Seminal Plasma Anti-Müllerian Hormone: A Potential AI-Boar Fertility Biomarker?
Abstract
1. Introduction
2. Materials and Methods
2.1. Reagents and Media
2.2. Animals and Ejaculates
2.3. Seminal Plasma Processing and Storage
2.4. Measurement of SP-AMH Concentration
2.5. Assessment of Sperm Quality and Functionality Parameters
2.6. Experimental Design
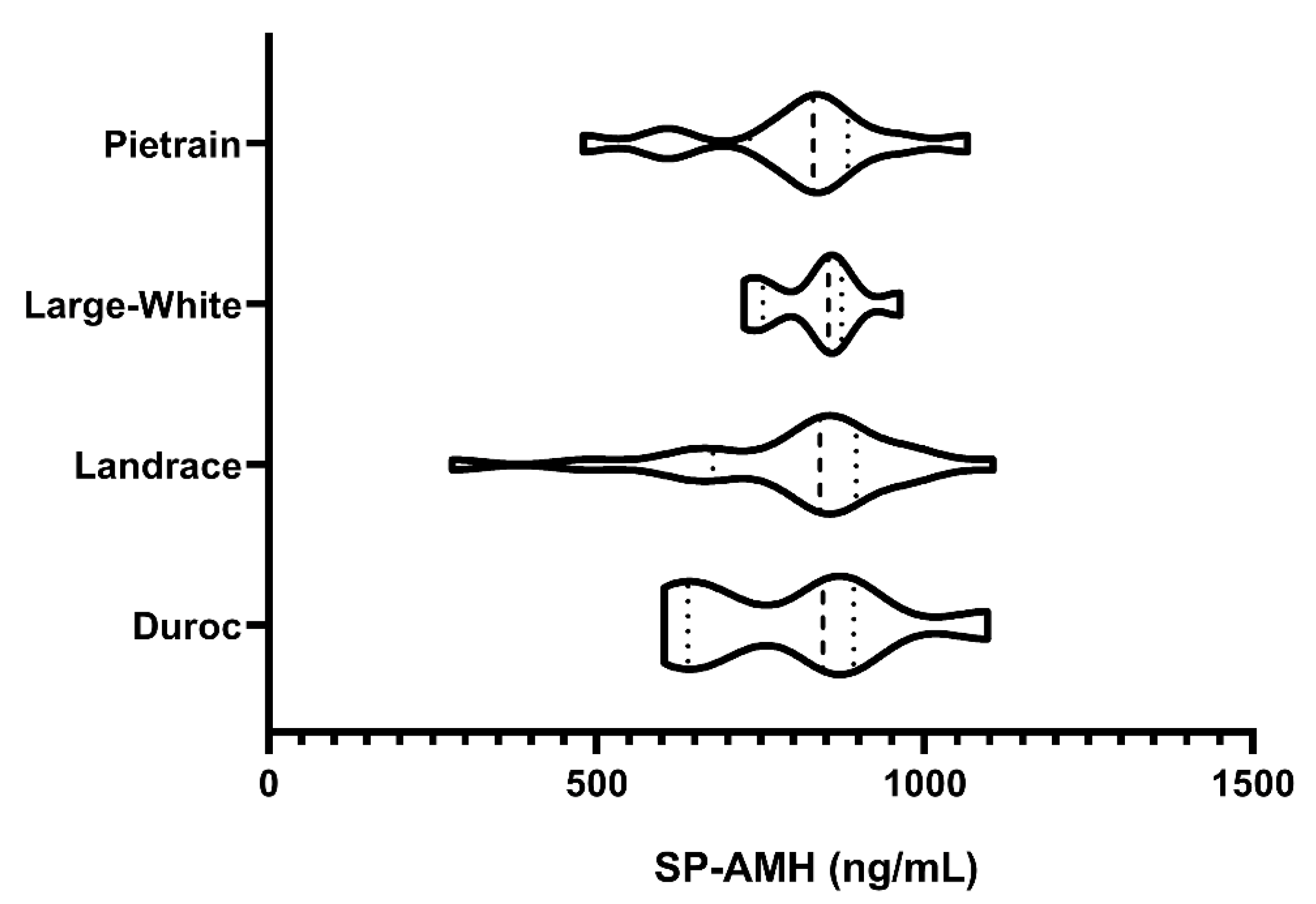
2.6.1. Experiment 1: SP-AMH Concentration Variability Among Breeds
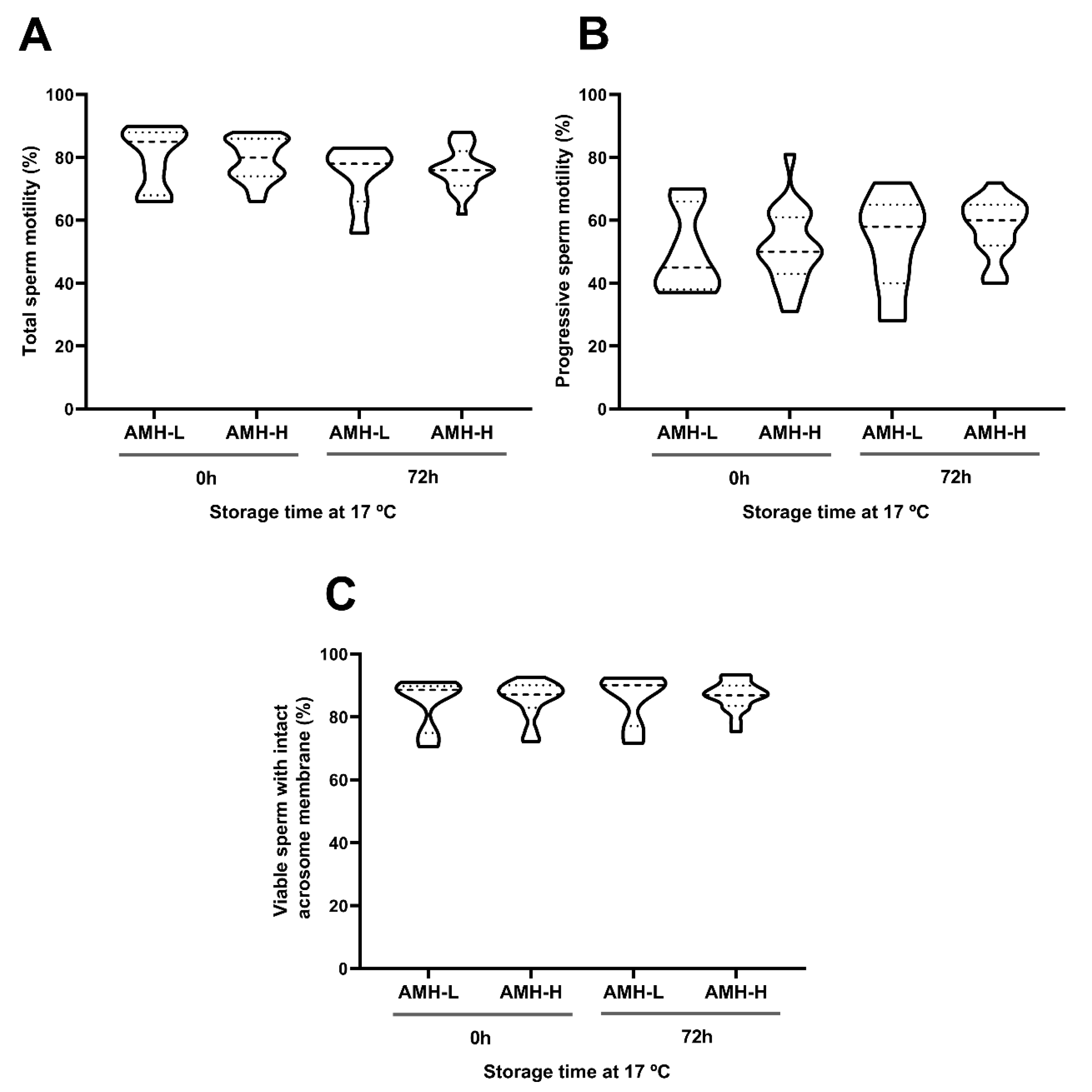
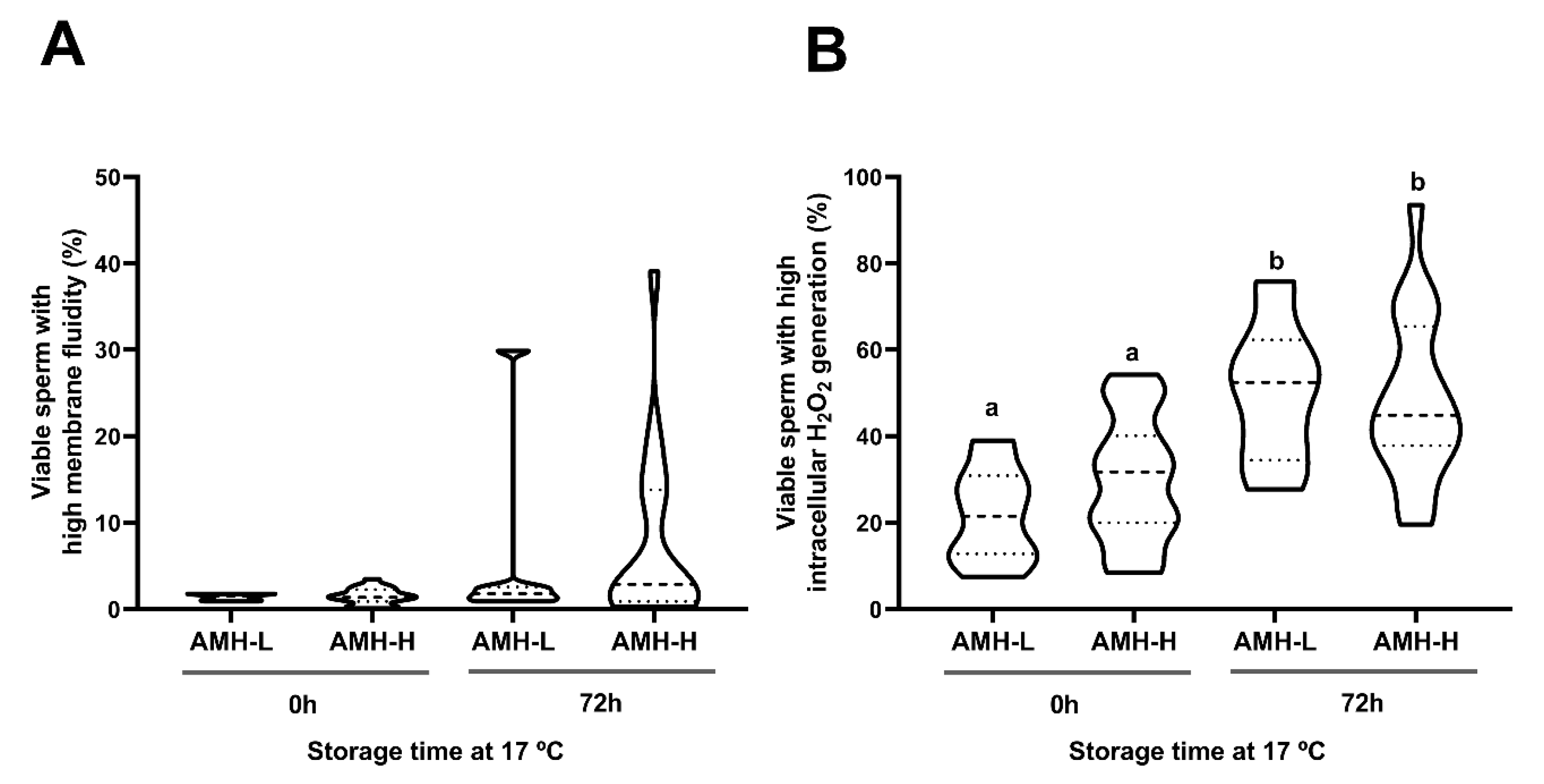
2.6.2. Experiment 2: Association Between SP-AMH Concentration and Sperm Quality and Functionality Parameters of Semen Samples Stored at 17 °C
2.6.3. Experiment 3: Association Between SP-AMH Concentration and In Vivo Fertility Outcomes of Semen Samples Stored at 17 °C
2.7. Statistical Analysis
3. Results
3.1. Experiment 1: SP-AMH Concentration Variability Among Breeds
3.2. Experiment 2: Association Between SP-AMH Concentration and Sperm Quality and Functionality Parameters of Semen Samples Stored at 17 °C
3.3. Experiment 3: Association Between SP-AMH Concentration and In Vivo Fertility Outcomes of Semen Samples Stored at 17 °C
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Roca, J.; Parrilla, I.; Bolarin, A.; Martinez, E.A.; Rodriguez-Martinez, H. Will AI in pigs become more efficient? Theriogenology 2016, 86, 187–193. [Google Scholar] [CrossRef] [PubMed]
- Flowers, W.L. Selection for boar fertility and semen quality—the way ahead. Soc. Reprod. Fertil. Suppl. 2009, 66, 67–78. [Google Scholar] [PubMed]
- Broekhuijse, M.L.W.J.; Feitsma, H.; Gadella, B.M. Field data analysis of boar semen quality. Reprod. Domest. Anim. 2011, 46, 59–63. [Google Scholar] [CrossRef] [PubMed]
- Broekhuijse, M.L.W.J.; Feitsma, H.; Gadella, B.M. Artificial insemination in pigs: Predicting male fertility. Vet. Q. 2012, 32, 151–157. [Google Scholar] [CrossRef][Green Version]
- Parrilla, I.; del Olmo, D.; Sijses, L.; Martinez-Alborcia, M.J.; Cuello, C.; Vazquez, J.M.; Martinez, E.A.; Roca, J. Differences in the ability of spermatozoa from individual boar ejaculates to withstand different semen-processing techniques. Anim. Reprod. Sci. 2012, 132, 66–73. [Google Scholar] [CrossRef]
- Roca, J.; Broekhuijse, M.L.W.J.; Parrilla, I.; Rodriguez-Martinez, H.; Martinez, E.A.; Bolarin, A. Boar Differences In Artificial Insemination Outcomes: Can They Be Minimized? Reprod. Domest. Anim. 2015, 50, 48–55. [Google Scholar] [CrossRef]
- Rodriguez-Martinez, H.; Kvist, U.; Ernerudh, J.; Sanz, L.; Calvete, J.J. Seminal plasma proteins: What role do they play? Am. J. Reprod. Immunol. 2011, 66, 11–22. [Google Scholar] [CrossRef]
- Barranco, I.; Tvarijonaviciute, A.; Perez-Patino, C.; Alkmin, D.V.; Ceron, J.J.; Martinez, E.A.; Rodriguez-Martinez, H.; Roca, J. The activity of paraoxonase type 1 (PON-1) in boar seminal plasma and its relationship with sperm quality, functionality, and in vivo fertility. Andrology 2015, 3, 315–320. [Google Scholar] [CrossRef]
- Barranco, I.; Tvarijonaviciute, A.; Perez-Patino, C.; Vicente-Carrillo, A.; Parrilla, I.; Ceron, J.J.; Martinez, E.A.; Rodriguez-Martinez, H.; Roca, J. Glutathione Peroxidase 5 Is Expressed by the Entire Pig Male Genital Tract and Once in the Seminal Plasma Contributes to Sperm Survival and In Vivo Fertility. PLoS ONE 2016, 11, e0162958. [Google Scholar] [CrossRef]
- Perez-Patino, C.; Barranco, I.; Ruber, M.; Padilla, L.; Alvarez-Rodriguez, M.; Parrilla, I.; Bolarin, A.; Martinez, E.A.; Rodriguez-Martinez, H.; Roca, J. TGF-β1 levels in seminal plasma relates positively to porcine in vivo fertility. Reprod. Domest. Anim. 2018, 53, 180–181. [Google Scholar]
- Barranco, I.; Padilla, L.; Perez-Patino, C.; Vazquez, J.M.; Martinez, E.A.; Rodriguez-Martinez, H.; Roca, J.; Parrilla, I. Seminal Plasma Cytokines Are Predictive of the Outcome of Boar Sperm Preservation. Front. Vet. Sci. 2019, 6, 436. [Google Scholar] [CrossRef] [PubMed]
- Fenichel, P.; Rey, R.; Poggioli, S.; Donzeau, M.; Chevallier, D.; Pointis, G. Anti-Mullerian hormone as a seminal marker for spermatogenesis in non-obstructive azoospermia. Hum. Reprod. 1999, 14, 2020–2024. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Mostafa, T.; Amer, M.K.; Abdel-Malak, G.; Nsser, T.A.; Zohdy, W.; Ashour, S.; El-Gayar, D.; Awad, H.H. Seminal plasma anti-Mullerian hormone level correlates with semen parameters but does not predict success of testicular sperm extraction (TESE). Asian J. Androl. 2007, 9, 265–270. [Google Scholar] [CrossRef] [PubMed]
- Zalata, A.A.; Hassan, A.H.; Nada, H.A.; Bragais, F.M.; Agarwal, A.; Mostafa, T. Follicle-stimulating hormone receptor polymorphism and seminal anti-Mullerian hormone in fertile and infertile men. Andrologia 2008, 40, 392–397. [Google Scholar] [CrossRef]
- Fujisawa, M.; Yamasaki, T.; Okada, H.; Kamidono, S. The significance of anti-Mullerian hormone concentration in seminal plasma for spermatogenesis. Hum. Reprod. 2002, 17, 968–970. [Google Scholar] [CrossRef]
- Josso, N.; Lamarre, I.; Picard, J.Y.; Berta, P.; Davies, N.; Morichon, N.; Peschanski, M.; Jeny, R. Anti-mullerian hormone in early human development. Early Hum. Dev. 1993, 33, 91–99. [Google Scholar] [CrossRef]
- Fallat, M.E.; Siow, Y.; Belker, A.M.; Boyd, J.K.; Yoffe, S.; MacLaughlin, D.T. The presence of mullerian inhibiting substance in human seminal plasma. Hum. Reprod. 1996, 11, 2165–2169. [Google Scholar] [CrossRef][Green Version]
- Nery, S.F.; Vieira, M.A.F.; Dela Cruz, C.; Lobach, V.N.M.; Del Puerto, H.L.; Torres, P.B.; Rocha, A.L.L.; Reis, A.B.; Reis, F.M. Seminal plasma concentrations of Anti-Mullerian hormone and inhibin B predict motile sperm recovery from cryopreserved semen in asthenozoospermic men: A prospective cohort study. Andrology 2014, 2, 918–923. [Google Scholar] [CrossRef]
- Andersen, J.M.; Herning, H.; Witczak, O.; Haugen, T.B. Anti-Mullerian hormone in seminal plasma and serum: Association with sperm count and sperm motility. Hum. Reprod. 2016, 31, 1662–1667. [Google Scholar] [CrossRef]
- Kucera, R.; Ulcova-Gallova, Z.; Windrichova, J.; Losan, P.; Topolcan, O. Anti-Mullerian hormone in serum and seminal plasma in comparison with other male fertility parameters. Syst. Biol. Reprod. Med. 2016, 62, 223–226. [Google Scholar] [CrossRef]
- La Marca, A.; Sighinolfi, G.; Radi, D.; Argento, C.; Baraldi, E.; Artenisio, A.C.; Stabile, G.; Volpe, A. Anti-Mullerian hormone (AMH) as a predictive marker in assisted reproductive technology (ART). Hum. Reprod. Update 2010, 16, 113–130. [Google Scholar] [CrossRef] [PubMed]
- Fallat, M.E.; Siow, Y.; Klar, E.A.; Belker, A.M.; MacLaughlin, D.T. The presence of Mullerian inhibiting substance binding sites in human sperm. J. Urol. 1998, 159, 2210–2214. [Google Scholar] [CrossRef]
- Guthrie, H.D.; Welch, G.R. Determination of intracellular reactive oxygen species and high mitochondrial membrane potential in Percoll-treated viable boar sperm using fluorescence-activated flow cytometry. J. Anim. Sci. 2006, 84, 2089–2100. [Google Scholar] [CrossRef] [PubMed]
- Broekhuijse, M.L.W.J.; Sostaric, E.; Feitsma, H.; Gadella, B.M. The value of microscopic semen motility assessment at collection for a commercial artificial insemination center, a retrospective study on factors explaining variation in pig fertility. Theriogenology 2012, 77, 1466–1479.e3. [Google Scholar] [CrossRef]
- Batista, E.O.S.; Macedo, G.G.; Sala, R.V.; Ortolan, M.D.D.V.; Sa Filho, M.F.; Del Valle, T.A.; Jesus, E.F.; Lopes, R.N.V.R.; Renno, F.P.; Baruselli, P.S. Plasma antimullerian hormone as a predictor of ovarian antral follicular population in Bos indicus (Nelore) and Bos taurus (Holstein) heifers. Reprod. Domest. Anim. 2014, 49, 448–452. [Google Scholar] [CrossRef]
- Broer, S.L.; Broekmans, F.J.M.; Laven, J.S.E.; Fauser, B.C.J.M. Anti-Mullerian hormone: Ovarian reserve testing and its potential clinical implications. Hum. Reprod. Update 2014, 20, 688–701. [Google Scholar] [CrossRef]
- Torres-Rovira, L.; Gonzalez-Bulnes, A.; Succu, S.; Spezzigu, A.; Manca, M.E.; Leoni, G.G.; Sanna, M.; Pirino, S.; Gallus, M.; Naitana, S.; et al. Predictive value of antral follicle count and anti-Mullerian hormone for follicle and oocyte developmental competence during the early prepubertal period in a sheep model. Reprod. Fertil. Dev. 2014, 26, 1094–1106. [Google Scholar] [CrossRef]
- Claes, A.; Ball, B.A.; Scoggin, K.E.; Esteller-Vico, A.; Kalmar, J.J.; Conley, A.J.; Squires, E.L.; Troedsson, M.H.T. The interrelationship between anti-Mullerian hormone, ovarian follicular populations and age in mares. Equine Vet. J. 2015, 47, 537–541. [Google Scholar] [CrossRef]
- Tanihara, F.; Hirata, M.; Iizuka, S.; Sairiki, S.; Nii, M.; Nguyen, N.T.; Le, Q.A.; Hirano, T.; Otoi, T. Relationship among ovarian follicular status, developmental competence of oocytes, and anti-Mullerian hormone levels: A comparative study in Japanese wild boar crossbred gilts and Large White gilts. Anim. Sci. J. 2019, 90, 712–718. [Google Scholar] [CrossRef]
- Matuszczak, E.; Hermanowicz, A.; Komarowska, M.; Debek, W. Serum AMH in Physiology and Pathology of Male Gonads. Int. J. Endocrinol. 2013, 2013, 128907. [Google Scholar] [CrossRef]
- Audet, I.; Berube, N.; Bailey, J.L.; Laforest, J.-P.; Quesnel, H.; Matte, J.J. Effects of dietary vitamin supplementation and semen collection frequency on hormonal profile during ejaculation in the boar. Theriogenology 2009, 71, 334–341. [Google Scholar] [CrossRef]
- Zhang, Q.; Bai, Q.; Yuan, Y.; Liu, P.; Qiao, J. Assessment of seminal estradiol and testosterone levels as predictors of human spermatogenesis. J. Androl. 2010, 31, 215–220. [Google Scholar] [CrossRef]
- Xu, H.-Y.; Zhang, H.-X.; Xiao, Z.; Qiao, J.; Li, R. Regulation of anti-Mullerian hormone (AMH) in males and the associations of serum AMH with the disorders of male fertility. Asian J. Androl. 2019, 21, 109–114. [Google Scholar]
- Lyet, L.; Louis, F.; Forest, M.G.; Josso, N.; Behringer, R.R.; Vigier, B. Ontogeny of reproductive abnormalities induced by deregulation of anti-mullerian hormone expression in transgenic mice. Biol. Reprod. 1995, 52, 444–454. [Google Scholar] [CrossRef][Green Version]
- Sriraman, V.; Niu, E.; Matias, J.R.; Donahoe, P.K.; MacLaughlin, D.T.; Hardy, M.P.; Lee, M.M. Mullerian inhibiting substance inhibits testosterone synthesis in adult rats. J. Androl. 2001, 22, 750–758. [Google Scholar]
- Duvilla, E.; Lejeune, H.; Trombert-Paviot, B.; Gentil-Perret, A.; Tostain, J.; Levy, R. Significance of inhibin B and anti-Mullerian hormone in seminal plasma: A preliminary study. Fertil. Steril. 2008, 89, 444–448. [Google Scholar] [CrossRef]
- Zura Zaja, I.; Samardzija, M.; Vince, S.; Vilic, M.; Majic-Balic, I.; Duricic, D.; Milinkovic-Tur, S. Differences in seminal plasma and spermatozoa antioxidative systems and seminal plasma lipid and protein levels among boar breeds and hybrid genetic traits. Anim. Reprod. Sci. 2016, 170, 75–82. [Google Scholar] [CrossRef]
- Perry, J.R.B.; McMahon, G.; Day, F.R.; Ring, S.M.; Nelson, S.M.; Lawlor, D.A. Genome-wide association study identifies common and low-frequency variants at the AMH gene locus that strongly predict serum AMH levels in males. Hum. Mol. Genet. 2016, 25, 382–388. [Google Scholar] [CrossRef]
- Rebourcet, D.; Darbey, A.; Monteiro, A.; Soffientini, U.; Tsai, Y.T.; Handel, I.; Pitetti, J.-L.; Nef, S.; Smith, L.B.; O’Shaughnessy, P.J. Sertoli Cell Number Defines and Predicts Germ and Leydig Cell Population Sizes in the Adult Mouse Testis. Endocrinology 2017, 158, 2955–2969. [Google Scholar] [CrossRef]
- Hutson, J.M.; Fallat, M.E.; Kamagata, S.; Donahoe, P.K.; Budzik, G.P. Phosphorylation events during Mullerian duct regression. Science 1984, 223, 586–589. [Google Scholar] [CrossRef]
- Burks, D.J.; Carballada, R.; Moore, H.D.; Saling, P.M. Interaction of a tyrosine kinase from human sperm with the zona pellucida at fertilization. Science 1995, 269, 83–86. [Google Scholar] [CrossRef]
- Al-Qahtani, A.; Muttukrishna, S.; Appasamy, M.; Johns, J.; Cranfield, M.; Visser, J.A.; Themmen, A.P.N.; Groome, N.P. Development of a sensitive enzyme immunoassay for anti-Mullerian hormone and the evaluation of potential clinical applications in males and females. Clin. Endocrinol. (Oxf). 2005, 63, 267–273. [Google Scholar] [CrossRef]
- Goulis, D.G.; Tsametis, C.; Iliadou, P.K.; Polychronou, P.; Kantartzi, P.-D.; Tarlatzis, B.C.; Bontis, I.N.; Papadimas, I. Serum inhibin B and anti-Mullerian hormone are not superior to follicle-stimulating hormone as predictors of the presence of sperm in testicular fine-needle aspiration in men with azoospermia. Fertil. Steril. 2009, 91, 1279–1284. [Google Scholar] [CrossRef]
- Carrarelli, P.; Rocha, A.L.L.; Belmonte, G.; Zupi, E.; Abrao, M.S.; Arcuri, F.; Piomboni, P.; Petraglia, F. Increased expression of antimullerian hormone and its receptor in endometriosis. Fertil. Steril. 2014, 101, 1353–1358. [Google Scholar] [CrossRef]
- Robertson, S.A.; Ingman, W.V.; O’Leary, S.; Sharkey, D.J.; Tremellen, K.P. Transforming growth factor beta-a mediator of immune deviation in seminal plasma. J. Reprod. Immunol. 2002, 57, 109–128. [Google Scholar] [CrossRef]


© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Barranco, I.; Fernandez-Fuertes, B.; Padilla, L.; Delgado-Bermúdez, A.; Tvarijonaviciute, A.; Yeste, M. Seminal Plasma Anti-Müllerian Hormone: A Potential AI-Boar Fertility Biomarker? Biology 2020, 9, 78. https://doi.org/10.3390/biology9040078
Barranco I, Fernandez-Fuertes B, Padilla L, Delgado-Bermúdez A, Tvarijonaviciute A, Yeste M. Seminal Plasma Anti-Müllerian Hormone: A Potential AI-Boar Fertility Biomarker? Biology. 2020; 9(4):78. https://doi.org/10.3390/biology9040078
Chicago/Turabian StyleBarranco, Isabel, Beatriz Fernandez-Fuertes, Lorena Padilla, Ariadna Delgado-Bermúdez, Asta Tvarijonaviciute, and Marc Yeste. 2020. "Seminal Plasma Anti-Müllerian Hormone: A Potential AI-Boar Fertility Biomarker?" Biology 9, no. 4: 78. https://doi.org/10.3390/biology9040078
APA StyleBarranco, I., Fernandez-Fuertes, B., Padilla, L., Delgado-Bermúdez, A., Tvarijonaviciute, A., & Yeste, M. (2020). Seminal Plasma Anti-Müllerian Hormone: A Potential AI-Boar Fertility Biomarker? Biology, 9(4), 78. https://doi.org/10.3390/biology9040078





