Phytochemical Profiles and Biological Activities of Plant Extracts from Aromatic Plants Cultivated in Cyprus
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Plant Material
2.2. Mineral Analysis and Nutritional Value
2.3. Phytochemical Analysis
2.3.1. Extraction and NMR Analysis
2.3.2. Preparation of Plant Extracts
2.3.3. Total Phenolic and Flavonoid Content and Antioxidant Activity
2.4. Antimicrobial Activity
2.4.1. Microorganisms and Cultivation Media
2.4.2. Microdilution Method
2.5. Cell Viability following Treatment with the Plant Extracts
2.5.1. Cell Culture
2.5.2. MTT Assay
2.6. Statistical Analysis
3. Results and Discussion
3.1. Mineral Content and Nutritional Value
3.2. NMR Analysis
3.3. Antioxidant Activity, Total Phenolics, and Flavonoid Content
3.4. Antimicrobial Activity
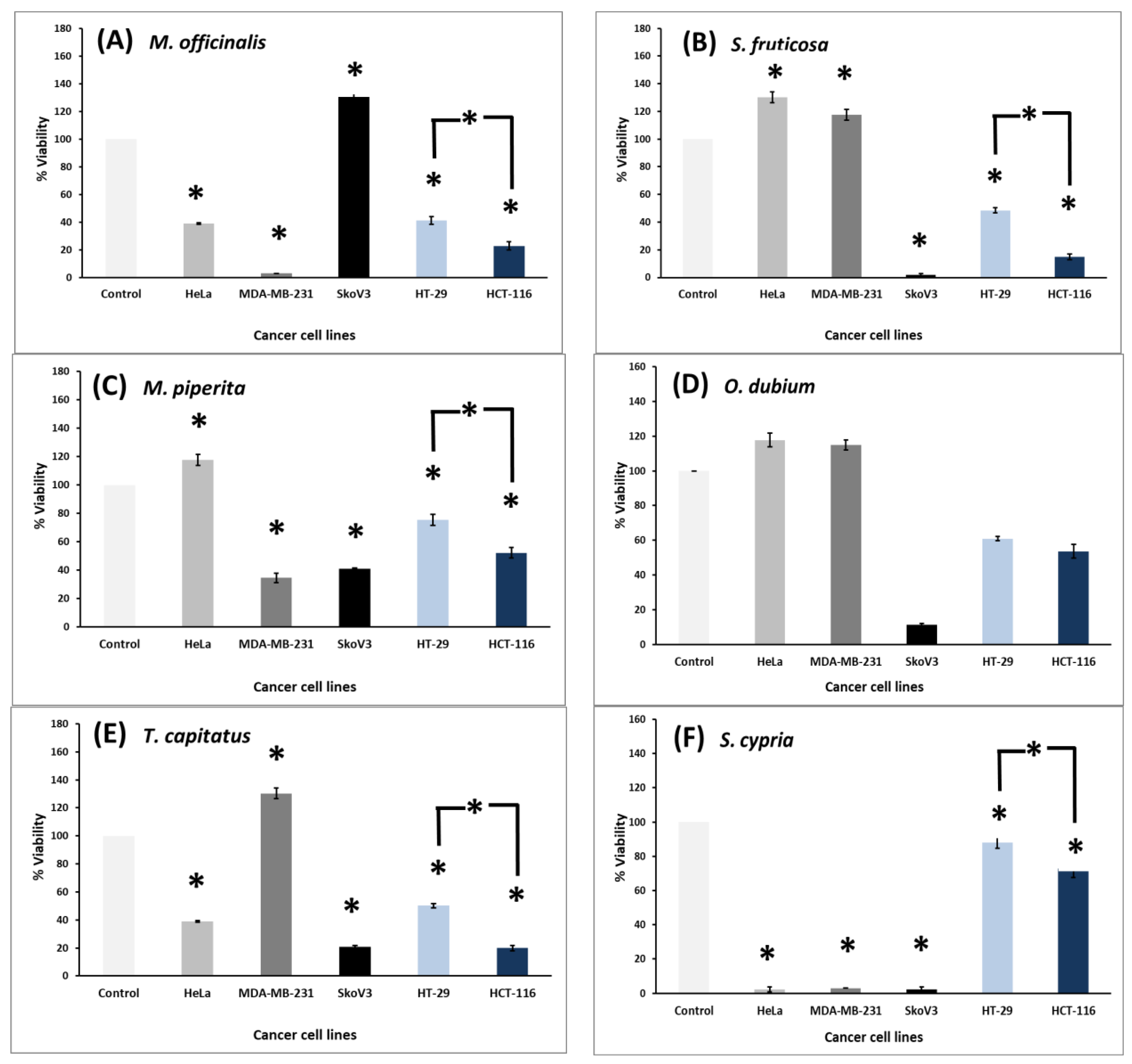
3.5. Reduced Cell Viability of Cancer Cell Lines following Treatment with the Plant Extracts
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Barata, A.M.; Rocha, F.; Lopes, V.; Carvalho, A.M. Conservation and sustainable uses of medicinal and aromatic plants genetic resources on the worldwide for human welfare. Ind. Crops Prod. 2016, 88, 8–11. [Google Scholar] [CrossRef]
- Aye, M.M.; Aung, H.T.; Sein, M.M.; Armijos, C. A review on the phytochemistry, medicinal properties and pharmacological activities of 15 selected myanmar medicinal plants. Molecules 2019, 24, 293. [Google Scholar] [CrossRef] [PubMed]
- Vujanović, M.; Zengin, G.; Đurović, S.; Mašković, P.; Cvetanović, A.; Radojković, M. Biological activity of extracts of traditional wild medicinal plants from the Balkan Peninsula. S. Afr. J. Bot. 2019, 120, 213–218. [Google Scholar] [CrossRef]
- Chrysargyris, A.; Kloukina, C.; Vassiliou, R.; Tomou, E.-M.; Skaltsa, H.; Tzortzakis, N. Cultivation strategy to improve chemical profile and anti-oxidant activity of Sideritis perfoliata L. subsp. perfoliata. Ind. Crops Prod. 2019, 140, 111694. [Google Scholar] [CrossRef]
- Capecka, E.; Mareczek, A.; Leja, M. Antioxidant activity of fresh and dry herbs of some Lamiaceae species. Food Chem. 2005, 93, 223–226. [Google Scholar]
- Ramírez Monsalve, J.; Jaramillo Gaviria, L.; Arango Correa, A. Use of freeze-dried aromatic herbs with quality organoleptic characteristics in gastronomic products. Int. J. Gastron. Food Sci. 2021, 24, 100341. [Google Scholar]
- Pavithra, T.; Geetha, P.; Selvan, P.; Karpoora Sundara Pandian, N. Proximate, Physicochemical and Functional Analysis of Herbal Frozen Dessert. Biol. Forum 2023, 15, 258–263. [Google Scholar]
- Bassolé, I.H.N.; Juliani, H.R. Essential oils in combination and their antimicrobial properties. Molecules 2012, 17, 3989–4006. [Google Scholar]
- Xylia, P.; Chrysargyris, A.; Botsaris, G.; Tzortzakis, N. Mint and pomegranate extracts/oils as antibacterial agents against Escherichia coli O157:H7 and Listeria monocytogenes on shredded carrots. J. Food Saf. 2018, 38, e12423. [Google Scholar] [CrossRef]
- Tzortzakis, N.G. Ethanol, vinegar and Origanum vulgare oil vapour suppress the development of anthracnose rot in tomato fruit. Int. J. Food Microbiol. 2010, 142, 14–18. [Google Scholar] [CrossRef]
- Petropoulos, S.A.; Fernandes, Â.; Calhelha, R.C.; Di Gioia, F.; Kolovou, P.; Barros, L.; Ferreira, I.C.F.R. Chemical composition and bioactive properties of Cichorium spinosum L. in relation to nitrate/ammonium nitrogen ratio. J. Sci. Food Agric. 2019, 99, 6741–6750. [Google Scholar] [CrossRef]
- Cocco, E.; Maccioni, D.; Sanjust, E.; Falconieri, D.; Farris, E.; Maxia, A. Ethnopharmacobotany and Diversity of Mediterranean Endemic Plants in Marmilla Subregion, Sardinia, Italy. Plants 2022, 11, 3165. [Google Scholar] [CrossRef]
- Isah, T. Stress and defense responses in plant secondary metabolites production. Biol. Res. 2019, 52, 39. [Google Scholar] [CrossRef]
- Lopez-Alvarado, J.; Farris, E. Ecology and Evolution of Plants in the Mediterranean Basin: Perspectives and Challenges. Plants 2022, 11, 1584. [Google Scholar] [CrossRef] [PubMed]
- Chrysargyris, A.; Goumenos, C.; Tzortzakis, N. Use of Medicinal and Aromatic Plant Residues for Partial Peat Substitution in Growing Media for Sonchus oleraceus Production. Agronomy 2023, 13, 1074. [Google Scholar] [CrossRef]
- Lall, N.; Chrysargyris, A.; Lambrechts, I.; Fibrich, B.; Staden, A.B.V.; Twilley, D.; Canha, M.N.; Oosthuizen, C.B.; Bodiba, D.; Tzortzakis, N.; et al. Sideritis perfoliata (subsp. perfoliata) nutritive value and its potential medicinal properties. Antioxidants 2019, 8, 521. [Google Scholar] [CrossRef] [PubMed]
- Kavoosi, G.; Rowshan, V. Chemical composition, antioxidant and antimicrobial activities of essential oil obtained from Ferula assa-foetida oleo-gum-resin: Effect of collection time. Food Chem. 2013, 138, 2180–2187. [Google Scholar] [CrossRef] [PubMed]
- Spiridon, I.; Bodirlau, R.; Teaca, C.A. Total phenolic content and antioxidant activity of plants used in traditional Romanian herbal medicine. Cent. Eur. J. Biol. 2011, 6, 388–396. [Google Scholar] [CrossRef]
- Oke, F.; Aslim, B.; Ozturk, S.; Altundag, S. Essential oil composition, antimicrobial and antioxidant activities of Satureja cuneifolia Ten. Food Chem. 2009, 112, 874–879. [Google Scholar] [CrossRef]
- Wojdyło, A.; Oszmiański, J.; Czemerys, R. Antioxidant activity and phenolic compounds in 32 selected herbs. Food Chem. 2007, 105, 940–949. [Google Scholar] [CrossRef]
- Petrović, J.; Kovalenko, V.; Svirid, A.; Stojković, D.; Ivanov, M.; Kostić, M. Individual stereoisomers of verbenol and verbenone express bioactive features. J. Mol. Struct. 2022, 1251, 131999. [Google Scholar] [CrossRef]
- Stojković, D.; Dias, M.I.; Drakulić, D.; Barros, L.; Stevanović, M.; Ferreira, I.C.F.R.; Soković, M.D. Methanolic extract of the herb Ononis spinosa L. Is an antifungal agent with no cytotoxicity to primary human cells. Pharmaceuticals 2020, 13, 78. [Google Scholar] [CrossRef]
- Christou, C.; Christodoulou, M.I.; Zaravinos, A.; Gkretsi, V. Ras suppressor 1 long form (RSU1L) silencing promotes apoptosis in invasive breast cancer cells. Cell. Signal. 2023, 101, 110522. [Google Scholar] [CrossRef]
- Afuape, A.O.; Afolayan, A.J.; Buwa-Komoreng, L.V. Proximate, Vitamins, Minerals and Anti-Nutritive Constituents of the Leaf and Stem of Helichrysum odoratissimum (L.) Sweet: A Folk Medicinal Plant in South Africa. Int. J. Plant Biol. 2022, 13, 463–472. [Google Scholar] [CrossRef]
- Kim, M.J.; Moon, Y.; Tou, J.C.; Mou, B.; Waterland, N.L. Nutritional value, bioactive compounds and health benefits of lettuce (Lactuca sativa L.). J. Food Compos. Anal. 2016, 49, 19–34. [Google Scholar] [CrossRef]
- Soetan, K.O.; Olaiya, C.O.; Oyewole, O.E. The importance of mineral elements for humans, domestic animals and plants: A review. African J. Food Sci. 2010, 4, 200–222. [Google Scholar]
- Rahmatollah, R.; Mahbobeh, R. Mineral contents of some plants used in Iran. Pharmacogn. Res. 2010, 2, 267–270. [Google Scholar] [CrossRef] [PubMed]
- Achi, N.K.; Onyeabo, C.; Ekeleme-Egedigwe, C.A.; Onyeanula, J.C. Phytochemical, proximate analysis, vitamin and mineral composition of aqueous extract of Ficus capensis leaves in South Eastern Nigeria. J. Appl. Pharm. Sci. 2017, 7, 117–122. [Google Scholar] [CrossRef]
- Radha; Kumar, M.; Puri, S.; Pundir, A.; Bangar, S.P.; Changan, S.; Choudhary, P.; Parameswari, E.; Alhariri, A.; Samota, M.K.; et al. Evaluation of Nutritional, Phytochemical, and Mineral Composition of Selected Medicinal Plants for Therapeutic Uses from Cold Desert of Western Himalaya. Plants 2021, 10, 1429. [Google Scholar]
- Kazemi, M.; Moheghi, M.M.; Tohidi, R. A study on the nutritional characteristics of some plants and their effects on ruminal microbial fermentation and protozoa population. AMB Express 2021, 11, 174. [Google Scholar] [CrossRef]
- Lytra, K.; Tomou, E.M.; Chrysargyris, A.; Christofi, M.D.; Miltiadous, P.; Tzortzakis, N.; Skaltsa, H. Bio-Guided Investigation of Sideritis cypria Methanol Extract Driven by in Vitro Antioxidant and Cytotoxic Assays. Chem. Biodivers. 2021, 18, e2000966. [Google Scholar] [CrossRef]
- Uysal, I.; Koçer, O.; Mohammed, F.S.; Lekesiz, Ö.; Doğan, M.; Şabik, A.E.; Sevindik, E.; Gerçeker, F.Ö.; Sevindik, M. Pharmacological and Nutritional Properties: Genus Salvia. Adv. Pharmacol. Pharm. 2023, 11, 140–155. [Google Scholar] [CrossRef]
- Mainasara, M.M.; Abu Bakar, M.F.; Waziri, A.H.; Musa, A.R. Comparison of Phytochemical, Proximate and Mineral Composition of Fresh and Dried Peppermint (Mentha piperita) Leaves. J. Sci. Technol. 2018, 10, 85–91. [Google Scholar] [CrossRef][Green Version]
- Khalil, E.; Esoh, R.; Rababah, T.; Almajwal, A.M.; Alu’datt, M.H. Minerals, proximate composition and their correlations of medicinal plants from Jordan. J. Med. Plants Res. 2012, 6, 5757–5762. [Google Scholar]
- Zeroual, A.; Sakar, E.H.; Eloutassi, N.; Mahjoubi, F.; Chaouch, M.; Chaqroune, A. Phytochemical profiling of essential oils isolated using hydrodistillation and microwave methods and characterization of some nutrients in Origanum compactum benth from central-northern Morocco. Biointerface Res. Appl. Chem. 2021, 11, 9358–9371. [Google Scholar] [CrossRef]
- Dimas, D.; Tomou, E.-M.; Karamani, C.; Sfiniadakis, I.; Siakavella, K.I.; Liakopoulou, A.; Hatziantoniou, S.; Rallis, M.; Skaltsa, H. Melissa officinalis ssp. altissima extracts: A therapeutic approach targeting psoriasis in mice. J. Ethnopharmacol. 2020, 246, 112208. [Google Scholar] [CrossRef]
- Shakeri, A.; Sahebkar, A.; Javadi, B. Melissa officinalis L.—A review of its traditional uses, phytochemistry and pharmacology. J. Ethnopharmacol. 2016, 188, 204–228. [Google Scholar] [CrossRef] [PubMed]
- EMA. Assessment Report on Melissa officinalis L., folium; EMA: Amsterdam, The Netherlands, 2013. [Google Scholar]
- Xu, J.; Wei, K.; Zhang, G.; Lei, L.; Yang, D.; Wang, W.; Han, Q.; Xia, Y.; Bi, Y.; Yang, M.; et al. Ethnopharmacology, phytochemistry, and pharmacology of Chinese Salvia species: A review. J. Ethnopharmacol. 2018, 225, 18–30. [Google Scholar] [CrossRef] [PubMed]
- Wu, Y.-B.; Ni, Z.-Y.; Shi, Q.-W.; Dong, M.; Kiyota, H.; Gu, Y.-C.; Cong, B. Constituents from Salvia Species and Their Biological Activities. Chem. Rev. 2012, 112, 5967–6026. [Google Scholar] [CrossRef] [PubMed]
- Lu, Y.; Foo, L. Polyphenolic in Salvia. Phytochemistry 2002, 59, 117–140. [Google Scholar] [CrossRef] [PubMed]
- Lu, Y.; Foo, L.Y. Flavonoid and phenolic glycosides from Salvia officinalis. Phytochemistry 2000, 55, 263–267. [Google Scholar] [CrossRef]
- Mahendran, G.; Rahman, L.U. Ethnomedicinal, phytochemical and pharmacological updates on Peppermint (Mentha × piperita L.)—A review. Phyther. Res. 2020, 34, 2088–2139. [Google Scholar] [CrossRef]
- McKay, D.; Blumberg, J. A Review of the Bioactivity and Potential Health Benefits of Peppermint Tea (Mentha piperita L.). Phyther. Res. 2006, 20, 619–633. [Google Scholar] [CrossRef]
- Bouyahya, A.; Chamkhi, I.; Benali, T.; Guaouguaou, F.E.; Balahbib, A.; El Omari, N.; Taha, D.; Belmehdi, O.; Ghokhan, Z.; El Menyiy, N. Traditional use, phytochemistry, toxicology, and pharmacology of Origanum majorana L. J. Ethnopharmacol. 2021, 265, 113318. [Google Scholar] [CrossRef] [PubMed]
- Sharifi-Rad, M.; Berkay Yılmaz, Y.; Antika, G.; Salehi, B.; Tumer, T.B.; Kulandaisamy Venil, C.; Das, G.; Patra, J.K.; Karazhan, N.; Akram, M.; et al. Phytochemical constituents, biological activities, and health-promoting effects of the genus Origanum. Phyther. Res. 2021, 35, 95–121. [Google Scholar] [CrossRef] [PubMed]
- Karioti, A.; Milošević-Ifantis, T.; Pachopos, N.; Niryiannaki, N.; Hadjipavlou-Litina, D.; Skaltsa, H. Antioxidant, anti-inflammatory potential and chemical constituents of Origanum dubium Boiss., growing wild in Cyprus. J. Enzyme Inhib. Med. Chem. 2015, 30, 38–43. [Google Scholar] [CrossRef] [PubMed]
- Bouyahya, A.; Chamkhi, I.; Guaouguaou, F.E.; Benali, T.; Balahbib, A.; El Omari, N.; Taha, D.; El-Shazly, M.; El Menyiy, N. Ethnomedicinal use, phytochemistry, pharmacology, and food benefits of Thymus capitatus. J. Ethnopharmacol. 2020, 259, 112925. [Google Scholar] [CrossRef] [PubMed]
- Żyżelewicz, D.; Kulbat-Warycha, K.; Oracz, J.; Żyżelewicz, K. Polyphenols and other bioactive compounds of sideritis plants and their potential biological activity. Molecules 2020, 25, 3763. [Google Scholar] [CrossRef] [PubMed]
- González-Burgos, E.; Carretero, M.E.; Gómez-Serranillos, M.P. Sideritis spp.: Uses, chemical composition and pharmacological activities—A review. J. Ethnopharmacol. 2011, 135, 209–225. [Google Scholar] [CrossRef]
- Lytra, K.; Tomou, E.; Chrysargyris, A.; Drouza, C.; Skaltsa, H.; Tzortzakis, N. Traditionally Used Sideritis cypria Post.: Phytochemistry, Nutritional Content, Bioactive Compounds of Cultivated Populations. Front. Pharmacol. 2020, 11, 650. [Google Scholar] [CrossRef]
- Hanoğlu, D.Y.; Hanoğlu, A.; Yusufoğlu, H.; Demirci, B.; Başer, K.H.C.; Çalış, İ.; Yavuz, D.Ö. Phytochemical investigation of endemic Sideritis cypria post. Rec. Nat. Prod. 2019, 14, 105–115. [Google Scholar] [CrossRef]
- Miraj, S.; Rafieian-Kopaei; Kiani, S. Melissa officinalis L: A Review Study with an Antioxidant Prospective. J. Evidence-Based Complement. Altern. Med. 2017, 22, 385–394. [Google Scholar] [CrossRef]
- Lee, H.J.; Choi, Y.J.; Choi, Y.I.; Lee, J.J. Effects of lemon balm on the oxidative stability and the quality properties of hamburger patties during refrigerated storage. Korean J. Food Sci. Anim. Resour. 2014, 34, 533–542. [Google Scholar] [CrossRef]
- Spiridon, I.; Colceru, S.; Anghel, N.; Teaca, C.A.; Bodirlau, R.; Armatu, A. Antioxidant capacity and total phenolic contents of oregano (Origanum vulgare), lavender (Lavandula angustifolia) and lemon balm (Melissa officinalis) from Romania. Nat. Prod. Res. 2011, 25, 1657–1661. [Google Scholar] [CrossRef] [PubMed]
- Uribe, E.; Marín, D.; Vega-Gálvez, A.; Quispe-Fuentes, I.; Rodríguez, A. Assessment of vacuum-dried peppermint (Mentha piperita L.) as a source of natural antioxidants. Food Chem. 2016, 190, 559–565. [Google Scholar] [CrossRef] [PubMed]
- Dorman, H.J.D.; Koşar, M.; Başer, K.H.C.; Hiltunen, R. Phenolic Profile and Antioxidant Evaluation of. Nat. Prod. Commun. 2009, 4, 535–542. [Google Scholar] [PubMed]
- Dawra, M.; Bouajila, J.; El Beyrouthy, M.; Rizk, A.A.; Taillandier, P.; Nehme, N.; El Rayess, Y. Chemical Characterization and Antioxidant, Antibacterial, Antiacetylcholinesterase and Antiproliferation Properties of Salvia fruticosa Miller Extracts. Molecules 2023, 28, 2429. [Google Scholar] [CrossRef] [PubMed]
- Benoutman, A.; Erbiai, E.H.; Edderdaki, F.Z.; Cherif, E.K.; Saidi, R.; Lamrani, Z.; Pintado, M.; Pinto, E.; Esteves da Silva, J.C.G.; Maouni, A. Phytochemical Composition, Antioxidant and Antifungal Activity of Thymus capitatus, a Medicinal Plant Collected from Northern Morocco. Antibiotics 2022, 11, 681. [Google Scholar] [CrossRef] [PubMed]
- de Oliveira, I.; Chrysargyris, A.; Heleno, S.A.; Carocho, M.; Calhelha, R.C.; Dias, M.I.; Petrović, J.; Soković, M.; Petropoulos, S.A.; Santos-Buelga, C.; et al. Effects of the extraction techniques on the chemical composition and bioactive properties of lemon balm (Melissa officinalis L.) plants grown under different cropping and irrigation regimes. Food Res. Int. 2023, 170, 113044. [Google Scholar] [CrossRef] [PubMed]
- Witkowski, M.; Grajeta, H.; Gomułka, K. Hypersensitivity Reactions to Food Additives—Preservatives, Antioxidants, Flavor Enhancers. Int. J. Environ. Res. Public Health 2022, 19, 11493. [Google Scholar] [CrossRef]
- Abdel-Naime, W.A.; Fahim, J.R.; Fouad, M.A.; Kamel, M.S. Antibacterial, antifungal, and GC–MS studies of Melissa officinalis. South African J. Bot. 2019, 124, 228–234. [Google Scholar] [CrossRef]
- Petrisor, G.; Motelica, L.; Craciun, L.N.; Oprea, O.C.; Ficai, D.; Ficai, A. Melissa officinalis: Composition, Pharmacological Effects and Derived Release Systems—A Review. Int. J. Mol. Sci. 2022, 23, 3591. [Google Scholar] [CrossRef]
- Abdulla-Eltawaty, S.I.; Abdalkader, G.M.H.; Hasan, H.M.; Eltaguri, H.M.A. Antibacterial activity and GC-MS analysis of chloroform extract of bark of the Antibacterial activity and GC-MS analysis of chloroform extract of bark of the Libyan Salvia fruticosa Mill. Int. J. Multidiscip. Sci. Adv. Technol. 2021, 715–721. [Google Scholar]
- Ecevit, K.; Barros, A.A.; Silva, J.M.; Reis, R.L. Preventing Microbial Infections with Natural Phenolic Compounds. Future Pharmacol. 2022, 2, 460–498. [Google Scholar] [CrossRef]
- Hanoğlu, D.Y.; Hanoğlu, A.; Güvenir, M.; Süer, K.; Demirci, B.; Başer, K.H.C.; Özkum Yavuz, D. Chemical composition and antimicrobial activity of the essential oil of Sideritis cypria Post endemic in Northern Cyprus. J. Essent. Oil Res. 2017, 29, 228–232. [Google Scholar] [CrossRef]
- Indrayudha, P. Antibacterial Activity of Combination of Ethanol Extract of Pepermine Leaves (Mentha piperita L.) and Amikacin Against Klebsiella pneumonia, Staphylococcus aureus, and Escherichia coli. J. Nutraceuticals Herb. Med. 2021, 4, 12–29. [Google Scholar] [CrossRef]
- Saravani, S.; Ghaffari, M.; Valizadeh, M.; Ali-Malayeri, F.; Biabangard, A. Antimicrobial Activity of Mentha piperita, Rosmarinus officinalis, and Withania somnifera Prepared by Ultrasound Against Escherichia coli Isolated from Poultry Stool. Gene Cell Tissue 2021, 9, e109104. [Google Scholar] [CrossRef]
- Siegel, R.L.; Miller, K.D.; Wagle, N.S.; Jemal, A. Cancer statistics, 2023. CA Cancer J. Clin. 2023, 73, 17–48. [Google Scholar] [CrossRef] [PubMed]
- Pape, J.; Magdeldin, T.; Ali, M.; Walsh, C.; Lythgoe, M.; Emberton, M.; Cheema, U. Cancer invasion regulates vascular complexity in a three-dimensional biomimetic model. Eur. J. Cancer 2019, 119, 179–193. [Google Scholar] [CrossRef] [PubMed]
- Tuy-On, T.; Itharat, A.; Maki, P.; Thongdeeying, P.; Pipatrattanaseree, W.; Ooraikul, B. In Vitro Cytotoxic Activity against Breast, Cervical, and Ovarian Cancer Cells and Flavonoid Content of Plant Ingredients Used in a Selected Thai Traditional Cancer Remedy: Correlation and Hierarchical Cluster Analysis. Evidence-based Complement. Altern. Med. 2020, 2020, 8884529. [Google Scholar] [CrossRef]
- Stewart, B.; Wild, C. World Cancer Report; World Health Organization: Lyon, France, 2014. [Google Scholar]
- Bitgen, N.; Baran, M.; Önder, G.Ö.; Alisa Suna, P.; Gurbuz, P.; Yay, A. Effect of Melissa officinalis L. on human breast cancer cell line via apoptosis and autophagy. Cukurova Med. J. 2022, 47, 765–775. [Google Scholar] [CrossRef]
- Somaida, A.; Tariq, I.; Ambreen, G.; Abdelsalam, A.M.M.; Ayoub, A.M.M.; Wojcik, M.; Dzoyem, J.P.P.; Bakowsky, U. Potent cytotoxicity of four cameroonian plant extracts on different cancer cell lines. Pharmaceuticals 2020, 13, 357. [Google Scholar] [CrossRef]
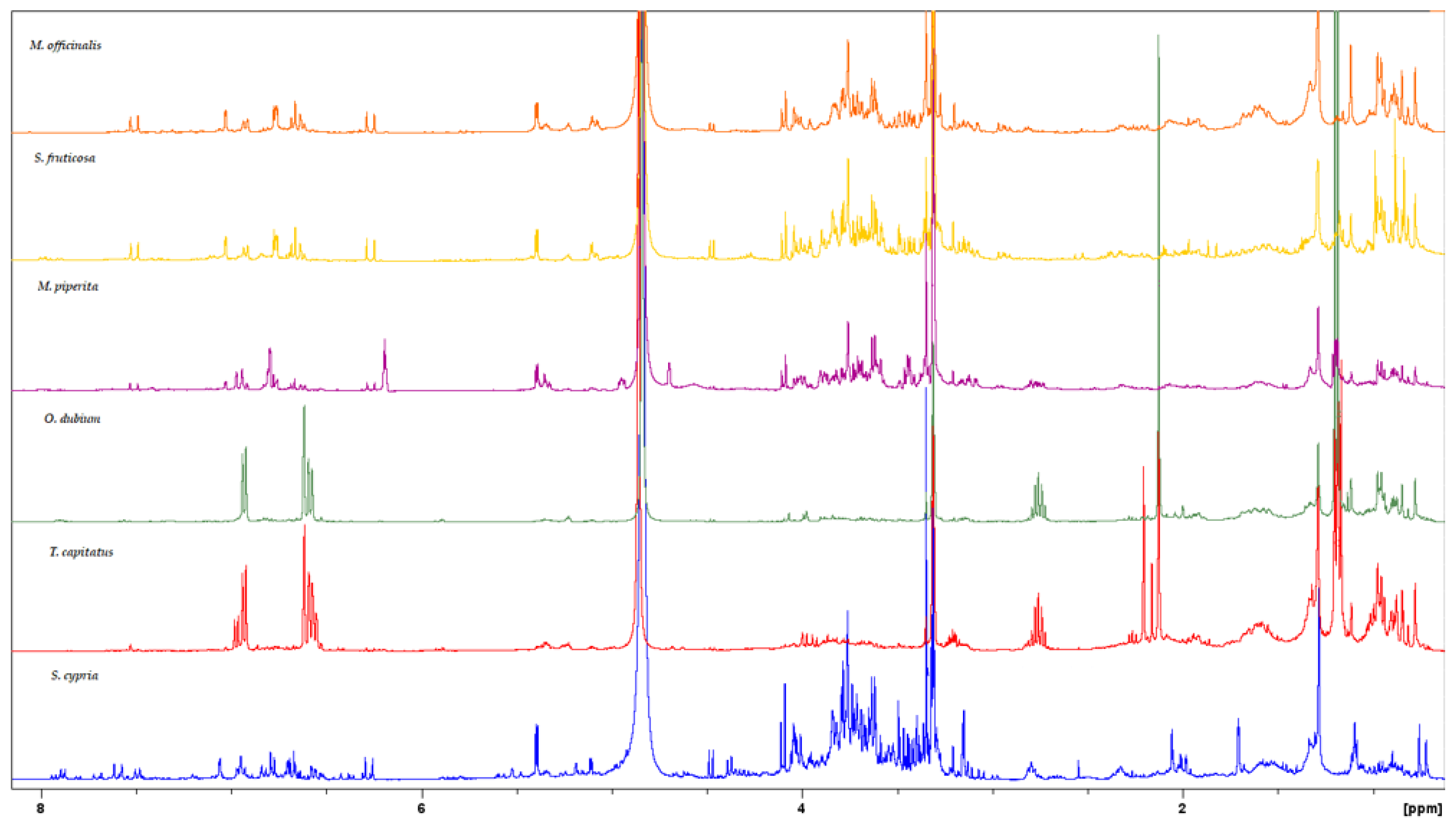
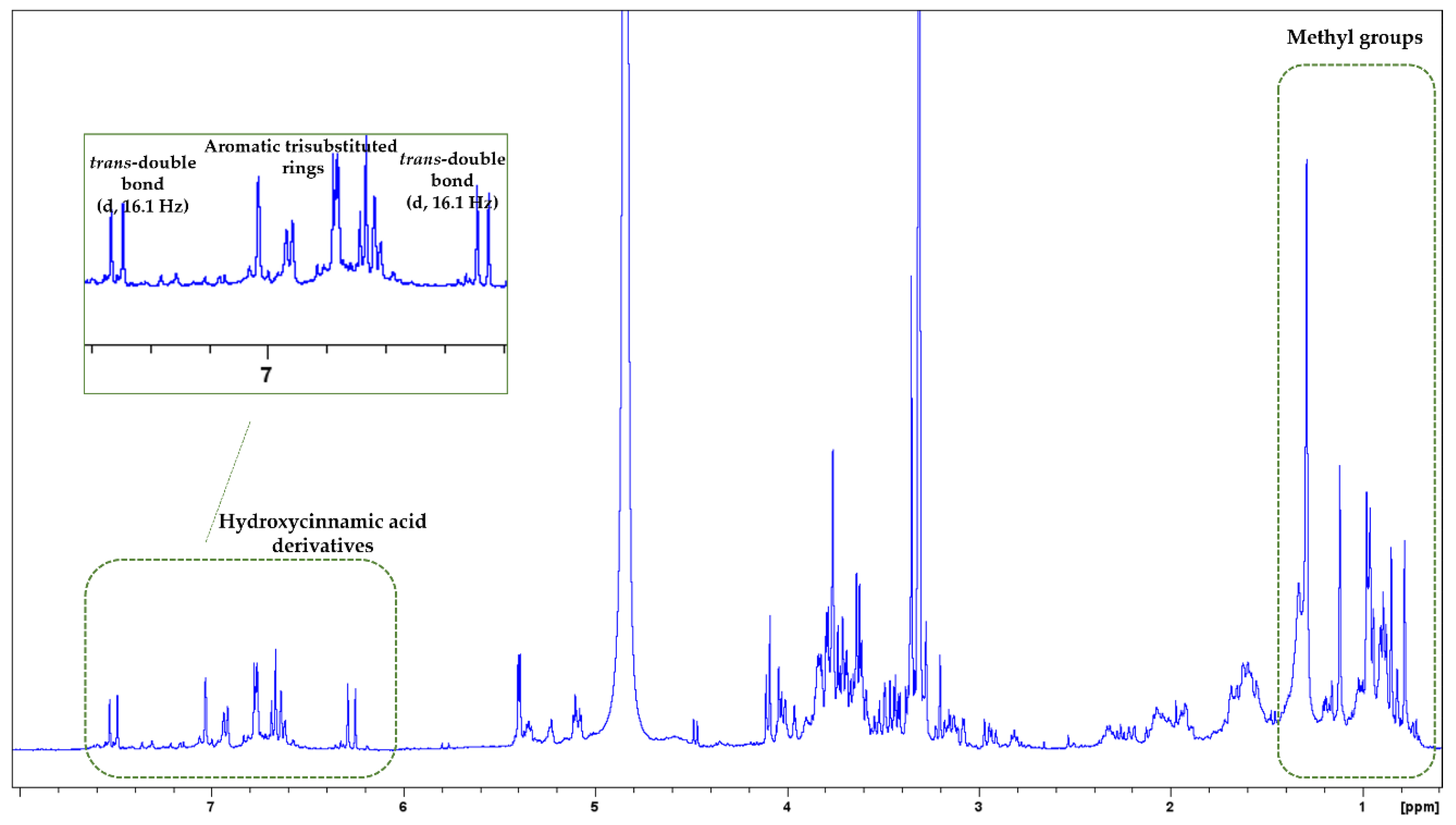
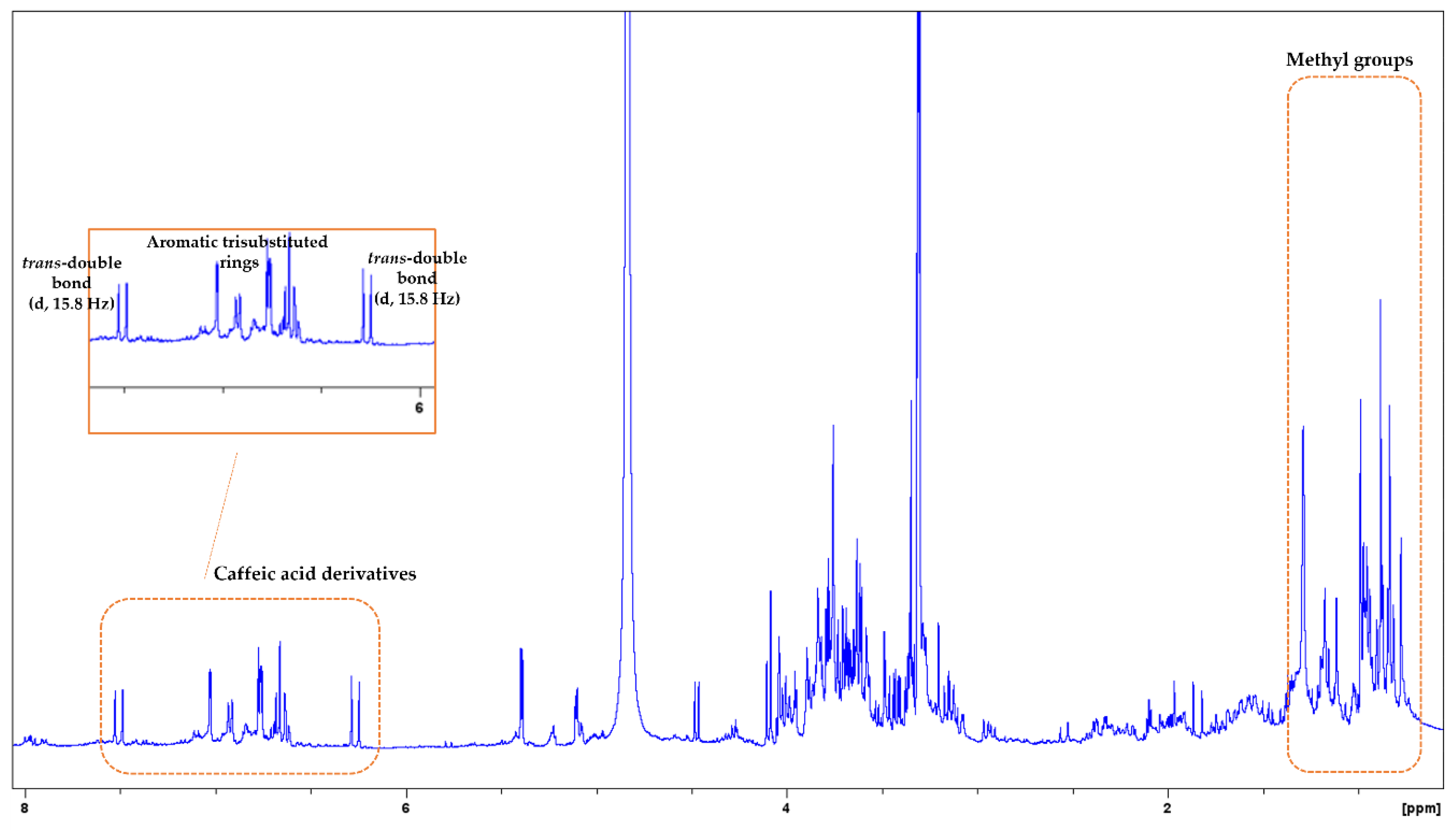
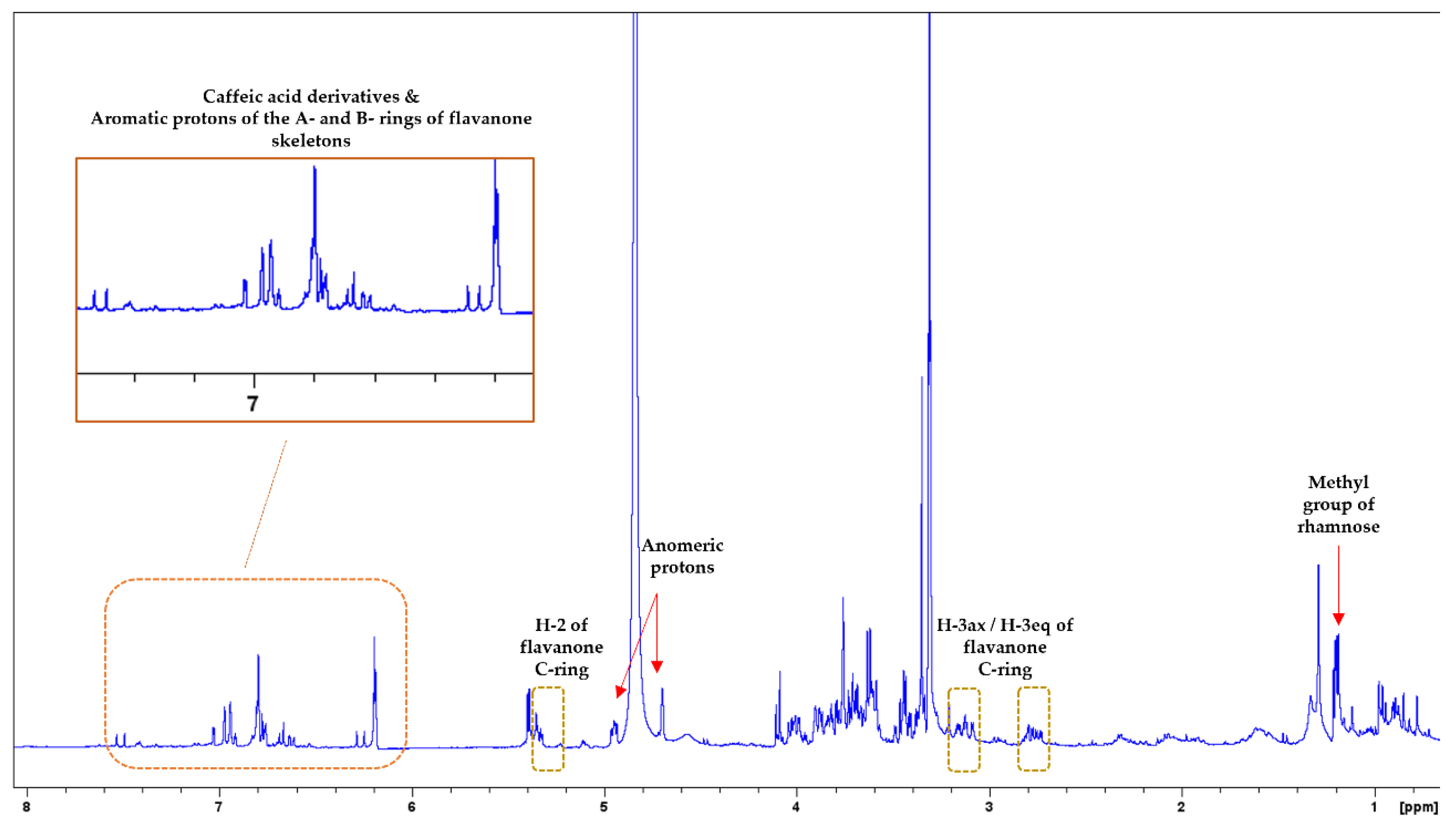
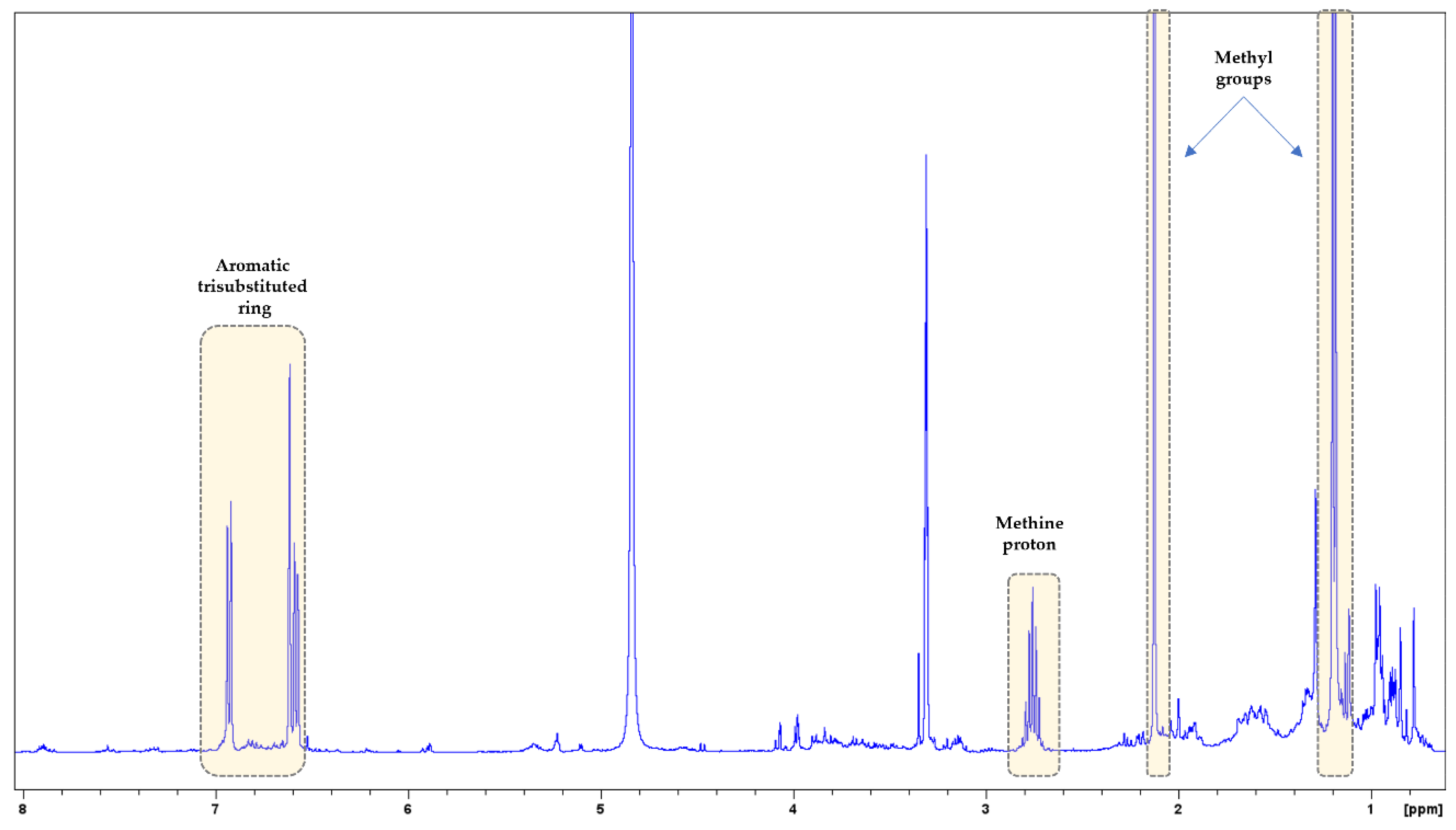
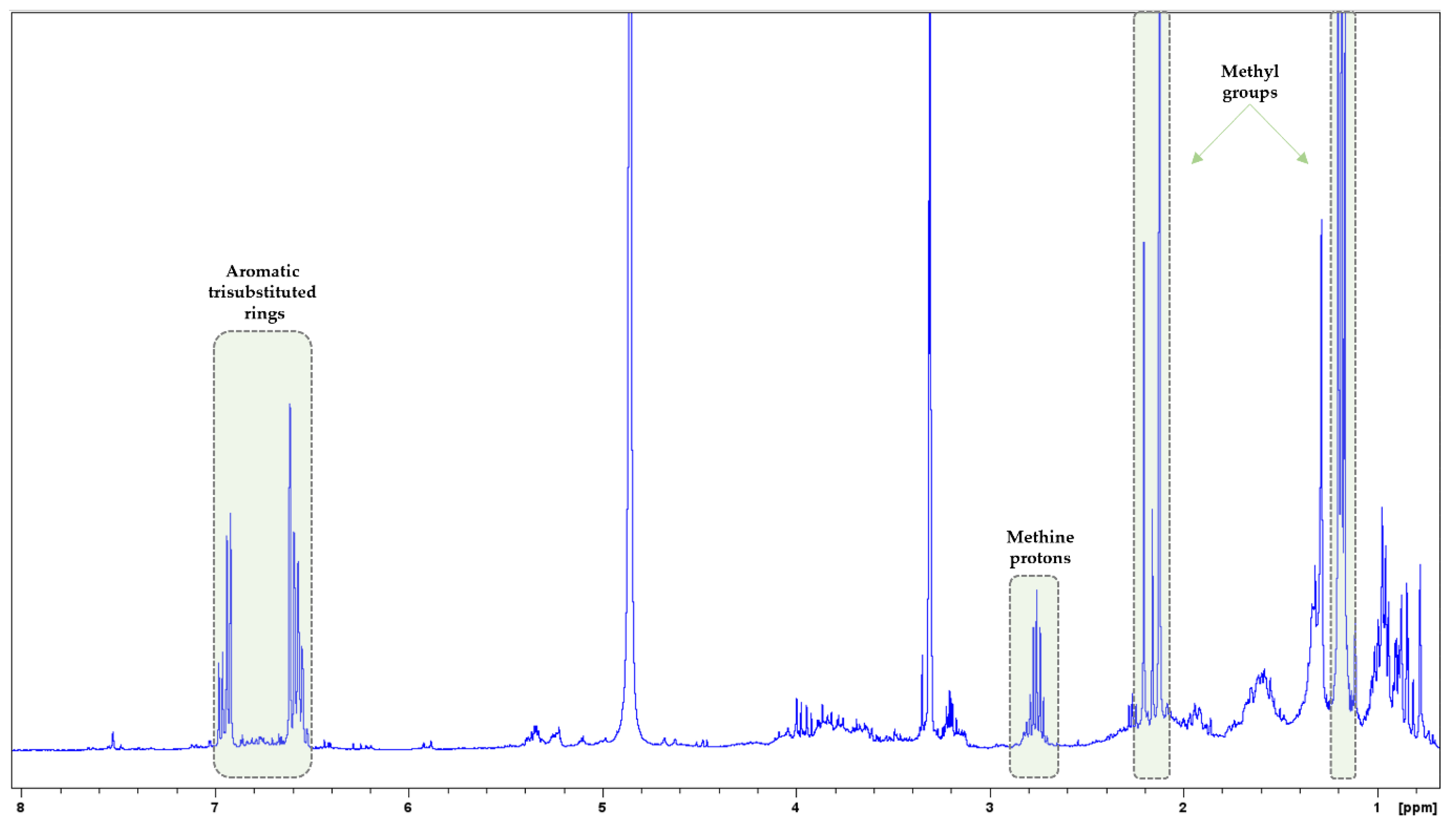
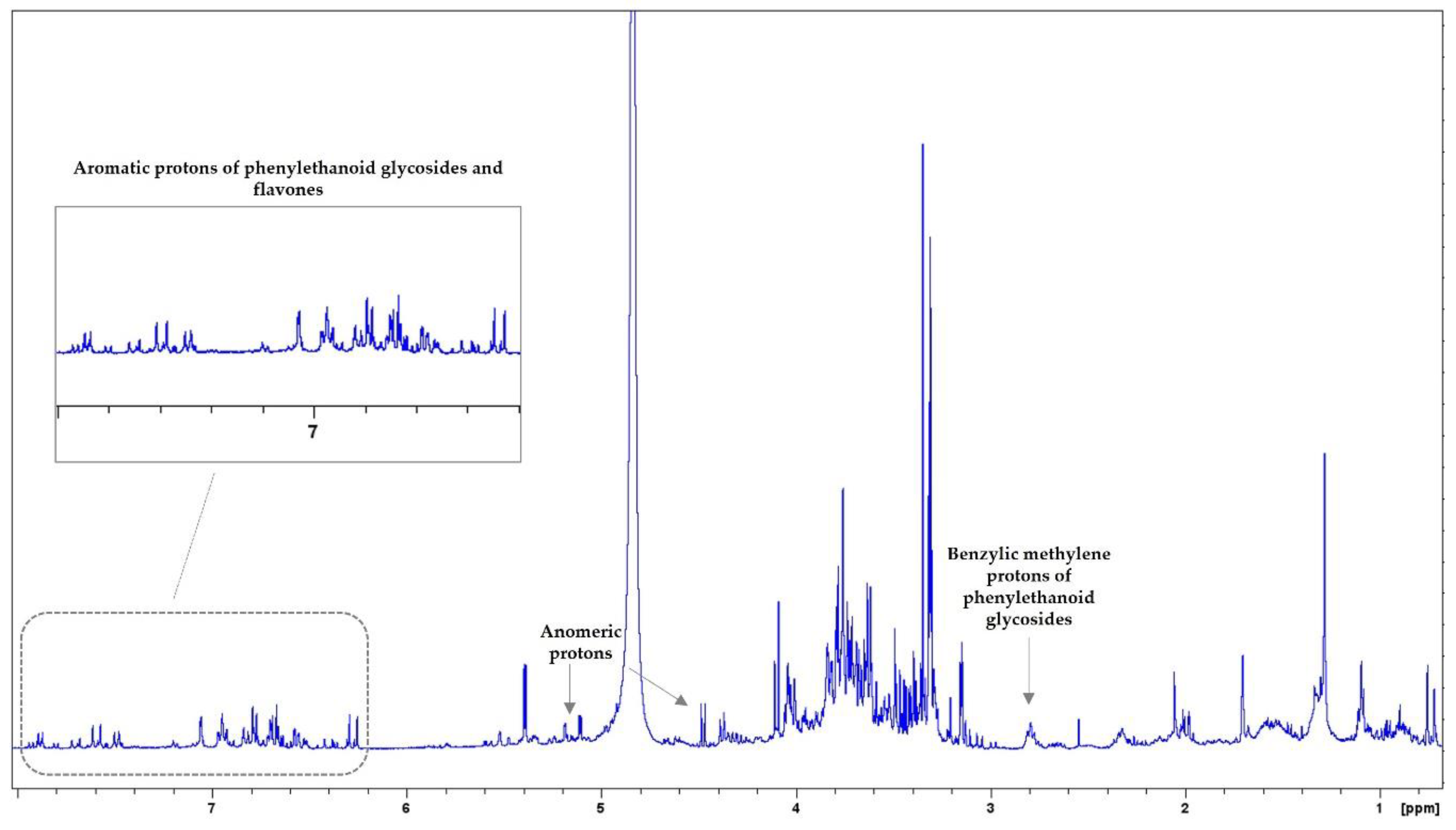
| MAP Species | Nitrogen (N) g/kg | Sodium (Na) g/kg | Potassium (K) g/kg | Magnesium (Mg) g/kg | Calcium (Ca) g/kg | Phosphorus (P) g/kg |
|---|---|---|---|---|---|---|
| Melissa officinalis | 15.42 ± 0.55 bc | 0.13 ± 0.04 d | 22.17 ± 0.15 a | 4.22 ± 0.06 b | 9.20 ± 0.13 d | 2.58 ± 0.04 bc |
| Salvia fruticosa | 16.46 ± 0.76 b | 0.59 ± 0.04 c | 15.20 ± 0.44 c | 3.12 ± 0.12 c | 9.54 ± 0.28 d | 1.57 ± 0.11 d |
| Mentha piperita | 20.55 ± 0.31 a | 0.66 ± 0.02 c | 16.77 ± 0.15 b | 5.82 ± 0.04 a | 14.20 ± 0.22 b | 2.43 ± 0.28 c |
| Origanum dubium | 11.46 ± 0.28 d | 0.13 ± 0.01 d | 12.69 ± 0.21 d | 4.31 ± 0.21 b | 10.22 ± 0.39 d | 2.99 ± 0.09 b |
| Thymus capitatus | 14.19 ± 0.13 c | 1.01 ± 0.03 b | 15.19 ± 0.28 c | 2.09 ± 0.05 d | 11.46 ± 0.22 c | 1.37 ± 0.05 d |
| Sideritis cypria | 13.67 ± 0.10 c | 1.28 ± 0.02 a | 22.07 ± 0.15 a | 2.30 ± 0.08 d | 17.49 ± 0.18 a | 3.75 ± 0.05 a |
| MAP Species | Dry Matter % | Humidity % | Ash % | Total Fats % | Proteins % | Carbohydrates % | Energy kcal/100 g |
|---|---|---|---|---|---|---|---|
| Melissa officinalis | 21.63 ± 0.36 c | 78.36 ± 0.36 a | 8.18 ± 0.36 b | 1.71 ± 0.04 d | 9.64 ± 0.69 bc | 80.45 ± 0.48 b | 375.86 ± 1.39 c |
| Salvia fruticosa | 29.36 ± 0.43 c | 70.63 ± 0.43 b | 6.97 ± 0.19 b | 5.89 ± 0.02 a | 10.28 ± 0.96 b | 76.84 ± 0.38 c | 401.54 ± 0.65 a |
| Mentha piperita | 19.14 ± 0.57 c | 80.85 ± 0.57 a | 11.28 ± 0.21 a | 2.81 ± 0.01 c | 12.84 ± 0.39 a | 73.06 ± 0.26 d | 368.94 ± 0.82 d |
| Origanum dubium | 21.12 ± 1.22 c | 78.87 ± 1.22 a | 7.02 ± 0.66 b | 4.03 ± 0.06 b | 7.16 ± 0.35 d | 81.78 ± 0.63 b | 392.07 ± 2.61 b |
| Thymus capitatus | 42.64 ± 0.79 a | 57.35 ± 0.79 c | 6.30 ± 0.56 b | 0.69 ± 0.01 e | 8.87 ± 0.16 c | 84.12 ± 0.61 a | 378.23 ± 2.27 c |
| Sideritis cypria | 28.90 ± 0.16 b | 70.76 ± 0.30 b | 11.84 ± 0.95 a | 2.92 ± 0.29 c | 8.54 ± 0.11 c | 76.68 ± 0.44 c | 367.26 ± 1.12 d |
| MAP Species | Total Phenolic Content (µmol GA/g DE) | DPPH (mg Trolox/g DE) | FRAP (mg Trolox/g DE) | ABTS (mg Trolox/g DE) | Total Flavonoid Content (mg Rutin/g DE) |
|---|---|---|---|---|---|
| Melissa officinalis | 138.37 ± 0.65 d | 195.02 ± 3.22 b | 262.65 ± 1.78 b | 188.87 ± 2.98 d | 116.71 ± 1.58 b |
| Salvia fruticosa | 91.32 ± 1.09 f | 91.90 ± 1.46 c | 182.03 ± 1.29 d | 171.48 ± 3.72 e | 59.22 ± 0.25 e |
| Mentha piperita | 196.96 ± 1.73 a | 295.18 ± 3.79 a | 389.63 ± 1.27 a | 252.41 ± 1.34 c | 207.74 ± 1.93 a |
| Origanum dubium | 162.02 ± 2.32 b | 90.12 ± 1.04 c | 206.84 ± 2.60 c | 427.86 ± 1.13 a | 85.89 ± 1.88 c |
| Thymus capitatus | 154.07 ± 0.77 c | 71.66 ± 1.43 d | 210.84 ± 1.87 c | 351.75 ± 4.64 b | 80.64 ± 0.97 c |
| Sideritis cypria | 100.74 ± 0.93 e | 79.25 ± 2.29 d | 160.02 ± 2.49 e | 155.99 ± 4.39 e | 72.52 ± 0.55 d |
| S. aureus (ATCC 11632) | B. cereus (Clinical Isolate) | L. monocytogenes (NCTC 7973) | S. enterica subsp. enterica serovar Typhimurium (ATCC 13311) | E. coli (ATCC 25922) | ||
|---|---|---|---|---|---|---|
| M. officinalis | MIC | 0.25 | 0.25 | 0.25 | 0.50 | 0.25 |
| MBC | 0.50 | 0.50 | 0.50 | 1.00 | 0.50 | |
| S. fruticosa | MIC | 0.50 | 0.25 | 0.25 | 0.25 | 0.25 |
| MBC | 1.00 | 0.50 | 0.50 | 0.50 | 0.50 | |
| M. piperita | MIC | 0.50 | 0.50 | 0.50 | 0.50 | 0.25 |
| MBC | 1.00 | 1.00 | 1.00 | 1.00 | 0.50 | |
| O. dubium | MIC | 0.25 | 0.25 | 0.125 | 0.125 | 0.125 |
| MBC | 0.50 | 0.50 | 0.25 | 0.25 | 0.25 | |
| T. capitatus | MIC | 0.50 | 0.25 | 0.125 | 0.125 | 0.125 |
| MBC | 1.00 | 0.50 | 0.25 | 0.25 | 0.25 | |
| S. cypria | MIC | 0.50 | 0.25 | 0.125 | 0.125 | 0.125 |
| MBC | 1.00 | 0.50 | 0.25 | 0.25 | 0.25 | |
| E211 | MIC | 4.00 | 0.50 | 1.00 | 1.00 | 1.00 |
| MBC | 4.00 | 0.50 | 2.00 | 2.00 | 2.00 | |
| E224 | MIC | 1.0 | 2.0 | 0.5 | 1.0 | 0.5 |
| MBC | 1.0 | 4.0 | 1.0 | 1.0 | 1.0 |
| A. fumigatus (ATCC 9197) | A. versicolor (ATCC 11730) | A. niger (ATCC 6275) | P. funiculosum (ATCC 36839) | P. verrucosum var. cyclopium (Food Isolate) | ||
|---|---|---|---|---|---|---|
| M. officinalis | MIC | 0.50 | 0.25 | 0.50 | 1.00 | 1.00 |
| MFC | 1.00 | 0.50 | 1.00 | 2.00 | 2.00 | |
| S. fruticosa | MIC | 1.00 | 1.00 | 0.50 | 1.00 | 1.00 |
| MFC | 2.00 | 2.00 | 1.00 | 2.00 | 2.00 | |
| M. piperita | MIC | 0.50 | 0.50 | 0.50 | 1.00 | 1.00 |
| MFC | 1.00 | 1.00 | 1.00 | 2.00 | 2.00 | |
| O. dubium | MIC | 0.50 | 1.00 | 0.50 | 0.25 | 0.50 |
| MFC | 1.00 | 2.00 | 1.00 | 0.50 | 1.00 | |
| T. capitatus | MIC | 0.50 | 1.00 | 1.00 | 0.50 | 0.50 |
| MFC | 1.00 | 2.00 | 2.00 | 1.00 | 1.00 | |
| S. cypria | MIC | 0.50 | 0.50 | 0.25 | 0.25 | 0.25 |
| MFC | 1.00 | 1.00 | 0.50 | 0.50 | 0.50 | |
| E211 | MIC | 1.00 | 2.00 | 1.00 | 1.00 | 2.00 |
| MBC | 2.00 | 4.00 | 2.00 | 2.00 | 4.00 | |
| E224 | MIC | 1.00 | 1.00 | 1.00 | 0.50 | 1.00 |
| MBC | 1.00 | 1.00 | 1.00 | 0.50 | 1.00 |
| Female-Derived Cancer Cells | Colon Cancer Cells | ||||
|---|---|---|---|---|---|
| Treatment: 1000 μg/mL of plant extracts for 72 h | MDA-MB231 | SkoV3 | HeLa | HT-29 | HCT-116 |
| Origanum dubium | 448.6 | >1000 | >1000 | ||
| Sideritis cypria | 166.8 | 352.3 | 264.3 | ||
| Thymus capitatus | 394.2 | 698.4 | 858.2 | 237.5 | |
| Melissa officinalis | 181.3 | 676.2 | 765.5 | 371.2 | |
| Mentha piperita | 642.3 | 756.5 | 225.6 | ||
| Salvia fruticosa | 184.9 | 814.4 | 106.3 | ||
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chrysargyris, A.; Petrovic, J.D.; Tomou, E.-M.; Kyriakou, K.; Xylia, P.; Kotsoni, A.; Gkretsi, V.; Miltiadous, P.; Skaltsa, H.; Soković, M.D.; et al. Phytochemical Profiles and Biological Activities of Plant Extracts from Aromatic Plants Cultivated in Cyprus. Biology 2024, 13, 45. https://doi.org/10.3390/biology13010045
Chrysargyris A, Petrovic JD, Tomou E-M, Kyriakou K, Xylia P, Kotsoni A, Gkretsi V, Miltiadous P, Skaltsa H, Soković MD, et al. Phytochemical Profiles and Biological Activities of Plant Extracts from Aromatic Plants Cultivated in Cyprus. Biology. 2024; 13(1):45. https://doi.org/10.3390/biology13010045
Chicago/Turabian StyleChrysargyris, Antonios, Jovana D. Petrovic, Ekaterina-Michaela Tomou, Kalia Kyriakou, Panayiota Xylia, Andria Kotsoni, Vasiliki Gkretsi, Panagiota Miltiadous, Helen Skaltsa, Marina D. Soković, and et al. 2024. "Phytochemical Profiles and Biological Activities of Plant Extracts from Aromatic Plants Cultivated in Cyprus" Biology 13, no. 1: 45. https://doi.org/10.3390/biology13010045
APA StyleChrysargyris, A., Petrovic, J. D., Tomou, E.-M., Kyriakou, K., Xylia, P., Kotsoni, A., Gkretsi, V., Miltiadous, P., Skaltsa, H., Soković, M. D., & Tzortzakis, N. (2024). Phytochemical Profiles and Biological Activities of Plant Extracts from Aromatic Plants Cultivated in Cyprus. Biology, 13(1), 45. https://doi.org/10.3390/biology13010045










