidence in rams by dietary fish oil. Reprod. Domest. Anim. 2014, 49, 599–605. [Google Scholar] [CrossRef]
- Mehraban, Z.; Ghaffari Novin, M.; Golmohammadi, M.G.; Sagha, M.; Pouriran, K.; Nazarian, H. Protective effect of gallic acid on apoptosis of sperm and in vitro fertilization in adult male mice treated with cyclophosphamide. J. Cell. Biochem. 2019, 120, 17250–17257. [Google Scholar] [CrossRef]
- Paul, C.; Teng, S.; Saunders, P.T.K. A Single, Mild, Transient Scrotal Heat Stress Causes Hypoxia and Oxidative Stress in Mouse Testes, Which Induces Germ Cell Death1. Biol. Reprod. 2009, 80, 913–919. [Google Scholar] [CrossRef]
- Pérez-Crespo, M.; Moreira, P.; Pintado, B.; Gutiérrez-Adán, A. Factors from damaged sperm affect its DNA integrity and its ability to promote embryo implantation in mice. J. Androl. 2008, 29, 47–54. [Google Scholar] [CrossRef]
- Sánchez-Gutiérrez, M.; García-Montalvo, E.A.; Izquierdo-Vega, J.A.; Del Razo, L.M. Effect of dietary selenium deficiency on the in vitro fertilizing ability of mice spermatozoa. Cell Biol. Toxicol. 2008, 24, 321–329. [Google Scholar] [CrossRef] [PubMed]
- Silva, P.F.N.; Gadella, B.M.; Colenbrander, B.; Roelen, B.A.J. Exposure of bovine sperm to pro-oxidants impairs the developmental competence of the embryo after the first cleavage. Theriogenology 2007, 67, 609–619. [Google Scholar] [CrossRef] [PubMed]
- Simões, R.; Feitosa, W.B.; Siqueira, A.F.P.; Nichi, M.; Paula-Lopes, F.F.; Marques, M.G.; Peres, M.A.; Barnabe, V.H.; Visintin, J.A.; Assumpçaõ, M.E.O. Influence of bovine sperm DNA fragmentation and oxidative stress on early embryo in vitro development outcome. Reproduction 2013, 146, 433–441. [Google Scholar] [CrossRef]
- Yamauchi, Y.; Shaman, J.A.; Ward, W.S. Topoisomerase II-mediated breaks in spermatozoa cause the specific degradation of paternal DNA in fertilized oocytes. Biol. Reprod. 2007, 76, 666–672. [Google Scholar] [CrossRef]
- Yi, Y.J.; Lee, I.K.; Lee, S.M.; Yun, B.S. An antioxidant davallialactone from Phellinus baumii enhances sperm penetration on in vitro fertilization of pigs. Mycobiology 2016, 44, 54–57. [Google Scholar] [CrossRef] [PubMed]
- Yi, Y.J.; Kamala-Kannan, S.; Lim, J.M.; Oh, B.T.; Lee, S.M. Effects of difructose dianhydride (DFA)-IV on in vitro fertilization in pigs. J. Biomed. Res. 2017, 31, 453–461. [Google Scholar] [CrossRef] [PubMed]
- Baskaran, S.; Finelli, R.; Agarwal, A.; Henkel, R. Diagnostic value of routine semen analysis in clinical andrology. Andrologia 2020, e13614. [Google Scholar] [CrossRef]
- Chen, Q.; Yan, M.; Cao, Z.; Li, X.; Zhang, Y.; Shi, J.; Feng, G.; Peng, H.; Zhang, X.; Zhang, Y.; et al. Sperm tsRNAs contribute to intergenerational inheritance of an acquired metabolic disorder. Science 2016, 351, 397–400. [Google Scholar] [CrossRef] [PubMed]
- Drevet, J.R.; Aitken, R.J. Oxidation of Sperm Nucleus in Mammals: A Physiological Necessity to Some Extent with Adverse Impacts on Oocyte and Offspring. Antioxidants 2020, 9, 95. [Google Scholar] [CrossRef] [PubMed]
- Barati, E.; Nikzad, H.; Karimian, M. Oxidative stress and male infertility: Current knowledge of pathophysiology and role of antioxidant therapy in disease management. Cell. Mol. Life Sci. 2020, 77, 93–113. [Google Scholar] [CrossRef]
- Thompson, D.M.; Lu, C.; Green, P.J.; Parker, R. tRNA cleavage is a conserved response to oxidative stress in eukaryotes. RNA 2008, 14, 2095–2103. [Google Scholar] [CrossRef]
- Zhang, Y.; Zhang, X.; Shi, J.; Tuorto, F.; Li, X.; Liu, Y.; Liebers, R.; Zhang, L.; Qu, Y.; Qian, J.; et al. Dnmt2 mediates intergenerational transmission of paternally acquired metabolic disorders through sperm small non-coding RNAs. Nat. Cell Biol. 2018, 20, 535–540. [Google Scholar] [CrossRef]
- Jenkins, T.G.; James, E.R.; Alonso, D.F.; Hoidal, J.R.; Murphy, P.J.; Hotaling, J.M.; Cairns, B.R.; Carrell, D.T.; Aston, K.I. Cigarette smoking significantly alters sperm DNA methylation patterns. Andrology 2017, 5, 1089–1099. [Google Scholar] [CrossRef] [PubMed]
- Murphy, P.J.; Guo, J.; Jenkins, T.G.; James, E.R.; Hoidal, J.R.; Huecksteadt, T.; Broberg, D.S.; Hotaling, J.M.; Alonso, D.F.; Carrell, D.T.; et al. NRF2 loss recapitulates heritable impacts of paternal cigarette smoke exposure. PLOS Genet. 2020, 16, e1008756. [Google Scholar] [CrossRef] [PubMed]
- Nätt, D.; Kugelberg, U.; Casas, E.; Nedstrand, E.; Zalavary, S.; Henriksson, P.; Nijm, C.; Jäderquist, J.; Sandborg, J.; Flinke, E.; et al. Human sperm displays rapid responses to diet. PLoS Biol. 2019, 17, e3000559. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Chen, Q. Human sperm RNA code senses dietary sugar. Nat. Rev. Endocrinol. 2020, 16, 200–201. [Google Scholar] [CrossRef] [PubMed]
- Hua, M.; Liu, W.; Chen, Y.; Zhang, F.; Xu, B.; Liu, S.; Chen, G.; Shi, H.; Wu, L. Identification of small non-coding RNAs as sperm quality biomarkers for in vitro fertilization. Cell Discov. 2019, 5, 20. [Google Scholar] [CrossRef] [PubMed]
- Yoshida, K.; Maekawa, T.; Ly, N.H.; Fujita, S.-I.; Muratani, M.; Ando, M.; Katou, Y.; Araki, H.; Miura, F.; Shirahige, K.; et al. ATF7-Dependent Epigenetic Changes Are Required for the Intergenerational Effect of a Paternal Low-Protein Diet. Mol. Cell 2020, 78, 445–458.e6. [Google Scholar] [CrossRef]
- Conti, M.; Franciosi, F. Acquisition of oocyte competence to develop as an embryo: Integrated nuclear and cytoplasmic events. Hum. Reprod. Update 2018, 24, 245–266. [Google Scholar] [CrossRef]
- Practice Committee of the American Society for Reproductive Medicine The clinical utility of sperm DNA integrity testing: A guideline. Fertil. Steril. 2013, 99, 673–677. [CrossRef]
- Barratt, C.L.R.; Aitken, R.J.; Björndahl, L.; Carrell, D.T.; de Boer, P.; Kvist, U.; Lewis, S.E.M.; Perreault, S.D.; Perry, M.J.; Ramos, L.; et al. Sperm DNA: Organization, protection and vulnerability: From basic science to clinical applications--A position report. Hum. Reprod. 2010, 25, 824–838. [Google Scholar] [CrossRef]
- Toyoshima, M. Analysis of p53 dependent damage response in sperm-irradiated mouse embryos. J. Radiat. Res. 2009, 50, 11–17. [Google Scholar] [CrossRef] [PubMed]
- Adiga, S.K.; Toyoshima, M.; Shiraishi, K.; Shimura, T.; Takeda, J.; Taga, M.; Nagai, H.; Kumar, P.; Niwa, O. p21 provides stage specific DNA damage control to preimplantation embryos. Oncogene 2007, 26, 6141–6149. [Google Scholar] [CrossRef] [PubMed]
| Animal | Total Studies | Fertilization Method | Oocyte Origin | |||
|---|---|---|---|---|---|---|
| IVF Studies | ICSI Studies | IVM | Superovulated | Nonspecified | ||
| Pig | 3 | 3 | 0 | 3 | - | - |
| Cattle | 9 | 8 | 1 | 7 | 1 | 1 |
| Mice | 13 | 7 | 6 | - | 12 | 1 |
| Mouse and Hamster | 1 | 0 | 1 | - | 1 | - |
| Sheep | 1 | 0 | 1 | 1 | - | - |
| Rat | 1 | 1 | 0 | - | 1 | - |
| Macaque | 1 | 0 | 1 | - | 1 | - |
| Horse | 1 | 0 | 1 | - | 1 (only ovulation induction) | - |
| ID | Reference | Aim | Animal | Treatment | Sperm Parameter Measured | Effects of Treatment on Sperm | Oocyte Origin | IVF or ICSI | Effects on IVF/ICSI | Conclusion |
|---|---|---|---|---|---|---|---|---|---|---|
| 1 | Ahmadi et al. 1999 [40] | To study the developmental capacity of spermatozoa at various levels of disintegration. | MouseHamster | Frozen twice without cryoprotectant plus DTT and gamma radiation | DNA damage: TUNEL | Increased DNA damage. | Super-ovulated | ICSI | - Similar fertilization rates - Lower blastocyst development - Lower live birth rate | Impairment of sperm cells causes a reduction in ICSI outcomes. |
| 2 | Arias et al. 2014 [41] | To evaluate the effects of pretreatment of bovine spermatozoa with NaOH and DTT on in vitro developmental potential. | Cattle | Incubation of sperm with 5 mM DTT for 20 min and 1 mM NaOH for 60 min | DNA fragmentation: TUNEL | Highest concentrations of NaOH led to high DNA damage. | Super-ovulated | ICSI | - High concentration of NaOH blocked blastocyst development | NaOH has a detrimental effect on embryo development after ICSI. |
| 3 | Bittner et al. 2018 [42] | To elucidate whether sperm oxidative stress results in increased DNA damage in the embryo. | Cattle | Incubation of sperm with H2O2 | DNA damage: SCSA | Exposure of sperm to H2O2 led to an increase of DNA damage in the embryo. | IVM | IVF | - Embryo development was delayed when sperm were incubated with H2O2 treatment - Reduced cleavage and blastocyst rates | Oxidative stress in spermatozoa induces developmental abnormalities in the embryo. |
| 4 | Burruel et al. 2013 [38] | To determine whether sperm oxidative damage induced by ROS affects embryo development. | Rhesus Macaque | Oxidation of sperm cells with xanthine oxidase. | - Lipid peroxidation | Treatment with xanthine oxidase led to an increase of lipid peroxidation. | Super-ovulated | ICSI | - Exposure to high levels of ROS leads to arrest of embryo development before eight-cell stage. Additionally, all stages were affected in the treated group. | Paternal oxidative stress influences early embryo development. |
| 5 | Castro et al. 2016 [43] | To assess the impact of sperm oxidative stress on embryo development. | Cattle | Incubation of sperm with increasing doses of H2O2 | - Oxidative status: CellROX green - DNA damage: SCSA | Incubation causes an increase of oxidative stress and alterations in chromatin integrity. | IVM | IVF | - Cleavage rates, development to 8 cells and percentages of blastocysts are inversely related to H2O2 concentration | Oxidative environment can impair bull sperm quality, affecting embryo development. |
| 6 | Castro et al. 2018 [44] | To predict in vitro bull fertility with the assessment of DNA integrity. | Cattle | High vs. low fertility bulls based on embryo development rate | - DNA damage: SCSA and Comet - DNA condensation: CMA3 | None. | Not specified | IVF | - SCSA and Comet did not show differences between high and low fertility groups - DNA condensation (CMA3 test) was lower in the high fertility group. | The differences found in DNA condensation through CMA3 were not sufficient to explain differences in fertility rates. |
| 7 | Cho et al. 2003 [45] | To determine whether a reduction in protamine content results in failure to transmit the male genome to next generation. | Mouse | Chimeric, Prm2−/>− mice | - DNA damage: Comet assay | DNA damage was observed in chimeras, affecting more than 70% spermatozoa. | Not specified | ICSI | - Arrested embryos at metaphase. - Less embryos developed to later stages. | Protamine 2 is essential to maintain sperm DNA integrity and to promote embryo development. |
| 8 | Ebadi Manas et al. 2013 [46] | To elucidate how pyridaben can affect sperm quality and in vitro fertilizing ability. | Mouse | Oral administration of pyridaben for 45 days at two different doses | - Chromatin integrity: Aniline blue and acridine orange | Increase of sperm DNA damage. | Super-ovulated | IVF | - Lower fertilization rates - Lower development to blastocyst rates | Pyridaben induces DNA damage, which decreases fertilization and blastocyst rates. |
| 9 | Fatehi et al. 2006 [47] | To investigate whether and at what level the paternal DNA damage influences fertilization and embryo development. | Cattle | Irradiation with X or Gamma rays | - DNA damage: TUNEL and Acridin Orange | Sperm DNA damage increased in a dose dependent manner. | IVM | IVF | - Similar rates of embryo cleavage at day 4 - Embryo development to blastocyst was severely impaired | DNA damage induced by radiations impair embryo development to blastocyst. |
| 10 | Gawecka et al. 2013 [19] | To test how zygotes respond to DNA damage during the first cell cycle. | Mouse | Induction of DNA damage with manganese and calcium | - DNA damage: Pulsed Field Gel Electrophoresis | Double strand DNA damage detected by Pulsed Field Gel Electrophoresis (PFGE). | Super-ovulated | ICSI | - Chromosomal alterations at first paternal pronucleus - No fertilization impairment - Embryo development was delayed after fertilization and arrested before reaching the blastocyst stage | DNA damage is important for proper embryo development at initial stages. |
| 11 | Gonçalves et al. 2010 [48] | To investigate the effects of antioxidants and a pro-oxidant on the quality and fertilizing ability of bull spermatozoa. | Cattle | Sperm were co-incubated with antioxidants (β-mercaptoethanol and Cysteamine) or pro-oxidant (buthionine sulfoximine) molecules | - DNA damage: Acridine Orange | No effect on sperm DNA damage, but reduction of plasma membrane integrity. | IVM | IVF | - Antioxidants and pro-oxidants reduced fertilization and blastocyst rates | Supplementation with antioxidants and with pro-oxidants during IVF procedures impaired sperm quality, normal pronuclear formation and development to blastocyst. |
| 12 | Gonzalez-Castro et al. 2018 [49] | To determine which sperm population characteristics are predictive of ICSI outcome. | Horse | Evaluation of DNA damage and plasma membrane integrity | - Membrane integrity: HOS test - DNA damage: SCD test | None. | Ovulation induction and follicle aspiration | ICSI | - DNA damage was not associated to any fertilization, embryo development or pregnancy | Plasma membrane integrity was the parameter with higher association to ICSI outcome |
| 13 | Hourcade et al. 2010 [50] | To investigate if female tract is able to select nonaffected sperm cells. | Mouse | Heat stress and Gamma radiation to animals. | - DNA damage: Comet assay | The extent of DNA damage in sperm obtained from female tract was higher following heat stress than after gamma radiation. | Super-ovulated | IVF and ICSI | - Heat stress reduced fertilization rates in both IVF and ICSI - Gamma radiation decreased blastocyst rates in gamma radiation in both IVF and ICSI | - Sperm DNA fragmentation affects IVF and ICSI. - ICSI should be performed with highly motile sperm. |
| 14 | Izquierdo-Vega et al. 2008 [51] | To evaluate the effects of fluoride on in vitro sperm fertilizing ability. | Rat | Rats administered with 5 mg fluoride/kg/body mass/24 h for eight weeks | - SOD activity - Intracellular superoxide anion levels - Lipid peroxidation | Fluoride increases oxidative stress, decreases the antioxidant activity of SOD, and increases lipid peroxidation. | Super-ovulated | IVF | - Lower ability to fertilize oocytes | Fluoride exposure causes a decrease in sperm fertilization capacity. |
| 15 | Jang et al. 2010 [52] | To examine the developmental rates of semen treated with and without melatonin in the presence of H2O2. | Pig | Treatment of semen with H2O2 with and without melatonin. | - Lipid peroxidation: Malondialdehyde levels | Exposure to H2O2 augmented lipid peroxidation, but this increase was mitigated by melatonin. | IVM | IVF | - Fertilization was not impaired by exposure of sperm to H2O2 - Exposing sperm to H2O2 arrested the development to blastocyst, but melatonin prevented that negative effect. | Supplementation with melatonin could improve sperm quality, increasing the developmental capacity of porcine embryos. |
| 16 | Li et al. 2009 [53] | To evaluate whether normal offspring can be generated after exposing sperm to high NaOH concentrations. | Mouse | High alkaline treatment of sperm cells | - DNA damage: Acridine Orange | Increase of DNA damage. | Super-ovulated | ICSI | - Decrease of fertilization rates - Reduction of implantation and live pups born - Increase of chromosomal damage in embryos | Sperm treated with NaOH retain their ability to activate the oocyte, but embryo development is lower as NaOH concentration increases. |
| 17 | Li et al. 2020 [54] | To assess the extent to which the measurement of DNA fragmentation index in sperm can predict their fertilizing ability. | Mouse | Freeze-thawing | - DNA damage: TUNEL | Cryopreservation increased DNA damage. | Super-ovulated | IVF and ICSI | - Sperm DNA damage reduced fertilization rates following both IVF and ICSI. - Samples with higher DNA damage resulted in a reduction of viable offspring. | DNA fragmentation index is an accurate parameter to determine sperm quality and fertility potential. |
| 18 | Llamas Luceño et al. 2020 [55] | To address the impact of natural heat stress on bull fertility. | Cattle | Comparison of different bulls exposed to separate levels of heat stress | - ROS production - Lipid peroxidation - DNA fragmentation: TUNEL | No differences in H2O2 production, lipid peroxidation and DNA damage. A tendency to significance in total ROS production was observed. | IVM | IVF | - No impact of exposing sperm to heat stress upon cleavage rates - A reduction of day 7 and 8 blastocyst rate and a delay of blastocyst hatching were observed. | Sperm fertilizing ability decreases due to male exposure to heat stress. |
| 19 | Makvan-di et al. 2019 [56] | To show the potential benefits of alpha lipoic acid as antioxidant in lipopolysaccharide- treated sperm cells. | Mouse | Sperm cells were incubated with lipopolysaccharide and alpha lipoic acid at different concentrations | - ROS production: DCFH-DA test - DNA damage: Acridine orange | Lipopolysaccharide augmented ROS and DNA damage, but the addition of alpha lipoic acid mitigated those increases. | Super-ovulated | IVF | While lipopolysaccharide led to lower fertilization, cleavage, compaction and blastocyst rates, this effect was reverted by alpha lipoic acid. | Alpha lipoic acid is a strong antioxidant and protective sperm factor. |
| 20 | Matini Behzad et al. 2014 [57] | To evaluate the effects of fish oil feeding on sperm parameters and its incidence in IVF. | Sheep | Fish oil diet for 70 days | Intracellular ROS | Superoxide anion was lower in the treated than in the control group. | IVM | IVF | - Cleavage rates were higher in the treated group. | Addition of fish oil to ram diet improves sperm quality and in vitro fertilization ability. |
| 21 | Mehraban et al. 2019 [58] | To evaluate the antioxidant effects of Gallic Acid on apoptotic-like changes in sperm. | Mouse | Cyclophosphamide and gallic acid | Apoptotic-like changes: Annexin V staining | Cyclophosphamide induces apoptotic-like changes compared to controls. Gallic Acid mitigates that increase. | Super-ovulated | IVF | - Cyclophosphamide reduces fertilization rates and proportions of cleaved embryos. | Gallic Acid suppresses ROS induced by Cyclophosphamide and help rescue fertility. |
| 22 | Paul et al. 2008 [59] | To explore the link between heat stress, DNA damage and the impact on cell function. | Mouse | Acute testicular heat stress at different temperatures | - Sperm DNA damage: SCSA - Spermatocyte DNA damage | Increase of DNA damage in spermatocytes and spermatozoa when heat stress was 40 °C/42 °C. | Super-ovulated | IVF | - Fertilization was similar between groups. - Development to 2-4 cells was similar to controls. - Development to blastocyst was severely reduced. | Spermatogenesis is impaired when scrotal temperatures increase, showing that exposing male to high temperatures reduces embryo development and pregnancy of the oocytes fertilized with those sperm. |
| 23 | Perez-Crespo et al. 2008 [60] | To evaluate whether factors released from membrane-damaged spermatozoa have a role in DNA damage and ICSI outcomes. | Mouse | Incubation of sperm in media containing factors released by damaged sperm | - DNA damage: TUNEL | Increase of sperm DNA damage. | Super-ovulated | ICSI | - Cleavage and blastocyst rates were not impaired by treatment - Implantation was reduced, regardless of whether embryos were transferred at 2-cell stage or at blastocyst stage. | Factors released from membrane-damaged sperm are capable of inducing DNA fragmentation in viable spermatozoa, and decrease implantation rates. |
| 24 | Sanchez-Gutierrez et al. 2008 [61] | To evaluate the effect of selenium deficiency on in vitro fertilizing ability. | Mouse | Selenium deficient diet for 4 months | - Glutathione peroxidase activity - Lipid peroxidation | Glutathione peroxidase activity was reduced and lipid peroxidation was increased in selenium free diet. | Super-ovulated | IVF | - Fertilization rates were reduced | Selenium deficiency leads to a reduction of sperm quality and fertilizing ability. |
| 25 | Silva et al. 2007 [62] | To describe the effects of exposing bull sperm to mild and intense ROS generation conditions. | Cattle | Sperm were incubated with pro-oxidant molecules | - Sperm DNA oxidation - Lipid peroxidation - Cytosol and mitochondrial oxidation | Increased intracellular and mitochondrial ROS, while no significant increase of DNA oxidation was observed. | IVM | IVF | - Reduced cleavage rates and blastocyst rates when high oxidation was applied. | Oxidative stress can result in damaged sperm cell structures, affecting embryo development. |
| 26 | Simoes et al. 2013 [63] | To evaluate the influence of sperm oxidative stress susceptibility on DNA fragmentation and IVF outcomes. | Cattle | Division in four groups according to oxidative stress susceptibility | - DNA damage: Comet assay, Acridine Orange and TUNEL | Sperm DNA was compromised in response to increased oxidative stress susceptibility. | IVM | IVF | - Embryo cleavage decreased as oxidative stress increased - No significant differences in blastocyst rates or number of blastocysts were observed | Increased sperm oxidative stress leads to a reduction of embryo quality. |
| 27 | Yamauchi et al. 2007 [37] | To test if embryo DNA synthesis is related to paternal DNA degradation in zygotes, which is induced by sperm DNA damage. | Mouse | Sperm DNA damage induction through manganese and calcium incubations | - DNA damage: Pulsed Field Gel Electrohporesis (PFGE) | Treatment caused DNA damage detected through PFGE. | Super-ovulated | ICSI | - Fertilization with vas deferens damaged sperm led to DNA degradation in paternal pronucleous. - Intiation of male and female pronucleus DNA replication was delayed. - Blastocyst development was arrested | Impairment of DNA synthesis in the embryo is related to DNA damage in sperm. |
| 28 | Yamauchi et al. 2007 [64] | To test embryo development from sperm cells to which DNA damage was induced. | Mouse | DNA damage induction through manganese and calcium incubations | - DNA damage: Pulsed Field Gel Electrohporesis | Treatment caused DNA damage detected through PFGE. | Super-ovulated | ICSI | - DNA damage caused a reduction in the percentage of embryos reaching two-cell stage - Development to blastocyst was impaired when sperm DNA was damaged | Embryo development is impaired when the fertilizing sperm presents chromatin fragmentation. |
| 29 | Yi et al. 2016 [65] | To examine the effects of Davallialactone (DAVA) on in vitro sperm fertilizing ability. | Pig | Spermatozoa were incubated with DAVA | - ROS production: intracellular H2O2 | DAVA reduced intracellular H2O2 in sperm cells. | IVM | IVF | - Fertilization was enhanced by DAVA | Addition of DAVA to fertilization medium reduces ROS and increases fertilization rates |
| 30 | Yi et al. 2017 [66] | To determine the impact of difructose dianhydride IV (DFA-IV) on in vitro sperm fertilizing ability. | Pig | Spermatozoa were incubated in DFA-IV at different concentrations for two hours | - Total oxidative stress. | Total intracellular ROS levels were decreased in samples incubated with DFA-IV. | IVM | IVF | - Higher fertilization rates - Higher cleavage and blastocyst rates | Addition of DFA-IV to sperm increases fertilization and blastocyst rates. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ribas-Maynou, J.; Yeste, M.; Salas-Huetos, A. The Relationship between Sperm Oxidative Stress Alterations and IVF/ICSI Outcomes: A Systematic Review from Nonhuman Mammals. Biology 2020, 9, 178. https://doi.org/10.3390/biology9070178
Ribas-Maynou J, Yeste M, Salas-Huetos A. The Relationship between Sperm Oxidative Stress Alterations and IVF/ICSI Outcomes: A Systematic Review from Nonhuman Mammals. Biology. 2020; 9(7):178. https://doi.org/10.3390/biology9070178
Chicago/Turabian StyleRibas-Maynou, Jordi, Marc Yeste, and Albert Salas-Huetos. 2020. "The Relationship between Sperm Oxidative Stress Alterations and IVF/ICSI Outcomes: A Systematic Review from Nonhuman Mammals" Biology 9, no. 7: 178. https://doi.org/10.3390/biology9070178
APA StyleRibas-Maynou, J., Yeste, M., & Salas-Huetos, A. (2020). The Relationship between Sperm Oxidative Stress Alterations and IVF/ICSI Outcomes: A Systematic Review from Nonhuman Mammals. Biology, 9(7), 178. https://doi.org/10.3390/biology9070178







