Venous Thrombo-Embolism in Hospitalized SARS-CoV-2 Patients Treated with Three Different Anticoagulation Protocols: Prospective Observational Study
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Outcomes
2.2. Laboratory Analysis and Imaging
2.3. Statistical Analysis
3. Results
3.1. VTE Prevalence
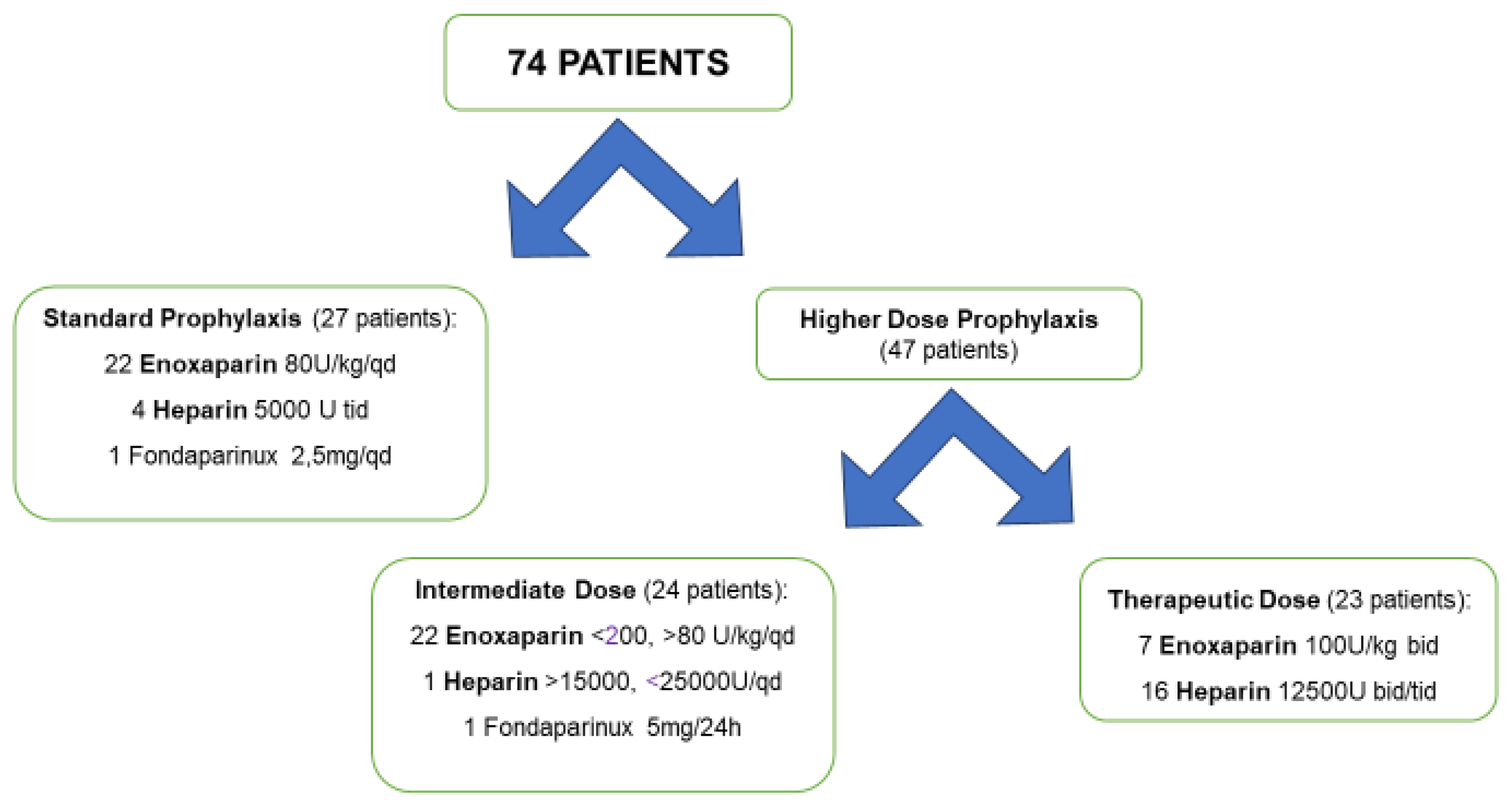
3.2. Antithrombotic Prophylaxis
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Ruan, Q.; Yang, K.; Wang, W.; Jiang, L.; Song, J. Clinical predictors of mortality due to COVID-19 based on an analysis of data of 150 patients from Wuhan, China. Intensive Care Med. 2020, 46, 846–848. [Google Scholar] [CrossRef]
- Han, H.; Yang, L.; Liu, R.; Liu, F.; Wu, K.L.; Li, J.; Liu, X.H.; Zhu, C.L. Prominent changes in blood coagulation of patients with SARS-CoV-2 infection. Clin. Chem. Lab. Med. 2020, 58, 1116–1120. [Google Scholar] [CrossRef] [PubMed]
- Huang, C.; Wang, Y.; Li, X.; Ren, L.; Zhao, J.; Hu, Y.; Zhang, L.; Fan, G.; Xu, J.; Gu, X.; et al. Clinical features of patients infected with 2019 novel coronavirus in Wuhan, China. Lancet 2020, 395, 497–506. [Google Scholar] [CrossRef]
- Chen, N.; Zhou, M.; Dong, X.; Qu, J.; Gong, F.; Han, Y.; Qiu, Y.; Wang, J.; Liu, Y.; Wei, Y.; et al. Epidemiological and clinical characteristics of 99 cases of 2019 novel coronavirus pneumonia in Wuhan, China: A descriptive study. Lancet 2020, 395, 507–513. [Google Scholar] [CrossRef]
- Tang, N.; Bai, H.; Chen, X.; Gong, J.; Li, D.; Sun, Z. Anticoagulant treatment is associated with decreased mortality in severe coronavirus disease 2019 patients with coagulopathy. J. Thromb. Haemost. 2020, 18, 1094–1099. [Google Scholar] [CrossRef]
- Varga, Z.; Flammer, A.J.; Steiger, P.; Haberecker, M.; Andermatt, R.; Zinkernagel, A.S.; Mehra, M.R.; Schuepbach, R.A.; Ruschitzka, F.; Moch, H. Endothelial cell infection and endotheliitis in COVID-19. Lancet 2020, 395, 1417–1418. [Google Scholar] [PubMed]
- Helms, J.; Tacquard, C.; Severac, F.; Leonard-Lorant, I.; Ohana, M.; Delabranche, X.; Merdji, H.; Clere-Jehl, R.; Schenck, M.; Fagot Gandet, F.; et al. CRICS TRIGGERSEP Group (Clinical Research in Intensive Care and Sepsis Trial Group for Global Evaluation and Research in Sepsis). High risk of thrombosis in patients with severe SARS-CoV-2 infection: A multicenter prospective cohort study. Intensive Care Med. 2020, 46, 1089–1098. [Google Scholar]
- Klok, F.A.; Kruip, M.J.H.A.; van der Meer, N.J.M.; Arbous, M.S.; Gommers, D.A.M.P.J.; Kant, K.M.; Kaptein, F.H.J.; van Paassen, J.; Stals, M.A.M.; Huisman, M.V.; et al. Incidence of thrombotic complications in critically ill ICU patients with COVID-19. Thromb. Res. 2020, 191, 145–147. [Google Scholar]
- Demelo-Rodríguez, P.; Cervilla-Muñoz, E.; Ordieres-Ortega, L.; Parra-Virto, A.; Toledano-Macías, M.; Toledo-Samaniego, N.; García-García, A.; García-Fernández-Bravo, I.; Ji, Z.; de-Miguel-Diez, J.; et al. Incidence of asymptomatic deep vein thrombosis in patients with COVID-19 pneumonia and elevated D-dimer levels. Thromb. Res. 2020, 192, 23–26. [Google Scholar] [CrossRef]
- Chi, G.; Lee, J.J.; Jamil, A.; Gunnam, V.; Najafi, H.; Memar Montazerin, S.; Shojaei, F.; Marszalek, J. Venous Thromboembolism among Hospitalized Patients with COVID-19 Undergoing Thromboprophylaxis: A Systematic Review and Meta-Analysis. J. Clin. Med. 2020, 9, 2489. [Google Scholar]
- Porfidia, A.; Pola, R. Venous thromboembolism and heparin use in COVID-19 patients: Juggling between pragmatic choices, suggestions of medical societies and the lack of guidelines. J. Thromb. Thrombolysis 2020, 50, 68–71. [Google Scholar] [CrossRef]
- Thachil, J.; Tang, N.; Gando, S.; Falanga, A.; Cattaneo, M.; Levi, M.; Clark, C.; Iba, T. ISTH interim guidance on recognition and management of coagulopathy in COVID-19. J. Thromb. Haemost. 2020, 18, 1023–1026. [Google Scholar] [CrossRef] [PubMed]
- Zhai, Z.; Li, C.; Chen, Y.; Gerotziafas, G.; Zhang, Z.; Wan, J.; Liu, P.; Elalamy, I.; Wang, C. Prevention Treatment of VTE Associated with COVID-19 Infection Consensus Statement Group. Prevention and Treatment of Venous Thromboembolism Associated with Coronavirus Disease 2019 Infection: A Consensus Statement before Guidelines. Thromb. Haemost. 2020, 120, 937–948. [Google Scholar] [CrossRef]
- Longhitano, Y.; Racca, F.; Zanza, C.; Piccioni, A.; Audo, A.; Muncinelli, M.; Santi, R.; Kozel, D.; Geraci, C.; Taverna, M.; et al. Venous thromboembolism in critically ill patients affected by ARDS related to Covid-19 in northen-west Italy. Eur. Rev. Med. Pharmacol. Sci. 2020, 24, 9154–9160. [Google Scholar] [PubMed]
- Alhazzani, W.; Møller, M.H.; Arabi, Y.M.; Loeb, M.; Gong, M.N.; Fan, E.; Oczkowski, S.; Levy, M.M.; Derde, L.; Dzierba, A.; et al. Surviving Sepsis Campaign: Guidelines on the management of critically ill adults with Coronavirus Disease 2019 (COVID-19). Intensive Care Med. 2020, 46, 854–887. [Google Scholar] [CrossRef] [PubMed]
- Lodigiani, C.; Iapichino, G.; Carenzo, L.; Cecconi, M.; Ferrazzi, P.; Sebastian, T.; Kucher, N.; Studt, J.D.; Sacco, C.; Alexia, B.; et al. Venous and arterial thromboembolic complications in COVID-19 patients admitted to an academic hospital in Milan, Italy. Thromb. Res. 2020, 191, 9–14. [Google Scholar] [CrossRef]
- Chi, G.; Goldhaber, S.; Hull, R.; Hernandez, A.; Kerneis, M.; Al Khalfan, F.; Cohen, A.; Harrington, R.; Gibson, C.M. Thrombus Burden of Deep Vein Thrombosis and Its Association with Thromboprophylaxis and D-Dimer Measurement: Insights from the APEX Trial. Thromb. Haemost. 2017, 117, 2389–2395. [Google Scholar] [CrossRef]
- Bikdeli, B.; Madhavan, M.V.; Jimenez, D.; Chuich, T.; Dreyfus, I.; Driggin, E.; Der Nigoghossian, C.; Ageno, W.; Madjid, M.; Guo, Y.; et al. COVID-19 and Thrombotic or Thromboembolic Disease: Implications for Prevention, Antithrombotic Therapy, and Follow-Up. J. Am. Coll. Cardiol. 2020, 75, 2950–2973. [Google Scholar] [CrossRef]
- Wichmann, D.; Sperhake, J.P.; Lütgehetmann, M.; Steurer, S.; Edler, C.; Heinemann, A.; Heinrich, F.; Mushumba, H.; Kniep, I.; Schröder, A.S.; et al. Autopsy findings and venous thromboembolism in patients with COVID-19: A prospective cohort study. Ann. Intern Med. 2020, 173, 268–277. [Google Scholar] [CrossRef]
- McKelvie, P.A. Autopsy evidence of pulmonary thromboembolism. Med. J. Aust. 1994, 160, 127–128. [Google Scholar] [CrossRef]
- Patel, R.; Cook, D.J.; Meade, M.O.; Griffith, L.E.; Mehta, G.; Rocker, G.M.; Marshall, J.C.; Hodder, R.; Martin, C.M.; Heyland, D.K.; et al. Burden of illness in venous thromboembolism in critical care: A multicenter observational study. J. Crit. Care 2005, 20, 341–347. [Google Scholar] [CrossRef] [PubMed]
- Chi, G.; Gibson, C.M.; Kalayci, A.; Cohen, A.T.; Hernandez, A.F.; Hull, R.D.; Kahe, F.; Jafarizade, M.; Sharfaei, S.; Liu, Y.; et al. Extended-duration betrixaban versus shorter-duration enoxaparin for venous thromboembolism prophylaxis in critically ill medical patients: An APEX trial substudy. Intensive Care Med. 2019, 45, 477–487. [Google Scholar] [CrossRef] [PubMed]
- Cui, S.; Chen, S.; Li, X.; Liu, S.; Wang, F. Prevalence of venous thromboembolism in patients with severe novel coronavirus pneumonia. J. Thromb. Haemost. 2020, 18, 1421–1424. [Google Scholar] [CrossRef] [PubMed]
- Poissy, J.; Goutay, J.; Caplan, M.; Parmentier, E.; Duburcq, T.; Lassalle, F.; Jeanpierre, E.; Rauch, A.; Labreuche, J.; Susen, S. Lille ICU Haemostasis COVID-19 Group. Pulmonary Embolism in COVID-19 Patients: Awareness of an Increased Prevalence. Circulation 2020, 142, 184–186. [Google Scholar] [CrossRef]
- Middeldorp, S.; Coppens, M.; van Haaps, T.F.; Foppen, M.; Vlaar, A.P.; Müller, M.C.A.; Bouman, C.C.S.; Beenen, L.F.M.; Kootte, R.S.; Heijmans, J.; et al. Incidence of venous thromboembolism in hospitalized patients with COVID-19. J. Thromb. Haemost. 2020, 18, 1995–2002. [Google Scholar] [CrossRef]
- Nahum, J.; Morichau-Beauchant, T.; Daviaud, F.; Echegut, P.; Fichet, J.; Maillet, J.M.; Thierry, S. Venous Thrombosis Among Critically Ill Patients with Coronavirus Disease 2019 (COVID-19). JAMA Netw. Open 2020, 3, e2010478. [Google Scholar] [CrossRef]
- Llitjos, J.F.; Leclerc, M.; Chochois, C.; Monsallier, J.M.; Ramakers, M.; Auvray, M.; Merouani, K. High incidence of venous thromboembolic events in anticoagulated severe COVID-19 patients. J. Thromb. Haemost. 2020, 18, 1743–1746. [Google Scholar] [CrossRef]
- Santoliquido, A.; Porfidia, A.; Nesci, A.; De Matteis, G.; Marrone, G.; Porceddu, E.; Cammà, G.; Giarretta, I.; Fantoni, M.; Landi, F.; et al. Incidence of deep vein thrombosis among non-ICU patients hospitalized for COVID-19 despite pharmacological thromboprophylaxis. J. Thromb. Haemost. 2020. [Google Scholar] [CrossRef]
- Artifoni, M.; Danic, G.; Gautier, G.; Gicquel, P.; Boutoille, D.; Raffi, F.; Néel, A.; Lecomte, R. Systematic assessment of venous thromboembolism in COVID-19 patients receiving thromboprophylaxis: Incidence and role of D-dimer as predictive factors. J. Thromb. Thrombolysis 2020, 50, 211–216. [Google Scholar] [CrossRef]
- Marongiu, F.; Grandone, E.; Barcellona, D. Pulmonary thrombosis in 2019-nCoV pneumonia? J. Thromb. Haemost. 2020, 18, 1511–1513. [Google Scholar] [CrossRef]
- Marongiu, F.; Mameli, A.; Grandone, E.; Barcellona, D. Pulmonary Thrombosis: A Clinical Pathological Entity Distinct from Pulmonary Embolism? Semin. Thromb. Hemost. 2019, 45, 778–783. [Google Scholar] [CrossRef] [PubMed]
- Terpos, E.; Ntanasis-Stathopoulos, I.; Elalamy, I.; Kastritis, E.; Sergentanis, T.N.; Politou, M.; Psaltopoulou, T.; Gerotziafas, G.; Dimopoulos, M.A. Hematological findings and complications of COVID-19. Am. J. Hematol. 2020, 95, 834–847. [Google Scholar] [CrossRef] [PubMed]
- Vitacca, M.; Nava, S.; Santus, P.; Harari, S. Early consensus management for non-ICU acute respiratory failure SARS-CoV-2 emergency in Italy: From ward to trenches. Eur. Respir. J. 2020, 55, 2000632. [Google Scholar] [CrossRef] [PubMed]
- Parke, R.L.; Eccleston, M.L.; McGuinness, S.P. The effects of flow on airway pressure during nasal high-flow oxygen therapy. Respir. Care 2011, 56, 1151–1155. [Google Scholar] [CrossRef]
- Kaplan, D.; Casper, T.C.; Elliott, C.G.; Men, S.; Pendleton, R.C.; Kraiss, L.W.; Weyrich, A.S.; Grissom, C.K.; Zimmerman, G.A.; Rondina, M.T. VTE incidence and risk factors in patients with severe sepsis and septic shock. Chest 2015, 148, 1224–1230. [Google Scholar] [CrossRef]
- Geerts, W.; Cook, D.; Selby, R.; Etchells, E. Venous thromboembolism and its prevention in critical care. J. Crit. Care 2002, 17, 95–104. [Google Scholar] [CrossRef]
- Caprini, J.A. Thrombosis risk assessment as a guide to quality patient care. Dis. Mon 2005, 51, 70–78. [Google Scholar] [CrossRef]
- White, D.; MacDonald, S.; Bull, T.; Hayman, M.; de Monteverde-Robb, R.; Sapsford, D.; Lavinio, A.; Varley, J.; Johnston, A.; Besser, M.; et al. Heparin resistance in COVID-19 patients in the intensive care unit. J. Thromb. Thrombolysis 2020, 50, 287–291. [Google Scholar] [CrossRef]
- Connors, J.M.; Levy, J.H. COVID-19 and its implications for thrombosis and anticoagulation. Blood 2020, 135, 2033–2040. [Google Scholar] [CrossRef]
- Ciavarella, A.; Peyvandi, F.; Martinelli, I. Where do we stand with antithrombotic prophylaxis in patients with COVID-19? Thromb. Res. 2020, 191, 29. [Google Scholar] [CrossRef]
| ICU (18 Patients) | General Ward (56 Patients) | Total (74 Patients) | |
|---|---|---|---|
| Gender, n (%) | |||
| Male | 15 (83.3%) | 29 (51.8%) | 44 (59.5%) |
| Female | 3 (16.6%) | 27 (48.2%) | 30 (40.5%) |
| Age, (years) | 60.22 (±10.49) | 71.46 (±15.45) | 68.65 (±15.12) p = 0.002 |
| Comorbidities, n (%) | |||
| AF | 1 (5.5%) | 2 (4.3%) | 3 (4%) |
| CRF | 3 (16.6%) | 4 (8.7%) | 7 (9.5%) |
| Obesity | 4 (22.2%) | 2 (4.3%) | 6 (8.1%) |
| DM | 5 (27.7%) | 12 (26.1%) | 17 (22.9%) |
| Cardiovascular disease | 9 (50%) | 23 (50%) | 32 (43.3%) |
| COPD | 2 (11.1%) | 6 (13.0%) | 8 (10.8%) |
| Asthma | 0 (0%) | 0 (0%) | 0 (0%) |
| Cancer | 2 (11.1%) | 6 (13%) | 8 (10.8%) |
| Days from hospital admission to CDUS | - | - | 9 <5–18.7> |
| Days from symptoms onset to CDUS | - | - | 14 <6–23.7> |
| Unilateral | Bilateral | Total | |
|---|---|---|---|
| DVT, proximal | 1 | 4 | 5 |
| DVT, distal | 2 | - | 2 |
| DVT proximal + superficial, lower limbs | 2 | - | 2 |
| Superficial, lower limbs | 4 | 5 | 9 |
| Superficial, lower limbs + jugular | 1 | - | 1 |
| Superficial, upper limbs + jugular | 1 | - | 1 |
| Jugular, isolated | 1 | - | 1 |
| Venous Thrombosis Positive (21 Patients) | Venous Thrombosis Negative (53 Patients) | ODD Ratio | p-Value | |
|---|---|---|---|---|
| Gender, n (%) | ||||
| Male | 15 (71.4%) | 29 (54.7%) | 2.07 <0.71–5.98> | 0.197 |
| Female | 6 (28.6%) | 24 (45.3%) | ||
| Age, (years) | 66.7 (±16.78) | 69.40 (±14.54) | - | 0.529 |
| Comorbidities, n (%) | ||||
| AF | 0 (0%) | 3 (5.7%) | - | 0.266 |
| CRF | 2 (9.5%) | 3 (5.7%) | 1.75 <0.32–9.65> | 0.551 |
| Obesity | 1 (4.8%) | 6 (11.3%) | 0.39 <0.06–2.49> | 0.385 |
| DM | 5 (23.8%) | 11 (20.7%) | 1.19 <0.37–3.82> | 0.774 |
| Cardiovascular disease | 8 (38.1%) | 24 (45.3%) | 0.74 <0.27–2.04> | 0.574 |
| COPD | 2 (9.5%) | 6 (11.3%) | 0.82 <0.17–3.89> | 0.822 |
| Asthma | 0 (0%) | 0 (0%) | - | - |
| Cancer | 1 (4.8%) | 9 (16.9%) | 0.24 <0.4–1.48> | 0.166 |
| PaO2/FiO2 | 204 <180–236> | 235 <184–281> | - | 0.280 |
| Patients underwent positive pressure respiratory assistance, n (%) | 19 (90.5%) | 34 (64.1%) | 5.31 <1.27–22.16> | 0.024 |
| Patients allocation, n (%) | ||||
| ICU | 12 (57.1%) | 6 (11.3%) | 10.44 <3.22–33.86> | <0.0001 |
| General ward | 9 (42.8%) | 47 (88.7%) | ||
| Laboratory tests results | ||||
| LDH, (U/L) | 779 <631–867> | 481 <359–697> | - | 0.001 |
| Ferritin, (mcg/L) | 1379 <644.4–1590> | 623.7 <356.5–1093> | - | 0.333 |
| CRP, (mg/dl) | 13.78 <4.2–21.6> | 1.56 <0.54–6.53> | - | 0.001 |
| Lymphocytes, (×10/mcL3) | 0.74 (±0.14) | 0.82 (±0.29) | - | 0.371 |
| Coagulation test results | ||||
| Fibrinogen, (mg/dL) | 645 <421.5–795.5> | 505 <413–633.5> | - | 0.352 |
| D-dimer, (mcg/mL) | 2.78 <1.42–5.73> | 1.08 <0.45–1.59> | - | <0.0001 |
| aPTT, (s) | 44.72 (±15.92) | 42.12 (±9.8) | - | 0.458 |
| PT, (s) | 15.3 (±2.6) | 14.3 (±2.6) | - | 0.158 |
| PLTS, (×103/mcL) | 299.6 (±145.3) | 254.3 (±116.3) | - | 0.171 |
| Hospital LOS, (days) | 45.7 (±15.3) | 29 (±19.5) | - | 0.152 |
| Mortality | 3 (14.3%) | 9 (16.9%) | 0.81 <0.21–3.11> | 0.777 |
| Standard Antithrombotic Prophylaxis (27 Patients) | Higher Dose Antithrombotic Prophylaxis (47 Patients) | ODD Ratio | p-Value | |
|---|---|---|---|---|
| Gender, n(%) | ||||
| Male | 14 (51.8%) | 30 (63.8%) | 1.639 <0.636–4.220> | 0.312 |
| Female | 13 (48.1%) | 17 (36.2%) | ||
| Age, | 65.69 (±12.77) | 70.33 (±16.19) | - | 0.092 |
| Comorbidities, n (%) | ||||
| AF | 0 (0%) | 3 (6.4%) | - | 0.180 |
| CRF | 1 (3.7%) | 4 (8.5%) | 2.419 <0.358–16.33> | 0.428 |
| Obesity | 3 (11.1%) | 4 (8.5%) | 0.744 <0.169–3.276> | 0.713 |
| DM | 4 (14.8%) | 12 (25.5%) | 1.971 <0.596–6.525> | 0.281 |
| Cardiovascular disease | 9 (33.3%) | 23 (48.9%) | 1.917 <0.73–5.034> | 0.192 |
| COPD | 2 (7.4%) | 6 (12.8%) | 1.829 <0.392–8.531> | 0.475 |
| Asthma | 0 (0%) | 0 (0%) | - | - |
| Cancer | 5 (18.5%) | 5 (10.6%) | 0.524 <0.145–1.898> | 0.34 |
| PaO2/FiO2, | 255 <220–285> | 214 <175–246> | - | 0.063 |
| Patients underwent positive pressure respiratory assistance, n (%) | 22 (81.4%) | 31 (65.9%) | 0.44 <0.15–1.33> | 0.154 |
| Thrombotic events, n (%) | 10 (37%) | 11 (23.4%) | 0.516 <0.189–1.429> | 0.210 |
| Patients allocation, n (%) | ||||
| General ward | 21 (77.7%) | 35 (74.5%) | 1.2 <0.40–3.56> | 0.749 |
| ICU | 6 (22.2%) | 12 (25.5%) | ||
| Laboratory test results | ||||
| CRP, (mg/dL) | 1.81 <0.66–12.51> | 3.48 <0.96–12.31> | - | 0.380 |
| LDH, (U/L) | 631 <360.5–818.5> | 371 <443.0–752.5> | - | 0.623 |
| D-Dimer, (mcg/mL) | 0.75 <0.34–2.73> | 1.44 <0.94–2.02> | - | 0.203 |
| Hospital LOS, (days) | 31.7 (±24.3) | 37.26 (±28.5) | - | 0.479 |
| Mortality | 2 (7.4%) | 10 (21.3%) | 3.38 <0.78–14.67> | 0.119 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Longhitano, Y.; Racca, F.; Zanza, C.; Muncinelli, M.; Guagliano, A.; Peretti, E.; Minerba, A.C.; Mari, M.; Boverio, R.; Salio, M.; et al. Venous Thrombo-Embolism in Hospitalized SARS-CoV-2 Patients Treated with Three Different Anticoagulation Protocols: Prospective Observational Study. Biology 2020, 9, 310. https://doi.org/10.3390/biology9100310
Longhitano Y, Racca F, Zanza C, Muncinelli M, Guagliano A, Peretti E, Minerba AC, Mari M, Boverio R, Salio M, et al. Venous Thrombo-Embolism in Hospitalized SARS-CoV-2 Patients Treated with Three Different Anticoagulation Protocols: Prospective Observational Study. Biology. 2020; 9(10):310. https://doi.org/10.3390/biology9100310
Chicago/Turabian StyleLonghitano, Yaroslava, Fabrizio Racca, Christian Zanza, Marina Muncinelli, Alberto Guagliano, Elisa Peretti, Anna Chiara Minerba, Marta Mari, Riccardo Boverio, Mario Salio, and et al. 2020. "Venous Thrombo-Embolism in Hospitalized SARS-CoV-2 Patients Treated with Three Different Anticoagulation Protocols: Prospective Observational Study" Biology 9, no. 10: 310. https://doi.org/10.3390/biology9100310
APA StyleLonghitano, Y., Racca, F., Zanza, C., Muncinelli, M., Guagliano, A., Peretti, E., Minerba, A. C., Mari, M., Boverio, R., Salio, M., Chichino, G., Franceschi, F., Piccioni, A., Abenavoli, L., Salvini, M., & Artico, M. (2020). Venous Thrombo-Embolism in Hospitalized SARS-CoV-2 Patients Treated with Three Different Anticoagulation Protocols: Prospective Observational Study. Biology, 9(10), 310. https://doi.org/10.3390/biology9100310







