Study on the Technology of Laparoscopic Ovum Pick-Up and In Vitro Embryo Production in Chongming Goats
Abstract
Simple Summary
Abstract
1. Introduction
2. Material and Methods
2.1. Ethics Statement
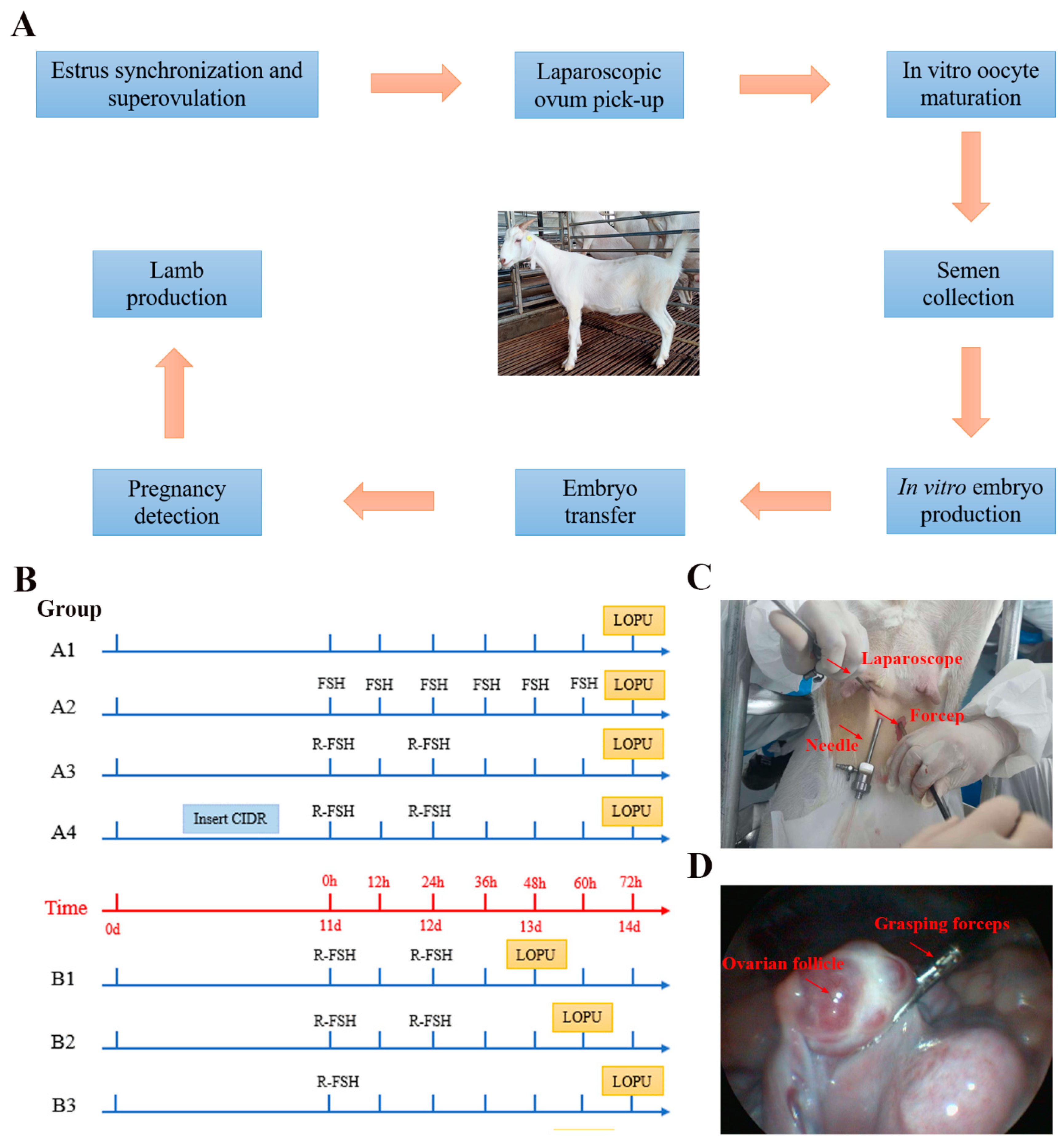
2.2. Experimental Design
2.2.1. Effect of Different Superovulation Regimens on LOPU Efficiency
2.2.2. Effect of Intravaginal Controlled Internal Drug Release on LOPU Efficiency
2.2.3. Effect of Follicular Development Period on the Efficiency of LOPU
2.2.4. Effect of Hormone Treatment Mode on LOPU Efficiency
2.3. Laparoscopic Ovum Pick-Up
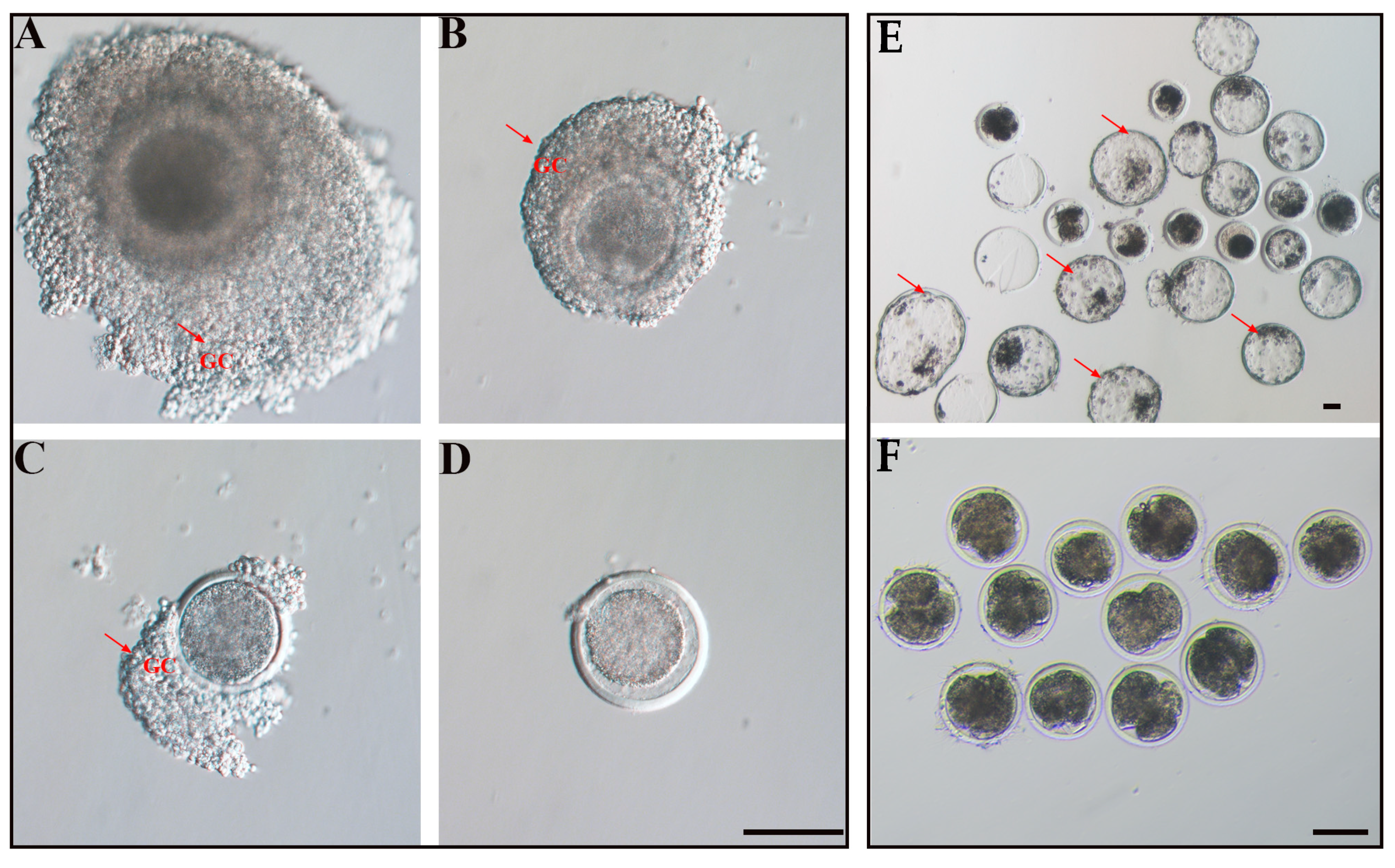
2.4. Grading of Oocyte Quality
2.5. In Vitro Embryo Production (IVEP)
2.6. Embryo Transfer
2.7. Statistical Analysis
3. Results
3.1. Effect of Different Superovulation Regimens on LOPU Efficiency
3.2. Effect of Intravaginal Controlled Internal Drug Release on LOPU Efficiency
3.3. Effect of Follicular Development Period on the Efficiency of LOPU
3.4. Effect of Hormone Treatment Mode on LOPU Efficiency
3.5. IVEP and Embryo Transfer in Goats
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Alkan, K.K.; Alkan, H.; Kaymaz, M.; Izgur, I.H. Multiple ovulation and embryo transfer during the breeding season in Angora goats: A comparison of fresh and vitrified-thawed embryo transfer. Vet. Res. Forum Int. Q. J. 2021, 12, 143–148. [Google Scholar] [CrossRef]
- Gonźalez-Bulnes, A.; Baird, D.T.; Campbell, B.K.; Cocero, M.J.; García-García, R.M.; Inskeep, E.K.; López-Sebastián, A.; McNeilly, A.S.; Santiago-Moreno, J.; Souza, C.J.; et al. Multiple factors affecting the efficiency of multiple ovulation and embryo transfer in sheep and goats. Reprod. Fertil. Dev. 2004, 16, 421–435. [Google Scholar] [CrossRef] [PubMed]
- Amiridis, G.S.; Cseh, S. Assisted reproductive technologies in the reproductive management of small ruminants. Anim. Reprod. Sci. 2012, 130, 152–161. [Google Scholar] [CrossRef]
- Baldassarre, H.J.A. Laparoscopic Ovum Pick-Up Followed by In Vitro Embryo Production and Transfer in Assisted Breeding Programs for Ruminants. Animals 2021, 11, 216. [Google Scholar] [CrossRef] [PubMed]
- Fonseca, J.F.; Oliveira, M.E.F.; Brandão, F.Z.; Batista, R.; Garcia, A.R.; Bartlewski, P.M.; Souza-Fabjan, J.M.G. Non-surgical embryo transfer in goats and sheep: The Brazilian experience. Reprod. Fertil. Dev. 2018, 31, 17–26. [Google Scholar] [CrossRef]
- Souza-Fabjan, J.M.G.; Leal, G.R.; Monteiro, C.A.S.; Batista, R.; Barbosa, N.O.; Freitas, V.J.F. In vitro embryo production in small ruminants: What is still missing? Anim. Reprod. 2023, 20, e20230055. [Google Scholar] [CrossRef]
- Paramio, M.T.; Izquierdo, D. Current status of in vitro embryo production in sheep and goats. Reprod. Domest. Anim. Zuchthygiene 2014, 49 (Suppl. S4), 37–48. [Google Scholar] [CrossRef]
- Fry, R.C. Gonadotropin priming before OPU: What are the benefits in cows and calves? Theriogenology 2020, 150, 236–240. [Google Scholar] [CrossRef]
- De Roo, C.; Tilleman, K. In Vitro Maturation of Oocytes Retrieved from Ovarian Tissue: Outcomes from Current Approaches and Future Perspectives. J. Clin. Med. 2021, 10, 4680. [Google Scholar] [CrossRef]
- Vogelsang, M.M.; Kreider, J.L.; Bowen, M.J.; Potter, G.D.; Forrest, D.W.; Kraemer, D.C. Methods for collecting follicular oocytes from mares. Theriogenology 1988, 29, 1007–1018. [Google Scholar] [CrossRef]
- Bousquet, D.; Twagiramungu, H.; Morin, N.; Brisson, C.; Carboneau, G.; Durocher, J. In vitro embryo production in the cow: An effective alternative to the conventional embryo production approach. Theriogenology 1999, 51, 59–70. [Google Scholar] [CrossRef] [PubMed]
- Renou, P.; Trounson, A.O.; Wood, C.; Leeton, J.F. The collection of human oocytes for in vitro fertilization. I. An instrument for maximizing oocyte recovery rate. Fertil. Steril. 1981, 35, 409–412. [Google Scholar] [CrossRef]
- Neto, P.N.J.; Requena, L.A.; Pizzutto, C.S.; Baldassarre, H.J.S. Laparoscopic Ovum Pick-Up (LOPU): From animal production to conservation. SPERMOVA 2018, 8, 61–67. [Google Scholar] [CrossRef]
- Souza-Fabjan, J.M.G.; Batista, R.; Freitas, V.J.F.; Mermillod, P. In Vitro Culture of Embryos from LOPU-Derived Goat Oocytes. Methods Mol. Biol. 2019, 2006, 141–153. [Google Scholar] [CrossRef] [PubMed]
- Baldassarre, H.; Currin, L.; Michalovic, L.; Bellefleur, A.M.; Gutierrez, K.; Mondadori, R.G.; Glanzner, W.G.; Schuermann, Y.; Bohrer, R.C.; Dicks, N.; et al. Interval of gonadotropin administration for in vitro embryo production from oocytes collected from Holstein calves between 2 and 6 months of age by repeated laparoscopy. Theriogenology 2018, 116, 64–70. [Google Scholar] [CrossRef]
- Baldassarre, H.; Karatzas, C.N. Advanced assisted reproduction technologies (ART) in goats. Anim. Reprod. Sci. 2004, 82, 255–266. [Google Scholar] [CrossRef]
- Baldassarre, H.; Keefer, C.; Wang, B.; Lazaris, A.; Karatzas, C.N.J.C.S.C. Nuclear transfer in goats using in vitro matured oocytes recovered by laparoscopic ovum pick-up. Cloning Stem Cells 2003, 5, 279–285. [Google Scholar] [CrossRef]
- Baldassarre, H.; Wang, B.; Kafidi, N.; Gauthier, M.; Neveu, N.; Lapointe, J.; Sneek, L.; Leduc, M.; Duguay, F.; Zhou, J.F.; et al. Production of transgenic goats by pronuclear microinjection of in vitro produced zygotes derived from oocytes recovered by laparoscopy. Theriogenology 2003, 59, 831–839. [Google Scholar] [CrossRef]
- Menchaca, A.; Anegon, I.; Whitelaw, C.B.; Baldassarre, H.; Crispo, M. New insights and current tools for genetically engineered (GE) sheep and goats. Theriogenology 2016, 86, 160–169. [Google Scholar] [CrossRef][Green Version]
- Wieczorek, J.; Koseniuk, J.; Skrzyszowska, M.; Cegła, M. L-OPU in Goat and Sheep-Different Variants of the Oocyte Recovery Method. Animals 2020, 10, 658. [Google Scholar] [CrossRef]
- Locatelli, Y.; Vallet, J.C.; Huyghe, F.P.; Cognié, Y.; Legendre, X.; Mermillod, P. Laparoscopic ovum pick-up and in vitro production of sika deer embryos: Effect of season and culture conditions. Theriogenology 2006, 66, 1334–1342. [Google Scholar] [CrossRef] [PubMed]
- Baldassarre, H.; Bordignon, V.J.A.R. Laparoscopic ovum pick-up for in vitro embryo production from dairy bovine and buffalo calves. Anim. Reprod. 2018, 15, 181. [Google Scholar] [CrossRef]
- Currin, L.; Michalovic, L.; Bellefleur, A.M.; Gutierrez, K.; Glanzner, W.; Schuermann, Y.; Bohrer, R.C.; Dicks, N.; Da Rosa, P.R.; De Cesaro, M.P.J.T. The effect of age and length of gonadotropin stimulation on the invitro embryo development of Holstein calf oocytes. Theriogenology 2017, 104, 87–93. [Google Scholar] [CrossRef] [PubMed]
- Cox, J.F.; Alfaro, V. In vitro fertilization and development of OPU derived goat and sheep oocytes. Reprod. Domest. Anim. Zuchthygiene 2007, 42, 83–87. [Google Scholar] [CrossRef] [PubMed]
- Zhang, T.; Fan, X.; Li, R.; Zhang, C.; Zhang, J. Effects of pre-incubation with C-type natriuretic peptide on nuclear maturation, mitochondrial behavior, and developmental competence of sheep oocytes. Biochem. Biophys. Res. Commun. 2018, 497, 200–206. [Google Scholar] [CrossRef]
- King, C.; Osborn, D.; Grupen, C.G. Multiple ovulation and embryo transfer in sheep: Effects of embryo developmental stage and quality on viability in vivo under farm conditions. Aust. Vet. J. 2022, 100, 451–458. [Google Scholar] [CrossRef]
- Baldassarre, H.; Wang, B.; Kafidi, N.; Keefer, C.; Lazaris, A.; Karatzas, C.N. Advances in the production and propagation of transgenic goats using laparoscopic ovum pick-up and in vitro embryo production technologies. Theriogenology 2002, 57, 275–284. [Google Scholar] [CrossRef]
- Rodríguez, C.; Anel, L.; Alvarez, M.; Anel, E.; Boixo, J.C.; Chamorro, C.A.; de Paz, P. Ovum pick-up in sheep: A comparison between different aspiration devices for optimal oocyte retrieval. Reprod. Domest. Anim. Zuchthygiene 2006, 41, 106–113. [Google Scholar] [CrossRef]
- Morton, K.M.; Maxwell, W.M.; Evans, G. Effect of aspiration pressure during oocyte harvesting on oocyte recovery and in vitro development of ovine oocytes. Reprod. Domest. Anim. Zuchthygiene 2008, 43, 106–110. [Google Scholar] [CrossRef]
- Baldassarre, H.; Furnus, C.C.; De Matos, D.G.; Pessi, H. In vitro production of sheep embryos using laparoscopic folliculocentesis: Alternative gonadotrophin treatments for stimulation of oocyte donors. Theriogenology 1996, 45, 707–717. [Google Scholar] [CrossRef]
- Cognié, Y.; Poulin, N.; Locatelli, Y.; Mermillod, P. State-of-the-art production, conservation and transfer of in-vitro-produced embryos in small ruminants. Reprod. Fertil. Dev. 2004, 16, 437–445. [Google Scholar] [CrossRef] [PubMed]
- de Castro, T.; Rubianes, E.; Menchaca, A.; Rivero, A. Ovarian dynamics, serum estradiol and progesterone concentrations during the interovulatory interval in goats. Theriogenology 1999, 52, 399–411. [Google Scholar] [CrossRef]
- Rivera, F.A.; Mendonça, L.G.; Lopes, G., Jr.; Santos, J.E.; Perez, R.V.; Amstalden, M.; Correa-Calderón, A.; Chebel, R.C. Reduced progesterone concentration during growth of the first follicular wave affects embryo quality but has no effect on embryo survival post transfer in lactating dairy cows. Reproduction 2011, 141, 333–342. [Google Scholar] [CrossRef]
- Nasser, L.F.; MF, S.F.; Reis, E.L.; Rezende, C.R.; Mapletoft, R.J.; Bó, G.A.; Baruselli, P.S. Exogenous progesterone enhances ova and embryo quality following superstimulation of the first follicular wave in Nelore (Bos indicus) donors. Theriogenology 2011, 76, 320–327. [Google Scholar] [CrossRef] [PubMed]
- Menchaca, A.; Cuadro, F.; Dos Santos-Neto, P.C.; Bosolasco, D.; Barrera, N.; de Brun, V.; Crispo, M. Oocyte developmental competence is improved by relatively greater circulating progesterone concentrations during preovulatory follicular growth. Anim. Reprod. Sci. 2018, 195, 321–328. [Google Scholar] [CrossRef] [PubMed]
- Baldassarre, H.; Matos, D.G.D.; Furnus, C.C.; Castro, T.E.; Fischer, E.I.C.J.A.R.S. Technique for efficient recovery of sheep oocytes by laparoscopic folliculocentesis. Anim. Reprod. Sci. 1994, 35, 145–150. [Google Scholar] [CrossRef]
- Thiangthientham, P.; Anakkul, N.; Suwimonteerabutr, J.; Kallayanathum, W.; Santiviparat, S.; Techakumphu, M.; Tharasanit, T. Ovarian stimulation with a low dose of follicle stimulating hormone improved recovery rates and developmental competence of goat oocytes. Thai J. Vet. Med. 2022, 52, 507–513. [Google Scholar] [CrossRef]
- Sanchez, D.J.D.; Melo, C.H.S.; Souza-Fabjan, J.M.G.; Sousa, F.C.; Rocha, A.A.; Campelo, I.S.; Teixeira, D.I.A.; Pereira, A.F.; Melo, L.M.; Freitas, V.J. Repeated hormonal treatment and laparoscopic ovum pick-up followed by in vitro embryo production in goats raised in the tropics. Livest. Sci. 2014, 165, 217–222. [Google Scholar] [CrossRef]
- Leoni, G.G.; Succu, S.; Satta, V.; Paolo, M.; Bogliolo, L.; Bebbere, D.; Spezzigu, A.; Madeddu, M.; Berlinguer, F.; Ledda, S.; et al. In vitro production and cryotolerance of prepubertal and adult goat blastocysts obtained from oocytes collected by laparoscopic oocyte-pick-up (LOPU) after FSH treatment. Reprod. Fertil. Dev. 2009, 21, 901–908. [Google Scholar] [CrossRef]
- Han, Z.B.; Lan, G.C.; Wu, Y.G.; Han, D.; Feng, W.G.; Wang, J.Z.; Tan, J.H. Interactive effects of granulosa cell apoptosis, follicle size, cumulus-oocyte complex morphology, and cumulus expansion on the developmental competence of goat oocytes: A study using the well-in-drop culture system. Reproduction 2006, 132, 749–758. [Google Scholar] [CrossRef][Green Version]
- Machatkova, M.; Krausova, K.; Jokesova, E.; Tomanek, M. Developmental competence of bovine oocytes: Effects of follicle size and the phase of follicular wave on in vitro embryo production. Theriogenology 2004, 61, 329–335. [Google Scholar] [CrossRef] [PubMed]
- Avelar, S.R.G.; Moura, R.R.; Sousa, F.C.D.; Pereira, A.F.; Almeida, K.C.; Melo, C.H.S.; Teles-Filho, A.C.A.; Baril, G.; Melo, L.M.; Teixeira, D.I.A.; et al. Oocyte production and in vitro maturation in Canindé goats following hormonal ovarian stimulation. Anim. Reprod. 2012, 9, 2012. [Google Scholar]
- Abdullah, R.B.; Liow, S.L.; Rahman, A.N.M.A.; Chan, W.K.; Wan-Khadijah, W.E.; Ng, S.C.J.T. Prolonging the interval from ovarian hyperstimulation to laparoscopic ovum pick-up improves oocyte yield, quality, and developmental competence in goats. Theriogenology 2008, 70, 765–771. [Google Scholar] [CrossRef] [PubMed]
- Shin, S.T.; Jang, S.K.; Yang, H.S.; Lee, O.K.; Shim, Y.H.; Choi, W.I.; Lee, D.S.; Lee, G.S.; Cho, J.K.; Lee, Y.W. Laparoscopy vs. laparotomy for embryo transfer to produce transgenic goats (Capra hircus). J. Vet. Sci. 2008, 9, 103–107. [Google Scholar] [CrossRef]
| Group | No. of Ovarian Follicles | No. of COCs | No. of Available COCs | COs Recovery Rate (%) | Average No. of Ovarian Follicles | Average No. of COCs | Average No. of Available COCs |
|---|---|---|---|---|---|---|---|
| A1 | 113 | 104 | 76 | 96.00 ± 5.65 | 8.69 ± 0.77 a | 8.00 ± 2.16 a | 5.85 ± 0.41 a |
| A2 | 71 | 73 | 69 | 102.50 ± 5.31 | 17.75 ± 0.62 c | 18.25 ± 2.99 c | 17.25 ± 1.03 c |
| A3 | 297 | 333 | 311 | 112.77 ± 6.52 | 22.85 ± 0.66 b | 25.62 ± 4.99 b | 23.92 ± 1.50 b |
| Group | No. of Ovarian Follicles | No. of COCs | No.of Available COCs | COC Recovery Rate (%) | Average No. of Ovarian Follicles | Average No. of COCs | Average No. of Availabile COCs |
|---|---|---|---|---|---|---|---|
| A3 | 146 | 160 | 131 | 107.81 ± 10.57 | 20.86 ± 1.74 | 22.86 ± 3.18 | 20.00 ± 3.05 |
| A4 | 136 | 138 | 129 | 103.57 ± 7.20 | 22.67 ± 1.69 | 23.00 ± 1.34 | 22.17 ± 2.30 |
| Group | No. of Ovarian Follicles | No. of COCs | No. of Available COCs | COC Recovery Rate (%) | Average No. of Ovarian Follicles | Average No. of COCs | Average No. of Available COCs |
|---|---|---|---|---|---|---|---|
| A3 | 297 | 287 | 267 | 96.87 ± 1.96 | 22.85 ± 0.52 | 22.08 ± 0.50 | 20.54 ± 0.39 |
| B1 | 94 | 91 | 85 | 96.95 ± 4.01 | 23.50 ± 065 | 22.75 ± 0.85 | 21.25 ± 0.94 |
| B2 | 142 | 133 | 126 | 93.69 ± 1.73 | 23.67 ± 0.84 | 22.17 ± 0.87 | 21.00 ± 0.52 |
| Group | No. of Ovarian Follicles | No. of COCs | No. of Available COCs | COC Recovery Rate (%) | Average No. of Ovarian Follicles | Average No. of COCs | Average No. of Available COCs |
|---|---|---|---|---|---|---|---|
| A3 | 297 | 287 | 267 | 96.87 ± 1.97 | 22.85 ± 0.52 a | 22.08 ± 0.50 a | 20.54 ± 0.39 a |
| B3 | 80 | 73 | 70 | 91.25 ± 5.15 | 20.00 ± 0.41 b | 18.25 ± 1.11 b | 17.50 ± 1.19 b |
| No. Recipients | No. Ovarian Follicles | Available COCs | No. Cleavage | Cleavage Rate (%) | No. Blastocyst | Blastocyst Rate (%) | Average No. of Blastocysts |
|---|---|---|---|---|---|---|---|
| 16 | 361 | 359 | 304 | 84.68 | 193 | 63.48 | 12.06 |
| Receptor Type | No. Recipients | No. Pregnancies | Pregnancy Rate (%) |
|---|---|---|---|
| Conventional recipients | 22 | 14 | 63.63 |
| Special recipients | 12 | 7 | 58.33 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wu, X.; Li, D.; Chen, Y.; Wu, Y.; Abudureyimu, G.; Zhang, W.; Deng, K.; Huang, Z.; Lin, J.; Wang, L. Study on the Technology of Laparoscopic Ovum Pick-Up and In Vitro Embryo Production in Chongming Goats. Biology 2024, 13, 699. https://doi.org/10.3390/biology13090699
Wu X, Li D, Chen Y, Wu Y, Abudureyimu G, Zhang W, Deng K, Huang Z, Lin J, Wang L. Study on the Technology of Laparoscopic Ovum Pick-Up and In Vitro Embryo Production in Chongming Goats. Biology. 2024; 13(9):699. https://doi.org/10.3390/biology13090699
Chicago/Turabian StyleWu, Xiangli, Dongxu Li, Ying Chen, Yangsheng Wu, Gulimire Abudureyimu, Wei Zhang, Kelu Deng, Zhen Huang, Jiapeng Lin, and Liqin Wang. 2024. "Study on the Technology of Laparoscopic Ovum Pick-Up and In Vitro Embryo Production in Chongming Goats" Biology 13, no. 9: 699. https://doi.org/10.3390/biology13090699
APA StyleWu, X., Li, D., Chen, Y., Wu, Y., Abudureyimu, G., Zhang, W., Deng, K., Huang, Z., Lin, J., & Wang, L. (2024). Study on the Technology of Laparoscopic Ovum Pick-Up and In Vitro Embryo Production in Chongming Goats. Biology, 13(9), 699. https://doi.org/10.3390/biology13090699






