The Dynamics of the Cell Wall Proteome of Developing Alfalfa Stems
Abstract
1. Introduction
2. Materials and Methods
2.1. Plant Material
2.2. Cell Wall Protein Enrichment
2.2.1. Extraction of Cell Wall Proteins
2.2.2. Concentration and Desalting of the Extracts.
2.3. Cell Wall Protein Analysis by 2D-DIGE
2.3.1. Protein Migration
2.3.2. Spot Selection
2.3.3. Spot Picking, Digestion, MS/MS Analysis
2.3.4. Post-Identification Statistical Procedure
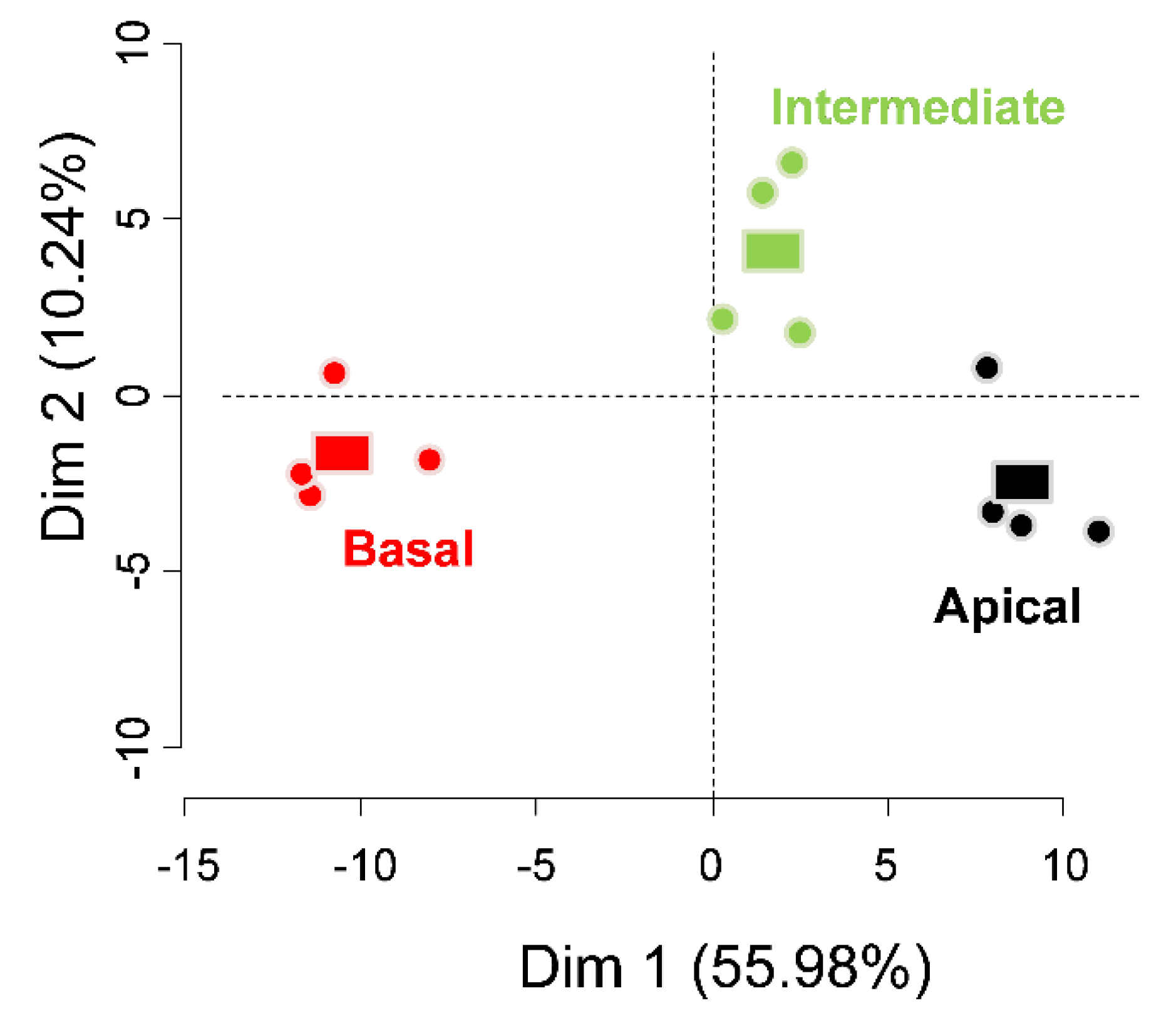
3. Results
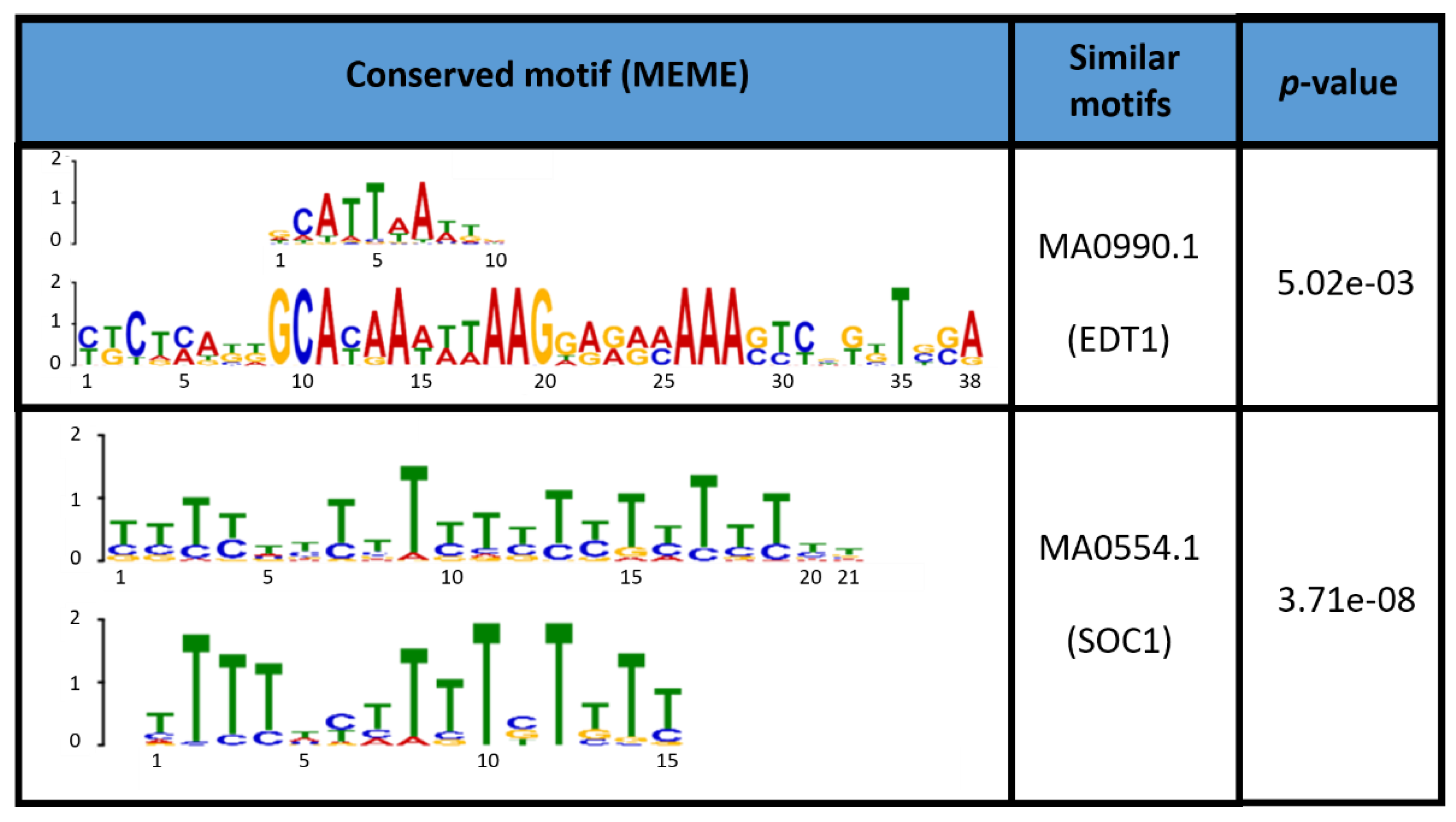
4. Discussion
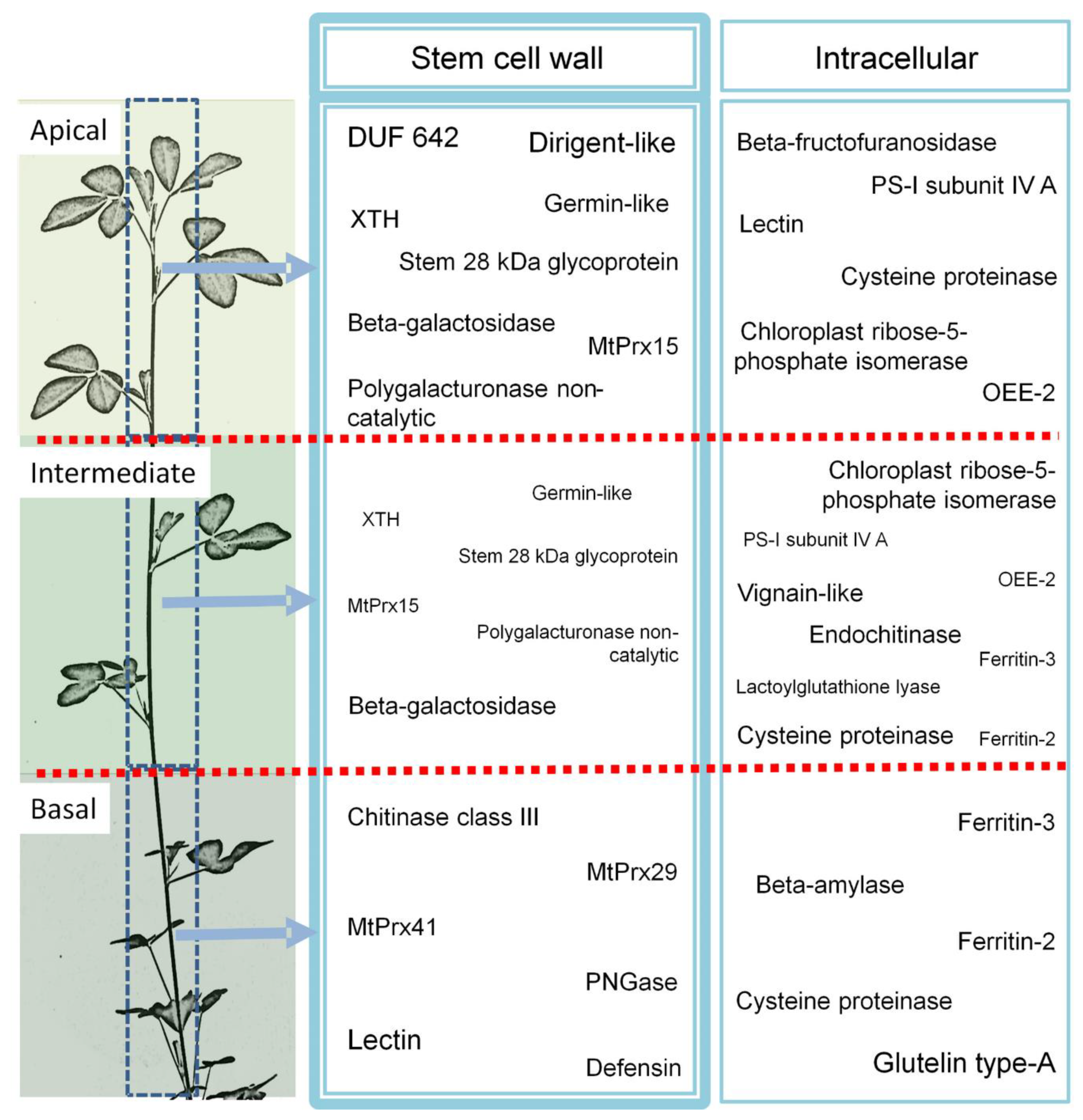
4.1. Carbohydrate-Interacting Proteins Accumulate in the Cell Wall of the Apical Region
4.2. β-galactosidases are Abundant in the Apical and Intermediate Stem Part
4.3. Class III Peroxidases MtPrx29, MtPrx41, and MtPrx15
4.4. Enzymes Involved in Protein Degradation/Maturation Accumulate in the Oldest Stem Region
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Guerriero, G.; Hausman, J.F.; Cai, G. No Stress! Relax! Mechanisms governing growth and shape in plant cells. Int. J. Mol. Sci. 2014, 15, 5094–5114. [Google Scholar] [CrossRef] [PubMed]
- Underwood, W. The plant cell wall: A dynamic barrier against pathogen invasion. Front. Plant Sci. 2012, 3, 85. [Google Scholar] [CrossRef] [PubMed]
- Cosgrove, D.J. Plant cell wall extensibility: Connecting plant cell growth with cell wall structure, mechanics, and the action of wall-modifying enzymes. J. Exp. Bot. 2016, 67, 463–476. [Google Scholar] [CrossRef] [PubMed]
- Houston, K.; Tucker, M.R.; Chowdhury, J.; Shirley, N.; Little, A. The plant cell wall: A complex and dynamic structure as revealed by the responses of genes under stress conditions. Front. Plant Sci. 2016, 7, 984. [Google Scholar] [CrossRef] [PubMed]
- Cassab, G.I.; Varner, J.E. Cell wall proteins. Annu. Rev. Plant Physiol. Plant Mol. Biol. 1988, 39, 321–353. [Google Scholar] [CrossRef]
- Boaretto, L.F.; Mazzafera, P. The proteomes of feedstocks used for the production of second-generation ethanol: A lacuna in the biofuel era. Ann. Appl. Biol. 2013, 163, 12–22. [Google Scholar] [CrossRef]
- Rose, J.K.C.; Lee, S.J. Straying off the highway: Trafficking of secreted plant proteins and complexity in the plant cell wall proteome. Plant Physiol. 2010, 153, 433–436. [Google Scholar] [CrossRef]
- Pinedo, M.; Regente, M.; Elizande, M.; Quiroga, I.Y.; Pagnussat, A.L.; Jorrin-Novo, J.; Maldonado, A.; de la Canal, L. Extracellular sunflower proteins: Evidence on non-classical secretion of a jacalin-related lectin. Protein Pep. Lett. 2012, 19, 270–276. [Google Scholar] [CrossRef]
- Rayon, C.; Lerouge, P.; Faye, L. The protein N-glycosylation in plants. J. Exp. Bot. 1998, 49, 1463–1472. [Google Scholar] [CrossRef]
- Hijazi, M.; Velasquez, S.M.; Jamet, E.; Estevez, J.M.; Albenne, C. An update on post-translational modifications of hydroxyproline-rich glycoproteins: Toward a model highlighting their contribution to plant cell wall architecture. Front. Plant Sci. 2014, 5, 395. [Google Scholar] [CrossRef]
- de la Canal, L.; Pinedo, M. Extracellular vesicles: A missing component in plant cell wall remodeling. J. Exp. Bot. 2018, 69, 4655–4658. [Google Scholar] [CrossRef] [PubMed]
- Yun, H.S.; Kwon, C.; Kang, B.G.; Lee, J.S.; Han, T.J.; Chang, S.C.; Kim, S.K. A xyloglucan endotransglycosylase/hydrolase1, VrXTH1, is associated with cell elongation in mungbean hypocotyls. Physiol. Plant. 2005, 125, 106–113. [Google Scholar] [CrossRef]
- Micheli, F. Pectin methylesterases: Cell wall enzymes with important roles in plant physiology. Trends Plant Sci. 2001, 6, 414–419. [Google Scholar] [CrossRef]
- Altmann, F.; Paschinger, K.; Dalik, T.; Vorauer, K. Characterisation of peptide-N4-(N-acetyl-β-glucosaminyl)asparagine amidase A and its N-glycans. Eur. J. Biochem. 1998, 252, 118–123. [Google Scholar] [CrossRef] [PubMed]
- Gantulga, D.; Ahn, Y.O.; Zhou, C.; Battogtokh, D.; Bevan, D.R.; Winkel, B.S.J.; Esen, A. Comparative characterization of the Arabidopsis subfamily a1 β-galactosidases. Phytochemistry 2009, 70, 1999–2009. [Google Scholar] [CrossRef]
- Vazquez-Lobo, A.; Roujol, D.; Zuniga-Sanchez, E.; Albenne, C.; Pinero, D.; Gamboa deBuen, A.; Jamet, E. The highly conserved spermatophyte cell wall DUF642 protein family: Phylogeny and first evidence of interaction with cell wall polysaccharides in vitro. Mol. Phylogenet. Evol. 2012, 63, 510–520. [Google Scholar] [CrossRef]
- Zhao, Q.; Nakashima, J.; Chen, F.; Yin, Y.; Fu, C.; Yun, J.; Shao, H.; Wang, X.; Wang, Z.Y.; Dixon, R.A. LACCASE is necessary and nonredundant with PEROXIDASE for lignin polymerization during vascular development in Arabidopsis. Plant Cell 2013, 25, 3976–3987. [Google Scholar] [CrossRef]
- Zúñiga-Sánchez, E.; Gamboa-deBuen, A. The two DUF642 At5g11420 and At4g32460-encoded proteins interact in vitro with the AtPME3 catalytic domain. In Protein Interactions; Cai, J., Wang, R.E., Eds.; InTechOpen: Rijeka, Croatia, 2012; Chapter 7; p. 464. [Google Scholar] [CrossRef]
- Verdonk, J.C.; Hatfield, R.D.; Sullivan, M.L. Proteomic analysis of cell walls of two developmental stages of alfalfa stems. Front. Plant Sci. 2012, 3, 279. [Google Scholar] [CrossRef]
- Feiz, L.; Irshad, M.; Pont-Lezica, R.F.; Canut, H.; Jamet, E. Evaluation of cell wall preparations for proteomics: A new procedure for purifying cell walls from Arabidopsis hypocotyls. Plant Methods 2006, 2, 10. [Google Scholar] [CrossRef]
- Printz, B.; Dos Santos Morais, R.; Wienkoop, S.; Sergeant, K.; Lutts, S.; Hausman, J.F.; Renaut, J. An improved protocol to study the plant cell wall proteome. Front. Plant Sci. 2015, 6, 237. [Google Scholar] [CrossRef]
- Barros, J.; Temple, S.; Dixon, R.A. Development and commercialization of reduced lignin alfalfa. Curr. Opin. Biotechnol. 2019, 56, 48–54. [Google Scholar] [CrossRef]
- Gutsch, A.; Keunen, E.; Guerriero, G.; Renaut, J.; Cuypers, A.; Hausman, J.F.; Sergeant, K. Long-term cadmium exposure influences the abundance of proteins that impact the cell wall structure in Medicago sativa stems. Plant Biol. 2018, 20, 1023–1035. [Google Scholar] [CrossRef]
- Gutsch, A.; Sergeant, K.; Keunen, E.; Prinsen, E.; Guerriero, G.; Renaut, J.; Hausman, J.F.; Cuypers, A. Does long-term cadmium exposure influence the composition of pectic polysaccharides in the cell wall of Medicago sativa stems? BMC Plant Biol. 2019, 19, 271. [Google Scholar] [CrossRef]
- Printz, B.; Guerriero, G.; Sergeant, K.; Renaut, J.; Lutts, S.; Hausman, J.F. Ups and downs in alfalfa: Proteomic and metabolic changes occurring in the growing stem. Plant Sci. 2015, 238, 13–25. [Google Scholar] [CrossRef][Green Version]
- Duruflé, H.; San Clemente, H.; Balliau, T.; Zivy, M.; Dunand, C.; Jamet, E. Cell wall proteome analysis of Arabidopsis thaliana mature stems. Proteomics 2017, 17, 1600449. [Google Scholar] [CrossRef]
- Calderan-Rodrigues, M.J.; Jamet, E.; Douché, T.; Bonassi, M.B.; Cataldi, T.R.; Fonseca, J.G.; San Clemente, H.; Pont-Lezica, R.; Labate, C.A. Cell wall proteome of sugarcane stems: Comparison of a destructive and a non-destructive extraction method showed differences in glycoside hydrolases and peroxidases. BMC Plant Biol. 2016, 16, 14. [Google Scholar] [CrossRef]
- Chabi, M.; Goulas, E.; Leclercq, C.C.; de Waele, I.; Rihouey, C.; Cenci, U.; Day, A.; Blervacq, A.S.; Neutelings, G.; Duponchel, L.; et al. A cell wall proteome and targeted cell wall analyses provide novel information on hemicellulose metabolism in flax. Mol. Cell. Proteom. 2017, 16, 1634–1651. [Google Scholar] [CrossRef]
- Komatsu, S.; Yanagawa, Y. Cell wall proteomics of crops. Front. Plant Sci. 2013, 4, 17. [Google Scholar] [CrossRef]
- Sergeant, K.; Printz, B.; Gutsch, A.; Behr, M.; Renaut, J.; Hausman, J.F. Didehydrophenylalanine, an abundant modification in the beta subunit of plant polygalacturonases. PLoS ONE 2017, 12, e0171990. [Google Scholar] [CrossRef]
- Shpak, E.; Leykam, J.F.; Kieliszewski, M.J. Synthetic genes for glycoprotein design and the elucidation of hydroxyproline-O-glycosylation codes. Proc. Natl. Acad. Sci. USA 1999, 96, 14736–14741. [Google Scholar] [CrossRef]
- Hoagland, D.R.; Arnon, D.I. The water-culture method for growing plants without soil. Circ. Calif. Agric. Exp. Stn. 1950, 347, 32. [Google Scholar]
- Printz, B.; Sergeant, K.; Lutts, S.; Guignard, C.; Renaut, J.; Hausman, J.F. From tolerance to acute metabolic deregulation: Contribution of proteomics to dig into the molecular response of alder-species under a polymetallic exposure. J. Proteome Res. 2013, 12, 5160–5179. [Google Scholar] [CrossRef]
- Almagro Armenteros, J.J.; Sønderby, C.K.; Sønderby, S.K.; Nielsen, H.; Winther, O. DeepLoc: Prediction of protein subcellular localization using deep learning. Bioinformatics 2017, 33, 3387–3395. [Google Scholar] [CrossRef]
- Lê, S.; Josse, J.; Husson, F. FactoMineR: An R package for multivariate analysis. J. Stat. Softw. 2008, 25, 1–18. [Google Scholar] [CrossRef]
- Fawal, N.; Li, Q.; Savelli, B.; Brette, M.; Passaia, G.; Fabre, M.; Mathé, C.; Dunand, C. PeroxiBase: A database for large-scale evolutionary analysis of peroxidases. Nucleic Acids Res. 2013, 41, D441–D444. [Google Scholar] [CrossRef]
- Watson, B.S.; Lei, Z.; Dixon, R.A.; Sumner, L.W. Proteomics of Medicago sativa cell walls. Phytochemistry 2004, 65, 1709–1720. [Google Scholar] [CrossRef]
- Irshad, M.; Canut, H.; Borderies, G.; Pont-Lezica, R.; Jamet, E. A new picture of cell wall protein dynamics in elongating cells of Arabidopsis thaliana: Confirmed actors and newcomers. BMC Plant Biol. 2008, 8, 94. [Google Scholar] [CrossRef]
- Douché, T.; San Clemente, H.; Burlat, V.; Roujol, D.; Valot, B.; Zivy, M.; Pont-Lezica, R.; Jamet, E. Brachypodium distachyon as a model plant toward improved biofuel crops: Search for secreted proteins involved in biogenesis and disassembly of cell wall polymers. Proteomics 2013, 13, 2438–2454. [Google Scholar] [CrossRef]
- Emanuelsson, O.; Brunak, S.; von Heijne, G.; Nielsen, H. Locating proteins in the cell using TargetP, SignalP and related tools. Nat. Protoc. 2007, 2, 953–971. [Google Scholar] [CrossRef]
- Neuhaus, J.M.; Pietrzak, M.; Boller, T. Mutation analysis of the C-terminal vacuolar targeting peptide of tobacco chitinase: Low specificity of the sorting system, and gradual transition between intracellular retention and secretion into the extracellular space. Plant J. 1994, 5, 45–54. [Google Scholar] [CrossRef]
- Jamet, E.; Albenne, C.; Boudart, G.; Irshad, M.; Canut, H.; Pont-Lezica, R. Recent advances in plant cell wall proteomics. Proteomics 2008, 8, 893–908. [Google Scholar] [CrossRef]
- Wasąg, P.; Suwińska, A.; Zakrzewski, P.; Walczewski, J.; Lenartowski, R.; Lenartowska, M. Calreticulin localizes to plant intra/extracellular peripheries of highly specialized cells involved in pollen-pistil interactions. Protoplasma 2018, 255, 57–67. [Google Scholar] [CrossRef]
- Jia, X.Y.; He, L.H.; Jing, R.L.; Li, R.Z. Calreticulin: Conserved protein and diverse functions in plants. Physiol. Plant. 2009, 136, 127–138. [Google Scholar] [CrossRef]
- Hojrup, P.; Roepstorff, P.; Houen, G. Human placental calreticulin characterization of domain structure and post-translational modifications. Eur. J. Biochem. 2001, 268, 2558–2565. [Google Scholar] [CrossRef]
- Cai, Y.-M.; Yu, J.; Ge, Y.; Mironov, A.; Gallois, P. Two proteases with caspase-3-like activity, cathepsin B and proteasome, antagonistically control ER-stress-induced programmed cell death in Arabidopsis. New Phytol. 2018, 218, 1143–1155. [Google Scholar] [CrossRef]
- Tarr, M.; Young, P.J.; Morse, R.; Shaw, D.J.; Haigh, R.; Petrov, P.G.; Johnson, S.J.; Winyard, P.G.; Eggleton, P. A mechanism of release of calreticulin from cells during apoptosis. J. Mol. Biol. 2010, 401, 799–812. [Google Scholar] [CrossRef]
- Toby, T.K.; Fornelli, L.; Kelleher, N.L. Progress in top-down proteomics and the analysis of proteoforms. Annu. Rev. Anal. Chem. 2016, 9, 499–519. [Google Scholar] [CrossRef]
- Printz, B.; Guerriero, G.; Sergeant, K.; Audinot, J.-N.; Guignard, C.; Renaut, J.; Lutts, S.; Hausman, J.F. Combining-omics to unravel the impact of copper nutrition on alfalfa (Medicago sativa) stem metabolism. Plant Cell Physiol. 2016, 57, 407–422. [Google Scholar] [CrossRef]
- O’Rourke, J.A.; Fu, F.; Bucciarelli, B.; Yang, S.S.; Samac, D.A.; Lamb, J.F.S.; Monteros, M.J.; Graham, M.A.; Gronwald, J.W.; Krom, N.; et al. The Medicago sativa gene index 1.2: A web-accessible gene expression atlas for investigating expression differences between Medicago sativa subspecies. BMC Genom. 2015, 16, 502. [Google Scholar] [CrossRef]
- Watson, C.F.; Zheng, L.; DellaPenna, D. Reduction of tomato polygalacturonase beta subunit expression affects pectin solubilization and degradation during fruit ripening. Plant Cell 1994, 6, 1623–1634. [Google Scholar] [CrossRef]
- Orozco-Cárdenas, M.L.; Ryan, C.A. Polygalacturonase β-Subunit antisense gene expression in tomato plants leads to a progressive enhanced wound response and necrosis in leaves and abscission of developing flowers. Plant Physiol. 2003, 133, 693–701. [Google Scholar] [CrossRef]
- Yuan, Z.; Yao, X.; Zhang, D.; Sun, Y.; Huang, H. Genome-wide expression profiling in seedlings of the Arabidopsis mutant uro that is defective in the secondary cell wall formation. J. Integr. Plant Biol. 2007, 49, 1754–1762. [Google Scholar] [CrossRef]
- Yun, H.S.; Kwon, C.; Kim, T.W.; Joo, S.H.; Cho, M.H.; Kang, B.G.; Chang, S.C.; Kim, S.K. Regulation of VrXTH1 expression in mungbean. J. Plant Biol. 2007, 50, 65–69. [Google Scholar] [CrossRef]
- Campbell, P.; Braam, J. In vitro activities of four xyloglucan endotransglycosylases from Arabidopsis. Plant J. 2002, 18, 371–382. [Google Scholar] [CrossRef]
- Morales-Quintana, L.; Carrasco-Orellana, C.; Beltran, D.; Moya-Leon, M.A.; Herrera, R. Molecular insights of a xyloglucan endo-transglycosylase/hydrolase of radiata pine (PrXTH1) expressed in response to inclination: Kinetics and computational study. Plant Physiol. Biochem. 2019, 136, 155–161. [Google Scholar] [CrossRef]
- Salazar-Iribe, A.; Cruz-Valderrama, J.E.; Jimenez-Duran, K.; Gomez-Maqueo, X.; Gamboa-deBuen, A. BIDDXI, a DUF642 cell wall protein, is involved in hypocotyl growth via auxin efflux. J. Plant Physiol. 2018, 231, 105–109. [Google Scholar] [CrossRef]
- Zúñiga-Sánchez, E.; Soriano, D.; Martínez-Barajas, E.; Orozco-Segovia, A.; Gamboa-deBuen, A. BIIDXI, the At4g32460 DUF642 gene, is involved in pectin methyl esterase regulation during Arabidopsis thaliana seed germination and plant development. BMC Plant Biol. 2014, 14, 338. [Google Scholar] [CrossRef]
- Dekkers, B.J.W.; Pearce, S.; van Bolderen-Veldkamp, R.P.; Marshall, A.; Widera, P.; Gilbert, J.; Drost, H.G.; Bassel, G.W.; Müller, K.; King, J.R.; et al. Transcriptional dynamics of two seed compartments with opposing roles in Arabidopsis seed germination. Plant Physiol. 2013, 163, 205–215. [Google Scholar] [CrossRef]
- Araki, R.; Mermod, M.; Yamasaki, H.; Kamiya, T.; Fuijwara, T.; Shikanai, T. SPL7 locally regulates copper-homeostasis-related genes in Arabidopsis. J. Plant Physiol. 2018, 224–225, 137–143. [Google Scholar] [CrossRef]
- Xu, P.; Cai, X.T.; Wang, Y.; Xing, L.; Chen, Q.; Xiang, C.-B. HDG11 upregulates cell-wall-loosening protein genes to promote root elongation in Arabidopsis. J. Exp. Bot. 2014, 65, 4285–4295. [Google Scholar] [CrossRef]
- Jung, J.-H.; Ju, Y.; Seo, P.J.; Lee, J.-H.; Park, C.-M. The SOC1-SPL module integrates photoperiod and gibberellic acid signals to control flowering time in Arabidopsis. Plant J. Cell. Mol. Biol. 2012, 69, 577–588. [Google Scholar] [CrossRef]
- Salazar-Iribe, A.; Agredano-Moreno, L.T.; Zúñiga-Sánchez, E.; Jiménez-Garcia, L.F.; Gamboa-deBuen, A. The cell wall DUF642 At2g41800 (TEB) protein is involved in hypocotyl cell elongation. Plant Sci. 2016, 253, 206–214. [Google Scholar] [CrossRef]
- Iglesias, N.; Abelenda, J.A.; Rodiño, M.; Sampedro, J.; Revilla, G.; Zarra, I. Apoplastic glycosidases active against xyloglucan oligosaccharides of Arabidopsis thaliana. Plant Cell Physiol. 2006, 47, 55–63. [Google Scholar] [CrossRef]
- Tesfaye, M.; Yang, S.S.; Lamb, J.F.S.; Jung, H.J.G.; Samac, D.A.; Vance, C.P.; Gronwald, J.W.; VandenBosch, K.A. Medicago truncatula as a model for dicot cell wall development. BioEnergy Res. 2009, 2, 59–76. [Google Scholar] [CrossRef]
- Cosio, C.; Dunand, C. Specific functions of individual class III peroxidase genes. J. Exp. Bot. 2009, 60, 391–408. [Google Scholar] [CrossRef]
- Francoz, E.; Ranocha, P.; Nguyen-Kim, H.; Jamet, E.; Burlat, V.; Dunand, C. Roles of cell wall peroxidases in plant development. Phytochemistry 2014, 112, 15–21. [Google Scholar] [CrossRef]
- Behr, M.; Legay, S.; Hausman, J.-F.; Guerriero, G. Analysis of cell wall-related genes in organs of Medicago sativa L. under different abiotic stresses. Int. J. Mol. Sci. 2015, 16, 16104–16124. [Google Scholar] [CrossRef]
- Rahman, M.A.; Alam, I.; Kim, Y.-G.; Ahn, N.Y.; Heo, S.H.; Lee, D.G.; Liu, G.; Lee, B.H. Screening for salt-responsive proteins in two contrasting alfalfa cultivars using a comparative proteome approach. Plant Physiol. Biochem. 2015, 89, 112–122. [Google Scholar] [CrossRef]
- Soares, N.C.; Wojtkowska, J.; Jackson, P.A. A proteomic analysis of the wound response in Medicago leaves reveals the early activation of a ROS-sensitive signal pathway. J. Proteom. 2011, 74, 1411–1420. [Google Scholar] [CrossRef]
- Oger, E.; Marino, D.; Guigonis, J.-M.; Pauly, N.; Puppo, A. Sulfenylated proteins in the Medicago truncatula–Sinorhizobium meliloti symbiosis. J. Proteom. 2012, 75, 4102–4113. [Google Scholar] [CrossRef]
- Li, N.; Zhao, M.; Liu, T.; Dong, L.; Cheng, Q.; Wu, J.; Wang, L.; Chen, X.; Zhang, C.; Lu, W.; et al. A novel soybean dirigent gene GmDIR22 contributes to promotion of lignan biosynthesis and enhances resistance to Phytophthora sojae. Front. Plant Sci. 2017, 8, 1185. [Google Scholar] [CrossRef]
- Bacete, L.; Melida, H.; Miedes, E.; Molina, A. Plant cell wall-mediated immunity: Cell wall changes trigger disease resistance responses. Plant J. 2018, 93, 614–636. [Google Scholar] [CrossRef]
- Song, M.; Peng, X. Genome-wide identification and characterization of DIR genes in Medicago truncatula. Biochem. Genet. 2019, 57, 1–20. [Google Scholar] [CrossRef]
- Francin-Allami, M.; Lollier, V.; Pavlovic, M.; San Clemente, H.; Rogniaux, H.; Jamet, E.; Guillon, F.; Larré, C. Understanding the remodelling of cell walls during Brachypodium distachyon grain development through a sub-cellular quantitative proteomic approach. Proteomes 2016, 4, 21. [Google Scholar] [CrossRef]
- Tarentino, A.L.; Gomez, C.M.; Plummer, T.H. Deglycosylation of asparagine-linked glycans by peptide:N-glycosidase F. Biochemistry 1985, 24, 4665–4671. [Google Scholar] [CrossRef]
- Maeda, M.; Kimura, Y. Structural features of free N-glycans occurring in plants and functional features of de-N-glycosylation enzymes, ENGase, and PNGase: The presence of unusual plant complex type N-glycans. Front. Plant Sci. 2014, 5, 429. [Google Scholar] [CrossRef]
- Boudart, G.; Jamet, E.; Rossignol, M.; Lafitte, C.; Borderies, G.; Jauneau, A.; Esquerré-Tugayé, M.T.; Pont-Lezica, R. Cell wall proteins in apoplastic fluids of Arabidopsis thaliana rosettes: Identification by mass spectrometry and bioinformatics. Proteomics 2005, 5, 212–221. [Google Scholar] [CrossRef]
- Tsai, A.Y.; Kunieda, T.; Rogalski, J.; Foster, L.J.; Ellis, B.E.; Haughn, G.W. Identification and characterization of Arabidopsis seed coat mucilage proteins. Plant Physiol. 2017, 173, 1059–1074. [Google Scholar] [CrossRef]
- Charmont, S.; Jamet, E.; Pont-Leziva, R.; Canut, H. Proteomics analysis of secreted proteins from Arabidopsis thaliana seedlings: Improved recovery following removal of phenolic compounds. Phytochemistry 2004, 66, 453–461. [Google Scholar] [CrossRef]
- Gu, C.; Shabab, M.; Strasser, R.; Wolters, P.J.; Shindo, T.; Niemer, M.; Kaschani, F.; Mach, L.; van der Hoorn, R.A. Post-translational regulation and trafficking of the granulin-containing protease RD21 of Arabidopsis thaliana. PLoS ONE 2012, 7, e32422. [Google Scholar] [CrossRef]
- Jashni, M.K.; Mehrabi, R.; Collemare, J.; Mesarich, C.H.; de Wit, P.J.G.M. The battle in the apoplast: Further insights into the roles of proteases and their inhibitors in plant-pathogen interactions. Front. Plant Sci. 2015, 6, 584. [Google Scholar] [CrossRef]
- Pogorelko, G.V.; Juvale, P.S.; Rutter, W.B.; Hütten, M.; Maier, T.R.; Hewezi, T.; Paulus, J.; van der Hoorn, R.A.L.; Grundler, F.M.W.; Siddique, S.; et al. Re-targeting of a plant defense protease by a cyst nematode effector. Plant J. 2019, 98, 1000–1014. [Google Scholar] [CrossRef]
- Cvitanich, C.; Przybylowicz, W.J.; Urbanski, D.F.; Jurkiewicz, A.M.; Mesjasz-Przybylowicz, J.; Blair, M.W.; Astudillo, C.; Jensen, E.Ø.; Stougaard, J. Iron and ferritin accumulate in separate cellular locations in Phaseolus seeds. BMC Plant Biol. 2010, 10, 26. [Google Scholar] [CrossRef]
- Ravet, K.; Touraine, B.; Boucherez, J.; Briat, J.F.; Gaymard, F.; Cellier, F. Ferritins control interaction between iron homeostasis and oxidative stress in Arabidopsis. Plant J. 2009, 57, 400–412. [Google Scholar] [CrossRef]
- Strozycki, P.M.; Szczurek, A.; Lotocka, B.; Figlerowicz, M.; Legocki, A.B. Ferritins and nodulation in Lupinus luteus: Iron management in indeterminate type nodules. J. Exp. Bot. 2007, 58, 3145–3153. [Google Scholar] [CrossRef]
- Hotte, N.; Deyholos, M. A flax fibre proteome: Identification of proteins enriched in bast fibres. BMC Plant Biol. 2008, 8, 52. [Google Scholar] [CrossRef]
- De Hoff, P.L.; Brill, L.M.; Hirsch, A.M. Plant lectins: The ties that bind in root symbiosis and plant defense. Mol. Genet. Genom. 2009, 282, 1–15. [Google Scholar] [CrossRef]
- Dias, R.O.; Machado, L.S.; Migliolo, L.; Franco, O.L. Insights into animal and plant lectins with antimicrobial activities. Molecules 2015, 20, 519–541. [Google Scholar] [CrossRef]
| Cluster | Spot | gi-Number | Name | Loc | Type | Base | Int. | Apex |
|---|---|---|---|---|---|---|---|---|
| 1 | CaCl2-2127 | gi|2951684 | lectin [M. sativa] | Extrac | Sol | 0.37 | −0.20 | −0.39 |
| 1 | CaCl2-2128 | gi|2951684 | lectin [M. sativa] | Extrac | Sol | 0.25 | −0.30 | −0.27 |
| 1 | CaCl2-2193 | gi|400180 | RecName: Full=Truncated lectin 2 | Extrac | Sol | 0.34 | −0.14 | −0.14 |
| 1 | EGTA-1167 | gi|388505450 | Glutelin type-A [M. truncatula] | Cytop | Sol | 0.25 | −0.14 | −0.23 |
| 1 | EGTA-1390 | gi|400180 | RecName: Full=Truncated lectin 2 | Extrac | Sol | 0.40 | −0.20 | −0.23 |
| 1 | EGTA-1411 | gi|2951684 | lectin [M. sativa] | Extrac | Sol | 0.20 | −0.29 | −0.30 |
| 1 | EGTA-1420 | gi|400180 | RecName: Full=Truncated lectin 2 | Extrac | Sol | 0.19 | −0.19 | −0.22 |
| 1 | LiCl-765 | gi|2951684 | lectin [M. sativa] | Extrac | Sol | 0.21 | −0.25 | −0.23 |
| 2 | CaCl2-1870 | gi|388505450 | Glutelin type-A [M. truncatula] | Cytop | Sol | 0.20 | −0.20 | −0.26 |
| 2 | CaCl2-2363 | gi|357468557 | Ferritin-3 [M. truncatula] | Plast | Sol | 0.24 | −0.04 | −0.44 |
| 2 | CaCl2-2627 | gi|357492793 | Ferritin-2 [M. truncatula] | Plast | Sol | 0.11 | −0.01 | −0.40 |
| 2 | EGTA-2050 | gi|50320305 | putative defensin 2.1 precursor [M. sativa] | Extrac | Sol | 0.15 | −0.14 | −0.19 |
| 3 | CaCl2-1013 | gi|922395483 | PNGase A [M. truncatula] | Extrac | Sol | 0.02 | −0.08 | −0.17 |
| 3 | CaCl2-2165 | gi|228204925 | chitinase class III-1 [M. sativa] | Extrac | Sol | 0.04 | −0.12 | −0.21 |
| 3 | CaCl2-2426 | gi|357468557 | Ferritin-3 [M. truncatula] | Plast | Sol | 0.09 | −0.08 | −0.14 |
| 3 | EGTA-1228 | gi|357437719 | Cysteine proteinase [M. truncatula] | Lyso | Sol | 0.16 | −0.12 | −0.14 |
| 3 | EGTA-1372 | gi|228204925 | chitinase class III-1 [M. sativa] | Extrac | Sol | 0.03 | −0.14 | −0.23 |
| 3 | EGTA-2117 | gi|3913031 | RecName: Full=Beta-amylase [M. sativa] | Cytop | Sol | 0.12 | −0.11 | −0.20 |
| 3 | EGTA-570 | gi|3913031 | RecName: Full=Beta-amylase [M. sativa] | Cytop | Sol | 0.11 | −0.10 | −0.22 |
| 3 | EGTA-573 | gi|3913031 | RecName: Full=Beta-amylase [M. sativa] | Cytop | Sol | 0.09 | −0.06 | −0.19 |
| 3 | EGTA-586 | gi|3913031 | RecName: Full=Beta-amylase [M. sativa] | Cytop | Sol | 0.12 | −0.09 | −0.18 |
| 3 | EGTA-587 | gi|3913031 | RecName: Full=Beta-amylase [M. sativa] | Cytop | Sol | 0.07 | −0.05 | −0.14 |
| 3 | EGTA-593 | gi|3913031 | RecName: Full=Beta-amylase [M. sativa] | Cytop | Sol | 0.08 | −0.13 | −0.22 |
| 3 | EGTA-595 | gi|3913031 | RecName: Full=Beta-amylase [M. sativa] | Cytop | Sol | 0.05 | −0.11 | −0.16 |
| 3 | EGTA-610 | gi|3913031 | RecName: Full=Beta-amylase [M. sativa] | Cytop | Sol | 0.12 | −0.09 | −0.28 |
| 3 | EGTA-615 | gi|3913031 | RecName: Full=Beta-amylase [M. sativa] | Cytop | Sol | 0.10 | −0.15 | −0.23 |
| 3 | EGTA-640 | gi|3913032 | RecName: Full=Beta-amylase [M. sativa] | Cytop | Sol | 0.13 | −0.07 | −0.14 |
| 4 | CaCl2-1716 | gi|388518933 | Endochitinase [M. truncatula] | Extrac | Sol | 0.26 | 0.10 | −0.12 |
| 4 | CaCl2-1880 | gi|357448991 | endo-1,3-beta-glucosidase [M. truncatula] | Extrac | Sol | 0.19 | 0.06 | −0.08 |
| 4 | CaCl2-2356 | gi|357468557 | Ferritin-3 [M. truncatula] | Plast | Sol | 0.14 | 0.08 | −0.20 |
| 4 | CaCl2-2425 | gi|357468557 | Ferritin-3 [M. truncatula] | Plast | Sol | 0.23 | 0.06 | −0.32 |
| 4 | CaCl2-2439 | gi|357468557 | Ferritin-3 [M. truncatula] | Plast | Sol | 0.28 | 0.04 | −0.21 |
| 4 | CaCl2-2553 | gi|357468557 | Ferritin-3 [M. truncatula] | Plast | Sol | 0.24 | −0.04 | −0.18 |
| 4 | CaCl2-2827 | gi|22901738 | high FW vegetative storage protein | Extrac | Sol | 0.21 | −0.03 | −0.23 |
| 4 | CaCl2-2828 | gi|357437721 | Cysteine proteinase [M. truncatula] | Lyso | Sol | 0.09 | 0.05 | −0.13 |
| 4 | EGTA-1111 | gi|71534882 | serine carboxypeptidase S10 [M. sativa] | Extrac | Sol | 0.21 | 0.00 | −0.16 |
| 4 | EGTA-1114 | gi|3913031 | RecName: Full=Beta-amylase [M. sativa] | Cytop | Sol | 0.18 | 0.06 | −0.08 |
| 4 | EGTA-1119 | gi|50317234 | Aspartic proteinase Asp1 [M. truncatula] | Extrac | Sol | 0.17 | 0.00 | −0.12 |
| 4 | EGTA-1143 | gi|357448997 | endo-1,3-beta-glucosidase [M. truncatula] | Extrac | Sol | 0.15 | −0.02 | −0.10 |
| 4 | EGTA-1166 | gi|298364452 | chitinase [M. sativa] | Extrac | Sol | 0.11 | 0.03 | −0.17 |
| 4 | EGTA-1168 | gi|657377089 | peroxidase family protein [M. truncatula] | Extrac | Sol | 0.16 | −0.03 | −0.12 |
| 4 | EGTA-1233 | gi|357437719 | Cysteine proteinase [M. truncatula] | Extrac | Sol | 0.12 | 0.03 | −0.14 |
| 4 | EGTA-1461 | gi|169147017 | putative thaumatin-like protein | Extrac | Sol | 0.19 | 0.05 | −0.04 |
| 4 | EGTA-1922 | gi|71534922 | thioredoxin h [M. sativa] | Cytop | Sol | 0.12 | −0.01 | −0.15 |
| 4 | EGTA-914 | gi|657371151 | eukaryotic aspartyl protease | Extrac | Sol | 0.19 | −0.02 | −0.17 |
| 4 | LiCl-1068 | gi|23049900 | put early nodulin-like 2 [T pratense] | CM | Memb | 0.10 | 0.09 | −0.08 |
| 5 | CaCl2-1769 | gi|357491415 | Peroxidase [M. truncatula] | Extrac | Sol | 0.40 | 0.29 | 0.11 |
| 5 | CaCl2-2269 | gi|3688528 | pre-pro-TPE4A protein [Pisum sativum] | Lyso | Sol | −0.01 | 0.17 | 0.05 |
| 5 | EGTA-1052 | gi|357517805 | Endochitinase [M. truncatula] | Lyso | Sol | 0.18 | 0.07 | 0.01 |
| 5 | EGTA-1057 | gi|388494834 | Endochitinase [M. truncatula] | Lyso | Sol | 0.23 | 0.09 | −0.01 |
| 5 | EGTA-1450 | gi|3688528 | cysteine proteinase [Vicia sativa] | Lyso | Sol | 0.00 | 0.21 | 0.06 |
| 5 | EGTA-1451 | gi|571536058 | PREDICTED: vignain-like [Glycine max] | Lyso | Sol | −0.02 | 0.15 | 0.00 |
| 5 | EGTA-1452 | gi|30141021 | cysteine protease-2 [Helianthus annuus] | Lyso | Sol | −0.04 | 0.14 | −0.01 |
| 5 | EGTA-2122 | gi|357448991 | endo-1,3-beta-glucosidase [M. truncatula] | Extrac | Sol | 0.26 | 0.10 | −0.03 |
| 6 | LiCl-885 | gi|357513969 | Germin-like protein [M. truncatula] | Extrac | Sol | −0.40 | −0.28 | −0.08 |
| 6 | LiCl-935 | gi|388502800 | Germin-like protein [Pisum sativum] | Extrac | Sol | −0.31 | −0.25 | −0.14 |
| 7 | CaCl2-1593 | gi|593701343 | Cyclophilin 38 isoform 1 [T. cacao] | Plast | Memb | −0.15 | −0.06 | 0.13 |
| 7 | EGTA-1388 | gi|357480321 | Lactoylglutathione lyase [M. truncatula] | Cytop | Sol | −0.15 | −0.02 | 0.04 |
| 7 | EGTA-1480 | gi|357512271 | Ribose-5-phosphate isomer. [M. truncatula] | Plast | Sol | −0.18 | 0.01 | 0.18 |
| 7 | EGTA-1586 | gi|388502800 | Germin-like protein [Pisum sativum] | Extrac | Sol | −0.23 | −0.07 | −0.01 |
| 7 | EGTA-196 | gi|357437609 | Beta-galactosidase [M. truncatula] | Extrac | Sol | −0.18 | 0.04 | 0.01 |
| 7 | EGTA-197 | gi|357437609 | Beta-galactosidase [M. truncatula] | Extrac | Sol | −0.19 | 0.03 | 0.03 |
| 7 | EGTA-201 | gi|357437609 | Beta-galactosidase [M. truncatula] | Extrac | Sol | −0.24 | 0.02 | 0.03 |
| 7 | EGTA-203 | gi|357437609 | Beta-galactosidase [M. truncatula] | Extrac | Sol | −0.23 | 0.05 | 0.05 |
| 7 | EGTA-205 | gi|357437609 | Beta-galactosidase [M. truncatula] | Extrac | Sol | −0.19 | 0.01 | 0.02 |
| 7 | EGTA-206 | gi|357437609 | Beta-galactosidase [M. truncatula] | Extrac | Sol | −0.19 | 0.02 | 0.03 |
| 7 | EGTA-2120 | gi|357437609 | Beta-galactosidase [M. truncatula] | Extrac | Sol | −0.16 | 0.05 | 0.03 |
| 7 | EGTA-214 | gi|357437609 | Beta-galactosidase [M. truncatula] | Extrac | Sol | −0.24 | 0.04 | 0.03 |
| 7 | EGTA-215 | gi|357437609 | Beta-galactosidase [M. truncatula] | Extrac | Sol | −0.23 | 0.05 | 0.05 |
| 7 | EGTA-216 | gi|357437609 | Beta-galactosidase [M. truncatula] | Extrac | Sol | −0.18 | 0.02 | 0.01 |
| 7 | EGTA-220 | gi|357437609 | Beta-galactosidase [M. truncatula] | Extrac | Sol | −0.21 | 0.06 | 0.06 |
| 7 | EGTA-221 | gi|357437609 | Beta-galactosidase [M. truncatula] | Extrac | Sol | −0.18 | 0.06 | 0.06 |
| 7 | EGTA-222 | gi|657400954 | beta-like galactosidase [M. truncatula] | Extrac | Sol | −0.23 | 0.00 | 0.04 |
| 7 | EGTA-240 | gi|357437609 | Beta-galactosidase [M. truncatula] | Extrac | Sol | −0.23 | 0.02 | 0.01 |
| 7 | EGTA-272 | gi|87241408 | Protease-associated PA [M. truncatula] | Extrac | Sol | −0.08 | −0.01 | 0.14 |
| 7 | EGTA-326 | gi|357437609 | Beta-galactosidase [M. truncatula] | Extrac | Sol | -0.22 | 0.04 | 0.02 |
| 7 | EGTA-333 | gi|357437609 | Beta-galactosidase [M. truncatula] | Extrac | Sol | −0.16 | 0.02 | 0.06 |
| 7 | EGTA-341 | gi|357437609 | Beta-galactosidase [M. truncatula] | Extrac | Sol | −0.16 | 0.03 | 0.04 |
| 7 | EGTA-342 | gi|357437609 | Beta-galactosidase [M. truncatula] | Extrac | Sol | −0.19 | 0.05 | 0.01 |
| 7 | EGTA-379 | gi|357437609 | Beta-galactosidase [M. truncatula] | Extrac | Sol | −0.20 | −0.02 | −0.05 |
| 8 | CaCl2-2612 | gi|357476945 | Acid beta-fructofuranosidase | Lyso | Sol | −0.16 | 0.02 | 0.27 |
| 8 | CaCl2-2968 | gi|388514479 | Oxygen-evolving enhancer protein 2 | Plast | Sol | −0.16 | -0.03 | 0.28 |
| 8 | EGTA-1296 | gi|357438145 | Cysteine proteinase [M. truncatula] | Lyso | Sol | −0.04 | 0.03 | 0.21 |
| 8 | EGTA-1485 | gi|357512271 | Ribose-5-phosphate isomer. [M. truncatula] | Plast | Sol | −0.16 | 0.04 | 0.24 |
| 8 | EGTA-1490 | gi|357512271 | Ribose-5-phosphate isomer. [M. truncatula] | Plast | Sol | -0.18 | 0.02 | 0.19 |
| 8 | EGTA-1794 | gi|157973737 | PSI reaction center IV A [Cicer arietinum] | Plast | Sol | −0.01 | 0.10 | 0.30 |
| 8 | LiCl-1156 | gi|537317 | peroxidase [M. sativa] | Extrac | Sol | −0.07 | 0.05 | 0.24 |
| 8 | LiCl-1163 | gi|537317 | peroxidase [M. sativa] | Extrac | Sol | 0.03 | 0.13 | 0.22 |
| 8 | LiCl-300 | gi|657374433 | polygalacturonase non-catalytic | Extrac | Sol | 0.00 | 0.06 | 0.20 |
| 8 | LiCl-340 | gi|537317 | peroxidase [M. sativa] | Extrac | Sol | −0.06 | −0.01 | 0.20 |
| 9 | CaCl2-1292 | gi|388508134 | Nod factor-bind. lectin [M. truncatula] | Golgi | Memb | −0.22 | −0.15 | 0.14 |
| 9 | CaCl2-1940 | gi|388502358 | Xylogluc. Endotransglucosylase | Extrac | Sol | −0.31 | −0.01 | 0.11 |
| 9 | CaCl2-1945 | gi|357508519 | Xylogluc. Endotransglucosylase | Extrac | Sol | −0.10 | 0.01 | 0.21 |
| 9 | CaCl2-2042 | gi|5777611 | cysteine protease [M. sativa] | Extrac | Sol | −0.02 | −0.08 | 0.18 |
| 9 | CaCl2-2511 | gi|388502800 | Germin-like protein [Pisum sativum] | Extrac | Sol | −0.22 | −0.02 | 0.03 |
| 9 | EGTA-1376 | gi|357513539 | Stem 28 kDa glycoprotein [M. truncatula] | Extrac | Sol | −0.09 | −0.07 | 0.18 |
| 9 | EGTA-1391 | gi|357513539 | Stem 28 kDa glycoprotein [M. truncatula] | Extrac | Sol | −0.17 | −0.02 | 0.30 |
| 9 | EGTA-1661 | gi|357476945 | Acid beta-fructofuranosidase | Lyso | Sol | −0.15 | 0.01 | 0.18 |
| 9 | EGTA-956 | gi|537313 | plant/F18G18-200 protein [M. truncatula] | Extrac | Sol | −0.31 | −0.09 | 0.24 |
| 9 | EGTA-965 | gi|537313 | plant/F18G18-200 protein [M. truncatula] | Extrac | Sol | −0.23 | −0.05 | 0.24 |
| 10 | CaCl2-1586 | gi|537313 | plant/F18G18-200 protein [M. truncatula] | Extrac | Sol | −0.38 | −0.14 | 0.37 |
| 10 | CaCl2-1609 | gi|537313 | plant/F18G18-200 protein [M. truncatula] | Extrac | Sol | −0.45 | −0.20 | 0.42 |
| 10 | CaCl2-2570 | gi|388509752 | unknown [Lotus japonicus] (pfam03018) | Extrac | Sol | −0.16 | −0.13 | 0.26 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sergeant, K.; Printz, B.; Guerriero, G.; Renaut, J.; Lutts, S.; Hausman, J.-F. The Dynamics of the Cell Wall Proteome of Developing Alfalfa Stems. Biology 2019, 8, 60. https://doi.org/10.3390/biology8030060
Sergeant K, Printz B, Guerriero G, Renaut J, Lutts S, Hausman J-F. The Dynamics of the Cell Wall Proteome of Developing Alfalfa Stems. Biology. 2019; 8(3):60. https://doi.org/10.3390/biology8030060
Chicago/Turabian StyleSergeant, Kjell, Bruno Printz, Gea Guerriero, Jenny Renaut, Stanley Lutts, and Jean-Francois Hausman. 2019. "The Dynamics of the Cell Wall Proteome of Developing Alfalfa Stems" Biology 8, no. 3: 60. https://doi.org/10.3390/biology8030060
APA StyleSergeant, K., Printz, B., Guerriero, G., Renaut, J., Lutts, S., & Hausman, J.-F. (2019). The Dynamics of the Cell Wall Proteome of Developing Alfalfa Stems. Biology, 8(3), 60. https://doi.org/10.3390/biology8030060









