Parameterization of the Differences in Neural Oscillations Recorded by Wearable Magnetoencephalography for Chinese Semantic Cognition
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Subjects and Experiment
2.2. Data Acquisition and Preprocessing
2.2.1. Acquisition and Preprocessing of OPM-MEG Signals
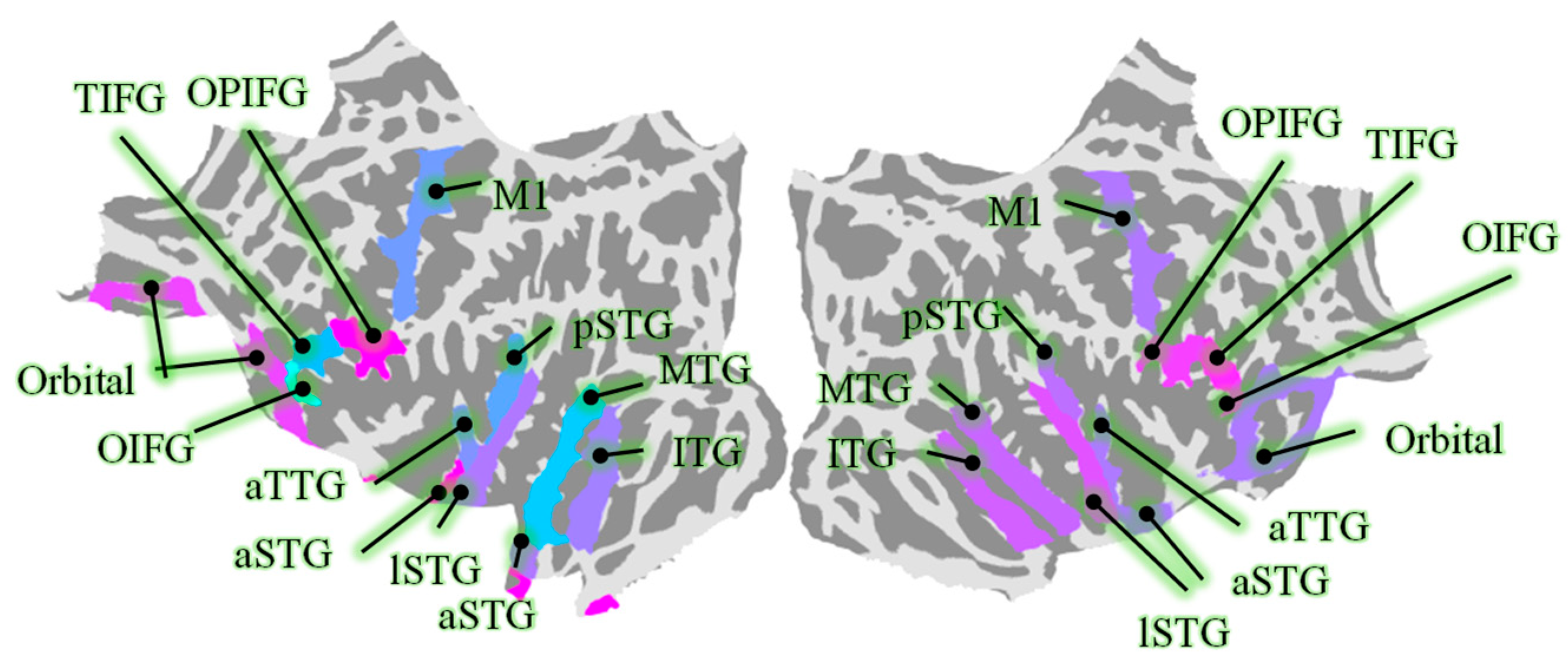
2.2.2. Source Reconstruction of ROI
2.3. The Time–Frequency Analysis of the Differences Between Neural Time Series Based on SLT and Cluster Depth Tests
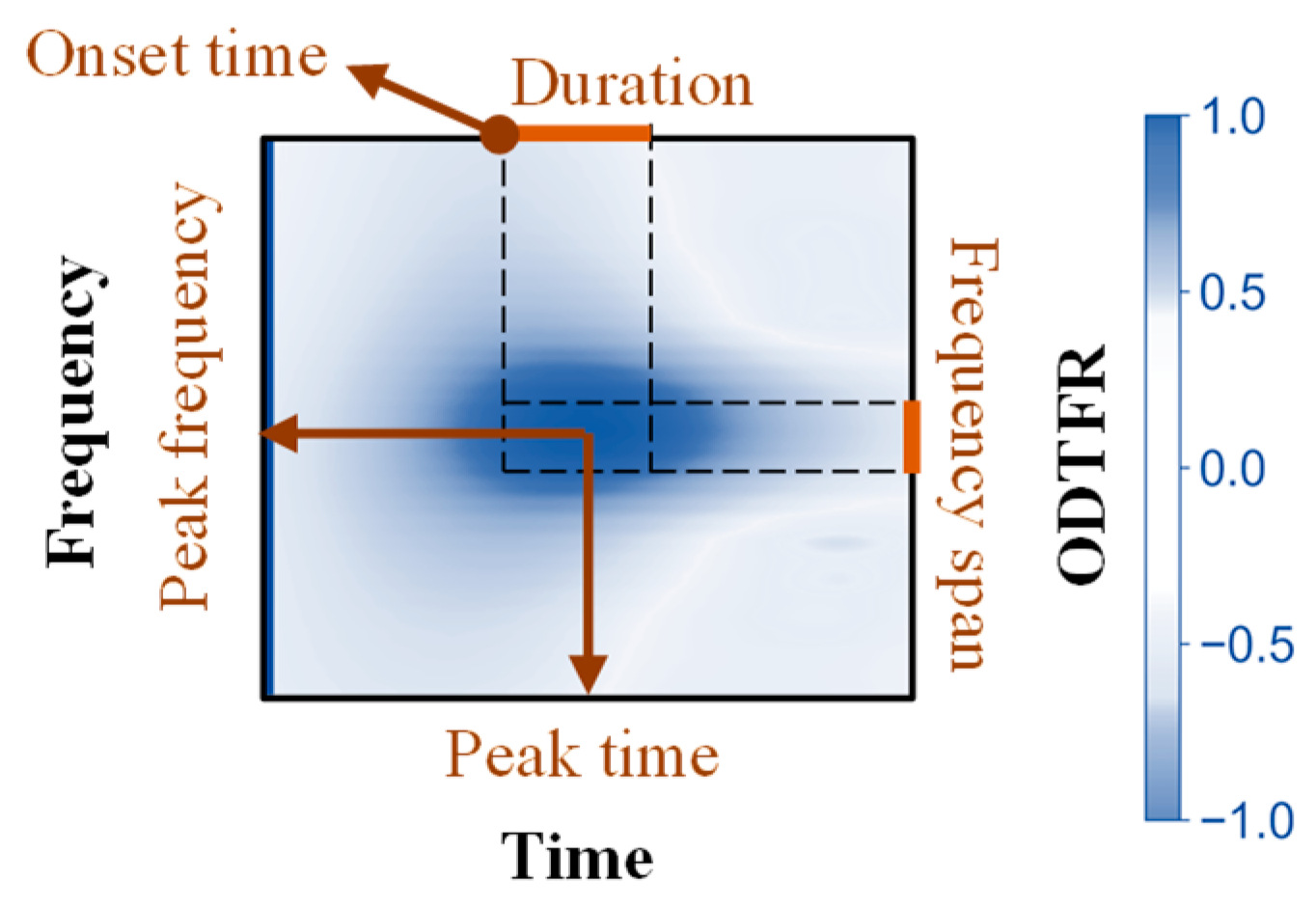
2.4. Parameterization of Local Oscillatory Differences
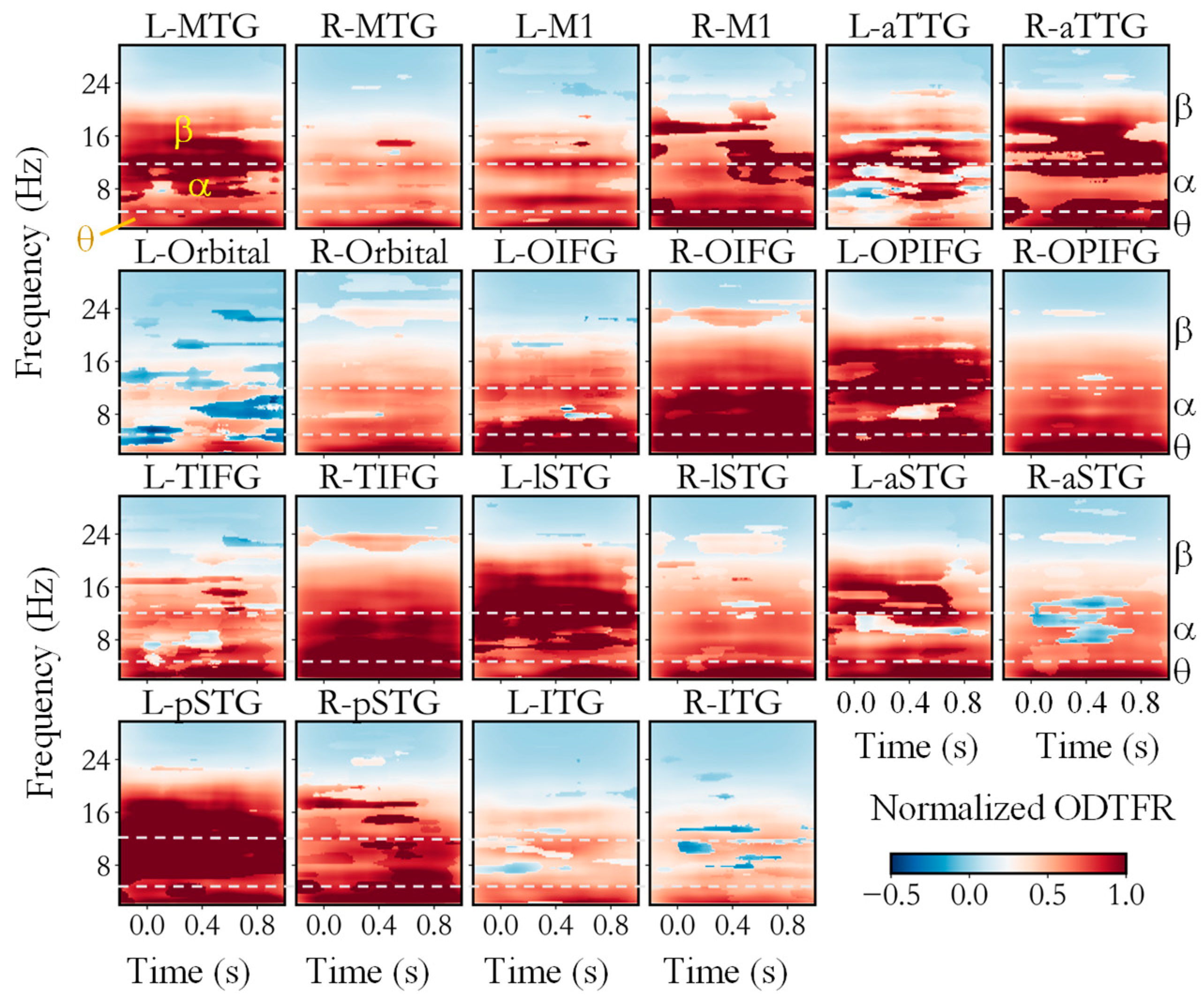
3. Results
4. Discussion
4.1. Neural Activities During Semantic Processing
4.2. The Techniques for Analyzing Neural Activity During Semantic Processing
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Kutas, M.; Hillyard, S.A. Reading Senseless Sentences: Brain Potentials Reflect Semantic Incongruity. Science 1980, 207, 203–205. [Google Scholar] [CrossRef] [PubMed]
- Wu, H.; Liang, X.; Wang, R.; Ma, Y.; Gao, Y.; Ning, X. A Multivariate Analysis on Evoked Components of Chinese Semantic Congruity: An OP-MEG Study with EEG. Cereb. Cortex 2024, 34, bhae108. [Google Scholar] [CrossRef] [PubMed]
- Toro-Hernández, F.D.; Migeot, J.; Marchant, N.; Olivares, D.; Ferrante, F.; González-Gómez, R.; González Campo, C.; Fittipaldi, S.; Rojas-Costa, G.M.; Moguilner, S.; et al. Neurocognitive Correlates of Semantic Memory Navigation in Parkinson’s Disease. npj Park. Dis. 2024, 10, 15. [Google Scholar] [CrossRef]
- Irish, M.; Addis, D.R.; Hodges, J.R.; Piguet, O. Considering the Role of Semantic Memory in Episodic Future Thinking: Evidence from Semantic Dementia. Brain 2012, 135, 2178–2191. [Google Scholar] [CrossRef] [PubMed]
- Broderick, M.P.; Anderson, A.J.; Lalor, E.C. Semantic Context Enhances the Early Auditory Encoding of Natural Speech. J. Neurosci. 2019, 39, 7564–7575. [Google Scholar] [CrossRef]
- Almeida, V.N. Neurophysiological Basis of the N400 Deflection, from Mismatch Negativity to Semantic Prediction Potentials and Late Positive Components. Int. J. Psychophysiol. 2021, 166, 134–150. [Google Scholar] [CrossRef] [PubMed]
- Heidlmayr, K.; Ferragne, E.; Isel, F. Neuroplasticity in the Phonological System: The PMN and the N400 as Markers for the Perception of Non-Native Phonemic Contrasts by Late Second Language Learners. Neuropsychologia 2021, 156, 107831. [Google Scholar] [CrossRef]
- Herman, D.; Baker, S.; Chow, R.; Cazes, J.; Alain, C.; Rosenbaum, R.S. Mismatch Negativity as a Marker of Auditory Pattern Separation. Cereb. Cortex 2023, 33, 10181–10193. [Google Scholar] [CrossRef] [PubMed]
- Lau, E.F.; Phillips, C.; Poeppel, D. A Cortical Network for Semantics: (De)Constructing the N400. Nat. Rev. Neurosci. 2008, 9, 920–933. [Google Scholar] [CrossRef]
- Morett, L.M.; Landi, N.; Irwin, J.; McPartland, J.C. N400 Amplitude, Latency, and Variability Reflect Temporal Integration of Beat Gesture and Pitch Accent during Language Processing. Brain Res. 2020, 1747, 147059. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Jensen, O.; van den Brink, D.; Weder, N.; Schoffelen, J.-M.; Magyari, L.; Hagoort, P.; Bastiaansen, M. Beta Oscillations Relate to the N400m during Language Comprehension. Hum. Brain Mapp. 2012, 33, 2898–2912. [Google Scholar] [CrossRef] [PubMed]
- Kutas, M.; Federmeier, K.D. Thirty Years and Counting: Finding Meaning in the N400 Component of the Event Related Brain Potential (ERP). Annu. Rev. Psychol. 2011, 62, 621–647. [Google Scholar] [CrossRef] [PubMed]
- Boto, E.; Holmes, N.; Leggett, J.; Roberts, G.; Shah, V.; Meyer, S.S.; Muñoz, L.D.; Mullinger, K.J.; Tierney, T.M.; Bestmann, S.; et al. Moving Magnetoencephalography towards Real-World Applications with a Wearable System. Nature 2018, 555, 657–661. [Google Scholar] [CrossRef] [PubMed]
- Wang, R.; Liang, X.; Wu, H.; Yang, Y.; Zhao, R.; Gao, Y.; Ning, X. Performance Evaluation of Interference Removal Methods Based on Subspace Projection with Wearable OPM-MEG. IEEE Trans. Instrum. Meas. 2024, 73, 9514922. [Google Scholar] [CrossRef]
- An, N.; Cao, F.; Li, W.; Wang, W.; Xu, W.; Wang, C.; Gao, Y.; Xiang, M.; Ning, X. Multiple Source Detection Based on Spatial Clustering and Its Applications on Wearable OPM-MEG. IEEE Trans. Bio. Med. Eng. 2022, 69, 3131–3141. [Google Scholar] [CrossRef] [PubMed]
- Geller, A.S.; Teale, P.; Kronberg, E.; Ebersole, J.S. Magnetoencephalography for Epilepsy Presurgical Evaluation. Curr. Neurol. Neurosci. Rep. 2023, 24, 35–46. [Google Scholar] [CrossRef] [PubMed]
- Ma, Y.; Gao, Y.; Liang, X.; Wu, H.; Gao, Z.; Cao, F.; Li, Y.; Lu, H.; Liu, C.; Ning, X. Evaluating the Performance of Optically Pumped Magnetometer Magnetoencephalography in Measuring Inter-Trial and Inter-Region Phase-Locking Value. Measurement 2024, 237, 115149. [Google Scholar] [CrossRef]
- Brookes, M.J.; Leggett, J.; Rea, M.; Hill, R.M.; Holmes, N.; Boto, E.; Bowtell, R. Magnetoencephalography with Optically Pumped Magnetometers (OPM-MEG): The next Generation of Functional Neuroimaging. Trends Neurosci. 2022, 45, 621–634. [Google Scholar] [CrossRef] [PubMed]
- Wu, H.; Wang, R.; Ma, Y.; Liang, X.; Liu, C.; Yu, D.; An, N.; Ning, X. Decoding N400m Evoked Component: A Tutorial on Multivariate Pattern Analysis for OP-MEG Data. Bioengineering 2024, 11, 609. [Google Scholar] [CrossRef]
- Doelling, K.B.; Assaneo, M.F. Neural Oscillations Are a Start toward Understanding Brain Activity Rather than the End. PLoS Biol. 2021, 19, e3001234. [Google Scholar] [CrossRef] [PubMed]
- Hincapié Casas, A.S.; Lajnef, T.; Pascarella, A.; Guiraud-Vinatea, H.; Laaksonen, H.; Bayle, D.; Jerbi, K.; Boulenger, V. Neural Oscillations Track Natural but Not Artificial Fast Speech: Novel Insights from Speech-Brain Coupling Using MEG. NeuroImage 2021, 244, 118577. [Google Scholar] [CrossRef] [PubMed]
- Düzel, E.; Penny, W.D.; Burgess, N. Brain Oscillations and Memory. Curr. Opin. Neurobiol. 2010, 20, 143–149. [Google Scholar] [CrossRef] [PubMed]
- Packard, P.A.; Steiger, T.K.; Fuentemilla, L.; Bunzeck, N. Neural Oscillations and Event-Related Potentials Reveal How Semantic Congruence Drives Long-Term Memory in Both Young and Older Humans. Sci. Rep. 2020, 10, 9116. [Google Scholar] [CrossRef] [PubMed]
- Friederici, A.D. Towards a Neural Basis of Auditory Sentence Processing. Trends Cogn. Sci. 2002, 6, 78–84. [Google Scholar] [CrossRef]
- Mamashli, F.; Khan, S.; Obleser, J.; Friederici, A.D.; Maess, B. Oscillatory Dynamics of Cortical Functional Connections in Semantic Prediction. Hum. Brain Mapp. 2019, 40, 1856–1866. [Google Scholar] [CrossRef] [PubMed]
- Shahin, A.J.; Picton, T.W.; Miller, L.M. Brain Oscillations during Semantic Evaluation of Speech. Brain Cogn. 2009, 70, 259–266. [Google Scholar] [CrossRef] [PubMed]
- Moca, V.V.; Bârzan, H.; Nagy-Dăbâcan, A.; Mureșan, R.C. Time-Frequency Super-Resolution with Superlets. Nat. Commun. 2021, 12, 337. [Google Scholar] [CrossRef] [PubMed]
- Tripathi, P.M.; Kumar, A.; Kumar, M.; Komaragiri, R.S. Automatic Seizure Detection and Classification Using Super-Resolution Superlet Transform and Deep Neural Network -A Preprocessing-Less Method. Comput. Methods Programs Biomed. 2023, 240, 107680. [Google Scholar] [CrossRef] [PubMed]
- Lewis, A.G.; Schoffelen, J.-M.; Hoffmann, C.; Bastiaansen, M.; Schriefers, H. Discourse-Level Semantic Coherence Influences Beta Oscillatory Dynamics and the N400 during Sentence Comprehension. Lang. Cogn. Neurosci. 2017, 32, 601–617. [Google Scholar] [CrossRef]
- Mai, G.; Minett, J.W.; Wang, W.S.-Y. Delta, Theta, Beta, and Gamma Brain Oscillations Index Levels of Auditory Sentence Processing. NeuroImage 2016, 133, 516–528. [Google Scholar] [CrossRef]
- Strauß, A.; Kotz, S.A.; Scharinger, M.; Obleser, J. Alpha and Theta Brain Oscillations Index Dissociable Processes in Spoken Word Recognition. NeuroImage 2014, 97, 387–395. [Google Scholar] [CrossRef] [PubMed]
- Noguchi, Y. Combinatorial Binding of Semantic Information through the Sharing of Neural Oscillatory Signals. bioRxiv 2023. [Google Scholar] [CrossRef]
- Piai, V.; Roelofs, A.; van der Meij, R. Event-Related Potentials and Oscillatory Brain Responses Associated with Semantic and Stroop-like Interference Effects in Overt Naming. Brain Res. 2012, 1450, 87–101. [Google Scholar] [CrossRef] [PubMed]
- Keitel, A.; Ince, R.A.A.; Gross, J.; Kayser, C. Auditory Cortical Delta-Entrainment Interacts with Oscillatory Power in Multiple Fronto-Parietal Networks. NeuroImage 2017, 147, 32–42. [Google Scholar] [CrossRef] [PubMed]
- Maris, E. Statistical Testing in Electrophysiological Studies: Statistical Testing in Electrophysiological Studies. Psychophysiology 2012, 49, 549–565. [Google Scholar] [CrossRef] [PubMed]
- Smith, S.M.; Nichols, T.E. Threshold-Free Cluster Enhancement: Addressing Problems of Smoothing, Threshold Dependence and Localisation in Cluster Inference. NeuroImage 2009, 44, 83–98. [Google Scholar] [CrossRef]
- Frossard, J.; Renaud, O. The Cluster Depth Tests: Toward Point-Wise Strong Control of the Family-Wise Error Rate in Massively Univariate Tests with Application to M/EEG. NeuroImage 2022, 247, 118824. [Google Scholar] [CrossRef]
- Cross, Z.R.; Corcoran, A.W.; Schlesewsky, M.; Kohler, M.J.; Bornkessel-Schlesewsky, I. Oscillatory and Aperiodic Neural Activity Jointly Predict Language Learning. J. Cogn. Neurosci. 2022, 34, 1630–1649. [Google Scholar] [CrossRef]
- Jafari, Z.; Kolb, B.E.; Mohajerani, M.H. Neural Oscillations and Brain Stimulation in Alzheimer’s Disease. Prog. Neurobiol. 2020, 194, 101878. [Google Scholar] [CrossRef] [PubMed]
- Calderone, D.J.; Lakatos, P.; Butler, P.D.; Castellanos, F.X. Entrainment of Neural Oscillations as a Modifiable Substrate of Attention. Trends Cogn. Sci. 2014, 18, 300–309. [Google Scholar] [CrossRef] [PubMed]
- Zhu, Y.; Xu, M.; Lu, J.; Hu, J.; Kwok, V.P.Y.; Zhou, Y.; Yuan, D.; Wu, B.; Zhang, J.; Wu, J.; et al. Distinct Spatiotemporal Patterns of Syntactic and Semantic Processing in Human Inferior Frontal Gyrus. Nat. Hum. Behav. 2022, 6, 1104–1111. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Q.; Wang, H.; Luo, C.; Zhang, J.; Jin, Z.; Li, L. The Neural Basis of Semantic Cognition in Mandarin Chinese: A Combined fMRI and TMS Study. Hum. Brain Mapp. 2019, 40, 5412–5423. [Google Scholar] [CrossRef] [PubMed]
- Ablin, P.; Cardoso, J.-F.; Gramfort, A. Faster Independent Component Analysis by Preconditioning With Hessian Approximations. IEEE Trans. Signal Process. 2018, 66, 4040–4049. [Google Scholar] [CrossRef]
- Fischl, B. FreeSurfer. NeuroImage 2012, 62, 774–781. [Google Scholar] [CrossRef]
- Gramfort, A.; Luessi, M.; Larson, E.; Engemann, D.A.; Strohmeier, D.; Brodbeck, C.; Parkkonen, L.; Hämäläinen, M.S. MNE Software for Processing MEG and EEG Data. NeuroImage 2014, 86, 446–460. [Google Scholar] [CrossRef] [PubMed]
- Destrieux, C.; Fischl, B.; Dale, A.; Halgren, E. Automatic Parcellation of Human Cortical Gyri and Sulci Using Standard Anatomical Nomenclature. NeuroImage 2010, 53, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Dale, A.M.; Liu, A.K.; Fischl, B.R.; Buckner, R.L.; Belliveau, J.W.; Lewine, J.D.; Halgren, E. Dynamic Statistical Parametric Mapping: Combining fMRI and MEG for High-Resolution Imaging of Cortical Activity. Neuron 2000, 26, 55–67. [Google Scholar] [CrossRef] [PubMed]
- Brady, B.; Bardouille, T. Periodic/Aperiodic Parameterization of Transient Oscillations (PAPTO)–Implications for Healthy Ageing. NeuroImage 2022, 251, 118974. [Google Scholar] [CrossRef] [PubMed]
- Shin, H.; Law, R.; Tsutsui, S.; Moore, C.I.; Jones, S.R. The Rate of Transient Beta Frequency Events Predicts Behavior across Tasks and Species. eLife 2017, 6, e29086. [Google Scholar] [CrossRef] [PubMed]
- Lundqvist, M.; Miller, E.K.; Nordmark, J.; Liljefors, J.; Herman, P. Beta: Bursts of Cognition. Trends Cogn. Sci. 2024, 28, 662–676. [Google Scholar] [CrossRef] [PubMed]
- Sauer, A.; Grent-’t-Jong, T.; Zeev-Wolf, M.; Singer, W.; Goldstein, A.; Uhlhaas, P.J. Spectral and Phase-Coherence Correlates of Impaired Auditory Mismatch Negativity (MMN) in Schizophrenia: A MEG Study. Schizophr. Res. 2023, 261, 60–71. [Google Scholar] [CrossRef] [PubMed]
- Hua, J.P.Y.; Roach, B.J.; Ford, J.M.; Mathalon, D.H. Mismatch Negativity and Theta Oscillations Evoked by Auditory Deviance in Early Schizophrenia. Biol. Psychiatry Cogn. Neurosci. Neuroimaging 2023, 8, 1186–1196. [Google Scholar] [CrossRef]
- Hughes, A.M.; Whitten, T.A.; Caplan, J.B.; Dickson, C.T. BOSC: A Better Oscillation Detection Method, Extracts Both Sustained and Transient Rhythms from Rat Hippocampal Recordings. Hippocampus 2012, 22, 1417–1428. [Google Scholar] [CrossRef] [PubMed]
- Hancock, R.; Pugh, K.R.; Hoeft, F. Neural Noise Hypothesis of Developmental Dyslexia. Trends Cogn. Sci. 2017, 21, 434–448. [Google Scholar] [CrossRef] [PubMed]
- Bertone, A.; Mottron, L.; Jelenic, P.; Faubert, J. Enhanced and Diminished Visuo-Spatial Information Processing in Autism Depends on Stimulus Complexity. Brain 2005, 128, 2430–2441. [Google Scholar] [CrossRef] [PubMed]
- Donoghue, T.; Haller, M.; Peterson, E.J.; Varma, P.; Sebastian, P.; Gao, R.; Noto, T.; Lara, A.H.; Wallis, J.D.; Knight, R.T.; et al. Parameterizing Neural Power Spectra into Periodic and Aperiodic Components. Nat. Neurosci. 2020, 23, 1655–1665. [Google Scholar] [CrossRef] [PubMed]
- Wilson, L.E.; da Silva Castanheira, J.; Baillet, S. Time-Resolved Parameterization of Aperiodic and Periodic Brain Activity. eLife 2022, 11, e77348. [Google Scholar] [CrossRef] [PubMed]
- Elouaham, S.; Dliou, A.; Laaboubi, M.; Latif, R.; Elkamoun, N.; Zougagh, H. Filtering and Analyzing Normal and Abnormal Electromyogram Signals. Indones. J. Electr. Eng. Comput. Sci. 2020, 20, 176–184. [Google Scholar] [CrossRef]
| ROI | Mean ± Std * | ROI | Mean ± Std * | ROI | Mean ± Std * | ROI | Mean ± Std * |
|---|---|---|---|---|---|---|---|
| L-MTG | 42.7 ± 8.7 | L-Orbital | 36.6 ± 2.7 | L-TIFG | 18.6 ± 3.2 | L-pSTG | 24.6 ± 4.2 |
| R-MTG | 50.8 ± 9.6 | R-Orbital | 43.7 ± 3.4 | R-TIFG | 15.8 ± 2.9 | R-pSTG | 21.7 ± 4.2 |
| L-M1 | 50.8 ± 7.1 | L-OIFG | 7.1 ± 2.4 | L-lSTG | 37.1 ± 5.3 | L-ITG | 40.6 ± 3.1 |
| R-M1 | 54.6 ± 7.2 | R-OIFG | 5.3 ± 0.7 | R-lSTG | 34 ± 5.3 | R-ITG | 37.6 ± 4.1 |
| L-aTTG | 10.4 ± 1.2 | L-OPIFG | 25.3 ± 1.8 | L-aSTG | 15.3 ± 2.6 | ||
| R-aTTG | 7.9 ± 0.6 | R-OPIFG | 26.1 ± 4.1 | R-aSTG | 20.6 ± 2.9 |
| Parameters | Definition |
|---|---|
| Peak Frequency | The frequency corresponding to local peaks in the ODTFR, where the difference in power is maximized. |
| Frequency span | The frequency span corresponding to the half-width of local peaks in the ODTFR and the frequency span over which the difference occurs. |
| Peak Time | The moment corresponding to local peaks in the ODTFR, where the difference in power is maximized. |
| Onset Time | The onset time corresponding to the half-width of local peaks in the ODTFR and the time at which the difference begins to appear. |
| Duration | The duration of time corresponding to the half-width of local peaks in the ODTFR and the duration over which the difference persists. |
| Band | ROI | Duration (ms) | Peak Frequency (Hz) | Peak Time (ms) | Onset Time (ms) |
|---|---|---|---|---|---|
| theta | L-MTG | 400 ± 37 | 7.58 ± 0.18 | 499 ± 30 | 301 ± 34 |
| L-Orbital | 300 ± 10 | 6 ± 0 | 371 ± 31 | 370 ± 31 | |
| L-TIFG | 116 ± 57 | 4.5 ± 1.13 | 234 ± 46 | 187 ± 23 | |
| L-aTTG | 566 ± 42 | 4.38 ± 0.57 | 536 ± 18 | 243 ± 21 | |
| L-lSTG | 146 ± 58 | 6.58 ± 0.08 | 502 ± 106 | 406 ± 103 | |
| L-pSTG | 230 ± 39 | 3.93 ± 0.44 | 363 ± 94 | 260 ± 111 | |
| R-M1 | 156 ± 30 | 4 ± 0.09 | 276 ± 1 | 191 ± 17 | |
| R-TIFG | 228 ± 76 | 6.81 ± 0.36 | 444 ± 37 | 351 ± 62 | |
| R-aTTG | 312 ± 55 | 3.74 ± 0.21 | 497 ± 44 | 308 ± 51 | |
| R-pSTG | 135 ± 34 | 4.3 ± 0.18 | 324 ± 41 | 219 ± 9 |
| Band | ROI | Duration (ms) | Peak Frequency (Hz) | Peak Time (ms) | Onset Time (ms) |
|---|---|---|---|---|---|
| alpha | L-MTG | 241 ± 67 | 8.56 ± 0.32 | 674 ± 84 | 627 ± 98 |
| L-OPIFG | 351 ± 58 | 11.79 ± 0.44 | 520 ± 37 | 332 ± 47 | |
| L-TIFG | 92 ± 23 | 12.38 ± 0.07 | 626 ± 2 | 580 ± 7 | |
| L-aSTG | 413 ± 34 | 14.73 ± 0.28 | 444 ± 27 | 244 ± 13 | |
| L-aTTG | 19 ± 5 | 9.00 ± 1.15 | 926 ± 8 | 925 ± 8 | |
| R-M1 | 262 ± 25 | 11.55 ± 0.3 | 536 ± 14 | 423 ± 15 | |
| R-TIFG | 328 ± 53 | 10.60 ± 0.49 | 318 ± 42 | 221 ± 30 | |
| R-aTTG | 463 ± 53 | 12.93 ± 0.71 | 363 ± 29 | 171 ± 29 | |
| R-pSTG | 154 ± 37 | 10.51 ± 0.15 | 535 ± 31 | 461 ± 42 |
| Band | ROI | Duration (ms) | Peak Frequency (Hz) | Peak Time (ms) | Onset Time (ms) |
|---|---|---|---|---|---|
| beta | L-ITG | 94 ± 52 | 25.94 ± 0.06 | 330 ± 44 | 293 ± 39 |
| L-M1 | 289 ± 45 | 24.10 ± 1.04 | 433 ± 39 | 289 ± 38 | |
| L-OIFG | 310 ± 11 | 26.75 ± 0.01 | 242 ± 3 | 241 ± 5 | |
| L-OPIFG | 84 ± 24 | 24.75 ± 1.46 | 707 ± 85 | 638 ± 91 | |
| L-TIFG | 249 ± 30 | 23.08 ± 0.93 | 436 ± 32 | 336 ± 33 | |
| L-aSTG | 371 ± 122 | 23.68 ± 2.21 | 499 ± 118 | 353 ± 144 | |
| L-aTTG | 128 ± 26 | 19.08 ± 1.17 | 563 ± 50 | 482 ± 51 | |
| L-lSTG | 57 ± 5 | 22.25 ± 0.01 | 620 ± 9 | 605 ± 10 | |
| L-pSTG | 241 ± 74 | 22.54 ± 0.08 | 275 ± 84 | 158 ± 96 | |
| R-M1 | 154 ± 36 | 22.96 ± 0.68 | 472 ± 67 | 415 ± 68 | |
| R-MTG | 143 ± 28 | 21.23 ± 1.43 | 422 ± 27 | 372 ± 25 | |
| R-OIFG | 273 ± 61 | 23.53 ± 0.63 | 512 ± 65 | 406 ± 82 | |
| R-OPIFG | 378 ± 35 | 23.20 ± 0.09 | 352 ± 54 | 119 ± 22 | |
| R-Orbital | 314 ± 67 | 24.53 ± 0.87 | 422 ± 45 | 272 ± 49 | |
| R-TIFG | 314 ± 47 | 23.22 ± 0.41 | 489 ± 45 | 352 ± 57 | |
| R-aSTG | 374 ± 59 | 24.35 ± 0.52 | 478 ± 37 | 295 ± 55 | |
| R-aTTG | 257 ± 39 | 22.44 ± 0.98 | 283 ± 35 | 206 ± 32 | |
| R-lSTG | 445 ± 50 | 21.66 ± 0.42 | 474 ± 33 | 240 ± 35 | |
| R-pSTG | 254 ± 29 | 23.72 ± 0.70 | 374 ± 23 | 264 ± 22 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Liang, X.; Wu, H.; Ma, Y.; Liu, C.; Ning, X. Parameterization of the Differences in Neural Oscillations Recorded by Wearable Magnetoencephalography for Chinese Semantic Cognition. Biology 2025, 14, 91. https://doi.org/10.3390/biology14010091
Liang X, Wu H, Ma Y, Liu C, Ning X. Parameterization of the Differences in Neural Oscillations Recorded by Wearable Magnetoencephalography for Chinese Semantic Cognition. Biology. 2025; 14(1):91. https://doi.org/10.3390/biology14010091
Chicago/Turabian StyleLiang, Xiaoyu, Huanqi Wu, Yuyu Ma, Changzeng Liu, and Xiaolin Ning. 2025. "Parameterization of the Differences in Neural Oscillations Recorded by Wearable Magnetoencephalography for Chinese Semantic Cognition" Biology 14, no. 1: 91. https://doi.org/10.3390/biology14010091
APA StyleLiang, X., Wu, H., Ma, Y., Liu, C., & Ning, X. (2025). Parameterization of the Differences in Neural Oscillations Recorded by Wearable Magnetoencephalography for Chinese Semantic Cognition. Biology, 14(1), 91. https://doi.org/10.3390/biology14010091





