Direct RNA Sequencing Reveals Sex-Biased Transcriptomic and Epitranscriptomic Regulation in Procambarus clarkii
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. P. clarkii Sample Collection
2.2. Total RNA Extraction and mRNA Enrichment
2.3. Nanopore Sequencing Library Construction and Sequencing
2.4. Preprocessing, Alignment, and Novel Gene/Transcript Analysis
2.5. Transcript Structure Analysis
2.6. Isoform Poly(A) Length Analysis
2.7. RNA Methylation Analysis
2.8. Statistical Analysis
3. Results
3.1. Statistics of DRS Data of the Gonads of P. clarkii
3.2. Differential Gene Expression Analysis Between Gonads
3.3. Structure Analysis of P. clarkii Gonads
3.4. RNA Modification Analysis of P. clarkii Gonads
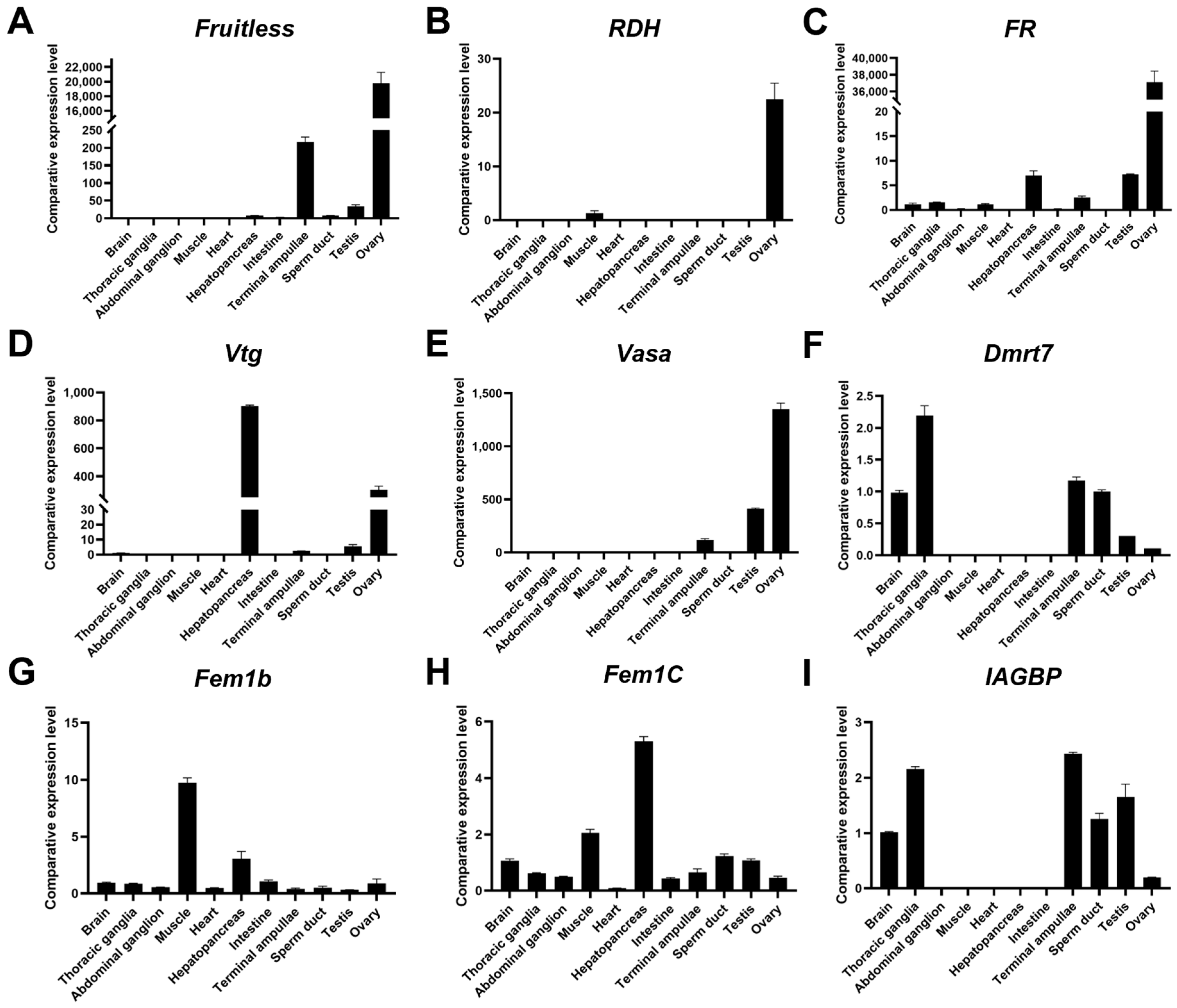
3.5. Verification of Potential Sex-Related Genes
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- National Aquatic Technology Promotion Station, China Fisheries Society. China’s Crayfish Industry Development Report (2025); Fishery and Fishery Administration Bureau, Ministry of Agriculture and Rural Affairs: Beijing, China, 2025.
- Shen, Y.; Wang, Q.; Wang, W.; Li, Y. Exploration of an XX/XY Sex Determination System and Development of PCR-Based Sex-Specific Markers in Procambarus clarkii Based on Next-Generation Sequencing Data. Front. Genet. 2022, 13, 850983. [Google Scholar] [CrossRef]
- Goudie, C.A.; Simco, B.A.; Davis, K.B.; Carmichael, G.J. Growth of Channel Catfish in Mixed Sex and Monosex Pond Culture. Aquaculture 1994, 128, 97–104. [Google Scholar] [CrossRef]
- Beardmore, J.A.; Mair, G.C.; Lewis, R.I. Monosex Male Production in Finfish as Exemplified by Tilapia: Applications, Problems, and Prospects. Aquaculture 2001, 197, 283–301. [Google Scholar] [CrossRef]
- Wahl, M.; Levy, T.; Ventura, T.; Sagi, A. Monosex Populations of the Giant Freshwater Prawn Macrobrachium rosenbergii—From a Pre-Molecular Start to the Next Generation Era. Int. J. Mol. Sci. 2023, 24, 17433. [Google Scholar] [CrossRef]
- Wahl, M.; Hongrath, K.; Thinbanmai, T.; Suriyaworakul, P.; Aflalo, E.D.; Shechter, A.; Sagi, A. Field Validation of an All-Female Monosex Biotechnology for the Freshwater Prawn Macrobrachium rosenbergii. Aquac. Rep. 2025, 42, 102733. [Google Scholar] [CrossRef]
- Ventura, T.; Manor, R.; Aflalo, E.D.; Weil, S.; Rosen, O.; Sagi, A. Timing Sexual Differentiation: Full Functional Sex Reversal Achieved Through Silencing of a Single Insulin-like Gene in the Prawn, Macrobrachium rosenbergii. Biol. Reprod. 2012, 86, 90. [Google Scholar] [CrossRef] [PubMed]
- Levy, T.; Rosen, O.; Manor, R.; Dotan, S.; Azulay, D.; Abramov, A.; Sklarz, M.Y.; Chalifa-Caspi, V.; Baruch, K.; Shechter, A.; et al. Production of WW Males Lacking the Masculine Z Chromosome and Mining the Macrobrachium rosenbergii Genome for Sex-Chromosomes. Sci. Rep. 2019, 9, 12408. [Google Scholar] [CrossRef] [PubMed]
- Miao, M.; Li, S.; Yuan, J.; Liu, P.; Fang, X.; Zhang, C.; Zhang, X.; Li, F. CRISPR/Cas9-Mediated Gene Mutation of EcIAG Leads to Sex Reversal in the Male Ridgetail White Prawn Exopalaemon carinicauda. Front. Endocrinol. 2023, 14, 1266641. [Google Scholar] [CrossRef] [PubMed]
- Cai, P.; Zhang, W.; Jiang, S.; Xiong, Y.; Yuan, H.; Gao, Z.; Gao, X.; Ma, C.; Zhou, Y.; Gong, Y.; et al. Insulin-like Androgenic Gland Hormone Induced Sex Reversal and Molecular Pathways in Macrobrachium nipponense: Insights into Reproduction, Growth, and Sex Differentiation. Int. J. Mol. Sci. 2023, 24, 14306. [Google Scholar] [CrossRef]
- Shi, L.; Han, S.; Fei, J.; Zhang, L.; Ray, J.W.; Wang, W.; Li, Y. Molecular Characterization and Functional Study of Insulin-like Androgenic Gland Hormone Gene in the Red Swamp Crayfish, Procambarus clarkii. Genes 2019, 10, 645. [Google Scholar] [CrossRef]
- Ge, H.-L.; Tan, K.; Shi, L.-L.; Sun, R.; Wang, W.-M.; Li, Y.-H. Comparison of Effects of DsRNA and SiRNA RNA Interference on Insulin-like Androgenic Gland Gene (IAG) in Red Swamp Crayfish Procambarus clarkii. Gene 2020, 752, 144783. [Google Scholar] [CrossRef]
- Xu, H.J.; Chen, Y.L.; Li, J.W.; Luo, J.Y.; Wang, Y.M.; Ma, W.M. A Novel Unique Terminal Ampullae-Expressed Insulin-like Peptide in Male White Shrimp, Penaeus vannamei. Aquac. Rep. 2022, 23, 101011. [Google Scholar] [CrossRef]
- Belote, J.M.; Baker, B.S. Sex Determination in Drosophila melanogaster: Analysis of Transformer-2, a Sex-Transforming Locus. Proc. Natl. Acad. Sci. USA 1982, 79, 1568–1572. [Google Scholar] [CrossRef]
- Kato, Y.; Kobayashi, K.; Watanabe, H.; Iguchi, T. Environmental Sex Determination in the Branchiopod Crustacean Daphnia magna: Deep Conservation of a Doublesex Gene in the Sex-Determining Pathway. PLoS Genet. 2011, 7, e1001345. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Colbourne, J.K. A Molecular Mechanism for Environmental Sex Determination. Trends Genet. 2024, 40, 817–818. [Google Scholar] [CrossRef]
- Xu, H.; Lin, W.; Ma, X.; Jawad, M.; Wu, M.; Qiu, J.; Li, M. A Timecourse Analysis of Gonadal Histology and Transcriptome during the Sexual Development of Yellow Catfish, Pelteobagrus fulvidraco. Comp. Biochem. Physiol. Part D Genom. Proteom. 2025, 56, 101576. [Google Scholar] [CrossRef] [PubMed]
- Shaked, S.A.; Levy, T.; Moscovitz, S.; Wattad, H.; Manor, R.; Ovadia, O.; Sagi, A.; Aflalo, E.D. All-Female Crayfish Populations for Biocontrol and Sustainable Aquaculture. Aquaculture 2024, 580, 740377. [Google Scholar] [CrossRef]
- Jiang, H.; Xing, Z.; Lu, W.; Qian, Z.; Yu, H.; Li, J. Transcriptome Analysis of Red Swamp Crawfish Procambarus clarkii Reveals Genes Involved in Gonadal Development. PLoS ONE 2014, 9, e105122. [Google Scholar] [CrossRef]
- Xu, Z.; Gao, T.; Xu, Y.; Li, X.; Li, J.; Lin, H.; Yan, W.; Pan, J.; Tang, J. A Chromosome-Level Reference Genome of Red Swamp Crayfish Procambarus clarkii Provides Insights into the Gene Families Regarding Growth or Development in Crustaceans. Genomics 2021, 113, 3274–3284. [Google Scholar] [CrossRef]
- Liao, M.; Xu, M.; Hu, R.; Xu, Z.; Bonvillain, C.; Li, Y.; Li, X.; Luo, X.; Wang, J.; Wang, J.; et al. The Chromosome-Level Genome Assembly of the Red Swamp Crayfish Procambarus clarkii. Sci. Data 2024, 11, 885. [Google Scholar] [CrossRef]
- Zhu, Z.; Younas, L.; Zhou, Q. Evolution and Regulation of Animal Sex Chromosomes. Nat. Rev. Genet. 2025, 26, 59–74. [Google Scholar] [CrossRef] [PubMed]
- Śnchez, L.; Granadino, B.; Torres, M. Sex Determination in Drosophila melanogaster: X-linked Genes Involved in the Initial Step of Sex-lethal Activation. Dev. Genet. 1994, 15, 251–264. [Google Scholar] [CrossRef]
- Kiuchi, T.; Koga, H.; Kawamoto, M.; Shoji, K.; Sakai, H.; Arai, Y.; Ishihara, G.; Kawaoka, S.; Sugano, S.; Shimada, T.; et al. A Single Female-Specific piRNA Is the Primary Determiner of Sex in the Silkworm. Nature 2014, 509, 633–636. [Google Scholar] [CrossRef] [PubMed]
- Salz, H.K. Sex Determination in Insects: A Binary Decision Based on Alternative Splicing. Curr. Opin. Genet. Dev. 2011, 21, 395–400. [Google Scholar] [CrossRef] [PubMed]
- Stévant, I.; Nef, S. Genetic Control of Gonadal Sex Determination and Development. Trends Genet. 2019, 35, 346–358. [Google Scholar] [CrossRef]
- Xiao, W.; Adhikari, S.; Dahal, U.; Chen, Y.S.; Hao, Y.J.; Sun, B.F.; Sun, H.Y.; Li, A.; Ping, X.L.; Lai, W.Y.; et al. Nuclear m6A Reader YTHDC1 Regulates mRNA Splicing. Mol. Cell 2016, 61, 507–519. [Google Scholar] [CrossRef]
- Haussmann, I.U.; Bodi, Z.; Sanchez-Moran, E.; Mongan, N.P.; Archer, N.; Fray, R.G.; Soller, M. m6A Potentiates Sxl Alternative Pre-mRNA Splicing for Robust Drosophila Sex Determination. Nature 2016, 540, 301–304. [Google Scholar] [CrossRef]
- Wang, X.; Lu, Z.; Gomez, A.; Hon, G.C.; Yue, Y.; Han, D.; Fu, Y.; Parisien, M.; Dai, Q.; Jia, G.; et al. N6-Methyladenosine-Dependent Regulation of Messenger RNA Stability. Nature 2014, 505, 117–120. [Google Scholar] [CrossRef]
- Wang, X.; Zhao, B.S.; Roundtree, I.A.; Lu, Z.; Han, D.; Ma, H.; Weng, X.; Chen, K.; Shi, H.; He, C. N6-Methyladenosine Modulates Messenger RNA Translation Efficiency. Cell 2015, 161, 1388–1399. [Google Scholar] [CrossRef]
- Perlegos, A.E.; Shields, E.J.; Shen, H.; Liu, K.F.; Bonini, N.M. Mettl3-Dependent m6A Modification Attenuates the Brain Stress Response in Drosophila. Nat. Commun. 2022, 13, 5387. [Google Scholar] [CrossRef]
- Jalloh, B.; Lancaster, C.L.; Rounds, J.C.; Brown, B.E.; Leung, S.W.; Banerjee, A.; Morton, D.J.; Bienkowski, R.S.; Fasken, M.B.; Kremsky, I.J.; et al. The Drosophila Nab2 RNA Binding Protein Inhibits M6A Methylation and Male-Specific Splicing of Sex Lethal Transcript in Female Neuronal Tissue. eLife 2023, 12, e64904. [Google Scholar] [CrossRef] [PubMed]
- Richter, J.D. Cytoplasmic Polyadenylation in Development and Beyond. Microbiol. Mol. Biol. Rev. 1999, 63, 446–456. [Google Scholar] [CrossRef] [PubMed]
- Chandler, J.C.; Fitzgibbon, Q.P.; Smith, G.; Elizur, A.; Ventura, T. Y-Linked iDmrt1 Paralogue (iDMY) in the Eastern Spiny Lobster, Sagmariasus verreauxi: The First Invertebrate Sex-Linked Dmrt. Dev. Biol. 2017, 430, 337–345. [Google Scholar] [CrossRef] [PubMed]
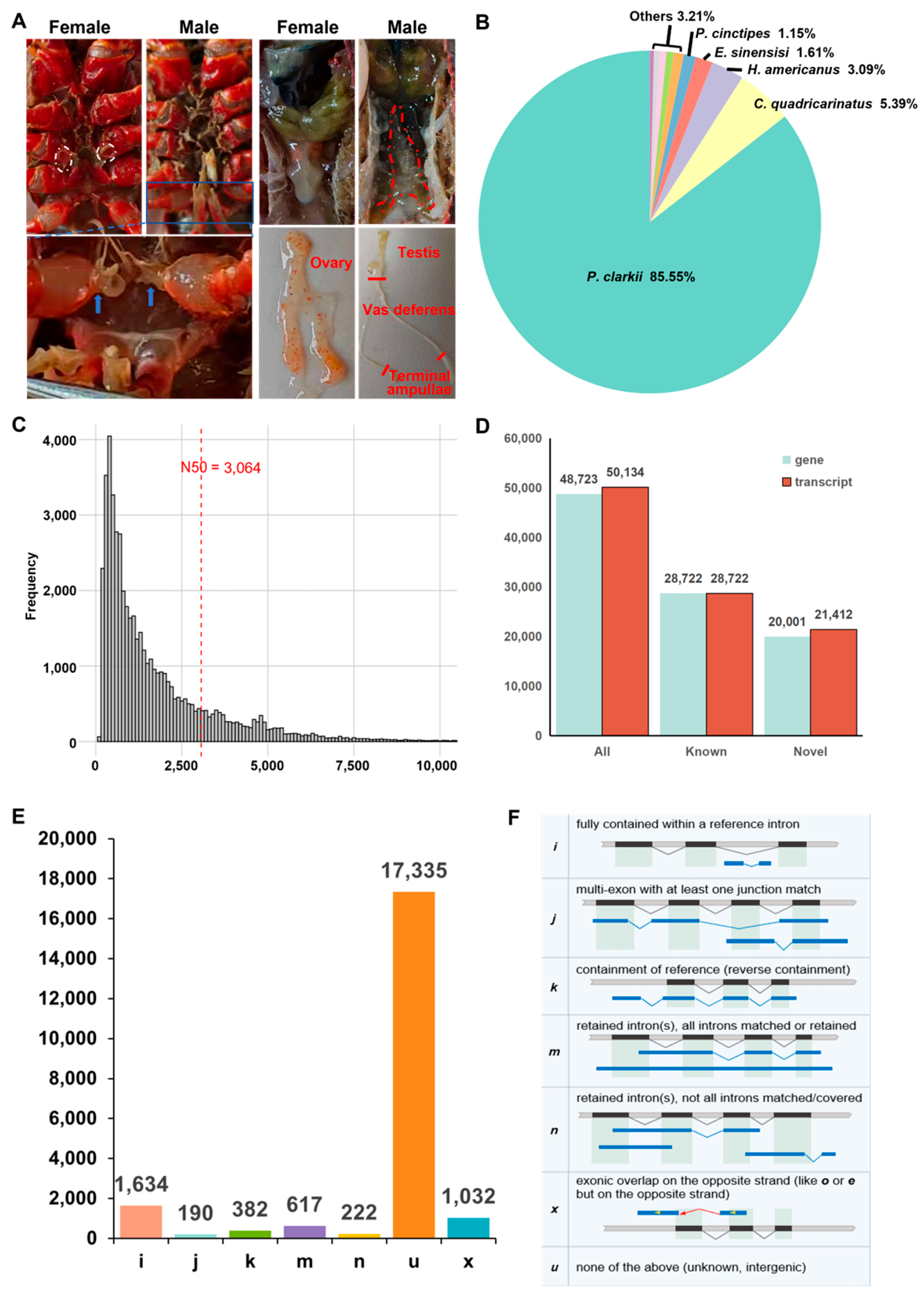
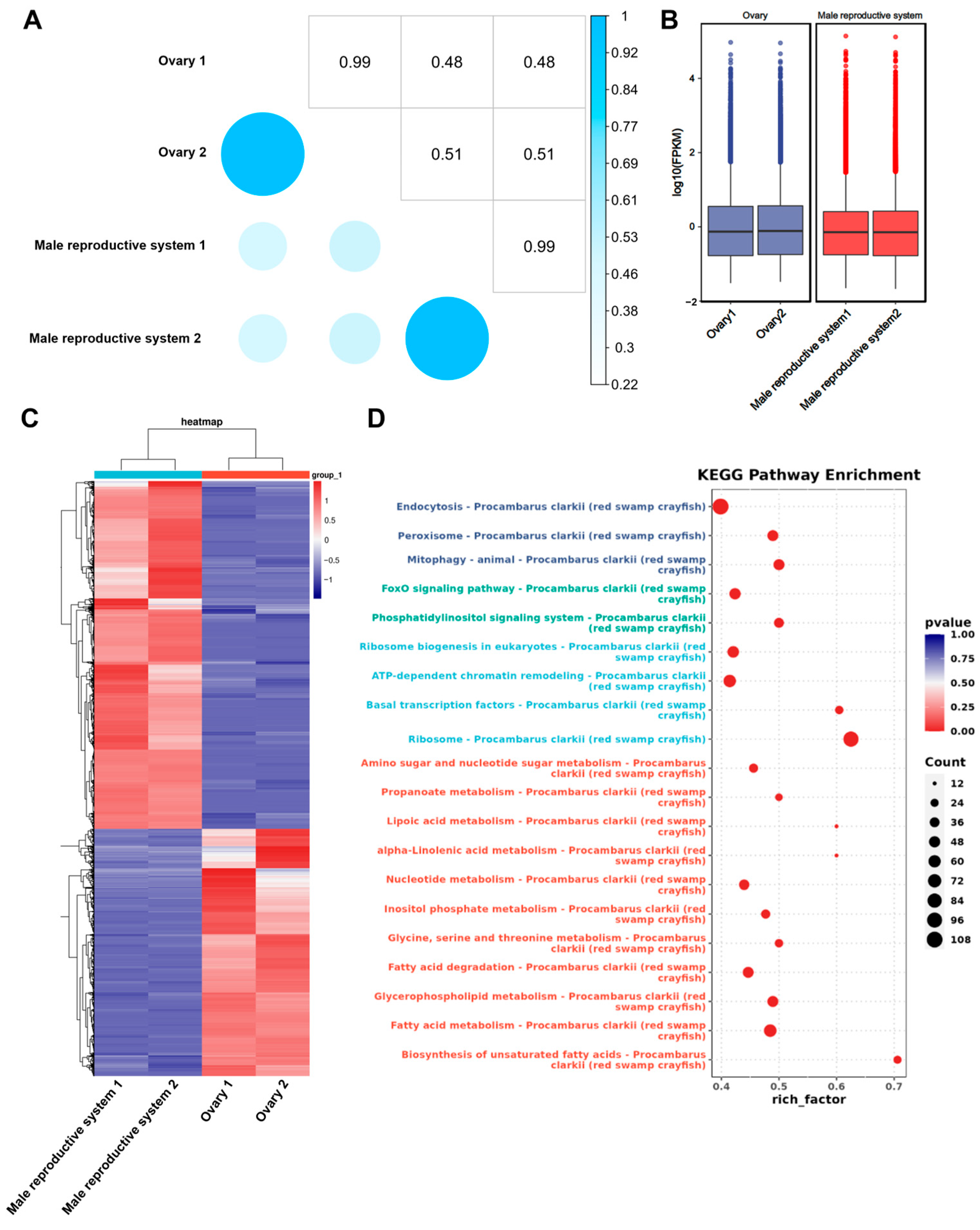
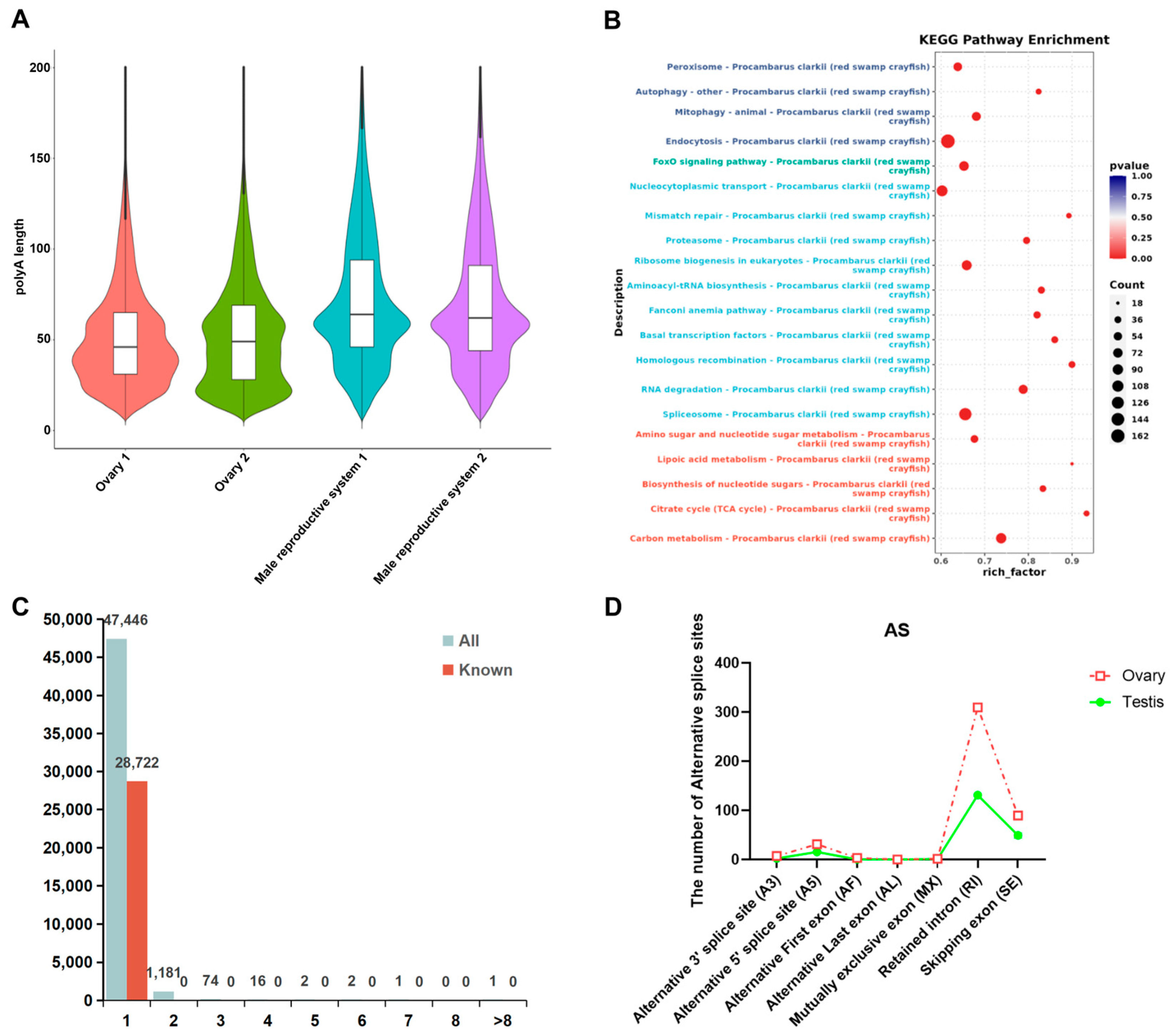
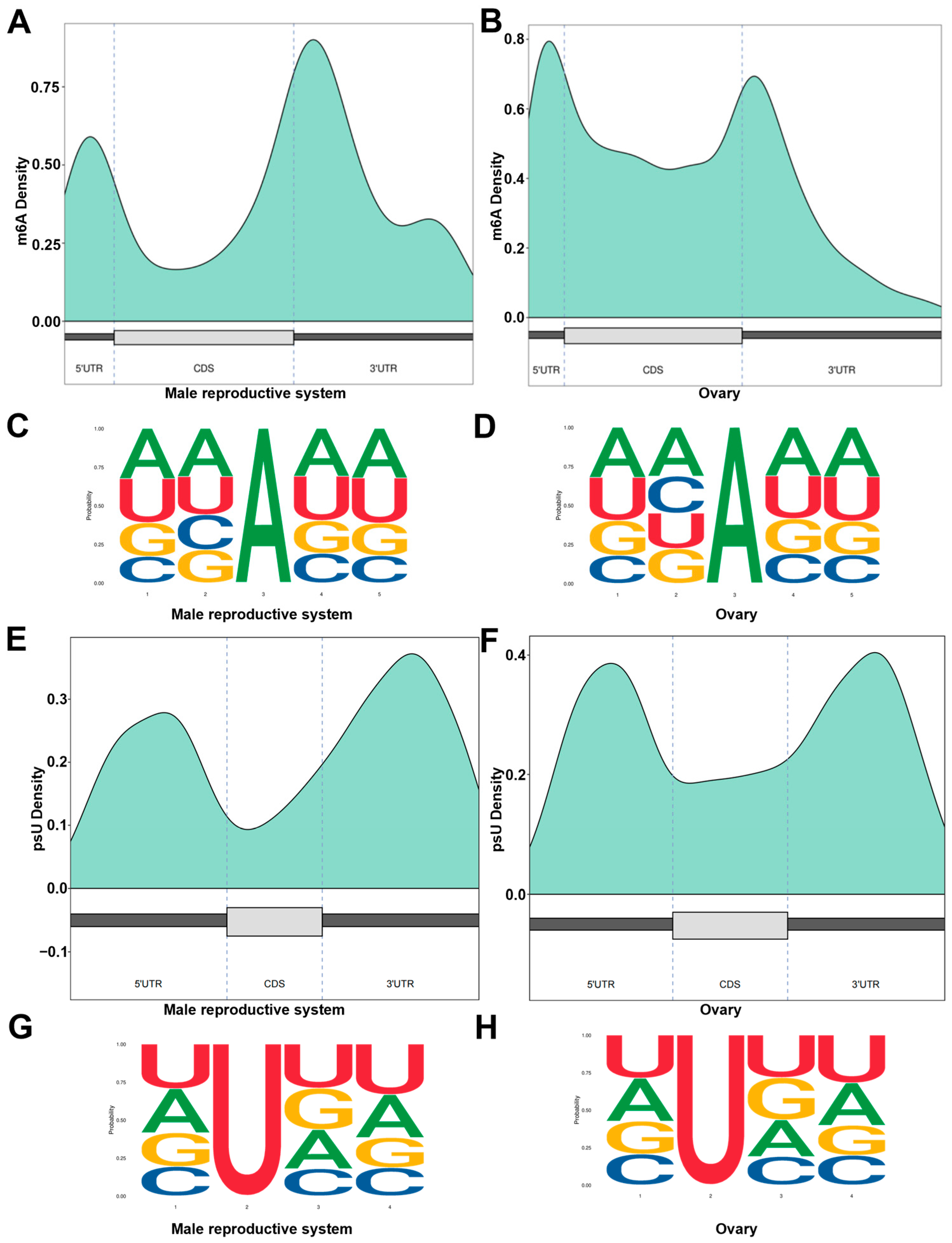
| Sample | Type | Total Base | Total Reads | Max Len. | Avg. Len. | N50 | L50 | N90 | L90 |
|---|---|---|---|---|---|---|---|---|---|
| Ovary 1 | all | 6.71 Gb | 6.86 Mb | 352,878 | 1001.8 | 1333 | 1.51 Mb | 587 | 4.47 Mb |
| Ovary 1 | pass | 6.55 Gb | 6.28 Mb | 352,878 | 1067.61 | 1337 | 1.48 Mb | 600 | 4.34 Mb |
| Ovary 2 | all | 6.39 Gb | 6.80 Mb | 447,796 | 961.94 | 1268 | 1.54 Mb | 568 | 4.48 Mb |
| Ovary 2 | pass | 6.22 Gb | 6.18 Mb | 447,796 | 1031.09 | 1270 | 1.50 Mb | 581 | 4.30 Mb |
| Male reproductive system 1 | all | 6.51 Gb | 11.13 Mb | 426,539 | 598.9 | 1007 | 2.01 Mb | 332 | 6.41 Mb |
| Male reproductive system 1 | pass | 6.26 Gb | 9.5 Mb | 426,539 | 673.61 | 1010 | 1.96 Mb | 347 | 6.11 Mb |
| Male reproductive system 2 | all | 6.79 Gb | 12.17 Mb | 429,741 | 571.24 | 962 | 2.24 Mb | 318 | 7.04 Mb |
| Male reproductive system 2 | pass | 6.57 Gb | 10.18 Mb | 429,741 | 661.1 | 971 | 2.17 Mb | 333 | 6.71 Mb |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Xu, H.; Song, G.; Luo, Y.; Zhang, H.; Jawad, M.; Zhang, W.; Li, T.; Zhao, D.; Yang, C.; Wang, A.; et al. Direct RNA Sequencing Reveals Sex-Biased Transcriptomic and Epitranscriptomic Regulation in Procambarus clarkii. Biology 2025, 14, 1757. https://doi.org/10.3390/biology14121757
Xu H, Song G, Luo Y, Zhang H, Jawad M, Zhang W, Li T, Zhao D, Yang C, Wang A, et al. Direct RNA Sequencing Reveals Sex-Biased Transcriptomic and Epitranscriptomic Regulation in Procambarus clarkii. Biology. 2025; 14(12):1757. https://doi.org/10.3390/biology14121757
Chicago/Turabian StyleXu, Haijing, Guangtong Song, Yichen Luo, Haoxuan Zhang, Muhammad Jawad, Wei Zhang, Tao Li, Dawei Zhao, Chunyan Yang, Aimin Wang, and et al. 2025. "Direct RNA Sequencing Reveals Sex-Biased Transcriptomic and Epitranscriptomic Regulation in Procambarus clarkii" Biology 14, no. 12: 1757. https://doi.org/10.3390/biology14121757
APA StyleXu, H., Song, G., Luo, Y., Zhang, H., Jawad, M., Zhang, W., Li, T., Zhao, D., Yang, C., Wang, A., & Li, M. (2025). Direct RNA Sequencing Reveals Sex-Biased Transcriptomic and Epitranscriptomic Regulation in Procambarus clarkii. Biology, 14(12), 1757. https://doi.org/10.3390/biology14121757







