DNA Damage Response in Plants: Conserved and Variable Response Compared to Animals
Abstract
:1. Introduction
2. Components of the DDR Pathway in Animals and Plants
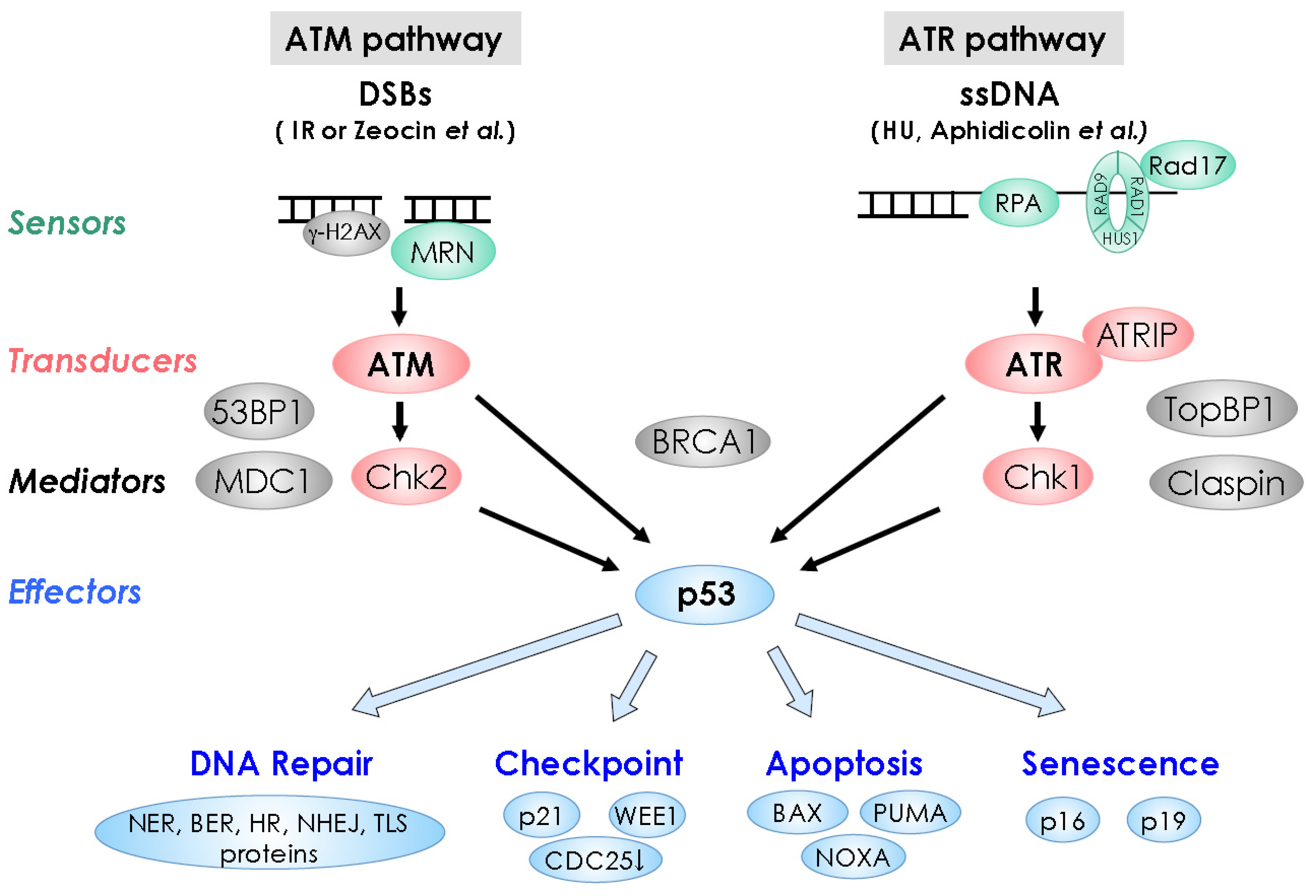
2.1. DNA Damage Sensors
2.2. Signal Transducers
2.3. Mediators
2.4. Effectors
2.4.1. Effectors for Cell-Cycle Arrest (Cell-Cycle Checkpoint)
2.4.2. Effectors for DNA Repair
2.4.3. Effectors for Programmed Cell Death (PCD) or Apoptosis
3. Plant-Specific DDR
3.1. Endoreduplication
3.2. Plant-Specific Factors of the DDR
3.2.1. SOG1 Function
3.2.2. The Mechanisms of SOG1 Regulation

4. What Growth Strategies Do Plants Have?
5. Conclusions
| Function | Class | Animal gene | Arabidopsis gene | AGI code |
|---|---|---|---|---|
| Sensors | DSB recognition/repair | MRE11 | MRE11 | At5g54260 |
| (MRN complex) | RAD51 | RAD51 | At2g31970 | |
| NBS1 | NBS1 | At3g02680 | ||
| ssDNA binding protein | RPA1 | RPA70a | At2g06510 | |
| PCNA-like | RAD9 | RAD9 | At3g05480 | |
| (9-1-1 complex) | RAD1 | RAD1 | At4g17760 | |
| HUS1 | HUS1 | At1g52530 | ||
| Compornet of RFC1-like | RAD17 | RAD17 | At5g66130 | |
| Transducers | PI3 kinase-like protein | ATM | ATM | At3g48190 |
| ATR | ATR | At5g40820 | ||
| PIKK binding protein | ATRIP | ATRIP | At5g45610 | |
| Protein kinase | CHK1 | Not found | ||
| CHK2 | Not found | |||
| Mediators | BRCT-containing | MDC1 | Not found | |
| 53BP1 | Not found | |||
| BRCA1 | BRCA1 | At4g21070 | ||
| TopBP1 | MEI1? | At1g77320 | ||
| CHK1 binding | Claspin | Not found | ||
| Histon variant | H2AX | H2AX | At1g08880 | |
| Effectors | Transcription factor | p53 | Not found | |
| Not found | SOG1 | At1g25580 | ||
| Cell cycle arrest | p21 | Not found | ||
| WEE1 | WEE1 | At1g02970 | ||
| CDC25 | CDC25-like? | At5g03455 | ||
| DNA repair | See reference [119,120] | See reference [84,85,121] | ||
| Apoptosis | PUMA | Not found | ||
| BAX | Not found | |||
| NOXA | Not found |
Acknowledgments
Conflicts of Interest
References
- Ciccia, A.; Elledge, S.J. The DNA damage response: Making it safe to play with knives. Mol. Cell 2010, 40, 179–204. [Google Scholar] [CrossRef]
- Triantaphylides, C.; Havaux, M. Singlet oxygen in plants: Production, detoxification and signaling. Trends Plant Sci. 2009, 14, 219–228. [Google Scholar] [CrossRef]
- Krieger-Liszkay, A. Singlet oxygen production in photosynthesis. J. Exp. Bot. 2005, 56, 337–346. [Google Scholar] [CrossRef]
- Asada, K. The water-water cycle in chloroplasts: Scavenging of active oxygens and dissipation of excess photons. Annu. Rev. Plant Physiol. Plant Mol. Biol. 1999, 50, 601–639. [Google Scholar] [CrossRef]
- Beck, C.B. An Introduction to Plant Structure and Development, 2nd ed.; Cambridge University Press: Cambridge, UK, 2010. [Google Scholar]
- Mannuss, A.; Trapp, O.; Puchta, H. Gene regulation in response to DNA damage. Biochim. Biophys. Acta 2012, 1819, 154–165. [Google Scholar] [CrossRef]
- Sirbu, B.M.; Cortez, D. DNA damage response: Three levels of DNA repair regulation. Cold Spring Harb. Perspect. Biol. 2013, 5. [Google Scholar] [CrossRef]
- Cimprich, K.A.; Cortez, D. Atr: An essential regulator of genome integrity. Nat. Rev. Mol. Cell Biol. 2008, 9, 616–627. [Google Scholar] [CrossRef]
- Sancar, A.; Lindsey-Boltz, L.A.; Unsal-Kacmaz, K.; Linn, S. Molecular mechanisms of mammalian DNA repair and the DNA damage checkpoints. Annu. Rev. Biochem. 2004, 73, 39–85. [Google Scholar] [CrossRef]
- Rupnik, A.; Lowndes, N.F.; Grenon, M. Mrn and the race to the break. Chromosoma 2010, 119, 115–135. [Google Scholar] [CrossRef]
- Dickey, J.S.; Redon, C.E.; Nakamura, A.J.; Baird, B.J.; Sedelnikova, O.A.; Bonner, W.M. H2ax: Functional roles and potential applications. Chromosoma 2009, 118, 683–692. [Google Scholar] [CrossRef]
- Zou, L.; Elledge, S.J. Sensing DNA damage through atrip recognition of RPA-ssDNA complexes. Science 2003, 300, 1542–1548. [Google Scholar] [CrossRef]
- Bermudez, V.P.; Lindsey-Boltz, L.A.; Cesare, A.J.; Maniwa, Y.; Griffith, J.D.; Hurwitz, J.; Sancar, A. Loading of the human 9-1-1 checkpoint complex onto DNA by the checkpoint clamp loader hrad17-replication factor c complex in vitro. Proc. Natl. Acad. Sci. USA 2003, 100, 1633–1638. [Google Scholar] [CrossRef]
- Griffith, J.D.; Lindsey-Boltz, L.A.; Sancar, A. Structures of the human rad17-replication factor c and checkpoint rad 9-1-1 complexes visualized by glycerol spray/low voltage microscopy. J. Biol. Chem. 2002, 277, 15233–15236. [Google Scholar] [CrossRef]
- Friesner, J.D.; Liu, B.; Culligan, K.; Britt, A.B. Ionizing radiation-dependent gamma-h2ax focus formation requires ataxia telangiectasia mutated and ataxia telangiectasia mutated and rad3-related. Mol. Biol. Cell 2005, 16, 2566–2576. [Google Scholar] [CrossRef]
- Hartung, H.P.F. Isolation of the complete cDNA of the Mre11 homolog of Arabidopsis (accession no. Aj243822) indicates conservation of DNA recombination mechanisms between plants and other eucaryotes. Plant Physiol. 1999, 121, 312. [Google Scholar]
- Gallego, M.E.; White, C.I. Rad50 function is essential for telomere maintenance in arabidopsis. Proc. Natl. Acad. Sci. USA 2001, 98, 1711–1716. [Google Scholar] [CrossRef]
- Akutsu, N.; Iijima, K.; Hinata, T.; Tauchi, H. Characterization of the plant homolog of nijmegen breakage syndrome 1: Involvement in DNA repair and recombination. Biochem. Biophys. Res. Commun. 2007, 353, 394–398. [Google Scholar] [CrossRef]
- Amiard, S.; Charbonnel, C.; Allain, E.; Depeiges, A.; White, C.I.; Gallego, M.E. Distinct roles of the atr kinase and the mre11-rad50-nbs1 complex in the maintenance of chromosomal stability in arabidopsis. Plant Cell 2010, 22, 3020–3033. [Google Scholar]
- Takashi, Y.; Kobayashi, Y.; Tanaka, K.; Tamura, K. Arabidopsis replication protein a 70a is required for DNA damage response and telomere length homeostasis. Plant Cell Physiol. 2009, 50, 1965–1976. [Google Scholar] [CrossRef]
- Heitzeberg, F.; Chen, I.P.; Hartung, F.; Orel, N.; Angelis, K.J.; Puchta, H. The rad17 homologue of arabidopsis is involved in the regulation of DNA damage repair and homologous recombination. Plant J. Cell Mol. Biol. 2004, 38, 954–968. [Google Scholar] [CrossRef]
- Bakkenist, C.J.; Kastan, M.B. DNA damage activates atm through intermolecular autophosphorylation and dimer dissociation. Nature 2003, 421, 499–506. [Google Scholar] [CrossRef]
- Warmerdam, D.O.; Kanaar, R.; Smits, V.A. Differential dynamics of atr-mediated checkpoint regulators. J. Nucleic Acids 2010, 2010, 319142:1–319142:6. [Google Scholar]
- Ball, H.L.; Cortez, D. Atrip oligomerization is required for atr-dependent checkpoint signaling. J. Biol. Chem. 2005, 280, 31390–31396. [Google Scholar] [CrossRef]
- Kastan, M.B.; Lim, D.S. The many substrates and functions of atm. Nat. Rev. Mol. Cell Biol. 2000, 1, 179–186. [Google Scholar] [CrossRef]
- Brown, E.J.; Baltimore, D. Atr disruption leads to chromosomal fragmentation and early embryonic lethality. Genes Dev. 2000, 14, 397–402. [Google Scholar]
- Xu, Y.; Ashley, T.; Brainerd, E.E.; Bronson, R.T.; Meyn, M.S.; Baltimore, D. Targeted disruption of atm leads to growth retardation, chromosomal fragmentation during meiosis, immune defects, and thymic lymphoma. Genes Dev. 1996, 10, 2411–2422. [Google Scholar] [CrossRef]
- Bartek, J.; Falck, J.; Lukas, J. Chk2 kinase—A busy messenger. Nat. Rev. Mol. Cell Biol. 2001, 2, 877–886. [Google Scholar] [CrossRef]
- Chen, Y.; Sanchez, Y. Chk1 in the DNA damage response: Conserved roles from yeasts to mammals. DNA Repair 2004, 3, 1025–1032. [Google Scholar] [CrossRef]
- Ahn, J.Y.; Schwarz, J.K.; Piwnica-Worms, H.; Canman, C.E. Threonine 68 phosphorylation by ataxia telangiectasia mutated is required for efficient activation of chk2 in response to ionizing radiation. Cancer Res. 2000, 60, 5934–5936. [Google Scholar]
- Matsuoka, S.; Rotman, G.; Ogawa, A.; Shiloh, Y.; Tamai, K.; Elledge, S.J. Ataxia telangiectasia-mutated phosphorylates chk2 in vivo and in vitro. Proc. Natl. Acad. Sci. USA 2000, 97, 10389–10394. [Google Scholar]
- Gatei, M.; Sloper, K.; Sorensen, C.; Syljuasen, R.; Falck, J.; Hobson, K.; Savage, K.; Lukas, J.; Zhou, B.B.; Bartek, J.; et al. Ataxia-telangiectasia-mutated (atm) and nbs1-dependent phosphorylation of chk1 on ser-317 in response to ionizing radiation. J. Biol. Chem. 2003, 278, 14806–14811. [Google Scholar] [CrossRef]
- Liu, Q.; Guntuku, S.; Cui, X.S.; Matsuoka, S.; Cortez, D.; Tamai, K.; Luo, G.; Carattini-Rivera, S.; DeMayo, F.; Bradley, A.; et al. Chk1 is an essential kinase that is regulated by atr and required for the g(2)/m DNA damage checkpoint. Genes Dev. 2000, 14, 1448–1459. [Google Scholar]
- Zhao, H.; Piwnica-Worms, H. Atr-mediated checkpoint pathways regulate phosphorylation and activation of human chk1. Mol. Cell. Biol. 2001, 21, 4129–4139. [Google Scholar] [CrossRef]
- Kim, M.A.; Kim, H.J.; Brown, A.L.; Lee, M.Y.; Bae, Y.S.; Park, J.I.; Kwak, J.Y.; Chung, J.H.; Yun, J. Identification of novel substrates for human checkpoint kinase chk1 and chk2 through genome-wide screening using a consensus chk phosphorylation motif. Exp. Mol. Med. 2007, 39, 205–212. [Google Scholar] [CrossRef]
- Garcia, V.; Bruchet, H.; Camescasse, D.; Granier, F.; Bouchez, D.; Tissier, A. Atatm is essential for meiosis and the somatic response to DNA damage in plants. Plant Cell 2003, 15, 119–132. [Google Scholar] [CrossRef]
- Culligan, K.; Tissier, A.; Britt, A. Atr regulates a g2-phase cell-cycle checkpoint in arabidopsis thaliana. Plant Cell 2004, 16, 1091–1104. [Google Scholar] [CrossRef]
- Culligan, K.M.; Robertson, C.E.; Foreman, J.; Doerner, P.; Britt, A.B. Atr and atm play both distinct and additive roles in response to ionizing radiation. Plant J. Cell Mol. Biol. 2006, 48, 947–961. [Google Scholar] [CrossRef]
- Elkon, R.; Rashi-Elkeles, S.; Lerenthal, Y.; Linhart, C.; Tenne, T.; Amariglio, N.; Rechavi, G.; Shamir, R.; Shiloh, Y. Dissection of a DNA-damage-induced transcriptional network using a combination of microarrays, rna interference and computational promoter analysis. Genome Biol. 2005, 6, R43. [Google Scholar] [CrossRef]
- Heinloth, A.N.; Shackelford, R.E.; Innes, C.L.; Bennett, L.; Li, L.; Amin, R.P.; Sieber, S.O.; Flores, K.G.; Bushel, P.R.; Paules, R.S. Atm-dependent and -independent gene expression changes in response to oxidative stress, gamma irradiation, and uv irradiation. Radiat. Res. 2003, 160, 273–290. [Google Scholar] [CrossRef]
- Sweeney, P.R.; Britt, A.B.; Culligan, K.M. The arabidopsis atrip ortholog is required for a programmed response to replication inhibitors. Plant J. Cell Mol. Biol. 2009, 60, 518–526. [Google Scholar] [CrossRef]
- Sakamoto, A.N.; Lan, V.T.; Puripunyavanich, V.; Hase, Y.; Yokota, Y.; Shikazono, N.; Nakagawa, M.; Narumi, I.; Tanaka, A. A uvb-hypersensitive mutant in arabidopsis thaliana is defective in the DNA damage response. Plant J. Cell Mol. Biol. 2009, 60, 509–517. [Google Scholar] [CrossRef]
- Inze, D.; de Veylder, L. Cell cycle regulation in plant development. Annu. Rev. Genet. 2006, 40, 77–105. [Google Scholar] [CrossRef]
- Lafarge, S. Characterization of arabidopsis thaliana ortholog of the human breast cancer susceptibility gene 1: Atbrca1, strongly induced by gamma rays. Nucleic Acids Res. 2003, 31, 1148–1155. [Google Scholar] [CrossRef]
- Stracker, T.H.; Usui, T.; Petrini, J.H. Taking the time to make important decisions: The checkpoint effector kinases chk1 and chk2 and the DNA damage response. DNA Repair 2009, 8, 1047–1054. [Google Scholar] [CrossRef]
- Stewart, G.S.; Wang, B.; Bignell, C.R.; Taylor, A.M.; Elledge, S.J. Mdc1 is a mediator of the mammalian DNA damage checkpoint. Nature 2003, 421, 961–966. [Google Scholar] [CrossRef]
- Stucki, M.; Jackson, S.P. Mdc1/nfbd1: A key regulator of the DNA damage response in higher eukaryotes. DNA Repair 2004, 3, 953–957. [Google Scholar] [CrossRef]
- Garcia, V.; Furuya, K.; Carr, A.M. Identification and functional analysis of topbp1 and its homologs. DNA Repair 2005, 4, 1227–1239. [Google Scholar] [CrossRef]
- Kumagai, A.; Kim, S.M.; Dunphy, W.G. Claspin and the activated form of atr-atrip collaborate in the activation of chk1. J. Biol. Chem. 2004, 279, 49599–49608. [Google Scholar] [CrossRef]
- Manke, I.A.; Lowery, D.M.; Nguyen, A.; Yaffe, M.B. Brct repeats as phosphopeptide-binding modules involved in protein targeting. Science 2003, 302, 636–639. [Google Scholar] [CrossRef]
- Yu, X.; Chini, C.C.; He, M.; Mer, G.; Chen, J. The brct domain is a phospho-protein binding domain. Science 2003, 302, 639–642. [Google Scholar] [CrossRef]
- Chini, C.C.; Chen, J. Human claspin is required for replication checkpoint control. J. Biol. Chem. 2003, 278, 30057–30062. [Google Scholar] [CrossRef]
- Kumagai, A.; Dunphy, W.G. Claspin, a novel protein required for the activation of chk1 during a DNA replication checkpoint response in xenopus egg extracts. Mol. Cell 2000, 6, 839–849. [Google Scholar] [CrossRef]
- Rogakou, E.P.; Pilch, D.R.; Orr, A.H.; Ivanova, V.S.; Bonner, W.M. DNA double-stranded breaks induce histone h2ax phosphorylation on serine 139. J. Biol. Chem. 1998, 273, 5858–5868. [Google Scholar]
- Lukas, C.; Melander, F.; Stucki, M.; Falck, J.; Bekker-Jensen, S.; Goldberg, M.; Lerenthal, Y.; Jackson, S.P.; Bartek, J.; Lukas, J. Mdc1 couples DNA double-strand break recognition by nbs1 with its h2ax-dependent chromatin retention. EMBO J. 2004, 23, 2674–2683. [Google Scholar] [CrossRef]
- Celeste, A.; Fernandez-Capetillo, O.; Kruhlak, M.J.; Pilch, D.R.; Staudt, D.W.; Lee, A.; Bonner, R.F.; Bonner, W.M.; Nussenzweig, A. Histone h2ax phosphorylation is dispensable for the initial recognition of DNA breaks. Nat. Cell Biol. 2003, 5, 675–679. [Google Scholar] [CrossRef]
- Lou, Z.; Minter-Dykhouse, K.; Franco, S.; Gostissa, M.; Rivera, M.A.; Celeste, A.; Manis, J.P.; van Deursen, J.; Nussenzweig, A.; Paull, T.T.; et al. Mdc1 maintains genomic stability by participating in the amplification of atm-dependent DNA damage signals. Mol. Cell 2006, 21, 187–200. [Google Scholar] [CrossRef]
- Aunoble, B.; Bernard-Gallon, D.; Bignon, Y.J. Regulation of brca1 and brca2 transcript in response to cisplatin, adriamycin, taxol and ionising radiation is correlated to p53 functional status in ovarian cancer cell lines. Oncol. Rep. 2001, 8, 663–668. [Google Scholar]
- Trapp, O.; Seeliger, K.; Puchta, H. Homologs of breast cancer genes in plants. Front. Plant Sci. 2011, 2, 19. [Google Scholar]
- Mathilde, G.; Ghislaine, G.; Daniel, V.; Georges, P. The arabidopsis mei1 gene encodes a protein with five brct domains that is involved in meiosis-specific DNA repair events independent of spo11-induced dsbs. Plant J. 2003, 35, 465–475. [Google Scholar] [CrossRef]
- Lavin, M.F.; Kozlov, S. Atm activation and DNA damage response. Cell Cycle 2007, 6, 931–942. [Google Scholar] [CrossRef]
- Helton, E.S.; Chen, X. P53 modulation of the DNA damage response. J. Cell. Biochem. 2007, 100, 883–896. [Google Scholar] [CrossRef]
- Appella, E.; Anderson, C.W. Post-translational modifications and activation of p53 by genotoxic stresses. Eur. J. Biochem. FEBS 2001, 268, 2764–2772. [Google Scholar] [CrossRef]
- Xirodimas, D.P.; Saville, M.K.; Bourdon, J.C.; Hay, R.T.; Lane, D.P. Mdm2-mediated nedd8 conjugation of p53 inhibits its transcriptional activity. Cell 2004, 118, 83–97. [Google Scholar] [CrossRef]
- Taira, N.; Yoshida, K. Post-translational modifications of p53 tumor suppressor: Determinants of its functional targets. Histol. Histopathol. 2012, 27, 437–443. [Google Scholar]
- Shieh, S.Y.; Ikeda, M.; Taya, Y.; Prives, C. DNA damage-induced phosphorylation of p53 alleviates inhibition by mdm2. Cell 1997, 91, 325–334. [Google Scholar] [CrossRef]
- Hoeberichts, F.A.; Woltering, E.J. Multiple mediators of plant programmed cell death: Interplay of conserved cell death mechanisms and plant-specific regulators. BioEssays News Rev. Mol. Cell. Dev. Biol. 2003, 25, 47–57. [Google Scholar] [CrossRef]
- Harrison, J.C.; Haber, J.E. Surviving the breakup: The DNA damage checkpoint. Annu. Rev. Genet. 2006, 40, 209–235. [Google Scholar] [CrossRef]
- Harper, J.W.; Elledge, S.J. The DNA damage response: Ten years after. Mol. Cell 2007, 28, 739–745. [Google Scholar] [CrossRef]
- Lazzaro, F.; Giannattasio, M.; Puddu, F.; Granata, M.; Pellicioli, A.; Plevani, P.; Muzi-Falconi, M. Checkpoint mechanisms at the intersection between DNA damage and repair. DNA Repair 2009, 8, 1055–1067. [Google Scholar] [CrossRef]
- Abbas, T.; Dutta, A. P21 in cancer: Intricate networks and multiple activities. Nat. Rev. Cancer 2009, 9, 400–414. [Google Scholar] [CrossRef]
- Sorensen, C.S.; Syljuasen, R.G. Safeguarding genome integrity: The checkpoint kinases atr, chk1 and wee1 restrain cdk activity during normal DNA replication. Nucleic Acids Res. 2012, 40, 477–486. [Google Scholar] [CrossRef]
- Reinhardt, H.C.; Schumacher, B. The p53 network: Cellular and systemic DNA damage responses in aging and cancer. Trends Genet. TIG 2012, 28, 128–136. [Google Scholar]
- McGowan, C.H.; Russell, P. Cell cycle regulation of human wee1. EMBO J. 1995, 14, 2166–2175. [Google Scholar]
- Heald, R.; McLoughlin, M.; McKeon, F. Human wee1 maintains mitotic timing by protecting the nucleus from cytoplasmically activated cdc2 kinase. Cell 1993, 74, 463–474. [Google Scholar] [CrossRef]
- Boutros, R.; Dozier, C.; Ducommun, B. The when and wheres of cdc25 phosphatases. Curr. Opin. Cell Biol. 2006, 18, 185–191. [Google Scholar] [CrossRef]
- Karlsson-Rosenthal, C.; Millar, J.B. Cdc25: Mechanisms of checkpoint inhibition and recovery. Trends Cell Biol. 2006, 16, 285–292. [Google Scholar] [CrossRef]
- Yoshiyama, K.; Conklin, P.A.; Huefner, N.D.; Britt, A.B. Suppressor of gamma response 1 (sog1) encodes a putative transcription factor governing multiple responses to DNA damage. Proc. Natl. Acad. Sci. USA 2009, 106, 12843–12848. [Google Scholar]
- Sorrell, D.A.; Marchbank, A.; McMahon, K.; Dickinson, J.R.; Rogers, H.J.; Francis, D. A wee1 homologue from arabidopsis thaliana. Planta 2002, 215, 518–522. [Google Scholar] [CrossRef]
- De Schutter, K.; Joubes, J.; Cools, T.; Verkest, A.; Corellou, F.; Babiychuk, E.; van Der Schueren, E.; Beeckman, T.; Kushnir, S.; Inze, D.; et al. Arabidopsis wee1 kinase controls cell cycle arrest in response to activation of the DNA integrity checkpoint. Plant Cell 2007, 19, 211–225. [Google Scholar] [CrossRef]
- Spadafora, N.D.; Doonan, J.H.; Herbert, R.J.; Bitonti, M.B.; Wallace, E.; Rogers, H.J.; Francis, D. Arabidopsis t-DNA insertional lines for cdc25 are hypersensitive to hydroxyurea but not to zeocin or salt stress. Ann. Bot. 2011, 107, 1183–1192. [Google Scholar] [CrossRef]
- Dissmeyer, N.; Weimer, A.K.; Pusch, S.; de Schutter, K.; Alvim Kamei, C.L.; Nowack, M.K.; Novak, B.; Duan, G.L.; Zhu, Y.G.; de Veylder, L.; et al. Control of cell proliferation, organ growth, and DNA damage response operate independently of dephosphorylation of the arabidopsis cdk1 homolog cdka;1. Plant Cell 2009, 21, 3641–3654. [Google Scholar] [CrossRef]
- Friedberg, E.C.; Walker, G.C.; Siede, W.; Wood, R.D.; Schultz, R.A.; Ellenberger, T. DNA Repair and Mutagenesis, 2nd ed.; American Society for Microbiology: Washington, DC, USA, 2005. [Google Scholar]
- Hays, J.B. Arabidopsis thaliana, a versatile model system for study of eukaryotic genome-maintenance functions. DNA Repair 2002, 1, 579–600. [Google Scholar] [CrossRef]
- Kimura, S.; Sakaguchi, K. DNA repair in plants. Chem. Rev. 2006, 106, 753–766. [Google Scholar] [CrossRef]
- Jasin, M.; Moynahan, M.E.; Richardson, C. Targeted transgenesis. Proc. Natl. Acad. Sci. USA 1996, 93, 8804–8808. [Google Scholar] [CrossRef]
- Lee, K.Y.; Lund, P.; Lowe, K.; Dunsmuir, P. Homologous recombination in plant cells after agrobacterium-mediated transformation. Plant Cell 1990, 2, 415–425. [Google Scholar]
- Offringa, R.; de Groot, M.J.; Haagsman, H.J.; Does, M.P.; van den Elzen, P.J.; Hooykaas, P.J. Extrachromosomal homologous recombination and gene targeting in plant cells after agrobacterium mediated transformation. EMBO J. 1990, 9, 3077–3084. [Google Scholar]
- Vousden, K.H.; Lu, X. Live or let die: The cell’s response to p53. Nat. Rev. Cancer 2002, 2, 594–604. [Google Scholar] [CrossRef]
- Fulcher, N.; Sablowski, R. Hypersensitivity to DNA damage in plant stem cell niches. Proc. Natl. Acad. Sci. USA 2009, 106, 20984–20988. [Google Scholar] [CrossRef]
- Rich, T.; Allen, R.L.; Wyllie, A.H. Defying death after DNA damage. Nature 2000, 407, 777–783. [Google Scholar] [CrossRef]
- Lacomme, C.; Santa Cruz, S. Bax-induced cell death in tobacco is similar to the hypersensitive response. Proc. Natl. Acad. Sci. USA 1999, 96, 7956–7961. [Google Scholar] [CrossRef]
- Yoshinaga, K.; Arimura, S.I.; Hirata, A.; Niwa, Y.; Yun, D.J.; Tsutsumi, N.; Uchimiya, H.; Kawai-Yamada, M. Mammalian bax initiates plant cell death through organelle destruction. Plant Cell Rep. 2005, 24, 408–417. [Google Scholar] [CrossRef]
- Watanabe, N.; Lam, E. Arabidopsis bax inhibitor-1 functions as an attenuator of biotic and abiotic types of cell death. Plant J. Cell Mol. Biol. 2006, 45, 884–894. [Google Scholar] [CrossRef]
- Uren, A.G.; O’Rourke, K.; Aravind, L.A.; Pisabarro, M.T.; Seshagiri, S.; Koonin, E.V.; Dixit, V.M. Identification of paracaspases and metacaspases: Two ancient families of caspase-like proteins, one of which plays a key role in malt lymphoma. Mol. Cell 2000, 6, 961–967. [Google Scholar]
- Lam, E.; Zhang, Y. Regulating the reapers: Activating metacaspases for programmed cell death. Trends Plant Sci. 2012, 17, 487–494. [Google Scholar] [CrossRef]
- Lee, H.O.; Davidson, J.M.; Duronio, R.J. Endoreplication: Polyploidy with purpose. Genes Dev. 2009, 23, 2461–2477. [Google Scholar] [CrossRef]
- Fox, D.T.; Duronio, R.J. Endoreplication and polyploidy: Insights into development and disease. Development 2013, 140, 3–12. [Google Scholar] [CrossRef]
- Endo, M.; Ishikawa, Y.; Osakabe, K.; Nakayama, S.; Kaya, H.; Araki, T.; Shibahara, K.; Abe, K.; Ichikawa, H.; Valentine, L.; et al. Increased frequency of homologous recombination and t-DNA integration in arabidopsis caf-1 mutants. EMBO J. 2006, 25, 5579–5590. [Google Scholar] [CrossRef]
- Ramirez-Parra, E.; Gutierrez, C. E2f regulates fasciata1, a chromatin assembly gene whose loss switches on the endocycle and activates gene expression by changing the epigenetic status. Plant Physiol. 2007, 144, 105–120. [Google Scholar] [CrossRef]
- Schonrock, N.; Exner, V.; Probst, A.; Gruissem, W.; Hennig, L. Functional genomic analysis of caf-1 mutants in arabidopsis thaliana. J. Biol. Chem. 2006, 281, 9560–9568. [Google Scholar]
- Takahashi, N.; Lammens, T.; Boudolf, V.; Maes, S.; Yoshizumi, T.; de Jaeger, G.; Witters, E.; Inze, D.; de Veylder, L. The DNA replication checkpoint aids survival of plants deficient in the novel replisome factor etg1. EMBO J. 2008, 27, 1840–1851. [Google Scholar] [CrossRef]
- Adachi, S.; Minamisawa, K.; Okushima, Y.; Inagaki, S.; Yoshiyama, K.; Kondou, Y.; Kaminuma, E.; Kawashima, M.; Toyoda, T.; Matsui, M.; et al. Programmed induction of endoreduplication by DNA double-strand breaks in arabidopsis. Proc. Natl. Acad. Sci. USA 2011, 108, 10004–10009. [Google Scholar] [CrossRef]
- Sugimoto-Shirasu, K.; Roberts, K. “Big it up”: Endoreduplication and cell-size control in plants. Curr. Opin. Plant Biol. 2003, 6, 544–553. [Google Scholar] [CrossRef]
- Hashimura, Y.; Ueguchi, C. The arabidopsis meristem disorganization 1 gene is required for the maintenance of stem cells through the reduction of DNA damage. Plant J. Cell Mol Biol. 2011, 68, 657–669. [Google Scholar] [CrossRef]
- Wenig, U.; Meyer, S.; Stadler, R.; Fischer, S.; Werner, D.; Lauter, A.; Melzer, M.; Hoth, S.; Weingartner, M.; Sauer, N. Identification of main, a factor involved in genome stability in the meristems of arabidopsis thaliana. Plant J. Cell Mol. Biol. 2013, 75, 469–483. [Google Scholar] [CrossRef]
- Wei, W.; Ba, Z.; Gao, M.; Wu, Y.; Ma, Y.; Amiard, S.; White, C.I.; Rendtlew Danielsen, J.M.; Yang, Y.G.; Qi, Y. A role for small rnas in DNA double-strand break repair. Cell 2012, 149, 101–112. [Google Scholar] [CrossRef]
- Preuss, S.B.; Britt, A.B. A DNA-damage-induced cell cycle checkpoint in arabidopsis. Genetics 2003, 164, 323–334. [Google Scholar]
- Yoshiyama, K.O.; Kobayashi, J.; Ogita, N.; Ueda, M.; Kimura, S.; Maki, H.; Umeda, M. Atm-mediated phosphorylation of sog1 is essential for the DNA damage response in arabidopsis. EMBO Rep. 2013, 14, 817–822. [Google Scholar] [CrossRef]
- Furukawa, T.; Curtis, M.J.; Tominey, C.M.; Duong, Y.H.; Wilcox, B.W.; Aggoune, D.; Hays, J.B.; Britt, A.B. A shared DNA-damage-response pathway for induction of stem-cell death by uvb and by gamma irradiation. DNA Repair 2010, 9, 940–948. [Google Scholar] [CrossRef]
- Rutkowski, R.; Hofmann, K.; Gartner, A. Phylogeny and function of the invertebrate p53 superfamily. Cold Spring Harb. Perspect. Biol. 2010, 2, a001131. [Google Scholar]
- Rozan, L.M.; El-Deiry, W.S. P53 downstream target genes and tumor suppression: A classical view in evolution. Cell Death Differ. 2007, 14, 3–9. [Google Scholar] [CrossRef]
- Elson, A.; Wang, Y.; Daugherty, C.J.; Morton, C.C.; Zhou, F.; Campos-Torres, J.; Leder, P. Pleiotropic defects in ataxia-telangiectasia protein-deficient mice. Proc. Natl. Acad. Sci. USA 1996, 93, 13084–13089. [Google Scholar]
- Easton, D.F. Cancer risks in a-t heterozygotes. Int. J. Radiat. Biol. 1994, 66, S177–S182. [Google Scholar] [CrossRef]
- Baer, R.; Ludwig, T. The brca1/bard1 heterodimer, a tumor suppressor complex with ubiquitin e3 ligase activity. Curr. Opin. Genet. Dev. 2002, 12, 86–91. [Google Scholar] [CrossRef]
- Miki, Y.; Swensen, J.; Shattuck-Eidens, D.; Futreal, P.A.; Harshman, K.; Tavtigian, S.; Liu, Q.; Cochran, C.; Bennett, L.M.; Ding, W.; et al. A strong candidate for the breast and ovarian cancer susceptibility gene brca1. Science 1994, 266, 66–71. [Google Scholar]
- Reidt, W.; Wurz, R.; Wanieck, K.; Chu, H.H.; Puchta, H. A homologue of the breast cancer-associated gene bard1 is involved in DNA repair in plants. EMBO J. 2006, 25, 4326–4337. [Google Scholar] [CrossRef]
- Doerner, P.; Jorgensen, J.E.; You, R.; Steppuhn, J.; Lamb, C. Control of root growth and development by cyclin expression. Nature 1996, 380, 520–523. [Google Scholar] [CrossRef]
- Kruman, I. DNA Repair; InTech: Rijeka, Croatia, 2011. [Google Scholar]
- Postel-Vinay, S.; Vanhecke, E.; Olaussen, K.A.; Lord, C.J.; Ashworth, A.; Soria, J.C. The potential of exploiting DNA-repair defects for optimizing lung cancer treatment. Nat. Rev. Clin. Oncol. 2012, 9, 144–155. [Google Scholar] [CrossRef]
- Britt, A.B. DNA damage and repair in plants. Annu. Rev. Plant Physiol. Plant Mol. Biol. 1996, 47, 75–100. [Google Scholar] [CrossRef]
- The Arabidopsis Information Resource. Available online: http://www.arabidopsis.org/ (accessed on 23 August 2013).
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Yoshiyama, K.O.; Sakaguchi, K.; Kimura, S. DNA Damage Response in Plants: Conserved and Variable Response Compared to Animals. Biology 2013, 2, 1338-1356. https://doi.org/10.3390/biology2041338
Yoshiyama KO, Sakaguchi K, Kimura S. DNA Damage Response in Plants: Conserved and Variable Response Compared to Animals. Biology. 2013; 2(4):1338-1356. https://doi.org/10.3390/biology2041338
Chicago/Turabian StyleYoshiyama, Kaoru Okamoto, Kengo Sakaguchi, and Seisuke Kimura. 2013. "DNA Damage Response in Plants: Conserved and Variable Response Compared to Animals" Biology 2, no. 4: 1338-1356. https://doi.org/10.3390/biology2041338
APA StyleYoshiyama, K. O., Sakaguchi, K., & Kimura, S. (2013). DNA Damage Response in Plants: Conserved and Variable Response Compared to Animals. Biology, 2(4), 1338-1356. https://doi.org/10.3390/biology2041338





